Controlled Decoration of [60]Fullerene with Polymannan Analogues and Amino Acid Derivatives through Malondiamide-Based Linkers
Abstract
1. Introduction
2. Results
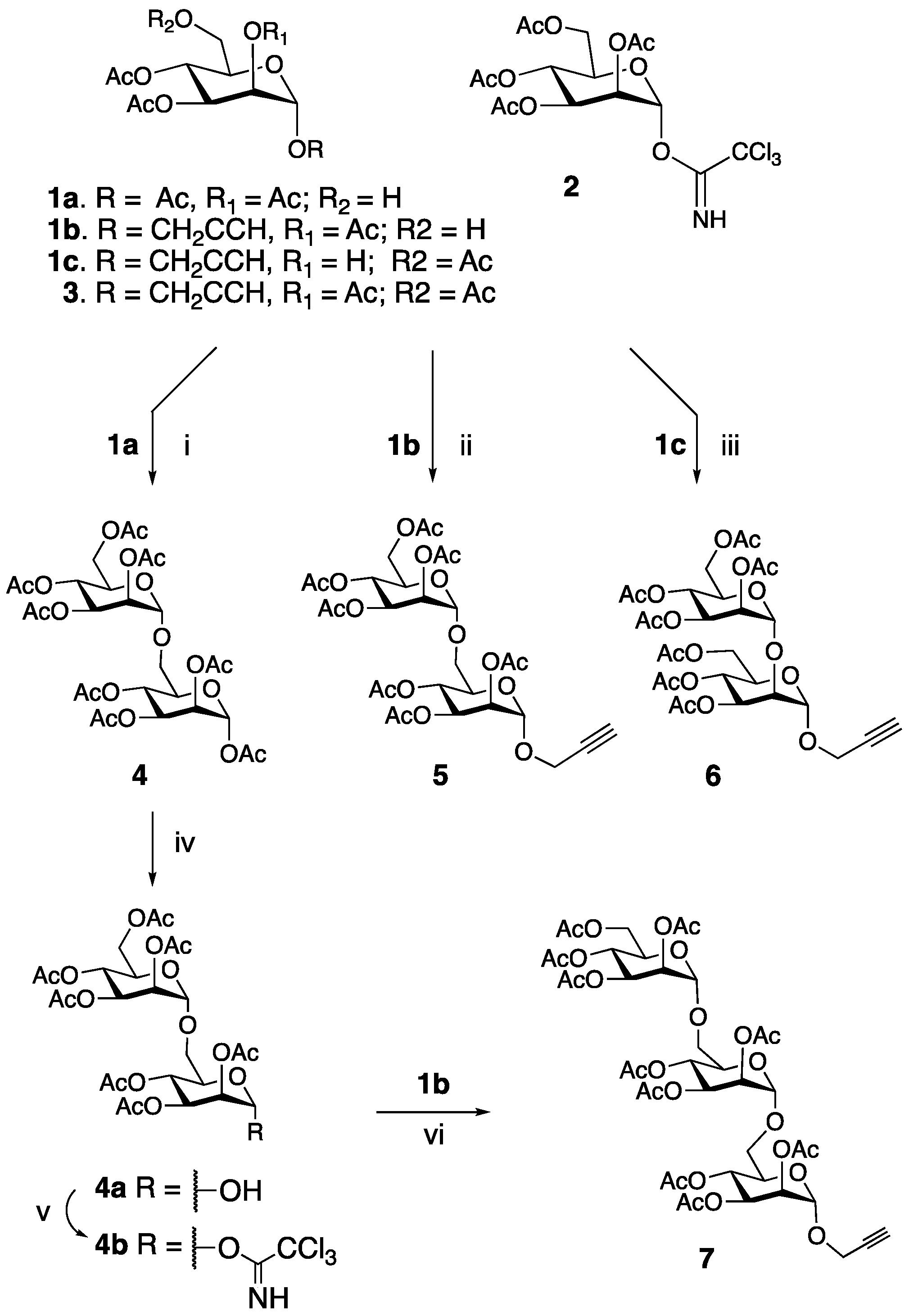
2.1. Synthesis of Propargyl-Mannose Glycans
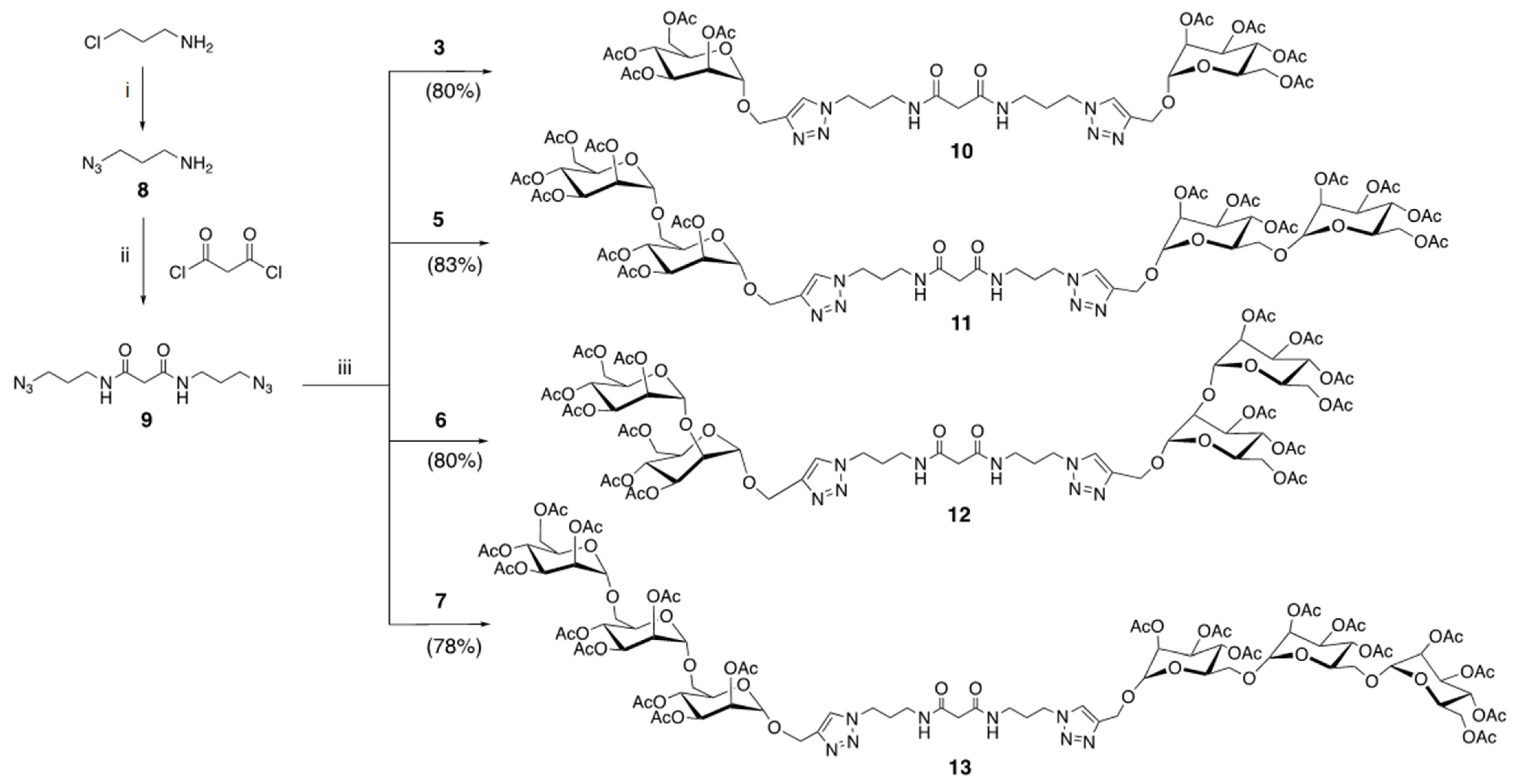
2.2. Preparation of Glycosylated C60 Derivatives
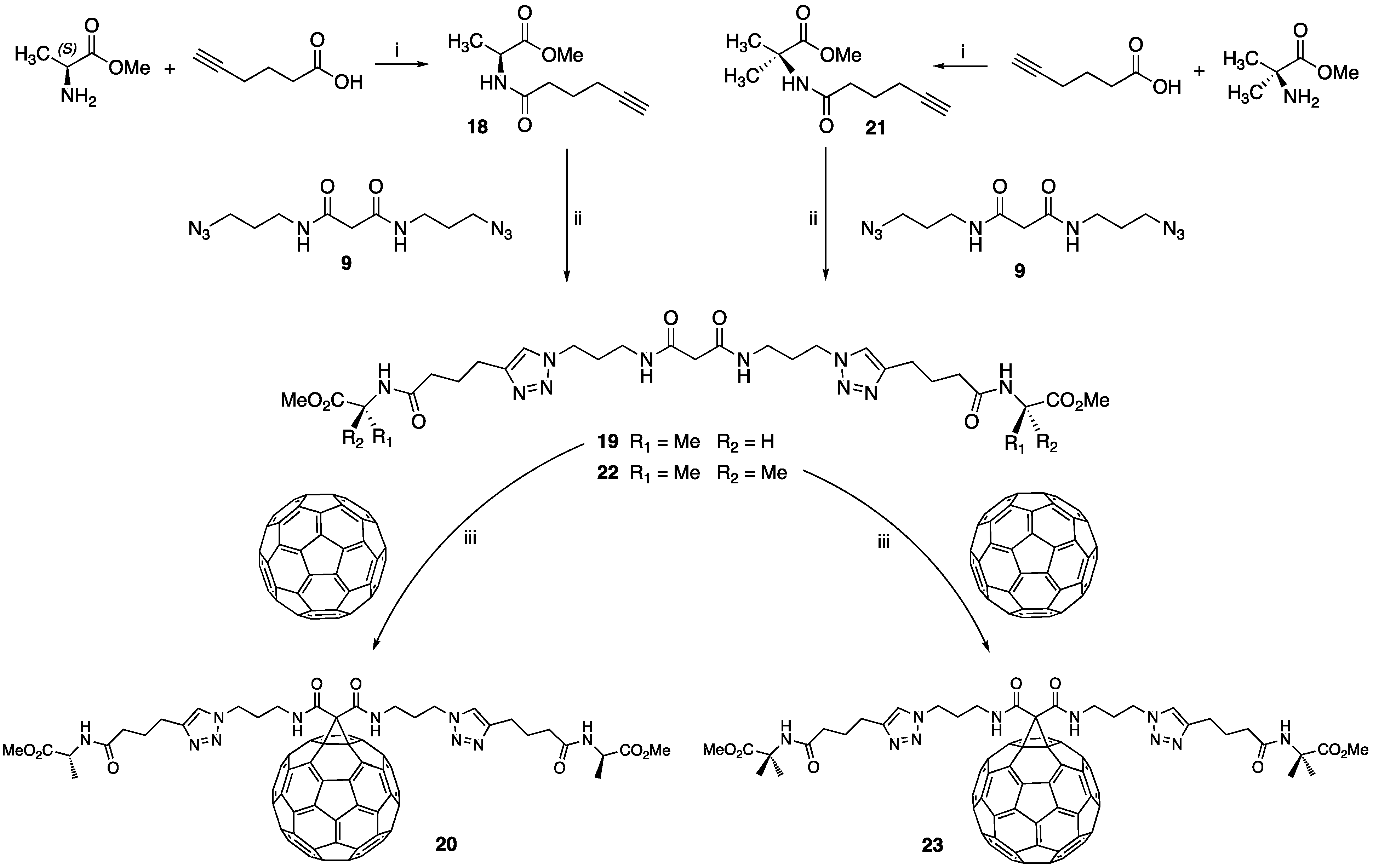
2.3. Conjugation of C60 with Amino Acid Derivatives
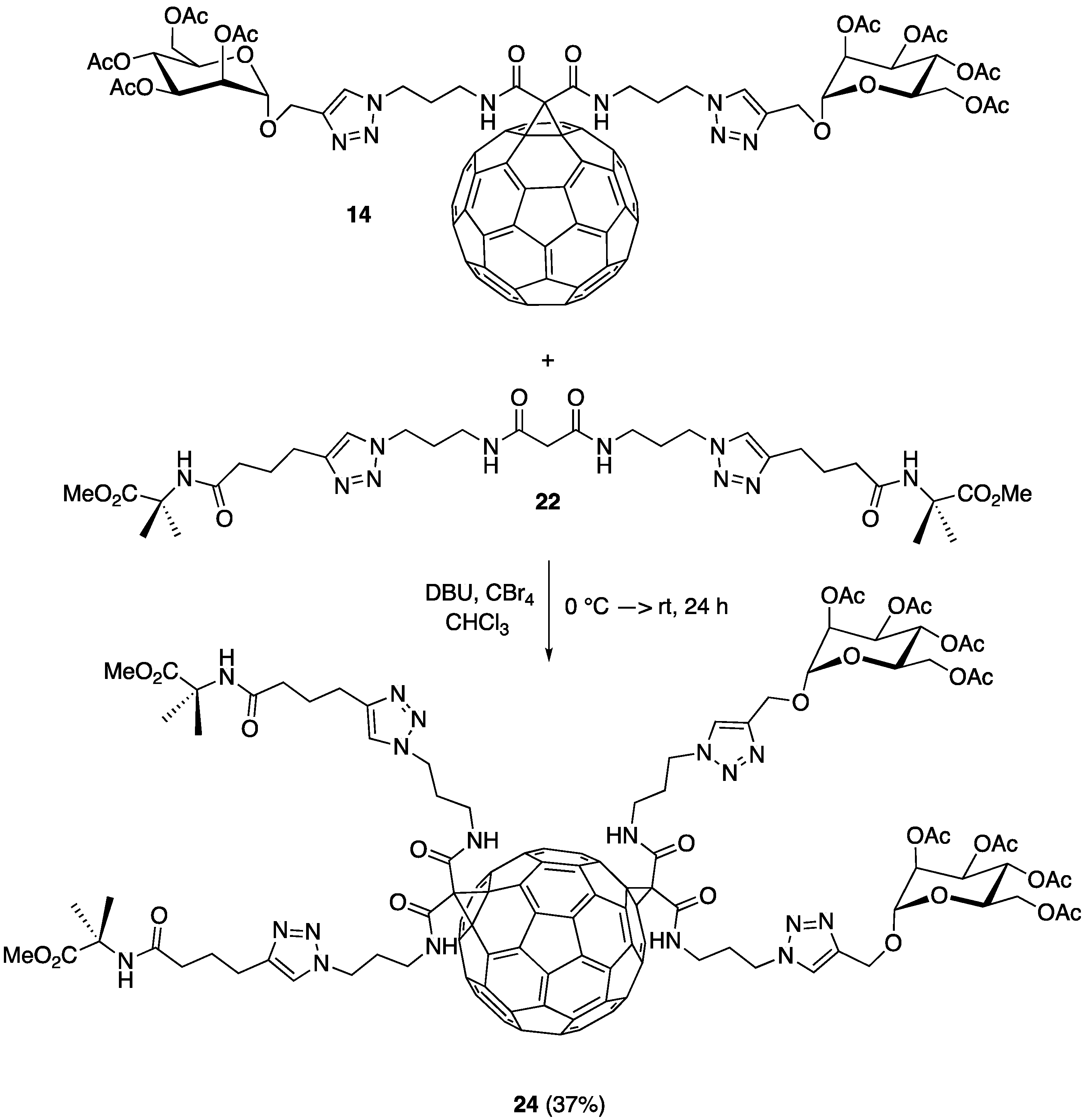
2.4. Double Conjugation of C60: Sugar and Amino Acid Derivative
3. Conclusions
4. Materials and Methods
4.1. Sugar Synthesis
- Propargyl 2″,3″,4″,6″-tetra-O-acetyl-α-d-mannopyranosyl-(1→6)-2′,3′,4′-tri-O-acetyl-α-d-mannopyranosyl-(1→6)-2,3,4-tri-O-acetyl-α-d-mannopyranoside (7)
4.2. Functionalization of C60 with Sugar Derivatives
- 3-azidopropan-1-amine (8) [54]
- N1,N3-bis(3-azidopropyl)malondiamide (9)
- Man malondiamide chain (10)
- Man(α1,6)man malondiamide chain (11)
- Man(α1,2)man malondiamide chain (12)
- Man(α1,6)man(α1,6)man malondiamide chain (13)
- Fullerene-man (14)
- Fullerene-man(α1,6)man (15)
- Fullerene-man(α1,2)man (16)
- Fullerene-man(α1,6)man(α1,6)man (17)
4.3. Functionalization of C60 with Amino Acid Derivatives
- Methyl hex-5-ynoyl-l-alaninate (18)
- Dimethyl 2,2′-((4,4′-(((malonylbis(azanediyl))bis(propane-3,1-diyl))bis(1H-1,2,3-triazole-1,4-diyl))bis(butanoyl))bis(azanediyl))dipropionate (19)
- Fullerene-l-alanine (20)
- Methyl 2-(hex-5-ynamido)-2-methylpropanoate (21)
- Dimethyl 2,2′-((4,4′-(((malonylbis(azanediyl))bis(propane-3,1-diyl))bis(1H-1,2,3-triazole-1,4-diyl))bis(butanoyl))bis(azanediyl))bis(2-methylpropanoate) (22)
- Fullerene–Aib (23)
4.4. Double Functionalization of C60
- Fullerene–man–Aib (24)
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Tanzi, L.; Terreni, M.; Zhang, Y. Synthesis and biological application of glyco- and peptide derivatives of fullerene C60. Eur. J. Med. Chem. 2022, 230, 114104. [Google Scholar] [CrossRef] [PubMed]
- Riley, P.R.; Narayan, R.J. Recent advances in carbon nanomaterials for biomedical applications: A review. Curr. Opin. Biomed. Eng. 2021, 17, 100262. [Google Scholar] [CrossRef] [PubMed]
- Sharoyko, V.V.; Ageev, S.V.; Podolsky, N.E.; Petrov, A.V.; Litasova, E.V.; Vlasov, T.D.; Vasina, L.V.; Murin, I.V.; Piotrovskiy, L.B.; Semenov, K.N. Biologically active water-soluble fullerene adducts: Das Glasperlenspiel (by H. Hesse)? J. Mol. Liq. 2021, 323, 114990. [Google Scholar] [CrossRef]
- Zhou, S. Sugar-Functionalized Fullerenes. Curr. Org. Chem. 2016, 20, 1490–1501. [Google Scholar] [CrossRef]
- Cecioni, S.; Oerthel, V.; Iehl, J.; Holler, M.; Goyard, D.; Praly, J.P.; Imberty, A.; Nierengarten, J.F.; Vidal, S. Synthesis of dodecavalent fullerene-based glycoclusters and evaluation of their binding properties towards a bacterial lectin. Chemistry—A Eur. J. 2011, 17, 3252–3261. [Google Scholar] [CrossRef]
- Ramos-Soriano, J.; Reina, J.J.; Illescas, B.M.; Rojo, J.; Martín, N. Maleimide and Cyclooctyne-Based Hexakis-Adducts of Fullerene: Multivalent Scaffolds for Copper-Free Click Chemistry on Fullerenes. J. Org. Chem. 2018, 83, 1727–1736. [Google Scholar] [CrossRef]
- Enes, R.F.; Tomé, A.C.; Cavaleiro, J.A.S.; El-Agamey, A.; McGarvey, D.J. Synthesis and solvent dependence of the photophysical properties of [60]fullerene-sugar conjugates. Tetrahedron 2005, 61, 11873–11881. [Google Scholar] [CrossRef]
- Uhlmann, P.; Harth, E.; Naughton, A.B.; Vasella, A. Glycosylidene Carbenes. Part 20. Synthesis of deprotected, spiro-linked C-glycosides of C60. Helv. Chim. Acta 1994, 77, 2335–2340. [Google Scholar] [CrossRef]
- Zhai, W.Q.; Jiang, S.P.; Peng, R.F.; Jin, B.; Wang, G.W. Facile access to novel [60]fullerenyl diethers and [60]fullerene-sugar conjugates via annulation of diol moieties. Org. Lett. 2015, 17, 1862–1865. [Google Scholar] [CrossRef]
- Yashiro, A.; Nishida, Y.; Ohno, M.; Eguchi, S.; Kobayashi, K. Fullerene glycoconjugates: A general synthetic approach via cycloaddition of per-O-acetyl glycosyl azides to [60]fullerene. Tetrahedron Lett. 1998, 39, 9031–9034. [Google Scholar] [CrossRef]
- Serda, M.; Ware, M.J.; Newton, J.M.; Sachdeva, S.; Krzykawska-Serda, M.; Nguyen, L.; Law, J.; Anderson, A.O.; Curley, S.A.; Wilson, L.J.; et al. Development of photoactive Sweet-C 60 for pancreatic cancer stellate cell therapy. Nanomedicine 2018, 13, 2981–2993. [Google Scholar] [CrossRef] [PubMed]
- Serda, M.; Szewczyk, G.; Krzysztyńska-Kuleta, O.; Korzuch, J.; Dulski, M.; Musioł, R.; Sarna, T. Developing [60]Fullerene Nanomaterials for Better Photodynamic Treatment of Non-Melanoma Skin Cancers. ACS Biomater. Sci. Eng. 2020, 6, 5930–5940. [Google Scholar] [CrossRef] [PubMed]
- Barańska, E.; Wiecheć-Cudak, O.; Rak, M.; Bienia, A.; Mrozek-Wilczkiewicz, A.; Krzykawska-Serda, M.; Serda, M. Interactions of a water-soluble glycofullerene with glucose transporter 1. Analysis of the cellular effects on a pancreatic tumor model. Nanomaterials 2021, 11, 513. [Google Scholar] [CrossRef] [PubMed]
- Strom, T.A.; Durdagi, S.; Ersoz, S.S.; Salmas, R.E.; Supuran, C.T.; Barron, A.R. Fullerene-based inhibitors of HIV-1 protease. J. Pept. Sci. 2015, 21, 862–870. [Google Scholar] [CrossRef]
- Tanimoto, S.; Takahashi, D.; Toshima, K. Chemical methods for degradation of target proteins using designed light-activatable organic molecules. Chem. Commun. 2012, 48, 7659–7671. [Google Scholar] [CrossRef]
- Tanimoto, S.; Sakai, S.; Kudo, E.; Okada, S.; Matsumura, S.; Takahashi, D.; Toshima, K. Target-selective photodegradation of HIV-1 protease and inhibition of HIV-1 replication in living cells by designed fullerene-sugar hybrids. Chemistry—Asian J. 2012, 7, 911–914. [Google Scholar] [CrossRef]
- Ishida, Y.; Tanimoto, S.; Takahashi, D.; Toshima, K. Photo-degradation of amyloid β by a designed fullerene-sugar hybrid. MedChemComm 2010, 1, 212–215. [Google Scholar] [CrossRef]
- Horie, M.; Fukuhara, A.; Saito, Y.; Yoshida, Y.; Sato, H.; Ohi, H.; Obata, M.; Mikata, Y.; Yano, S.; Niki, E. Antioxidant action of sugar-pendant C60 fullerenes. Bioorg. Med. Chem. Lett. 2009, 19, 5902–5904. [Google Scholar] [CrossRef]
- Muñoz, A.; Sigwalt, D.; Illescas, B.M.; Luczkowiak, J.; Rodríguez-Pérez, L.; Nierengarten, I.; Holler, M.; Remy, J.S.; Buffet, K.; Vincent, S.P.; et al. Synthesis of giant globular multivalent glycofullerenes as potent inhibitors in a model of Ebola virus infection. Nat. Chem. 2016, 8, 50–57. [Google Scholar] [CrossRef]
- Ramos-Soriano, J.; Rojo, J. Glycodendritic structures as DC-SIGN binders to inhibit viral infections. Chem. Commun. 2021, 57, 5111–5126. [Google Scholar] [CrossRef]
- Nierengarten, J.F.; Schneider, J.P.; Trinh, T.M.N.; Joosten, A.; Holler, M.; Lepage, M.L.; Bodlenner, A.; García-Moreno, M.I.; Ortiz Mellet, C.; Compain, P. Giant Glycosidase Inhibitors: First- and Second-Generation Fullerodendrimers with a Dense Iminosugar Shell. Chem.—A Eur. J. 2018, 24, 2483–2492. [Google Scholar] [CrossRef] [PubMed]
- Durka, M.; Buffet, K.; Iehl, J.; Holler, M.; Nierengarten, J.F.; Vincent, S.P. The inhibition of liposaccharide heptosyltransferase WaaC with multivalent glycosylated fullerenes: A new mode of glycosyltransferase inhibition. Chem.—A Eur. J. 2012, 18, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Jennepalli, S.; Pyne, S.G.; Keller, P.A. [60] Fullerenyl amino acids and peptides: A review of their synthesis and applications. RSC Adv. 2014, 4, 46383–46398. [Google Scholar] [CrossRef][Green Version]
- Burley, G.A.; Keller, P.A.; Pyne, S.G. [60]Fullerene Amino Acids and Related Derivatives. Fuller. Sci. Technol. 1999, 7, 973–1001. [Google Scholar] [CrossRef]
- Barron, A.R. [60]Fullerene-peptides: Bio-nano conjugates with structural and chemical diversity. J. Enzym. Inhib. Med. Chem. 2016, 31, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Kotelńikova, R.A.; Faingoĺd, I.I.; Poletaeva, D.A.; Mishchenko, D.V.; Romanova, V.S.; Shtoĺko, V.N.; Bogdanov, G.N.; Rybkin, A.Y.; Frog, E.S.; Smolina, A.V.; et al. Antioxidant properties of water-soluble amino acid derivatives of fullerenes and their role in the inhibition of herpes virus infection. Russ. Chem. Bull. 2011, 60, 1172–1176. [Google Scholar] [CrossRef]
- Friedman, S.H.; DeCamp, D.L.; Kenyon, G.L.; Sijbesma, R.P.; Srdanov, G.; Wudl, F. Inhibition of the HIV-1 Protease by Fullerene Derivatives: Model Building Studies and Experimental Verification. J. Am. Chem. Soc. 1993, 115, 6506–6509. [Google Scholar] [CrossRef]
- Mashino, T.; Shimotohno, K.; Ikegami, N.; Nishikawa, D.; Okuda, K.; Takahashi, K.; Nakamura, S.; Mochizuki, M. Human immunodeficiency virus-reverse transcriptase inhibition and hepatitis C virus RNA-dependent RNA polymerase inhibition activities of fullerene derivatives. Bioorg. Med. Chem. Lett. 2005, 15, 1107–1109. [Google Scholar] [CrossRef]
- Xie, L.; Luo, Y.; Lin, D.; Xi, W.; Yang, X.; Wei, G. The molecular mechanism of fullerene-inhibited aggregation of Alzheimer’s β-amyloid peptide fragment. Nanoscale 2014, 6, 9752–9762. [Google Scholar] [CrossRef]
- Sofou, P.; Elemes, Y.; Panou-Pomonis, E.; Stavrakoudis, A.; Tsikaris, V.; Sakarellos, C.; Sakarellos-Daitsiotis, M.; Maggini, M.; Formaggio, F.; Toniolo, C. Synthesis of a proline-rich [60]fullerene peptide with potential biological activity. Tetrahedron 2004, 60, 2823–2828. [Google Scholar] [CrossRef]
- Fillmore, H.L.; Shultz, M.D.; Henderson, S.C.; Cooper, P.; Broaddus, W.C.; Chen, Z.J.; Shu, C.Y.; Zhang, J.; Ge, J.; Dorn, H.C.; et al. Conjugation of functionalized gadolinium metallofullerenes with IL-13 peptides for targeting and imaging glial tumors. Nanomedicine 2011, 6, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Yang, D.; Lu, W.; Hu, X.; Hong, H.; Cai, T. Positron emission tomography (PET) guided glioblastoma targeting by a fullerene-based nanoplatform with fast renal clearance. Acta Biomater. 2017, 61, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F. Vaccine delivery systems. Hum. Vaccines Immunother. 2017, 13, 17–18. [Google Scholar] [CrossRef] [PubMed]
- Wendorf, J. Molecular Nanomedicine Towards Cancer. J. Pharm. Sci. 2006, 95, 2738–2750. [Google Scholar] [CrossRef]
- Liu, J.; Feng, X.; Chen, Z.; Yang, X.; Shen, Z.; Guo, M.; Deng, F.; Liu, Y.; Zhang, H.; Chen, C. The adjuvant effect of C60 (OH)22 nanoparticles promoting both humoral and cellular immune responses to HCV recombinant proteins. Mater. Sci. Eng. C 2019, 97, 753–759. [Google Scholar] [CrossRef]
- Masalova, V.; Shepelev, A.V.; Atanadze, S.N.; Parnes, Z.N.; Romanova, V.S.; Vo, O.M.; Yu, A.; Kushch, A.A. Immunostimulation Effect of Water-Soluble Fullerene Derivatives; Potential Adjuvants for the New Generation of Vaccines. Doklandy Biochem. 1999, 369, 180–182. [Google Scholar]
- Tan, M.C.A.A.; Mommaas, A.M.; Drijfhout, J.W.; Jordens, R.; Onderwater, J.J.M.; Verwoerd, D.; Mulder, A.A.; Van Der Heiden, A.N.; Scheidegger, D.; Oomen, L.C.J.M.; et al. Mannose receptor-mediated uptake of antigens strongly enhances HLA class II-restricted antigen presentation by cultured dendritic cells. Eur. J. Immunol. 1997, 27, 2426–2435. [Google Scholar] [CrossRef]
- McIntosh, J.D.; Brimble, M.A.; Brooks, A.E.S.; Dunbar, P.R.; Kowalczyk, R.; Tomabechi, Y.; Fairbanks, A.J. Convergent chemo-enzymatic synthesis of mannosylated glycopeptides; targeting of putative vaccine candidates to antigen presenting cells. Chem. Sci. 2015, 6, 4636–4642. [Google Scholar] [CrossRef]
- Biglova, Y.N.; Mustafin, A.G. Nucleophilic cyclopropanation of [60]fullerene by the addition-elimination mechanism. RSC Adv. 2019, 9, 22428–22498. [Google Scholar] [CrossRef]
- Li, G.; Jin, B.; Chai, Z.; Liao, L.; Chu, S.; Peng, R. Synthesis and stabilization mechanism of novel stabilizers for fullerene-malonamide derivatives in nitrocellulose-based propellants. Polym. Test. 2020, 86, 106493. [Google Scholar] [CrossRef]
- Wang, K.; Wu, Y.S.; Wang, G.T.; Wang, R.X.; Jiang, X.K.; Fu, H.B.; Li, Z.T. Hydrogen bonding-mediated foldamer-bridged zinc porphyrin-C60 dyads: Ideal face-to-face orientation and tunable donor-acceptor interacion. Tetrahedron 2009, 65, 7718–7729. [Google Scholar] [CrossRef]
- Wharton, T.; Wilson, L.J. Highly-iodinated fullerene as a contrast agent for X-ray imaging. Bioorg. Med. Chem. 2002, 10, 3545–3554. [Google Scholar] [CrossRef]
- Kato, H.; Böttcher, C.; Hirsch, A. Sugar balls: Synthesis and supramolecular assembly of [60]fullerene glycoconjugates. Eur. J. Org. Chem. 2007, 4, 2659–2666. [Google Scholar] [CrossRef]
- Gulumkar, V.; Tähtinen, V.; Ali, A.; Rahkila, J.; Valle-Delgado, J.J.; Äärelä, A.; Österberg, M.; Yliperttula, M.; Virta, P. Synthesis of an Azide- and Tetrazine-Functionalized [60]Fullerene and Its Controlled Decoration with Biomolecules. ACS Omega 2022, 7, 1329–1336. [Google Scholar] [CrossRef]
- Fensterbank, H.; Baczko, K.; Constant, C.; Idttalbe, N.; Bourdreux, F.; Vallée, A.; Goncalves, A.M.; Méallet-Renault, R.; Clavier, G.; Wright, K.; et al. Sequential Copper-Catalyzed Alkyne-Azide Cycloaddition and Thiol-Maleimide Addition for the Synthesis of Photo- and/or Electroactive Fullerodendrimers and Cysteine-Functionalized Fullerene Derivatives. J. Org. Chem. 2016, 81, 8222–8233. [Google Scholar] [CrossRef] [PubMed]
- Tanzi, L.; Robescu, M.S.; Marzatico, S.; Recca, T.; Zhang, Y.; Terreni, M.; Bavaro, T. Developing a Library of Mannose-Based Mono- and Disaccharides: A General Chemoenzymatic Approach to Monohydroxylated Building Blocks. Molecules 2020, 25, 5764. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.R.; Michel, J. Facile Synthesis of α- and β-O-Glycosyl Imidates; Preparation of Glycosides and Disaccharides. Angew. Chem. Int. Ed. Engl. 1980, 19, 731–732. [Google Scholar] [CrossRef]
- Van Berkel, S.S.; Van Der Lee, B.; Van Delft, F.L.; Rutjes, F.P.J.T. A macrocyclic coumarin-containing tripeptide via CuAAC chemistry. Chem. Commun. 2009, 9, 4272–4274. [Google Scholar] [CrossRef]
- Camps, X.; Hirsch, A.; Hirsh, A. Efficient cyclopropanation of C60 starting from malonates. J. Chem. Soc. Perkin Trans. 1997, 8, 1595–1596. [Google Scholar] [CrossRef]
- Djojo, F.; Herzog, A.; Lamparth, I.; Hampel, F.; Hirsch, A. Regiochemistry of Twofold Additions to [6,6] Bonds in C60: Influence of the Addend-Independent Cage Distortion in 1,2-Monoadducts. Chem. Eur. J. 1996, 2, 1537–1547. [Google Scholar] [CrossRef]
- Trinh, T.M.N.; Schillinger, F.; Guerra, S.; Meichsner, E.; Nierengarten, I.; Hahn, U.; Holler, M.; Nierengarten, J.F. Regioselective Preparation of Fullerene Bis-adducts from Cleavable Macrocyclic Bis-malonates. Eur. J. Org. Chem. 2021, 2021, 3770–3786. [Google Scholar] [CrossRef]
- Djojo, F.; Hirsch, A. Synthesis and chiroptical properties of enantiomericaily pure bis- and trisadducts of C60 with an inherent chiral addition pattern. Chemistry—A Eur. J. 1998, 4, 344–356. [Google Scholar] [CrossRef]
- Armarego, W.L.F.; Chai, C.L.L. Purification Of Laboratory Chemicals, 7th ed.; Elsevier: Oxford, UK, 2013; ISBN 9780123821614. [Google Scholar] [CrossRef]
- Hodgson, S.M.; McNelles, S.A.; Abdullahu, L.; Marozas, I.A.; Anseth, K.S.; Adronov, A. Reproducible Dendronized PEG Hydrogels via SPAAC Cross-Linking. Biomacromolecules 2017, 18, 4054–4059. [Google Scholar] [CrossRef] [PubMed]

| Compound | Entry | Ratio (C60:Sugar) | Temperature | Time | Yield |
|---|---|---|---|---|---|
| 14 | 1 | 1:1 | rt | 1.5 h | 17% |
| 2 | 1:1 | rt | 1 h | 30% | |
| 3 | 1:1 | rt | 45 min | 41% | |
| 15 | 4 | 1:1 | rt | 1.5 h | 10% |
| 5 | 1:1 | rt | 1 h | 12% | |
| 6 | 1:1 | rt | 30 min | 16% | |
| 7 | 1:1 | rt | 15 min | 22% | |
| 8 | 1:1 | rt | 7 min | 24% | |
| 9 | 1:1 | 0 °C | 20 min | 26% | |
| 10 | 1:1 | 0 °C | 1 h | 30% | |
| 11 | 1:1 | 0 °C | 2 h | 31% | |
| 16 | 12 | 1:1 | 0 °C | 1 h | 8% |
| 13 | 1:1 | −20 °C | 3 h | 8% | |
| 14 | 1:1 | −20 °C | 1.5 h | <5% | |
| 15 | 1:1 | rt | 3 h | 11% | |
| 16 | 1:1 | rt | 1.5 h | 10% | |
| 17 | 1:1 | rt | 6 h | 5% | |
| 18 | 1:0.7 | rt | 1 h | 5% | |
| 17 | 19 | 1:1 | 0 °C | 1 h | 12% |
| 20 | 1:1 | 0 °C | 20 min | 15% | |
| 21 | 1:1 | 0 °C | 10 min | 13% | |
| 22 | 1:1 | rt | 15 min | 12% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanzi, L.; Rubes, D.; Bavaro, T.; Sollogoub, M.; Serra, M.; Zhang, Y.; Terreni, M. Controlled Decoration of [60]Fullerene with Polymannan Analogues and Amino Acid Derivatives through Malondiamide-Based Linkers. Molecules 2022, 27, 2776. https://doi.org/10.3390/molecules27092776
Tanzi L, Rubes D, Bavaro T, Sollogoub M, Serra M, Zhang Y, Terreni M. Controlled Decoration of [60]Fullerene with Polymannan Analogues and Amino Acid Derivatives through Malondiamide-Based Linkers. Molecules. 2022; 27(9):2776. https://doi.org/10.3390/molecules27092776
Chicago/Turabian StyleTanzi, Lisa, Davide Rubes, Teodora Bavaro, Matthieu Sollogoub, Massimo Serra, Yongmin Zhang, and Marco Terreni. 2022. "Controlled Decoration of [60]Fullerene with Polymannan Analogues and Amino Acid Derivatives through Malondiamide-Based Linkers" Molecules 27, no. 9: 2776. https://doi.org/10.3390/molecules27092776
APA StyleTanzi, L., Rubes, D., Bavaro, T., Sollogoub, M., Serra, M., Zhang, Y., & Terreni, M. (2022). Controlled Decoration of [60]Fullerene with Polymannan Analogues and Amino Acid Derivatives through Malondiamide-Based Linkers. Molecules, 27(9), 2776. https://doi.org/10.3390/molecules27092776









