Chiral Separation of Apremilast by Capillary Electrophoresis Using Succinyl-β-Cyclodextrin—Reversal of Enantiomer Elution Order by Cationic Capillary Coating
Abstract
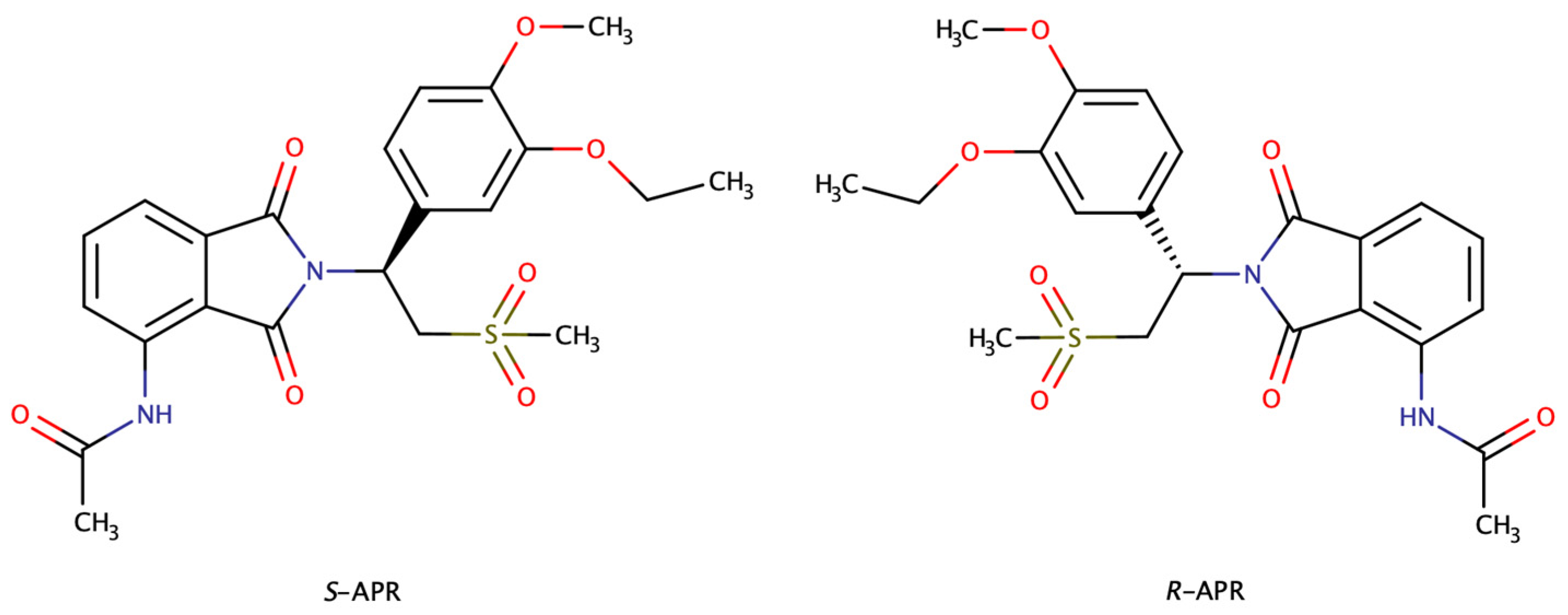
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Enantioselective CE Conditions
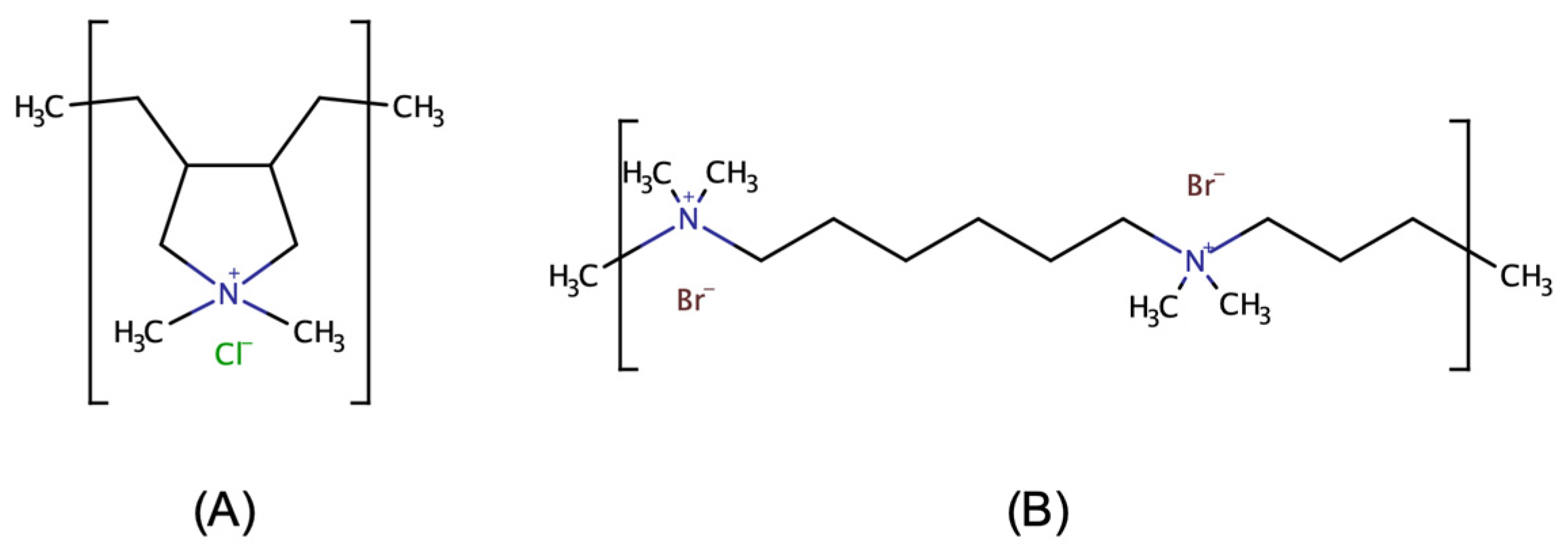
3.3. Dynamic Coating Techniques
3.3.1. Polydiallyldimethylammonium Chloride (PDADMAC) Monolayer Coating
3.3.2. Polybrene (PB; Known as Hexadimethrine Bromide) Monolayer Coating
3.4. Preparation of BGE and Sample Solutions
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Bianchi, L.; Del Duca, E.; Romanelli, M.; Saraceno, R.; Chimenti, S.; Chiricozzi, A. Pharmacodynamic Assessment of Apremilast for the Treatment of Moderate-to-Severe Plaque Psoriasis. Expert Opin. Drug Metab. Toxicol. 2016, 12, 1121–1128. [Google Scholar] [CrossRef]
- Gao, J.C.; Wu, A.G.; Contento, M.N.; Maher, J.M.; Cline, A. Apremilast in the Treatment of Plaque Psoriasis: Differential Use in Psoriasis. Clin. Cosmet. Investig. Dermatol. 2022, 15, 395. [Google Scholar] [CrossRef] [PubMed]
- Torres, T.; Puig, L. Apremilast: A Novel Oral Treatment for Psoriasis and Psoriatic Arthritis. Am. J. Clin. Dermatol. 2018, 19, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Schafer, P.H.; Parton, A.; Capone, L.; Cedzik, D.; Brady, H.; Evans, J.F.; Man, H.-W.; Muller, G.W.; Stirling, D.I.; Chopra, R. Apremilast Is a Selective PDE4 Inhibitor with Regulatory Effects on Innate Immunity. Cell Signal. 2014, 26, 2016–2029. [Google Scholar] [CrossRef]
- Millrine, D.; Kishimoto, T. A Brighter Side to Thalidomide: Its Potential Use in Immunological Disorders. Trends Mol. Med. 2017, 23, 348–361. [Google Scholar] [CrossRef] [PubMed]
- Man, H.-W.; Schafer, P.; Wong, L.M.; Patterson, R.T.; Corral, L.G.; Raymon, H.; Blease, K.; Leisten, J.; Shirley, M.A.; Tang, Y.; et al. Discovery of (S)-N-[2-[1-(3-Ethoxy-4-Methoxyphenyl)-2-Methanesulfonylethyl]-1,3-Dioxo-2,3-Dihydro-1H-Isoindol-4-Yl] Acetamide (Apremilast), a Potent and Orally Active Phosphodiesterase 4 and Tumor Necrosis Factor-α Inhibitor. J. Med. Chem. 2009, 52, 1522–1524. [Google Scholar] [CrossRef]
- Narode, H.; Gayke, M.; Eppa, G.; Yadav, J.S. A Review on Synthetic Advances toward the Synthesis of Apremilast, an Anti-Inflammatory Drug. Org. Process Res. Dev. 2021, 25, 1512–1523. [Google Scholar] [CrossRef]
- European Medicines Agency. Otezla (Apremilast): European Public Assessment Report [Online]. Available online: https://www.ema.europa.eu/en/documents/assessment-report/otezla-epar-public-assessment-report_en.pdf (accessed on 13 February 2023).
- Xiong, K.; Ma, X.; Cao, N.; Liu, L.; Sun, L.; Zou, Q.; Wei, P. Identification, Characterization and HPLC Quantification of Impurities in Apremilast. Anal. Methods 2016, 8, 1889–1897. [Google Scholar] [CrossRef]
- Lu, Y.; Shen, X.; Hang, T.; Song, M. Identification and Characterization of Process-Related Substances and Degradation Products in Apremilast: Process Optimization and Degradation Pathway Elucidation. J. Pharm. Biomed. Anal. 2017, 141, 70–78. [Google Scholar] [CrossRef]
- Faigl, F.; Doubsky, J.; Klvana, R.; Richter, J.; Fogassy, E.; Matravolgyi, B.; Mizsak, A. A Method of Chiral Resolution of the Key Intermediate of the Synthesis of Apremilast and Its Use for the Preparation of Pure Apremilast. European Patent EP3280701B1, 16 October 2019. [Google Scholar]
- The Apremilast of High Chiral Purity. Chinese Patent CN107628983A, 26 January 2018.
- Foroughbakhshfasaei, M.; Szabó, Z.-I.; Tóth, G. Validated LC Method for Determination of Enantiomeric Purity of Apremilast Using Polysaccharide-Type Stationary Phases in Polar Organic Mode. Chromatographia 2018, 81, 1613–1621. [Google Scholar] [CrossRef]
- Foroughbakhshfasaei, M.; Dobó, M.; Boda, F.; Szabó, Z.-I.; Tóth, G. Comparative Chiral Separation of Thalidomide Class of Drugs Using Polysaccharide-Type Stationary Phases with Emphasis on Elution Order and Hysteresis in Polar Organic Mode. Molecules 2022, 27, 111. [Google Scholar] [CrossRef] [PubMed]
- Vijaykumar, C.; Kumar, Y.R.; Aparna, P.; Marisetti, V.M. Enantioselective Green HPLC Method for Simultaneous Determination of Enantiomer, and Potential Impurities in Apremilast Drug Substance. Anal. Chem. Lett. 2022, 12, 691–714. [Google Scholar] [CrossRef]
- Dombi, G.; Horváth, P.; Fiser, B.; Mirzahosseini, A.; Dobó, M.; Szabó, Z.-I.; Tóth, G. Enantioselective Human Serum Albumin Binding of Apremilast: Liquid Chromatographic, Fluorescence and Molecular Docking Study. Int. J. Mol. Sci. 2023, 24, 2168. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.B.; Quirino, J.P. Chiral Selectors in Capillary Electrophoresis: Trends during 2017–2018. Molecules 2019, 24, 1135. [Google Scholar] [CrossRef]
- Orlandini, S.; Hancu, G.; Szabó, Z.-I.; Modroiu, A.; Papp, L.-A.; Gotti, R.; Furlanetto, S. New Trends in the Quality Control of Enantiomeric Drugs: Quality by Design-Compliant Development of Chiral Capillary Electrophoresis Methods. Molecules 2022, 27, 7058. [Google Scholar] [CrossRef]
- Scriba, G.K.E.; Konjaria, M.-L.; Krait, S. Cyclodextrins. In Chiral Separations and Stereochemical Elucidation: Fundamentals, Methods, and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2023; pp. 273–323. [Google Scholar] [CrossRef]
- Scriba, G.K.E. Chiral Recognition in Separation Sciences. Part I: Polysaccharide and Cyclodextrin Selectors. TrAC—Trends Anal. Chem. 2019, 120, 115639. [Google Scholar] [CrossRef]
- Peluso, P.; Chankvetadze, B. Native and Substituted Cyclodextrins as Chiral Selectors for Capillary Electrophoresis Enantioseparations: Structures, Features, Application, and Molecular Modeling. Electrophoresis 2021, 42, 1676–1708. [Google Scholar] [CrossRef]
- Chankvetadze, B. Enantiomer Migration Order in Chiral Capillary Electrophoresis. Electrophoresis 2002, 23, 4022–4035. [Google Scholar] [CrossRef]
- Okamoto, M. Reversal of Elution Order during the Chiral Separation in High Performance Liquid Chromatography. J. Pharm. Biomed. Anal. 2002, 27, 401–407. [Google Scholar] [CrossRef]
- Horváth, S.; Németh, G. Hysteresis of Retention and Enantioselectivity on Amylose Tris(3,5-Dimethylphenylcarbamate) Chiral Stationary Phases in Mixtures of 2-Propanol and Methanol. J. Chromatogr. A 2018, 1568, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Horváth, S.; Eke, Z.; Németh, G. Utilization of the Hysteresis Phenomenon for Chiral High-Performance Liquid Chromatographic Method Selection in Polar Organic Mode. J. Chromatogr. A 2020, 1625, 461280. [Google Scholar] [CrossRef]
- Chankvetadze, B. Recent Trends in Preparation, Investigation and Application of Polysaccharide-Based Chiral Stationary Phases for Separation of Enantiomers in High-Performance Liquid Chromatography. TrAC—Trends Anal. Chem. 2020, 122, 115709. [Google Scholar] [CrossRef]
- Szabó, Z.-I.; Gál, R.; Szőcs, L.; Ludmerczki, R.; Muntean, D.-L.; Noszál, B.; Tóth, G. Validated Capillary Electrophoretic Method for the Enantiomeric Quality Control of R-Praziquantel. Electrophoresis 2017, 38, 1886–1894. [Google Scholar] [CrossRef]
- Szabó, Z.-I.; Ludmerczki, R.; Fiser, B.; Noszál, B.; Tóth, G. Chiral Separation of Rasagiline Using Sulfobutylether-β-Cyclodextrin: Capillary Electrophoresis, NMR and Molecular Modeling Study. Electrophoresis 2019, 40, 1897–1903. [Google Scholar] [CrossRef]
- Horvath, J.; Dolník, V. Polymer Wall Coatings for Capillary Electrophoresis. Electrophoresis 2001, 22, 644–655. [Google Scholar] [CrossRef] [PubMed]
- Hajba, L.; Guttman, A. Recent Advances in Column Coatings for Capillary Electrophoresis of Proteins. TrAC—Trends Anal. Chem. 2017, 90, 38–44. [Google Scholar] [CrossRef]
- Znaleziona, J.; Petr, J.; Knob, R.; Maier, V.; Ševčík, J. Dynamic Coating Agents in CE. Chromatographia 2008, 67, 5–12. [Google Scholar] [CrossRef]
- Meyring, M.; Chankvetadze, B.; Blaschke, G. Enantioseparation of Thalidomide and Its Hydroxylated Metabolites Using Capillary Electrophoresis with Various Cyclodextrins and Their Combinations as Chiral Buffer Additives. Electrophoresis 1999, 20, 2425–2431. [Google Scholar] [CrossRef]
- Szabó, Z.-I.; Szőcs, L.; Muntean, D.-L.; Noszál, B.; Tóth, G. Chiral Separation of Uncharged Pomalidomide Enantiomers Using Carboxymethyl-β-Cyclodextrin: A Validated Capillary Electrophoretic Method. Chirality 2016, 28, 199–203. [Google Scholar] [CrossRef]
- Szabó, Z.I.; Foroughbakhshfasaei, M.; Gál, R.; Horváth, P.; Komjáti, B.; Noszál, B.; Tóth, G. Chiral Separation of Lenalidomide by Liquid Chromatography on Polysaccharide-Type Stationary Phases and by Capillary Electrophoresis Using Cyclodextrin Selectors. J. Sep. Sci. 2018, 41, 1414–1423. [Google Scholar] [CrossRef] [PubMed]
- Szemán, J.; Ganzler, K.; Salgó, A.; Szejtli, J. Effect of the Degree of Substitution of Cyclodextrin Derivatives on Chiral Separations by High-Performance Liquid Chromatography and Capillary Electrophoresis. J. Chromatogr. A 1996, 728, 423–431. [Google Scholar] [CrossRef]
- Skanchy, D.J.; Xie, G.H.; Tait, R.J.; Luna, E.; Demarest, C.; Stobaugh, J.F. Application of Sulfobutylether-Beta-Cyclodextrin with Specific Degrees of Substitution for the Enantioseparation of Pharmaceutical Mixtures by Capillary Electrophoresis. Electrophoresis 1999, 20, 2638–2649. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, Q.; Luo, L.; Sun, T.; Guo, X. Comparison of Three S-β-CDs with Different Degrees of Substitution for the Chiral Separation of 12 Drugs in Capillary Electrophoresis. Chirality 2017, 29, 558–565. [Google Scholar] [CrossRef]
- Theurillat, R.; Knobloch, M.; Schmitz, A.; Lassahn, P.-G.; Mevissen, M.; Thormann, W. Enantioselective Analysis of Ketamine and Its Metabolites in Equine Plasma and Urine by CE with Multiple Isomer Sulfated β-CD. Electrophoresis 2007, 28, 2748–2757. [Google Scholar] [CrossRef]
- Kim, H.; Choi, Y.; Lim, J.; Paik, S.-R.; Jung, S. Chiral Separation of Catechin by Capillary Electrophoresis Using Mono-, Di-, Tri-Succinyl-Beta-Cyclodextrin as Chiral Selectors. Chirality 2009, 21, 937–942. [Google Scholar] [CrossRef] [PubMed]
- Chankvetadze, B.; Schulte, G.; Blaschke, G. Nature and Design of Enantiomer Migration Order in Chiral Capillary Electrophoresis. Enantiomer 1997, 2, 157–179. [Google Scholar]
- Chankvetadze, B. Capillary Electrophoresis in Chiral Analysis; John Wiley & Sons: Hoboken, NJ, USA, 1997. [Google Scholar]
- Chankvetadze, B.; Blaschke, G. Enantioseparations in Capillary Electromigration Techniques: Recent Developments and Future Trends. J. Chromatogr. A 2001, 906, 309–363. [Google Scholar] [CrossRef]
- Roca, S.; Dhellemmes, L.; Leclercq, L.; Cottet, H. Polyelectrolyte Multilayers in Capillary Electrophoresis. ChemPlusChem 2022, 87, e202200028. [Google Scholar] [CrossRef]
- Kpaibe, A.P.S.; Ben-Ameur, R.; Coussot, G.; Ladner, Y.; Montels, J.; Ake, M.; Perrin, C. Evaluation of Capillary Zone Electrophoresis for the Quality Control of Complex Biologic Samples: Application to Snake Venoms. Electrophoresis 2017, 38, 2050–2059. [Google Scholar] [CrossRef] [PubMed]
- Haselberg, R.; De Jong, G.J.; Somsen, G.W. Capillary Electrophoresis—Mass Spectrometry of Intact Basic Proteins Using Polybrene--Dextran Sulfate--Polybrene-Coated Capillaries: System Optimization and Performance. Anal. Chim. Acta 2010, 678, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Haselberg, R.; Brinks, V.; Hawe, A.; de Jong, G.J.; Somsen, G.W. Capillary Electrophoresis-Mass Spectrometry Using Noncovalently Coated Capillaries for the Analysis of Biopharmaceuticals. Anal. Bioanal. Chem. 2011, 400, 295–303. [Google Scholar] [CrossRef] [PubMed]


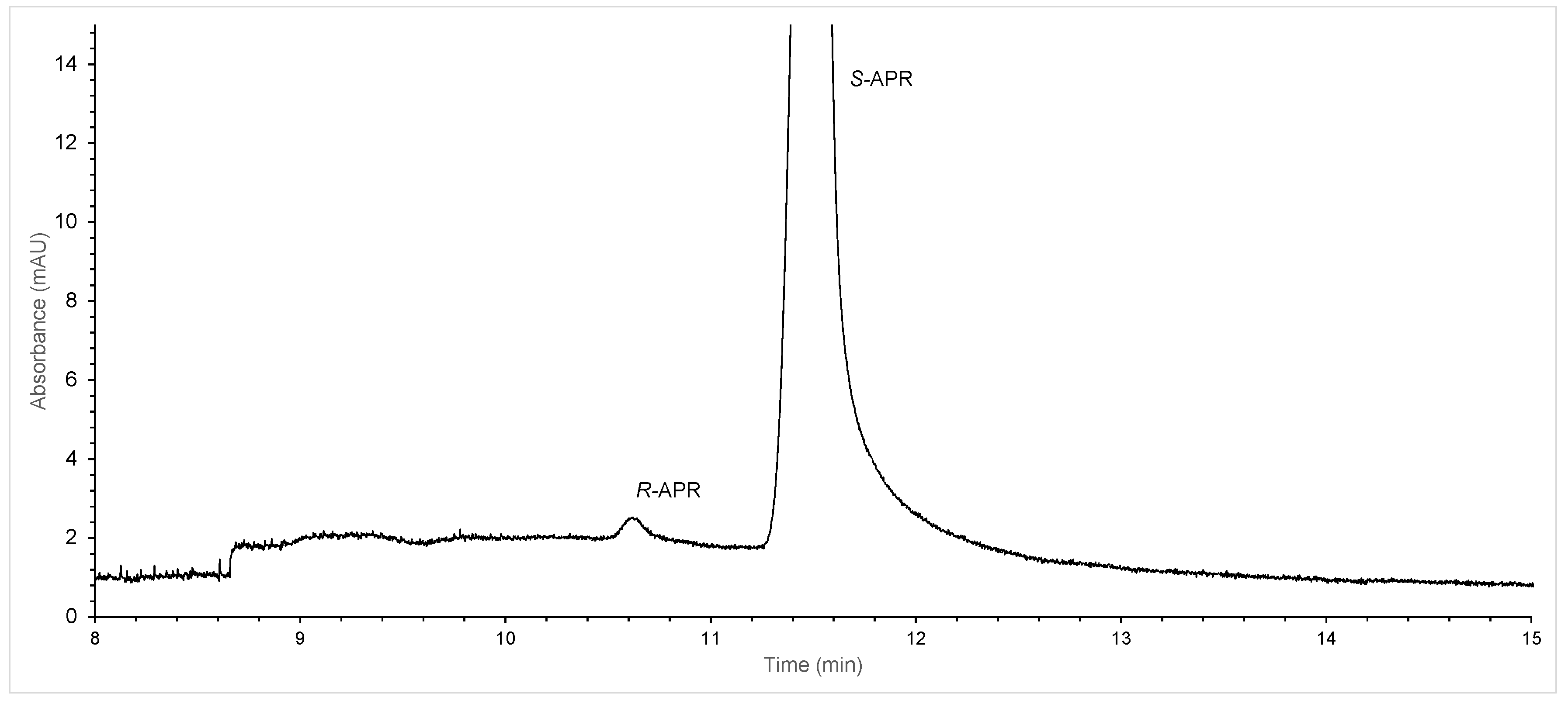
| Temperature | tR-APR (min) | tS-APR (min) | Rs |
|---|---|---|---|
| 20 °C | 10.92 | 11.80 | 4.35 |
| 30 °C | 10.30 | 11.06 | 3.72 |
| 40 °C | 9.19 | 9.75 | 2.76 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szabó, Z.-I.; Benkő, B.-M.; Bartalis-Fábián, Á.; Iványi, R.; Varga, E.; Szőcs, L.; Tóth, G. Chiral Separation of Apremilast by Capillary Electrophoresis Using Succinyl-β-Cyclodextrin—Reversal of Enantiomer Elution Order by Cationic Capillary Coating. Molecules 2023, 28, 3310. https://doi.org/10.3390/molecules28083310
Szabó Z-I, Benkő B-M, Bartalis-Fábián Á, Iványi R, Varga E, Szőcs L, Tóth G. Chiral Separation of Apremilast by Capillary Electrophoresis Using Succinyl-β-Cyclodextrin—Reversal of Enantiomer Elution Order by Cationic Capillary Coating. Molecules. 2023; 28(8):3310. https://doi.org/10.3390/molecules28083310
Chicago/Turabian StyleSzabó, Zoltán-István, Beáta-Mária Benkő, Ágnes Bartalis-Fábián, Róbert Iványi, Erzsébet Varga, Levente Szőcs, and Gergő Tóth. 2023. "Chiral Separation of Apremilast by Capillary Electrophoresis Using Succinyl-β-Cyclodextrin—Reversal of Enantiomer Elution Order by Cationic Capillary Coating" Molecules 28, no. 8: 3310. https://doi.org/10.3390/molecules28083310
APA StyleSzabó, Z.-I., Benkő, B.-M., Bartalis-Fábián, Á., Iványi, R., Varga, E., Szőcs, L., & Tóth, G. (2023). Chiral Separation of Apremilast by Capillary Electrophoresis Using Succinyl-β-Cyclodextrin—Reversal of Enantiomer Elution Order by Cationic Capillary Coating. Molecules, 28(8), 3310. https://doi.org/10.3390/molecules28083310










