BI-5756 Reduces Graft-Versus-Host Disease Through CB1-Mediated Treg Upregulation
Abstract
1. Introduction
2. Results
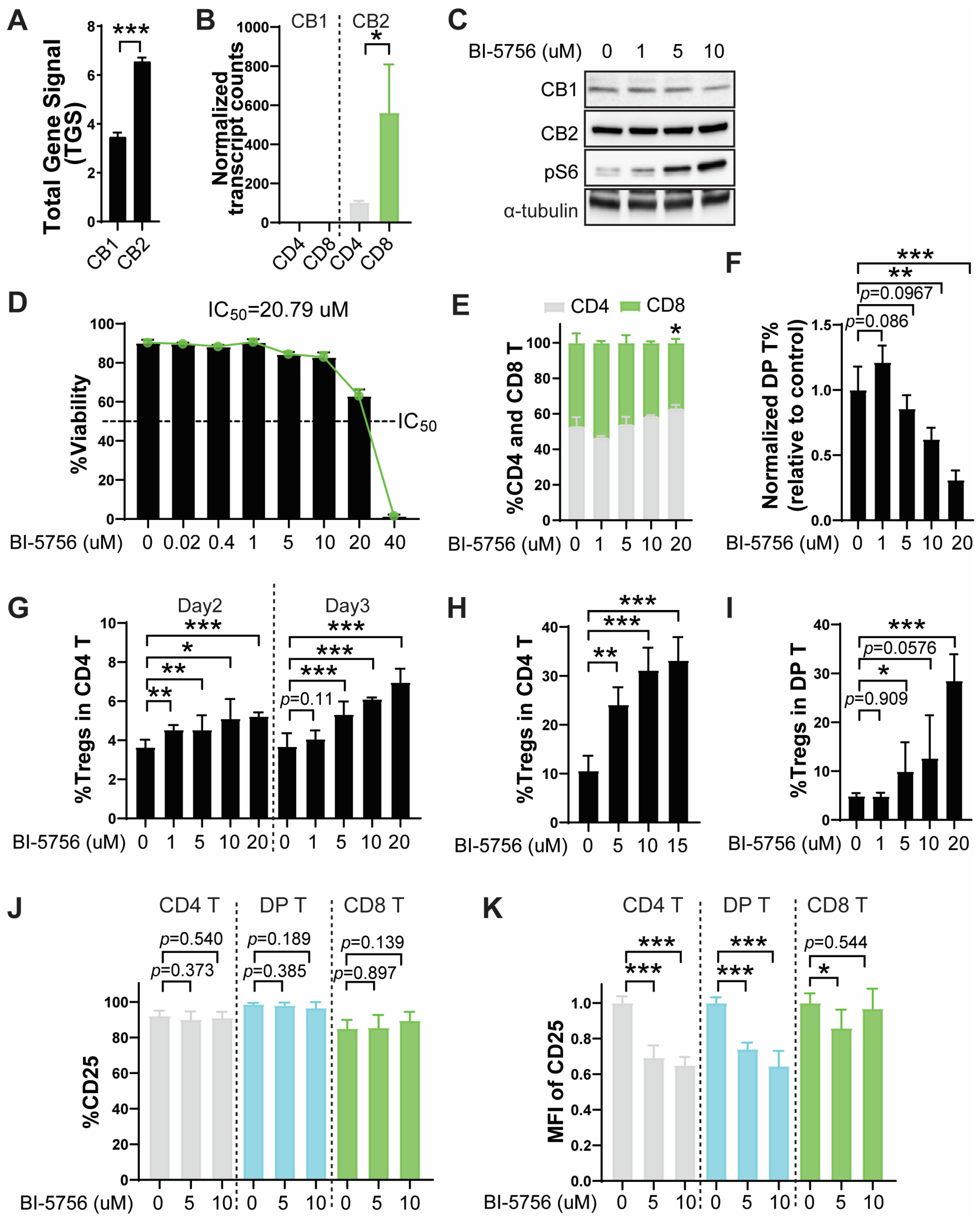
2.1. BI-5756 Upregulates Regulatory T Cells
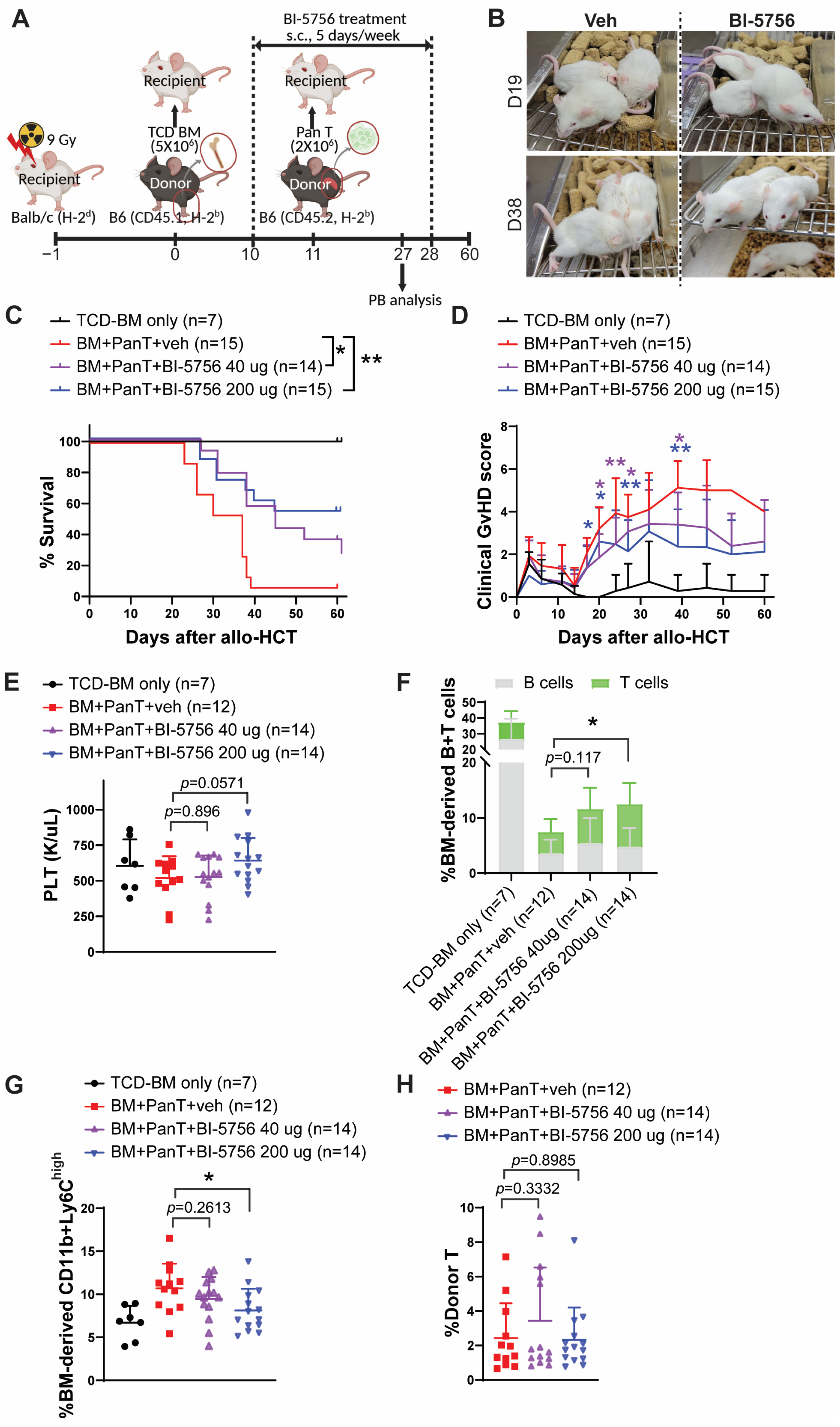
2.2. The Effect of BI-5756 on GvHD in a Preclinical Mouse Model of Allo-HCT
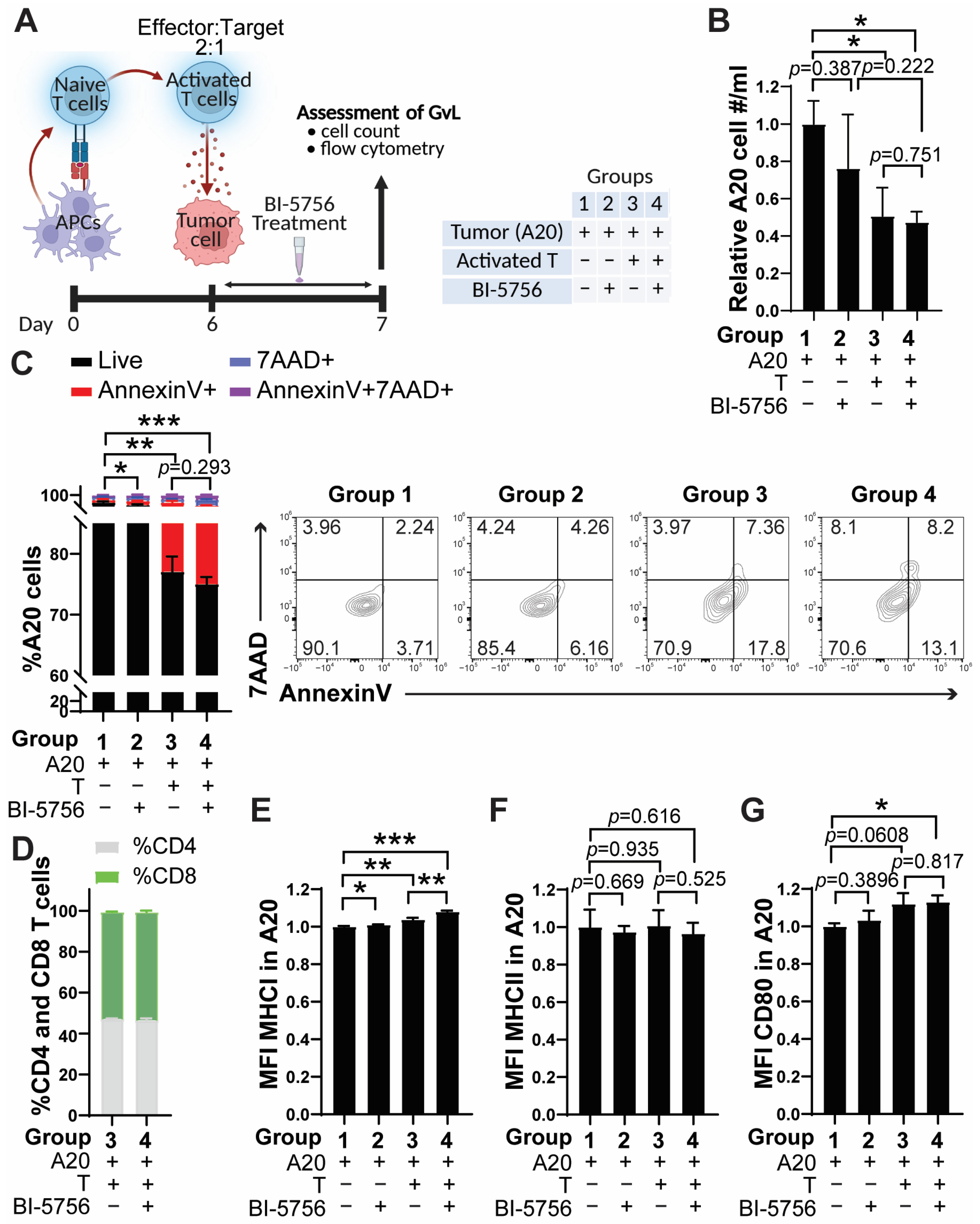
2.3. BI-5756 Directly Inhibits Tumor Cell Growth and Enhances Antigen Presentation in Murine B Cell Lymphoma Cells, A20
2.4. BI-5756 Does Not Compromise T Cell-Mediated Anti-Tumor Activities
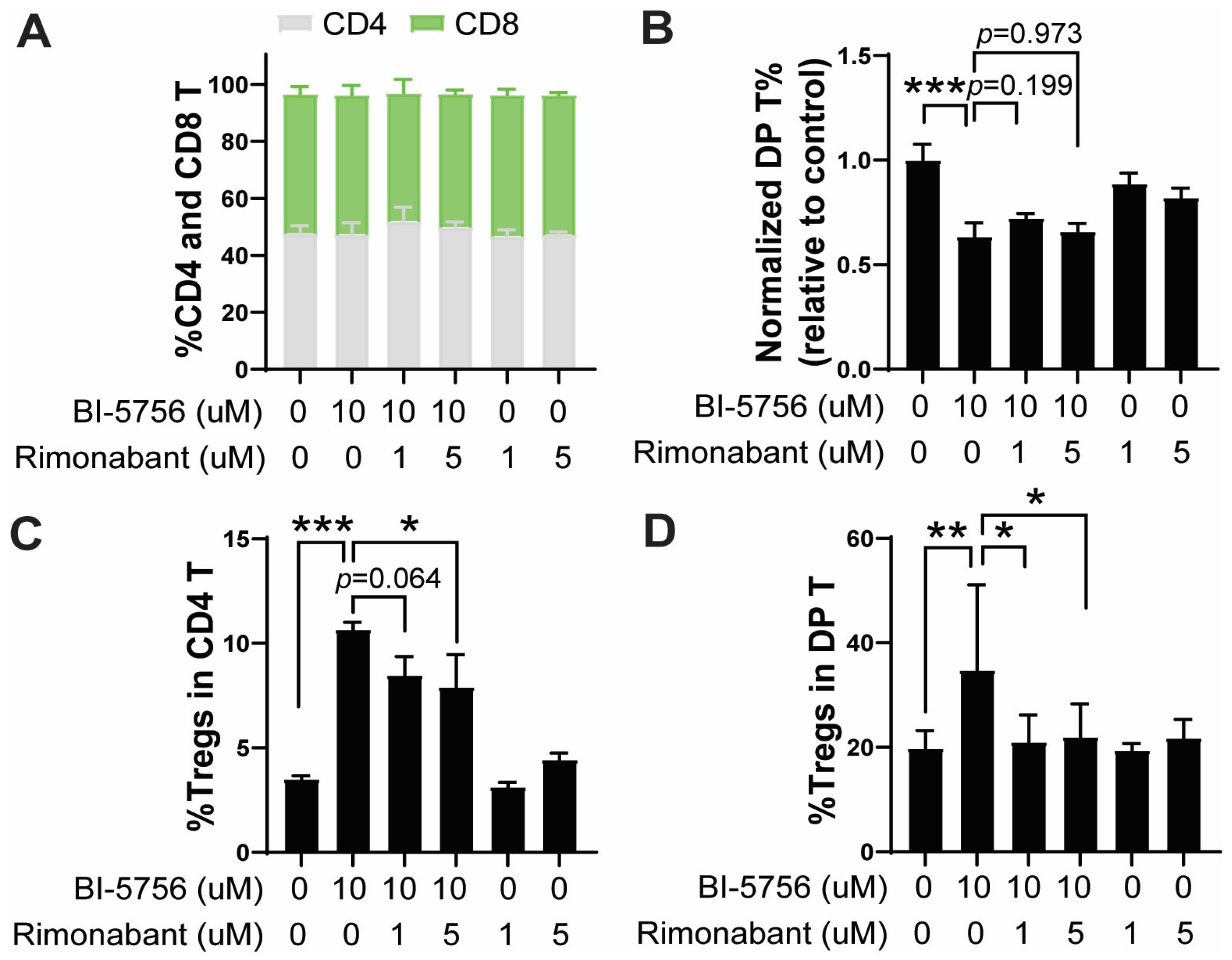
2.5. CB1 Signaling Contributes to BI-5756-Mediated GvHD Reduction by Upregulating Tregs
3. Discussion
4. Materials and Methods
4.1. Compounds
4.2. Mice
4.3. RNA Sequencing
4.4. Real-Time Quantitative PCR (qPCR)
4.5. Primary Murine T Cell Culture
4.6. Preclinical Mouse Model of Allo-HCT
4.7. Western Blotting
4.8. Flow Cytometry Analysis
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maurer, K.; Soiffer, R.J. The delicate balance of graft versus leukemia and graft versus host disease after allogeneic hematopoietic stem cell transplantation. Expert Rev. Hematol. 2023, 16, 943–962. [Google Scholar] [CrossRef]
- Smigiel, K.S.; Srivastava, S.; Stolley, J.M.; Campbell, D.J. Regulatory T-cell homeostasis: Steady-state maintenance and modulation during inflammation. Immunol. Rev. 2014, 259, 40–59. [Google Scholar] [CrossRef]
- Rocamora-Reverte, L.; Melzer, F.L.; Wurzner, R.; Weinberger, B. The Complex Role of Regulatory T Cells in Immunity and Aging. Front. Immunol. 2020, 11, 616949. [Google Scholar] [CrossRef]
- Meyer, E.H.; Pavlova, A.; Villar-Prados, A.; Bader, C.; Xie, B.; Muffly, L.; Kim, P.; Sutherland, K.; Bharadwaj, S.; Dahiya, S.; et al. Donor regulatory T-cell therapy to prevent graft-versus-host disease. Blood 2025, 145, 2012–2024. [Google Scholar] [CrossRef]
- Hoffmann, P.; Ermann, J.; Edinger, M.; Fathman, C.G.; Strober, S. Donor-type CD4(+)CD25(+) regulatory T cells suppress lethal acute graft-versus-host disease after allogeneic bone marrow transplantation. J. Exp. Med. 2002, 196, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.A.; Lees, C.J.; Blazar, B.R. The infusion of ex vivo activated and expanded CD4(+)CD25(+) immune regulatory cells inhibits graft-versus-host disease lethality. Blood 2002, 99, 3493–3499. [Google Scholar] [CrossRef] [PubMed]
- Edinger, M.; Hoffmann, P.; Ermann, J.; Drago, K.; Fathman, C.G.; Strober, S.; Negrin, R.S. CD4+CD25+ regulatory T cells preserve graft-versus-tumor activity while inhibiting graft-versus-host disease after bone marrow transplantation. Nat. Med. 2003, 9, 1144–1150. [Google Scholar] [CrossRef]
- Choi, J.; Ritchey, J.; Prior, J.L.; Holt, M.; Shannon, W.D.; Deych, E.; Piwnica-Worms, D.R.; DiPersio, J.F. In vivo administration of hypomethylating agents mitigate graft-versus-host disease without sacrificing graft-versus-leukemia. Blood 2010, 116, 129–139. [Google Scholar] [CrossRef]
- Elias, S.; Rudensky, A.Y. Therapeutic use of regulatory T cells for graft-versus-host disease. Br. J. Haematol. 2019, 187, 25–38. [Google Scholar] [CrossRef]
- Guo, W.W.; Su, X.H.; Wang, M.Y.; Han, M.Z.; Feng, X.M.; Jiang, E.L. Regulatory T Cells in GVHD Therapy. Front. Immunol. 2021, 12, 697854. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, O.; Ganguly, D. Endocannabinoids in immune regulation and immunopathologies. Immunology 2021, 164, 242–252. [Google Scholar] [CrossRef]
- Klein, T.W.; Newton, C.; Larsen, K.; Lu, L.; Perkins, I.; Nong, L.; Friedman, H. The cannabinoid system and immune modulation. J. Leukoc. Biol. 2003, 74, 486–496. [Google Scholar] [CrossRef]
- Yeshurun, M.; Shpilberg, O.; Herscovici, C.; Shargian, L.; Dreyer, J.; Peck, A.; Israeli, M.; Levy-Assaraf, M.; Gruenewald, T.; Mechoulam, R.; et al. Cannabidiol for the Prevention of Graft-versus-Host-Disease after Allogeneic Hematopoietic Cell Transplantation: Results of a Phase II Study. Biol. Blood Marrow Transplant. 2015, 21, 1770–1775. [Google Scholar] [CrossRef]
- Pandey, R.; Hegde, V.L.; Nagarkatti, M.; Nagarkatti, P.S. Targeting cannabinoid receptors as a novel approach in the treatment of graft-versus-host disease: Evidence from an experimental murine model. J. Pharmacol. Exp. Ther. 2011, 338, 819–828. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.H.; Meissler, J.J.; Fan, X.; Yu, D.; Adler, M.W.; Eisenstein, T.K. A CB2-Selective Cannabinoid Suppresses T-Cell Activities and Increases Tregs and IL-10. J. Neuroimmune Pharmacol. 2015, 10, 318–332. [Google Scholar] [CrossRef] [PubMed]
- Cabral, G.A.; Griffin-Thomas, L. Emerging role of the cannabinoid receptor CB2 in immune regulation: Therapeutic prospects for neuroinflammation. Expert Rev. Mol. Med. 2009, 11, e3. [Google Scholar] [CrossRef] [PubMed]
- Milligan, A.L.; Szabo-Pardi, T.A.; Burton, M.D. Cannabinoid Receptor Type 1 and Its Role as an Analgesic: An Opioid Alternative? J. Dual Diagn. 2020, 16, 106–119. [Google Scholar] [CrossRef]
- Mackie, K. Distribution of cannabinoid receptors in the central and peripheral nervous system. Handb. Exp. Pharmacol. 2005, 168, 299–325. [Google Scholar]
- Takheaw, N.; Jindaphun, K.; Pata, S.; Laopajon, W.; Kasinrerk, W. Cannabinoid Receptor 1 Agonist ACEA and Cannabinoid Receptor 2 Agonist GW833972A Attenuates Cell-Mediated Immunity by Different Biological Mechanisms. Cells 2023, 12, 848. [Google Scholar] [CrossRef]
- Angelina, A.; Perez-Diego, M.; Lopez-Abente, J.; Ruckert, B.; Nombela, I.; Akdis, M.; Martin-Fontecha, M.; Akdis, C.; Palomares, O. Cannabinoids induce functional Tregs by promoting tolerogenic DCs via autophagy and metabolic reprograming. Mucosal Immunol. 2022, 15, 96–108. [Google Scholar] [CrossRef]
- Chen, C.C.; Chang, Z.Y.; Tsai, F.J.; Chen, S.Y. Cannabinoid receptor type 1 antagonist inhibits progression of obesity-associated nonalcoholic steatohepatitis in a mouse model by remodulating immune system disturbances. Immun. Inflamm. Dis. 2020, 8, 544–558. [Google Scholar] [CrossRef]
- An, D.; Peigneur, S.; Hendrickx, L.A.; Tytgat, J. Targeting Cannabinoid Receptors: Current Status and Prospects of Natural Products. Int. J. Mol. Sci. 2020, 21, 5064. [Google Scholar] [CrossRef]
- Iannotti, F.A. Cannabinoids, Endocannabinoids, and Synthetic Cannabimimetic Molecules in Neuromuscular Disorders. Int. J. Mol. Sci. 2023, 25, 238. [Google Scholar] [CrossRef]
- Coronado-Alvarez, A.; Romero-Cordero, K.; Macias-Triana, L.; Tatum-Kuri, A.; Vera-Barron, A.; Budde, H.; Machado, S.; Yamamoto, T.; Imperatori, C.; Murillo-Rodriguez, E. The synthetic CB(1) cannabinoid receptor selective agonists: Putative medical uses and their legalization. Prog. Neuropsychopharmacol Biol. Psychiatry 2021, 110, 110301. [Google Scholar] [CrossRef]
- Pertwee, R.G. Receptors and channels targeted by synthetic cannabinoid receptor agonists and antagonists. Curr. Med. Chem. 2010, 17, 1360–1381. [Google Scholar] [CrossRef] [PubMed]
- OpnMe by Boehringer Ingelheim. Available online: https://www.opnme.com/molecules/cetp-inhibitor-bi-5756 (accessed on 8 August 2025).
- Nong, L.; Newton, C.; Friedman, H.; Klein, T.W. CB1 and CB2 receptor mRNA expression in human peripheral blood mononuclear cells (PBMC) from various donor types. Adv. Exp. Med. Biol. 2001, 493, 229–233. [Google Scholar] [PubMed]
- Borner, C.; Hollt, V.; Kraus, J. Activation of human T cells induces upregulation of cannabinoid receptor type 1 transcription. Neuroimmunomodulation 2007, 14, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Bando, Y.; Xiao, S.; Yang, K.; Anderson, A.C.; Kuchroo, V.K.; Khoury, S.J. CD11b+Ly-6C(hi) suppressive monocytes in experimental autoimmune encephalomyelitis. J. Immunol. 2007, 179, 5228–5237. [Google Scholar] [CrossRef]
- Ribeiro-Gomes, F.L.; Peters, N.C.; Debrabant, A.; Sacks, D.L. Efficient capture of infected neutrophils by dendritic cells in the skin inhibits the early anti-leishmania response. PLoS Pathog. 2012, 8, e1002536. [Google Scholar] [CrossRef]
- Chapman, N.M.; Zeng, H.; Nguyen, T.M.; Wang, Y.; Vogel, P.; Dhungana, Y.; Liu, X.; Neale, G.; Locasale, J.W.; Chi, H. mTOR coordinates transcriptional programs and mitochondrial metabolism of activated T(reg) subsets to protect tissue homeostasis. Nat. Commun. 2018, 9, 2095. [Google Scholar] [CrossRef]
- Nurmohamed, N.S.; Ditmarsch, M.; Kastelein, J.J.P. Cholesteryl ester transfer protein inhibitors: From high-density lipoprotein cholesterol to low-density lipoprotein cholesterol lowering agents? Cardiovasc. Res. 2022, 118, 2919–2931. [Google Scholar] [CrossRef] [PubMed]
- Brousseau, M.E.; Schaefer, E.J.; Wolfe, M.L.; Bloedon, L.T.; Digenio, A.G.; Clark, R.W.; Mancuso, J.P.; Rader, D.J. Effects of an inhibitor of cholesteryl ester transfer protein on HDL cholesterol. N. Engl. J. Med. 2004, 350, 1505–1515. [Google Scholar] [CrossRef]
- Chague, C.; Gautier, T.; Dal Zuffo, L.; Pais de Barros, J.P.; Wetzel, A.; Tarris, G.; Pallot, G.; Martin, L.; Valmary-Degano, S.; Deckert, V.; et al. High-density lipoprotein infusion protects from acute graft-versus-host disease in experimental allogeneic hematopoietic cell transplantation. Am. J. Transplant. 2022, 22, 1350–1361. [Google Scholar] [CrossRef]
- Bonacina, F.; Pirillo, A.; Catapano, A.L.; Norata, G.D. HDL in Immune-Inflammatory Responses: Implications beyond Cardiovascular Diseases. Cells 2021, 10, 1061. [Google Scholar] [CrossRef]
- Linton, M.F.; Yancey, P.G.; Tao, H.; Davies, S.S. HDL Function and Atherosclerosis: Reactive Dicarbonyls as Promising Targets of Therapy. Circ. Res. 2023, 132, 1521–1545. [Google Scholar] [CrossRef]
- Rivera-Franco, M.M.; Leon-Rodriguez, E.; Lastra-German, I.K.; Mendoza-Farias, A.A. Association of recipient and donor hypercholesterolemia prior allogeneic stem cell transplantation and graft-versus-host disease. Leuk. Res. 2018, 72, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lim, S.; Kim, B.; Ritchey, J.; Vij, K.; Prior, J.; Marsala, L.; Stoner, A.; Gao, F.; Achilefu, S.; et al. S100A9 upregulated by IFNGR signaling blockade functions as a novel GVHD suppressor without compromising GVL in mice. Blood 2023, 141, 945–950. [Google Scholar] [CrossRef]
- Kim, S.; Ashami, K.; Lim, S.; Staser, K.; Vij, K.; Santhanam, S.; Ritchey, J.; Peterson, S.; Gao, F.; Ciorba, M.A.; et al. Baricitinib prevents GvHD by increasing Tregs via JAK3 and treats established GvHD by promoting intestinal tissue repair via EGFR. Leukemia 2022, 36, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lim, S.; Razmkhah, F.; Choi, J. Overexpression of S100A9 in donor T cells is associated with reconstitution of gut microbiota and outcome of allogeneic hematopoietic stem cell transplantation. Blood Res. 2023, 58, 105–111. [Google Scholar] [CrossRef]
- Kim, S.; Ruminski, P.; Singh, M.; Staser, K.; Ashami, K.; Ritchey, J.; Lim, S.; DiPersio, J.F.; Choi, J. Novel JAK Inhibitors to Reduce Graft-Versus-Host Disease after Allogeneic Hematopoietic Cell Transplantation in a Preclinical Mouse Model. Molecules 2024, 29, 1801. [Google Scholar] [CrossRef]
- Choi, J.; Cooper, M.L.; Staser, K.; Ashami, K.; Vij, K.R.; Wang, B.; Marsala, L.; Niswonger, J.; Ritchey, J.; Alahmari, B.; et al. Baricitinib-induced blockade of interferon gamma receptor and interleukin-6 receptor for the prevention and treatment of graft-versus-host disease. Leukemia 2018, 32, 2483–2494. [Google Scholar] [CrossRef] [PubMed]
- Cooke, K.R.; Kobzik, L.; Martin, T.R.; Brewer, J.; Delmonte, J., Jr.; Crawford, J.M.; Ferrara, J.L. An experimental model of idiopathic pneumonia syndrome after bone marrow transplantation: I. The roles of minor H antigens and endotoxin. Blood 1996, 88, 3230–3239. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.; Dania, A.-J.; Lim, S.; Choi, J. BI-5756 Reduces Graft-Versus-Host Disease Through CB1-Mediated Treg Upregulation. Molecules 2025, 30, 3517. https://doi.org/10.3390/molecules30173517
Kim S, Dania A-J, Lim S, Choi J. BI-5756 Reduces Graft-Versus-Host Disease Through CB1-Mediated Treg Upregulation. Molecules. 2025; 30(17):3517. https://doi.org/10.3390/molecules30173517
Chicago/Turabian StyleKim, Sena, Abdul-Jalil Dania, Sora Lim, and Jaebok Choi. 2025. "BI-5756 Reduces Graft-Versus-Host Disease Through CB1-Mediated Treg Upregulation" Molecules 30, no. 17: 3517. https://doi.org/10.3390/molecules30173517
APA StyleKim, S., Dania, A.-J., Lim, S., & Choi, J. (2025). BI-5756 Reduces Graft-Versus-Host Disease Through CB1-Mediated Treg Upregulation. Molecules, 30(17), 3517. https://doi.org/10.3390/molecules30173517





