The Antiviral Effect of Panax Notoginseng Polysaccharides by Inhibiting PRV Adsorption and Replication In Vitro
Abstract
:1. Introduction
2. Results
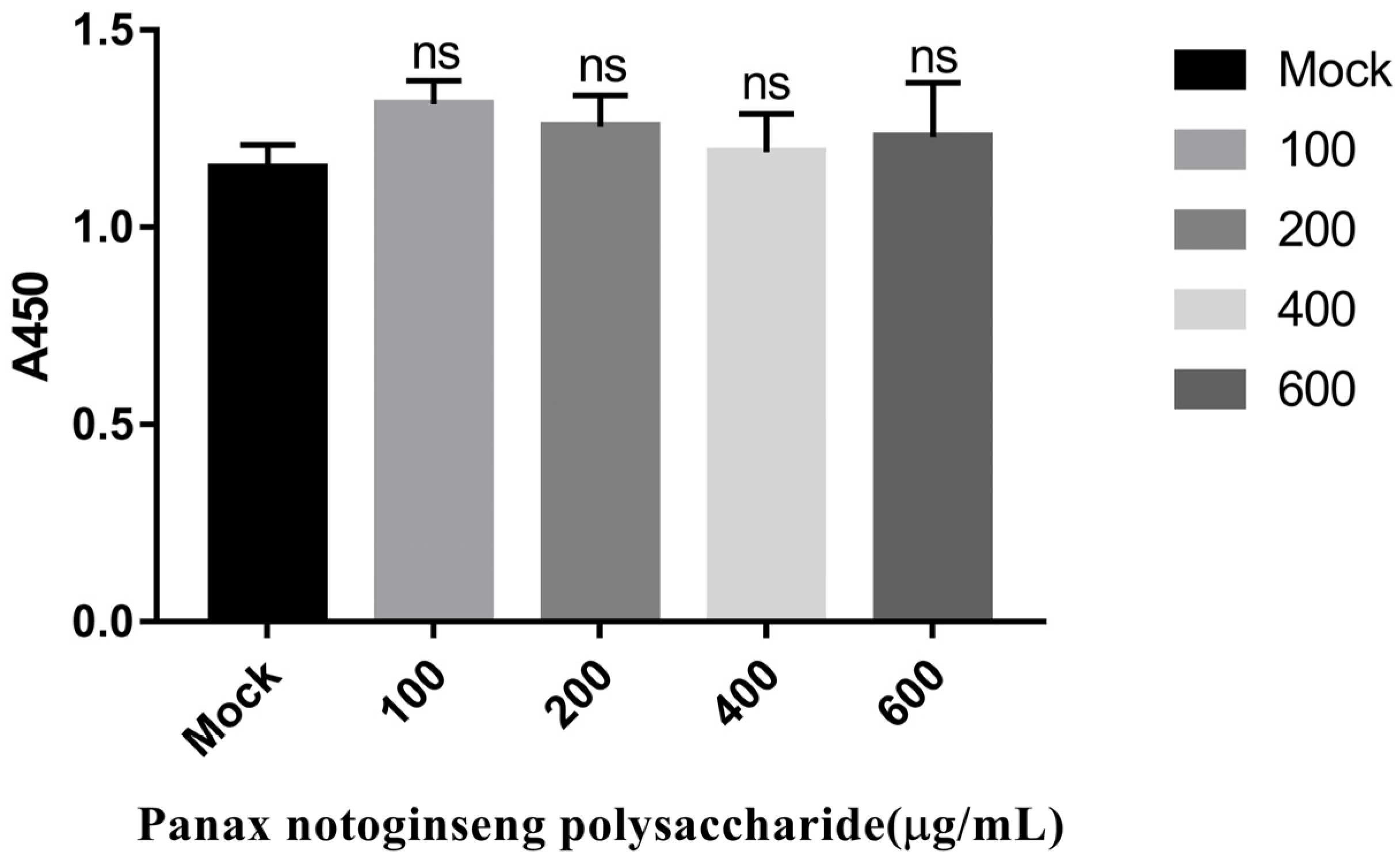
2.1. Panax Notoginseng Polysaccharides Have No Cytotoxic Effect on PK15 Cells
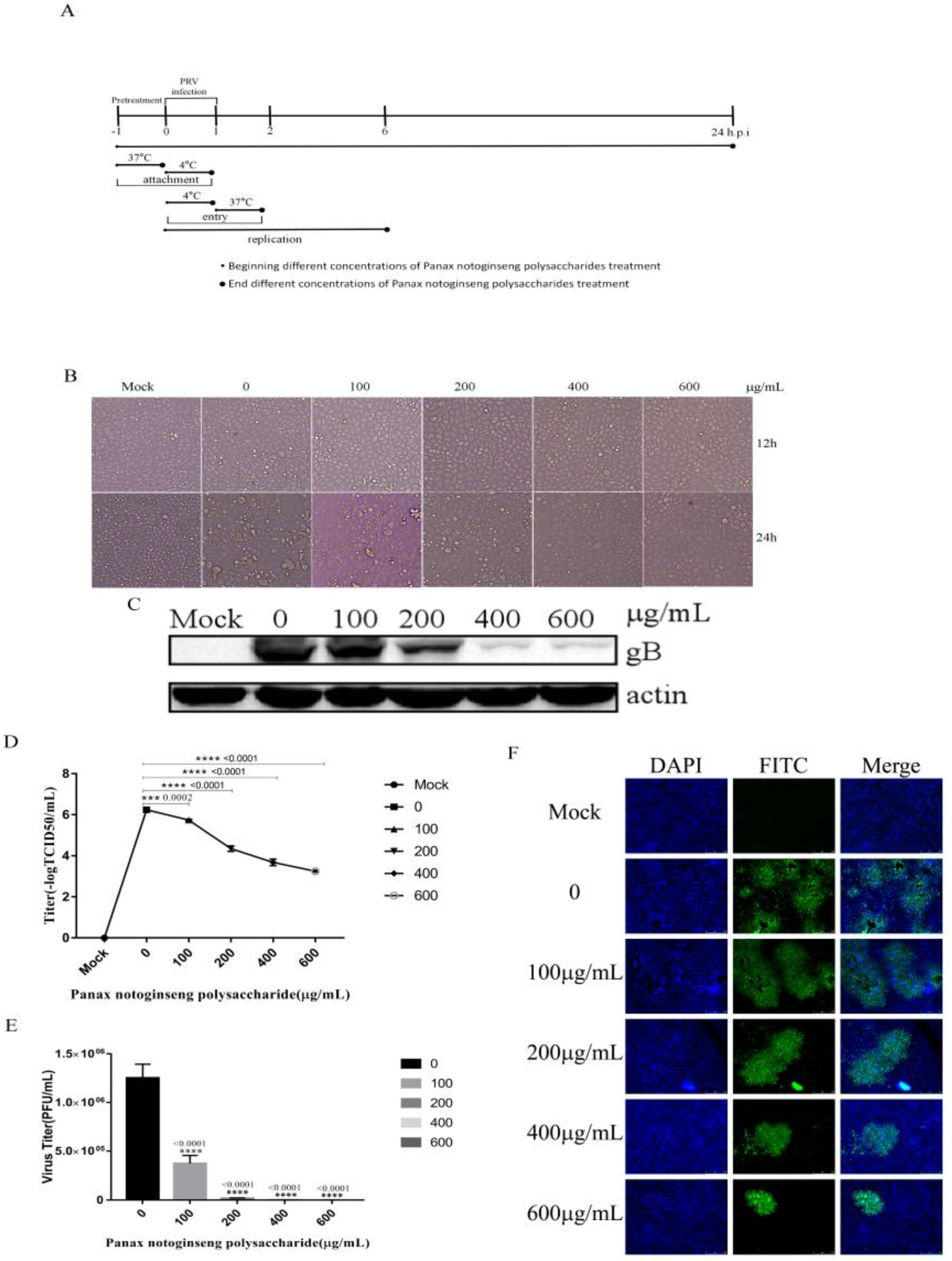
2.2. Panax Notoginseng Polysaccharides Can Inhibit PRV Infection in PK15 Cells
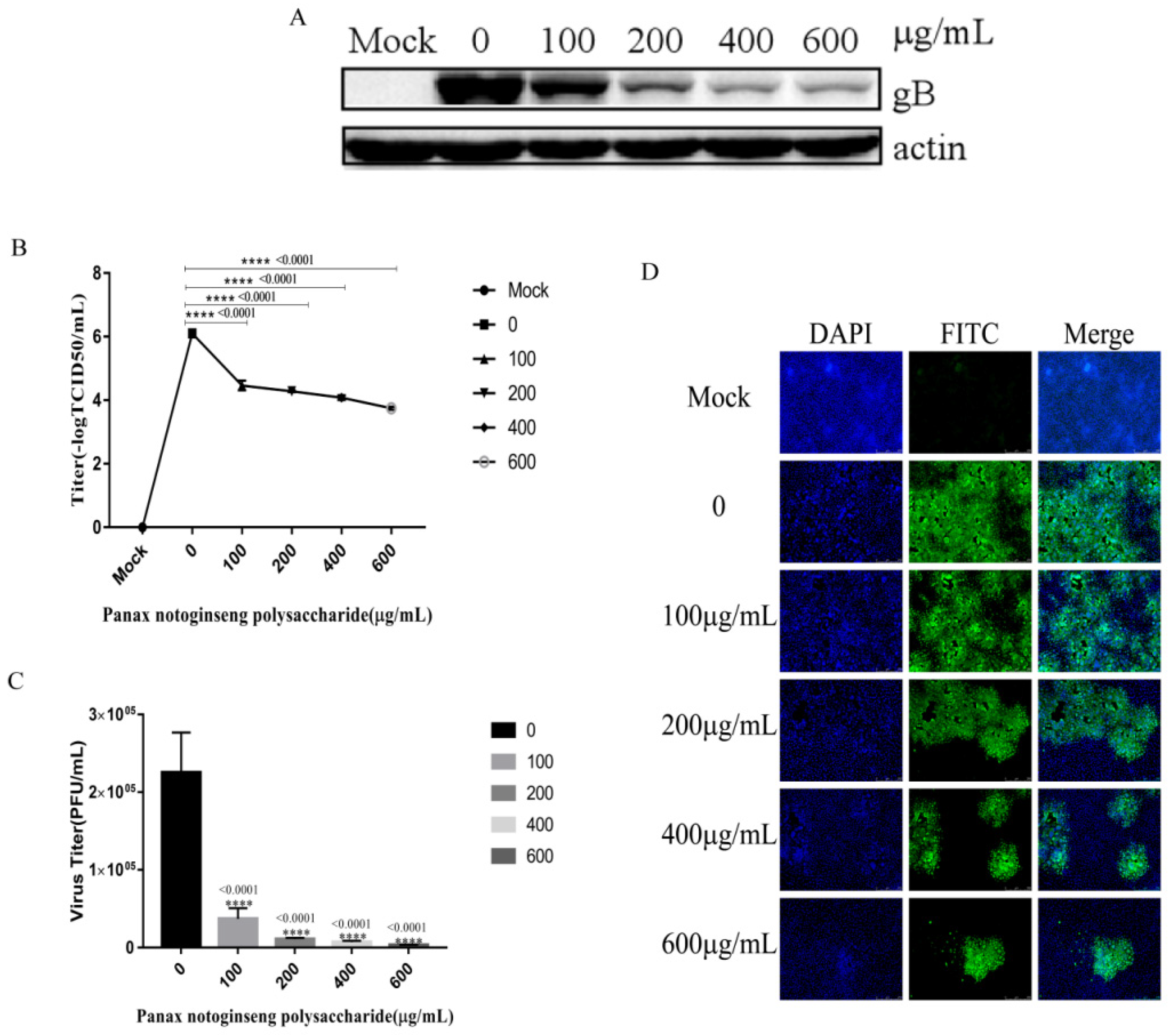
2.3. Panax Notoginseng Polysaccharides Inhibit the Adsorption and Entry of PRV to PK15 Cells
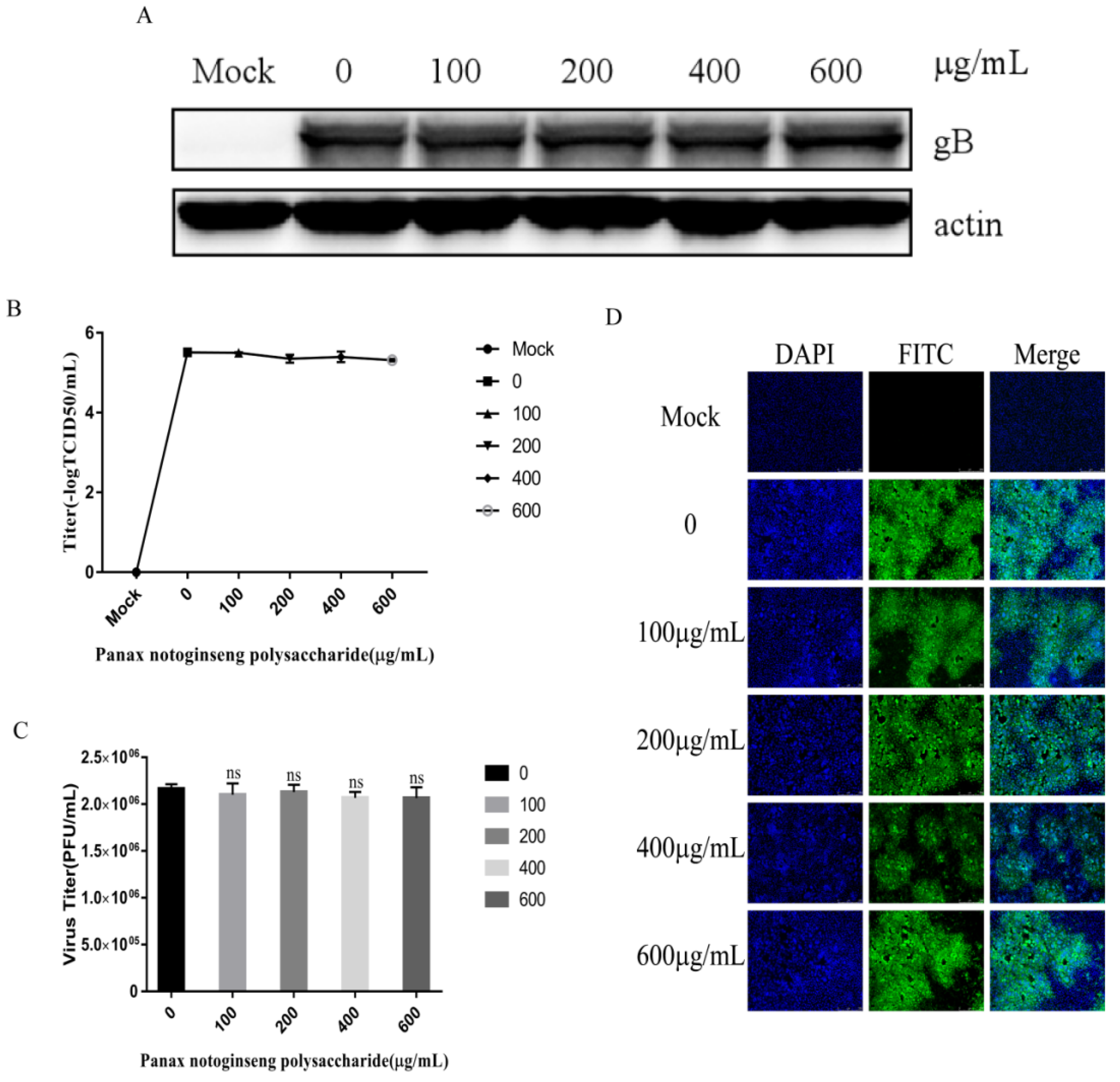
2.4. Panax Notoginseng Polysaccharides Inhibit the Adsorption of PRV to PK15 cells
2.5. Panax Notoginseng Polysaccharide Can’t Inhibit the Entry of PRV to PK15 Cells
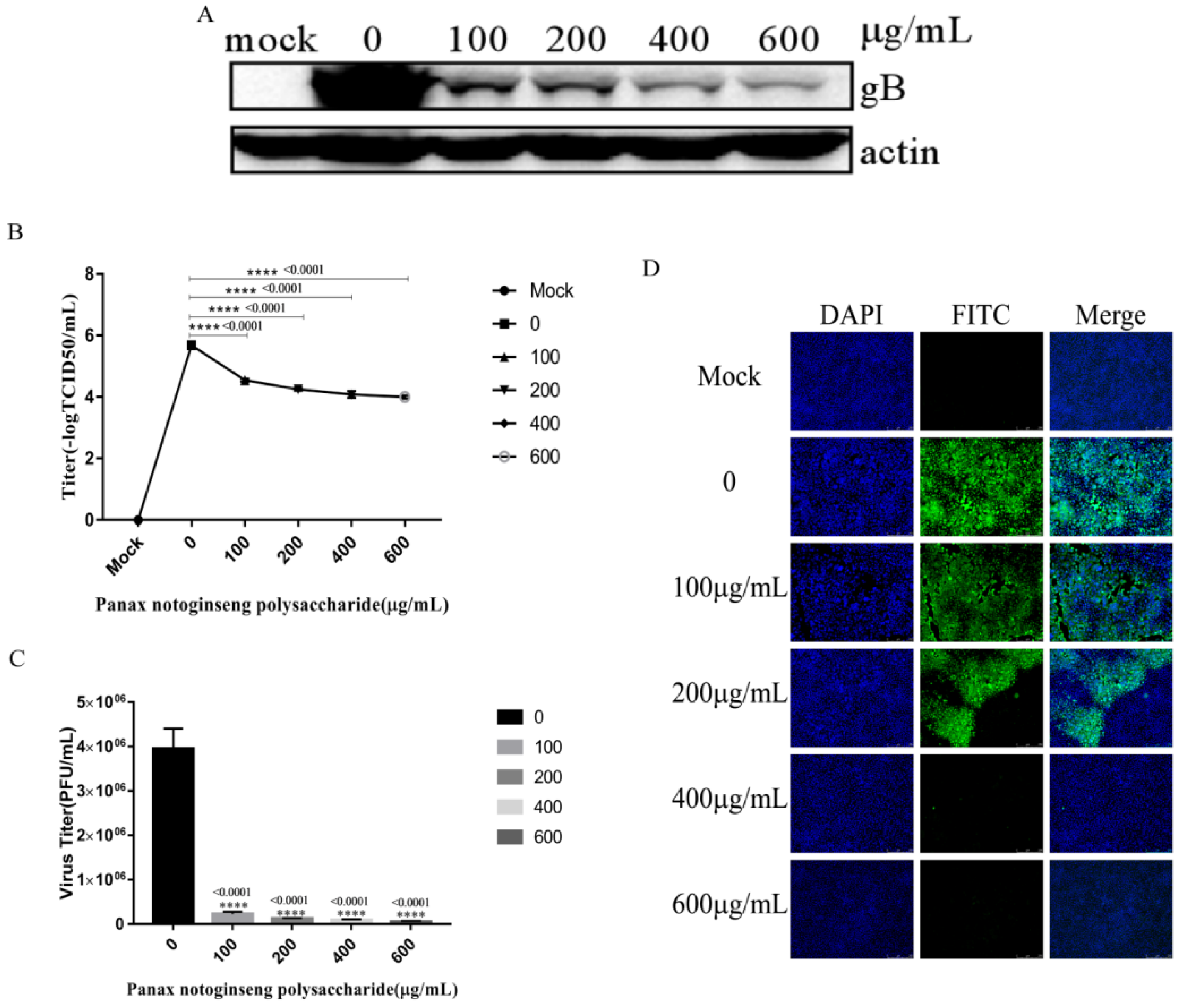
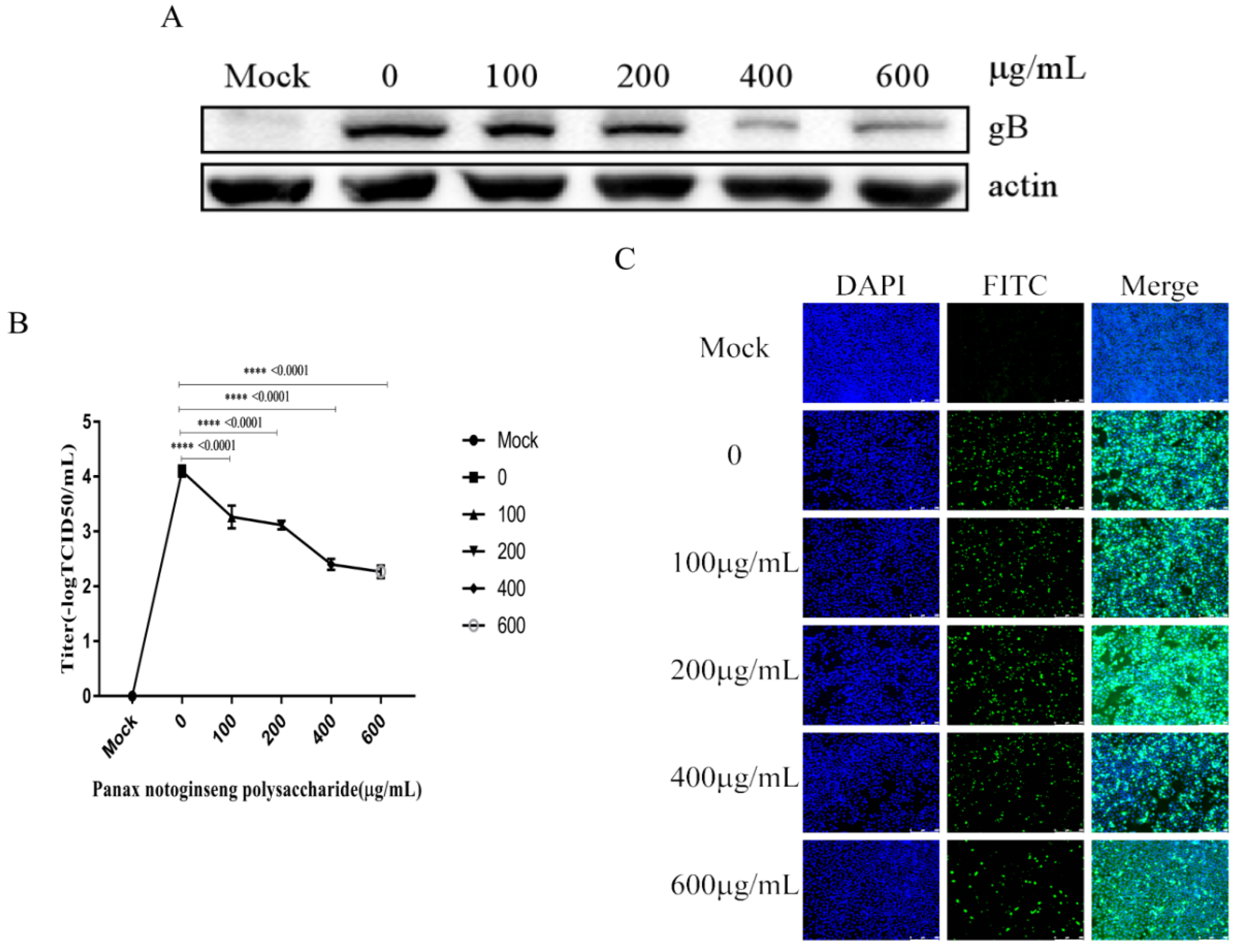
2.6. Panax Notoginseng Polysaccharides Reduce the Replication of PRV in PK15 Cells
3. Discussion
4. Materials and Methods
4.1. Cells
4.2. Virus
4.3. Reagents and Antibodies
4.4. Cytotoxic effect of Panax Notoginseng Polysaccharide on PK15 Cells
4.5. Culture of Cells
4.6. Effect of Panax Notoginseng Polysaccharide on Inhibition of PRV Infection in PK15 Cells
4.7. Effect of Panax Notoginseng Polysaccharide on Adsorption and Entry of Porcine Pseudorabies Virus
4.8. Effect of Panax Notoginseng Polysaccharide on Adsorption of PRV
4.9. Effect of Panax Notoginseng Polysaccharide on PRV Entry
4.10. Effect of Panax Notoginseng Polysaccharide on Replication of Porcine Pseudorabies Virus
4.11. Western Blotting
4.12. Virus Titer Assays
4.13. Plaque Assays
4.14. Indirect Immunofluorescent Assay (IFA)
4.15. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Shimabukuro-Vornhagen, A.; Godel, P.; Subklewe, M.; Stemmler, H.J.; Schlosser, H.A.; Schlaak, M.; Kochanek, M.; Boll, B.; von Bergwelt-Baildon, M.S. Cytokine release syndrome. J. Immunother. Cancer 2018, 6, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, G.; Lu, J.; Zhang, W.; Gao, G.F. Pseudorabies virus: A neglected zoonotic pathogen in humans? Emerg. Microbes. Infect. 2019, 8, 150–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pensaert, M. Virus Infections of Porcines; Elsevier: Amsterdam, The Netherlands; New York, NY, USA, 1989; p. xviii. 283p. [Google Scholar]
- Zhang, C.L.; Guo, L.H.; Jia, X.R.; Wang, T.Y.; Wang, J.; Sun, Z.; Wang, L.L.; Li, X.D.; Tan, F.F.; Tian, K.G. Construction of a triple gene-deleted Chinese Pseudorabies virus variant and its efficacy study as a vaccine candidate on suckling piglets. Vaccine 2015, 33, 2432–2437. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Bai, C.; Sun, J.; Chang, S.; Zhang, X. Emergence of virulent pseudorabies virus infection in northern China. J. Vet. Sci. 2013, 14, 363–365. [Google Scholar] [CrossRef] [Green Version]
- An, T.Q.; Peng, J.M.; Tian, Z.J.; Zhao, H.Y.; Li, N.; Liu, Y.M.; Chen, J.Z.; Leng, C.L.; Sun, Y.; Chang, D.; et al. Pseudorabies virus variant in Bartha-K61-vaccinated pigs, China, 2012. Emerg. Infect. Dis. 2013, 19, 1749–1755. [Google Scholar] [CrossRef]
- Ai, J.-W.; Weng, S.-S.; Cheng, Q.; Cui, P.; Li, Y.-J.; Wu, H.-L.; Zhu, Y.-M.; Xu, B.; Zhang, W.-H. Human Endophthalmitis Caused By Pseudorabies Virus Infection, China, 2017. Emerg. Infect. Dis. 2018, 24, 1087–1090. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Wang, X.; Xie, C.; Ding, S.; Yang, H.; Guo, S.; Li, J.; Qin, L.; Ban, F.; Wang, D.; et al. A novel human acute encephalitis caused by pseudorabies virus variant strain. Clin. Infect. Dis 2021, 73, e3690–e3700. [Google Scholar] [CrossRef]
- Yang, X.; Guan, H.; Li, C.; Li, Y.; Wang, S.; Zhao, X.; Zhao, Y.; Liu, Y. Characteristics of human encephalitis caused by pseudorabies virus: A case series study. Int. J. Infect. Dis. 2019, 87, 92–99. [Google Scholar] [CrossRef] [Green Version]
- Yuan, C.S.; Wang, C.Z.; Wicks, S.M.; Qi, L.W. Chemical and pharmacological studies of saponins with a focus on American ginseng. J. Ginseng Res. 2010, 34, 160–167. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Guo, R.; Zhou, G.; Zhou, X.; Kou, Z.; Sui, F.; Li, C.; Tang, L.; Wang, Z. Traditional uses, botany, phytochemistry, pharmacology and toxicology of Panax notoginseng (Burk.) F.H. Chen: A review. J. Ethnopharmacol. 2016, 188, 234–258. [Google Scholar] [CrossRef]
- Lee, D.Y.; Park, C.W.; Lee, S.J.; Park, H.R.; Kim, S.H.; Son, S.U.; Park, J.; Shin, K.S. Anti-Cancer Effects of Panax ginseng Berry Polysaccharides via Activation of Immune-Related Cells. Front. Pharmacol 2019, 10, 1411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, D.Y.; Park, C.W.; Lee, S.J.; Park, H.R.; Seo, D.B.; Park, J.Y.; Park, J.; Shin, K.S. Immunostimulating and Antimetastatic Effects of Polysaccharides Purified from Ginseng Berry. Am. J. Chin. Med. 2019, 47, 823–839. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, N.; Feng, Q.; Li, H.; Wang, D.; Ma, L.; Liu, S.; Chen, C.; Wu, W.; Jiao, L. The core structure characterization and of ginseng neutral polysaccharide with the immune-enhancing activity. Int. J. Biol. Macromol. 2019, 123, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, R.; Bryant, D.L.; Farone, A.L. Panax quinquefolius (North American Ginseng) Polysaccharides as Immunomodulators: Current Research Status and Future Directions. Molecules 2020, 25, 5854. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; In, G.; Han, S.T.; Lee, M.H.; Lee, J.W.; Shin, K.S. Structural characteristics of a red ginseng acidic polysaccharide rhamnogalacturonan I with immunostimulating activity from red ginseng. J. Ginseng. Res. 2020, 44, 570–579. [Google Scholar] [CrossRef]
- Kim, M.H.; Byon, Y.Y.; Ko, E.J.; Song, J.Y.; Yun, Y.S.; Shin, T.; Joo, H.G. Immunomodulatory activity of ginsan, a polysaccharide of panax ginseng, on dendritic cells. Korean J. Physiol. Pharmacol. 2009, 13, 169–173. [Google Scholar] [CrossRef] [Green Version]
- Cicero, A.F.; Vitale, G.; Savino, G.; Arletti, R. Panax notoginseng (Burk.) effects on fibrinogen and lipid plasma level in rats fed on a high-fat diet. Phytother. Res. 2003, 17, 174–178. [Google Scholar] [CrossRef]
- Calixto, J.B.; Campos, M.M.; Otuki, M.F.; Santos, A.R. Anti-inflammatory compounds of plant origin. Part II. modulation of pro-inflammatory cytokines, chemokines and adhesion molecules. Planta Med. 2004, 70, 93–103. [Google Scholar]
- Choi, J.G.; Jin, Y.H.; Lee, H.; Oh, T.W.; Yim, N.H.; Cho, W.K.; Ma, J.Y. Protective Effect of Panax notoginseng Root Water Extract against Influenza A Virus Infection by Enhancing Antiviral Interferon-Mediated Immune Responses and Natural Killer Cell Activity. Front. Immunol. 2017, 8, 1542. [Google Scholar] [CrossRef]
- Lam, S.K.; Ng, T.B. A xylanase from roots of sanchi ginseng (Panax notoginseng) with inhibitory effects on human immunodeficiency virus-1 reverse transcriptase. Life Sci. 2002, 70, 3049–3058. [Google Scholar] [CrossRef]
- Ohtani, K.; Mizutani, K.; Hatono, S.; Kasai, R.; Sumino, R.; Shiota, T.; Ushijima, M.; Zhou, J.; Fuwa, T.; Tanaka, O. Sanchinan-A, a reticuloendothelial system activating arabinogalactan from sanchi-ginseng (roots of Panax notoginseng). Planta Med. 1987, 53, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Wang, F.; Lien, E.J.; Trousdale, M.D. Immunostimulating polysaccharides from Panax notoginseng. Pharm. Res. 1996, 13, 1196–1200. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, D. Structural characterization and DPPH radical scavenging activity of an arabinoglucogalactan from Panax notoginseng root. J. Nat. Prod. 2008, 71, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Gu, L.; Zhong, Y.; Chen, Y.; Zhang, L.; Zhang, A.R.; Sobol, R.W.; Chen, T.; Li, J. Administration of polysaccharide from Panax notoginseng prolonged the survival of H22 tumor-bearing mice. Onco. Targets Ther. 2016, 9, 3433–3441. [Google Scholar]
- Wang, P.; Zhang, L.; Yao, J.; Shi, Y.; Li, P.; Ding, K. An arabinogalactan from flowers of Panax notoginseng inhibits angiogenesis by BMP2/Smad/Id1 signaling. Carbohydr. Polym. 2015, 121, 328–335. [Google Scholar] [CrossRef]
- Zhu, Y.; Pettolino, F.; Mau, S.L.; Shen, Y.C.; Chen, C.F.; Kuo, Y.C.; Bacic, A. Immunoactive polysaccharide-rich fractions from Panax notoginseng. Planta Med. 2006, 72, 1193–1199. [Google Scholar] [CrossRef]
- You, S.; Shi, X.; Yu, D.; Zhao, D.; An, Q.; Wang, D.; Zhang, J.; Li, M.; Wang, C. Fermentation of Panax notoginseng root extract polysaccharides attenuates oxidative stress and promotes type I procollagen synthesis in human dermal fibroblast cells. BMC Complement. Med. Ther. 2021, 21, 34. [Google Scholar] [CrossRef]
- Wang, C.; Zheng, L.; Liu, S.; Guo, X.; Qu, Y.; Gao, M.; Cui, X.; Yang, Y. A novel acidic polysaccharide from the residue of Panax notoginseng and its hepatoprotective effect on alcoholic liver damage in mice. Int. J. Biol. Macromol. 2020, 149, 1084–1097. [Google Scholar] [CrossRef]
- Jia, D.; Deng, Y.; Gao, J.; Liu, X.; Chu, J.; Shu, Y. Neuroprotective effect of Panax notoginseng plysaccharides against focal cerebral ischemia reperfusion injury in rats. Int. J. Biol. Macromol. 2014, 63, 177–180. [Google Scholar] [CrossRef]
- Zhu, B.; Xu, F.; Li, J.; Shuai, J.; Li, X.; Fang, W. Porcine circovirus type 2 explores the autophagic machinery for replication in PK-15 cells. Virus Res. 2012, 163, 476–485. [Google Scholar] [CrossRef]
- Gao, R.; Zhang, Y.; Kang, Y.; Xu, W.; Jiang, L.; Guo, T.; Huan, C. Glycyrrhizin Inhibits PEDV Infection and Proinflammatory Cytokine Secretion via the HMGB1/TLR4-MAPK p38 Pathway. Int. J. Mol. Sci. 2020, 21, 2961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huan, C.C.; Wang, H.X.; Sheng, X.X.; Wang, R.; Wang, X.; Mao, X. Glycyrrhizin inhibits porcine epidemic diarrhea virus infection and attenuates the proinflammatory responses by inhibition of high mobility group box-1 protein. Arch. Virol. 2017, 162, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huan, C.; Zhou, Z.; Yao, J.; Ni, B.; Gao, S. The Antiviral Effect of Panax Notoginseng Polysaccharides by Inhibiting PRV Adsorption and Replication In Vitro. Molecules 2022, 27, 1254. https://doi.org/10.3390/molecules27041254
Huan C, Zhou Z, Yao J, Ni B, Gao S. The Antiviral Effect of Panax Notoginseng Polysaccharides by Inhibiting PRV Adsorption and Replication In Vitro. Molecules. 2022; 27(4):1254. https://doi.org/10.3390/molecules27041254
Chicago/Turabian StyleHuan, Changchao, Ziyan Zhou, Jingting Yao, Bo Ni, and Song Gao. 2022. "The Antiviral Effect of Panax Notoginseng Polysaccharides by Inhibiting PRV Adsorption and Replication In Vitro" Molecules 27, no. 4: 1254. https://doi.org/10.3390/molecules27041254
APA StyleHuan, C., Zhou, Z., Yao, J., Ni, B., & Gao, S. (2022). The Antiviral Effect of Panax Notoginseng Polysaccharides by Inhibiting PRV Adsorption and Replication In Vitro. Molecules, 27(4), 1254. https://doi.org/10.3390/molecules27041254






