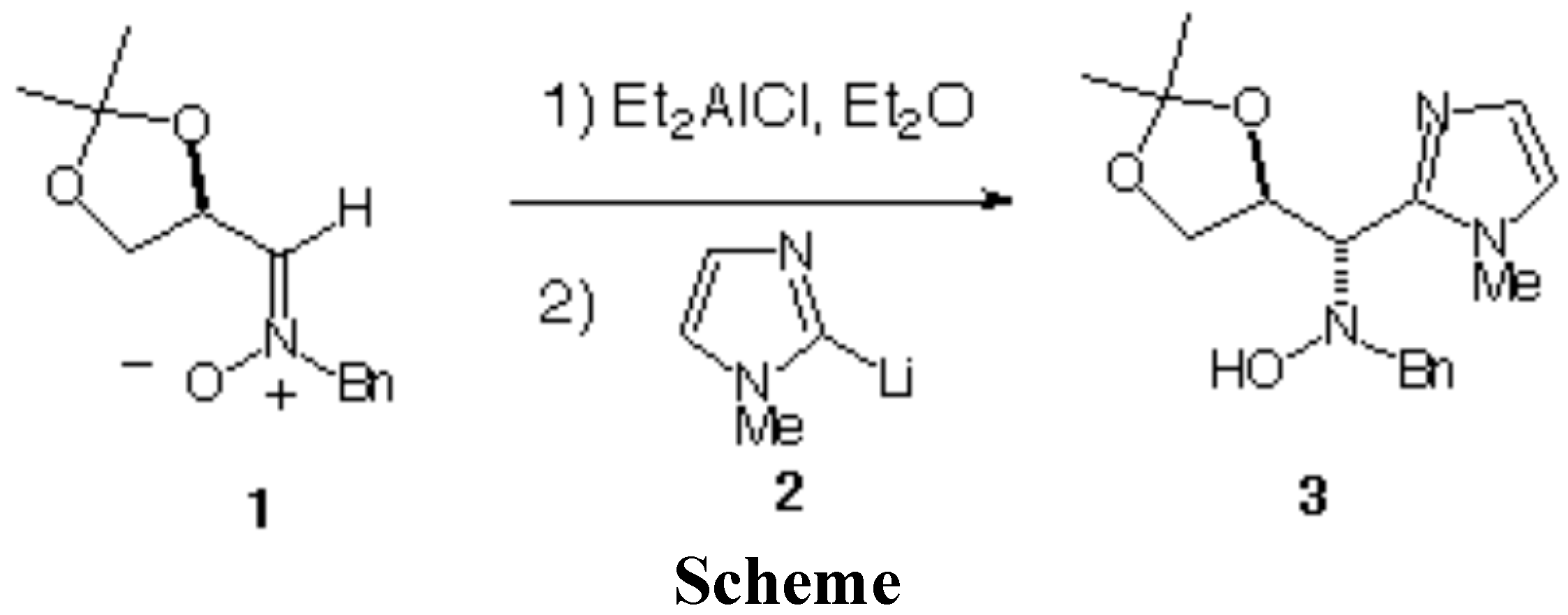
(1S,2S)-N-Benzyl-N-[(2,2-dimethyl-1,3-dioxolan-4-yl)-(1-methylimidazol-2-yl)methyl] Hydroxylamine
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References and Notes
- Merino, P.; Merchan, F. L.; Tejero, T. Acta Cryst. Sect. C 1995, 51, 2400–2401.
- Sample Availability: Available from the authors and MDPI.
© 1998 MDPI. All rights reserved. Molecules website http://www.mdpi.org/molecules/
Share and Cite
Merino, P.; Franco, S.; Merchan, F.L.; Tejero, T. (1S,2S)-N-Benzyl-N-[(2,2-dimethyl-1,3-dioxolan-4-yl)-(1-methylimidazol-2-yl)methyl] Hydroxylamine. Molecules 1998, 3, M83. https://doi.org/10.3390/M83
Merino P, Franco S, Merchan FL, Tejero T. (1S,2S)-N-Benzyl-N-[(2,2-dimethyl-1,3-dioxolan-4-yl)-(1-methylimidazol-2-yl)methyl] Hydroxylamine. Molecules. 1998; 3(5):M83. https://doi.org/10.3390/M83
Chicago/Turabian StyleMerino, Pedro, Santiago Franco, Francisco L. Merchan, and Tomas Tejero. 1998. "(1S,2S)-N-Benzyl-N-[(2,2-dimethyl-1,3-dioxolan-4-yl)-(1-methylimidazol-2-yl)methyl] Hydroxylamine" Molecules 3, no. 5: M83. https://doi.org/10.3390/M83
APA StyleMerino, P., Franco, S., Merchan, F. L., & Tejero, T. (1998). (1S,2S)-N-Benzyl-N-[(2,2-dimethyl-1,3-dioxolan-4-yl)-(1-methylimidazol-2-yl)methyl] Hydroxylamine. Molecules, 3(5), M83. https://doi.org/10.3390/M83



