Advanced Nanostructured Coatings Based on Doped TiO2 for Various Applications
Abstract
:1. Introduction
2. Properties of TiO2 Films
2.1. Structural Properties
2.2. Morphological Properties
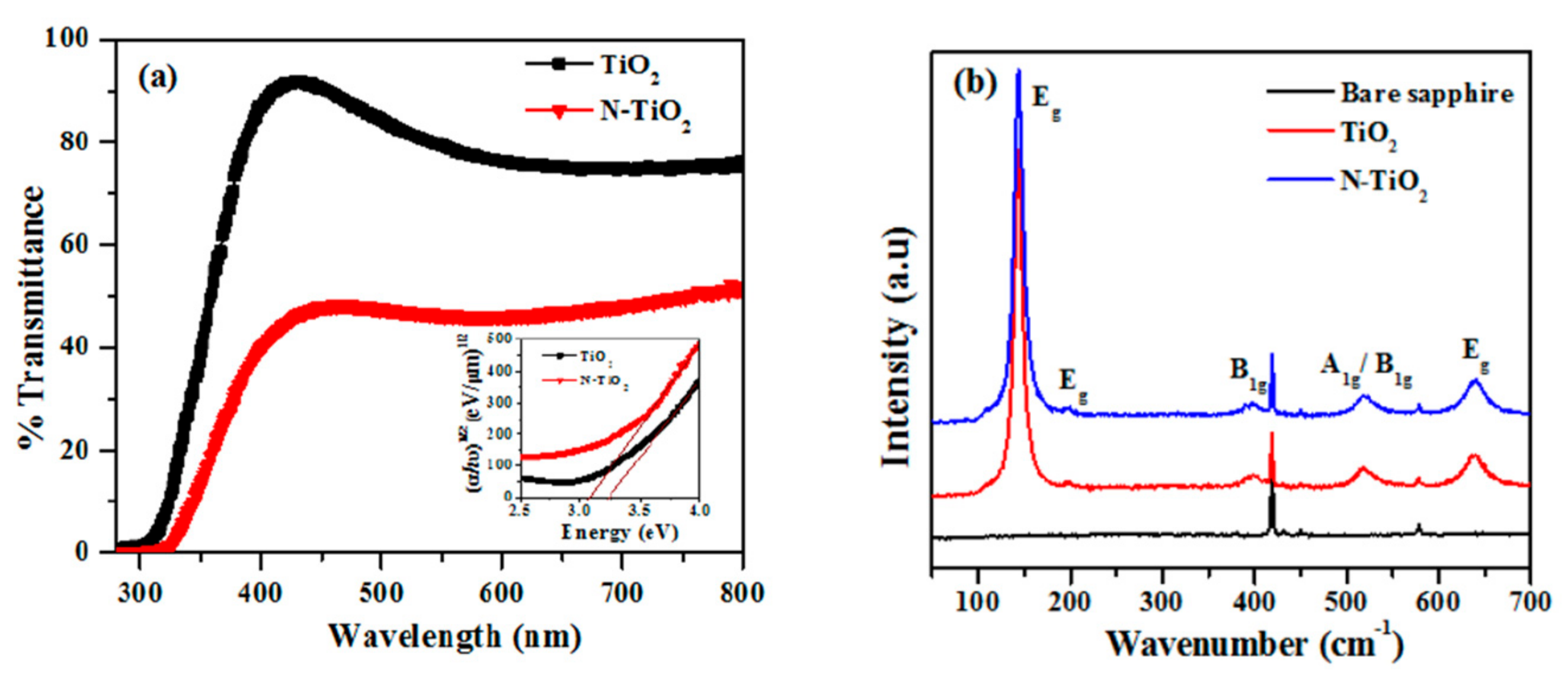
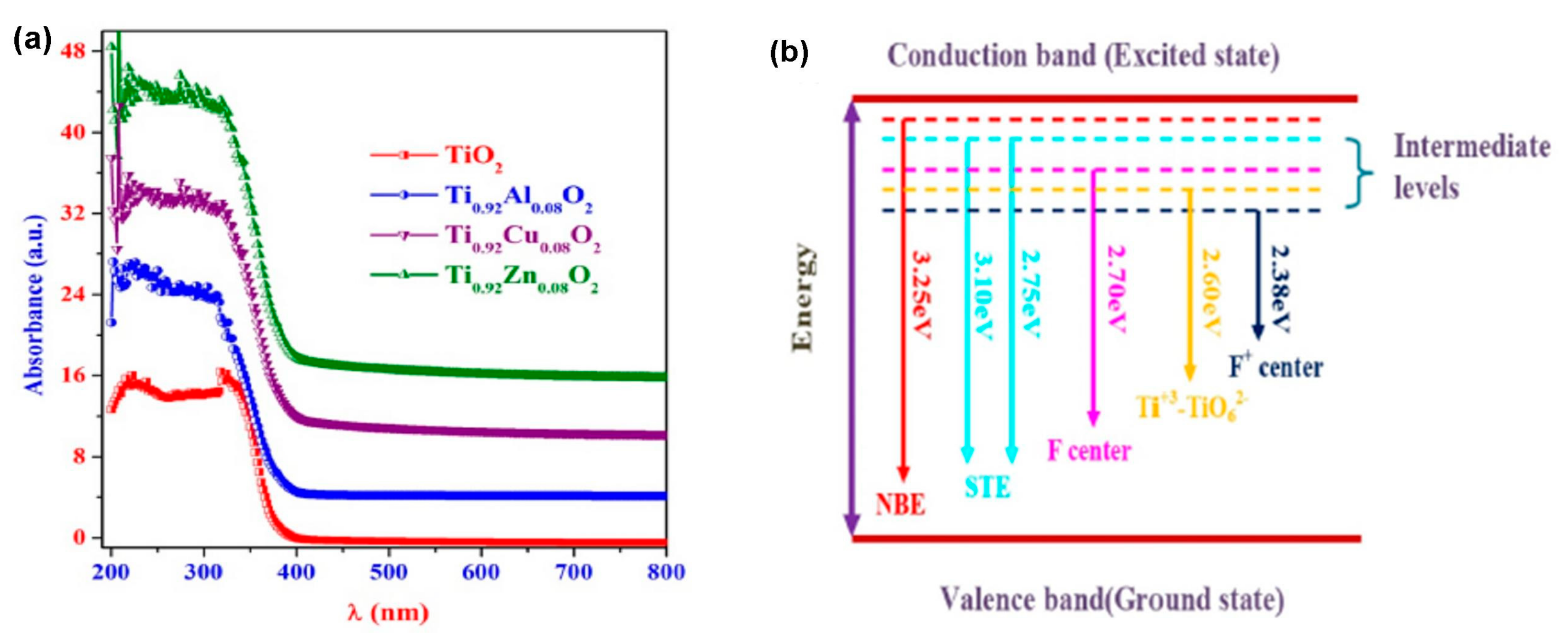
2.3. Optical Properties
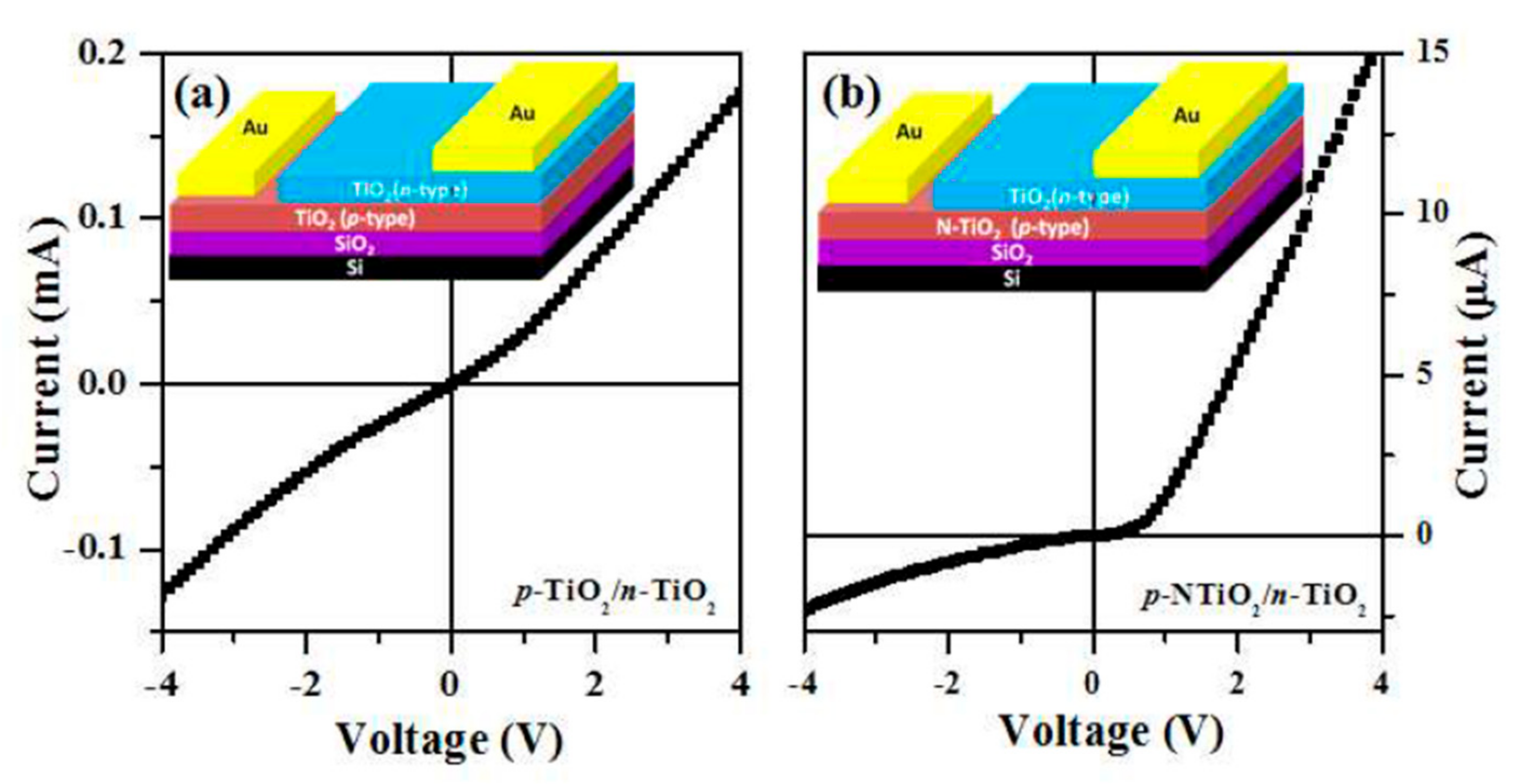
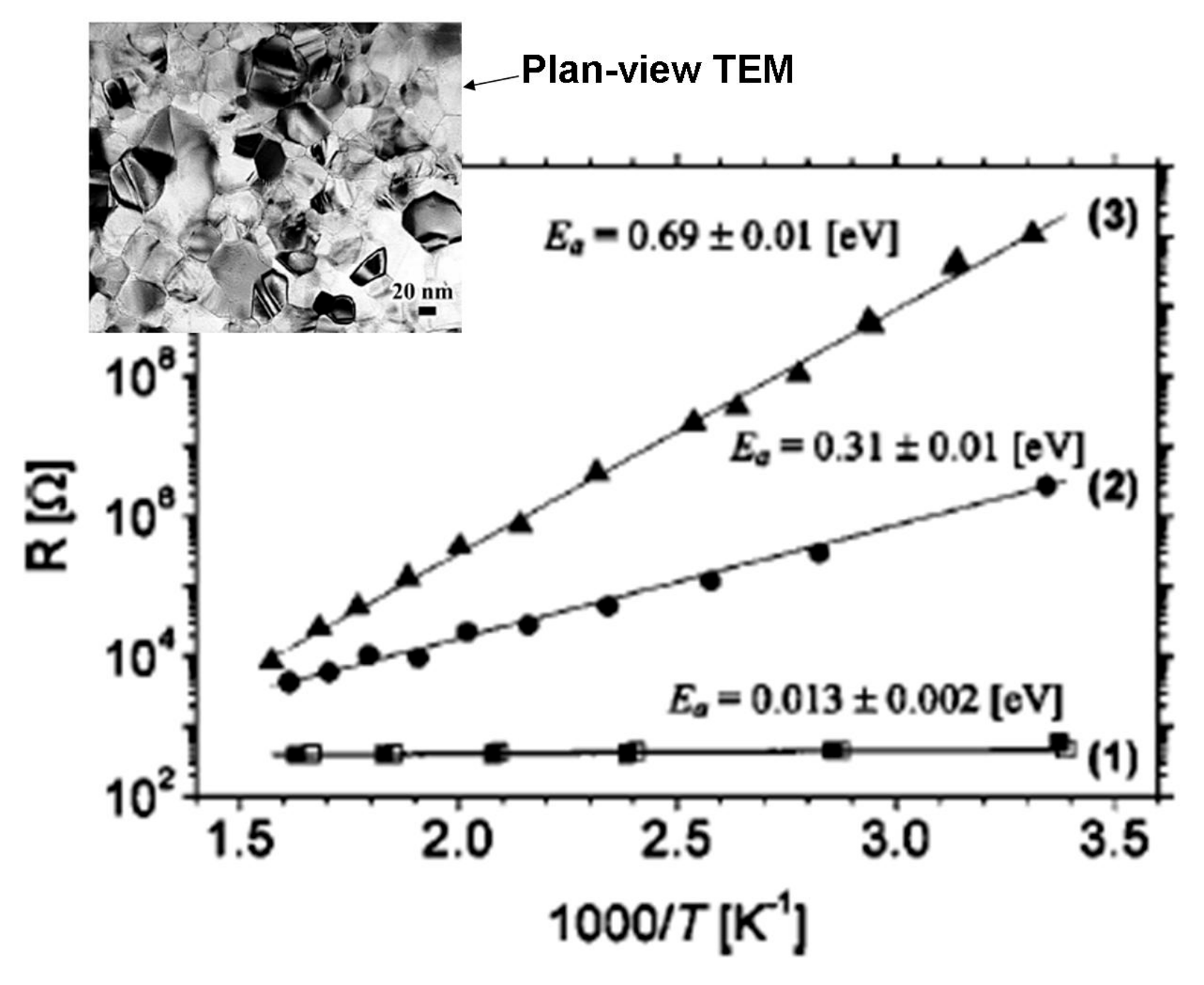
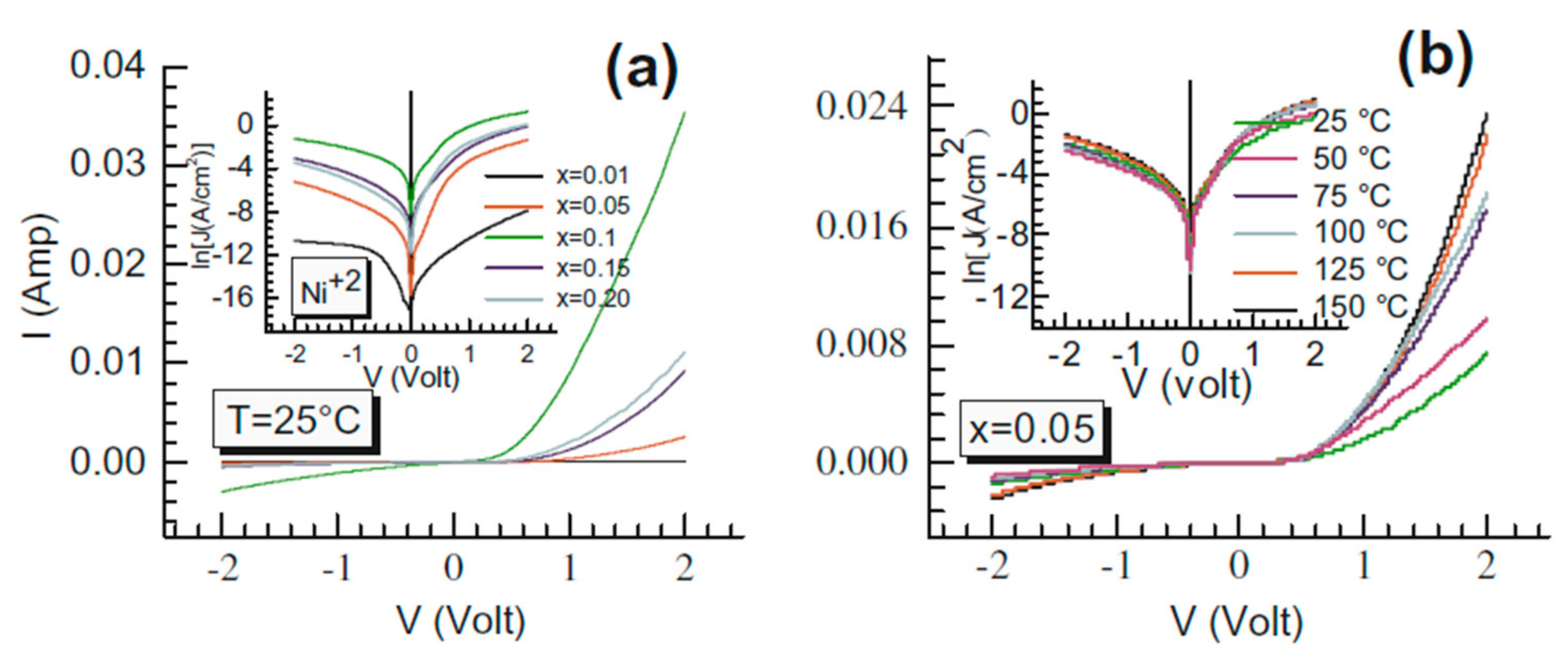
2.4. Electrical Properties
3. TiO2 as Photocatalyst
- -
- The pollutant molecules are adsorbed on the photocatalytic surface. This is not dependent on irradiation. The photocatalyst is placed in contact with the pollutant solution (with or without stirring) in the dark until an adsorption–desorption equilibrium is reached. Adsorption can be reinforced by pH control of the medium in order to favor electrostatic interactions between the pollutant molecules and the photocatalyst.
- -
- Upon activation of the photocatalyst by irradiation, electrons from the valence band jump to the conduction band, leaving behind holes. This step proceeds only if the radiation source energy is higher than the bandgap energy of the photocatalyst. Usually, semiconductor oxides have good photocatalytic properties. TiO2 is the most commonly used for this purpose.
- -
- The formation of reactive oxygen species (ROS) occurs when the holes from the valence band react with previously adsorbed water molecules on the surface of the photocatalyst and form highly reactive hydroxyl radicals. At the conduction band level, the electrons react with the previously adsorbed oxygen molecules, leading to the formation of highly reactive superoxide species.
- -
- The created ROS then attack the pollutant molecules, causing them to break down into smaller organic compounds, ideally CO2 and H2O (mineralization).
- -
- Finally, the decomposition products are desorbed from the photocatalyst surface, and the process can start over again.
3.1. Water Disinfection
- -
- To generate the long-lifespan reactive species (•O2− and H2O2);
- -
- To enhance the interaction between the bacteria and the photocatalyst;
- -
- As photocatalysts in particle form to provide more reactive surfaces for contacting bacteria.
- -
- Fullerenes are hydrophobic, and therefore, the formation of TiO2–fullerene junctions is not easy;
- -
- CNT may aggregate during the composite synthesis, limiting the available surface for TiO2 grafting;
- -
- Graphene is only stable around 160 °C, and therefore, it can lead to the formation of low-degree crystalline composites;
- -
- g-C3N4 is, so far, the most promising carbon derivative, as it maintains its 2D structure even at high temperatures and does not release any toxic elements into the environment.

3.2. Wastewater Treatment
3.3. Self-Sterilizing Coatings
3.4. Self-Cleaning Coatings for Built Environment
3.5. Food Preservation
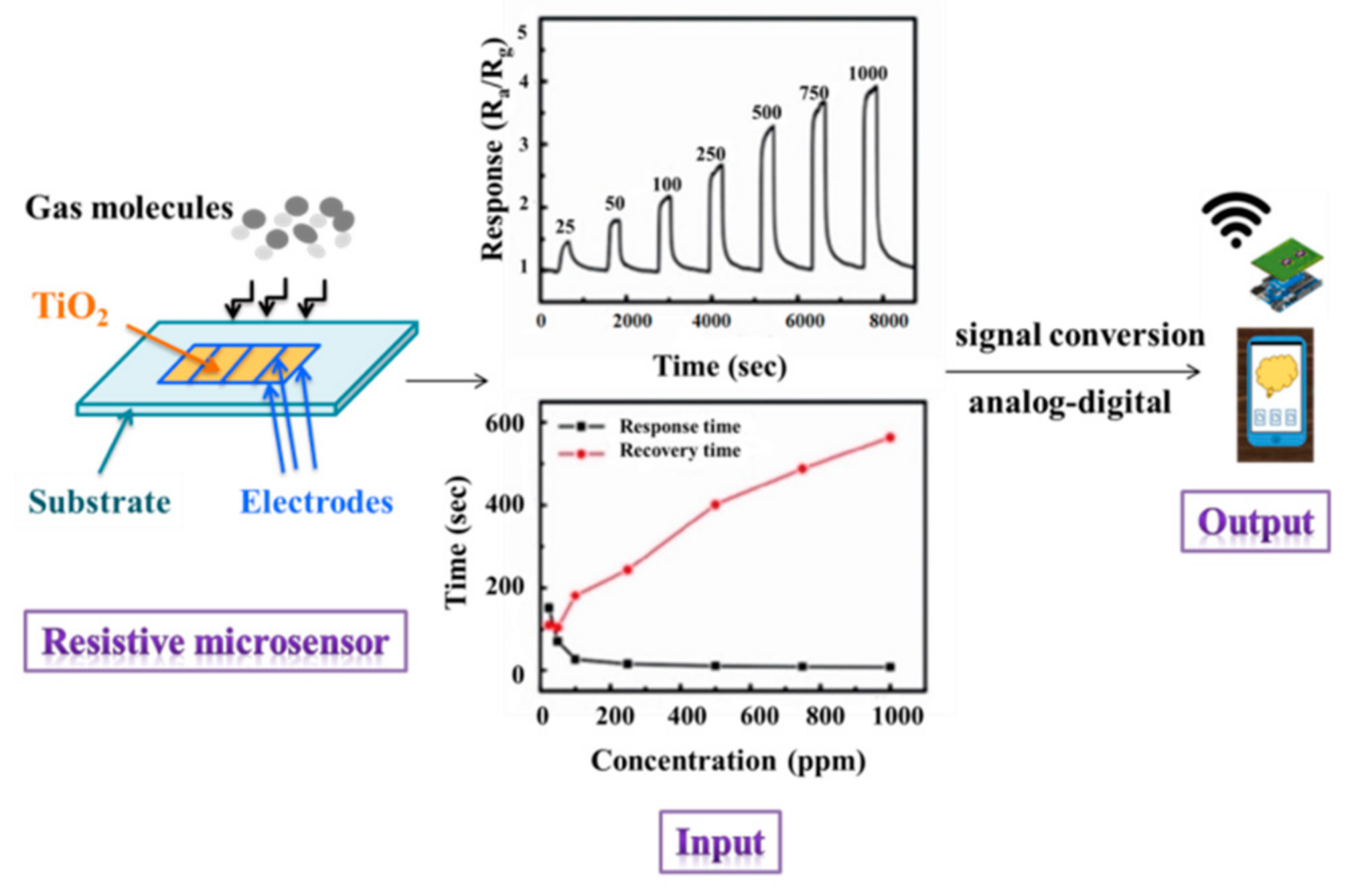
4. TiO2 as Sensor
4.1. Gas Sensors
- -
- The substrate material, especially for the electrolytic deposition of TiO2. The main problem is the side reaction of water oxidation occurring at the substrate–electrolyte interface, releasing oxygen [132].
- -
- The number and thickness of layers in the film.
- -
- The preparation method would provide the porous structure and the rough surface of the films. It is well known, for example, that the SG is a chemical method that produces the most porous films.
- -
- The type and concentration of the dopants/composites added to the TiO2 films.
- -
- The preservation of the anatase phase after the consolidation of the film.
- -
- The grain size must be small enough to significantly increase the surface area.
- -
- Tannin sulfonic acid-doped polyaniline–TiO2 composite [150];
- -
- Cellulose/TiO2/PANI composite nanofibers that incorporate p-n heterojunctions [151];
- -
- TiO2-NiO nanostructured bilayer thin films [152];
- -
- Protonated porphyrin/TiO2 composite thin films [153];
- -
- rGO-decorated TiO2 microspheres [154];
- -
- TiO2/ZnO and ZnO/TiO2 core/shell nanofibers [155];
- -
- PANI-TiO2-Au ternary self-assembly nanocomposite thin film [156];
- -
- Graphene/titanium dioxide hybrid [157];
- -
- PANI/TiO2 core–shell nanofibers [145];
- -
- TiO2/SnO2 and TiO2/CuO thin film nano-heterostructures [141].
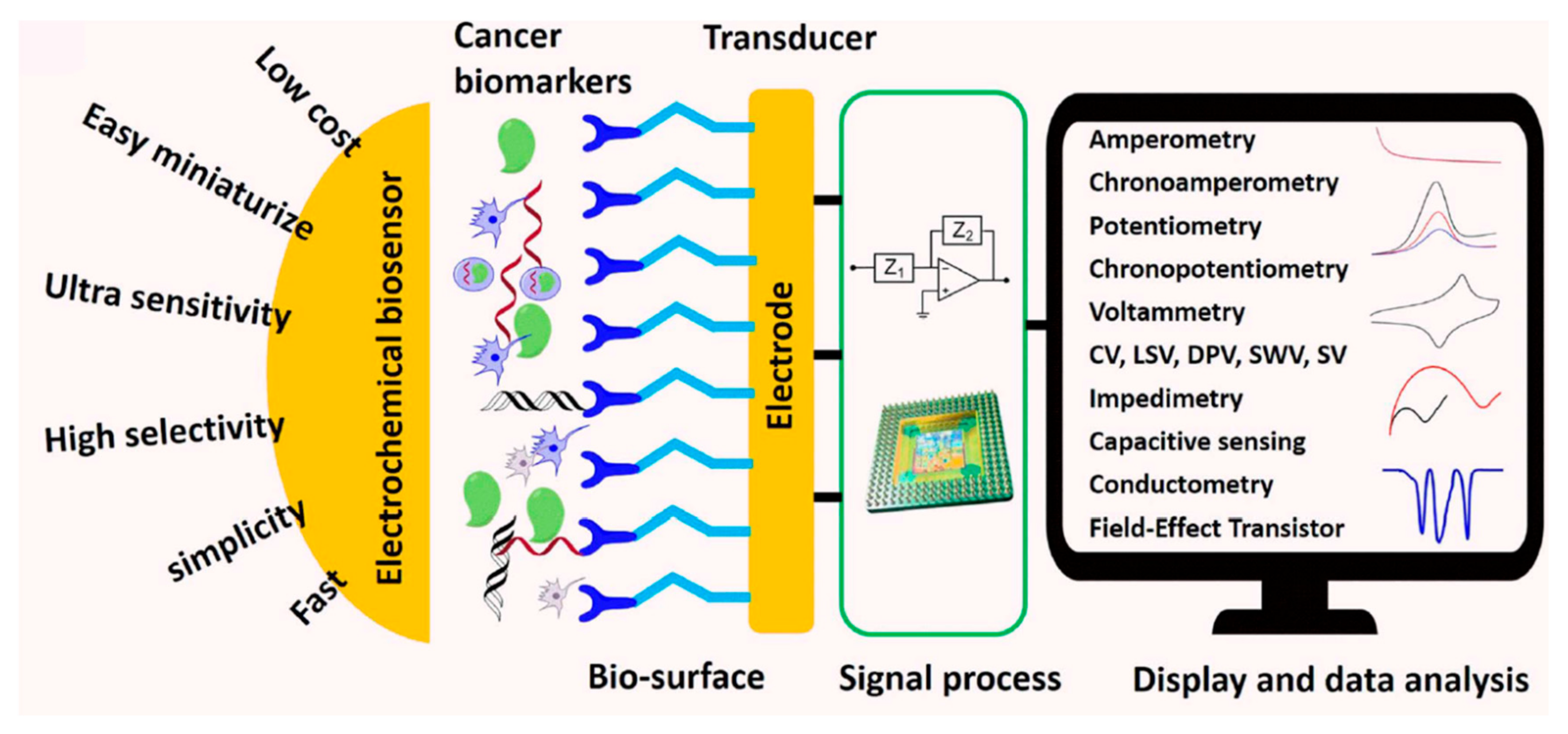
4.2. Biosensors
- -
- -
- Field effect transistor (FET) biosensor [158]. The FET acts as a transducer for the signal obtained through the biointeraction of the biorecognition element and the biomarker, especially those from early-stage cancers [166]. Azizah et al. [167] reported on a biosensor for the detection of Human Papillomavirus (HPV), a precursor to cervical cancer [167].
- -
- -
- -
- Electrical biosensors [159] are based on the trapping of an analyte by the interface of an electrode, which generates an electrical signal (current, potential, capacitance, impedance, and resistivity). There are two major classes of electrical biosensors, namely amperometric [171,172] and impedance [173] biosensors. The former measures the electrical current emmited in redox reactions, whereas the latter measures the conductance or capacitance. They are used in microbiology to detect, correctly identify, and even quantify different types of bacteria.
- -
- (Photo)electrochemical biosensors (PEC) [174,175,176,177] use photoelectrical materials as electrodes. These are able to convert light into electrical current that, when processed, is used for the detection and quantification of biomolecules. PEC sensors are sensitive and have good analytical performance, as well as a low background signal [178,179,180].
4.2.1. Cancer Detection
4.2.2. Implantology and Osteogeny
5. Conclusions
- -
- Photoactivity loss of doped TiO2 during recycling and long-term storage. To this end, extending the activation domain to a larger range of visible light is imperative.
- -
- Choosing the optimal type and concentration of dopant ions or carbon derivatives to engineer the bandgap energy of TiO2 is vital in order to attain photocatalytic and antimicrobial Vis activation.
- -
- Regarding the use of TiO2 in the field of medicine, in-depth studies on the compatibility of the TiO2-based materials (implants and stents) with the components of the human body (tissues, cartilages, and bones) are needed.
- -
- More intensive work is required to determine the TiO2 film’s cross-selectivity in the case of gas sensor applications.
- -
- Preventing fluorescence extinction in the case of fluorescent biosensors.
- -
- Oncological diseases appear suddenly and develop rapidly; therefore, early diagnosis in the initial stage is extremely important. In this respect, the development of rapid, safe, and precise diagnostic methods based on biosensors is expected.
- -
- The development of portable sensors still remains a challenge.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALA | 5-aminolevulinic acid |
| ALD | atomic layer deposition |
| AFM | atomic force microscopy |
| BBB | blood–brain barrier |
| C₆H₅CH₃ | toluene |
| C6H6 | benzene |
| CCVD | catalytic chemical vapour deposition |
| DNA | deoxyribonucleic acid |
| DSSC | Dye-sensitized solar cell |
| E. coli | Escherichia coli |
| Eg | energy bandgap |
| Ec | conduction band |
| EF | Fermi energy level |
| Ev | valence band |
| ENX | enoxacin |
| ES | Efros–Shklovskii |
| FET | field-effect transistor |
| GBs | grain boundaries |
| H2O2 | hydrogen peroxide |
| HPV | Human Papillomavirus |
| HT | hydrothermal |
| IDEs | interdigital electrodes |
| IPA | isopropyl alcohol |
| I–V | current-voltage |
| ML | molecular layering method |
| MOS | metal–oxide–semiconductor |
| MOX | metal oxide |
| MWCNTs | multi-walled carbon nanotubes |
| NIR | near infrared |
| NPs | nanoparticles |
| NTs | nanotubes (TNT titania nanotubes) |
| NTAs | nanotubes arrays |
| OPW | oilfield-produced water |
| PANI | polyaniline |
| PDD | photodynamic diagnosis |
| PEC | (photo)electrochemical biosensors |
| PEG | polyethylene glycol |
| PEI | polyethyleneimine |
| PHT | polyhexahydrotriazine |
| PL | photoluminescence |
| PLD | pulsed laser deposition |
| PPy | polypyrrole |
| rGO | reduced graphene oxide |
| RMS | root mean square roughness |
| RNA | ribonucleic acid |
| ROS | reactive oxygen species |
| RT | room temperature |
| SEM | scanning electron microscopy |
| SG | sol–gel |
| SP | spray pyrolysis |
| SPR | plasmon resonance biosensor |
| STO | strontium titanate (SrTiO3) |
| TCO | transparent conductive oxide |
| TEM | transmission electron microscopy |
| TNA | titanium dioxide nanotube arrays |
| UV | ultraviolet |
| Vis | visible |
| VOCs | volatile organic components |
| VRH | variable range hopping |
| XRD | X-ray diffraction |
References
- Kodithuwakku, P.; Jayasundara, D.R.; Munaweera, I.; Jayasinghe, R.; Thoradeniya, T.; Weerasekera, M.; Ajayan, P.M.; Kottegoda, N. A Review on Recent Developments in Structural Modification of TiO2 for Food Packaging Applications. Prog. Solid State Chem. 2022, 67, 100369. [Google Scholar] [CrossRef]
- EUR-Lex. Available online: https://eur-lex.europa.eu/eli/reg/2022/63/oj (accessed on 22 November 2023).
- Rashid, M.M.; Forte Tavčer, P.; Tomšič, B. Influence of Titanium Dioxide Nanoparticles on Human Health and the Environment. Nanomaterials 2021, 11, 2354. [Google Scholar] [CrossRef] [PubMed]
- Zaleska, A. Doped-TiO2: A Review. Recent Pat. Eng. 2008, 2, 157–164. [Google Scholar] [CrossRef]
- Li, Z.; Yao, Z.; Haidry, A.A.; Plecenik, T.; Xie, L.; Sun, L.; Fatima, Q. Resistive-Type Hydrogen Gas Sensor Based on TiO2: A Review. Int. J. Hydrogen Energy 2018, 43, 21114–21132. [Google Scholar] [CrossRef]
- Haider, A.J.; Jameel, Z.N.; Al-Hussaini, I.H.M. Review on: Titanium Dioxide Applications. Energy Procedia 2019, 157, 17–29. [Google Scholar] [CrossRef]
- Yan, J.; Li, X.; Jin, B.; Zeng, M.; Peng, R. Synthesis of TiO2/Pd and TiO2/PdO Hollow Spheres and Their Visible Light Photocatalytic Activity. Int. J. Photoenergy 2020, 2020, 4539472. [Google Scholar] [CrossRef]
- Malallah Rzaij, J.; Mohsen Abass, A. Review on: TiO2 Thin Film as a Metal Oxide Gas Sensor. J. Chem. Rev. 2020, 2, 114–121. [Google Scholar] [CrossRef]
- He, J.; Kumar, A.; Khan, M.; Lo, I.M.C. Critical Review of Photocatalytic Disinfection of Bacteria: From Noble Metals- and Carbon Nanomaterials-TiO2 Composites to Challenges of Water Characteristics and Strategic Solutions. Sci. Total Environ. 2021, 758, 143953. [Google Scholar] [CrossRef]
- Dharma, H.N.C.; Jaafar, J.; Widiastuti, N.; Matsuyama, H.; Rajabsadeh, S.; Othman, M.H.D.; Rahman, M.A.; Jafri, N.N.M.; Suhaimin, N.S.; Nasir, A.M.; et al. A Review of Titanium Dioxide (TiO2)-Based Photocatalyst for Oilfield-Produced Water Treatment. Membranes 2022, 12, 345. [Google Scholar] [CrossRef]
- Wang, J.; Guo, R.; Bi, Z.; Chen, X.; Hu, X.; Pan, W. A Review on TiO2−x-Based Materials for Photocatalytic CO2 Reduction. Nanoscale 2022, 14, 11512–11528. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Bilal, M.; Hou, J.; Butt, F.K.; Ahmad, J.; Ali, S.; Hussain, A. Photocatalytic CO2 Reduction Using TiO2-Based Photocatalysts and TiO2 Z-Scheme Heterojunction Composites: A Review. Molecules 2022, 27, 2069. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.C.; Koh, P.W.; Ponnusamy, V.K.; Lee, S.L. Titanium Dioxide and Other Nanomaterials Based Antimicrobial Additives in Functional Paints and Coatings: Review. Prog. Org. Coat. 2022, 163, 106660. [Google Scholar] [CrossRef]
- Chen, Y.; Fu, X.; Peng, Z. A Review on Oxygen-Deficient Titanium Oxide for Photocatalytic Hydrogen Production. Metals 2023, 13, 1163. [Google Scholar] [CrossRef]
- Reyes-Coronado, D.; Rodríguez-Gattorno, G.; Espinosa-Pesqueira, M.E.; Cab, C.; de Coss, R.; Oskam, G. Phase-Pure TiO2 Nanoparticles: Anatase, Brookite and Rutile. Nanotechnology 2008, 19, 145605. [Google Scholar] [CrossRef] [PubMed]
- Lance, R. Optical Analysis of Titania: Band Gaps of Brookite, Rutile and Anatase; Oregon State University: Corvallis, OR, USA, 2018. [Google Scholar]
- Kaur, A.; Umar, A.; Kansal, S.K. Sunlight-Driven Photocatalytic Degradation of Non-Steroidal Anti-Inflammatory Drug Based on TiO2 Quantum Dots. J. Colloid Interface Sci. 2015, 459, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Yang, N.; Chen, Z.; Jiang, B.; Sun, Y.; Yang, X.; Zhang, L. Fabrication of Meshes with Inverse Wettability Based on the TiO2 Nanowires for Continuous Oil/Water Separation. Chem. Eng. J. 2020, 380, 122524. [Google Scholar] [CrossRef]
- Zhang, L.; Bai, H.; Liu, L.; Sun, D.D. Dimension Induced Intrinsic Physio-Electrical Effects of Nanostructured TiO2 on Its Antibacterial Properties. Chem. Eng. J. 2018, 334, 1309–1315. [Google Scholar] [CrossRef]
- Cho, K.; Lee, S.; Kim, H.; Kim, H.-E.; Son, A.; Kim, E.; Li, M.; Qiang, Z.; Hong, S.W. Effects of Reactive Oxidants Generation and Capacitance on Photoelectrochemical Water Disinfection with Self-Doped Titanium Dioxide Nanotube Arrays. Appl. Catal. B Environ. 2019, 257, 117910. [Google Scholar] [CrossRef]
- Şennik, E.; Alev, O.; Öztürk, Z.Z. The Effect of Pd on the H2 and VOC Sensing Properties of TiO2 Nanorods. Sens. Actuators B Chem. 2016, 229, 692–700. [Google Scholar] [CrossRef]
- Lan, K.; Liu, Y.; Zhang, W.; Liu, Y.; Elzatahry, A.; Wang, R.; Xia, Y.; Al-Dhayan, D.; Zheng, N.; Zhao, D. Uniform Ordered Two-Dimensional Mesoporous TiO2 Nanosheets from Hydrothermal-Induced Solvent-Confined Monomicelle Assembly. J. Am. Chem. Soc. 2018, 140, 4135–4143. [Google Scholar] [CrossRef]
- Gopal, N.O.; Basha, M.H. TiO2 Nano-Flakes with High Activity Obtained from Phosphorus Doped TiO2 Nanoparticles by Hydrothermal Method. Ceram. Int. 2018, 44, 22129–22134. [Google Scholar] [CrossRef]
- Tang, H.; Zhang, D.; Tang, G.; Ji, X.; Li, C.; Yan, X.; Wu, Q. Low Temperature Synthesis and Photocatalytic Properties of Mesoporous TiO2 Nanospheres. J. Alloys Compd. 2014, 591, 52–57. [Google Scholar] [CrossRef]
- Lu, S.; Yang, S.; Hu, X.; Liang, Z.; Guo, Y.; Xue, Y.; Cui, H.; Tian, J. Fabrication of TiO2 Nanoflowers with Bronze (TiO2(B))/Anatase Heterophase Junctions for Efficient Photocatalytic Hydrogen Production. Int. J. Hydrogen Energy 2019, 44, 24398–24406. [Google Scholar] [CrossRef]
- Kondamareddy, K.K.; Neena, D.; Lu, D.; Peng, T.; Lopez, M.A.M.; Wang, C.; Yu, Z.; Cheng, N.; Fu, D.J.; Zhao, X.Z. Ultra-Trace (Parts per Million-Ppm) W6+ Dopant Ions Induced Anatase to Rutile Transition (ART) of Phase Pure Anatase TiO2 Nanoparticles for Highly Efficient Visible Light-Active Photocatalytic Degradation of Organic Pollutants. Appl. Surf. Sci. 2018, 456, 676–693. [Google Scholar] [CrossRef]
- Zhu, Z.; Wu, S.; Long, Y.; Zhang, L.; Xue, X.; Yin, Y.; Xu, B. Phase-Transition Kinetics of Silicon-Doped Titanium Dioxide Based on High-Temperature X-Ray-Diffraction Measurements. J. Solid State Chem. 2021, 303, 122544. [Google Scholar] [CrossRef]
- Zhang, L.; Luo, X.; Zhang, J.-D.; Long, Y.-F.; Xue, X.; Xu, B.-J. Kinetic Study on the Crystal Transformation of Fe-Doped TiO2 via In Situ High-Temperature X-Ray Diffraction and Transmission Electron Microscopy. ACS Omega 2021, 6, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Kayani, Z.N.; Intizar, T.; Riaz, S.; Naseem, S. Antibacterial, magnetic and dielectric properties of nano-structured V doped TiO2 thin films deposited by dip coating technique. Mater. Chem. Phys. 2021, 267, 124659. [Google Scholar] [CrossRef]
- Park, J.T.; Kim, D.J.; Kim, D.H.; Kim, J.H. A Facile Graft Polymerization Approach to n-Doped TiO2 Heterostructures with Enhanced Visible-Light Photocatalytic Activity. Mater. Lett. 2017, 202, 66–69. [Google Scholar] [CrossRef]
- Elmehasseb, I.; Kandil, S.; Elgendy, K. Advanced Visible-Light Applications Utilizing Modified Zn-Doped TiO2 Nanoparticles via Non-Metal in Situ Dual Doping for Wastewater Detoxification. Optik 2020, 213, 164654. [Google Scholar] [CrossRef]
- Sotelo-Vazquez, C.; Noor, N.; Kafizas, A.; Quesada-Cabrera, R.; Scanlon, D.O.; Taylor, A.; Durrant, J.R.; Parkin, I.P. Multifunctional p-Doped TiO2 Films: A New Approach to Self-Cleaning, Transparent Conducting Oxide Materials. Chem. Mater. 2015, 27, 3234–3242. [Google Scholar] [CrossRef]
- Asrafuzzaman, F.N.U.; Amin, K.F.; Gafur, M.A.; Gulshan, F. Mangifera Indica Mediated Biogenic Synthesis of Undoped and Doped TiO2 Nanoparticles and Evaluation of Their Structural, Morphological, and Photocatalytic Properties. Results Mater. 2023, 17, 100384. [Google Scholar] [CrossRef]
- Avilés-García, O.; Espino-Valencia, J.; Romero-Romero, R.; Rico-Cerda, J.; Arroyo-Albiter, M.; Solís-Casados, D.; Natividad-Rangel, R. Enhanced Photocatalytic Activity of Titania by Co-Doping with Mo and W. Catalysts 2018, 8, 631. [Google Scholar] [CrossRef]
- Wang, Y.; He, Y.; Lai, Q.; Fan, M. Review of the Progress in Preparing Nano TiO2: An Important Environmental Engineering Material. J. Environ. Sci. 2014, 26, 2139–2177. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Z.; Shi, J.; Yu, Y. One-Dimensional Titanium Dioxide Nanomaterials: Nanowires, Nanorods, and Nanobelts. Chem. Rev. 2014, 114, 9346–9384. [Google Scholar] [CrossRef] [PubMed]
- Bhandarkar, S.A.; Prathvi; Kompa, A.; Murari, M.S.; Kekuda, D.; Mohan, R.K. Investigation of Structural and Optical Properties of Spin Coated TiO2:Mn Thin Films. Opt. Mater. 2021, 118, 111254. [Google Scholar] [CrossRef]
- Anitha, V.C.; Banerjee, A.N.; Joo, S.W. Recent Developments in TiO2 as n- and p-Type Transparent Semiconductors: Synthesis, Modification, Properties, and Energy-Related Applications. J. Mater. Sci. 2015, 50, 7495–7536. [Google Scholar] [CrossRef]
- Youssef, A.M.; Yakout, S.M.; Mousa, S.M. High Relative Permittivity and Excellent Dye Photo-Elimination: Pure and (Zr4+, Y3+, Sb5+) Multi-Doped Anatase TiO2 Structure. Opt. Mater. 2023, 135, 113261. [Google Scholar] [CrossRef]
- Carp, O. Photoinduced Reactivity of Titanium Dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Vasu, K.; Sreedhara, M.B.; Ghatak, J.; Rao, C.N.R. Atomic Layer Deposition of p-Type Epitaxial Thin Films of Undoped and n-Doped Anatase TiO2. ACS Appl. Mater. Interfaces 2016, 8, 7897–7901. [Google Scholar] [CrossRef]
- Yao, W.; Luo, C.; Wu, J.B.; Hou, G. Degradation of Acid Yellow 36 Azo Dye from Textile Wastewater Using Vanadium-Doped TiO2 Photonanocatalyst. Int. J. Electrochem. Sci. 2022, 17, 220916. [Google Scholar] [CrossRef]
- Peighambardoust, N.S.; Khameneh Asl, S.; Mohammadpour, R.; Asl, S.K. Band-Gap Narrowing and Electrochemical Properties in n-Doped and Reduced Anodic TiO2 Nanotube Arrays. Electrochim. Acta 2018, 270, 245–255. [Google Scholar] [CrossRef]
- Sayed, M.; Arooj, A.; Shah, N.S.; Khan, J.A.; Shah, L.A.; Rehman, F.; Arandiyan, H.; Khan, A.M.; Khan, A.R. Narrowing the Band Gap of TiO2 by Co-Doping with Mn2+ and Co2+ for Efficient Photocatalytic Degradation of Enoxacin and Its Additional Peroxidase like Activity: A Mechanistic Approach. J. Mol. Liq. 2018, 272, 403–412. [Google Scholar] [CrossRef]
- Joshi, P.; Tiwari, S.; Punia, K.; Kumar, S. Defect Mediated Mechanism in Greenly Synthesized Undoped, Al+3, Cu+2 and Zn+2 Doped TiO2 Nanoparticles for Tailoring Bandgap, Luminescence, Magnetic and Electrical Properties. Opt. Mater. 2022, 132, 112778. [Google Scholar] [CrossRef]
- Yan, H.; Wang, R.; Liu, R.; Xu, T.; Sun, J.; Liu, L.; Wang, J. Recyclable and Reusable Direct Z-Scheme Heterojunction CeO2/TiO2 Nanotube Arrays for Photocatalytic Water Disinfection. Appl. Catal. B Environ. 2021, 291, 120096. [Google Scholar] [CrossRef]
- Roose, B.; Pathak, S.; Steiner, U. Doping of TiO2 for Sensitized Solar Cells. Chem. Soc. Rev. 2015, 44, 8326–8349. [Google Scholar] [CrossRef] [PubMed]
- Lettieri, S.; Pavone, M.; Fioravanti, A.; Santamaria Amato, L.; Maddalena, P. Charge Carrier Processes and Optical Properties in TiO2 and TiO2-Based Heterojunction Photocatalysts: A Review. Materials 2021, 14, 1645. [Google Scholar] [CrossRef]
- Nowotny, M.K.; Bak, T.; Nowotny, J. Electrical Properties and Defect Chemistry of TiO2 Single Crystal. I. Electrical Conductivity. J. Phys. Chem. B 2006, 110, 16270–16282. [Google Scholar] [CrossRef]
- Nowotny, J.; Bak, T.; Nowotny, M.; Sheppard, L. Titanium Dioxide for Solar-Hydrogen II. Defect Chemistry. Int. J. Hydrogen Energy 2007, 32, 2630–2643. [Google Scholar] [CrossRef]
- Bi, Z.; Li, K.; Jiang, C.; Zhang, J.; Ma, S.; Alberto, C.; Sun, M.; Bu, Y.; Barati, M.; Ren, S. New Insights into the Traditional Charge Compensation Theory: Amphoteric Behavior of TiO2 under the Guidance of Supply–Demand Relationship. ACS Omega 2022, 7, 21225–21232. [Google Scholar] [CrossRef]
- Chen, X.; Mao, S.S. Titanium Dioxide Nanomaterials: Synthesis, Properties, Modifications, and Applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef]
- Morgan, B.J.; Watson, G.W. Intrinsic n-Type Defect Formation in TiO2: A Comparison of Rutile and Anatase from GGA+ U Calculations. J. Phys. Chem. C 2010, 114, 2321–2328. [Google Scholar] [CrossRef]
- Nakamura, I.; Negishi, N.; Kutsuna, S.; Ihara, T.; Sugihara, S.; Takeuchi, K. Role of Oxygen Vacancy in the Plasma-Treated TiO2 Photocatalyst with Visible Light Activity for NO Removal. J. Mol. Catal. A Chem. 2000, 161, 205–212. [Google Scholar] [CrossRef]
- Pan, X.; Yang, M.-Q.; Fu, X.; Zhang, N.; Xu, Y.-J. Defective TiO2 with Oxygen Vacancies: Synthesis, Properties and Photocatalytic Applications. Nanoscale 2013, 5, 3601. [Google Scholar] [CrossRef]
- Simeonov, S.; Szekeres, A.; Covei, M.; Spassov, D.; Kitin, G.; Predoana, L.; Calderon-Moreno, J.M.; Nicolescu, M.; Preda, S.; Stroescu, H.; et al. Inter-Trap Tunneling in Vanadium Doped TiO2 Sol-Gel Films. Mater. Res. Bull. 2020, 127, 110854. [Google Scholar] [CrossRef]
- Wang, S.; Pan, L.; Song, J.-J.; Mi, W.; Zou, J.-J.; Wang, L.; Zhang, X. Titanium-Defected Undoped Anatase TiO2 with p-Type Conductivity, Room-Temperature Ferromagnetism, and Remarkable Photocatalytic Performance. J. Am. Chem. Soc. 2015, 137, 2975–2983. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.J.; Paul, M.D.; Miller, D.L.; Waldrop, J.R.; Harris, J.S. Schottky Barrier Behavior in Polycrystal GaAs. J. Vac. Sci. Technol. 1980, 17, 899–903. [Google Scholar] [CrossRef]
- McPherson, J.W.; Collis, W.; Stefanakos, E.; Safavi, A.; Abul-Fadl, A. Band Bending and Passivation Studies of GaAs Grain Boundaries. J. Electrochem. Soc. 1980, 127, 2713–2721. [Google Scholar] [CrossRef]
- Möller, H.J.; Strunk, H.P.; Werner, J.H. Polycrystalline Semiconductors; Möller, H.J., Strunk, H.P., Werner, J.H., Eds.; Springer Proceedings in Physics; Springer: Berlin/Heidelberg, Germany, 1989; Volume 35, ISBN 978-3-642-93415-5. [Google Scholar]
- Greuter, F.; Blatter, G. Electrical Properties of Grain Boundaries in Polycrystalline Compound Semiconductors. Semicond. Sci. Technol. 1990, 5, 111–137. [Google Scholar] [CrossRef]
- Rathee, D.; Arya, S.K.; Kumar, M. Analysis of TiO2 for Microelectronic Applications: Effect of Deposition Methods on Their Electrical Properties. Front. Optoelectron. China 2011, 4, 349–358. [Google Scholar] [CrossRef]
- Simeonov, S.; Kafedjiiska, E.; Szekeres, A.; Parlog, C.; Gartaer, M. Electrical Characterization of MIS Structures with Sol-Gel TiO2(La) Dielectric Films. J. Optoelectron. Adv. Mater. 2005, 7, 545–548. [Google Scholar]
- Kumar, A.; Mondal, S.; Rao, K.S.R.K. DLTS Analysis of Amphoteric Interface Defects in High-TiO2 MOS Structures Prepared by Sol-Gel Spin-Coating. AIP Adv. 2015, 5, 117122. [Google Scholar] [CrossRef]
- Gyanan; Mondal, S.; Kumar, A. Tunable Dielectric Properties of TiO2 Thin Film Based MOS Systems for Application in Microelectronics. Superlattices Microstruct. 2016, 100, 876–885. [Google Scholar] [CrossRef]
- Kumar, A.; Mondal, S.; Koteswara Rao, K.S.R. Critical Investigation of High Performance Spin-Coated High-κ Titania Thin Films Based MOS Capacitor. J. Mater. Sci. Mater. Electron. 2016, 27, 5264–5270. [Google Scholar] [CrossRef]
- Mech, B.C.; Kumar, J. Effect of High-k Dielectric on the Performance of Si, InAs and CNT FET. Micro Nano Lett. 2017, 12, 624–629. [Google Scholar] [CrossRef]
- Rothschild, A.; Komem, Y.; Levakov, A.; Ashkenasy, N.; Shapira, Y. Electronic and Transport Properties of Reduced and Oxidized Nanocrystalline TiO2 Films. Appl. Phys. Lett. 2003, 82, 574–576. [Google Scholar] [CrossRef]
- Mehdizadeh, P.; Tavangar, Z.; Shabani, N.; Hamadanian, M. Visible Light Activity of Nitrogen-Doped TiO2 by Sol-Gel Method Using Various Nitrogen Sources. J. Nanostruct. 2020, 10, 307–316. [Google Scholar] [CrossRef]
- Das, S.; Liu, D.; Park, J.B.; Hahn, Y.B. Metal-Ion Doped p-Type TiO2 Thin Films and Their Applications for Heterojunction Devices. J. Alloys Compd. 2013, 553, 188–193. [Google Scholar] [CrossRef]
- Yildiz, A.; Lisesivdin, S.B.; Kasap, M.; Mardare, D. Electrical Properties of TiO2 Thin Films. J. Non. Cryst. Solids 2008, 354, 4944–4947. [Google Scholar] [CrossRef]
- Kneiß, M.; Jenderka, M.; Brachwitz, K.; Lorenz, M.; Grundmann, M. Modeling the Electrical Transport in Epitaxial Undoped and Ni-, Cr-, and W-Doped TiO2 Anatase Thin Films. Appl. Phys. Lett. 2014, 105, 3–8. [Google Scholar] [CrossRef]
- Mott, N.F. Conduction in Glasses Containing Transition Metal Ions. J. Non. Cryst. Solids 1968, 1, 1–17. [Google Scholar] [CrossRef]
- Shklovskii, B.I.; Efros, A.L. Electronic Properties of Doped Semiconductors; Springer: Berlin, Germany, 1984; ISBN 9783662024058. [Google Scholar]
- Zhao, Y.L.; Lv, W.M.; Liu, Z.Q.; Zeng, S.W.; Motapothula, M.; Dhar, S.; Ariando; Wang, Q.; Venkatesan, T. Variable Range Hopping in TiO2 Insulating Layers for Oxide Electronic Devices. AIP Adv. 2012, 2, 012129. [Google Scholar] [CrossRef]
- Monteagudo, J.M.; Durán, A.; Martín, I.S.; Acevedo, A.M. A Novel Combined Solar Pasteurizer/TiO2 Continuous-Flow Reactor for Decontamination and Disinfection of Drinking Water. Chemosphere 2017, 168, 1447–1456. [Google Scholar] [CrossRef] [PubMed]
- Nyangaresi, P.O.; Qin, Y.; Chen, G.; Zhang, B.; Lu, Y.; Shen, L. Comparison of UV-LED Photolytic and UV-LED/TiO2 Photocatalytic Disinfection for Escherichia Coli in Water. Catal. Today 2019, 335, 200–207. [Google Scholar] [CrossRef]
- De Pasquale, I.; Lo Porto, C.; Dell’Edera, M.; Curri, M.L.; Comparelli, R. TiO2-Based Nanomaterials Assisted Photocatalytic Treatment for Virus Inactivation: Perspectives and Applications. Curr. Opin. Chem. Eng. 2021, 34, 100716. [Google Scholar] [CrossRef] [PubMed]
- Khaiboullina, S.; Uppal, T.; Dhabarde, N.; Subramanian, V.R.; Verma, S.C. Inactivation of Human Coronavirus by Titania Nanoparticle Coatings and UVC Radiation: Throwing Light on SARS-CoV-2. Viruses 2020, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-C.; Lin, T.-H.; Hsu, K.-H.; Hsu, J.-F. Photo-Induced Disinfection Property and Photocatalytic Activity Based on the Synergistic Catalytic Technique of Ag Doped TiO2 Nanofibers. Appl. Surf. Sci. 2019, 484, 326–334. [Google Scholar] [CrossRef]
- Gadgil, D.J.; Shetty Kodialbail, V. Suspended and Polycaprolactone Immobilized Ag@TiO2/Polyaniline Nanocomposites for Water Disinfection and Endotoxin Degradation by Visible and Solar Light-Mediated Photocatalysis. Environ. Sci. Pollut. Res. 2021, 28, 12780–12791. [Google Scholar] [CrossRef] [PubMed]
- Torres-Limiñana, J.; Feregrino-Pérez, A.A.; Vega-González, M.; Escobar-Alarcón, L.; Cervantes-Chávez, J.A.; Esquivel, K. Green Synthesis via Eucalyptus globulus L. Extract of Ag-TiO2 Catalyst: Antimicrobial Activity Evaluation toward Water Disinfection Process. Nanomaterials 2022, 12, 1944. [Google Scholar] [CrossRef]
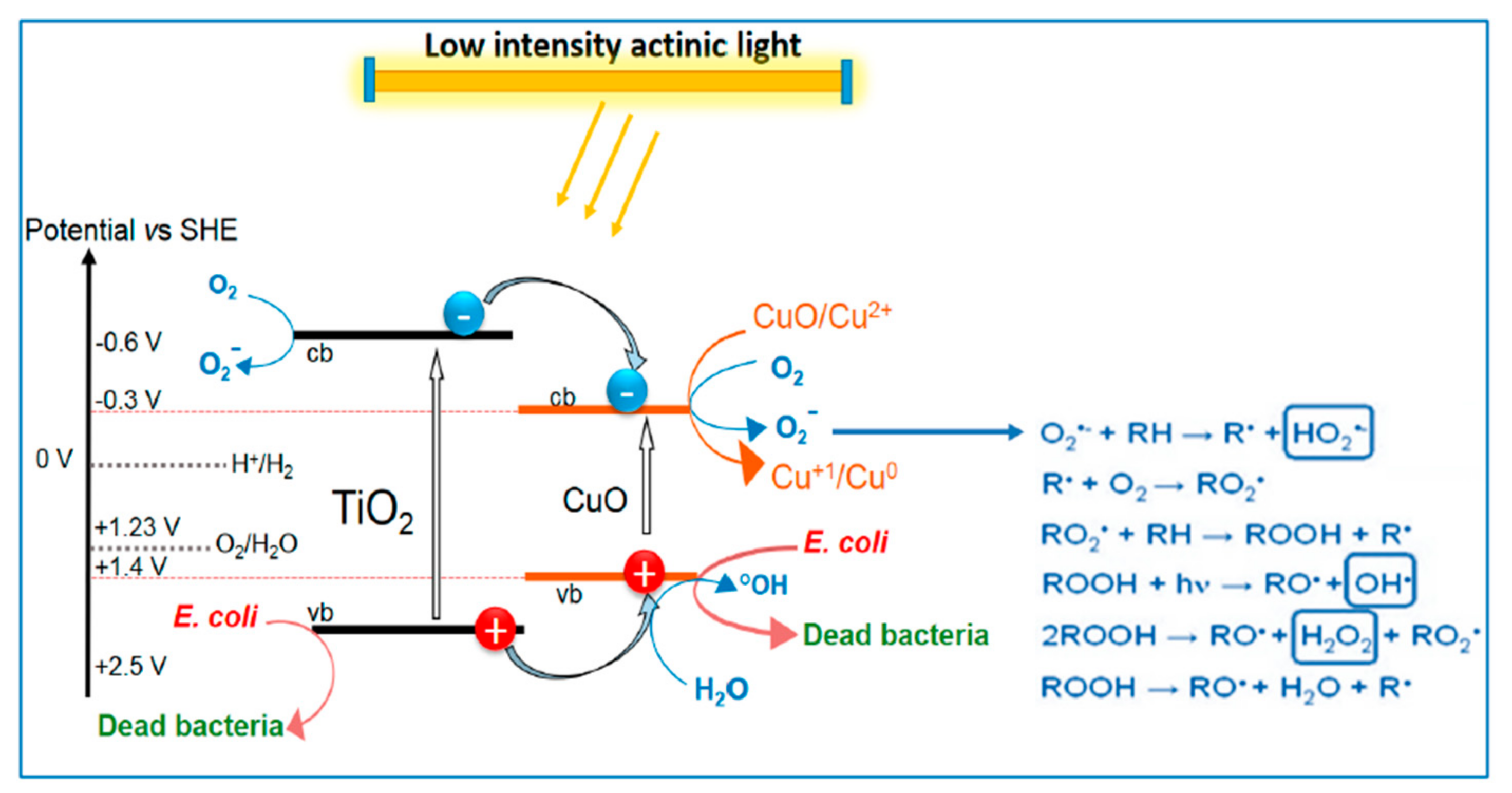
- Miao, Y.; Xu, X.; Liu, K.; Wang, N. Preparation of Novel Cu/TiO2 Mischcrystal Composites and Antibacterial Activities for Escherichia Coli under Visible Light. Ceram. Int. 2017, 43, 9658–9663. [Google Scholar] [CrossRef]
- Pablos, C.; Marugán, J.; van Grieken, R.; Dunlop, P.; Hamilton, J.; Dionysiou, D.; Byrne, J. Electrochemical Enhancement of Photocatalytic Disinfection on Aligned TiO2 and Nitrogen Doped TiO2 Nanotubes. Molecules 2017, 22, 704. [Google Scholar] [CrossRef]
- Makropoulou, T.; Panagiotopoulou, P.; Venieri, D. n-doped TiO2 Photocatalysts for Bacterial Inactivation in Water. J. Chem. Technol. Biotechnol. 2018, 93, 2518–2526. [Google Scholar] [CrossRef]
- Levchuk, I.; Homola, T.; Moreno-Andrés, J.; Rueda-Márquez, J.J.; Dzik, P.; Moríñigo, M.Á.; Sillanpää, M.; Manzano, M.A.; Vahala, R. Solar Photocatalytic Disinfection Using Ink-Jet Printed Composite TiO2/SiO2 Thin Films on Flexible Substrate: Applicability to Drinking and Marine Water. Sol. Energy 2019, 191, 518–529. [Google Scholar] [CrossRef]
- Keeley, M.; Kisslinger, K.; Adamson, C.; Furlan, P.Y. Magnetically Recoverable and Reusable Titanium Dioxide Nanocomposite for Water Disinfection. J. Mar. Sci. Eng. 2021, 9, 943. [Google Scholar] [CrossRef]
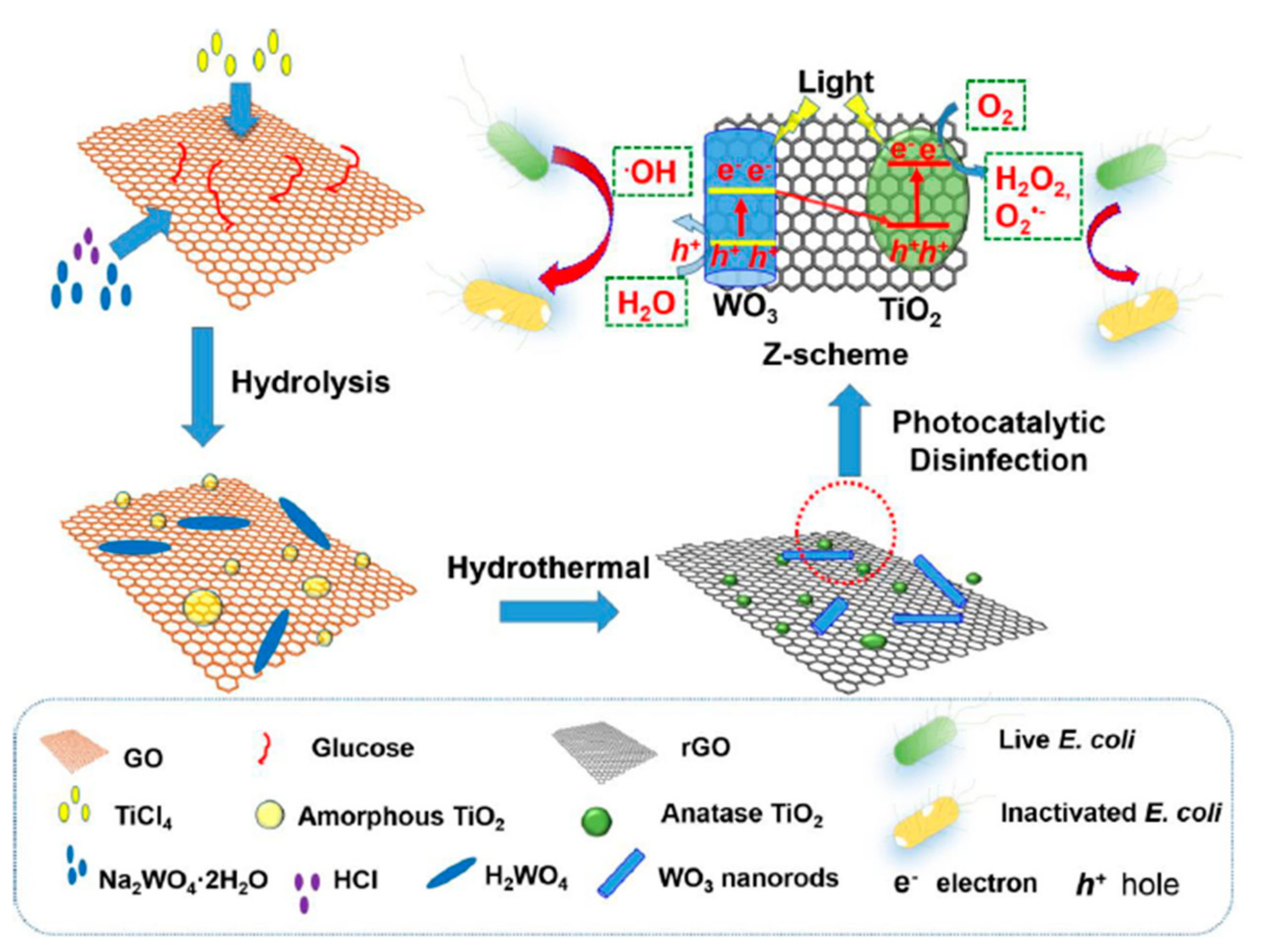
- Zeng, X.; Wang, Z.; Wang, G.; Gengenbach, T.R.; McCarthy, D.T.; Deletic, A.; Yu, J.; Zhang, X. Highly Dispersed TiO2 Nanocrystals and WO3 Nanorods on Reduced Graphene Oxide: Z-Scheme Photocatalysis System for Accelerated Photocatalytic Water Disinfection. Appl. Catal. B Environ. 2017, 218, 163–173. [Google Scholar] [CrossRef]
- Berberidou, C.; Kyzas, G.Z.; Paspaltsis, I.; Sklaviadis, T.; Poulios, I. Photocatalytic Disinfection and Purification of Water Employing Reduced Graphene Oxide/TiO2 Composites. J. Chem. Technol. Biotechnol. 2019, 94, 3905–3914. [Google Scholar] [CrossRef]
- Dey, B.; Bulou, S.; Ravisy, W.; Gautier, N.; Richard-Plouet, M.; Granier, A.; Choquet, P. Low-Temperature Deposition of Self-Cleaning Anatase TiO2 Coatings on Polymer Glazing via Sequential Continuous and Pulsed PECVD. Surf. Coat. Technol. 2022, 436, 128256. [Google Scholar] [CrossRef]
- Covei, M.; Bogatu, C.; Gheorghita, S.; Duta, A.; Stroescu, H.; Nicolescu, M.; Calderon-Moreno, J.M.; Atkinson, I.; Bratan, V.; Gartner, M. Influence of the Deposition Parameters on the Properties of TiO2 Thin Films on Spherical Substrates. Materials 2023, 16, 4899. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Sahu, K.; Pandey, A.; Kumar, M.; Ghosh, T.; Satpati, B.; Som, T.; Varma, S.; Avasthi, D.K.; Mohapatra, S. Atom Beam Sputtered Ag-TiO2 Plasmonic Nanocomposite Thin Films for Photocatalytic Applications. Appl. Surf. Sci. 2017, 411, 347–354. [Google Scholar] [CrossRef]
- Scarisoreanu, M.; Ilie, A.G.; Goncearenco, E.; Banici, A.M.; Morjan, I.P.; Dutu, E.; Tanasa, E.; Fort, I.; Stan, M.; Mihailescu, C.N.; et al. Ag, Au and Pt Decorated TiO2 Biocompatible Nanospheres for UV&Vis Photocatalytic Water Treatment. Appl. Surf. Sci. 2020, 509, 145217. [Google Scholar] [CrossRef]
- Cao, T.; Dong, W.; Liang, Y.; Bao, Q.; Xu, C.; Bai, M.; Luo, T.; Gu, X. A Simple Solvothermal Preparation of Mg-Doped Anatase TiO2 and Its Self-Cleaning Application. Sol. Energy 2023, 249, 12–20. [Google Scholar] [CrossRef]
- Mahanta, U.; Khandelwal, M.; Deshpande, A.S. TiO2@SiO2 Nanoparticles for Methylene Blue Removal and Photocatalytic Degradation under Natural Sunlight and Low-Power UV Light. Appl. Surf. Sci. 2022, 576, 151745. [Google Scholar] [CrossRef]
- Akhter, P.; Nawaz, S.; Shafiq, I.; Nazir, A.; Shafique, S.; Jamil, F.; Park, Y.-K.; Hussain, M. Efficient Visible Light Assisted Photocatalysis Using ZnO/TiO2 Nanocomposites. Mol. Catal. 2023, 535, 112896. [Google Scholar] [CrossRef]
- Bouziani, A.; Park, J.; Ozturk, A. Synthesis of α-Fe2O3/TiO2 Heterogeneous Composites by the Sol-Gel Process and Their Photocatalytic Activity. J. Photochem. Photobiol. A Chem. 2020, 400, 112718. [Google Scholar] [CrossRef]
- Wang, K.; Ruan, J.; Song, H.; Zhang, J.; Wo, Y.; Guo, S.; Cui, D. Biocompatibility of Graphene Oxide. Nanoscale Res. Lett. 2010, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zhi, L. Graphene-based Transparent Conductive Films: Material Systems, Preparation and Applications. Small Methods 2019, 3, 1800199. [Google Scholar] [CrossRef]
- Rauwel, P.; Galeckas, A.; Ducroquet, F.; Rauwel, E. Selective Photocurrent Generation in HfO2 and Carbon Nanotube Hybrid Nanocomposites under Ultra-Violet and Visible Photoexcitations. Mater. Lett. 2019, 246, 45–48. [Google Scholar] [CrossRef]
- Koli, V.B.; Dhodamani, A.G.; Raut, A.V.; Thorat, N.D.; Pawar, S.H.; Delekar, S.D. Visible Light Photo-Induced Antibacterial Activity of TiO2-MWCNTs Nanocomposites with Varying the Contents of MWCNTs. J. Photochem. Photobiol. A Chem. 2016, 328, 50–58. [Google Scholar] [CrossRef]
- Tian, H.; Shen, K.; Hu, X.; Qiao, L.; Zheng, W. N, S Co-Doped Graphene Quantum Dots-Graphene-TiO2 Nanotubes Composite with Enhanced Photocatalytic Activity. J. Alloys Compd. 2017, 691, 369–377. [Google Scholar] [CrossRef]
- Afzal, M.J.; Pervaiz, E.; Farrukh, S.; Ahmed, T.; Bingxue, Z.; Yang, M. Highly Integrated Nanocomposites of RGO/TiO2 Nanotubes for Enhanced Removal of Microbes from Water. Environ. Technol. 2019, 40, 2567–2576. [Google Scholar] [CrossRef]
- Xu, W.; Xie, W.; Huang, X.; Chen, X.; Huang, N.; Wang, X.; Liu, J. The Graphene Oxide and Chitosan Biopolymer Loads TiO2 for Antibacterial and Preservative Research. Food Chem. 2017, 221, 267–277. [Google Scholar] [CrossRef]
- Raja, A.; Selvakumar, K.; Rajasekaran, P.; Arunpandian, M.; Ashokkumar, S.; Kaviyarasu, K.; Asath Bahadur, S.; Swaminathan, M. Visible Active Reduced Graphene Oxide Loaded Titania for Photodecomposition of Ciprofloxacin and Its Antibacterial Activity. Colloids Surfaces A Physicochem. Eng. Asp. 2019, 564, 23–30. [Google Scholar] [CrossRef]
- Gillespie, P.N.O.; Martsinovich, N. Origin of Charge Trapping in TiO2/Reduced Graphene Oxide Photocatalytic Composites: Insights from Theory. ACS Appl. Mater. Interfaces 2019, 11, 31909–31922. [Google Scholar] [CrossRef] [PubMed]
- Giovannetti, R.; Rommozzi, E.; Zannotti, M.; D’Amato, C.A. Recent Advances in Graphene Based TiO2 Nanocomposites (GTiO2Ns) for Photocatalytic Degradation of Synthetic Dyes. Catalysts 2017, 7, 305. [Google Scholar] [CrossRef]
- Sharma, M.; Behl, K.; Nigam, S.; Joshi, M. TiO2-GO Nanocomposite for Photocatalysis and Environmental Applications: A Green Synthesis Approach. Vacuum 2018, 156, 434–439. [Google Scholar] [CrossRef]
- Covei, M.; Perniu, D.; Bogatu, C.; Duta, A.; Visa, I. Photocatalytic Composite Thin Films with Controlled Optical Properties Based on TiO2, WO3 and RGO. Surfaces Interfaces 2022, 31, 102075. [Google Scholar] [CrossRef]
- Covei, M.; Visa, I.; Duta, A. Self-Cleaning Photocatalytic Ceramic Coatings. In Advanced Ceramic Coatings; Elsevier: Amsterdam, The Netherlands, 2023; pp. 137–155. [Google Scholar]
- Lin, N.; Verma, D.; Saini, N.; Arbi, R.; Munir, M.; Jovic, M.; Turak, A. Antiviral Nanoparticles for Sanitizing Surfaces: A Roadmap to Self-Sterilizing against COVID-19. Nano Today 2021, 40, 101267. [Google Scholar] [CrossRef] [PubMed]
- Sadowski, R.; Wach, A.; Buchalska, M.; Kuśtrowski, P.; Macyk, W. Photosensitized TiO2 Films on Polymers–Titania-Polymer Interactions and Visible Light Induced Photoactivity. Appl. Surf. Sci. 2019, 475, 710–719. [Google Scholar] [CrossRef]
- Rtimi, S.; Giannakis, S.; Pulgarin, C. Self-Sterilizing Sputtered Films for Applications in Hospital Facilities. Molecules 2017, 22, 1074. [Google Scholar] [CrossRef]
- Rtimi, S.; Pulgarin, C.; Robyr, M.; Aybush, A.; Shelaev, I.; Gostev, F.; Nadtochenko, V.; Kiwi, J. Insight into the Catalyst/Photocatalyst Microstructure Presenting the Same Composition but Leading to a Variance in Bacterial Reduction under Indoor Visible Light. Appl. Catal. B Environ. 2017, 208, 135–147. [Google Scholar] [CrossRef]
- Rtimi, S.; Robyr, M.; Pulgarin, C.; Lavanchy, J.C.; Kiwi, J. A New Perspective in the Use of FeOx-TiO2 Photocatalytic Films: Indole Degradation in the Absence of Fe-Leaching. J. Catal. 2016, 342, 184–192. [Google Scholar] [CrossRef]
- Syafiq, A.; Vengadaesvaran, B.; Pandey, A.K.; Rahim, N.A. Superhydrophilic Smart Coating for Self-Cleaning Application on Glass Substrate. J. Nanomater. 2018, 2018, 6412601. [Google Scholar] [CrossRef]
- Luna, M.; Delgado, J.; Gil, M.; Mosquera, M. TiO2-SiO2 Coatings with a Low Content of AuNPs for Producing Self-Cleaning Building Materials. Nanomaterials 2018, 8, 177. [Google Scholar] [CrossRef] [PubMed]
- Bergamonti, L.; Predieri, G.; Paz, Y.; Fornasini, L.; Lottici, P.P.; Bondioli, F. Enhanced Self-Cleaning Properties of N-Doped TiO2 Coating for Cultural Heritage. Microchem. J. 2017, 133, 1–12. [Google Scholar] [CrossRef]
- Veziroglu, S.; Hwang, J.; Drewes, J.; Barg, I.; Shondo, J.; Strunskus, T.; Polonskyi, O.; Faupel, F.; Aktas, O.C. PdO Nanoparticles Decorated TiO2 Film with Enhanced Photocatalytic and Self-Cleaning Properties. Mater. Today Chem. 2020, 16, 100251. [Google Scholar] [CrossRef]
- Padmanabhan, N.T.; John, H. Titanium Dioxide Based Self-Cleaning Smart Surfaces: A Short Review. J. Environ. Chem. Eng. 2020, 8, 104211. [Google Scholar] [CrossRef]
- Kavitha, M.K.; Rolland, L.; Johnson, L.; John, H.; Jayaraj, M.K. Visible Light Responsive Superhydrophilic TiO2/Reduced Graphene Oxide Coating by Vacuum-Assisted Filtration and Transfer Method for Self-Cleaning Application. Mater. Sci. Semicond. Process. 2020, 113, 105011. [Google Scholar] [CrossRef]
- Salarbashi, D.; Tafaghodi, M.; Bazzaz, B.S.F. Soluble Soybean Polysaccharide/TiO2 Bionanocomposite Film for Food Application. Carbohydr. Polym. 2018, 186, 384–393. [Google Scholar] [CrossRef]
- Dias, V.; Maciel, H.; Fraga, M.; Lobo, A.; Pessoa, R.; Marciano, F. Atomic Layer Deposited TiO2 and Al2O3 Thin Films as Coatings for Aluminum Food Packaging Application. Materials 2019, 12, 682. [Google Scholar] [CrossRef]
- Athir, N.; Shah, S.A.A.; Shehzad, F.K.; Cheng, J.; Zhang, J.; Shi, L. Rutile TiO2 Integrated Zwitterion Polyurethane Composite Films as an Efficient Photostable Food Packaging Material. React. Funct. Polym. 2020, 157, 104733. [Google Scholar] [CrossRef]
- Anaya-Esparza, L.M.; Ruvalcaba-Gómez, J.M.; Maytorena-Verdugo, C.I.; González-Silva, N.; Romero-Toledo, R.; Aguilera-Aguirre, S.; Pérez-Larios, A.; Montalvo-González, E. Chitosan-TiO2: A Versatile Hybrid Composite. Materials 2020, 13, 811. [Google Scholar] [CrossRef]
- Abutalib, M.M.; Rajeh, A. Enhanced Structural, Electrical, Mechanical Properties and Antibacterial Activity of Cs/PEO Doped Mixed Nanoparticles (Ag/TiO2) for Food Packaging Applications. Polym. Test. 2021, 93, 107013. [Google Scholar] [CrossRef]
- Mesgari, M.; Aalami, A.H.; Sahebkar, A. Antimicrobial Activities of Chitosan/Titanium Dioxide Composites as a Biological Nanolayer for Food Preservation: A Review. Int. J. Biol. Macromol. 2021, 176, 530–539. [Google Scholar] [CrossRef] [PubMed]
- Van Nguyen, S.; Lee, B.-K. PVA/CNC/TiO2 Nanocomposite for Food-Packaging: Improved Mechanical, UV/Water Vapor Barrier, and Antimicrobial Properties. Carbohydr. Polym. 2022, 298, 120064. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Rhim, J.-W. Titanium Dioxide (TiO2) for the Manufacture of Multifunctional Active Food Packaging Films. Food Packag. Shelf Life 2022, 31, 100806. [Google Scholar] [CrossRef]
- Sonker, R.K.; Yadav, B.C.C.; Gupta, V.; Tomar, M. Fabrication and Characterization of ZnO-TiO2-PANI (ZTP) Micro/Nanoballs for the Detection of Flammable and Toxic Gases. J. Hazard. Mater. 2019, 370, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Chesler, P.; Vladut, C.M. Senzori Chimici Rezistivi; Berg: Bucuresti, Romania, 2020; ISBN 978-606-9036-60-0. [Google Scholar]
- Decroly, A.; Krumpmann, A.; Debliquy, M.; Lahem, D. Nanostructured TiO2 Layers for Photovoltaic and Gas Sensing Applications. In Green Nanotechnology-Overview and Further Prospects; IntechOpen: London, UK, 2016. [Google Scholar] [CrossRef]
- Chesler, P.; Hornoiu, C.; Bratan, V.; Munteanu, C.; Gartner, M.; Ionescu, N.I. Carbon Monoxide Sensing Properties of TiO2. Rev. Roum. Chim. 2015, 60, 227–232. [Google Scholar]
- Sabri, Y.M.; Kandjani, A.E.; Rashid, S.S.A.A.H.; Harrison, C.J.; Ippolito, S.J.; Bhargava, S.K. Soot Template TiO2 Fractals as a Photoactive Gas Sensor for Acetone Detection. Sens. Actuators B Chem. 2018, 275, 215–222. [Google Scholar] [CrossRef]
- Wilson, R.L.; Simion, C.E.; Blackman, C.S.; Carmalt, C.J.; Stanoiu, A.; Di Maggio, F.; Covington, J.A. The Effect of Film Thickness on the Gas Sensing Properties of Ultra-Thin TiO2 Films Deposited by Atomic Layer Deposition. Sensors 2018, 18, 735. [Google Scholar] [CrossRef]
- Chinh, N.D.; Kim, C.; Kim, D. UV-Light-Activated H2S Gas Sensing by a TiO2 Nanoparticulate Thin Film at Room Temperature. J. Alloys Compd. 2019, 778, 247–255. [Google Scholar] [CrossRef]
- Sertel, B.C.; Sonmez, N.A.; Kaya, M.D.; Ozcelik, S. Development of MgO:TiO2 Thin Films for Gas Sensor Applications. Ceram. Int. 2019, 45, 2917–2921. [Google Scholar] [CrossRef]
- Mokrushin, A.S.; Simonenko, E.P.; Simonenko, N.P.; Akkuleva, K.T.; Antipov, V.V.; Zaharova, N.V.; Malygin, A.A.; Bukunov, K.A.; Sevastyanov, V.G.; Kuznetsov, N.T. Oxygen Detection Using Nanostructured TiO2 Thin Films Obtained by the Molecular Layering Method. Appl. Surf. Sci. 2019, 463, 197–202. [Google Scholar] [CrossRef]
- Nagmani; Pravarthana, D.; Tyagi, A.; Jagadale, T.C.; Prellier, W.; Aswal, D.K. Highly Sensitive and Selective H2S Gas Sensor Based on TiO2 Thin Films. Appl. Surf. Sci. 2021, 549, 149281. [Google Scholar] [CrossRef]
- Alev, O.; Şennik, E.; Öztürk, Z.Z. Improved Gas Sensing Performance of p-Copper Oxide Thin Film/n-TiO2 Nanotubes Heterostructure. J. Alloys Compd. 2018, 749, 221–228. [Google Scholar] [CrossRef]
- Patil, S.M.; Dhodamani, A.G.; Vanalakar, S.A.; Deshmukh, S.P.; Delekar, S.D. Multi-Applicative Tetragonal TiO2/SnO2 Nanocomposites for Photocatalysis and Gas Sensing. J. Phys. Chem. Solids 2018, 115, 127–136. [Google Scholar] [CrossRef]
- Kim, S.M.; Kim, H.J.; Jung, H.J.; Park, J.Y.; Seok, T.J.; Choa, Y.H.; Park, T.J.; Lee, S.W. High-Performance, Transparent Thin Film Hydrogen Gas Sensor Using 2D Electron Gas at Interface of Oxide Thin Film Heterostructure Grown by Atomic Layer Deposition. Adv. Funct. Mater. 2019, 29, 1807760. [Google Scholar] [CrossRef]
- Hsu, K.-C.; Fang, T.-H.; Hsiao, Y.-J.; Wu, P.-C. Response and Characteristics of TiO2/Perovskite Heterojunctions for CO Gas Sensors. J. Alloys Compd. 2019, 794, 576–584. [Google Scholar] [CrossRef]
- Kaushik, P.; Eliáš, M.; Michalička, J.; Hegemann, D.; Pytlíček, Z.; Nečas, D.; Zajíčková, L. Atomic Layer Deposition of Titanium Dioxide on Multi-Walled Carbon Nanotubes for Ammonia Gas Sensing. Surf. Coat. Technol. 2019, 370, 235–243. [Google Scholar] [CrossRef]
- Seif, A.; Nikfarjam, A.; Ghassem, H.H.; Safe, A.M.; Nikfarjam, A.; Hajghassem, H. UV Enhanced Ammonia Gas Sensing Properties of PANI/TiO2 Core-Shell Nanofibers. Sens. Actuators B Chem. 2019, 298, 126906. [Google Scholar] [CrossRef]
- Maziarz, W. TiO2/SnO2 and TiO2/CuO Thin Film Nano-Heterostructures as Gas Sensors. Appl. Surf. Sci. 2019, 480, 361–370. [Google Scholar] [CrossRef]
- Mintcheva, N.; Srinivasan, P.; Rayappan, J.B.B.; Kuchmizhak, A.A.; Gurbatov, S.; Kulinich, S.A. Room-Temperature Gas Sensing of Laser-Modified Anatase TiO2 Decorated with Au Nanoparticles. Appl. Surf. Sci. 2020, 507, 145169. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, D.; Mi, Q. A High-Performance Room Temperature Benzene Gas Sensor Based on CoTiO3 Covered TiO2 Nanospheres Decorated with Pd Nanoparticles. Sens. Actuators B Chem. 2022, 350, 130830. [Google Scholar] [CrossRef]
- Duta, M.; Predoana, L.; Calderon-Moreno, J.M.; Preda, S.; Anastasescu, M.; Marin, A.; Dascalu, I.; Chesler, P.; Hornoiu, C.; Zaharescu, M.; et al. Nb-Doped TiO2 Sol–Gel Films for CO Sensing Applications. Mater. Sci. Semicond. Process. 2016, 42, 397–404. [Google Scholar] [CrossRef]
- Bairi, V.G.; Bourdo, S.E.; Sacre, N.; Nair, D.; Berry, B.C.; Biris, A.S.; Viswanathan, T. Ammonia Gas Sensing Behavior of Tanninsulfonic Acid Doped Polyaniline-TiO2 Composite. Sensors 2015, 15, 26415–26429. [Google Scholar] [CrossRef]
- Pang, Z.; Yang, Z.; Chen, Y.; Zhang, J.; Wang, Q.; Huang, F.; Wei, Q. A Room Temperature Ammonia Gas Sensor Based on Cellulose/TiO2/PANI Composite Nanofibers. Colloids Surfaces A Physicochem. Eng. Asp. 2016, 494, 248–255. [Google Scholar] [CrossRef]
- Kumar, A.; Sanger, A.; Kumar, A.; Chandra, R. Fast Response Ammonia Sensors Based on TiO2 and NiO Nanostructured Bilayer Thin Films. RSC Adv. 2016, 6, 77636–77643. [Google Scholar] [CrossRef]
- Castillero, P.; Roales, J.; Lopes-Costa, T.; Sánchez-Valencia, J.R.; Barranco, A.; González-Elipe, A.R.; Pedrosa, J.M. Optical Gas Sensing of Ammonia and Amines Based on Protonated Porphyrin/TiO2 Composite Thin Films. Sensors 2017, 17, 24. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhao, Y.; Wang, X.; Wang, J.; Gaskov, A.M.; Akbar, S.A. Reduced Graphene Oxide (RGO) Decorated TiO2 Microspheres for Selective Room-Temperature Gas Sensors. Sens. Actuators B Chem. 2016, 230, 330–336. [Google Scholar] [CrossRef]
- Boyadjiev, S.I.; Kéri, O.; Bárdos, P.; Firkala, T.; Gáber, F.; Nagy, Z.K.; Baji, Z.; Takács, M.; Szilágyi, I.M. TiO2/ZnO and ZnO/TiO2Core/Shell Nanofibers Prepared by Electrospinning and Atomic Layer Deposition for Photocatalysis and Gas Sensing. Appl. Surf. Sci. 2017, 424, 190–197. [Google Scholar] [CrossRef]
- Liu, C.; Tai, H.; Zhang, P.; Ye, Z.; Su, Y.; Jiang, Y. Enhanced Ammonia-Sensing Properties of PANI-TiO2-Au Ternary Self-Assembly Nanocomposite Thin Film at Room Temperature. Sens. Actuators B Chem. 2017, 246, 85–95. [Google Scholar] [CrossRef]
- Ye, Z.; Tai, H.; Guo, R.; Yuan, Z.; Liu, C.; Su, Y.; Chen, Z.; Jiang, Y. Excellent Ammonia Sensing Performance of Gas Sensor Based on Graphene/Titanium Dioxide Hybrid with Improved Morphology. Appl. Surf. Sci. 2017, 419, 84–90. [Google Scholar] [CrossRef]
- Pourmadadi, M.; Rajabzadeh-Khosroshahi, M.; Eshaghi, M.M.; Rahmani, E.; Motasadizadeh, H.; Arshad, R.; Rahdar, A.; Pandey, S. TiO2-Based Nanocomposites for Cancer Diagnosis and Therapy: A Comprehensive Review. J. Drug Deliv. Sci. Technol. 2023, 82, 104370. [Google Scholar] [CrossRef]
- Nadzirah, S.; Gopinath, S.C.B.; Parmin, N.A.; Hamzah, A.A.; Mohamed, M.A.; Chang, E.Y.; Dee, C.F. State-of-the-Art on Functional Titanium Dioxide-Integrated Nano-Hybrids in Electrical Biosensors. Crit. Rev. Anal. Chem. 2022, 52, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Brainina, K.Z. Sensors and Sample Preparation in Stripping Voltammetry. Anal. Chim. Acta 1995, 305, 146–153. [Google Scholar] [CrossRef]
- Mathew, S.; Kumar Prasad, A.; Benoy, T.; Rakesh, P.P.; Hari, M.; Libish, T.M.; Radhakrishnan, P.; Nampoori, V.P.N.; Vallabhan, C.P.G. UV-Visible Photoluminescence of TiO2 Nanoparticles Prepared by Hydrothermal Method. J. Fluoresc. 2012, 22, 1563–1569. [Google Scholar] [CrossRef]
- Ariño, C.; Serrano, N.; Díaz-Cruz, J.M.; Esteban, M. Voltammetric Determination of Metal Ions beyond Mercury Electrodes. A Review. Anal. Chim. Acta 2017, 990, 11–53. [Google Scholar] [CrossRef] [PubMed]
- Brainina, K.Z. Film Stripping Voltammetry. Talanta 1971, 18, 513–539. [Google Scholar] [CrossRef] [PubMed]
- Brainina, K.Z. Inverse Voltammetry (Stripping Analysis) in the Investigation of Biologically Important Compounds. J. Electroanal. Chem. Interfacial Electrochem. 1981, 128, 479–485. [Google Scholar] [CrossRef]
- Kanehira, K.; Yano, Y.; Hasumi, H.; Fukuhara, H.; Inoue, K.; Hanazaki, K.; Yao, M. Fluorescence Enhancement Effect of TiO2 Nanoparticles and Application for Photodynamic Diagnosis. Int. J. Mol. Sci. 2019, 20, 3698. [Google Scholar] [CrossRef]
- Adzhri, R.; Arshad, M.M.; Gopinath, S.C.; Ruslinda, A.R.; Fathil, M.F.; Ibau, C.; Nuzaihan, M. Enhanced Sensitivity Mediated Ambipolar Conduction with p-Type TiO2 Anatase Transducer for Biomarker Capturing. Sens. Actuators A Phys. 2017, 259, 57–67. [Google Scholar] [CrossRef]
- Azizah, N.; Hashim, U.; Nadzirah, S.; Ruslinda, A.R. Rapid and Sensitive Strategy for Human Papillomavirus (HPV) Detection Using a Gene-Based DNA Nanobiosensor. In Proceedings of the 2014 IEEE Conference on Biomedical Engineering and Sciences (IECBES), Kuala Lumpur, Malaysia, 8–10 December 2014; IEEE: Piscataway, NJ, USA, 2014; pp. 960–963. [Google Scholar]
- Xu, C.; Thakur, A.; Li, Z.; Yang, T.; Zhao, C.; Li, Y.; Lee, Y.; Wu, C.-M.L. Determination of Glioma Cells’ Malignancy and Their Response to TMZ via Detecting Exosomal BIGH3 by a TiO2-CTFE-AuNIs Plasmonic Biosensor. Chem. Eng. J. 2021, 415, 128948. [Google Scholar] [CrossRef]
- Yan, S.R.; Foroughi, M.M.; Safaei, M.; Jahani, S.; Ebrahimpour, N.; Borhani, F.; Rezaei Zade Baravati, N.; Aramesh-Boroujeni, Z.; Foong, L.K. A Review: Recent Advances in Ultrasensitive and Highly Specific Recognition Aptasensors with Various Detection Strategies. Int. J. Biol. Macromol. 2020, 155, 184–207. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.S.; Wang, F.; Ge, Y.; Lo, P.K. Recent Developments in Aptasensors for Diagnostic Applications. ACS Appl. Mater. Interfaces 2021, 13, 9329–9358. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chen, X.; Wang, X.; Han, X.; Liu, S.; Zhao, C. Electrochemical Synthesis of Gold Nanostructure Modified Electrode and Its Development in Electrochemical DNA Biosensor. Biosens. Bioelectron. 2011, 30, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Skotadis, E.; Voutyras, K.; Chatzipetrou, M.; Tsekenis, G.; Patsiouras, L.; Madianos, L.; Chatzandroulis, S.; Zergioti, I.; Tsoukalas, D. Label-Free DNA Biosensor Based on Resistance Change of Platinum Nanoparticles Assemblies. Biosens. Bioelectron. 2016, 81, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Stojko, N.Y.; Brainina, K.; Faller, C.; Henze, G. Stripping Voltammetric Determination of Mercury at Modified Solid Electrodes. Anal. Chim. Acta 1998, 371, 145–153. [Google Scholar] [CrossRef]
- Liu, W.; Yao, C.; Cui, H.; Cang, Y.; Zhang, Z.; Miao, Y.; Xin, Y. A Nano-Enzymatic Photoelectrochemical L-Cysteine Biosensor Based on Bi2MoO6 Modified Honeycomb TiO2 Nanotube Arrays Composite. Microchem. J. 2022, 175, 107200. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, M.; Su, W.; Wu, B.; Yang, Z.; Wang, X.; Qiao, B.; Pei, H.; Tu, J.; Chen, D.; et al. Photoelectrochemical Enzyme Biosensor Based on TiO2 Nanorod/TiO2 Quantum Dot/Polydopamine/Glucose Oxidase Composites with Strong Visible-Light Response. Langmuir 2022, 38, 751–761. [Google Scholar] [CrossRef]
- Yang, Z.; Xu, W.; Yan, B.; Wu, B.; Ma, J.; Wang, X.; Qiao, B.; Tu, J.; Pei, H.; Chen, D.; et al. Gold and Platinum Nanoparticle-Functionalized TiO2 Nanotubes for Photoelectrochemical Glucose Sensing. ACS Omega 2022, 7, 2474–2483. [Google Scholar] [CrossRef]
- Kozitsina, A.N.; Svalova, T.S.; Malysheva, N.N.; Okhokhonin, A.V.; Vidrevich, M.B.; Brainina, K.Z. Sensors Based on Bio and Biomimetic Receptors in Medical Diagnostic, Environment, and Food Analysis. Biosensors 2018, 8, 35. [Google Scholar] [CrossRef]
- Almeida, E.S.; Silva, L.A.J.; Sousa, R.M.F.; Richter, E.M.; Foster, C.W.; Banks, C.E.; Munoz, R.A.A. Organic-Resistant Screen-Printed Graphitic Electrodes: Application to on-Site Monitoring of Liquid Fuels. Anal. Chim. Acta 2016, 934, 1–8. [Google Scholar] [CrossRef]
- Dzyadevych, S.V.; Arkhypova, V.N.; Soldatkin, A.P.; El’skaya, A.V.; Martelet, C.; Jaffrezic-Renault, N. Amperometric Enzyme Biosensors: Past, Present and Future. IRBM 2008, 29, 171–180. [Google Scholar] [CrossRef]
- Chaubey, A.; Malhotra, B.D. Mediated Biosensors. Biosens. Bioelectron. 2002, 17, 441–456. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zhang, R.; Sun, N.; Li, X.; Wang, J.; Cao, Y.; Pei, R. Near-Infrared Light-Driven Photoelectrochemical Aptasensor Based on the Upconversion Nanoparticles and TiO2/CdTe Heterostructure for Detection of Cancer Cells. ACS Appl. Mater. Interfaces 2016, 8, 25834–25839. [Google Scholar] [CrossRef] [PubMed]
- Mavrič, T.; Benčina, M.; Imani, R.; Junkar, I.; Valant, M.; Kralj-Iglič, V.; Iglič, A. Electrochemical Biosensor Based on TiO2 Nanomaterials for Cancer Diagnostics. Adv. Biomembr. Lipid Self-Assem. 2018, 27, 63–105. [Google Scholar] [CrossRef]
- Mostufa, S.; Akib, T.B.A.; Rana, M.M.; Islam, M.R. Highly Sensitive TiO2/Au/Graphene Layer-Based Surface Plasmon Resonance Biosensor for Cancer Detection. Biosensors 2022, 12, 603. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Wang, X.; Chen, S. Ho2O3-TiO2 Nanobelts Electrode for Highly Selective and Sensitive Detection of Cancer MiRNAs. Biosensors 2022, 12, 800. [Google Scholar] [CrossRef]
- Khaliq, N.; Rasheed, M.A.; Khan, M.; Maqbool, M.; Ahmad, M.; Karim, S.; Nisar, A.; Schmuki, P.; Cho, S.O.; Ali, G. Voltage-Switchable Biosensor with Gold Nanoparticles on TiO2 Nanotubes Decorated with CdS Quantum Dots for the Detection of Cholesterol and H2O2. ACS Appl. Mater. Interfaces 2021, 13, 3653–3668. [Google Scholar] [CrossRef]
- Eksin, E.; Erdem, A. Recent Progress on Optical Biosensors Developed for Nucleic Acid Detection Related to Infectious Viral Diseases. Micromachines 2023, 14, 295. [Google Scholar] [CrossRef]
- Vadlamani, B.S.; Uppal, T.; Verma, S.C.; Misra, M. Functionalized TiO2 Nanotube-Based Electrochemical Biosensor for Rapid Detection of SARS-CoV-2. Sensors 2020, 20, 5871. [Google Scholar] [CrossRef]
- Sakib, S.; Pandey, R.; Soleymani, L.; Zhitomirsky, I. Surface Modification of TiO2 for Photoelectrochemical DNA Biosensors. Med. Devices Sens. 2020, 3, e10066. [Google Scholar] [CrossRef]
- Yoo, J.; Jeong, H.; Park, S.K.; Park, S.; Lee, J.S. Interdigitated Electrode Biosensor Based on Plasma-Deposited TiO2 Nanoparticles for Detecting DNA. Biosensors 2021, 11, 212. [Google Scholar] [CrossRef]
- Song, K.; Lin, J.; Zhuang, Y.; Han, Z.; Chen, J. Construction of Photoelectrochemical DNA Biosensors Based on TiO2@Carbon Dots@Black Phosphorous Quantum Dots. Micromachines 2021, 12, 1523. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Sun, N.; Wang, S.; Xu, M.; Zuo, C.; Xu, X.; Li, Z.; Sun, Q.; Wang, Y.; Liu, P.; et al. A Label-Free Electrochemiluminescence Sensing for Detection of Dopamine Based on TiO2 Electrospun Nanofibers. Electroanalysis 2022, 34, 122–129. [Google Scholar] [CrossRef]
- Wang, Y.; Yin, L.; Wu, J.; Li, N.; He, N.; Zhao, H.; Wu, Q.; Li, X. Perovskite-SrTiO3/TiO2/PDA as Photoelectrochemical Glucose Biosensor. Ceram. Int. 2021, 47, 29807–29814. [Google Scholar] [CrossRef]
- Sun, Y.; Li, P.; Zhu, Y.; Zhu, X.; Zhang, Y.; Liu, M.; Liu, Y. In Situ Growth of TiO2 Nanowires on Ti3C2 MXenes Nanosheets as Highly Sensitive Luminol Electrochemiluminescent Nanoplatform for Glucose Detection in Fruits, Sweat and Serum Samples. Biosens. Bioelectron. 2021, 194, 113600. [Google Scholar] [CrossRef] [PubMed]
- Kadian, S.; Arya, B.D.; Kumar, S.; Sharma, S.N.; Chauhan, R.P.; Srivastava, A.; Chandra, P.; Singh, S.P. Synthesis and Application of PHT-TiO2 Nanohybrid for Amperometric Glucose Detection in Human Saliva Sample. Electroanalysis 2018, 30, 2793–2802. [Google Scholar] [CrossRef]
- Ognjanović, M.; Stanković, V.; Knežević, S.; Antić, B.; Vranješ-Djurić, S.; Stanković, D.M. TiO2/APTES Cross-Linked to Carboxylic Graphene Based Impedimetric Glucose Biosensor. Microchem. J. 2020, 158, 105150. [Google Scholar] [CrossRef]
- Kumar, N.; Chauhan, N.S.; Mittal, A.; Sharma, S. TiO2 and Its Composites as Promising Biomaterials: A Review. BioMetals 2018, 31, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Farajpour, N.; Deivanayagam, R.; Phakatkar, A.; Narayanan, S.; Shahbazian-Yassar, R.; Shokuhfar, T. A Novel Antimicrobial Electrochemical Glucose Biosensor Based on Silver–Prussian Blue-modified TiO2 Nanotube Arrays. Med. Devices Sens. 2020, 3, e10061. [Google Scholar] [CrossRef]
- Yang, W.; Xu, W.; Wang, Y.; Chen, D.; Wang, X.; Cao, Y.; Wu, Q.; Tu, J.; Zhen, C. Photoelectrochemical Glucose Biosensor Based on the Heterogeneous Facets of Nanocrystalline TiO2/Au/Glucose Oxidase Films. ACS Appl. Nano Mater. 2020, 3, 2723–2732. [Google Scholar] [CrossRef]
- Akhbari Varkani, R.; Rafiee-Pour, H.A.; Noormohammadi, M. One Step Immobilization of Glucose Oxidase on TiO2 Nanotubes towards Glucose Biosensing. Microchem. J. 2021, 170, 106712. [Google Scholar] [CrossRef]
- Wu, B.; Cheng, Z.; Hou, Y.; Chen, Q.; Wang, X.; Qiao, B.; Chen, D.; Tu, J. Engineering Exposed Vertical Nano- TiO2(001) Facets/BiOI Nanosheet Heterojunction Film for Constructing a Satisfactory PEC Glucose Oxidase Biosensor. RSC Adv. 2022, 12, 19495–19504. [Google Scholar] [CrossRef]
- Hussain, M.; Khaliq, N.; Khan, A.A.; Khan, M.; Ali, G.; Maqbool, M. Synthesis, Characterization and Electrochemical Analysis of TiO2 Nanostructures for Sensing L-Cysteine and Hydrogen Peroxide. Phys. E Low-Dimens. Syst. Nanostruct. 2021, 128, 114541. [Google Scholar] [CrossRef]
- Saeed, A.A.; Abbas, M.N.; El-Hawary, W.F.; Issa, Y.M.; Singh, B. A Core–Shell Au@TiO2 and Multi-Walled Carbon Nanotube-Based Sensor for the Electroanalytical Determination of H2O2 in Human Blood Serum and Saliva. Biosensors 2022, 12, 778. [Google Scholar] [CrossRef] [PubMed]
- Çakıroğlu, B.; Demirci, Y.C.; Gökgöz, E.; Özacar, M. A Photoelectrochemical Glucose and Lactose Biosensor Consisting of Gold Nanoparticles, MnO2 and g-C3N4 Decorated TiO2. Sens. Actuators B Chem. 2019, 282, 282–289. [Google Scholar] [CrossRef]
- Gunatilake, U.B.; Garcia-Rey, S.; Ojeda, E.; Basabe-Desmonts, L.; Benito-Lopez, F. TiO2 Nanotubes Alginate Hydrogel Scaffold for Rapid Sensing of Sweat Biomarkers: Lactate and Glucose. ACS Appl. Mater. Interfaces 2021, 13, 37734–37745. [Google Scholar] [CrossRef] [PubMed]
- Umar, A.; Haque, M.; Ansari, S.G.; Seo, H.K.; Ibrahim, A.A.; Alhamami, M.A.M.; Algadi, H.; Ansari, Z.A. Label-Free Myoglobin Biosensor Based on Pure and Copper-Doped Titanium Dioxide Nanomaterials. Biosensors 2022, 12, 1151. [Google Scholar] [CrossRef]
- Huang, L.; Liang, Z.; Zhang, F.; Luo, H.; Liang, R.; Han, F.; Wu, Z.; Han, D.; Shen, J.; Niu, L. Upconversion NaYF4:Yb/Er-TiO2-Ti3C2 Heterostructure-Based Near-Infrared Light-Driven Photoelectrochemical Biosensor for Highly Sensitive and Selective d-Serine Detection. Anal. Chem. 2022, 94, 16246–16253. [Google Scholar] [CrossRef]
- Jiang, M.; Chen, J.S. A Lable-Free ECL Biosensor for the Detection of Uric Acid Based on Au NRs at TiO2 Nanocomposite. Int. J. Electrochem. Sci. 2019, 14, 2333–2344. [Google Scholar] [CrossRef]
- Parthasarathy, P.; Vivekanandan, S. Biocompatible TiO2-CeO2 Nano-Composite Synthesis, Characterization and Analysis on Electrochemical Performance for Uric Acid Determination. Ain Shams Eng. J. 2020, 11, 777–785. [Google Scholar] [CrossRef]
- Ramalingam, M.; Jaisankar, A.; Cheng, L.; Krishnan, S.; Lan, L.; Hassan, A.; Sasmazel, H.T.; Kaji, H.; Deigner, H.P.; Pedraz, J.L.; et al. Impact of Nanotechnology on Conventional and Artificial Intelligence-Based Biosensing Strategies for the Detection of Viruses. Discov. Nano 2023, 18, 58. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Rong, J.; Zhan, Z.; Yu, X. Glucose Biosensor Based on Glucose Oxidase Immobilized on Multi-Vacancy TiO2 Nanotube Arrays. Int. J. Electrochem. Sci. 2019, 14, 9661–9670. [Google Scholar] [CrossRef]
- Jafari, S.; Mahyad, B.; Hashemzadeh, H.; Janfaza, S.; Gholikhani, T.; Tayebi, L. Biomedical Applications of TiO2 Nanostructures: Recent Advances. Int. J. Nanomed. 2020, 15, 3447–3470. [Google Scholar] [CrossRef] [PubMed]
- Yasri, S.; Wiwanitkit, V. Sustainable Materials and COVID-19 Detection Biosensor: A Brief Review. Sens. Int. 2022, 3, 100171. [Google Scholar] [CrossRef] [PubMed]
- Kanwar, V.S.; Sharma, A.; Rinku; Kanwar, M.; Srivastav, A.L.; Soni, D.K. An Overview for Biomedical Waste Management during Pandemic like COVID-19. Int. J. Environ. Sci. Technol. 2023, 20, 8025–8040. [Google Scholar] [CrossRef] [PubMed]
- Hashemzadeh, H.; Javadi, H.; Darvishi, M.H. Study of Structural Stability and Formation Mechanisms in DSPC and DPSM Liposomes: A Coarse-Grained Molecular Dynamics Simulation. Sci. Rep. 2020, 10, 1837. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, X.; Lu, X.; Tian, H.; Jiang, H.; Lv, G.; Guo, D.; Wu, C.; Chen, B. The Incorporation of Daunorubicin in Cancer Cells through the Use of Titanium Dioxide Whiskers. Biomaterials 2009, 30, 4708–4715. [Google Scholar] [CrossRef]
- Kim, C.; Kim, S.; Oh, W.; Choi, M.; Jang, J. Efficient Intracellular Delivery of Camptothecin by Silica/Titania Hollow Nanoparticles. Chem. A Eur. J. 2012, 18, 4902–4908. [Google Scholar] [CrossRef]
- Chen, J.-L.; Zhang, H.; Huang, X.-Q.; Wan, H.-Y.; Li, J.; Fan, X.-X.; Luo, K.Q.; Wang, J.; Zhu, X.-M.; Wang, J. Antiangiogenesis-Combined Photothermal Therapy in the Second Near-Infrared Window at Laser Powers below the Skin Tolerance Threshold. Nano-Micro Lett. 2019, 11, 93. [Google Scholar] [CrossRef]
- Mushtaq, A.; Hou, Y.; Tian, C.; Deng, T.; Xu, C.; Sun, Z.; Kong, X.; Zubair Iqbal, M. Facile Synthesis of Mn Doped TiO2 Rhombic Nanocomposites for Enhanced T1-Magnetic Resonance Imaging and Photodynamic Therapy. Mater. Res. Bull. 2021, 144, 111481. [Google Scholar] [CrossRef]
- Fei Yin, Z.; Wu, L.; Gui Yang, H.; Hua Su, Y. Recent Progress in Biomedical Applications of Titanium Dioxide. Phys. Chem. Chem. Phys. 2013, 15, 4844. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jeong, J.-S.; Kim, S.Y.; Park, M.-K.; Choi, S.-D.; Kim, U.-J.; Park, K.; Jeong, E.J.; Nam, S.-Y.; Yu, W.-J. Titanium Dioxide Nanoparticles Oral Exposure to Pregnant Rats and Its Distribution. Part. Fibre Toxicol. 2019, 16, 31. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Sui, B.; Sun, J. Size- and Shape-Dependent Effects of Titanium Dioxide Nanoparticles on the Permeabilization of the Blood–Brain Barrier. J. Mater. Chem. B 2017, 5, 9558–9570. [Google Scholar] [CrossRef] [PubMed]
- Abdulnasser Harfoush, S.; Hannig, M.; Le, D.D.; Heck, S.; Leitner, M.; Omlor, A.J.; Tavernaro, I.; Kraegeloh, A.; Kautenburger, R.; Kickelbick, G.; et al. High-Dose Intranasal Application of Titanium Dioxide Nanoparticles Induces the Systemic Uptakes and Allergic Airway Inflammation in Asthmatic Mice. Respir. Res. 2020, 21, 168. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.-O.; Lee, S.-J.; Kim, W.-I.; Pak, S.-W.; Moon, C.; Shin, I.-S.; Heo, J.-D.; Ko, J.-W.; Kim, J.-C. Titanium Dioxide Nanoparticles Exacerbate Allergic Airway Inflammation via TXNIP Upregulation in a Mouse Model of Asthma. Int. J. Mol. Sci. 2021, 22, 9924. [Google Scholar] [CrossRef] [PubMed]
- Cui, F.; Zhou, Z.; Zhou, H.S. Review—Measurement and Analysis of Cancer Biomarkers Based on Electrochemical Biosensors. J. Electrochem. Soc. 2020, 167, 037525. [Google Scholar] [CrossRef]
- Liu, P.; Huo, X.; Tang, Y.; Xu, J.; Liu, X.; Wong, D.K.Y. A TiO2 Nanosheet-g-C3N4 Composite Photoelectrochemical Enzyme Biosensor Excitable by Visible Irradiation. Anal. Chim. Acta 2017, 984, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, A.; Khurshid, Z.; Khan, M.T.; Mansoor, E.; Butt, F.A.; Jamal, A.; Palma, P.J. Medical and Dental Applications of Titania Nanoparticles: An Overview. Nanomaterials 2022, 12, 3670. [Google Scholar] [CrossRef]
- Azzawi, Z.G.M.; Hamad, T.I.; Kadhim, S.A.; Naji, G.A.-H. Osseointegration Evaluation of Laser-Deposited Titanium Dioxide Nanoparticles on Commercially Pure Titanium Dental Implants. J. Mater. Sci. Mater. Med. 2018, 29, 96. [Google Scholar] [CrossRef]
- Souza, J.C.M.; Sordi, M.B.; Kanazawa, M.; Ravindran, S.; Henriques, B.; Silva, F.S.; Aparicio, C.; Cooper, L.F. Nano-Scale Modification of Titanium Implant Surfaces to Enhance Osseointegration. Acta Biomater. 2019, 94, 112–131. [Google Scholar] [CrossRef]
- Chen, W.; Shen, X.; Hu, Y.; Xu, K.; Ran, Q.; Yu, Y.; Dai, L.; Yuan, Z.; Huang, L.; Shen, T.; et al. Surface Functionalization of Titanium Implants with Chitosan-Catechol Conjugate for Suppression of ROS-Induced Cells Damage and Improvement of Osteogenesis. Biomaterials 2017, 114, 82–96. [Google Scholar] [CrossRef]
- Ueno, T.; Ikeda, T.; Tsukimura, N.; Ishijima, M.; Minamikawa, H.; Sugita, Y.; Yamada, M.; Wakabayashi, N.; Ogawa, T. Novel Antioxidant Capability of Titanium Induced by UV Light Treatment. Biomaterials 2016, 108, 177–186. [Google Scholar] [CrossRef]
- Yu, Y.; Shen, X.; Luo, Z.; Hu, Y.; Li, M.; Ma, P.; Ran, Q.; Dai, L.; He, Y.; Cai, K. Osteogenesis Potential of Different Titania Nanotubes in Oxidative Stress Microenvironment. Biomaterials 2018, 167, 44–57. [Google Scholar] [CrossRef]
- Yu, W.; Jiang, X.; Zhang, F.; Xu, L. The Effect of Anatase TiO2 Nanotube Layers on MC3T3-E1 Preosteoblast Adhesion, Proliferation, and Differentiation. J. Biomed. Mater. Res. Part A 2010, 94A, 1012–1022. [Google Scholar] [CrossRef]


| No. | Year | Materials | Application | Main Results | Refs. |
|---|---|---|---|---|---|
| 1 | 2018 | Pristine TiO2, metal-assisted TiO2, organic–TiO2 composites, carbon–TiO2 composites, and MOX–TiO2 composites. | H2 gas sensor | The enhancement of the gas-sensing performance of TiO2-based materials through the synthesis route (doping, surface modification, and nano-fabrication) in terms of its effects on the properties and surface reaction mechanisms as a hydrogen sensor. | [5] |
| 2 | 2019 | TiO2 NPs | Self-cleaning, self-sterilizing, air purification, water disinfection, and antitumor activity | Explanation of the photocatalytic mechanism of TiO2 materials and their applicability in different fields; special attention was given to TiO2 NPs with antibacterial and self-cleaning properties to develop transparent coatings (super-hydrophobic and super-hydrophilic coatings) for windows in outdoor applications. | [6] |
| 3 | 2020 | TiO2/Pd TiO2/PdO | Visible light photocatalyst | Photocatalytic degradation of rhodamine B under visible light irradiation. The TiO2/PdO photocatalyst exhibits higher photocatalytic activity compared with TiO2/Pd, which makes it suitable for pollutant removal in water and wastewater treatments. | [7] |
| 4 | 2020 | TiO2 thin films, doped or in composites, and nanotubes. | Metal oxide gas sensor for H2, CO, NO, O2, NO2, C3H4, C2H6O, C3H6O, NH3, and C₆H₅CH₃ | Effect of crystal structure, operating temperature, and doping with semiconductor oxides on the sensor properties of TiO2 thin films. | [8] |
| 5 | 2021 | Noble metals–TiO2 and carbon nanomaterials–TiO2 composites. | Water disinfection | Synthesis of TiO2 and TiO2-based composites as photocatalysts for water disinfection. The enhancement of the photocatalytic efficiency was achieved by adding noble metals and carbon materials to the TiO2 matrix. The impact of water properties on photocatalytic disinfection was also studied. Two important strategies to improve the photocatalytic efficiency of the materials were proposed: (1) the modification of the conduction band of the semiconductor to promote reactive species with longer lifetimes; (2) improving the bacteria and photocatalysts interactions. | [9] |
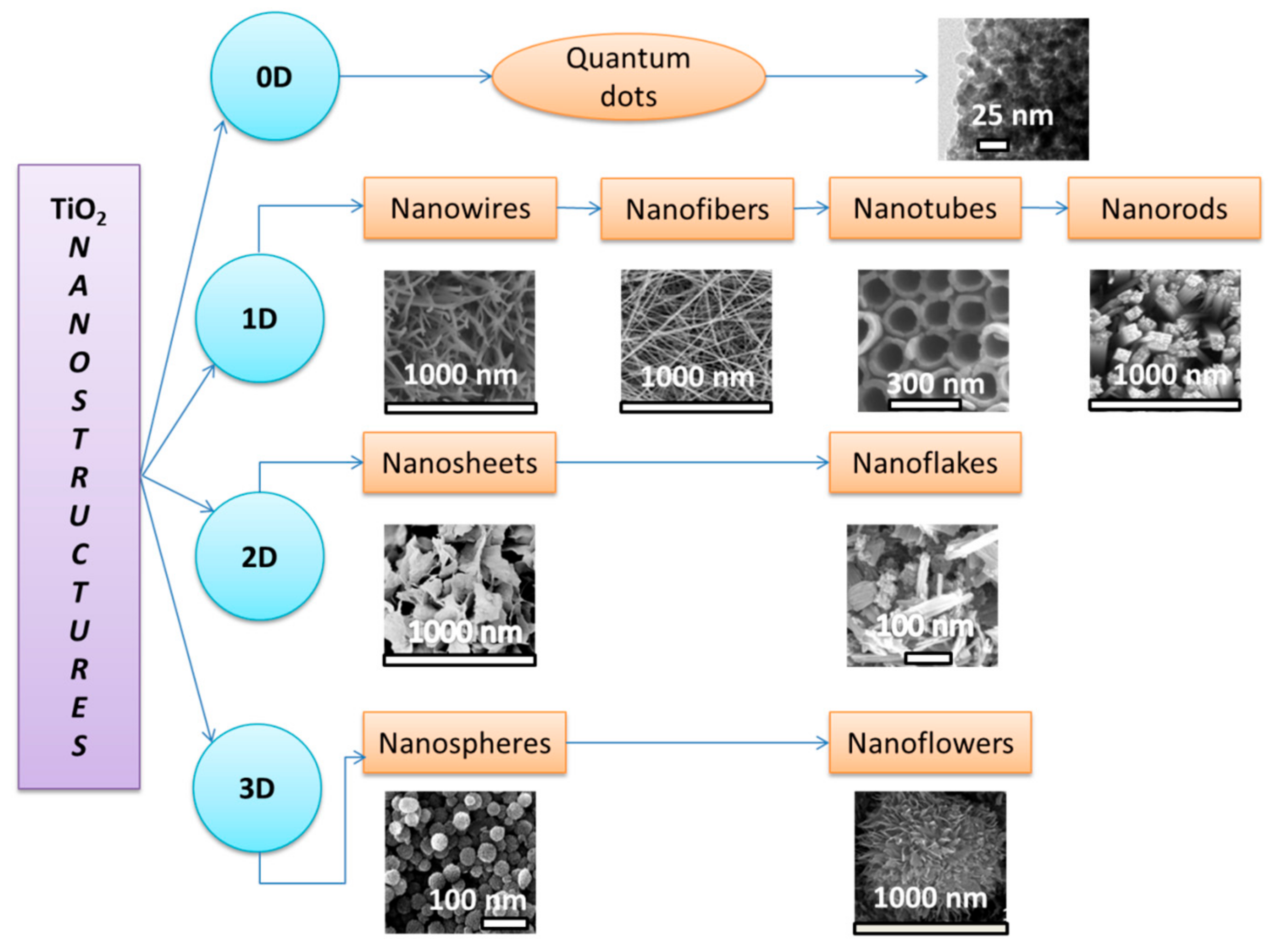
| 6 | 2022 | Hierarchical TiO2 nanostructure forms: 0D, 1D, 2D, and 3D. | Photocatalyst for oilfield-produced water (OPW) treatment | The possibility to enhance the photocatalytic activity for oilfield-produced water treatment (OPW) through the following pathways: energy band tailoring, obtaining and modifying TiO2 nanostructures by doping, and the development of photocatalytic membranes (PMs) based on TiO2. | [10] |
| 7 | 2022 | TiO2 nanohybrids (doping with metals, non-metals, co-doping, and graphene). | Food packaging | Food preservation and post-harvest loss mitigation applications. Nanotechnology has been explored as a scalable solution. | [1] |
| 8 | 2022 | Defective/reduced titanium dioxide (TiO2−x) and TiO2−x–carbon-based photocatalysts. | Photocatalytic CO2 reduction | Synthesis of TiO2−x with different morphologies and TiO2−x-based materials. This study presents the changes in TiO2 properties according to the amount of oxygen vacancies and their performance as photocatalysts in CO2 reduction. | [11] |
| 9 | 2022 | TiO2 composites; metal- and non-metal TiO2-based photocatalysts. | Photocatalytic CO2 reduction | Synthesis methods to prepare photocatalysts for CO2 reduction into green products. | [12] |
| 10 | 2022 | TiO2 and other nanomaterial-based antimicrobial additives. | Functional paints and coatings | The incorporation of TiO2 nanoparticles in paint and coatings due to their interesting properties being used as additives with antibacterial properties, as inorganic binders to prevent organic binder photodegradation. | [13] |
| 11 | 2023 | Oxygen-deficient titanium oxide films with an average composition of TiO2−δ. | Photocatalytic hydrogen production by water splitting | Explanation of the mechanism of photocatalytic hydrogen production by water splitting over TiO2. The introduction of oxygen defects into TiO2 through ion doping, the deposition of noble metals, and dye sensitization to improve the photocatalytic activity of TiO2-based materials. | [14] |
| Year | Material | Method | Substrate | Gas Detected | Main Results | Refs. |
|---|---|---|---|---|---|---|
| 2018 | Soot template TiO2 fractals | Chemical vapor deposition (CVD) | Deposited soot template (a layer of candle soot was deposited on Ti/Pt electrodes) | Acetone vapour | Novel structural design/diabetic concentrations in the breath. | [134] |
| 2018 | TiO2 films | Atomic layer deposition (ALD) | Alumina sensors and microscope slides | NH3 and CH4 | Sensitivity toward NH3 varied with thickness. | [135] |
| 2019 | TiO2 NP | Bar coating | Alumina | H2S | Works at RT under UV. | [136] |
| 2019 | MgO:TiO2 thin films | Co-sputtering (confocal sputtering) | Pt/SiO2/Si | CH4 | All the results showed that the dopant can improve the electrical performance and sensor properties. | [137] |
| 2019 | Nanostructured TiO2 | Molecular layering (ML) | Al2O3 substrates with platinum electrodes | O2 | ML-deposited TiO2 film shows good selectivity to oxygen. | [138] |
| 2021 | TiO2 thin film | Pulsed laser deposition (PLD) | -(100) silicon (Si/SiO2) -(100) SrTiO3 (STO) -polycrystalline Al2O3 | H2S | The surface of PLD TiO2 film on Si/SiO2 has a higher roughness than that on the STO substrate. | [139] |
| Composite heterostructure | ||||||
| 2018 | p-copper oxide thin film/n-TiO2Nts heterostructure | Anodization/ oxidation | Ti foil | H2, ethanol, acetone, chloroform, and NO2 | The improvement in the sensing properties is attributed to the heterojunction between the CuO thin film and the TiO2 NTs. | [140] |
| 2018 | TiO2 + SnO2 NPs | SG+ | Glass | NH3 | The improvement in the sensor performance is due to the increased active surface area and the efficient electron–hole charge separation and transfer. | [141] |
| 2019 | Pd/Al2O3/TiO2 thin film heterostructure | Atomic layer deposition (ALD) | Quartz | H2 | Development of flexible gas sensors. | [142] |
| 2019 | TiO2/perovskite heterojunctions | Electrochemical method, Sol–gel | Ti foil | CO | The heterojunction is more sensitive than a single film and could operate at lower temperatures. | [143] |
| 2019 | TiO2 film/carboxyl PP film/MWCNTs | ALD/CCVD | Si/SiO2 | CO, H2, and NH3 | Better sensor response, lower detection limit, and lower operation temperature against H2 compared to the separate sensors. | [144] |
| 2019 | Polyaniline (PANI)/TiO2 core–shell nanofibers | In situ chemical polymerization on electrospun TiO2 nanofibers | Glass | NH3 | Under UV at RT at different humidity levels. | [145] |
| 2019 | TiO2/SnO2 and TiO2/CuO thin film nano-heterostructures | Reactive magnetron sputtering | Metallic target and a-SiO2 | NO2 | The response of TiO2/SnO2 to NO2 at 150 °C is double than in the case of pure SnO2. | [146] |
| 2019 | ZnO (n-type)-TiO2 (n-type)-PANI (p-type) Micro/nanoballs | Chemical deposition technique | Glass | LPG, NO2, acetone, NH3, and CO2 | Very good selectivity. | [130] |
| 2020 | TiO2 decorated with Au NPs | Irradiation with laser | Si and interdigital electrodes | Volatile organic compounds | Very selective and stable over time; works at RT. | [147] |
| 2022 | Pd doped CoTiO3/TiO2 (Pd-CTT) | HT | Interdigital electrodes | Benzene | Works at RT; good resistance to humidity interference. | [148] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gartner, M.; Szekeres, A.; Stroescu, H.; Mitrea, D.; Covei, M. Advanced Nanostructured Coatings Based on Doped TiO2 for Various Applications. Molecules 2023, 28, 7828. https://doi.org/10.3390/molecules28237828
Gartner M, Szekeres A, Stroescu H, Mitrea D, Covei M. Advanced Nanostructured Coatings Based on Doped TiO2 for Various Applications. Molecules. 2023; 28(23):7828. https://doi.org/10.3390/molecules28237828
Chicago/Turabian StyleGartner, Mariuca, Anna Szekeres, Hermine Stroescu, Daiana Mitrea, and Maria Covei. 2023. "Advanced Nanostructured Coatings Based on Doped TiO2 for Various Applications" Molecules 28, no. 23: 7828. https://doi.org/10.3390/molecules28237828
APA StyleGartner, M., Szekeres, A., Stroescu, H., Mitrea, D., & Covei, M. (2023). Advanced Nanostructured Coatings Based on Doped TiO2 for Various Applications. Molecules, 28(23), 7828. https://doi.org/10.3390/molecules28237828