The Prognostication of Columnar Mesophases - Synthesis and Mesomorphism of Some Porphyrin Derivatives
Abstract
:Introduction
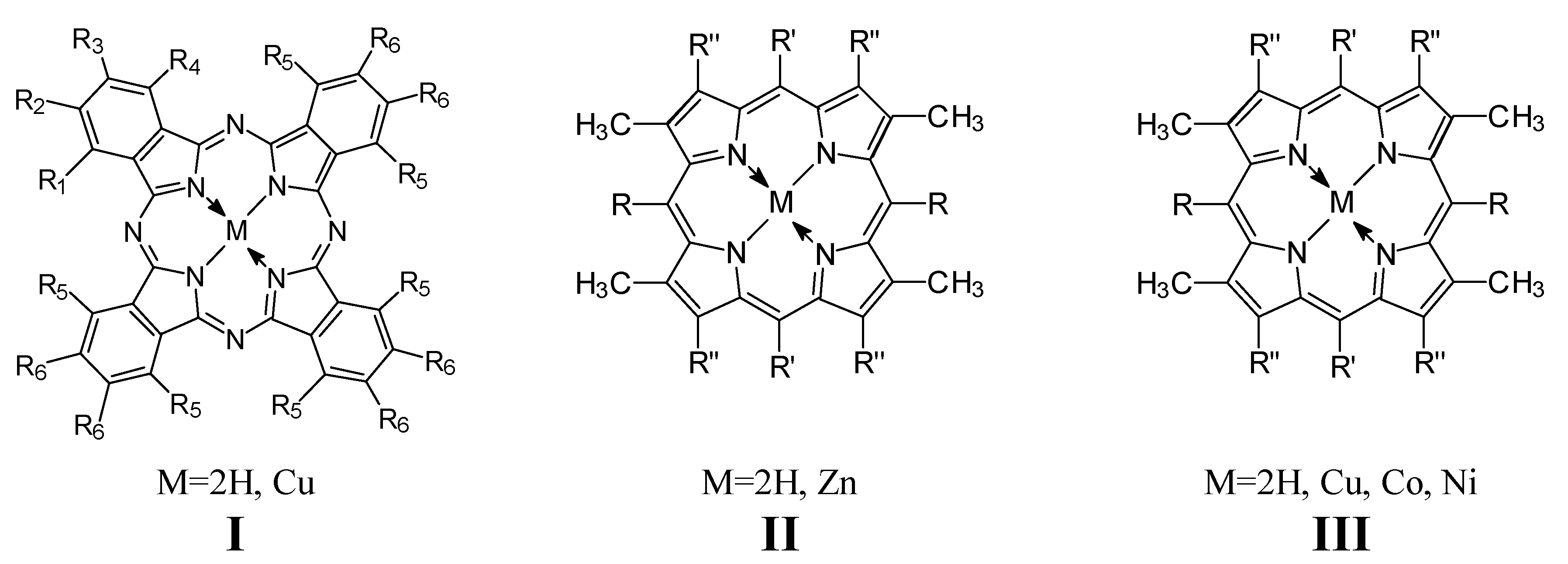
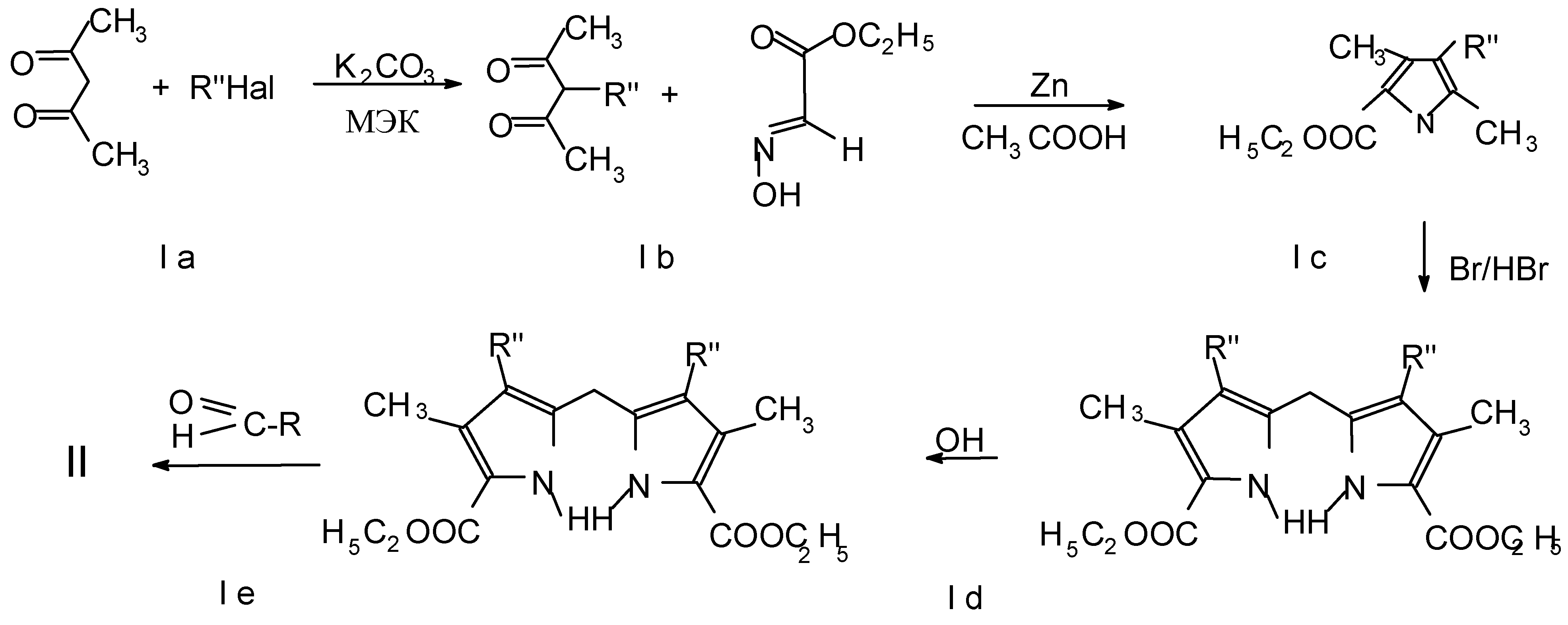
Results and Discussion
Conclusion
Experimental
General
5,15-Dialkyl- or 5,15-diaryl-2,8,12,18-tetraalkyl-3,7,13,17-tetramethylporphyrin, zinc complexes (II-18, 26, 28, 30)
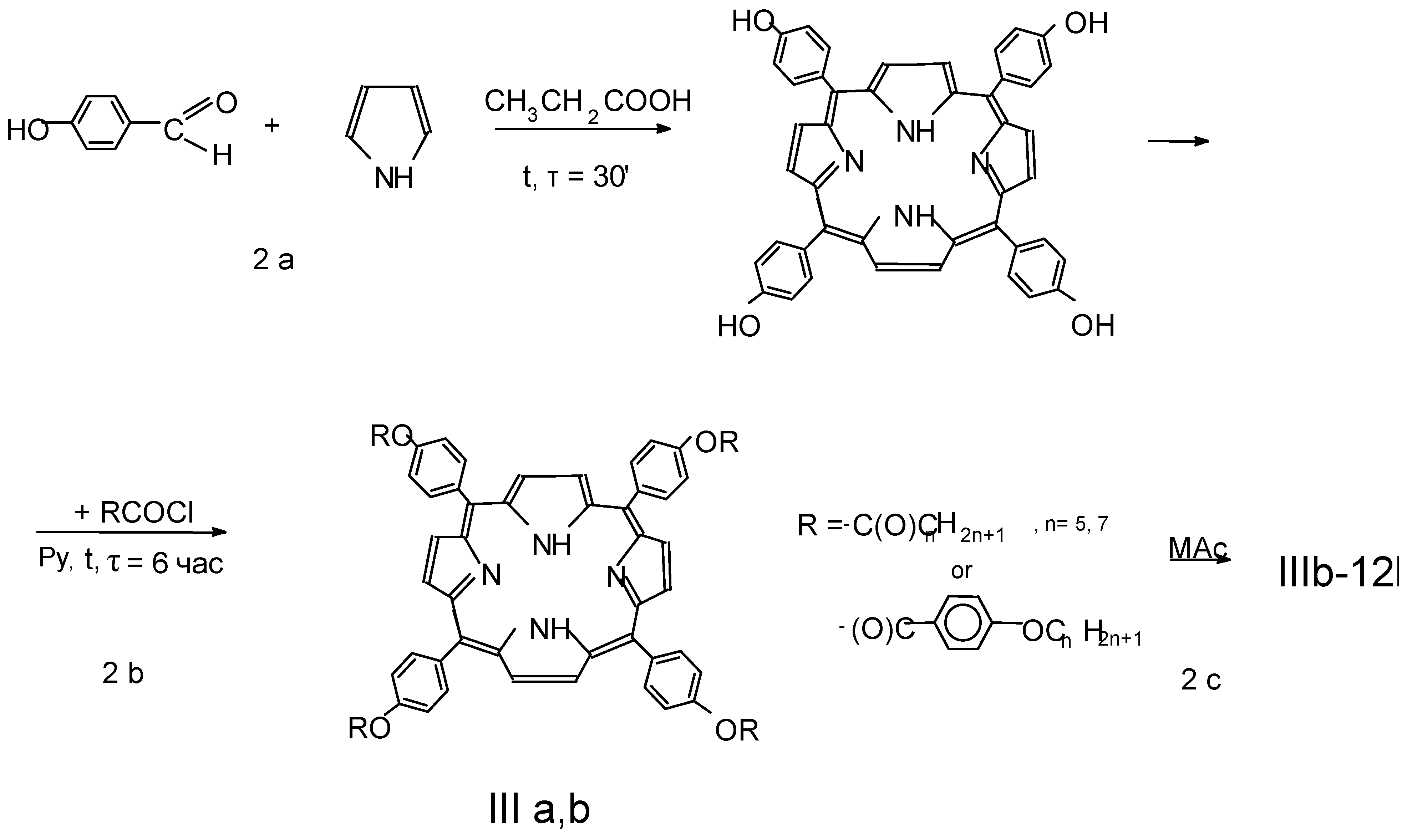
5,10,15,20-Tetra(p-hydroxyphenyl)porphyrin (Scheme 2, step 1)
Copper and cobalt metallocomplexes IIIb-12Cu, IIIb-12Co
Acknowledgements:
References and Notes
- Chandrasakhar, S.; Shadashiva, B. K.; Suresh, K. A. Pramana 1977, 9, 471.
- Goodby, J.W.; Gray, G.W. Mol. Cryst. Liq. Cryst. 1980, 56, 303–309. [CrossRef]
- Piechocki, C.; Simon, J.; Skoulios; Guillon, D.; Weber, P. J. Am. Chem. Soc. 1980, 102, 5245.
- Piechocki, C.; Simon, J. Nouv. J. Chim. 1985, 9, 159–166.
- Shimizu, Y.; Higashiyama, T. Proceedings of SPIE. Liq. Cryst.: Chemistry and Structure 1997, 3319, 177.
- Shimizu, Y.; Santiago, J.; Tantrawong, S.; Sugino, T. Abs. CCMM’98; Bayreuth: Germany, 1998; p. L.28. [Google Scholar]
- Usol’tseva, N. V.; Bykova, V. V.; Ananieva, G. A.; Maizlish, V. E.; Kudrik, E. V.; Shaposhnikov, G. P. Abs. CCMM’98; Bayreuth: Germany, 1998; p. L 27. [Google Scholar]
- Bykova, V. V.; Usol’tseva, N. V.; Ananieva, G. A.; Shaposhnikov, G. P.; Maizlish, V. E. Izvves- tiya Akademii nauk SSSR. Seriya phizitcheskaya 1998, 62, 1647–1651.
- Akopova, O. B.; Zdanovich, S. A.; Akopov, A. A.; Kotovich, L. N.; Usol’tseva, N. V. Izvvestiya Akademii nauk. Seriya phizitcheskaya 1997, 61, 623.
- Akopova, O. B.; Bobrov, V. I.; Erikalov, U. G. Russian J. of Physical Chem. 1990, 64, 1460.
- Akopova, O. B.; Jukova, L. V.; Chabichev, L. S. Russian J. of Physical Chem. 1995, 69, 96.
- Akopova, O. B.; Bronnikova, A. A.; Kruvchinnski, A.; Kotovich, L. N.; Chabichev, L. S.; Valk- ova, L. A. Russian J. Struct. Chem. 1998, 39, 464–472.
- Akopova, O. B.; Bronnikova, A. A. Russian J. Struct. Chem. 1998, 39, 473–477.
- Akopova, O. B.; Akopov, D. A.; Usol’tseva, N. V. Russian J. Physical Chem. 1999. (in print).
- Akopova, O. B.; Bobrov, V. I.; Chabichev, L. S.; Onichenko, A. A. Russian J. of Physical Chem. 1992, 66, 292–299.
- Billard, J.; Dubois, J. C.; Tinh, N. H.; Zann, A. Nouv. J. Chim. 1979, 2, 535–540.
- Zdanovich, S. A.; Mamardashvili, N. Zh.; Golubchikov, O. A. Russian J. of Organic Chem. 1996, 32, 788–792.
- Kuzmin, N. G.; Semeikin, A. S.; Koiphman, O. I. Izv. VUZov. Chimia i chim. technolog. 1988, 31, 39–44.
- Fischer, H.; Orth, H. Die chemie des Pyrrols; Leipzig: Akad. Verlages, 1934; Vol. 1, p. 766. [Google Scholar]
- Gray, G. W.; Goodby, W. Smectic Liquid Crystals: Textures and Structures; Leonard Hill: Glas- gow, 1984. [Google Scholar]
- Samples Availability: Available from the authors.

| Compound | K | Kp | Kar | Mm | Mr | Prognosis CM | Experiment [5] |
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| 1 | 9.01 | 1.190 | 0.119 | 2.410 | 0.601 | − | − |
| 2 | 8.90 | 0.981 | 0.131 | 1.940 | 0.486 | − | − |
| 3 | 11.47 | 0.808 | 0.129 | 1.630 | 0.408 | − | − |
| 4 | 13.06 | 0.680 | 0.127 | 1.400 | 0.351 | − | + |
| 5 | 13.62 | 0.591 | 0.124 | 1.230 | 0.308 | − | |
| 6 | 13.96 | 0.520 | 0.118 | 1.099 | 0.275 | − | |
| 7 | 18.24 | 0.454 | 0.110 | 0.992 | 0.248 | − | + |
| 8 | 16.19 | 0.421 | 0.109 | 0.903 | 0.226 | − | |
| 9 | 17.76 | 0.384 | 0.105 | 0.829 | 0.207 | − | + |
| 10 | 20.08 | 0.353 | 0.101 | 0.766 | 0.192 | − | |
| 11 | 19.79 | 0.327 | 0.097 | 0.712 | 0.178 | − | + |
| 12 | 20.62 | 0.304 | 0.093 | 0.666 | 0.166 | − | |
| 13 | 23.11 | 0.285 | 0.090 | 0.625 | 0.156 | − | |
| 14 | 24.68 | 0.267 | 0.087 | 0.588 | 0.147 | − | |
| 15 | 24.36 | 0.252 | 0.084 | 0.556 | 0.139 | − | |
| 16 | 24.08 | 0.238 | 0.081 | 0.527 | 0.132 | − | + |
| Compound | K | Kp | Kar | Mm | Mr | Prognosis CM | Experiment [5] |
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| 1 | 3.00 | 1.190 | 0.119 | 2.410 | 0.601 | − | − |
| 2 | 2.97 | 0.981 | 0.131 | 1.940 | 0.486 | − | − |
| 3 | 3.82 | 0.808 | 0.129 | 1.630 | 0.408 | − | − |
| 4 | 4.35 | 0.680 | 0.127 | 1.400 | 0.351 | + | + |
| 5 | 4.54 | 0.591 | 0.124 | 1.230 | 0.308 | + | |
| 6 | 4.65 | 0.520 | 0.118 | 1.099 | 0.275 | + | |
| 7 | 6.08 | 0.454 | 0.110 | 0.992 | 0.248 | + | + |
| 8 | 5.40 | 0.421 | 0.109 | 0.903 | 0.226 | + | |
| 9 | 5.92 | 0.384 | 0.105 | 0.829 | 0.207 | + | + |
| 10 | 6.70 | 0.353 | 0.101 | 0.766 | 0.192 | + | |
| 11 | 6.60 | 0.327 | 0.097 | 0.712 | 0.178 | + | + |
| 12 | 6.87 | 0.304 | 0.093 | 0.666 | 0.166 | + | |
| 13 | 7.70 | 0.285 | 0.090 | 0.625 | 0.156 | + | |
| 14 | 8.23 | 0.267 | 0.087 | 0.588 | 0.147 | ± | |
| 15 | 8.12 | 0.252 | 0.084 | 0.556 | 0.139 | ± | |
| 16 | 8.03 | 0.238 | 0.081 | 0.527 | 0.132 | ± | + |
| Compound | K | Kp | Kar | Mm | Mr | Prognosis CM | Experiment | Tp.t.,oC |
|---|---|---|---|---|---|---|---|---|
| 1-5/5 | 4.66 | 0.479 | 0.316 | 0.617 | 0.514 | + | ||
| 3-8/8 | 2.04 | 0.336 | 0.270 | 0.406 | 0.337 | + | ||
| 4-10/10 | 2.04 | 0.272 | 0.232 | 0.331 | 0.276 | + | ||
| 5-15/15 | 2.12 | 0.184' | 0.174 | 0.226 | 0.188' | − | ||
| 6-5/5 | 4.34 | 0.503 | 0.341 | 0.617 | 0.514 | + | ||
| 7-7/7 | 4.43 | 0.381 | 0.295 | 0.458 | 0.382 | + | ||
| 8-8/8 | 4.42 | 0.328 | 0.244 | 0.406 | 0.338 | + | ||
| 9-10/10 | 4.62 | 0.267 | 0.226 | 0.331 | 0.276 | + | ||
| 10-15/15 | 5.17 | 0.182' | 0.171 | 0.226’ | 0.188' | − | ||
| 11-4/6 | 2.39 | 0.398 | 0.259 | 0.654 | 0.545 | + | ||
| 12- Zn4/6 | 2.46 | 0.404 | 0.257 | 0.680 | 0.567 | + | ||
| 13-4/OBC9 | 3.21 | 0.306 | 0.098' | 0.578 | 0.482 | − | ||
| 14-Zn4/OBC9 | 5.56 | 0.277 | 0.084’ | 0.602 | 0.502 | − | ||
| 15-8/ OBC 9 | 3.19 | 0.278 | 0.130’ | 0.349 | 0.291 | − | ||
| 16-Zn8/ OBC | 3.07 | 0.292 | 0.140’ | 0.364 | 0.303 | − | ||
| 9 | ||||||||
| 17-8/7 | 4.58 | 0.329 | 0.236 | 0.429 | 0.357 | + | CM? | I |
| 18-Zn8/7 | 2.19 | 0.297 | 0.230 | 0.445 | 0.371 | + | ||
| 19-8/11 | 1.97' | 0.227’ | 0.172 | 0.369 | 0.308 | − | − | 187 |
| 20-Zn8/11 | 1.94' | 0.232’ | 0.176 | 0.384 | 0.320 | − | ||
| 23-8/ OBC | 1.94' | 0.452 | 0.197 | 0.498 | 0.415 | − | ||
| 25-4/ BC 9 | 3.64 | 0.219’ | 0.109' | 0,522 | 0.435 | − | ||
| 26-Zn4/BC 9 | 3.55 | 0.227’ | 0.115' | 0.520 | 0.430 | − | − | 160 |
| 27-8/BC | 2.04 | 0.316 | 0.219 | 0.450 | 0.375 | + | ||
| 28-Zn8/BC | 1.86' | 0.310 | 0.220 | 0.468 | 0.390 | − | − | 170 |
| 29-8/BC 9 | 2.72 | 0.223’ | 0.172 | 0.328 | 0.273 | − | − | 140 |
| 30-Zn8/BC9 | 2,40 | 0.275 | 0.165 | 0.315 | 0.262 | + | + | to 115 |
| 31-O7/O7/ 6 | 3.28 | 0.433 | 0.366 | 0.444 | 0.370 | + | ||
| 32-O6/O6/6 | 2.94 | 0.497 | 0.387' | 0.502 | 0.419 | − | ||
| 33-O6/O6/5 | 2.18 | 0.630 | 0.449' | 0.520 | 0.433 | − | ||
| 34-O6/O6/5/5 | 2.31 | 0.571 | 0.459' | 0.442 | 0.442 | − |
| N | n | K | Kp | Kar | Mm | Mr | Prognosis | Experiment | Tp.t., °C |
|---|---|---|---|---|---|---|---|---|---|
| 1 | IIIa -1 | 4.15 | 0.586 | 0.123 | 0.567 | 0.189 | + | ||
| 2 | 2 | 4.59 | 0.440 | 0.131 | 0.513 | 0.171 | + | ||
| 3 | 3 | 4.38 | 0.406 | 0.133 | 0.462 | 0.156 | + | ||
| 4 | 4 | 5.38 | 0.347 | 0.118 | 0.432 | 0.144’ | +, − | ||
| 5 | 5 | 5.87 | 0.317 | 0.114 | 0.400 | 0.134’ | − | − | subl. |
| 6 | 7 | 6.88 | 0.268 | 0.104 | 0.349 | 0.116’ | − | − | 300, |
| destr. | |||||||||
| 7 | 5-Cu | 4.11 | 0.342 | 0.127 | 0.481 | 0.160 | + | ||
| 8 | 5-Zn | 4.12 | 0.348 | 0.129 | 0.483 | 0.161 | + | ||
| 9 | 5 -Ni | 4.12 | 0.340 | 0.126 | 0.474 | 0.158 | + | ||
| 10 | 5-Co | 4.12 | 0.346 | 0.129 | 0.475 | 0.158 | + | ||
| 11 | IIIb -1 | 5.84 | 0.309 | 0.105 | 0.337 | 0.112’ | − | − | 120 |
| 12 | 2 | 6.07 | 0.285 | 0.102 | 0.317 | 0.106’ | − | ||
| 13 | 8 | 8.13 | 0.192’ | 0.083 | 0.235’ | 0.078’ | − | ||
| 14 | 9 | 8.38 | 0.182’ | 0.080 | 0.226’ | 0.075’ | − | ||
| 16 | 10 | 8.72 | 0.173’ | 0.078 | 0.217’ | 0.072’ | − | ||
| 17 | 11 | 8.71’ | 0.165’ | 0.075’ | 0.208’ | 0.069’ | − | ||
| 18 | 12 | 9.20’ | 0.158’ | 0.073’ | 0.201’ | 0.067’ | − | ||
| 19 | 12-Cu | 9.08’ | 0.160’ | 0.075’ | 0.241’ | 0.080’ | − | − | 165 |
| 20 | 12-Zn | 9.02’ | 0.161’ | 0.074’ | 0.242’ | 0.081’ | − | ||
| 21 | 12 -Ni | 9.06’ | 0.160’ | 0.075’ | 0.238’ | 0.079’ | − | ||
| 22 | 12-Co | 9.04’ | 0.161’ | 0.075’ | 0.238’ | 0.079’ | − | + | 190-220 |
| N | Compound | λ1, / lg ε1 | λ2, / lg ε2 | λ3, / lg ε3 | λ4, / lg ε4 | Solvent |
|---|---|---|---|---|---|---|
| 1 | II-25-4/BC9 | 617/3.54 | 578/4.22 | 541/4.12 | 510/4.53 | CHCl 3 |
| 2 | II-26- | 617/3.89 | - | 546/4.18 | - | ‘’------‘’ |
| Zn4/BC9 | ||||||
| 3 | II-28-Zn8/BC | 617/3.86 | - | 549/4.00 | - | ‘’------‘’ |
| 4 | II-29-8/BC9 | 637/3.58 | 588/4.09 | 543/4.00 | 513/4.47 | hexane |
| 5 | II-30- | 606/4.33 | - | 541/4.34 | - | ‘’------‘’ |
| Zn8/BC9 | ||||||
| 6 | III-a5 | 694/4.07 | 610/4.21 | 562/4.40 | 521/4.83 | CH2Cl 2 |
| 7 | III-a7 | 667/4.28 | 606/4.22 | 562/4.55 | 524/4.74 | ‘’------‘’ |
| 8 | III-b12 | 645/3.32 | 595/3.47 | 556/3.69 | 519/3.99 | ‘’------‘’ |
| 9 | III-bCo12 | - | 549/4.28 | 439/5.11 | 418/5.16 | ‘’------‘’ |
| 10 | III-bCu12 | - | 549/3.79 | - | 417/4.97 | ‘’------‘’ |
| N | Compound | Gross formula | MM | CALC, % | FOUND % | ||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | N | C | H | N | ||||
| 1 | II-18-Zn8/7 | C70H102N4Zn | 1013.61 | 82.94 | 10.16 | 5.52 | 83.03 | 10.44 | 5.35 |
| 2 | II-25-4BC9 | C58H90O2N4 | 875.35 | 79.58 | 10.38 | 6.40 | 79.96 | 10.49 | 6.55 |
| 3 | II-26-Zn4BC9 | C58H90O2N4Zn | 887.37 | 78.50 | 10.24 | 6.31 | 78.81 | 10.61 | 6.72 |
| 4 | II-28-Zn8/BC | C68H82N4Zn | 1020.82 | 80.00 | 8.11 | 5.49 | 80.48 | 8.63 | 5.21 |
| 5 | II-29-8/BC9 | C86H130O2N4 | 1252.00 | 82.50 | 10.49 | 4.47 | 83.07 | 9.70 | 4.03 |
| 6 | II-30-Zn8/BC9 | C86H128O2N4Zn | 1263.99 | 82.53 | 10.23 | 4.43 | 82.02 | 9.88 | 4.59 |
| 7 | III-a5 | C68H70O8N4 | 1071.33 | 76.23 | 6.60 | 5.23 | 76.01 | 6.06 | 5.03 |
| 8 | III-a7 | C76H86O8N4 | 1183.58 | 77.12 | 7.34 | 4.73 | 76.57 | 7.99 | 4.82 |
| 9 | III-b12 | C120H142O12N4 | 1832.47 | 78.65 | 7.83 | 3.06 | 79.92 | 8.37 | 3.20 |
| 10 | III-bCo12 | C120H140O12N4Co | 1889.38 | 76.28 | 7.48 | 2.97 | 75.20 | 7.89 | 3.15 |
| 11 | III-bCu12 | C120H140O12N4Cu | 1894.00 | 76.09 | 7.47 | 2.96 | 74.89 | 7.52 | 2.79 |
© 2000 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Akopova, O.B.; Zdanovich, S.A. The Prognostication of Columnar Mesophases - Synthesis and Mesomorphism of Some Porphyrin Derivatives. Molecules 2000, 5, 797-808. https://doi.org/10.3390/50600797
Akopova OB, Zdanovich SA. The Prognostication of Columnar Mesophases - Synthesis and Mesomorphism of Some Porphyrin Derivatives. Molecules. 2000; 5(6):797-808. https://doi.org/10.3390/50600797
Chicago/Turabian StyleAkopova, Olga B., and Sergei A. Zdanovich. 2000. "The Prognostication of Columnar Mesophases - Synthesis and Mesomorphism of Some Porphyrin Derivatives" Molecules 5, no. 6: 797-808. https://doi.org/10.3390/50600797
APA StyleAkopova, O. B., & Zdanovich, S. A. (2000). The Prognostication of Columnar Mesophases - Synthesis and Mesomorphism of Some Porphyrin Derivatives. Molecules, 5(6), 797-808. https://doi.org/10.3390/50600797




