Elucidating Sequence and Structural Determinants of Carbohydrate Esterases for Complete Deacetylation of Substituted Xylans
Abstract
:1. Introduction
2. Results and Discussion
2.1. Candidate Selection and Recombinant Protein Production
2.2. General Biochemical Properties
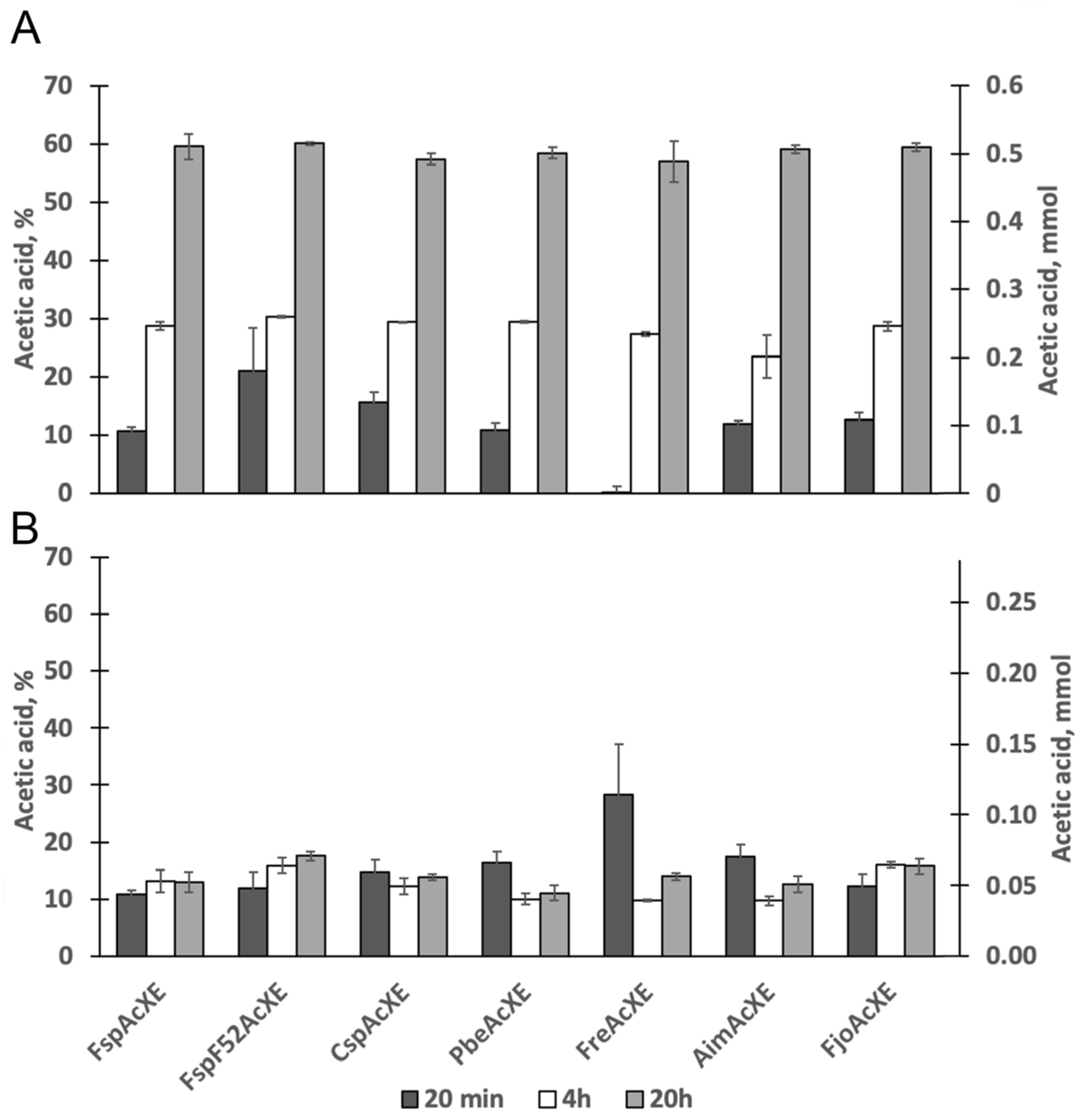
2.3. Activity on Acetylated Oligosaccharides
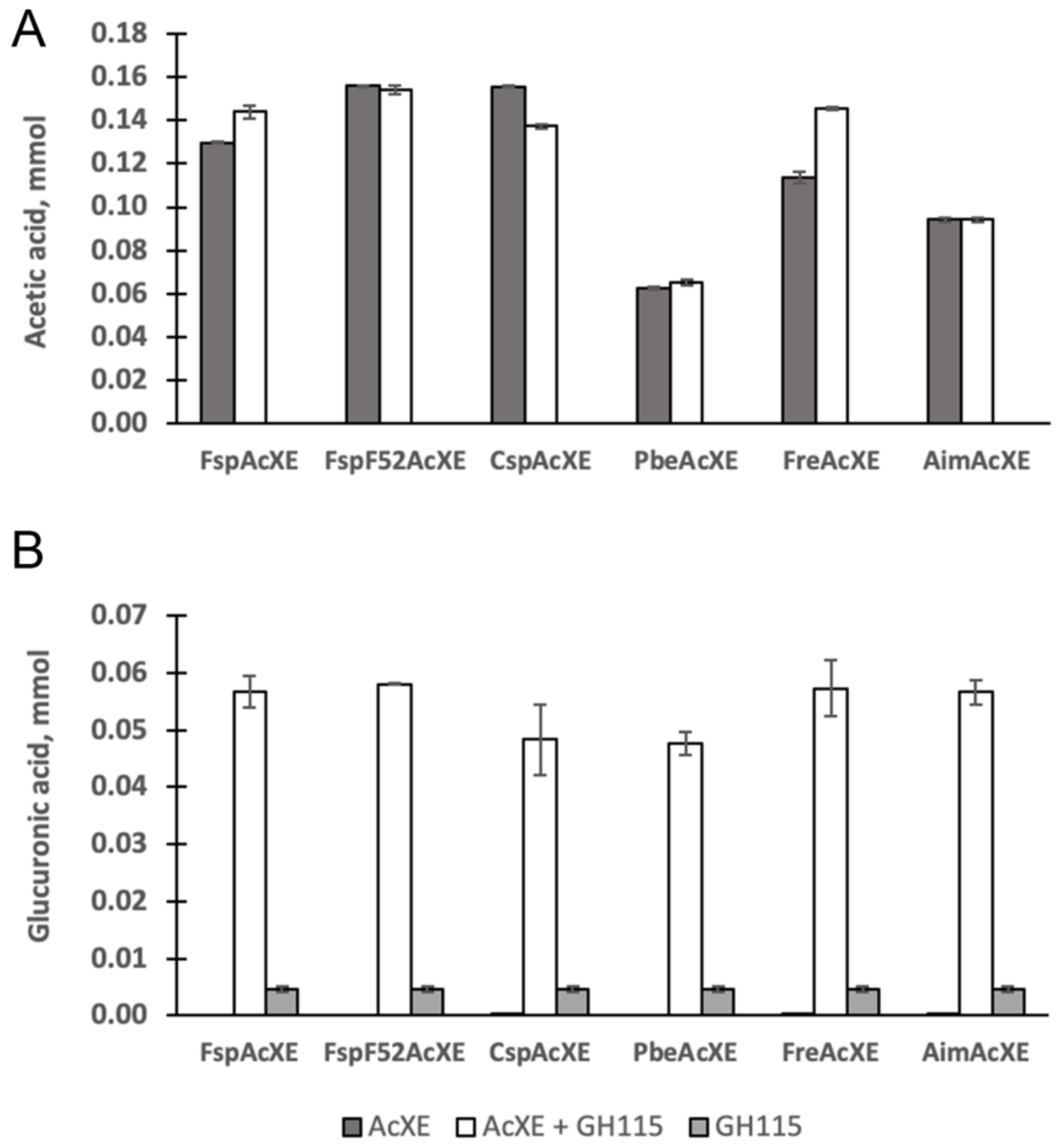
2.4. Complementation of a-Glucuronidase Activity
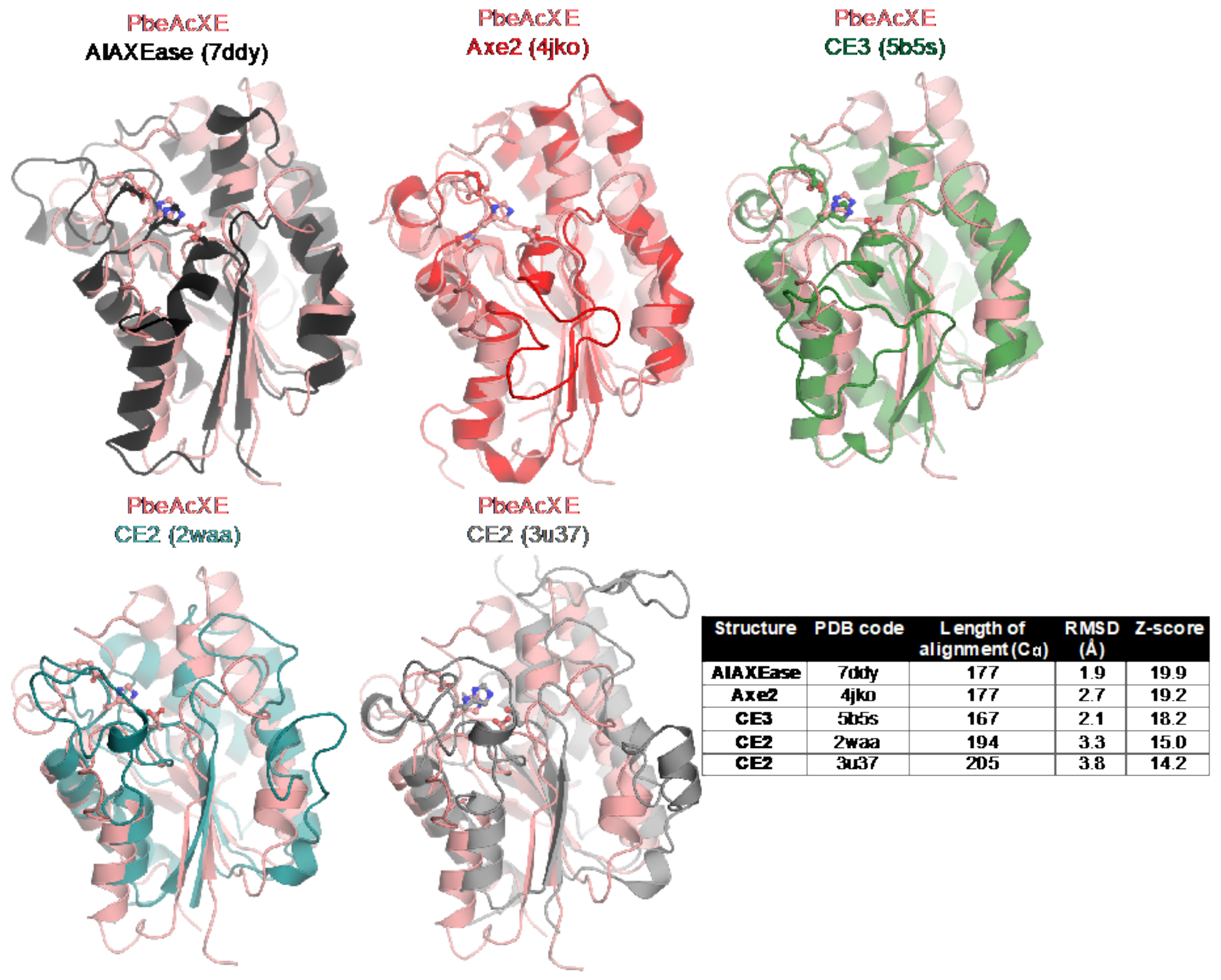
2.5. Structural Determinants of the New CE Family
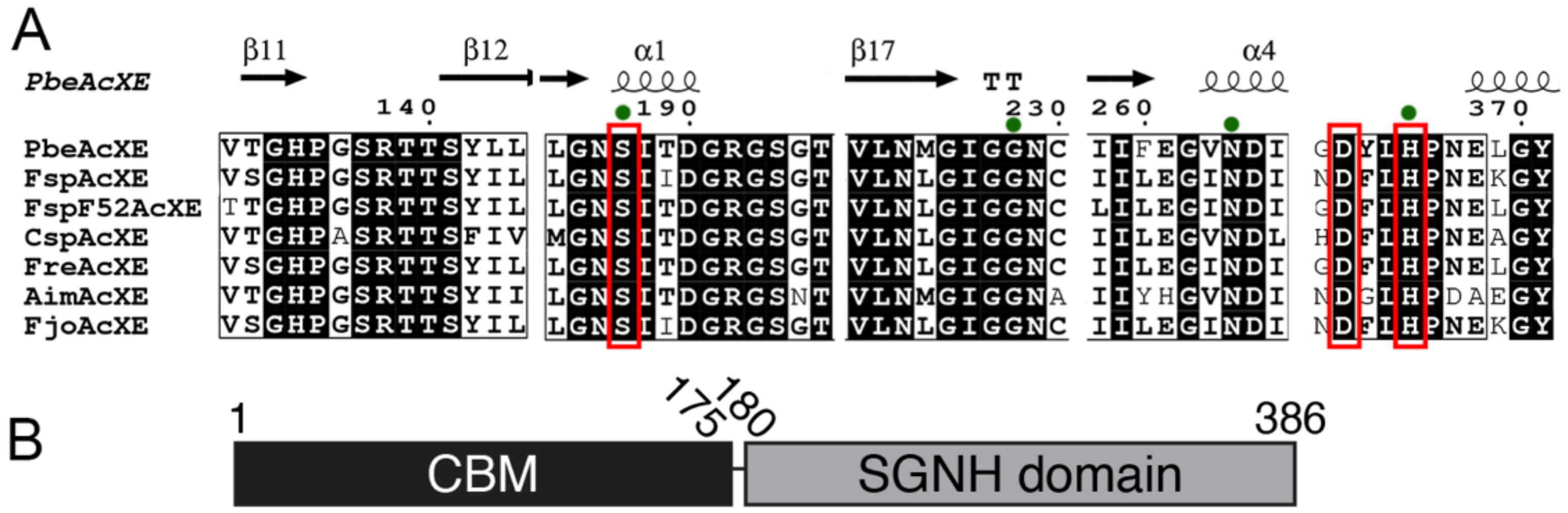
2.6. Sequence Determinants of the New CE Family
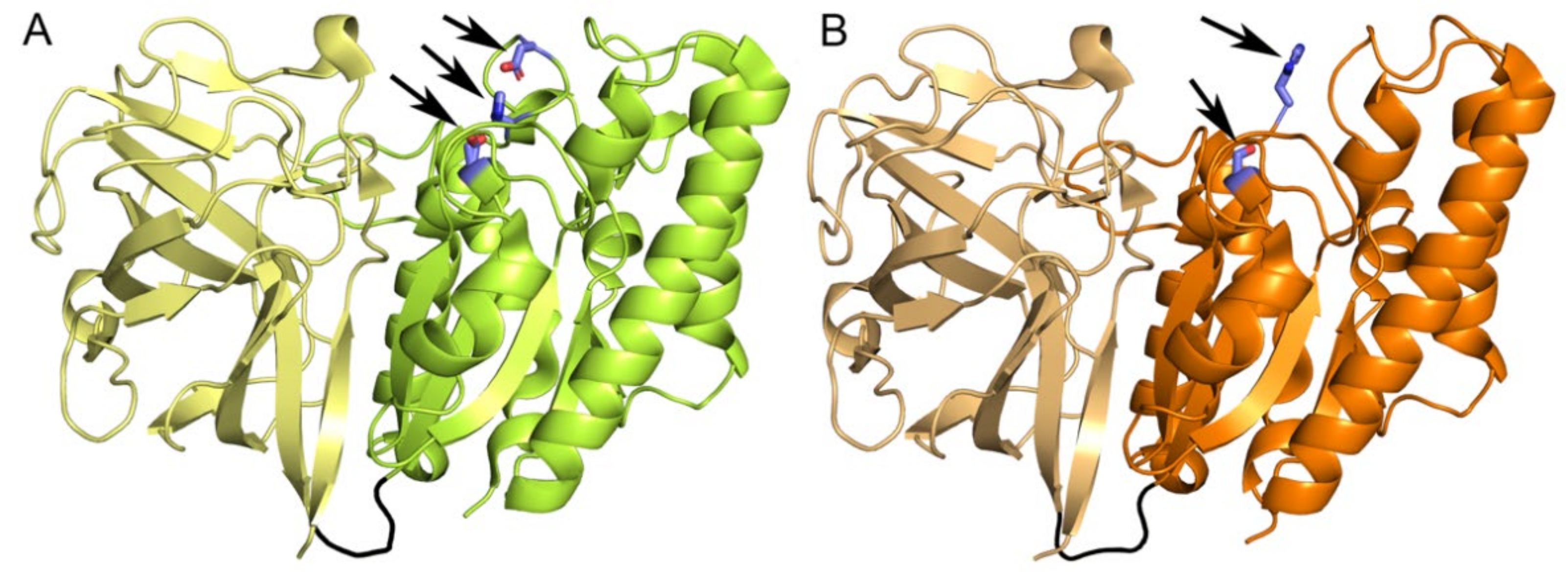
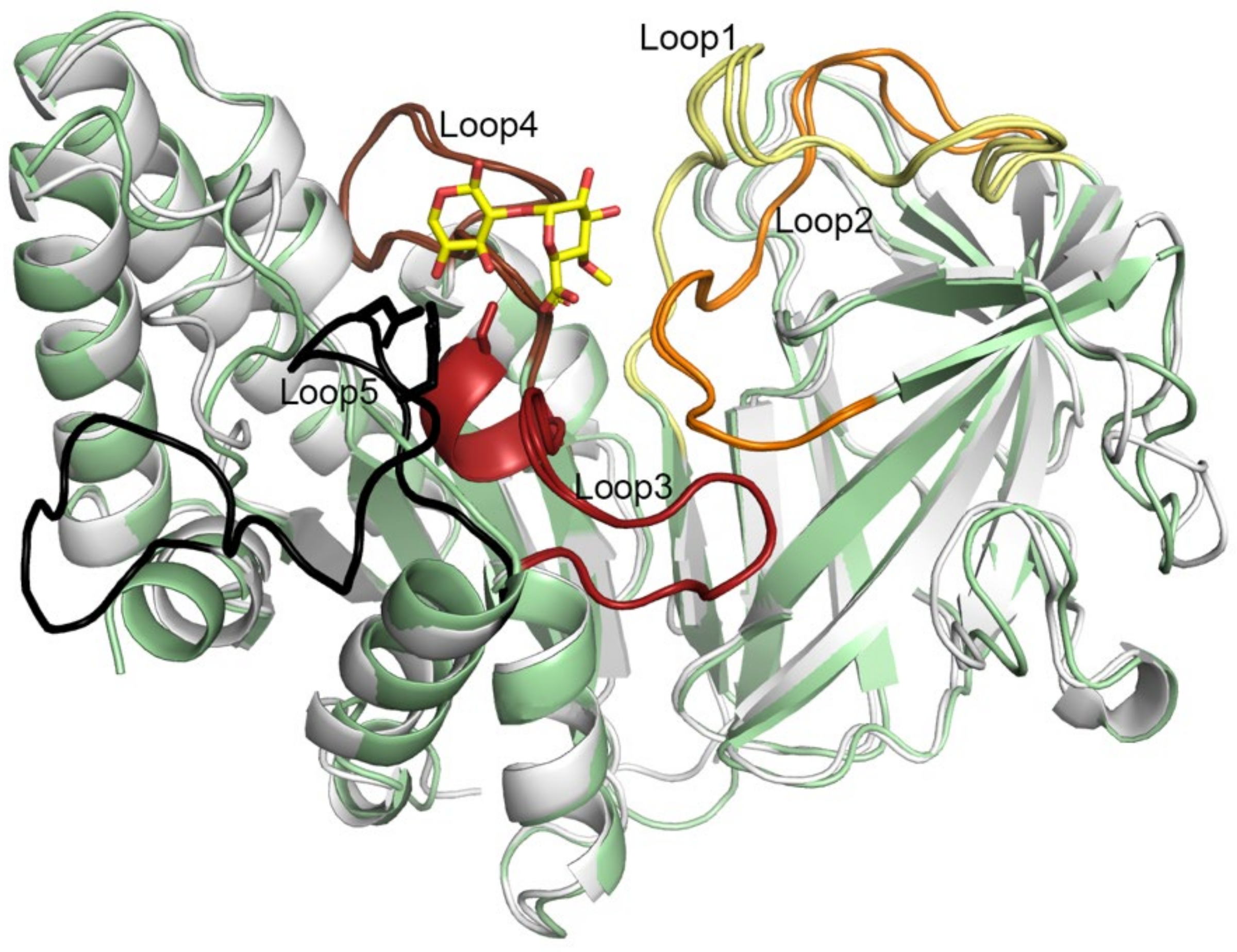
2.7. Five Conserved Loops Shape the Active Site
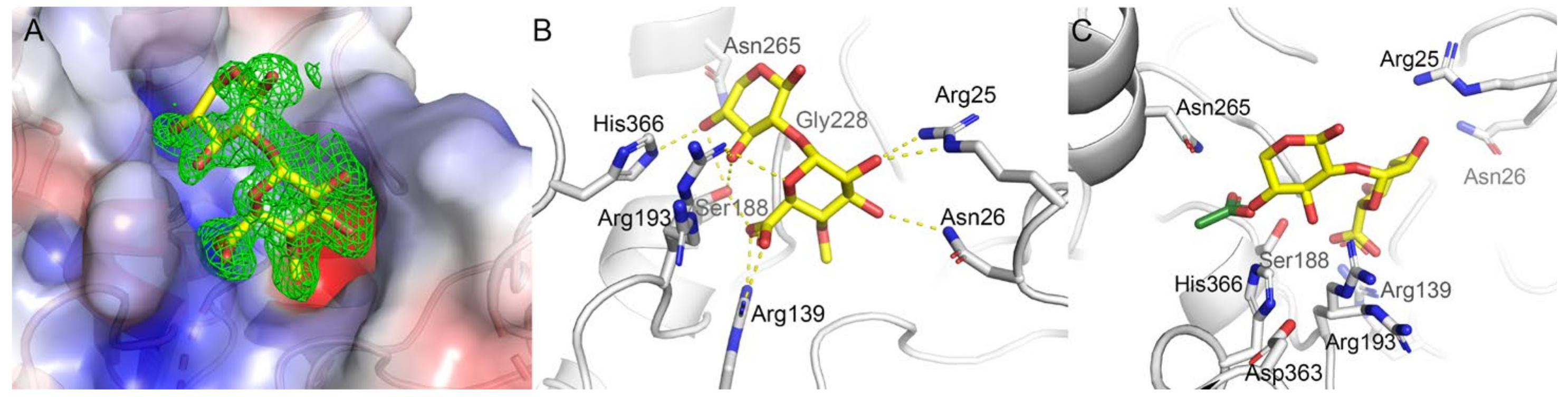
2.8. Substrate Binding at the Active Site
3. Materials and Methods
3.1. Materials
3.2. Candidate Selection
3.3. Protein Expression and Purification
3.4. Enzyme Activity Screens
3.5. Optimum Reaction Conditions
3.6. Activity on Acetylated Xylooligosaccharides
3.7. Complementation Assay Using AcXE and α-Glucuronidase
3.8. Protein Crystallization, Data Collection, and Structure Determination
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ebringerová, A.; Kramár, A.; Rendoš, F.; Domanský, R. Die Stufenextraktion Der Hemicellulosen Aus Dem Holz Der Hagebuche (Carpinus betulus L.). Holzforschung 1967, 21, 74–77. [Google Scholar] [CrossRef]
- Biely, P.; Singh, S.; Puchart, V. Towards Enzymatic Breakdown of Complex Plant Xylan Structures: State of the Art. Biotechnol. Adv. 2016, 34, 1260–1274. [Google Scholar] [CrossRef] [PubMed]
- Teleman, A.; Lundqvist, J.; Tjerneld, F.; Stålbrand, H.; Dahlman, O. Characterization of Acetylated 4-O-Methylglucuronoxylan Isolated from Aspen Employing 1H and 13C NMR Spectroscopy. Carbohydr. Res. 2000, 329, 807–815. [Google Scholar] [CrossRef]
- Teleman, A.; Tenkanen, M.; Jacobs, A.; Dahlman, O. Characterization of O-Acetyl-(4-O-Methylglucurono)Xylan Isolated from Birch and Beech. Carbohydr. Res. 2002, 337, 373–377. [Google Scholar] [CrossRef]
- Ebringerová, A.; Heinze, T. Xylan and Xylan Derivatives—Biopolymers with Valuable Properties, 1. Naturally Occurring Xylans Structures, Isolation Procedures and Properties. Macromol. Rapid Commun. 2000, 21, 542–556. [Google Scholar] [CrossRef]
- Ebringerová, A. Structural Diversity and Application Potential of Hemicelluloses. Macromol. Symp. 2005, 232, 1–12. [Google Scholar] [CrossRef]
- Shrestha, U.R.; Smith, S.; Pingali, S.V.; Yang, H.; Zahran, M.; Breunig, L.; Wilson, L.A.; Kowali, M.; Kubicki, J.D.; Cosgrove, D.J.; et al. Arabinose Substitution Effect on Xylan Rigidity and Self-Aggregation. Cellulose 2019, 26, 2267–2278. [Google Scholar] [CrossRef] [Green Version]
- Xiang, Z.; Jin, X.; Huang, C.; Li, L.; Wu, W.; Qi, H.; Nishiyama, Y. Water Cast Film Formability of Sugarcane Bagasse Xylans Favored by Side Groups. Cellulose 2020, 27, 7307–7320. [Google Scholar] [CrossRef]
- Tiwari, U.P.; Fleming, S.A.; Rasheed, M.S.A.; Jha, R.; Dilger, R.N. The Role of Oligosaccharides and Polysaccharides of Xylan and Mannan in Gut Health of Monogastric Animals. J. Nutr. Sci. 2020, 9, e21. [Google Scholar] [CrossRef]
- Mikkonen, K.S.; Tenkanen, M. Sustainable Food-Packaging Materials Based on Future Biorefinery Products: Xylans and Mannans. Trends Food Sci. Technol. 2012, 28, 90–102. [Google Scholar] [CrossRef]
- Chen, G.-G.; Qi, X.-M.; Guan, Y.; Peng, F.; Yao, C.-L.; Sun, R.-C. High Strength Hemicellulose-Based Nanocomposite Film for Food Packaging Applications. ACS Sustain. Chem. Eng. 2016, 4, 1985–1993. [Google Scholar] [CrossRef]
- Farhat, W.; Venditti, R.A.; Hubbe, M.; Taha, M.; Becquart, F.; Ayoub, A. A Review of Water-Resistant Hemicellulose-Based Materials: Processing and Applications. ChemSusChem 2017, 10, 305–323. [Google Scholar] [CrossRef] [PubMed]
- Deutschmann, R.; Dekker, R.F.H. From Plant Biomass to Bio-Based Chemicals: Latest Developments in Xylan Research. Biotechnol. Adv. 2012, 30, 1627–1640. [Google Scholar] [CrossRef] [PubMed]
- Vuong, T.V.; Master, E.R. Enzymatic Upgrading of Heteroxylans for Added-Value Chemicals and Polymers. Curr. Opin. Biotechnol. 2022, 73, 51–60. [Google Scholar] [CrossRef]
- Lombard, V.; Golaconda Ramulu, H.; Drula, E.; Coutinho, P.M.; Henrissat, B. The Carbohydrate-Active Enzymes Database (CAZy) in 2013. Nucleic Acids Res. 2014, 42, D490–D495. [Google Scholar] [CrossRef] [Green Version]
- Yan, R.; Wang, W.; Vuong, T.V.; Xiu, Y.; Skarina, T.; Di Leo, R.; Gatenholm, P.; Toriz, G.; Tenkanen, M.; Stogios, P.J.; et al. Structural Characterization of the Family GH115 α-Glucuronidase from Amphibacillus Xylanus Yields Insight into Its Coordinated Action with α-Arabinofuranosidases. New Biotechnol. 2021, 62, 49–56. [Google Scholar] [CrossRef]
- Yan, R.; Vuong, T.V.; Wang, W.; Master, E.R. Action of a GH115 α-Glucuronidase from Amphibacillus Xylanus at Alkaline Condition Promotes Release of 4- O -Methylglucopyranosyluronic Acid from Glucuronoxylan and Arabinoglucuronoxylan. Enzym. Microb. Technol. 2017, 104, 22–28. [Google Scholar] [CrossRef]
- McKee, L.S.; Sunner, H.; Anasontzis, G.E.; Toriz, G.; Gatenholm, P.; Bulone, V.; Vilaplana, F.; Olsson, L. A GH115 α-Glucuronidase from Schizophyllum Commune Contributes to the Synergistic Enzymatic Deconstruction of Softwood Glucuronoarabinoxylan. Biotechnol. Biofuels 2016, 9, 2. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Yan, R.; Nocek, B.P.; Vuong, T.V.; Di Leo, R.; Xu, X.; Cui, H.; Gatenholm, P.; Toriz, G.; Tenkanen, M.; et al. Biochemical and Structural Characterization of a Five-Domain GH115 α-Glucuronidase from the Marine Bacterium Saccharophagus degradans 2-40T. J. Biol. Chem. 2016, 291, 14120–14133. [Google Scholar] [CrossRef] [Green Version]
- Aalbers, F.; Turkenburg, J.P.; Davies, G.J.; Dijkhuizen, L.; van Bueren, A.L. Structural and Functional Characterization of a Novel Family GH115 4-O-Methyl-α-Glucuronidase with Specificity for Decorated Arabinogalactans. J. Mol. Biol. 2015, 427, 3935–3946. [Google Scholar] [CrossRef]
- Nurizzo, D.; Nagy, T.; Gilbert, H.J.; Davies, G.J. The Structural Basis for Catalysis and Specificity of the Pseudomonas Cellulosa α-Glucuronidase, GlcA67A. Structure 2002, 10, 547–556. [Google Scholar] [CrossRef] [Green Version]
- Ryabova, O.; Vršanská, M.; Kaneko, S.; van Zyl, W.H.; Biely, P. A Novel Family of Hemicellulolytic α-Glucuronidase. FEBS Lett. 2009, 583, 1457–1462. [Google Scholar] [CrossRef] [Green Version]
- Martínez, P.M.; Appeldoorn, M.M.; Gruppen, H.; Kabel, M.A. The Two Rasamsonia Emersonii α-Glucuronidases, ReGH67 and ReGH115, Show a Different Mode-of-Action towards Glucuronoxylan and Glucuronoxylo-Oligosaccharides. Biotechnol. Biofuels 2016, 9, 105. [Google Scholar] [CrossRef] [Green Version]
- Neumüller, K.G.; de Souza, A.; van Rijn, J.; Streekstra, H.; Gruppen, H.; Schols, H.A. Positional Preferences of Acetyl Esterases from Different CE Families towards Acetylated 4-O-Methyl Glucuronic Acid-Substituted Xylo-Oligosaccharides. Biotechnol. Biofuels 2015, 8, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koutaniemi, S.; van Gool, M.P.; Juvonen, M.; Jokela, J.; Hinz, S.W.; Schols, H.A.; Tenkanen, M. Distinct Roles of Carbohydrate Esterase Family CE16 Acetyl Esterases and Polymer-Acting Acetyl Xylan Esterases in Xylan Deacetylation. J. Biotechnol. 2013, 168, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Biely, P. Microbial Carbohydrate Esterases Deacetylating Plant Polysaccharides. Biotechnol. Adv. 2012, 30, 1575–1588. [Google Scholar] [CrossRef]
- Razeq, F.M.; Jurak, E.; Stogios, P.J.; Yan, R.; Tenkanen, M.; Kabel, M.A.; Wang, W.; Master, E.R. A Novel Acetyl Xylan Esterase Enabling Complete Deacetylation of Substituted Xylans. Biotechnol. Biofuels 2018, 11, 74. [Google Scholar] [CrossRef] [Green Version]
- Puchart, V.; Gjermansen, M.; Mastihubová, M.; Krogh, K.B.R.M.; Biely, P. Positional Specificity of Flavobacterium Johnsoniae Acetylxylan Esterase and Acetyl Group Migration on Xylan Main Chain. Carbohydr. Polym. 2020, 232, 115783. [Google Scholar] [CrossRef]
- Terrapon, N.; Lombard, V.; Drula, É.; Lapébie, P.; Al-Masaudi, S.; Gilbert, H.J.; Henrissat, B. PULDB: The Expanded Database of Polysaccharide Utilization Loci. Nucleic Acids Res. 2018, 46, D677–D683. [Google Scholar] [CrossRef]
- Mai-Gisondi, G.; Master, E.R. Colorimetric Detection of Acetyl Xylan Esterase Activities. Methods Mol. Biol. 2017, 1588, 45–57. [Google Scholar]
- Appeldoorn, M.M.; de Waard, P.; Kabel, M.A.; Gruppen, H.; Schols, H.A. Enzyme Resistant Feruloylated Xylooligomer Analogues from Thermochemically Treated Corn Fiber Contain Large Side Chains, Ethyl Glycosides and Novel Sites of Acetylation. Carbohydr. Res. 2013, 381, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Kovačić, F.; Granzin, J.; Wilhelm, S.; Kojić-Prodić, B.; Batra-Safferling, R.; Jaeger, K.-E. Structural and Functional Characterisation of TesA—A Novel Lysophospholipase A from Pseudomonas Aeruginosa. PLoS ONE 2013, 8, e69125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lansky, S.; Alalouf, O.; Solomon, H.V.; Alhassid, A.; Govada, L.; Chayen, N.E.; Belrhali, H.; Shoham, Y.; Shoham, G. A Unique Octameric Structure of Axe2, an Intracellular Acetyl-Xylooligosaccharide Esterase from Geobacillus Stearothermophilus. Acta Crystallogr. Sect. D Biol. Crystallogr. 2014, 70, 261–278. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Fukada, H.; Inoue, H.; Ishikawa, K. Crystal Structure of an Acetylesterase from Talaromyces Cellulolyticus and the Importance of a Disulfide Bond near the Active Site. FEBS Lett. 2015, 589, 1200–1206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montanier, C.; Money, V.A.; Pires, V.M.R.; Flint, J.E.; Pinheiro, B.A.; Goyal, A.; Prates, J.A.M.; Izumi, A.; Stålbrand, H.; Morland, C.; et al. The Active Site of a Carbohydrate Esterase Displays Divergent Catalytic and Noncatalytic Binding Functions. PLoS Biol. 2009, 7, e71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Till, M.; Goldstone, D.C.; Attwood, G.T.; Moon, C.D.; Kelly, W.J.; Arcus, V.L. Structure and Function of an Acetyl Xylan Esterase (Est2A) from the Rumen Bacterium Butyrivibrio Proteoclasticus. Proteins 2013, 81, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Chekan, J.R.; Dodd, D.; Hong, P.-Y.; Radlinski, L.; Revindran, V.; Nair, S.K.; Mackie, R.I.; Cann, I. Xylan Utilization in Human Gut Commensal Bacteria Is Orchestrated by Unique Modular Organization of Polysaccharide-Degrading Enzymes. Proc. Natl. Acad. Sci. USA 2014, 111, E3708–E3717. [Google Scholar] [CrossRef] [Green Version]
- Fujimoto, Z.; Suzuki, N.; Kishine, N.; Ichinose, H.; Momma, M.; Kimura, A.; Funane, K. Carbohydrate-Binding Architecture of the Multi-Modular α-1,6-Glucosyltransferase from Paenibacillus Sp. 598K, Which Produces α-1,6-Glucosyl-α-Glucosaccharides from Starch. Biochem. J. 2017, 474, 2763–2778. [Google Scholar] [CrossRef]
- Pluvinage, B.; Grondin, J.M.; Amundsen, C.; Klassen, L.; Moote, P.E.; Xiao, Y.; Thomas, D.; Pudlo, N.A.; Anele, A.; Martens, E.C.; et al. Molecular Basis of an Agarose Metabolic Pathway Acquired by a Human Intestinal Symbiont. Nat. Commun. 2018, 9, 1043. [Google Scholar] [CrossRef]
- Itoh, T.; Panti, N.; Hayashi, J.; Toyotake, Y.; Matsui, D.; Yano, S.; Wakayama, M.; Hibi, T. Crystal Structure of the Catalytic Unit of Thermostable GH87 α-1,3-Glucanase from Streptomyces Thermodiastaticus Strain HF3-3. Biochem. Biophys. Res. Commun. 2020, 533, 1170–1176. [Google Scholar] [CrossRef]
- Ollis, D.L.; Cheah, E.; Cygler, M.; Dijkstra, B.; Frolow, F.; Franken, S.M.; Harel, M.; Remington, S.J.; Silman, I.; Schrag, J.; et al. The α/β Hydrolase Fold. Protein Eng. Des. Sel. 1992, 5, 197–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Upton, C.; Buckley, J.T. A New Family of Lipolytic Enzymes? Trends Biochem. Sci. 1995, 20, 178–179. [Google Scholar] [CrossRef]
- Akoh, C.C.; Lee, G.-C.; Liaw, Y.-C.; Huang, T.-H.; Shaw, J.-F. GDSL Family of Serine Esterases/Lipases. Prog. Lipid Res. 2004, 43, 534–552. [Google Scholar] [CrossRef] [PubMed]
- Alalouf, O.; Balazs, Y.; Volkinshtein, M.; Grimpel, Y.; Shoham, G.; Shoham, Y. A New Family of Carbohydrate Esterases Is Represented by a GDSL Hydrolase/Acetylxylan Esterase from Geobacillus Stearothermophilus. J. Biol. Chem. 2011, 286, 41993–42001. [Google Scholar] [CrossRef] [Green Version]
- Soni, S.; Sathe, S.S.; Odaneth, A.A.; Lali, A.M.; Chandrayan, S.K. SGNH Hydrolase-Type Esterase Domain Containing Cbes-AcXE2: A Novel and Thermostable Acetyl Xylan Esterase from Caldicellulosiruptor Bescii. Extremophiles 2017, 21, 687–697. [Google Scholar] [CrossRef]
- Polgár, L. The Catalytic Triad of Serine Peptidases. Cell. Mol. Life Sci. 2005, 62, 2161–2172. [Google Scholar] [CrossRef]
- Brumlik, M.J.; Buckley, J.T. Identification of the Catalytic Triad of the Lipase/Acyltransferase from Aeromonas Hydrophila. J. Bacteriol. 1996, 178, 2060–2064. [Google Scholar] [CrossRef] [Green Version]
- Hameleers, L.; Penttinen, L.; Ikonen, M.; Jaillot, L.; Fauré, R.; Terrapon, N.; Deuss, P.J.; Hakulinen, N.; Master, E.R.; Jurak, E. Polysaccharide Utilization Loci-Driven Enzyme Discovery Reveals BD-FAE: A Bifunctional Feruloyl and Acetyl Xylan Esterase Active on Complex Natural Xylans. Biotechnol. Biofuels 2021, 14, 127. [Google Scholar] [CrossRef]
- Gullón, P.; González-Muñoz, M.J.; Domínguez, H.; Parajó, J.C. Membrane Processing of Liquors from Eucalyptus Globulus Autohydrolysis. J. Food Eng. 2008, 87, 257–265. [Google Scholar] [CrossRef]
- Studier, F.W. Protein Production by Auto-Induction in High-Density Shaking Cultures. Protein Expr. Purif. 2005, 41, 207–234. [Google Scholar] [CrossRef]
- Lever, M. Carbohydrate Determination with 4-Hydroxybenzoic Acid Hydrazide (PAHBAH): Effect of Bismuth on the Reaction. Anal. Biochem. 1977, 81, 21–27. [Google Scholar] [CrossRef]
- Minor, W.; Cymborowski, M.; Otwinowski, Z.; Chruszcz, M. HKL -3000: The Integration of Data Reduction and Structure Solution—From Diffraction Images to an Initial Model in Minutes. Acta Crystallogr. Sect. D Biol. Crystallogr. 2006, 62, 859–866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liebschner, D.; Afonine, P.V.; Baker, M.L.; Bunkóczi, G.; Chen, V.B.; Croll, T.I.; Hintze, B.; Hung, L.-W.; Jain, S.; McCoy, A.J.; et al. Macromolecular Structure Determination Using X-Rays, Neutrons and Electrons: Recent Developments in Phenix. Acta Crystallogr. Sect. D Struct. Biol. 2019, 75, 861–877. [Google Scholar] [CrossRef] [Green Version]
- Casañal, A.; Lohkamp, B.; Emsley, P. Current Developments in Coot for Macromolecular Model Building of Electron Cryo-microscopy and Crystallographic Data. Protein Sci. 2020, 29, 1055–1064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Substrate | pH | FspAcXE | FspF52AcXE | CspAcXE | PbeAcXE | FreAcXE | AimAcXE |
|---|---|---|---|---|---|---|---|
| pNPA | 5.5 | 23 | 27 | 52 | 42 | 47 | 24 |
| 7 | 47 | 47 | 57 | 55 | 50 | 53 | |
| 4-MUA | 5.5 | 35 | 32 | 33 | 31 | 25 | 25 |
| 7 | 45 | 42 | 48 | 47 | 40 | 54 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Penttinen, L.; Kouhi, V.; Fauré, R.; Skarina, T.; Stogios, P.; Master, E.; Jurak, E. Elucidating Sequence and Structural Determinants of Carbohydrate Esterases for Complete Deacetylation of Substituted Xylans. Molecules 2022, 27, 2655. https://doi.org/10.3390/molecules27092655
Penttinen L, Kouhi V, Fauré R, Skarina T, Stogios P, Master E, Jurak E. Elucidating Sequence and Structural Determinants of Carbohydrate Esterases for Complete Deacetylation of Substituted Xylans. Molecules. 2022; 27(9):2655. https://doi.org/10.3390/molecules27092655
Chicago/Turabian StylePenttinen, Leena, Vera Kouhi, Régis Fauré, Tatiana Skarina, Peter Stogios, Emma Master, and Edita Jurak. 2022. "Elucidating Sequence and Structural Determinants of Carbohydrate Esterases for Complete Deacetylation of Substituted Xylans" Molecules 27, no. 9: 2655. https://doi.org/10.3390/molecules27092655
APA StylePenttinen, L., Kouhi, V., Fauré, R., Skarina, T., Stogios, P., Master, E., & Jurak, E. (2022). Elucidating Sequence and Structural Determinants of Carbohydrate Esterases for Complete Deacetylation of Substituted Xylans. Molecules, 27(9), 2655. https://doi.org/10.3390/molecules27092655






