Red Horse Chestnut and Horse Chestnut Flowers and Leaves: A Potential and Powerful Source of Polyphenols with High Antioxidant Capacity
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Flowers’ and Leaves’ Origin
4.2. Plant Material Preparation
4.3. Chemical Analysis
4.3.1. Dry Matter Analysis
4.3.2. Polyphenols—Extraction Parameters
4.3.3. Polyphenols—Equipment Description
4.3.4. Polyphenols—Results Calculation
4.3.5. Carotenoids and Chlorophylls—Extraction Parameters
4.3.6. Carotenoids—Equipment Description
4.3.7. Carotenoids—Results Calculation
4.3.8. Antioxidant Activity Measurement and Calculation
ABTS Reagent Preparation
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Deng, Y.; Zhao, Y.; Padilla-Zakour, O.; Yang, G. Polyphenols, antioxidant and antimicrobial activities of leaf and bark extracts of Solidago canadensis L. Ind. Crops Prod. 2015, 74, 803–809. [Google Scholar] [CrossRef]
- Song, Y.; Desta, K.T.; Kim, G.-S.; Lee, S.J.; Lee, W.S.; Kim, Y.-H.; Jin, J.S.; El-Aty, A.; Shin, H.-C.; Shim, J.-H.; et al. Polyphenolic profile and antioxidant effects of various parts of Artemisia annua L. Biomed. Chromatogr. 2015, 30, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Papuc, C.; Goran, G.V.; Predescu, C.N.; Nicorescu, V.; Stefan, G. Plant polyphenols as antioxidant and antibacterial agents for shelf-life extension of meat and meat products: Classification, structures, sources, and action mechanisms. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1243–1268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef] [Green Version]
- Bunea, C.-I.; Pop, N.; Babeş, A.C.; Matea, C.; Dulf, F.V.; Bunea, A. Carotenoids, total polyphenols and antioxidant activity of grapes (Vitis vinifera) cultivated in organic and conventional systems. Chem. Centr. J. 2012, 6, 66. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.; Kaur, I.; Kariyat, R. The multifunctional roles of polyphenols in plant-herbivore interactions. Int. J. Mol. Sci. 2021, 22, 1442. [Google Scholar] [CrossRef]
- Oszmiański, J.; Kalisz, S.; Wojdyło, A. The content of phenolic compounds in leaf tissues of white (Aesculus hippocastanum L.) and red horse chestnut (Aesculus carea H.) colonized by the horse chestnut leaf miner (Cameraria ohridella Deschka & Dimić). Molecules 2014, 19, 14625–14636. [Google Scholar]
- Percival, G.C.; Holmes, S.P. The influence of systemic inducing agents on horse chestnut leaf miner (Cameraria ohridella) severity in white flowering horse chestnut (Aesculus hoppicastanum L.). Urban For. Urban Green. 2016, 20, 97–102. [Google Scholar] [CrossRef]
- Nguyen, C.T.T.; Lim, S.; Lee, J.G.; Lee, E.J. VcBBX, VcMYB21, and VcR2R3MYB Transcription factors are involved in UV–B-induced anthocyanin biosynthesis in the peel of harvested blueberry fruit. J. Agric. Food Chem. 2017, 65, 2066–2073. [Google Scholar] [CrossRef]
- Morales, L.O.; Tegelberg, R.; Brosche, M.; Keinanen, M.; Lindfors, A.; Aphalo, P.J. Effects of solar UV-A and UV-B radiation on gene expression and phenolic accumulation in Betula pendula leaves. Tree Phys. 2010, 30, 923–934. [Google Scholar] [CrossRef] [Green Version]
- Gagić, T.; Knez, Ž.; Škerget, M. Subcritical water extraction of horse chestnut (Aesculus hippocastanum) tree parts. J. Serb. Chem. Soc. 2021, 86, 603–613. [Google Scholar] [CrossRef]
- Kędzierski, B.; Kukula-Koch, W.; Widelski, J.; Głowniak, K. Impact of harvest time of Aesculus hippocastanum seeds on the composition, antioxidant capacity and total phenolic content. Ind. Crops Prod. 2016, 86, 68–72. [Google Scholar] [CrossRef]
- Owczarek, A.; Kołodziejczyk-Czepas, J.; Marczuk, P.; Siwek, J.; Wąsowicz, K.; Olszewska, M.A. Bioactivity potential of Aesculus hippocastanum L. flower: Phytochemical profile, antiradical capacity and protective effects on human plasma components under oxidative/nitrative stress in vitro. Pharmaceuticals 2021, 14, 1301. [Google Scholar] [CrossRef] [PubMed]
- Owczarek, A.; Kolodziejczyk-Czepas, J.; Woźniak-Serwata, J.; Magiera, A.; Kobiela, N.; Wąsowicz, K.; Olszewska, M.A. Potential activity mechanisms of Aesculus hippocastanum bark: Antioxidant effects in chemical and biological in vitro models. Antioxidants 2021, 10, 995. [Google Scholar] [CrossRef]
- Chen, G.-L.; Chen, S.-G.; Xie, Y.-Q.; Chen, F.; Zhao, Y.-Y.; Luo, C.-X.; Gao, Y.-Q. Total phenolic, flavonoid and antioxidant activity of 23 edible flowers subjected to in vitro digestion. J. Funct. Foods 2015, 17, 243–259. [Google Scholar] [CrossRef]
- Kaisoon, O.; Siriamornpun, S.; Weerapreeyakul, N.; Meeso, N. Phenolic compounds and antioxidant activities of edible flowers from Thailand. J. Funct. Foods 2011, 3, 88–99. [Google Scholar] [CrossRef]
- Hallmann, E. Quantitative and qualitative identification of bioactive compounds inedible flowers of black and bristly locust and their antioxidant activity. Biomolecules 2020, 10, 1603. [Google Scholar] [CrossRef]
- Grzeszczuk, M.; Stefaniak, A.; Pachlewska, A. Biological value of various edible flower species. Acta Sci. Pol. Hortorum Cultus 2016, 15, 109–119. [Google Scholar]
- Cruz, R.; Baptista, P.; Cunha, S.; Pereira, J.A.; Casal, S. Carotenoids of lettuce (Lactuca sativa L.) grown on soil enriched with spent coffee grounds. Molecules 2012, 17, 1535–1547. [Google Scholar] [CrossRef] [Green Version]
- Bunea, A.; Andjelkovic, M.; Socaciu, C.; Bobis, O.; Neacsu, M.; Verhé, R.; Camp, J.V. Total and individual carotenoids and phenolic acids content in fresh, refrigerated and processed spinach (Spinacia oleracea L.). Food Chem. 2008, 108, 649–656. [Google Scholar] [CrossRef]
- Hsu, C.-Y.; Chao, P.-Y.; Hu, S.-P.; Yang, C.-M. The antioxidant and free radical scavenging activities of chlorophylls and pheophytins. Food Sci. Nutr. 2013, 4, 35234. [Google Scholar] [CrossRef] [Green Version]
- Nihal, A.; Mithun, P.R.; Praveen, N. Effect of heavy metals (Hg, As and La) on biochemical constituents of Spinacia oleracea. J. Pharmacogn. Phytochem. 2019, 8, 669–674. [Google Scholar]
- Spinardi, A.; Cocetta, G.; Baldassarre, V.; Ferrante, A.; Mignani, I. Quality changes during storage of spinach and lettuce baby leaf. Acta Hortic. 2010, 877, 571–576. [Google Scholar] [CrossRef]
- Rasouli, H.; Farzaei, M.H.; Khodarahmi, R. Polyphenols and their benefits: A review. Int. J. Food Prop. 2017, 1700–1741. [Google Scholar] [CrossRef] [Green Version]
- Yan, Z.; Zhong, Y.; Duan, Y.; Chen, Q.; Li, F. Antioxidant mechanism of tea polyphenols and its impact on health benefits. Anim. Nutr. 2020, 6, 115–123. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.-P.; Li, S.; Chen, Y.-M.; Li, H.-B. Natural polyphenols for prevention and treatment of cancer. Nutrients 2016, 8, 515. [Google Scholar] [CrossRef]
- Lewandowska, H.; Kalinowska, M.; Lewandowski, W.; Stępkowski, T.M.; Brzóska, K. The role of natural polyphenols in cell signaling and cytoprotection against cancer development. J. Nutr. Biochem. 2016, 32, 1–19. [Google Scholar] [CrossRef]
- Giglio, R.V.; Patti, A.M.; Cicero, A.F.G.; Lippi, G.; Rizzo, M.; Toth, P.P.; Banach, M. Polyphenols: Potential use in the prevention and treatment of cardiovascular diseases. Curr. Pharm. Des. 2018, 24, 239–258. [Google Scholar] [CrossRef]
- Chensom, S.; Okumura, H.; Mishima, T. Primary screening of antioxidant activity, total polyphenol content, carotenoid content, and nutritional composition of 13 edible flowers from Japan. Prev. Nutr. Food Sci. 2019, 24, 171–178. [Google Scholar] [CrossRef]
- Kalemba-Drożdż, M.; Cierniak, A. Antioxidant and genoprotective properties of extracts from edible flowers. J. Food Nutr. Res. 2019, 58, 42–50. [Google Scholar]
- Zeng, C.; Luo, S.; Feng, S.; Chen, T.; Zhou, L.; Yuan, M.; Huang, Y.; Liao, Y.; Ding, C. Phenolic composition, antioxidant and anticancer potentials of extracts from Rosa banksiae Ait. Flower. Molecules 2020, 58, 3068. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.S.; Silva, A.M.; Nunes, F.M. Sambucus nigra L. Fruits and flowers: Chemical composition and related bioactivities. Food Rev. Int. 2020, 1–29. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Psomas, A.; Zovoili, A.; Siatis, V. Polyphenolic profile and antioxidant activity of five apple cultivars grown under organic and conventional agricultural practices. Int. J. Food Sci. 2009, 44, 1167–1175. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, C.; Bucheli, P.; Wei, D. Citrus flavonoids in fruit and traditional Chinese medicinal food ingredients in China. Plant Foods Hum. Nut. 2006, 61, 55–63. [Google Scholar] [CrossRef]
- Veberic, R.; Slatnar, A.; Bizjak, J.; Stampar, F.; Mikulic-Petkovsek, M. Anthocyanin composition of different wild and cultivated berry species. LWT Food Sci. Techol. 2015, 60, 509–517. [Google Scholar] [CrossRef]
- Sharipov, A.; Tursunov, K.; Fazliev, S.; Azimova, B.; Razzokov, J. Hypoglycemic and anti-inflammatory effects of triterpene glycoside fractions from Aeculus hippocastanum seeds. Molecules 2021, 26, 3784. [Google Scholar] [CrossRef]
- Dudek-Makuch, M.; Studzińska-Sroka, E.; Korybalska, K.; Czepulis, N.; Łuczak, J.; Rutkowski, R.; Witowski, J. Biological activity of Aesculus hippocastanum flower extracts on vascular endothelial cells cultured in vitro. Phytochem. Lett. 2019, 30, 367–375. [Google Scholar] [CrossRef]
- Polish Standard Committee. Polish Norm PN-R-04013: The Estimation of Dry Matter in Fruits and Vegetables; Polish Standard Committee: Warszawa, Poland, 1988; pp. 1–5. [Google Scholar]
- Średnicka-Tober, D.; Ponder, A.; Hallmann, E.; Głowacka, A.; Rozpara, E. The profile and content of polyphenols and carotenoids in local and commercial sweet cherry fruits (Prunus avium L.) and their antioxidant activity in vitro. Antioxidants 2019, 8, 534. [Google Scholar] [CrossRef] [Green Version]
- Ponder, A.; Hallmann, E. Phenolics and carotenoid contents in the leaves of different organic and conventional raspberry (Rubus idaeus L.) cultivars and their in vitro activity. Antioxidants 2019, 8, 458. [Google Scholar] [CrossRef] [Green Version]
- Re, R.; Pellegrini, N.; Proteggente, A.; Nala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]



| 2018 | Flowers | Leaves | ||
| Horse Chestnut | Red Horse Chestnut | Horse Chestnut | Red Horse Chestnut | |
| dry matter (g/100 g FW) | 11.73 ± 0.27 | 11.01 ± 0.32 | 11.94 ± 0.16 | 13.83 ± 0.17 |
| total carotenoids (µg/g FW) | 36.55 ± 0.16 | 40.64 ± 0.27 | 750.27 ± 2.92 | 885.37 ± 4.29 |
| total chlorophylls (µg/g FW) | 18.95 ± 0.16 | 36.89 ± 0.28 | 1186.84 ± 13.76 | 1615.57 ± 12.96 |
| total polyphenols (mg/g FW) | 9.45 ± 0.05 | 8.25 ± 0.05 | 4.45 ± 0.03 | 5.72 ± 0.04 |
| total phenolic acids (mg/g FW) | 2.16 ± 0.05 | 0.35 ± 0.01 | 0.66 ± 0.01 | 0.46 ± 0.01 |
| total flavonols (mg/g FW) | 3.98 ± 0.04 | 2.49 ± 0.04 | 3.79 ± 0.02 | 5.27 ± 0.05 |
| total anthocyanins (mg/g FW) | 3.32 ± 0.04 | 5.41 ± 0.03 | ||
| 2019 | Flowers | Leaves | ||
| Horse Chestnut | Red Horse Chestnut | Horse Chestnut | Red Horse Chestnut | |
| dry matter (g/100 g FW) | 17.30 ± 0.70 | 15.30 ± 0.30 | 17.29 ± 0.43 | 18.29 ± 0.27 |
| total carotenoids (µg/g FW) | 38.69 ± 0.38 | 36.03 ± 0.07 | 697.55 ± 5.06 | 962.76 ± 4.59 |
| total chlorophylls (µg/g FW) | 25.88 ± 0.12 | 38.61 ± 0.18 | 1192.57 ± 13.69 | 1806.88 ± 15.39 |
| total polyphenols (mg/g FW) | 8.68 ± 0.26 | 8.96 ± 0.17 | 15.97 ± 0.94 | 13.31 ± 0.09 |
| total phenolic acids (mg/g FW) | 2.14 ± 0.17 | 1.26 ± 0.01 | 10.86 ± 0.84 | 7.36 ± 0.02 |
| total flavonols (mg/g FW) | 3.75 ± 0.11 | 2.70 ± 0.16 | 5.11 ± 0.10 | 5.95 ± 0.07 |
| total anthocyanins (mg/g FW) | 2.80 ± 0.12 | 5.00 ± 0.06 | ||
| p-Value | ||||
| Species | Organs | Years | ||
| dry matter | N.S. | N.S. | N.S. | |
| total carotenoids | <0.0001 | <0.0001 | <0.0001 | |
| total chlorophylls | <0.0001 | <0.0001 | <0.0001 | |
| total polyphenols | N.S. | N.S. | N.S. | |
| total phenolic acids | N.S. | 0.026 | N.S. | |
| total flavonols | N.S. | <0.0001 | <0.0001 | |
| total anthocyanins | <0.0001 | <0.0001 | <0.0001 | |
| 2018 | Flowers | Leaves | ||
| Horse Chestnut | Red Horse Chestnut | Horse Chestnut | Red Horse Chestnut | |
| lutein | 9.32 ± 0.03 | 10.73 ± 0.06 | 4.44 ± 0.09 | 9.71 ± 0.05 |
| zeaxanthin | 12.58 ± 0.12 | 14.44 ± 0.10 | 22.55 ± 0.04 | 31.60 ± 0.04 |
| beta-carotene | 14.65 ± 0.08 | 15.47 ± 0.15 | 723.27 ± 2.93 | 844.05 ± 4.33 |
| chlorophyll a | 7.46 ± 0.16 | 8.51 ± 0.16 | 616.01 ± 7.46 | 765.54 ± 12.43 |
| chlorophyll b | 11.46 ± 0.15 | 28.38 ± 0.12 | 570.83 ± 6.31 | 850.03 ± 3.34 |
| 2019 | Flowers | Leaves | ||
| Horse Chestnut | Red Horse Chestnut | Horse Chestnut | Red Horse Chestnut | |
| lutein | 11.43 ± 0.60 | 11.49 ± 0.12 | 3.35 ± 0.07 | 8.68 ± 0.10 |
| zeaxanthin | 13.78 ± 0.07 | 11.19 ± 0.11 | 21.56 ± 0.08 | 30.38 ± 0.15 |
| beta-carotene | 13.48 ± 0.21 | 13.35 ± 0.14 | 672.63 ± 5.13 | 923.70 ± 4.76 |
| chlorophyll a | 6.32 ± 0.15 | 7.29 ± 0.07 | 556.86 ± 4.05 | 854.21 ± 13.26 |
| chlorophyll b | 19.56 ± 0.14 | 31.31 ± 0.11 | 635.71 ± 15.91 | 952.68 ± 2.63 |
| p-Value | ||||
| Species | Organs | Years | ||
| lutein | <0.0001 | <0.0001 | <0.0001 | |
| zeaxanthin | <0.0001 | <0.0001 | <0.0001 | |
| beta-carotene | <0.0001 | <0.0001 | <0.0001 | |
| chlorophyll a | <0.0001 | <0.0001 | <0.0001 | |
| chlorophyll b | <0.0001 | <0.0001 | <0.0001 | |
| Flowers | 2018 | 2019 | ||
| Horse Chestnut | Red Horse Chestnut | Horse Chestnut | Red Horse Chestnut | |
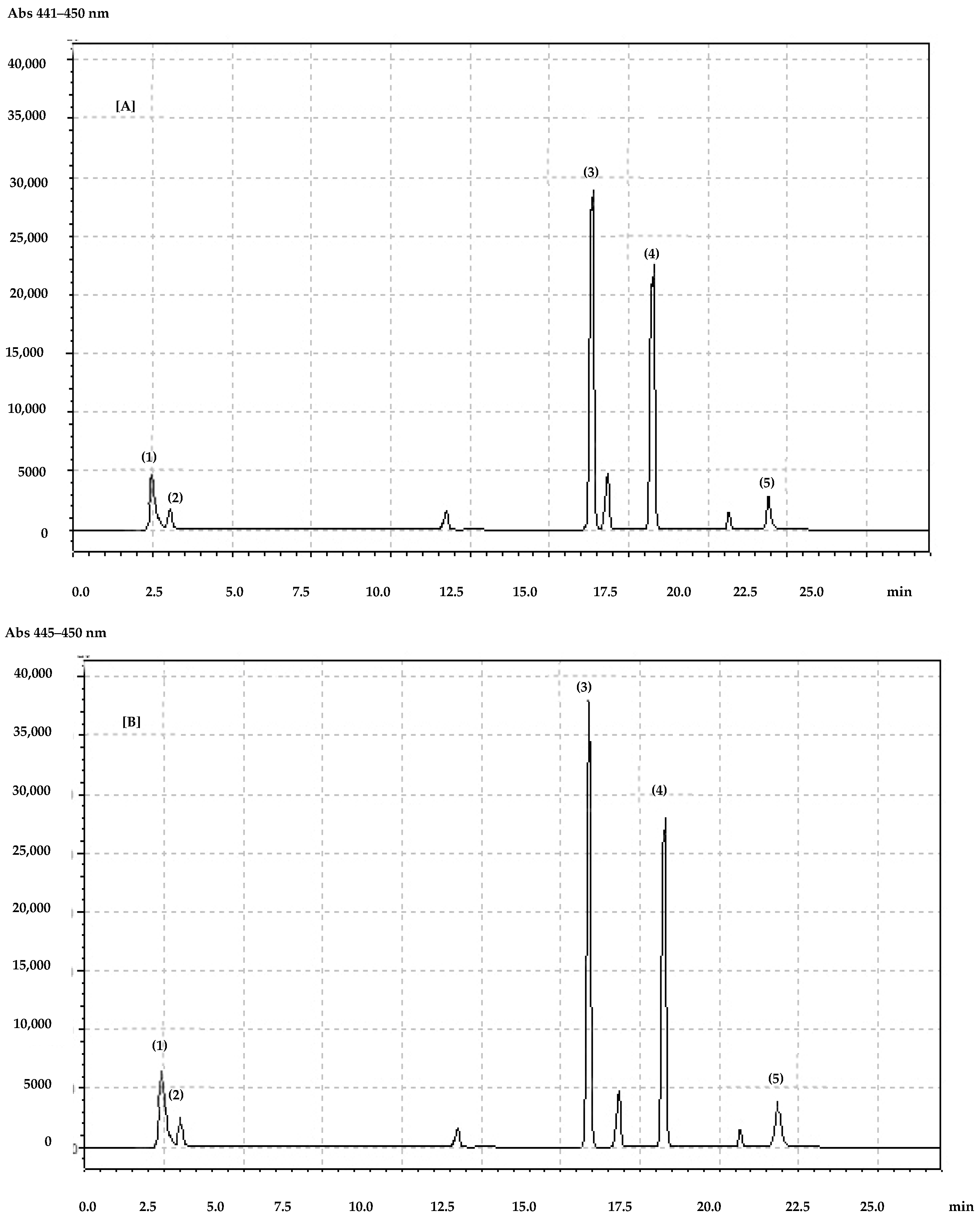
| cyanidin-3-O-glucoside | 0.19 ± 0.01 | 0.46 ± 0.01 | 0.14 ± 0.01 | 0.36 ± 0.01 |
| pelargonidin-3-O-glucoside | 1.55 ± 0.01 | 2.92 ± 0.02 | 1.34 ± 0.05 | 2.81 ± 0.06 |
| delphinidin-3-O-glucoside | 0.13 ± 0.01 | 0.18 ± 0.01 | 0.21 ± 0.01 | 0.15 ± 0.01 |
| malvidin-3-O-glucoside | 0.54 ± 0.02 | 0.25 ± 0.01 | 0.38 ± 0.03 | 0.26 ± 0.01 |
| peonidin-3-O-glucoside | 0.67 ± 0.01 | 0.85 ± 0.02 | 0.40 ± 0.02 | 0.77 ± 0.01 |
| petunin-3-O-glucoside | 0.24 ± 0.01 | 0.75 ± 0.01 | 0.33 ± 0.01 | 0.65 ± 0.01 |
| p-Value | ||||
| Species | Years | |||
| cyanidin-3-O-glucoside | <0.0001 | <0.0001 | ||
| pelargonidin-3-O-glucoside | <0.0001 | <0.0001 | ||
| delphinidin-3-O-glucoside | N.S. | N.S. | ||
| malvidin-3-O-glucoside | 0.0061 | 0.0061 | ||
| peonidin-3-O-glucoside | 0.0004 | 0.0004 | ||
| petunin-3-O-glucoside | <0.0001 | <0.0001 | ||
| 2018 | Flowers | Leaves | ||
| Horse Chestnut | Red Horse Chestnut | Horse Chestnut | Red Horse Chestnut | |
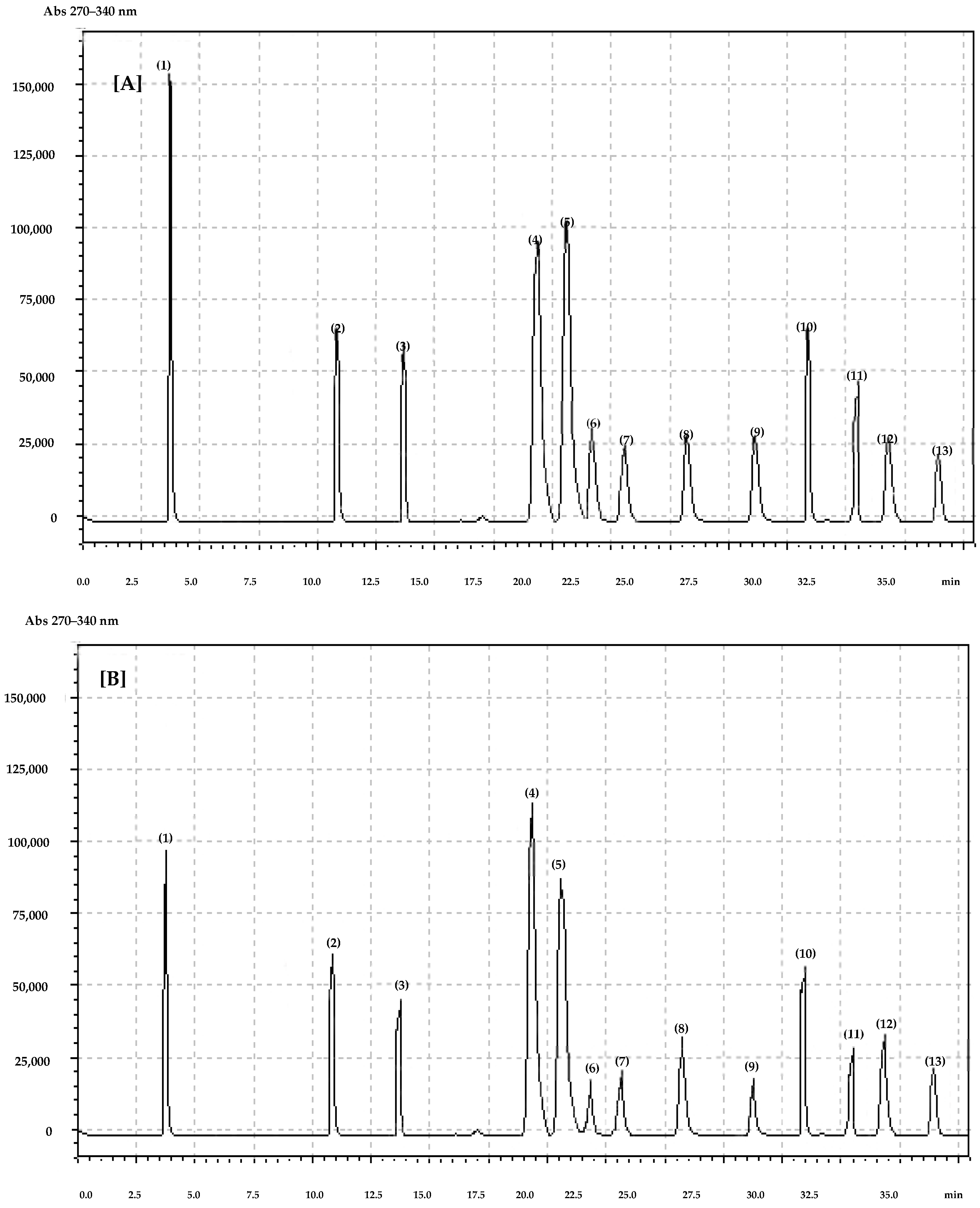
| gallic | 1.43 ± 0.04 | 0.31 ± 0.01 | 11.60 ± 0.12 | 8.35 ± 0.05 |
| chlorogenic | 0.24 ± 0.01 | 0.11 ± 0.01 | 0.14 ± 0.01 | 0.15 ± 0.01 |
| caffeic | 0.12 ± 0.01 | 0.16 ± 0.01 | 0.19 ± 0.01 | 0.06 ± 0.01 |
| p-coumaric | 0.68 ± 0.01 | 0.20 ± 0.01 | 0.46 ± 0.01 | 0.64 ± 0.01 |
| ferulic | 0.63 ± 0.01 | 0.10 ± 0.01 | 0.36 ± 0.02 | 0.08 ± 0.01 |
| quercetin-3-O-rutinoside | 0.75 ± 0.01 | 0.85 ± 0.01 | 1.23 ± 0.01 | 1.57 ± 0.01 |
| kaempferol-3-O-glucoside | 0.70 ± 0.02 | 0.64 ± 0.02 | 0.11 ± 0.01 | 0.78 ± 0.01 |
| quercetin-3-O-glucoside | 0.54 ± 0.01 | 0.23 ± 0.01 | 0.16 ± 0.01 | 0.36 ± 0.01 |
| myricetin | 0.19 ± 0.01 | 0.25 ± 0.01 | 0.35 ± 0.01 | 0.55 ± 0.02 |
| luteolin | 1.56 ± 0.01 | 0.25 ± 0.01 | 1.26 ± 0.01 | 1.66 ± 0.01 |
| quercetin | 0.11 ± 0.01 | 0.11 ± 0.01 | 0.12 ± 0.01 | 0.16 ± 0.01 |
| kaempferol | 0.12 ± 0.01 | 0.14 ± 0.01 | 0.56 ± 0.01 | 0.18 ± 0.01 |
| ABTS | 24.43 ± 0.07 | 29.57 ± 0.10 | 102.82 ± 0.21 | 115.73 ± 0.22 |
| 2019 | Flowers | Leaves | ||
| Horse Chestnut | Red Horse Chestnut | Horse Chestnut | Red Horse Chestnut | |
| gallic | 1.46 ± 0.15 | 0.98 ± 0.01 | 10.15 ± 0.08 | 7.11 ± 0.04 |
| chlorogenic | 0.13 ± 0.01 | 0.11 ± 0.01 | 0.23 ± 0.01 | 0.17 ± 0.01 |
| caffeic | 0.17 ± 0.01 | 0.11 ± 0.01 | 0.16 ± 0.01 | 0.03 ± 0.01 |
| p-coumaric | 0.77 ± 0.01 | 0.35 ± 0.01 | 0.54 ± 0.01 | 0.76 ± 0.01 |
| ferulic | 0.55 ± 0.01 | 0.07 ± 0.01 | 0.22 ± 0.01 | 0.06 ± 0.01 |
| quercetin-3-O-rutinoside | 0.87 ± 0.01 | 0.95 ± 0.01 | 2.35 ± 0.01 | 2.54 ± 0.08 |
| kaempferol-3-O-glucoside | 0.86 ± 0.01 | 0.78 ± 0.01 | 0.13 ± 0.01 | 0.87 ± 0.01 |
| quercetin-3-O-glucoside | 0.42 ± 0.01 | 0.13 ± 0.01 | 0.13 ± 0.01 | 0.24 ± 0.01 |
| myricetin | 0.16 ± 0.01 | 0.18 ± 0.01 | 0.49 ± 0.02 | 0.45 ± 0.02 |
| luteolin | 1.19 ± 0.03 | 0.45 ± 0.22 | 1.03 ± 0.05 | 1.57 ± 0.01 |
| quercetin | 0.14 ± 0.01 | 0.09 ± 0.01 | 0.19 ± 0.01 | 0.16 ± 0.01 |
| kaempferol | 0.10 ± 0.01 | 0.12 ± 0.01 | 0.79 ± 0.04 | 0.12 ± 0.01 |
| ABTS | 28.40 ± 0.06 | 26.59 ± 0.12 | 98.41 ± 0.11 | 125.64 ± 0.29 |
| p-Value | ||||
| Species | Organs | Years | ||
| gallic | <0.0001 | <0.0001 | 0.0001 | |
| chlorogenic | 0.0072 | N.S. | N.S. | |
| caffeic | <0.0001 | 0.0018 | <0.0001 | |
| p-coumaric | 0.0001 | 0.0011 | <0.0001 | |
| ferulic | <0.0001 | <0.0001 | <0.0001 | |
| quercetin-3-O-rutinoside | N.S. | <0.0001 | N.S. | |
| kaempferol-3-O-glucoside | <0.0001 | <0.0001 | <0.0001 | |
| quercetin-3-O-glucoside | 0.0052 | 0.0002 | <0.0001 | |
| myricetin | 0.0158 | <0.0001 | N.S. | |
| luteolin | 0.0037 | <0.0001 | <0.0001 | |
| quercetin | N.S. | 0.0003 | N.S. | |
| kaempferol | <0.0001 | <0.0001 | <0.0001 | |
| ABTS | <0.0001 | <0.0001 | <0.0001 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bielarska, A.M.; Jasek, J.W.; Kazimierczak, R.; Hallmann, E. Red Horse Chestnut and Horse Chestnut Flowers and Leaves: A Potential and Powerful Source of Polyphenols with High Antioxidant Capacity. Molecules 2022, 27, 2279. https://doi.org/10.3390/molecules27072279
Bielarska AM, Jasek JW, Kazimierczak R, Hallmann E. Red Horse Chestnut and Horse Chestnut Flowers and Leaves: A Potential and Powerful Source of Polyphenols with High Antioxidant Capacity. Molecules. 2022; 27(7):2279. https://doi.org/10.3390/molecules27072279
Chicago/Turabian StyleBielarska, Agnieszka Monika, Jakub Wojciech Jasek, Renata Kazimierczak, and Ewelina Hallmann. 2022. "Red Horse Chestnut and Horse Chestnut Flowers and Leaves: A Potential and Powerful Source of Polyphenols with High Antioxidant Capacity" Molecules 27, no. 7: 2279. https://doi.org/10.3390/molecules27072279
APA StyleBielarska, A. M., Jasek, J. W., Kazimierczak, R., & Hallmann, E. (2022). Red Horse Chestnut and Horse Chestnut Flowers and Leaves: A Potential and Powerful Source of Polyphenols with High Antioxidant Capacity. Molecules, 27(7), 2279. https://doi.org/10.3390/molecules27072279