Photoprotective Effect of Annona muricata L. Extracts in Rats Exposed to Ultraviolet Radiation via P53 and RB Gene Expression
Abstract
1. Introduction
2. Results
2.1. Phenolic Compounds and Acetogenins
2.2. Sun Protection Factor (SPF)
2.3. HPLC Characterization of Phenolic Compounds
2.4. Histologic Analysis
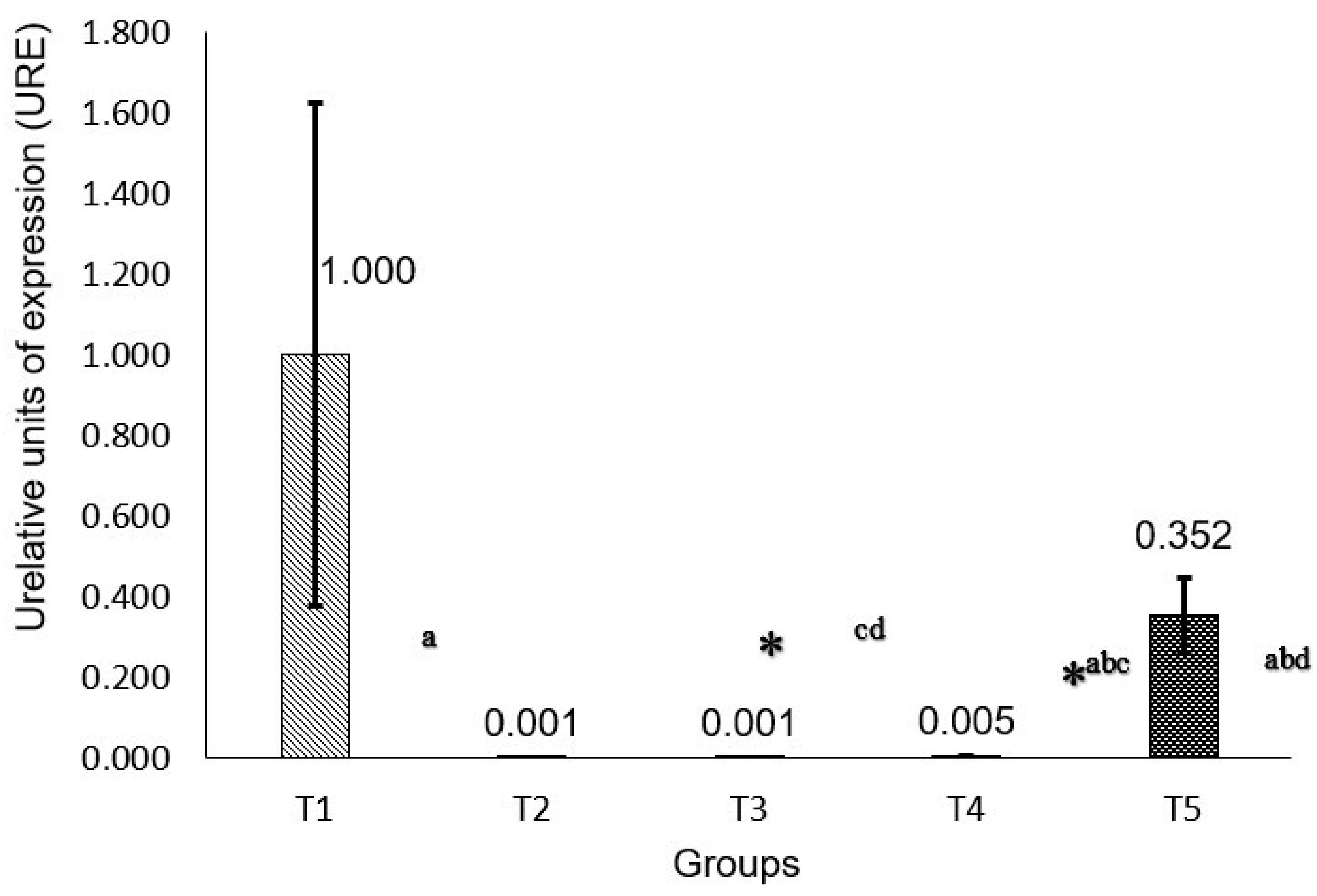
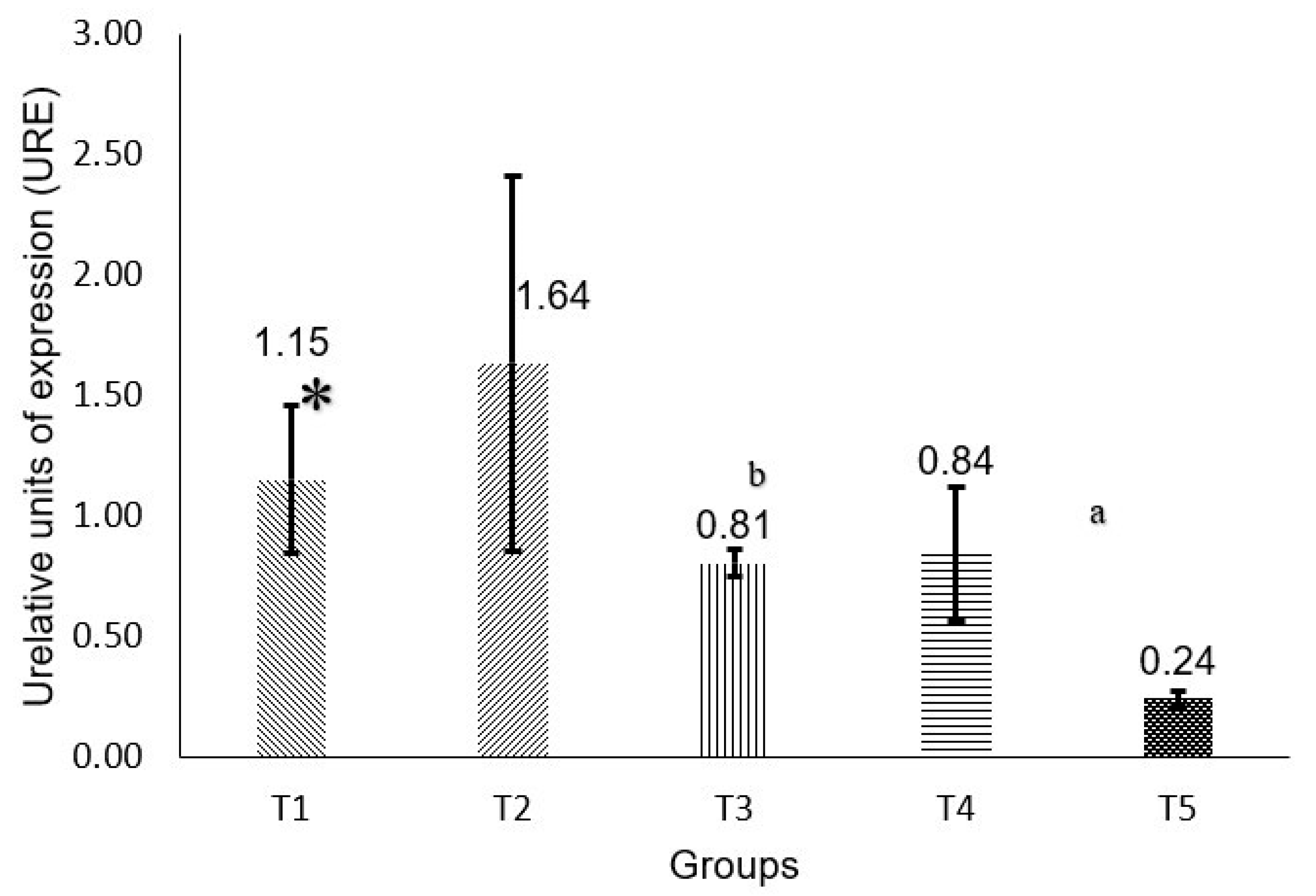
2.5. P53 and RB Gene Expression
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Extraction of Phenolic Compounds from Annona Muricata Leaves
4.3. Phenolic Compounds Quantification
4.4. Flavonoid Quantification
4.5. In Vitro Sun Protection Factor (SPF) Evaluation
4.6. Antioxidant Capacity Assay
4.7. Characterization of Phenolic Extract by HPLC
4.8. Extraction and Identification of Acetogenins
4.9. Animal Model
- -
- T1, not exposed to UV light and without treatment.
- -
- T2, exposed to UV light and untreated.
- -
- T3, treated with a commercial sunscreen with SPF 50.
- -
- T4, treated with acetogenins extract.
- -
- T5, treated with phenolic compound extract.
4.10. UV Light Irradiation Model
4.11. Histologic Examination
4.12. Gene Expression Quantification
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fahradyan, A.; Howell, A.C.; Wolfswinkel, E.M.; Tsuha, M.; Sheth, P.; Wong, A.K. Updates on the Management of Non-Melanoma Skin Cancer (NMSC). Healthcare 2017, 5, 82. [Google Scholar] [CrossRef]
- Chamcheu, J.C.; Rady, I.; Chamcheu, R.-C.N.; Siddique, A.B.; Bloch, M.B.; Banang Mbeumi, S.; Babatunde, A.S.; Uddin, M.B.; Noubissi, F.K.; Jurutka, P.W.; et al. Graviola (Annona muricata) Exerts Anti-Proliferative, Anti-Clonogenic and Pro-Apoptotic Effects in Human Non-Melanoma Skin Cancer UW-BCC1 and A431 Cells In Vitro: Involvement of Hedgehog Signaling. Int. J. Mol. Sci. 2018, 19, 1791. [Google Scholar] [CrossRef]
- Aguilar-Hernández, G.; García-Magaña, M.; Vivar-Vera, M.; Sáyago-Ayerdi, S.; Sánchez-Burgos, J.; Morales-Castro, J.; Anaya-Esparza, L.; Montalvo González, E. Optimization of Ultrasound-Assisted Extraction of Phenolic Compounds from Annona muricata By-Products and Pulp. Molecules 2019, 24, 904. [Google Scholar] [CrossRef]
- Adefegha, S.A.; Oyeleye, S.I.; Oboh, G. Distribution of Phenolic Contents, Antidiabetic Potentials, Antihypertensive Properties, and Antioxidative Effects of Soursop (Annona muricata L.) Fruit Parts In Vitro. Biochem. Res. Int. 2015, 2015, 347673. [Google Scholar] [CrossRef]
- Ilango, S.; Sahoo, D.K.; Paital, B.; Kathirvel, K.; Gabriel, J.I.; Subramaniam, K.; Jayachandran, P.; Dash, R.K.; Hati, A.K.; Behera, T.R.; et al. A Review on Annona muricata and Its Anticancer Activity. Cancers 2022, 14, 4539. [Google Scholar] [CrossRef] [PubMed]
- Rady, I.; Bloch, M.B.; Chamcheu, R.C.N.; Banang Mbeumi, S.; Anwar, M.R.; Mohamed, H.; Babatunde, A.S.; Kuiate, J.R.; Noubissi, F.K.; El Sayed, K.A.; et al. Anticancer Properties of Graviola (Annona muricata): A Comprehensive Mechanistic Review. Oxid. Med. Cell. Longev. 2018, 2018, 1826170. [Google Scholar] [CrossRef]
- Mutakin, M.; Fauziati, R.; Fadhilah, F.N.; Zuhrotun, A.; Amalia, R.; Hadisaputri, Y.E. Pharmacological Activities of Soursop (Annona muricata Lin.). Molecules 2022, 27, 1201. [Google Scholar] [CrossRef]
- Zorofchian-Moghadamtousi, S.; Karimian, H.; Rouhollahi, E.; Paydar, M.; Fadaeinasab, M.; Abdul Kadir, H. Annona Muricata Leaves Induce G1 Cell Cycle Arrest and Apoptosis through Mitochondria-Mediated Pathway in Human HCT-116 and HT-29 Colon Cancer Cells. J. Ethnopharmacol. 2014, 156, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Leal, F.C.; Farias, F.O.; do Amaral, W.; Toci, A.T.; Mafra, M.R.; Igarashi-Mafra, L. Green solvents to value Annona muricata L. leaves as antioxidants source: Process optimization and potential as a natural food additive. Waste Biomass. Valoriz. 2022, 13, 1233–1241. [Google Scholar] [CrossRef]
- Nolasco-González, Y.; Chacón-López, M.A.; Ortiz-Basurto, R.I.; Aguilera-Aguirre, S.; González-Aguilar, G.A.; Rodríguez-Aguayo, C.; Navarro-Cortez, M.C.; García-Galindo, H.S.; García-Magaña, M.d.L.; Meza-Espinoza, L.; et al. Annona muricata Leaves as a Source of Bioactive Compounds: Extraction and Quantification Using Ultrasound. Horticulturae 2022, 8, 560. [Google Scholar] [CrossRef]
- Giampieri, F.; Alvarez-Suarez, J.M.; Tulipani, S.; Gonzàles-Paramàs, A.M.; Santos-Buelga, C.; Bompadre, S.; Quiles, J.L.; Mezzetti, B.; Battino, M. Photoprotective Potential of Strawberry (Fragaria × Ananassa) Extract against UV-A Irradiation Damage on Human Fibroblasts. J. Agric. Food Chem. 2012, 60, 2322–2327. [Google Scholar] [CrossRef] [PubMed]
- Zubaidi, S.N.; Mohd Nani, H.; Ahmad Kamal, M.S.; Abdul Qayyum, T.; Maarof, S.; Afzan, A.; Mohmad Misnan, N.; Hamezah, H.S.; Baharum, S.N.; Mediani, A. Annona muricata: Comprehensive Review on the Ethnomedicinal, Phytochemistry, and Pharmacological Aspects Focusing on Antidiabetic Properties. Life 2023, 13, 353. [Google Scholar] [CrossRef]
- Roduan, M.D.; Hamid, R.A.; Cheah, Y.K.; Mohtarrudin, N. Cytotoxicity, Antitumor-Promoting and Antioxidant Activities of Annona muricata In Vitro. J. Herb. Med. 2019, 15, 100219. [Google Scholar] [CrossRef]
- Kaur, C.D.; Saraf, S. In Vitro Sun Protection Factor Determination of Herbal Oils Used in Cosmetics. Pharmacogn. Res. 2010, 2, 22–25. [Google Scholar] [CrossRef]
- Bae, J.T.; Ko, H.J.; Kim, G.B.; Pyo, H.B.; Lee, G.S. Protective Effects of Fermented Citrus unshiu Peel Extract against Ultraviolet-A-Induced Photoageing in Human Dermal Fibrobolasts. Phytother. Res. 2012, 26, 1851–1856. [Google Scholar] [CrossRef]
- Moghadamtousi, S.; Fadaeinasab, M.; Nikzad, S.; Mohan, G.; Ali, H.; Kadir, H. Annona muricata (Annonaceae): A Review of Its Traditional Uses, Isolated Acetogenins and Biological Activities. Int. J. Mol. Sci. 2015, 16, 15625–15658. [Google Scholar] [CrossRef]
- Ferreira, L.E.; Castro, P.M.N.; Chagas, A.C.S.; França, S.C.; Beleboni, R.O. In Vitro Anthelmintic Activity of Aqueous Leaf Extract of Annona muricata L. (Annonaceae) against Haemonchus contortus from Sheep. Exp. Parasitol. 2013, 134, 327–332. [Google Scholar] [CrossRef]
- Jiménez, V.M.; Gruschwitz, M.; Schweiggert, R.M.; Carle, R.; Esquivel, P. Identification of Phenolic Compounds in Soursop (Annona muricata) Pulp by High-Performance Liquid Chromatography with Diode Array and Electrospray Ionization Mass Spectrometric Detection. Food Res. Int. 2014, 65, 42–46. [Google Scholar] [CrossRef]
- Jacobo-Herrera, N.; Pérez-Plasencia, C.; Castro-Torres, V.A.; Martínez-Vázquez, M.; González-Esquinca, A.R.; Zentella-Dehesa, A. Selective Acetogenins and Their Potential as Anticancer Agents. Front. Pharmacol. 2019, 10, 783. [Google Scholar] [CrossRef]
- Nawwar, M.; Ayoub, N.; Hussein, S.; Hashim, A.; El-Sharawy, R.; Wende, K.; Harms, M.; Lindequist, U. Flavonol Triglycoside and Investigation of the Antioxidant and Cell Stimulating Activities of Annona muricata Linn. Arch. Pharm. Res. 2012, 35, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Gubern, A.; Joaquin, M.; Marquès, M.; Maseres, P.; Garcia-Garcia, J.; Amat, R.; González-Nuñez, D.; Oliva, B.; Real, F.X.; de Nadal, E.; et al. The N-Terminal Phosphorylation of RB by P38 Bypasses Its Inactivation by CDKs and Prevents Proliferation in Cancer Cells. Mol. Cell 2016, 64, 25–36. [Google Scholar] [CrossRef]
- Mnich, C.D.; Hoek, K.S.; Virkki, L.V.; Farkas, A.; Dudli, C.; Laine, E.; Urosevic, M.; Dummer, R. Green Tea Extract Reduces Induction of P53 and Apoptosis in UVB-Irradiated Human Skin Independent of Transcriptional Controls. Exp. Dermatol. 2009, 18, 69–77. [Google Scholar] [CrossRef]
- Saric, S.; Sivamani, R.K. Polyphenols and Sunburn. Int. J. Mol. Sci. 2016, 17, 1521. [Google Scholar] [CrossRef] [PubMed]
- Serafini, M.R.; Detoni, C.B.; Menezes, P.D.P.; Pereira Filho, R.N.; Fortes, V.S.; Vieira, M.J.F.; Guterres, S.S.; de Albuquerque Junior, R.L.C.; Araújo, A.A.D.S. UVA-UVB Photoprotective Activity of Topical Formulations Containing Morinda citrifolia Extract. BioMed Res. Int. 2014, 2014, 587819. [Google Scholar] [CrossRef]
- Plikus, M.v.; Wang, X.; Sinha, S.; Forte, E.; Thompson, S.M.; Herzog, E.L.; Driskell, R.R.; Rosenthal, N.; Biernaskie, J.; Horsley, V. Fibroblasts: Origins, Definitions, and Functions in Health and Disease. Cell 2021, 184, 3852–3872. [Google Scholar] [CrossRef] [PubMed]
- Czemplik, M.; Korzun-Chłopicka, U.; Szatkowski, M.; Działo, M.; Szopa, J.; Kulma, A. Optimization of Phenolic Compounds Extraction from Flax Shives and Their Effect on Human Fibroblasts. Evid.-Based Complement. Altern. Med. 2017, 2017, 3526392. [Google Scholar] [CrossRef]
- Merecz-Sadowska, A.; Sitarek, P.; Kucharska, E.; Kowalczyk, T.; Zajdel, K.; Cegliński, T.; Zajdel, R. Antioxidant Properties of Plant-Derived Phenolic Compounds and Their Effect on Skin Fibroblast Cells. Antioxidants 2021, 10, 726. [Google Scholar] [CrossRef]
- Haywood, R.; Wardman, P.; Sanders, R.; Linge, C. Sunscreens Inadequately Protect Against Ultraviolet-A-Induced Free Radicals in Skin: Implications for Skin Aging and Melanoma? J. Investig. Dermatol. 2003, 121, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Hamizah, S.; Roslida, A.H.; Fezah, O.; Tan, K.L.; Tor, Y.S.; Tan, C.I. Chemopreventive Potential of Annona muricata L Leaves on Chemically-Induced Skin Papillomagenesis in Mice. Asian Pac. J. Cancer Prev. 2012, 13, 2533–2539. [Google Scholar] [CrossRef]
- Coria-Téllez, A.V.; Montalvo-Gónzalez, E.; Yahia, E.M.; Obledo-Vázquez, E.N. Annona muricata: A Comprehensive Review on Its Traditional Medicinal Uses, Phytochemicals, Pharmacological Activities, Mechanisms of Action and Toxicity. Arab. J. Chem. 2018, 11, 662–691. [Google Scholar] [CrossRef]
- Nichols, J.A.; Katiyar, S.K. Skin Photoprotection by Natural Polyphenols: Anti-Inflammatory, Antioxidant and DNA Repair Mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Agati, G.; Brunetti, C.; Di Ferdinando, M.; Ferrini, F.; Pollastri, S.; Tattini, M. Functional Roles of Flavonoids in Photoprotection: New Evidence, Lessons from the Past. Plant Physiol. Biochem. 2013, 72, 35–45. [Google Scholar] [CrossRef]
- Korać, R.R.; Khambholja, K.M. Potential of Herbs in Skin Protection from Ultraviolet Radiation. Pharmacogn. Rev. 2011, 5, 164. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Velázquez, E.E.; Rivas-Díaz, K.; Loarca-Piña, M.G.F.; Mendoza-Díaz, S.; Reynoso-Camacho, R.; Ramos-Gómez, M. Comparación Del Contenido Fenólico, Capacidad Antioxidante y Actividad Antiinflamatoria de Infusiones Herbales Comerciales. Rev. Mex Cienc. Agric. 2012, 3, 481–495. [Google Scholar]
- Feresin, R.G.; Pourafshar, S.; Huang, J.; Zhao, Y.; Arjmandi, B.H.; Salazar, G. Extraction and Purification of Polyphenols from Freeze-Dried Berry Powder for the Treatment of Vascular Smooth Muscle Cells In Vitro. J. Vis. Exp. 2017, 2017, 55605. [Google Scholar] [CrossRef]
- Atanassova, M.; Georgieva, S.; Ivancheva, K. Total Phenolic and Total Flavonoid Contents, Antioxidant Capacity and Biological Contaminants in Medicinal Herbs. J. Univ. Chem. Technol. Metall. 2011, 46, 81–88. [Google Scholar]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Ragasa, C.Y.; Soriano, G.; Torres, O.B.; Don, M.J.; Shen, C.C. Acetogenins from Annona muricata. Pharmacogn. J. 2012, 4, 32–37. [Google Scholar] [CrossRef]
- Reynaga-Pereda, S.M. Efecto de La Relación Muestra: Solvente En El Rendimiento de Extracción de Acetogeninas Presentes En La Hoja de La Guanábana (Annona muricata), Provincia de Ascope, La Libertad. Tesis de Ingeniero Agroindustrial, Universidad Cesar Vallejo, Trujillo, Peru, 2018. [Google Scholar]
- Mathur, S.; Kaur, P.; Sharma, M.; Katyal, A.; Singh, B.; Tiwari, M.; Chandra, R. The Treatment of Skin Carcinoma, Induced by UV B Radiation, Using 1-Oxo-5β, 6β-Epoxy-Witha-2-Enolide, Isolated from the Roots of Withania somnifera, in a Rat Model. Phytomedicine 2004, 11, 452–460. [Google Scholar] [CrossRef]
- Xian, D.; Gao, X.; Xiong, X.; Xu, J.; Yang, L.; Pan, L.; Zhong, J. Photoprotection against UV-induced damage by skin-derived precursors in hairless mice. J. Photochem. Photobiol. B 2017, 175, 73–82. [Google Scholar] [CrossRef] [PubMed]
| Total Phenolic mg GAE/100 g | Total Flavonoids mg QE/100 g | DPPH % Inhibition | FRAP mmol Eq Trolox/mL | |
|---|---|---|---|---|
| Leaf | 61.5 ± 2.26 | 50.3 ± 3 | 64.37 ± 1.19 | 28.88 ± 0.278 |
| Phenolic Compound | Wavelength | ATR | Concentration (mg/mL) | mg/g Sample | |
|---|---|---|---|---|---|
| 1 | Shikimic Ac. | 270 | 2.429 | 12.251 | 612.533 |
| 2 | Protocatechuic Ac. | 270 | 15.671 | 0.006 | 0.305 |
| 3 | Gallocatechin | 270 | 17.573 | 0.159 | 7.970 |
| 4 | 4-Hydroxybenzoyl Ac. | 270 | 18.6805 | 0.008 | 0.425 |
| 5 | Chlorogenic Ac. | 320 | 19.5575 | 0.017 | 0.858 |
| 6 | 4-Hydroxyphenylacetic Ac. | 280 | 19.6455 | 0.103 | 5.141 |
| 7 | Vanillic Ac. | 270 | 20.2105 | 0.011 | 0.571 |
| 8 | Siryngic Ac. | 280 | 20.9 | 0.010 | 0.508 |
| 9 | 3-Hydroxybenzoic Ac. | 300 | 20.6245 | 0.037 | 1.867 |
| 10 | Caffeic Ac. | 320 | 21.0605 | 0.011 | 0.536 |
| 11 | Catechin | 280 | 21.386 | 0.039 | 1.925 |
| 12 | Rutin | 270 | 23.3335 | 0.062 | 3.089 |
| 13 | p-Coumaric Ac. | 310 | 23.405 | 0.047 | 2.343 |
| 14 | Trans-ferulic Ac. | 320 | 24.353 | 0.055 | 2.753 |
| 15 | Ellagic Ac. | 270 | 24.9165 | 0.037 | 1.828 |
| Total | 642.649 |
| Groups | N | Media ± SD (µm) | Media ± SD (mm2) |
|---|---|---|---|
| T2 | 45 | 61.58 ± 27.26 a | 2.689 ± 0.67 a |
| T3 | 45 | 49.66 ±15.82 ab | 2.061 ± 0.80 b |
| T4 | 45 | 43.02 ±12.43 b | 2.80 ± 0.86 a |
| T5 | 45 | 28.56 ± 7.91c | 2.566 ± 0.83 a |
| T1 | 45 | 18.66 ± 8.87 d | 1.5770 ± 0.64 c |
| Gene | Sense | Anti-Sense |
|---|---|---|
| RB | CCAGCGGAGTCCAAATTCCA | TCCCGAGGGTCTACAGTGTT |
| P53 | CCCCTGAAGACTGGATAACTGT | AGTTCCAGGTTCCTGTGCTG |
| GAPDH | AGACAGCCGCATCTTCTTGT | TACGGCCAAATCCGTTCACA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pizano-Andrade, J.C.; Vargas-Guerrero, B.; Vargas-Radillo, J.; Domínguez-Rosales, J.A.; Montalvo-González, E.; Rodriguez-Macias, R.; Garcia-López, P.M.; del Rocio Romero-Verdín, M.; Ruiz López, M.A. Photoprotective Effect of Annona muricata L. Extracts in Rats Exposed to Ultraviolet Radiation via P53 and RB Gene Expression. Molecules 2025, 30, 3518. https://doi.org/10.3390/molecules30173518
Pizano-Andrade JC, Vargas-Guerrero B, Vargas-Radillo J, Domínguez-Rosales JA, Montalvo-González E, Rodriguez-Macias R, Garcia-López PM, del Rocio Romero-Verdín M, Ruiz López MA. Photoprotective Effect of Annona muricata L. Extracts in Rats Exposed to Ultraviolet Radiation via P53 and RB Gene Expression. Molecules. 2025; 30(17):3518. https://doi.org/10.3390/molecules30173518
Chicago/Turabian StylePizano-Andrade, Juan Carlos, Belinda Vargas-Guerrero, Jesus Vargas-Radillo, José Alfredo Domínguez-Rosales, Efigenia Montalvo-González, Ramon Rodriguez-Macias, Pedro Macedonio Garcia-López, Margarita del Rocio Romero-Verdín, and Mario Alberto Ruiz López. 2025. "Photoprotective Effect of Annona muricata L. Extracts in Rats Exposed to Ultraviolet Radiation via P53 and RB Gene Expression" Molecules 30, no. 17: 3518. https://doi.org/10.3390/molecules30173518
APA StylePizano-Andrade, J. C., Vargas-Guerrero, B., Vargas-Radillo, J., Domínguez-Rosales, J. A., Montalvo-González, E., Rodriguez-Macias, R., Garcia-López, P. M., del Rocio Romero-Verdín, M., & Ruiz López, M. A. (2025). Photoprotective Effect of Annona muricata L. Extracts in Rats Exposed to Ultraviolet Radiation via P53 and RB Gene Expression. Molecules, 30(17), 3518. https://doi.org/10.3390/molecules30173518








