Recent Advancements in Photocatalytic Synthesis of Five Membered Nitrogen Heterocycles and Their Derivatives
Abstract
1. Introduction
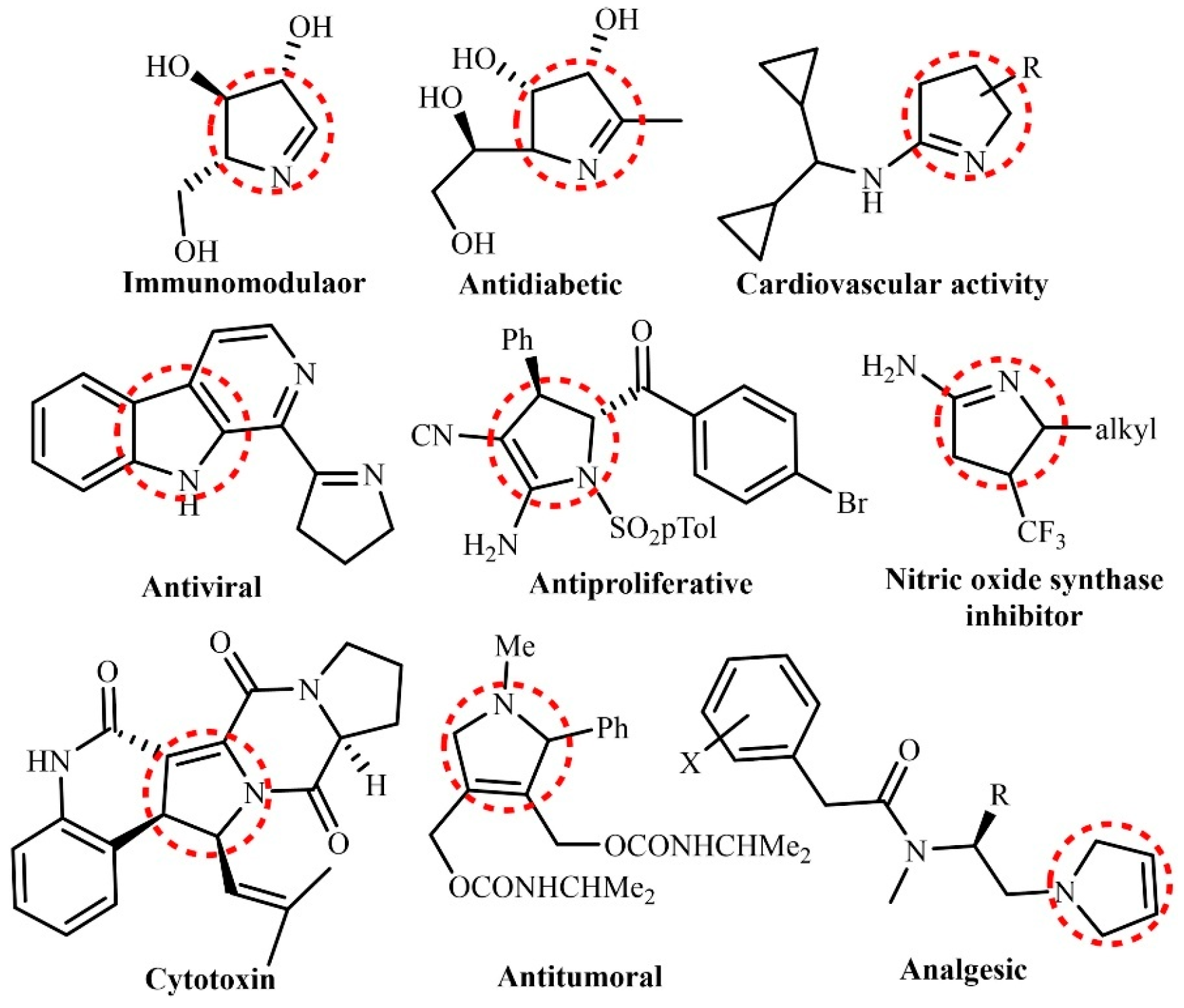
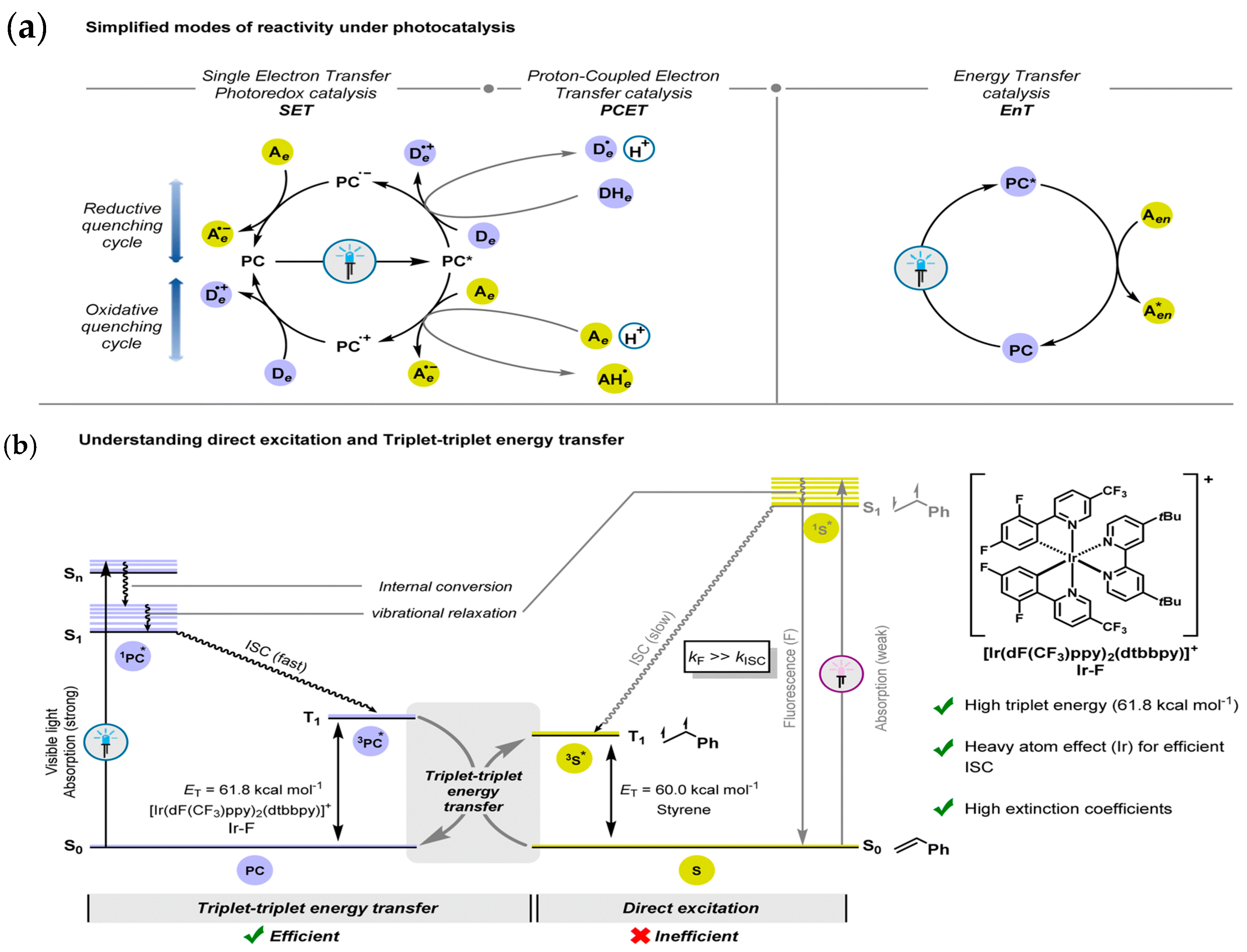
2. Light-Driven Synthesis of Heterocycles
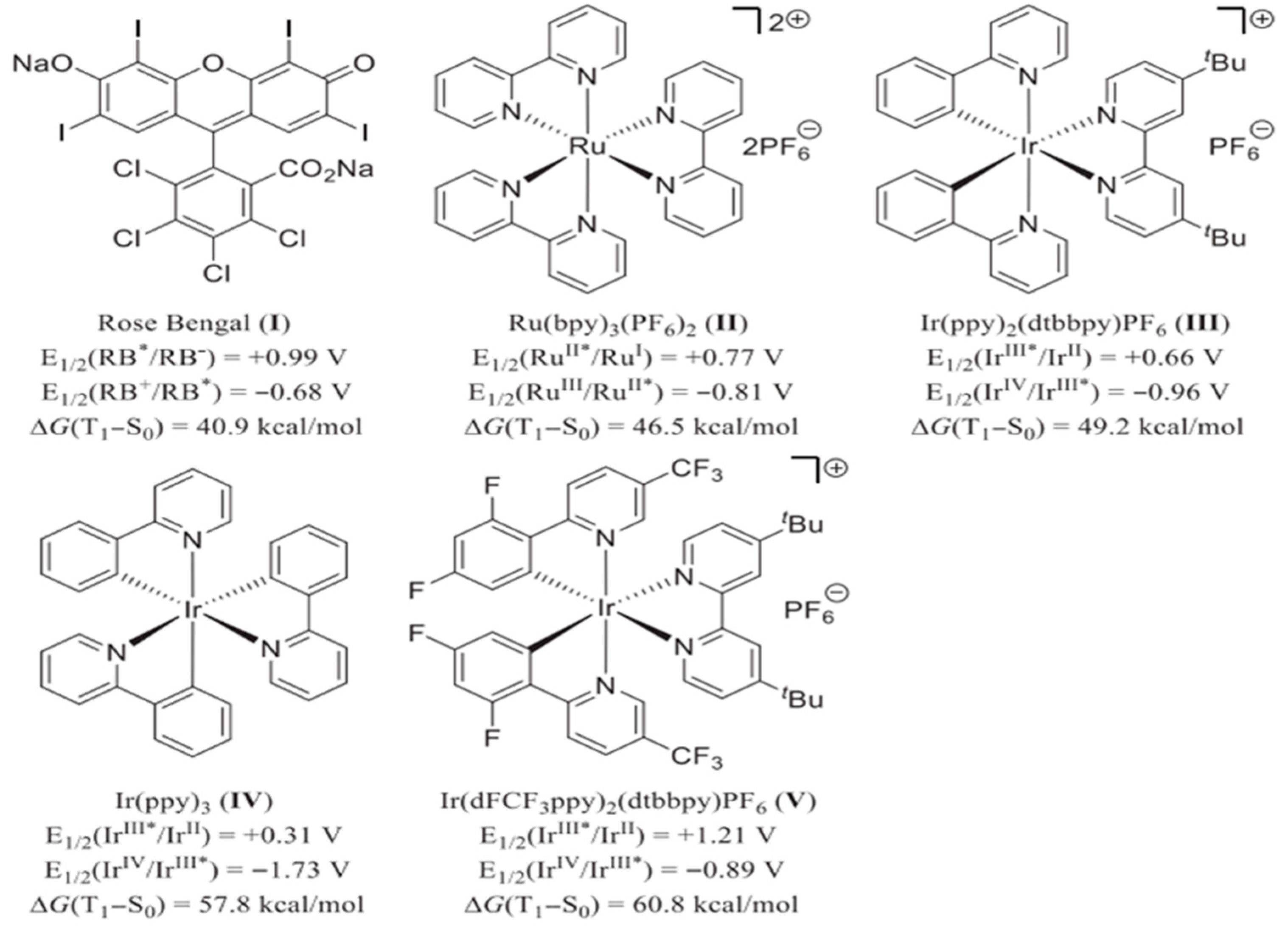
2.1. Photocatalysts for Driving Heterocycle Synthesis
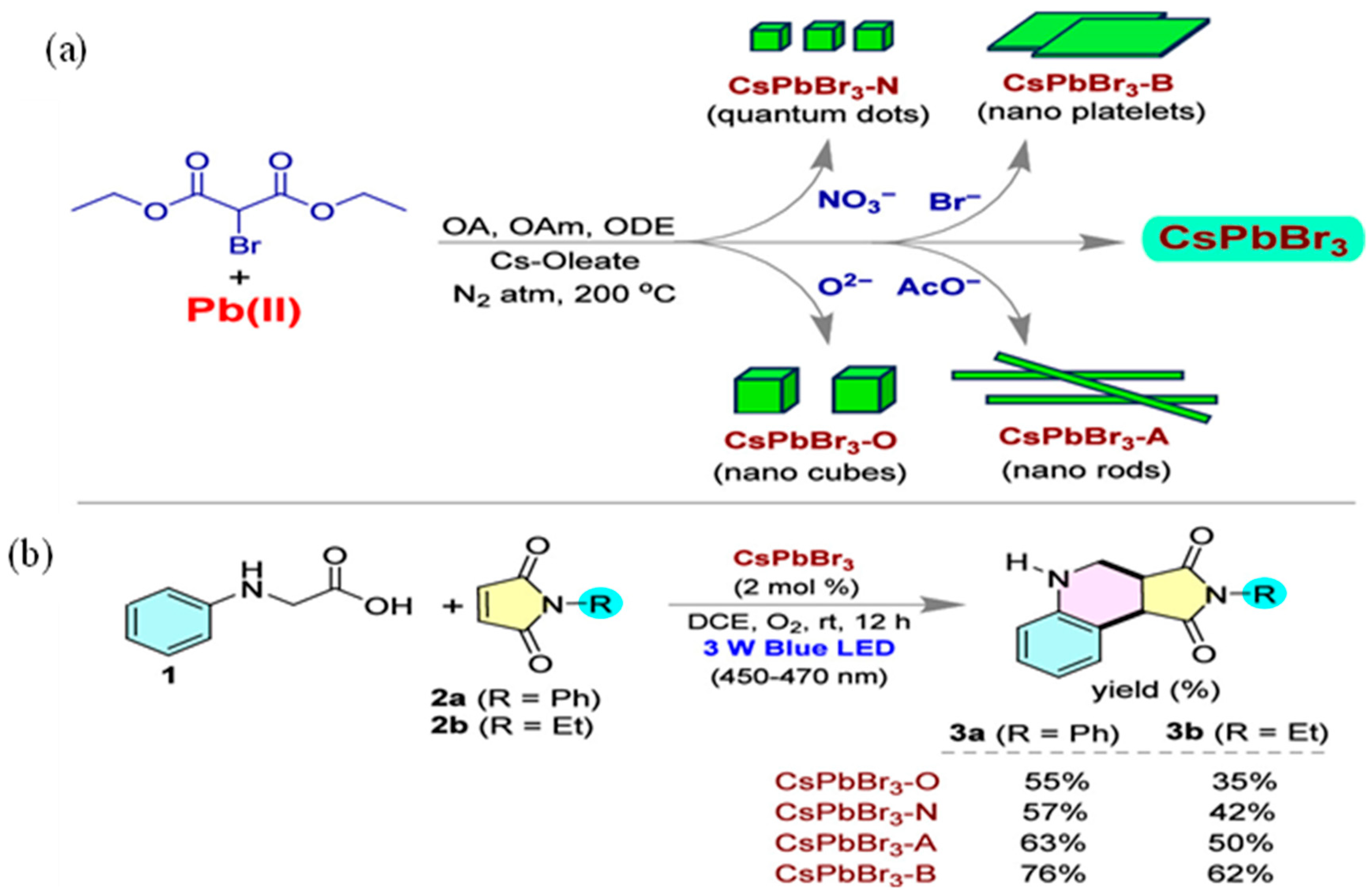
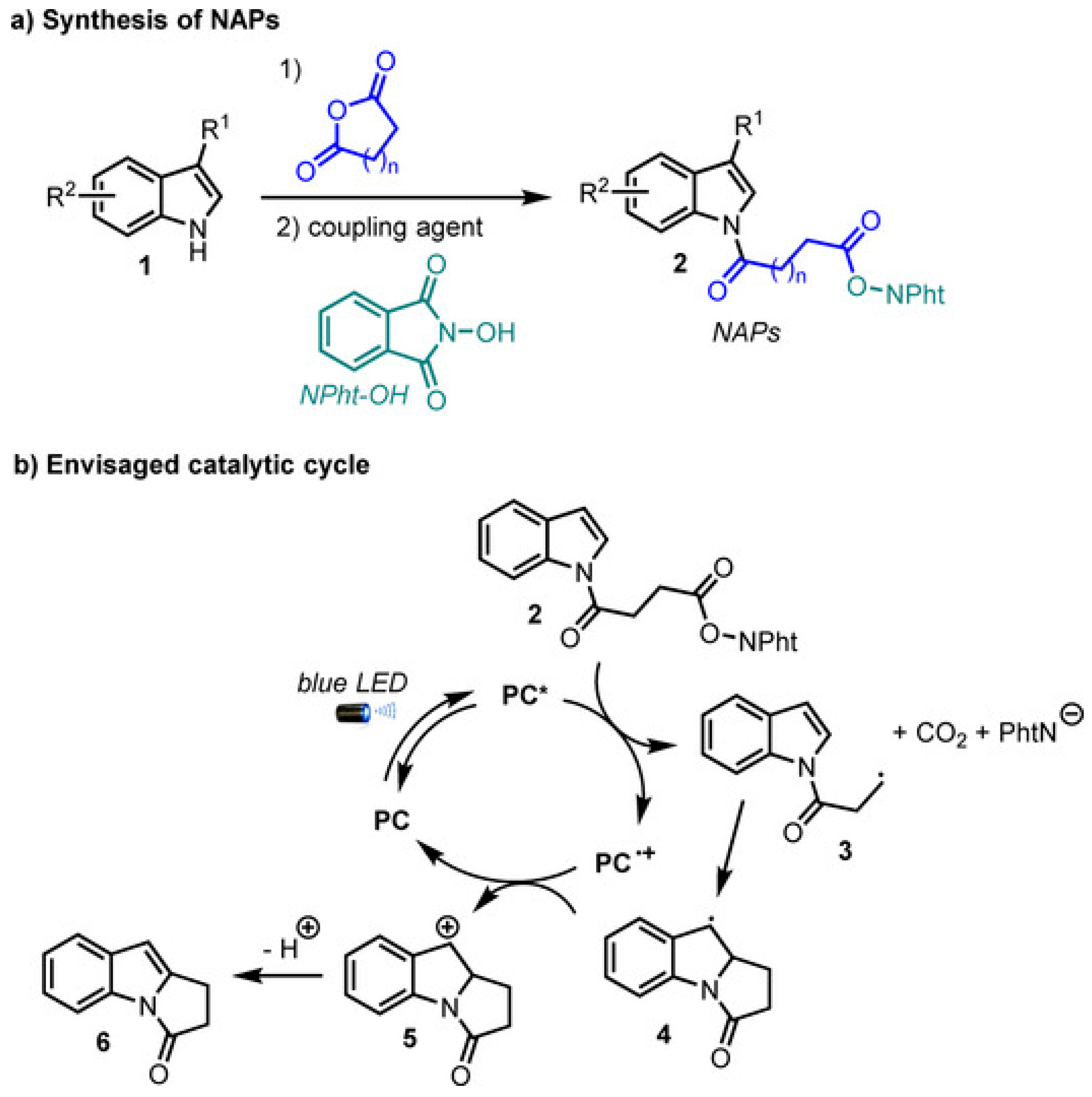
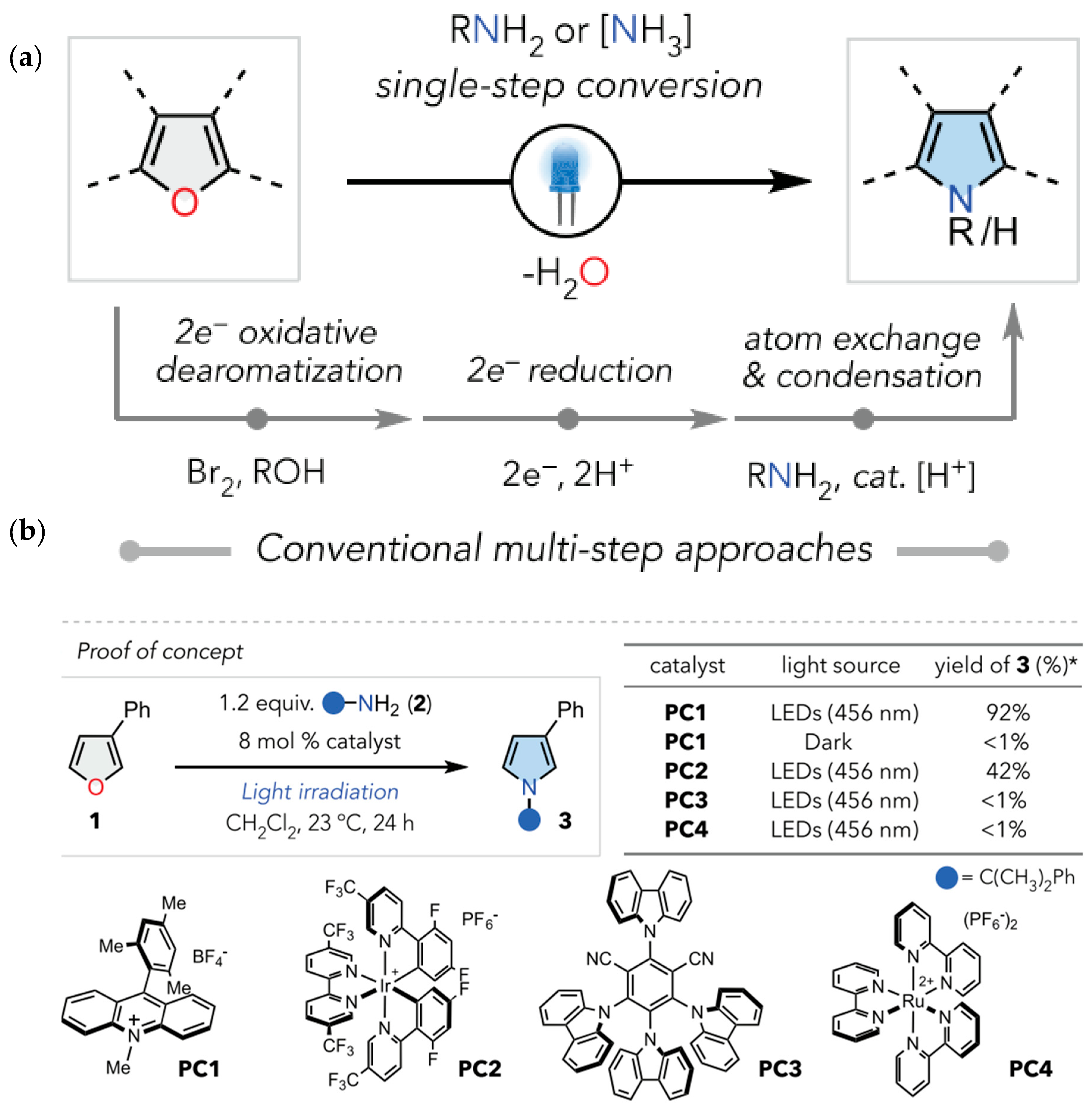
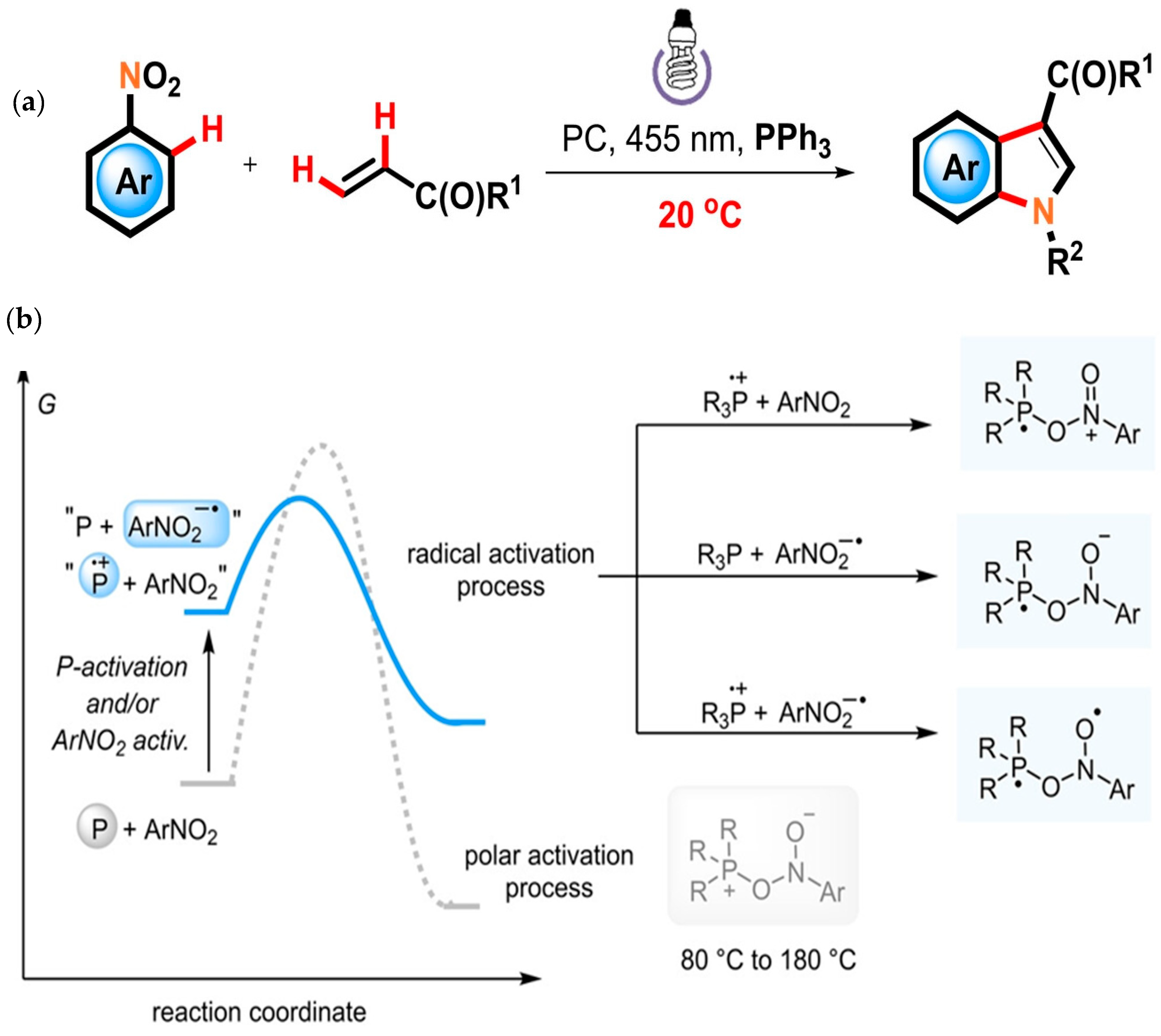
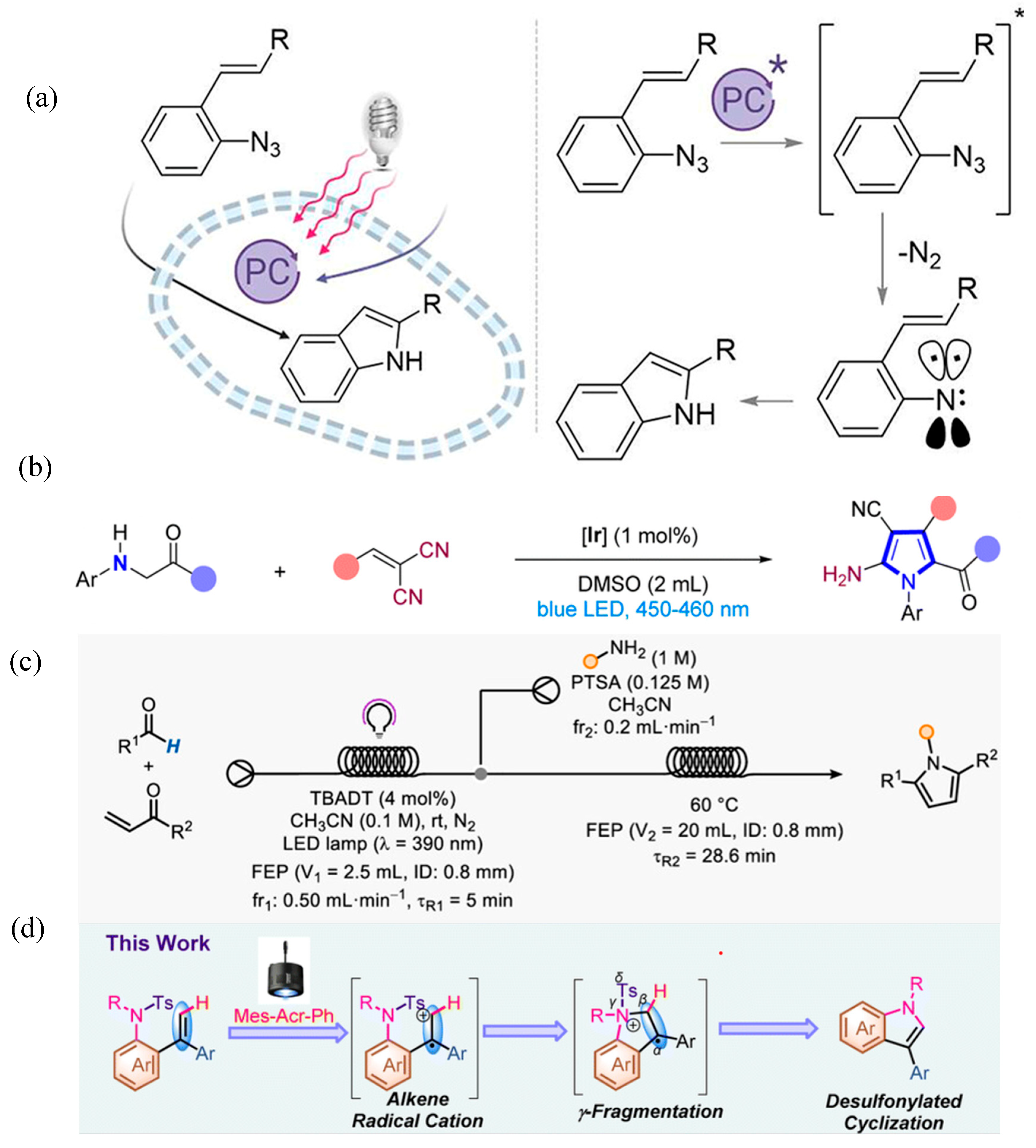
2.2. Advancement in Heterocyclic Synthesis
Five Membered Nitrogen-Containing Heterocycles
2.3. Comparison of Various Pathways
3. Challenges, Limitations and Future Outlook
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Han, Z.; Ma, S. Understanding the working principle of photocatalysts for designing high-performance antibacterial agents. Coord. Chem. Rev. 2025, 537, 216715. [Google Scholar] [CrossRef]
- Haider, Z.; Zheng, J.Y.; Kang, Y.S. Surfactant free fabrication and improved charge carrier separation induced enhanced photocatalytic activity of {001} facet exposed unique octagonal BiOCl nanosheets. Phys. Chem. Chem. Phys. 2016, 18, 19595–19604. [Google Scholar] [CrossRef]
- Zeeshan, H.; Soo, K.Y. Facile Preparation of Hierarchical TiO2 Nano Structures: Growth Mechanism and Enhanced Photocatalytic H2 Production from Water Splitting Using Methanol as a Sacrificial Reagent. ACS Appl. Mater. Interfaces 2014, 6, 10342–10352. [Google Scholar] [CrossRef]
- Zheng, J.Y.; Haider, Z.; Van, T.K.; Pawar, A.U.; Kang, M.J.; Kim, C.W.; Kang, Y.S. Tuning of the crystal engineering and photoelectrochemical properties of crystalline tungsten oxide for optoelectronic device applications. CrystEngComm 2015, 17, 6070–6093. [Google Scholar] [CrossRef]
- Zheng, J.Y.; Song, G.; Hong, J.; Van, T.K.; Pawar, A.U.; Kim, D.Y.; Kim, C.W.; Haider, Z.; Kang, Y.S. Facile fabrication of WO3 nanoplates thin films with dominant crystal facet of (002) for water splitting. Cryst. Growth Des. 2014, 14, 6057–6066. [Google Scholar] [CrossRef]
- Khan, M.D.; Fareed, I.; Khan, M.F.; Rehman, Z.U.; Ayoub, U.; Ahmed, A.; Butt, F.K. Novel N-doped ZnO and O-doped g-C3N4 heterojunction: Enhanced photocatalytic degradation and robust electrochemical biosensing of ascorbic acid. Diam. Relat. Mater. 2025, 151, 111752. [Google Scholar] [CrossRef]
- Rabeel, M.; Javed, S.; Khan, R.; Akram, M.A.; Rehman, S.; Kim, D.-K.; Khan, M.F. Controlling the wettability of ZnO thin films by spray pyrolysis for photocatalytic applications. Materials 2022, 15, 3364. [Google Scholar] [CrossRef] [PubMed]
- Haider, Z.; Cho, H.-I.; Moon, G.-H.; Kim, H.-I. Minireview: Selective production of hydrogen peroxide as a clean oxidant over structurally tailored carbon nitride photocatalysts. Catal. Today 2019, 335, 55–64. [Google Scholar] [CrossRef]
- Kumar, P.S.M.; Lee, D.-Y.; Cho, H.; Son, S.H.; Jae, J.; Kim, J.R.; Kwon, O.S.; Haider, Z.; Kim, H.-I. Biomass-derived carbon for solar H2O2 production: Current trends and future directions. Mater. Today Energy 2025, 49, 101840. [Google Scholar] [CrossRef]
- Lee, D.-Y.; Haider, Z.; Krishnan, S.K.; Kanagaraj, T.; Son, S.H.; Jae, J.; Kim, J.R.; Kumar, P.S.M.; Kim, H.-I. Oxygen-enriched carbon quantum dots from coffee waste: Extremely active organic photocatalyst for sustainable solar-to-H2O2 conversion. Chemosphere 2024, 361, 142330. [Google Scholar] [CrossRef]
- Muhmood, T.; Ahmad, I.; Haider, Z.; Haider, S.K.; Shahzadi, N.; Aftab, A.; Ahmed, S.; Ahmad, F. Graphene-like graphitic carbon nitride (g-C3N4) as a semiconductor photocatalyst: Properties, classification, and defects engineering approaches. Mater. Today Sustain. 2024, 25, 100633. [Google Scholar] [CrossRef]
- Son, S.; Haider, Z.; Lee, D.-Y.; Kim, S.; Thai, N.V.; Kim, H.-I. Ambivalent photocatalytic strategies for plastic waste conversion: Alkalinized carbon nitride for H2O2 production and complete mineralization. Appl. Catal. B Environ. Energy 2025, 365, 124847. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, X.; Gao, F.; Jiang, L.; Wang, D.; Pan, H. From single atom photocatalysts to synergistic photocatalysts: Design principles and applications. Adv. Funct. Mater. 2025, 35, 2418427. [Google Scholar] [CrossRef]
- Han, M.; Han, M.; Ma, J.; Du, M.; Zhu, Q.; Qiu, B. Photocatalytic Upcycling of Plastic Waste into Value-Added Chemicals. Electron 2025, 3, e70005. [Google Scholar] [CrossRef]
- Aziz, M.H.; Latif, M.; Hassan, R.U.; Naeem, T.; Asif, M.; Alam, M.; Ali, S.M.; Zeng, Q. Photocatalytic breakdown of tetracycline via Z-scheme BiFeO3/Ag/Cr2O3 nanocomposite under visible light irradiation: Degradation mechanism, toxicity evaluation and antibacterial activity. Surf. Interfaces 2025, 61, 106151. [Google Scholar] [CrossRef]
- Rasool, B.; Aziz, M.H.; Asif, M.; Hassan, R.U.; Fatima, M.; Latif, M.; Alanazi, F.K.; Alshehri, N.A.; Almarri, H.M.; Huang, Q. Optimizing the degradation of tetracycline antibiotics with an efficient rGO/Cu0.5Mn0.5Fe2O4/ZnO photocatalyst under visible light irradiation. Mater. Lett. 2025, 382, 137859. [Google Scholar] [CrossRef]
- Aziz, M.H.; Latif, M.; Hassan, R.U.; Asif, M.; Wahab, R.; Huang, Q. ZnFe2O4/Cr2O3/MXene nanocomposite photocatalyst stimulates tetracycline antibiotic degradation under visible light irradiation: Toxicity evaluation and degradation mechanism. Sep. Purif. Technol. 2025, 354, 128866. [Google Scholar] [CrossRef]
- Yang, L.; Sivasankaran, R.P.; Song, M.K.; Pawar, A.U.; Lee, D.K.; Kang, Y.S. Highly Selective Solar CO2 Conversion into Formic Acid in Nickel-Perylene-C3N4 Semiconductor Photocatalyst. Adv. Energy Mater. 2025, 15, 2402798. [Google Scholar] [CrossRef]
- Naz, G.; Khursheed, H.; Hussain, A.; Sharif, H.M.A.; Arshad, M.; Butt, F.K.; Nawaz, M.Z.; Hendi, A.A.; Almoneef, M.M.; Awad, M.A. Silver nanogranules-decorated ZnO hybrid nanostructures with enhanced UV photoresponses. Mater. Today Commun. 2024, 40, 110106. [Google Scholar] [CrossRef]
- Yang, C.; Hou, L.; Sharif, H.M.A.; Wang, Y.; Cai, Y.; Li, C.; Hou, L.A. Tailored anthraquinone-based covalent organic frameworks boosted atrazine degradation through peroxymonosulfate activation driven by visible light. Sep. Purif. Technol. 2024, 350, 127941. [Google Scholar] [CrossRef]
- Sasikumar, K.; Ju, H. Recent advances in vanadate-based materials for photocatalytic hydrogen production. Molecules 2025, 30, 789. [Google Scholar] [CrossRef] [PubMed]
- Sasikumar, K.; Rajamanikandan, R.; Ju, H. Z-scheme charge transfer heterostructure with NiV2O6@ gC3N4 nanocomposite: A competent photocatalyst for boosting the photodegradation of antibiotics. J. Taiwan Inst. Chem. Eng. 2025, 168, 105960. [Google Scholar] [CrossRef]
- Sasikumar, K.; Rajamanikandan, R.; Ju, H. Construction of Z-Scheme ZIF67/NiMoO4 heterojunction for enhanced photocatalytic degradation of antibiotic pollutants. Materials 2024, 17, 6225. [Google Scholar] [CrossRef]
- Sasikumar, K.; Rajamanikandan, R.; Ju, H. A high-performance NiMoO4/g-C3N4 direct Z-scheme heterojunction photocatalyst for the degradation of organic pollutants. Surf. Interfaces 2023, 42, 103389. [Google Scholar] [CrossRef]
- Luo, X.-N.; Yang, D.-Q.; Wu, H.-L.; Feng, J.-Y.; Du, S.; He, Y.; Yi, R.; Zhang, J.-Q.; Tang, K.; Wei, W. The construction of aromatic rings by photocatalytic radical-induced cyclization reactions. Org. Chem. Front. 2025; advance article. [Google Scholar]
- Xia, H.; Yang, H.; Yue, J.; Ma, C.; Fan, X.; Chen, R. Visible-Light-Driven Photocatalytic Synthesis of Polysubstituted Pyrrolidines via Desulfurative Tandem Cyclization of Thiols with 1, 6-Enynes. J. Org. Chem. 2025, 90, 9063–9075. [Google Scholar] [CrossRef]
- Qin, H.-N.; Jiang, H.-W.; Zhao, Y.; Qurban, S.; Wang, K.-C.; Xu, P.-F. Photocatalytic [3+ 2]-annulation via sodium tetraarylborate: A fundamental approach for synthesizing 1, 4, 2-diazaborole analogs. Chem. Sci. 2025, 16, 2837–2842. [Google Scholar] [CrossRef]
- Ghara, S.; Barik, P.; Ghosh, S.; Ghosh, S.; Mandal, A.; Pramanik, C.; Ikbal, M.; Dhara, S.; Samanta, S. UV/visible light-promoted external photocatalyst-free transformations: A Decade’s Journey of N-heterocycles and their functionalisation. Org. Chem. Front. 2025, 12, 2790–2837. [Google Scholar] [CrossRef]
- Zhao, Y.; Yuan, J.; Zhu, L.; Fang, Y. Photocatalytic synthesis of small-molecule drugs by porous framework materials. Chin. Chem. Lett. 2024, 35, 109065. [Google Scholar] [CrossRef]
- Li, S.; Ma, R.; Tu, C.; Zhang, W.; Li, R.; Zhao, Y.; Zhang, K.A. Programmed Charge Transfer in Conjugated Polymers with Pendant Benzothiadiazole Acceptor for Simultaneous Photocatalytic H2O2 Production and Organic Synthesis. Angew. Chem. Int. Ed. 2025, 64, e202421040. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhou, D.; Tian, K.; Chen, G.; Li, Y.; Liu, S.F.; Lee, S.-T.; Yan, J. Asymmetric Zn0. 5Cd0. 5S Loading of CoS x for Full-Space Electric Field Photocatalytic Hydrogen Production and Synergistic Organic Synthesis. ACS Catal. 2025, 15, 3660–3673. [Google Scholar] [CrossRef]
- Singh, S.; Hamid, Z.; Babu, R.; Gómez-Graña, S.; Hu, X.; McCulloch, I.; Hoye, R.L.; Rao, V.G.; Polavarapu, L. Halide Perovskite Photocatalysts for Clean Fuel Production and Organic Synthesis: Opportunities and Challenges. Adv. Mater. 2025, 37, e2419603. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.-C.; Cai, W.; Ji, H.-T.; Ou, L.-J.; He, W.-M. Recent advances in semi-heterogenous photocatalysis in organic synthesis. Chin. Chem. Lett. 2025, 36, 110469. [Google Scholar] [CrossRef]
- Mohamadpour, F.; Amani, A.M. Photocatalytic systems: Reactions; mechanism; applications. RSC Adv. 2024, 14, 20609–20645. [Google Scholar] [CrossRef]
- Yang, S.; Tan, X.; Liu, D.; Jiang, H.; Wu, W. Photoredox-Catalyzed Radical Alkylation/Cyclization of Isocyanides with Organoboron Reagents for Diversified Synthesis of N-Heteroaromatics. Adv. Synth. Catal. 2025, 367, e202500031. [Google Scholar] [CrossRef]
- Bhuyan, K.; Borang, L.; Jamatia, R. Visible Light-Mediated Radical Decarboxylative Strategies for the Synthesis of Heterocyclic Compounds. Asian J. Org. Chem. 2025, 14, e202400493. [Google Scholar] [CrossRef]
- Li, F.X.; Liu, Y.; Zhang, K.; Wang, C.; Yu, J.X.; He, X.J.; Liang, G.; Hou, H.Y.; Tung, C.H.; Wu, L.Z. Multifunctionalization of N-Heterocyclic Carbene Boranes by a Single Metal-free Photocatalyst. Adv. Synth. Catal. 2025, 367, e202401440. [Google Scholar] [CrossRef]
- He, J.; Han, B.; Xian, C.; Hu, Z.; Fang, T.; Zhang, Z. Hydrogen-Bond-Mediated Formation of C−N or C=N Bond during Photocatalytic Reductive Coupling Reaction over CdS Nanosheets. Angew. Chem. Int. Ed. 2024, 63, e202404515. [Google Scholar] [CrossRef]
- Feng, J.; Zhang, Y.; Wang, X.; Liu, J.; Benazzi, V.; Lu, K.; Zhao, X.; Protti, S. Recent Advances in Visible-Light-Driven C−S Bond Formation. Adv. Synth. Catal. 2023, 365, 3413–3431. [Google Scholar] [CrossRef]
- Velasco-Rubio, Á.; Martínez-Balart, P.; Álvarez-Constantino, A.M.; Fañanás-Mastral, M. C–C bond formation via photocatalytic direct functionalization of simple alkanes. Chem. Commun. 2023, 59, 9424–9444. [Google Scholar] [CrossRef]
- D’Avino, C.; Gutiérrez, S.; Feldhaus, M.J.; Tomás-Gamasa, M.; Mascareñas, J.L. Intracellular synthesis of indoles enabled by visible-light photocatalysis. J. Am. Chem. Soc. 2024, 146, 2895–2900. [Google Scholar] [CrossRef]
- Wu, C.-J.; Shao, M.-Z.; Kan, J.-L.; Liang, W.-J.; Li, T.-R.; Niu, L.-J.; Wang, H.-J.; Geng, Y.; Dong, Y.-B. Photocatalytic Synthesis of Quinoline-Linked Covalent Organic Frameworks via Three-Component Povarov Reaction. ACS Mater. Lett. 2024, 6, 5016–5022. [Google Scholar] [CrossRef]
- Kim, D.; You, J.; Lee, D.H.; Hong, H.; Kim, D.; Park, Y. Photocatalytic furan-to-pyrrole conversion. Science 2024, 386, 99–105. [Google Scholar] [CrossRef]
- Das, A.; Gouthaman, S.; Thomas, K.J. Photocatalysis as a green alternative toolkit for the construction of nitrogen-enriched heterocycles via the direct and indirect activation of alkynes/alkenes. Green Chem. 2024, 26, 1223–1280. [Google Scholar] [CrossRef]
- Abdellah, I.M.; Barqi, M.M.; Zaky, O.S.; Al-Ghorbani, M.; Eletmany, M.R. Short Review on the Synthesis and Applications of Heterocyclic Quinones. Int. J. Innov. Sci. Res. Technol. 2024, 9, 1804–1809. [Google Scholar]
- Emanuele, L.; D’Auria, M. The Use of Heterocyclic Azo Dyes on Different Textile Materials: A Review. Organics 2024, 5, 277–289. [Google Scholar] [CrossRef]
- Reischauer, S.; Pieber, B. Emerging concepts in photocatalytic organic synthesis. iScience 2021, 24, 102209. [Google Scholar] [CrossRef]
- Kesharwani, N.; Garima, K.; Srivastava, V.; Singh, P.P.; Singh, P.K. Visible-Light Driven Photocatalytic Strategies for the Synthesis of Pyrrolines. Asian J. Org. Chem. 2025, 14, e202400561. [Google Scholar] [CrossRef]
- Dutta, S.; Erchinger, J.E.; Strieth-Kalthoff, F.; Kleinmans, R.; Glorius, F. Energy transfer photocatalysis: Exciting modes of reactivity. Chem. Soc. Rev. 2024, 53, 1068–1089. [Google Scholar] [CrossRef]
- Ghosh, T.; Santra, S.; Zyryanov, G.V.; Ranu, B.C. Recent Developments on the Synthesis of Oxygen-and Sulfur-containing Heterocycles and their Derivatives under Visible Light Induced Reactions. Curr. Top. Med. Chem. 2025, 25, 124–140. [Google Scholar] [CrossRef]
- Bissonnette, N.B.; Ellis, J.M.; Hamann, L.G.; Romanov-Michailidis, F. Expedient access to saturated nitrogen heterocycles by photoredox cyclization of imino-tethered dihydropyridines. Chem. Sci. 2019, 10, 9591–9596. [Google Scholar] [CrossRef] [PubMed]
- Strieth-Kalthoff, F.; Glorius, F. Triplet energy transfer photocatalysis: Unlocking the next level. Chem 2020, 6, 1888–1903. [Google Scholar] [CrossRef]
- Ma, N.; Zhao, M.; Liu, H.; Zhang, C.; Liv, P.; Zhang, J. Novel magnetic photocatalyst Fe3O4/AgSbO3 for the efficient synthesis of a range of 4H-chromenes as important pharmaceuticals in treating diabetes. Mater. Technol. 2025, 40, 2512418. [Google Scholar] [CrossRef]
- López-Magano, A.; Daliran, S.; Oveisi, A.R.; Mas-Ballesté, R.; Dhakshinamoorthy, A.; Alemán, J.; Garcia, H.; Luque, R. Recent advances in the use of covalent organic frameworks as heterogenous photocatalysts in organic synthesis. Adv. Mater. 2023, 35, 2209475. [Google Scholar] [CrossRef]
- Arunachalapandi, M.; Duraivathi, C. Simple, Effective and Green Synthesis of N-Heterocycles Using g-C3N4 Nanosheets as Photocatalyst Under Visible Light Irradiation. Catal. Lett. 2025, 155, 222. [Google Scholar] [CrossRef]
- Martynova, E.A.; Voloshkin, V.A.; Guillet, S.G.; Bru, F.; Beliš, M.; Van Hecke, K.; Cazin, C.S.; Nolan, S.P. Energy transfer (EnT) photocatalysis enabled by gold-N-heterocyclic carbene (NHC) complexes. Chem. Sci. 2022, 13, 6852–6857. [Google Scholar] [CrossRef]
- Zhao, Y.; Voloshkin, V.A.; Martynova, E.A.; Maity, B.; Cavallo, L.; Nolan, S.P. Synthesis of cyclohepta [b] indoles via gold mediated energy transfer photocatalysis. Chem. Commun. 2024, 60, 3174–3177. [Google Scholar] [CrossRef]
- Zhu, M.; Zheng, C.; Zhang, X.; You, S.-L. Synthesis of cyclobutane-fused angular tetracyclic spiroindolines via visible-light-promoted intramolecular dearomatization of indole derivatives. J. Am. Chem. Soc. 2019, 141, 2636–2644. [Google Scholar] [CrossRef]
- Manna, A.; Nayek, P.; Mal, P. Tuning Dimensions of CsPbBr3 Nanocrystals through Pb (II) Counter Anions: A Dance of Dimensions and Product Selectivity in Visible-Light Photocatalysis. ACS Energy Lett. 2025, 10, 1499–1507. [Google Scholar] [CrossRef]
- Li, S.; Li, L.; Li, Y.; Dai, L.; Liu, C.; Liu, Y.; Li, J.; Lv, J.; Li, P.; Wang, B. Fully Conjugated Donor–Acceptor Covalent Organic Frameworks for Photocatalytic Oxidative Amine Coupling and Thioamide Cyclization. ACS Catal. 2020, 10, 8717–8726. [Google Scholar] [CrossRef]
- Yang, M.; Lian, R.; Zhang, X.; Wang, C.; Cheng, J.; Wang, X. Photocatalytic cyclization of nitrogen-centered radicals with carbon nitride through promoting substrate/catalyst interaction. Nat. Commun. 2022, 13, 4900. [Google Scholar] [CrossRef] [PubMed]
- Kleinmans, R.; Pinkert, T.; Dutta, S.; Paulisch, T.O.; Keum, H.; Daniliuc, C.G.; Glorius, F. Intermolecular [2 π+ 2 σ]-photocycloaddition enabled by triplet energy transfer. Nature 2022, 605, 477–482. [Google Scholar] [CrossRef]
- Hoving, M.; Haaksma, J.J.; Stoppel, A.; Chronc, L.; Hoffmann, J.; Beil, S.B. Triplet Energy Transfer Mechanism in Copper Photocatalytic N-and O-Methylation. Chem.–A Eur. J. 2024, 30, e202400560. [Google Scholar] [CrossRef]
- Chemmangat, A.; Chakkamalayath, J.; DuBose, J.T.; Kamat, P.V. Tuning energy transfer pathways in halide perovskite–dye hybrids through bandgap engineering. J. Am. Chem. Soc. 2024, 146, 3352–3362. [Google Scholar] [CrossRef]
- Kayal, A.; De, M. Organo-Soluble MoS2 Quantum Dots Mediated Photocatalytic Generation of Imminium Intermediate for Effective Synthesis of Bis (indolyl) Methanes Derivatives. ChemCatChem 2024, 30, e00977. [Google Scholar] [CrossRef]
- Liu, L.-J.; Lei, X.; Guo, J.; Mo, X.; Lao, Y.; Zhuang, S.; Zeng, H.; Yang, S.; Zhao, Y.; Xu, W.W. Johnson Solid Gold Nanoclusters as Heterogeneous Visible-Light Photocatalysts for Fukuyama Indole Synthesis. J. Am. Chem. Soc. 2025, 147, 27981–27991. [Google Scholar] [CrossRef]
- Dai, L.-F.; Jiang, Y.-X.; Yu, D.-L.; Sun, G.-Q.; Yan, S.-S.; Zhang, W.; Ye, J.-H.; Yu, D.-G. Photocatalytic Sequential Dimerization and Skeletal Rearrangement of Quinolines: Facile Synthesis of Indole–Methylquinoline Hybrids. ACS Catal. 2025, 15, 7792–7799. [Google Scholar] [CrossRef]
- Saget, T.; König, B. Photocatalytic synthesis of polycyclic indolones. Chem. A Eur. J. 2020, 26, 7004–7007. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Mück-Lichtenfeld, C.; Wiethoff, M.A.; Studer, A. Photocatalytic PPh3-Mediated Synthesis of C3-Functionalized Indoles via Radical Annulation of Nitroarenes and Alkenes. Angew. Chem. Int. Ed. 2024, 63, e202416726. [Google Scholar] [CrossRef] [PubMed]
- Sheng, H.; Guo, D.; Shi, Y.; Xie, H.; Kong, W.; Li, W.; Yu, Y.; Li, T. Radical [3+2] cycloaddition enables polysubstituted pyrrole synthesis via a visible-light-mediated reaction. Chem. Commun. 2025, 61, 8867–8870. [Google Scholar] [CrossRef] [PubMed]
- Sacchelli, F.; Quadri, E.; Raineri, L.; Jorea, A.; Pessina, M.; Presti, A.L.; Della Ca’, N.; Ravelli, D.; Capaldo, L. A Telescoped Strategy for the Preparation of Five-Membered Hetero-and Carbocycles via Hydrogen Atom Transfer Photocatalysis in Flow. ChemSusChem 2025, 18, e202501012. [Google Scholar] [CrossRef]
- Li, Q.; Gu, X.; Wei, Y.; Shi, M. Visible-light-induced indole synthesis via intramolecular C–N bond formation: Desulfonylative C (sp 2)–H functionalization. Chem. Sci. 2022, 13, 11623–11632. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Chang, R.; Guan, F.; Yang, W.; Zhou, C.-Y.; Guo, Z. Heterogeneous Photocatalytic C–H Functionalization of Indoles with Diazo Compounds Enabled by Carbon Nitride. J. Org. Chem. 2023, 88, 8703–8708. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.S.; Bhagat, P.R. Visible-light mediated reaction of indole with aryl halides using porphyrin photocatalyst: A feasible technique to afford N-functionalized indoles. Catal. Lett. 2024, 154, 4176–4200. [Google Scholar] [CrossRef]
- Zuo, H.-D.; Chen, X.; Zhang, Y.; Liu, J.-W.; Yan, S.-H.; Li, G.; Wang, J.-Y. Photocatalytic Thio/Selenosulfonylation–Bicyclization of Indole-Tethered 1, 6-Enynes Leading to Substituted Benzo [c] pyrrolo [1, 2, 3-lm] carbazoles. Org. Lett. 2024, 26, 3828–3833. [Google Scholar] [CrossRef]
- Yu, Y.; Lin, R.-D.; Yao, Y.; Shi, M.-L.; Lu, W.-F.; Wang, N.; Yu, X.-Q. Development of a metal-and oxidant-free enzyme–photocatalyst hybrid system for highly efficient C-3 acylation reactions of indoles with aldehydes. ACS Catal. 2022, 12, 12543–12554. [Google Scholar] [CrossRef]
- Zhang, Z.; Yi, D.; Zhang, M.; Wei, J.; Lu, J.; Yang, L.; Wang, J.; Hao, N.; Pan, X.; Zhang, S. Photocatalytic intramolecular cycloaddition of indole derivatives via energy transfer: A method for late-stage skeletal transformation. ACS Catal. 2020, 10, 10149–10156. [Google Scholar] [CrossRef]
- Xiong, Y.; Zong, Z.; Xie, W.; Chen, J.-Q.; Wu, J. Photocatalytic regioselective alkoxycarbonylation/cyclization of; dienes: Access to ester-containing pyrrolin-2-ones. Org. Lett. 2025, 27, 3037–3042. [Google Scholar] [CrossRef]
- Li, J.-J.; Liu, J.-Y.; Gao, S.-Y.; Yang, X.; Cao, R. A Tailored Quinoline-Locked Covalent Organic Framework for Metal-Free Photocatalytic α-Amino C–H Annulation. J. Am. Chem. Soc. 2025, 147, 21754–21763. [Google Scholar] [CrossRef] [PubMed]
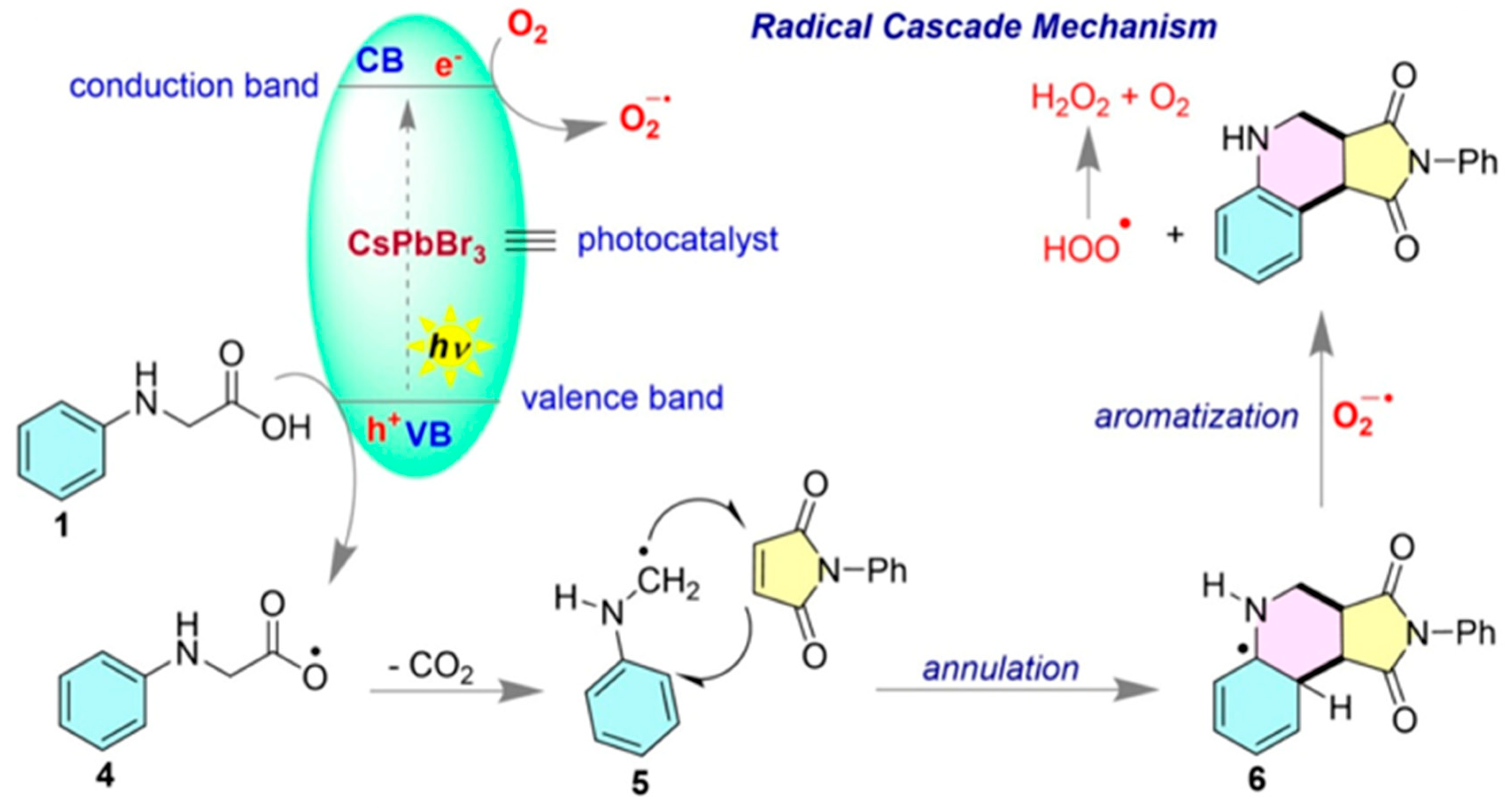
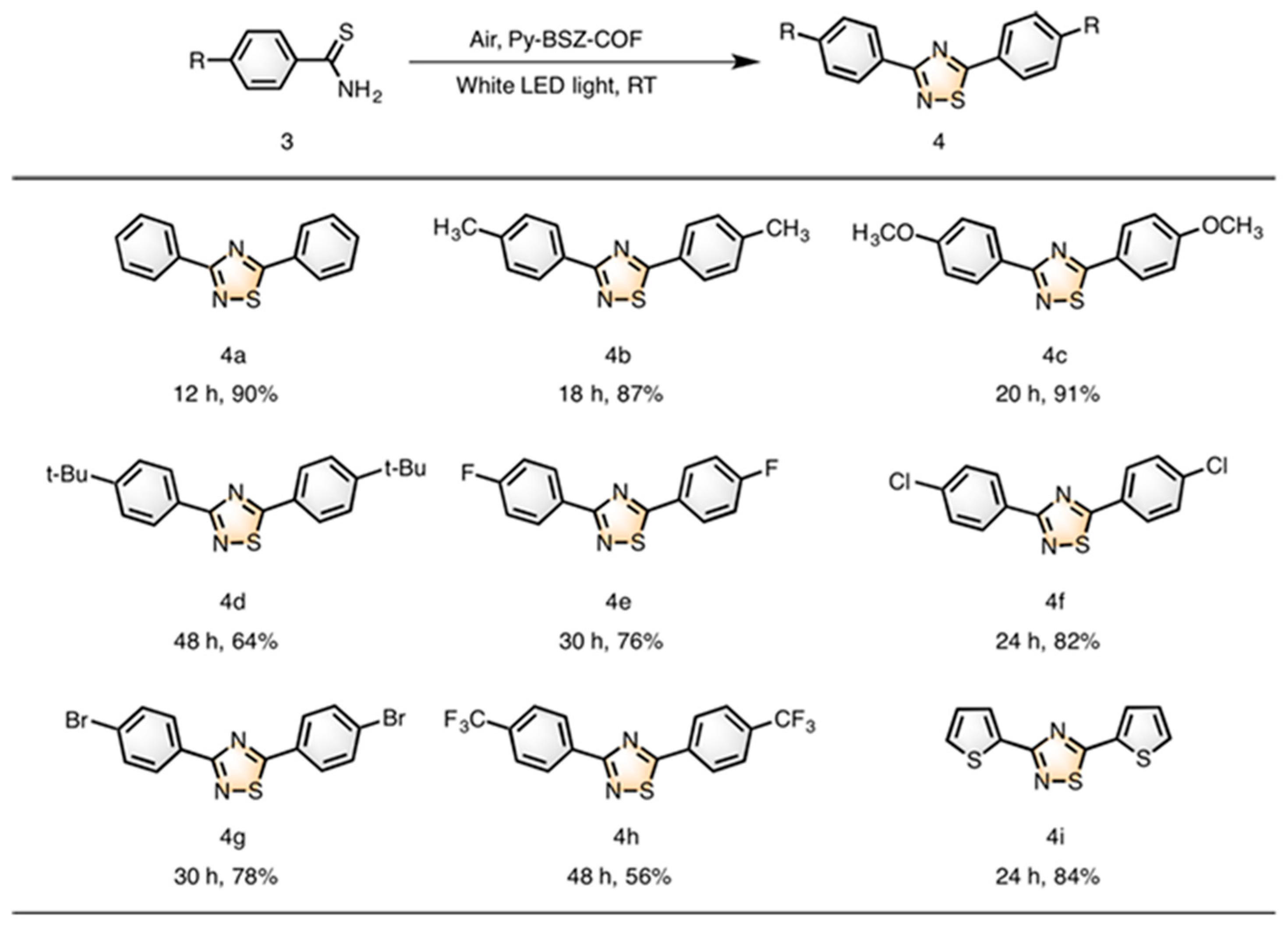
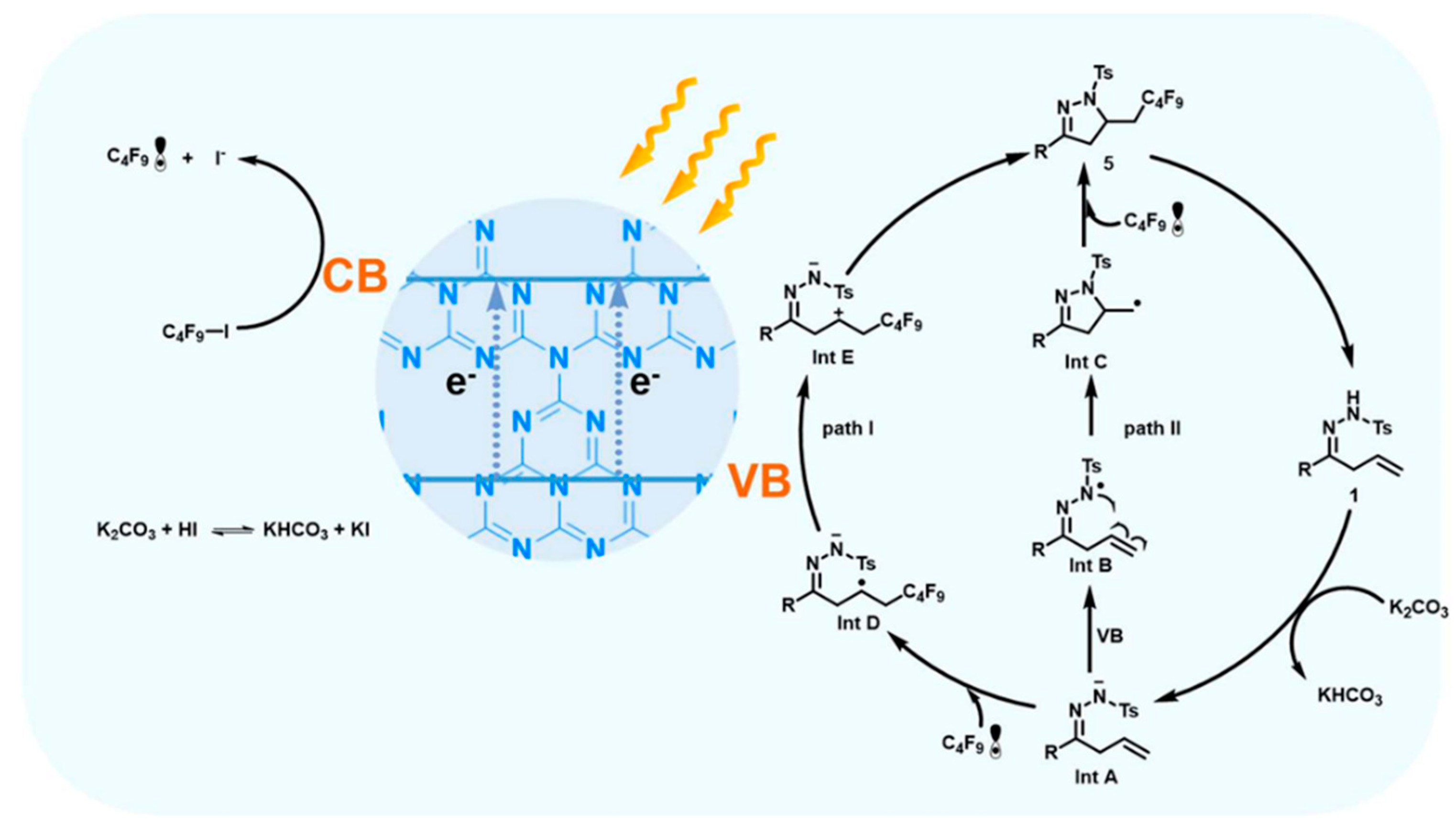
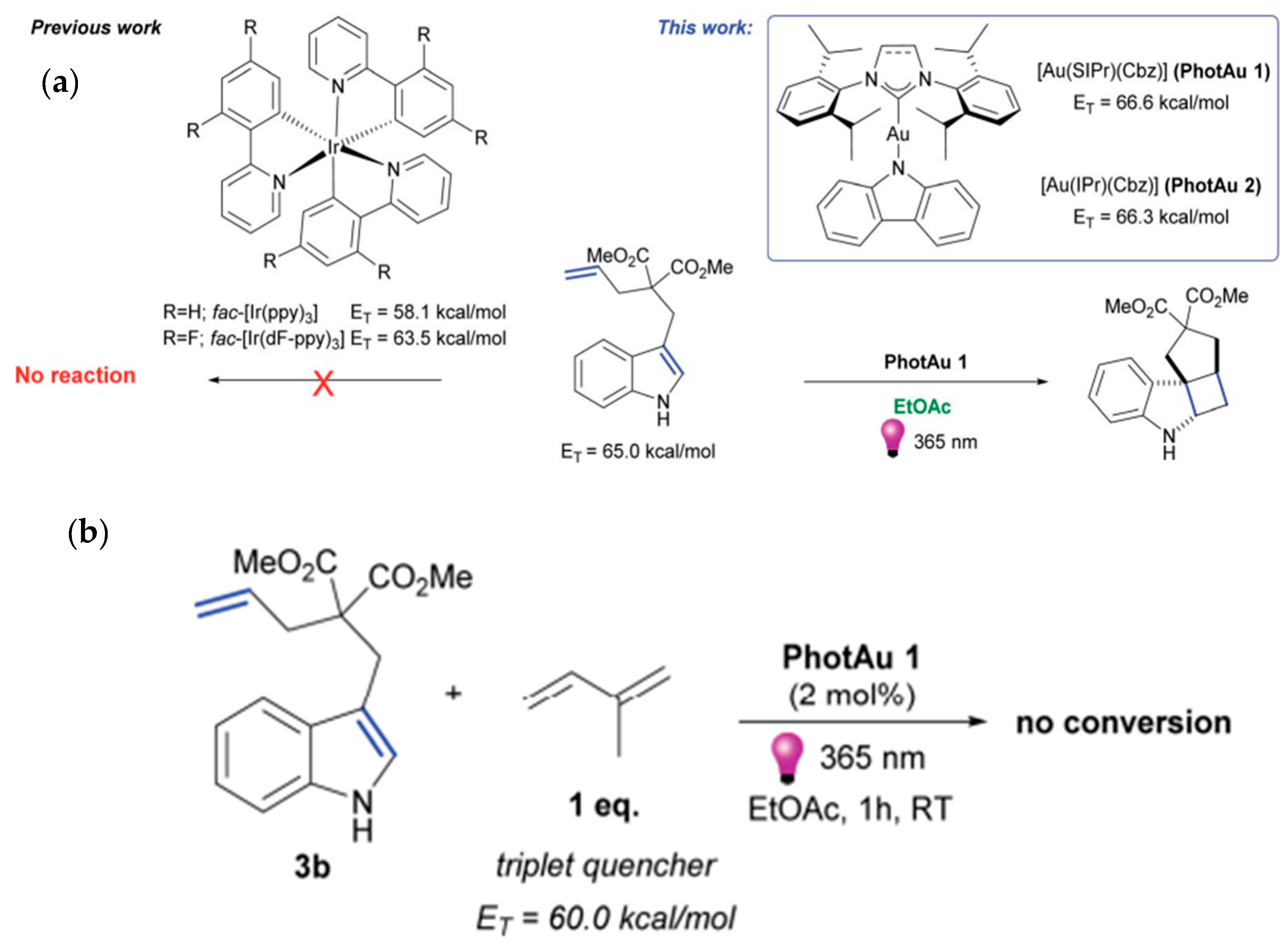
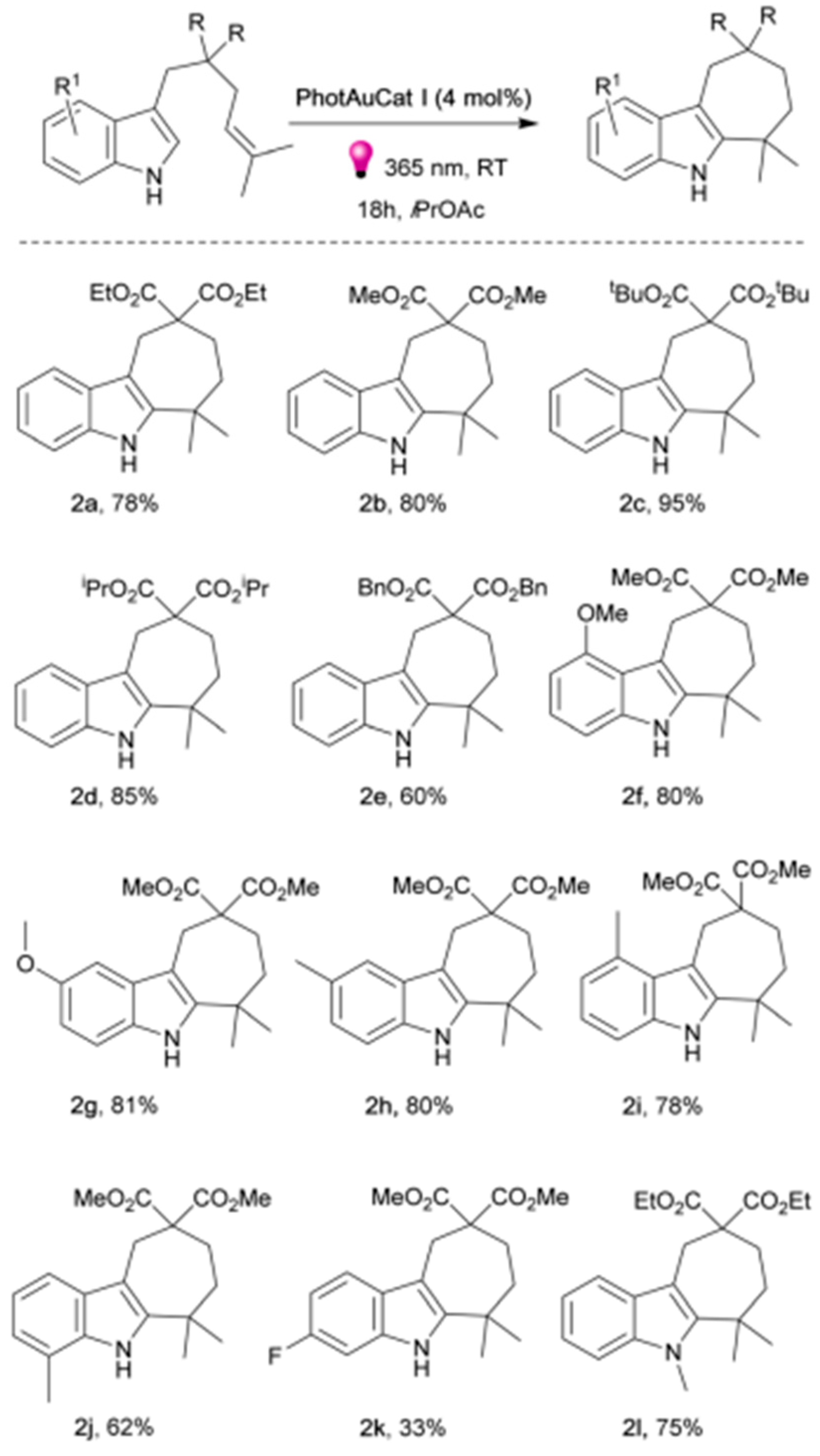
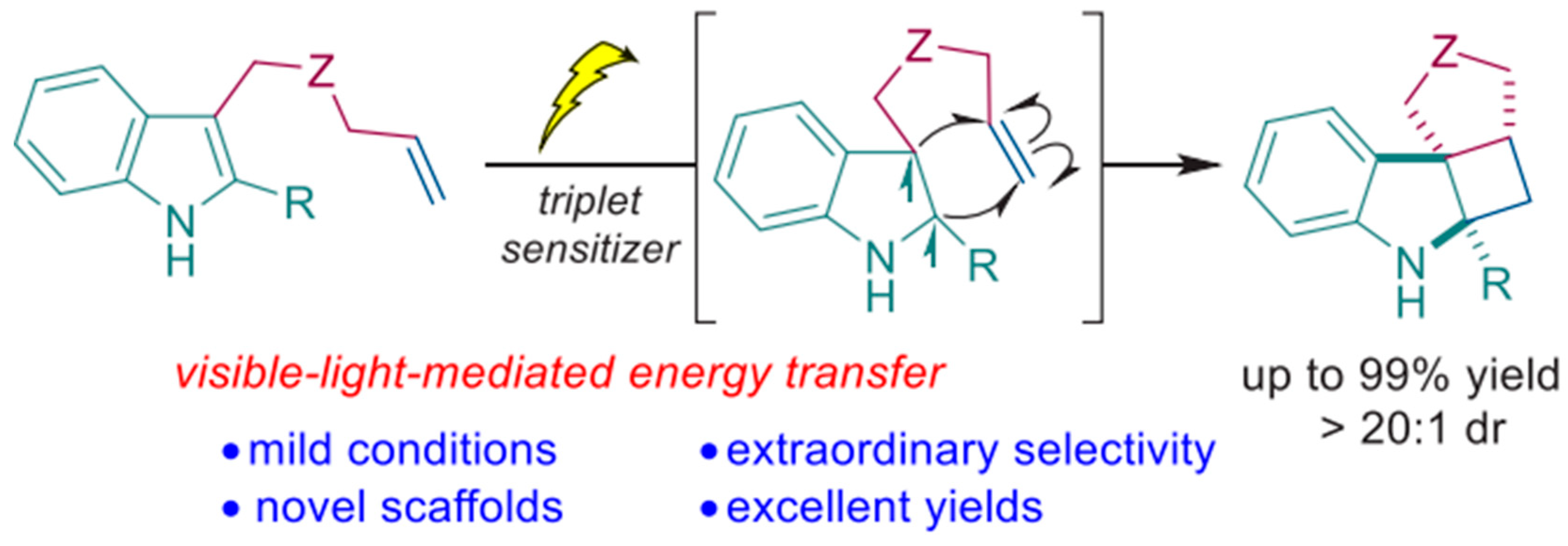
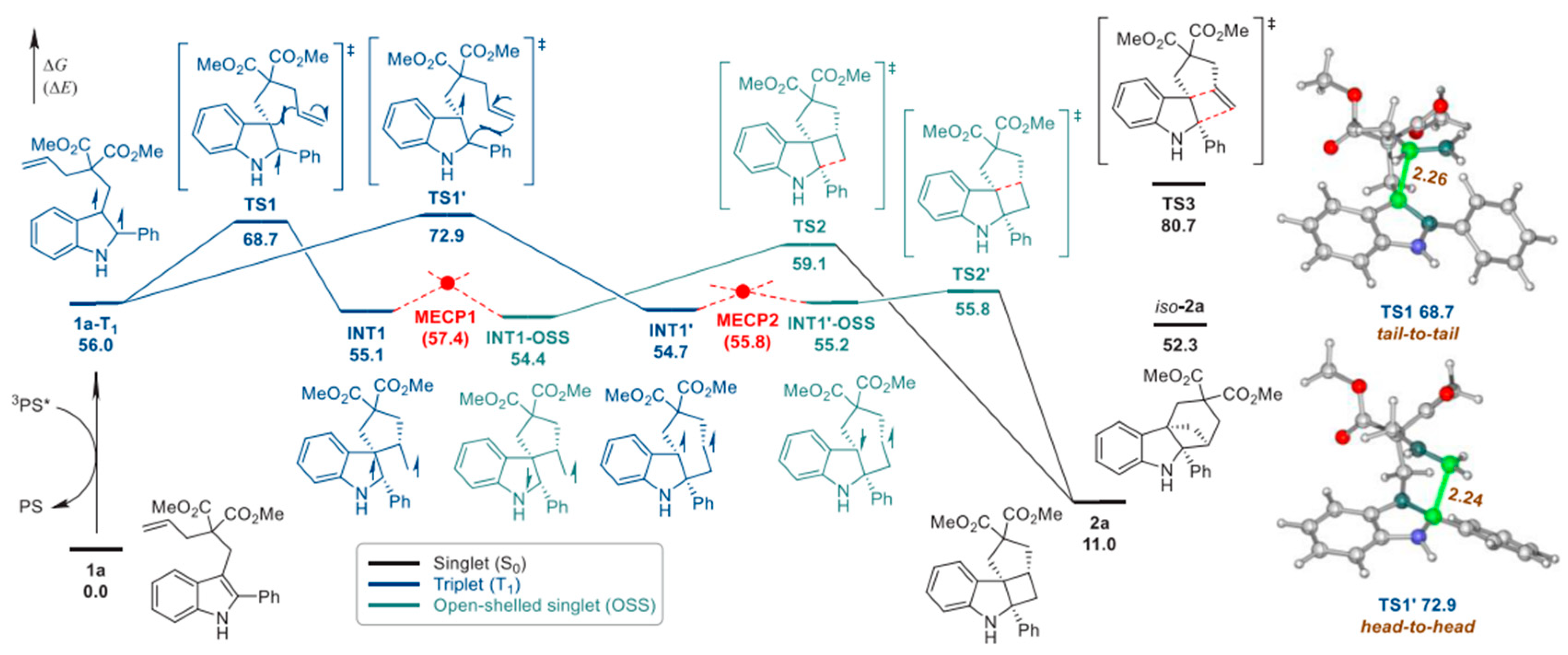
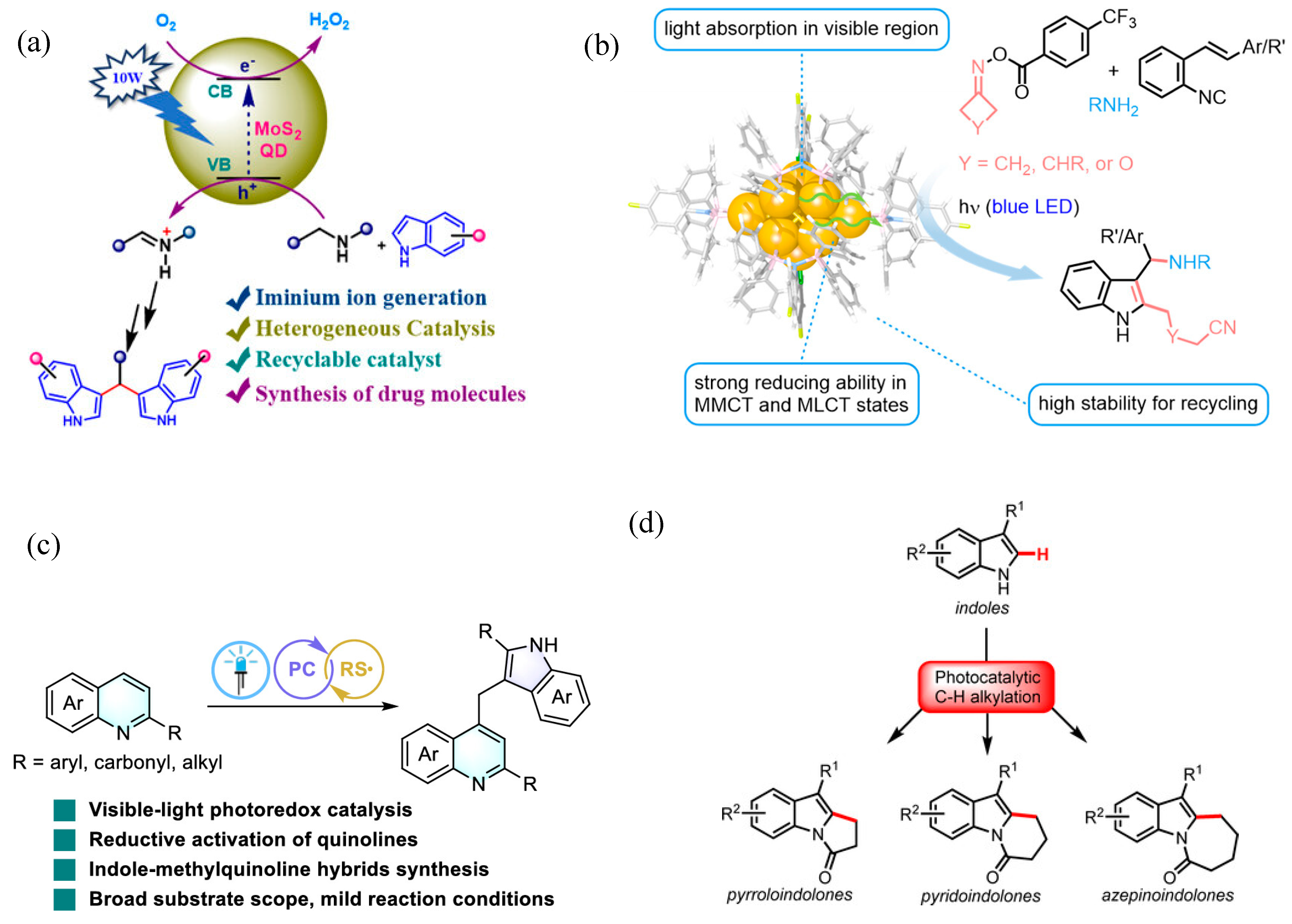
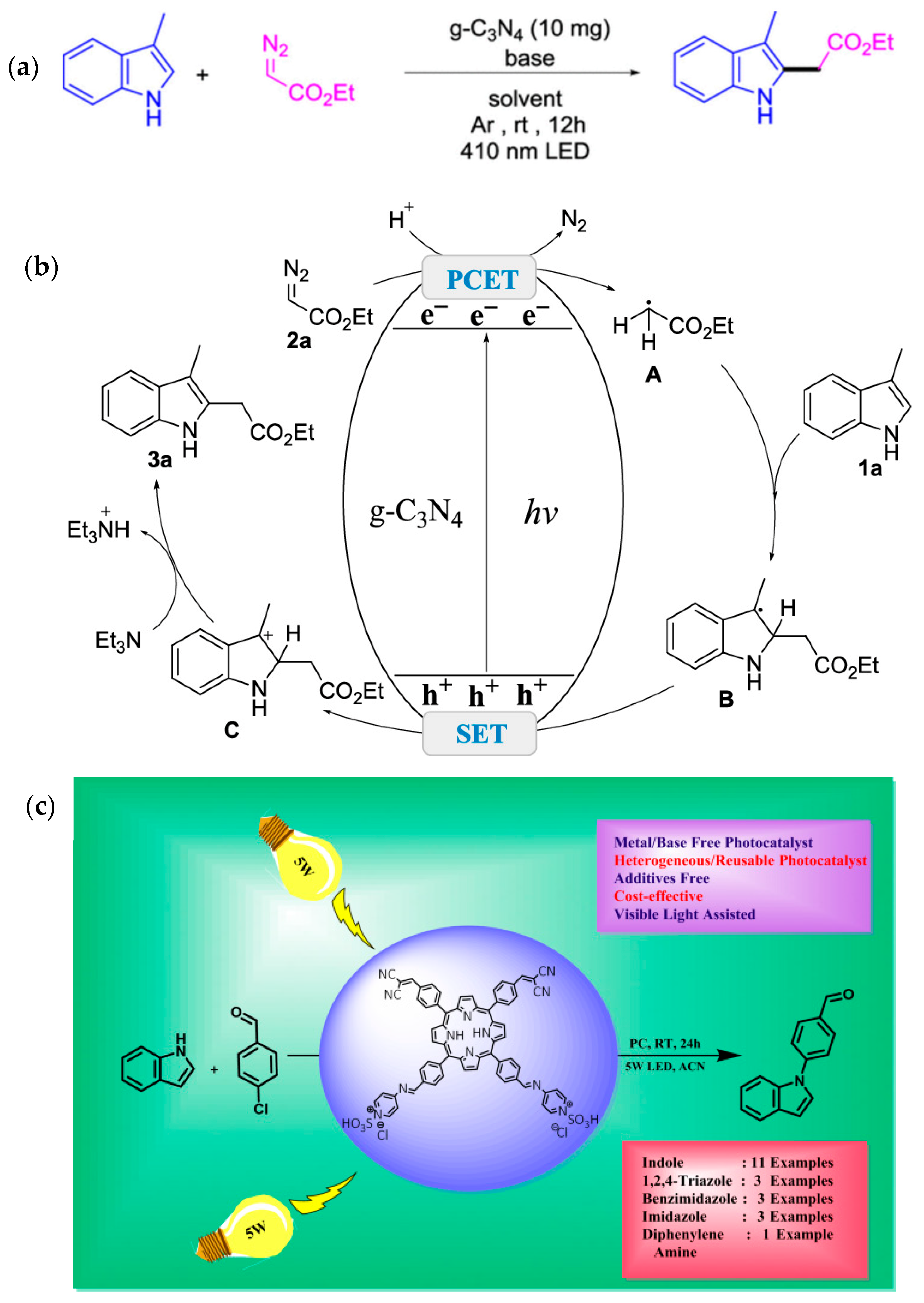
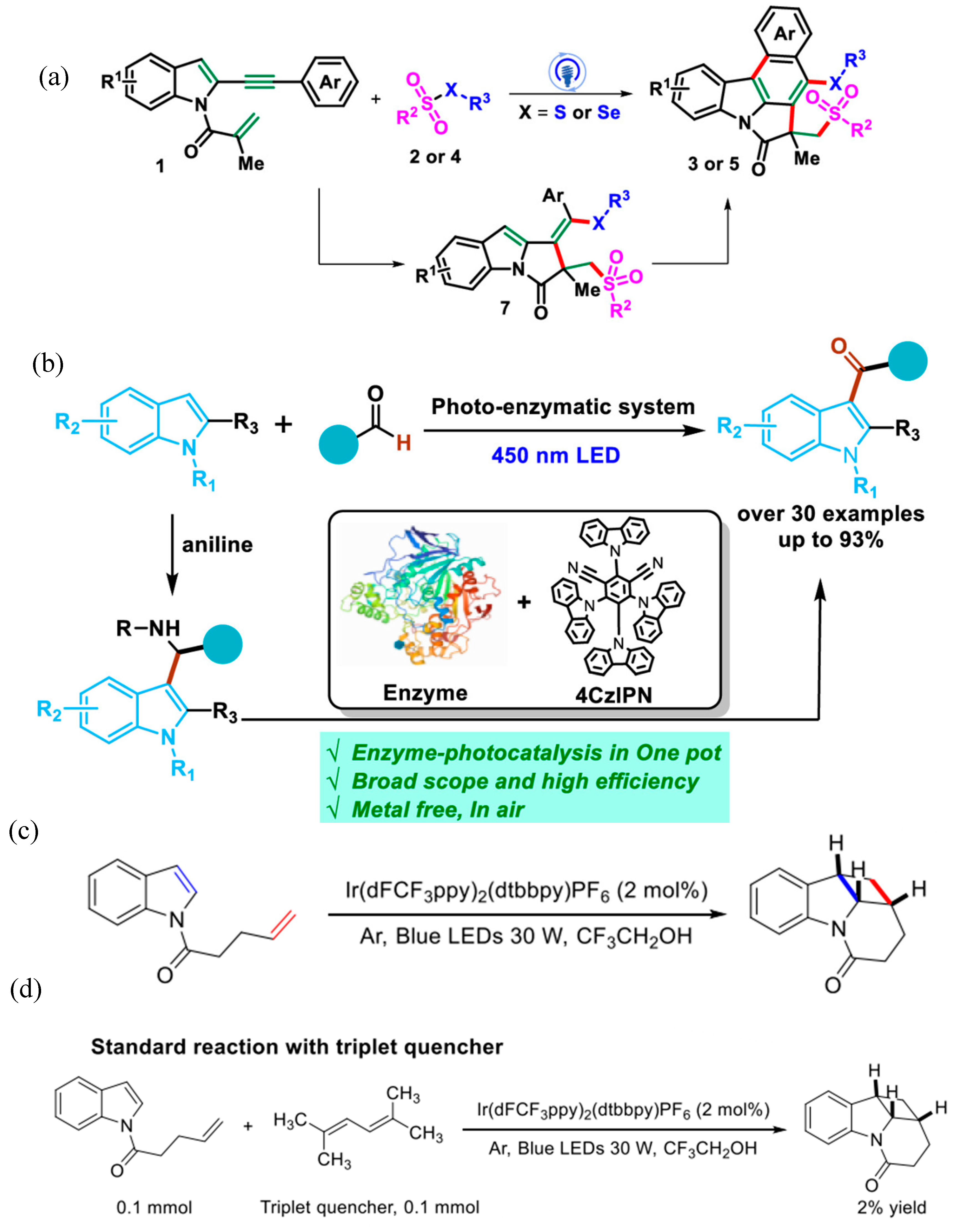
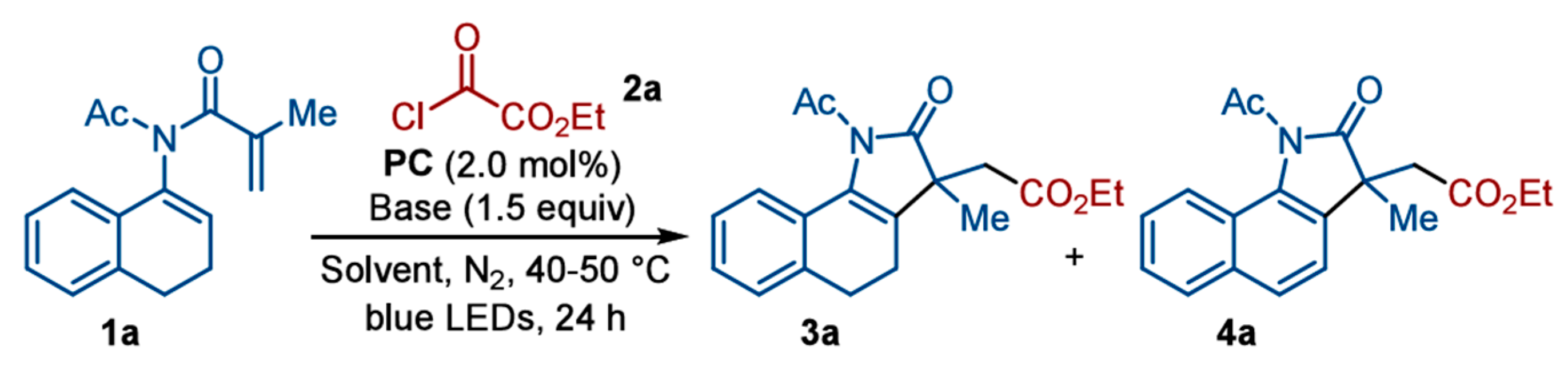

| Pathway | Key Mechanistic Aspects | Commonly Applied Photocatalysts | Specialty in Driving Heterocycles | Advantages | Limitations |
|---|---|---|---|---|---|
| Photoredox catalysis | Involving SET based radical intermediates | Semiconductor oxides, Organic dyes | C-C C-hetero bond formation | Mild reactions conditions Broader scope of application | Competing side reactions Need precise tuning of redox potentials |
| EnT | Transfer of Energy instead of electrons Can involve both singlet or triplet energy transfer | Complexes of Ir/Ru, organic dyes | [4 + 2] cycloadditions | Broader scope of application compared to TTEnT | Inappropriate for radical-induced cyclization |
| TTEnT | Triplet to triplet energy transfer | Complexes of Ir/Ru | [2 + 2] cycloadditions | Suitable for intermolecular cyclization Mild synthesis condition can drive the reaction Higher tolerance to functional groups | Lower efficiency for ISC Limited to substrate having matching triplet state energy levels Triplet state of photocatalyst should be higher than triplet state of substrate |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haider, Z.; Archana, R.; Ju, H. Recent Advancements in Photocatalytic Synthesis of Five Membered Nitrogen Heterocycles and Their Derivatives. Molecules 2025, 30, 3490. https://doi.org/10.3390/molecules30173490
Haider Z, Archana R, Ju H. Recent Advancements in Photocatalytic Synthesis of Five Membered Nitrogen Heterocycles and Their Derivatives. Molecules. 2025; 30(17):3490. https://doi.org/10.3390/molecules30173490
Chicago/Turabian StyleHaider, Zeeshan, Ravi Archana, and Heongkyu Ju. 2025. "Recent Advancements in Photocatalytic Synthesis of Five Membered Nitrogen Heterocycles and Their Derivatives" Molecules 30, no. 17: 3490. https://doi.org/10.3390/molecules30173490
APA StyleHaider, Z., Archana, R., & Ju, H. (2025). Recent Advancements in Photocatalytic Synthesis of Five Membered Nitrogen Heterocycles and Their Derivatives. Molecules, 30(17), 3490. https://doi.org/10.3390/molecules30173490







