Gelatin Coating for the Improvement of Stability and Cell Uptake of Hydrophobic Drug-Containing Liposomes
Abstract
:1. Introduction
2. Material and Methods
2.1. Materials
2.2. Preparation of PTX-Encapsulating Plain Liposomes
2.3. Encapsulation Efficiency
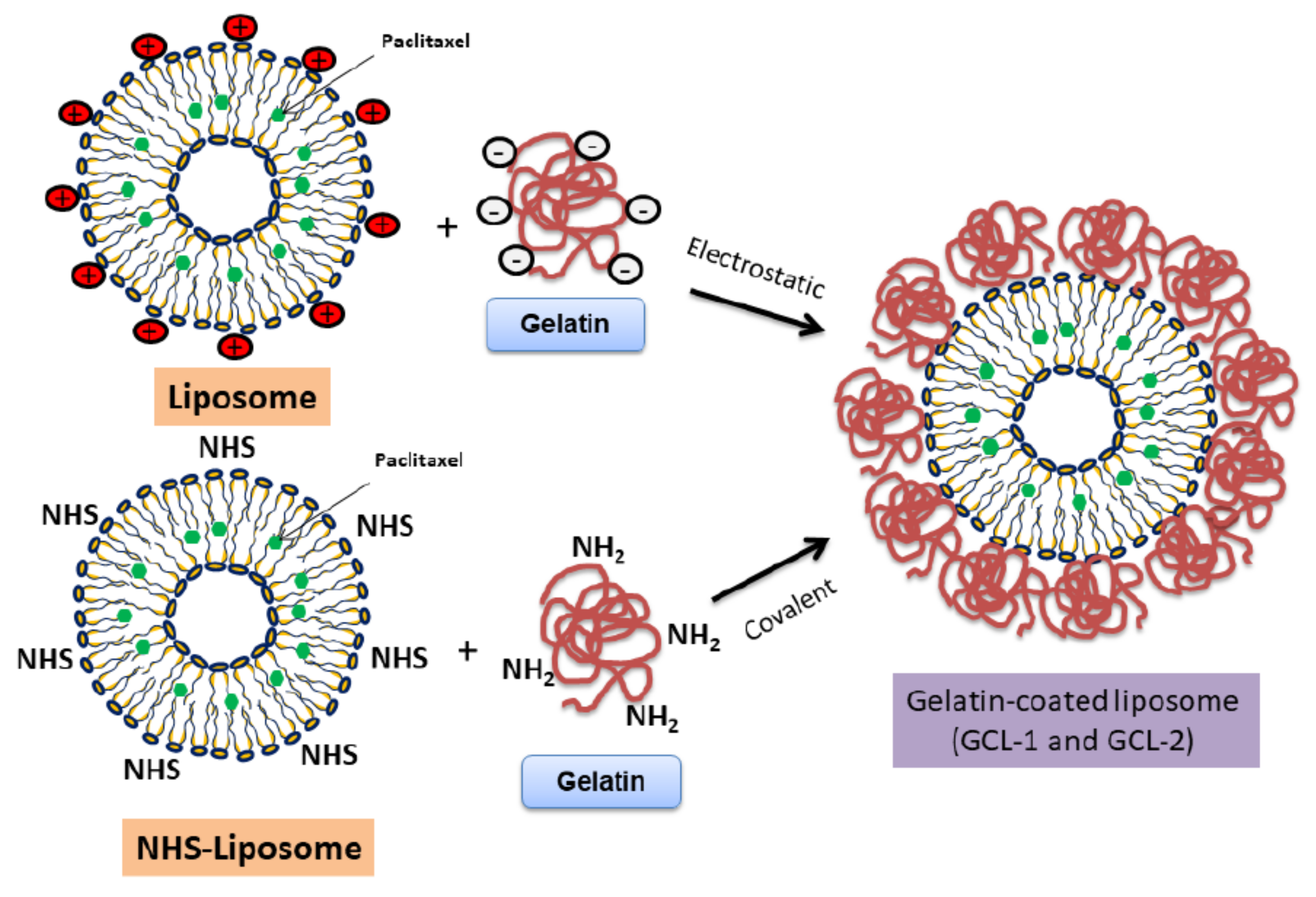
2.4. Fabrication of Gelatin-Coated Liposomes (GCL)
2.5. Differential Scanning Calorimetry (DSC)
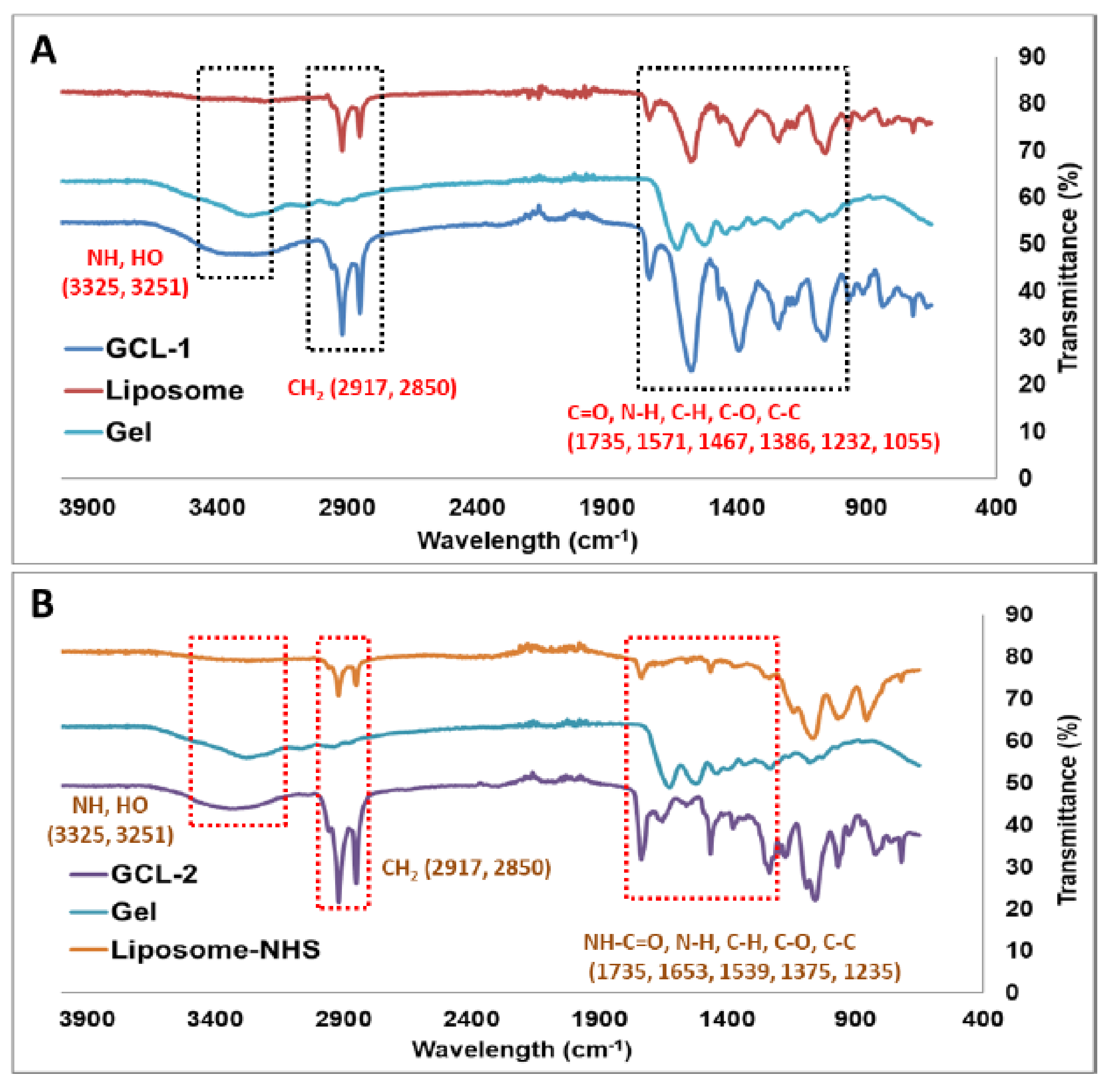
2.6. Fourier-Transform Infrared Spectroscopy (FT-IR)
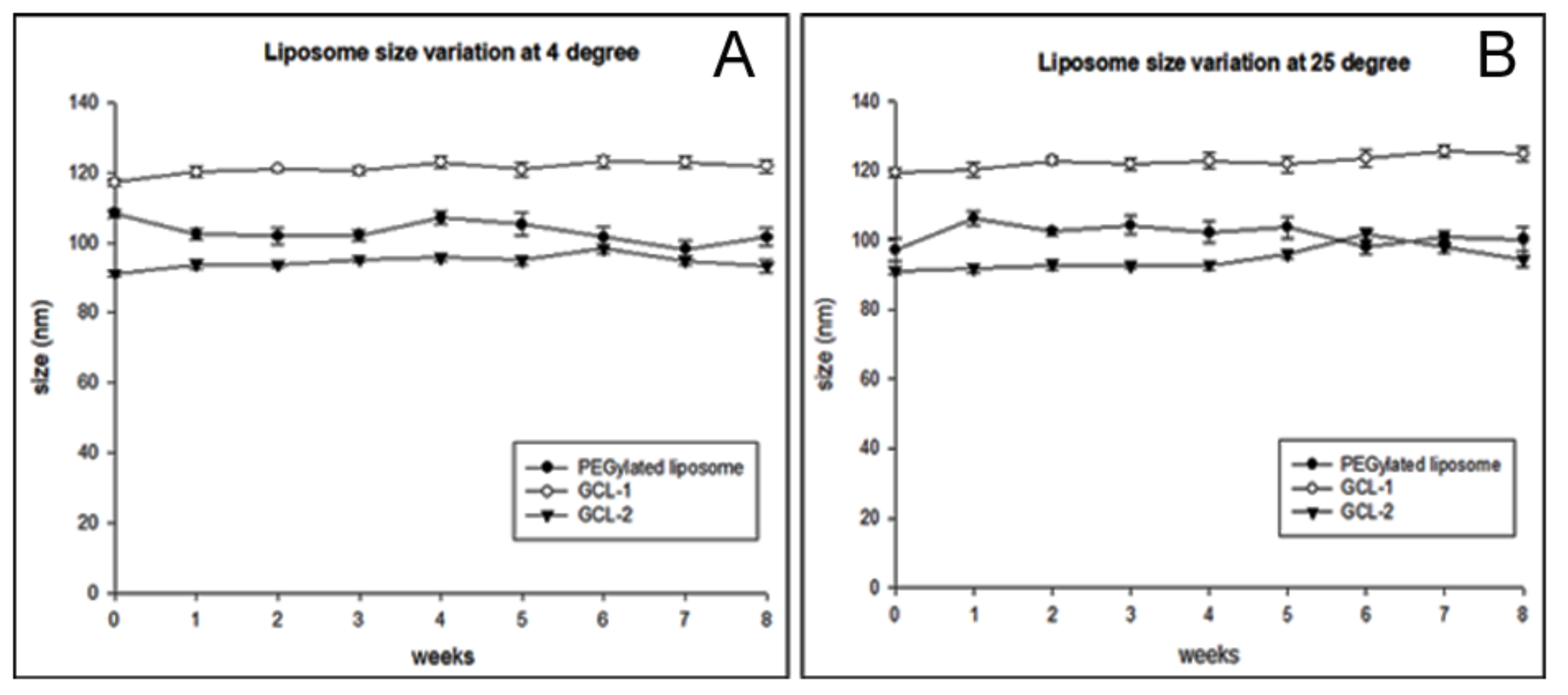
2.7. Physical Stability Analysis
2.8. In Vitro Release Study
2.9. Cell Culture and Cellular Uptake Study
2.10. Statistical Analysis
3. Results and Discussion
3.1. Preparation of PTX-Encapsulating Plain Liposomes
3.2. Fabrication of Gelatin-Coated Liposome (GCL)
3.3. Characterization of GCL-1 and GCL-2
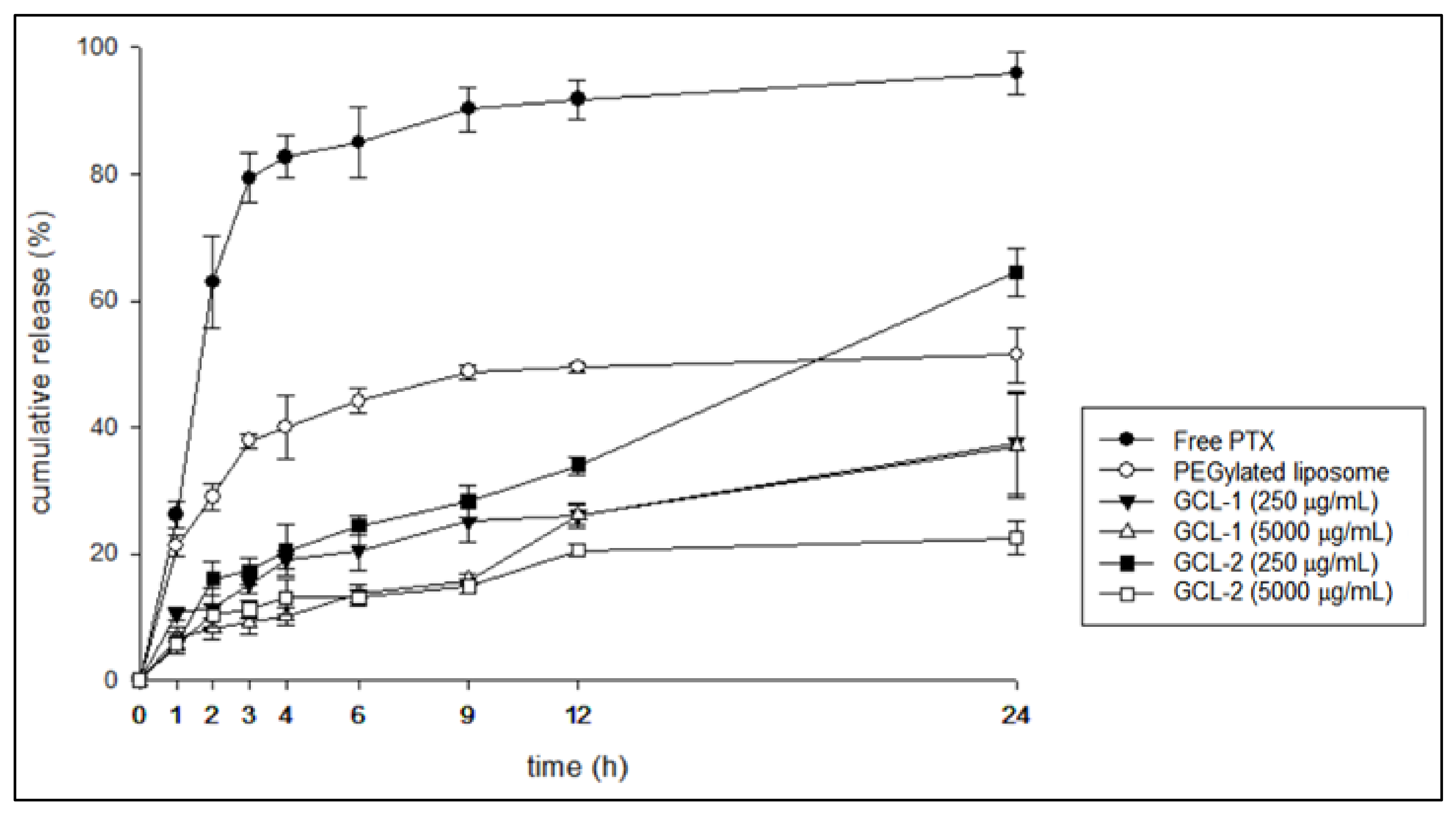
3.4. In Vitro Release Study
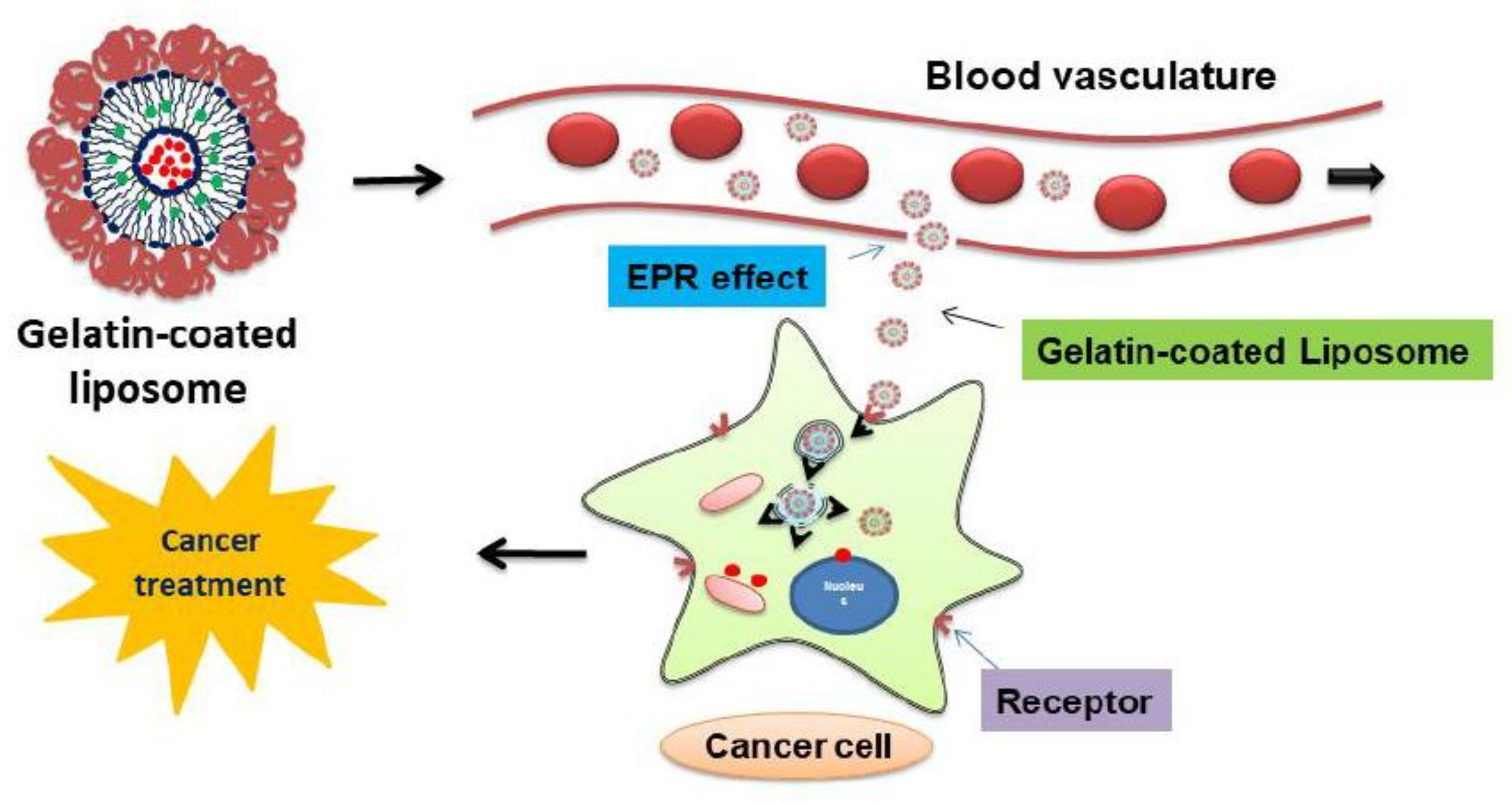
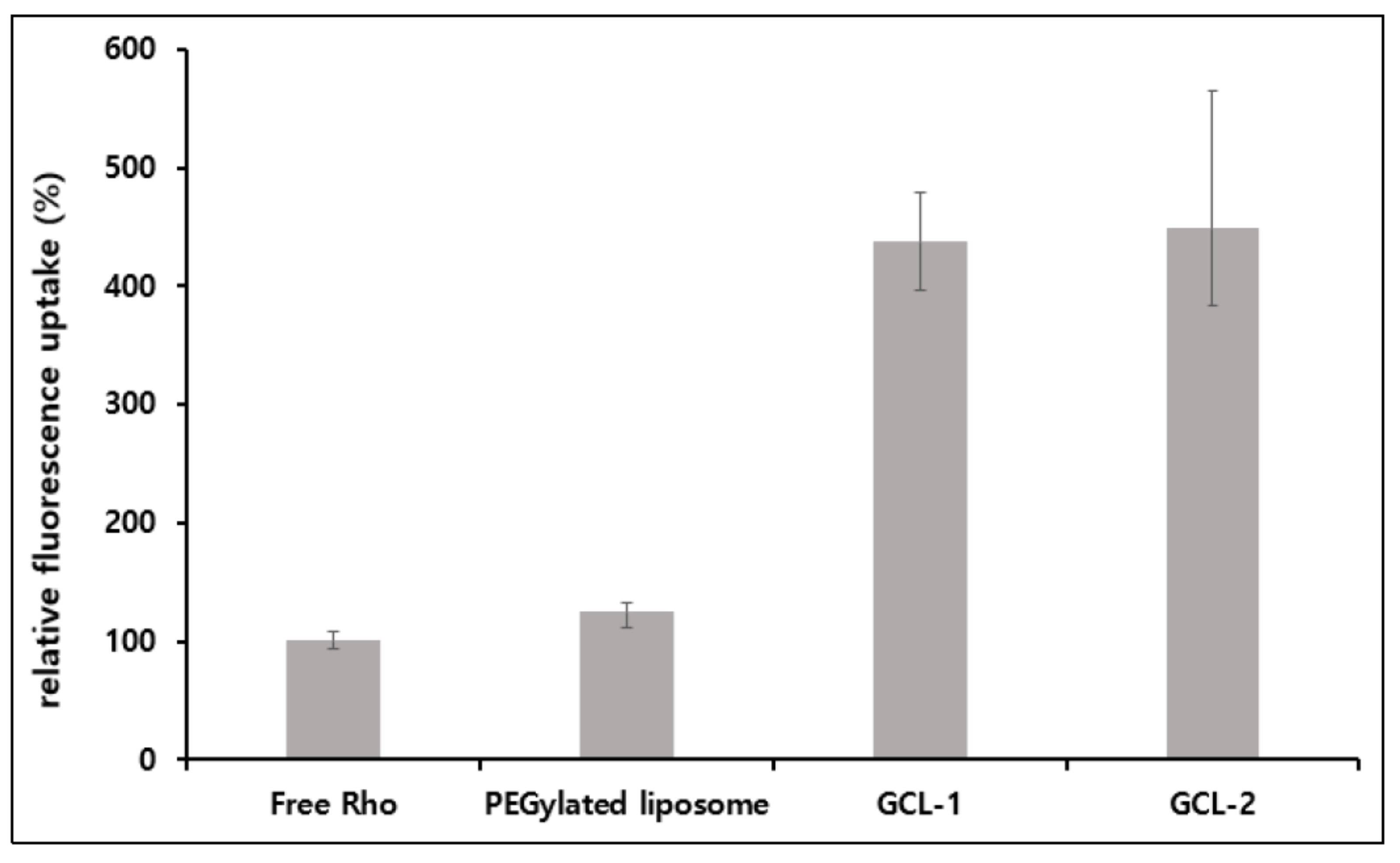
3.5. Cell Uptake Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Sample Availability
References
- Amarnath, S.; Urma, S.S. Liposomes in drug delivery; progress and limitations. Int. J. Pharm. 1997, 154, 123–140. [Google Scholar]
- Pandey, H.; Rani, R.; Agarwal, V. Liposome and Their Applications in Cancer Therapy. Braz. Arch. Biol. Technol. 2016, 59. [Google Scholar] [CrossRef] [Green Version]
- Nakhaei, P.; Margiana, R.; Bokov, D.O.; Abdelbasset, W.K.; Kouhbanani, M.A.J.; Varma, R.S.; Marofi, F.; Jarahian, M.; Beheshtkhoo, N. Liposomes: Structure, Biomedical Applications, and Stability Parameters With Emphasis on Cholesterol. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P.; Trubetskoy, V.S. Which polymers can make nanoparticulate drug carriers long-circulating? Adv. Drug Deliv. Rev. 1995, 16, 141–155. [Google Scholar] [CrossRef]
- Romberg, B.; Josber, M.M.; de Vringer, T.; Motonaga, K.; Kettenes-van den Bosch, J.J.; Oussoren, C.; Storm, G.; Hennink, W.E. Enzymatic Degradation of Liposome-Grafted Poly(hydroxyethyl L-glutamine). Bioconjug. Chem. 2005, 16, 767–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatakeyama, H.; Akita, H.; Kogure, K.; Oishi, M.; Nagasaki, Y.; Kihira, Y.; Ueno, M.; Kobayashi, H.; Kikuchi, H.; Harashima, H. Development of a novel systemic gene delivery system for cancer therapy with a tumor-specific cleavable PEG-lipid. Gene Ther. 2006, 14, 68–77. [Google Scholar] [CrossRef] [Green Version]
- Gomaa, A.I.; Martinent, C.; Hammami, R.; Fliss, I.; Subirade, M. Dual Coating of Liposomes as Encapsulating Matrix of Antimicrobial Peptides: Development and Characterization. Front. Chem. 2017, 5, 103. [Google Scholar] [CrossRef] [Green Version]
- Takeuchi, H.; Kojima, H.; Yamamoto, H.; Kawashima, Y. Polymer coating of liposomes with a modified polyvinyl alcohol and their systemic circulation and RES uptake in rats. J. Control. Release 2000, 68, 195–205. [Google Scholar] [CrossRef]
- Chen, K.; Di Sabatino, M.; Albertini, B.; Passerini, N.; Kett, V. The effect of polymer coatings on physicochemical properties of spray-dried liposomes for nasal delivery of BSA. Eur. J. Pharm. Sci. 2013, 50, 312–322. [Google Scholar] [CrossRef] [Green Version]
- Jung, S.H.; Kim, S.K.; Jung, S.H.; Kim, E.H.; Cho, S.H.; Jeong, K.-S.; Seong, H.; Shin, B.C. Increased stability in plasma and enhanced cellular uptake of thermally denatured albumin-coated liposomes. Colloids Surf. B Biointerfaces 2010, 76, 434–440. [Google Scholar] [CrossRef]
- Furumoto, K.; Yokoe, J.; Ogawara, K.; Amano, S.; Takaguchi, M.; Higaki, K.; Kai, T.; Kimura, T. Effect of coupling of albumin onto surface of PEG liposome on its in vivo disposition. Int. J. Pharm. 2007, 329, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Lohcharoenkal, W.; Wang, L.; Chen, Y.C.; Rojanasakul, Y. Protein Nanoparticles as Drug Delivery Carriers for Cancer Therapy. BioMed Res. Int. 2014, 2014, 180549. [Google Scholar] [CrossRef] [Green Version]
- Mohaddeseh, M.S. Strategies for surface modification of gelatin-based nanoparticles. Colloids Surf. B Biointerfaces 2019, 183, 110407. [Google Scholar]
- Morán, M.C.; Carazo, J.; Busquets, M.A. Dual responsive gelatin-based nanoparticles for enhanced 5-fluorouracil efficiency. Colloids Surf. B Biointerfaces 2018, 172, 646–654. [Google Scholar] [CrossRef] [PubMed]
- Nieberler, M.; Reuning, U.; Reichart, F.; Notni, J.; Wester, H.-J.; Schwaiger, M.; Weinmüller, M.; Räder, A.; Steiger, K.; Kessler, H. Exploring the Role of RGD-Recognizing Integrins in Cancer. Cancers 2017, 9, 116. [Google Scholar] [CrossRef]
- Sercombe, L.; Veerati, T.; Moheimani, F.; Wu, S.Y.; Sood, A.K.; Hua, S. Advances and Challenges of Liposome Assisted Drug Delivery. Front. Pharmacol. 2015, 6, 286. [Google Scholar] [CrossRef] [Green Version]
- Nikolić, V.; Savic, I.; Nikolić, L.; Stankovic, M.; Marinkovic, V. Paclitaxel as an anticancer agent: Isolation, activity, synthesis and stability. Open Med. 2011, 6, 527–536. [Google Scholar] [CrossRef]
- Ingle, S.G.; Pai, R.; Monpara, J.D.; Vavia, P.R. Liposils: An effective strategy for stabilizing Paclitaxel loaded liposomes by surface coating with silica. Eur. J. Pharm. Sci. 2018, 122, 51–63. [Google Scholar] [CrossRef]
- Luo, L.-M.; Huang, Y.; Zhao, B.-X.; Zhao, X.; Duan, Y.; Du, R.; Yu, K.-F.; Song, P.; Zhao, Y.; Zhang, X.; et al. Anti-tumor and anti-angiogenic effect of metronomic cyclic NGR-modified liposomes containing paclitaxel. Biomaterials 2013, 34, 1102–1114. [Google Scholar] [CrossRef]
- Yang, T.; Cui, F.-D.; Choi, M.-K.; Lin, H.; Chung, S.-J.; Shim, C.-K.; Kim, D.-D. Liposome Formulation of Paclitaxel with Enhanced Solubility and Stability. Drug Deliv. 2007, 14, 301–308. [Google Scholar] [CrossRef]
- Crosasso, P.; Ceruti, M.; Brusa, P.; Arpicco, S.; Dosio, F.; Cattel, L. Preparation, characterization and properties of sterically stabilized paclitaxel-containing liposomes. J. Control. Release 2000, 63, 19–30. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, J.; Zhang, H.; Liu, D.; Ming, L.; Liu, L.; Dong, Y.; Jian, B.; Cai, D. The anticancer efficacy of paclitaxel liposomes modified with low-toxicity hydrophobic cell-penetrating peptides in breast cancer: An in vitro and in vivo evaluation. RSC Adv. 2018, 8, 24084–24093. [Google Scholar] [CrossRef] [Green Version]
- Battogtokh, G.; Kang, J.H.; Ko, Y.T. Long-circulating self-assembled cholesteryl albumin nanoparticles enhance tumor accumulation of hydrophobic anticancer drug. Eur. J. Pharm. Biopharm. 2015, 96, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.-S.; Yiao, S.-Y.; Lim, K.; Jensen, R.V.; Hsiao, L.-L. Interpretation of biological and mechanical variations between the Lowry versus Bradford method for protein quantification. N. Am. J. Med. Sci. 2010, 2, 325–328. [Google Scholar] [PubMed]
- Liu, Y.; Ran, R.; Chen, J.; Kuang, Q.; Tang, J.; Mei, L.; Zhang, Q.; Gao, H.; Zhang, Z.; He, Q. Paclitaxel loaded liposomes decorated with a multifunctional tandem peptide for glioma targeting. Biomaterials 2014, 35, 4835–4847. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, Y.; Wang, W.; Liu, K. Effect of integrin receptor-targeted liposomal paclitaxel for hepatocellular carcinoma targeting and therapy. Oncol. Lett. 2015, 10, 77–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, S.; Kumar, D.; Swarnakar, N.K.; Thanki, K. Polyelectrolyte stabilized multilayered liposomes for oral delivery of paclitaxel. Biomaterials 2012, 33, 6758–6768. [Google Scholar] [CrossRef]
- Koudelka, S.; Turánek, J. Liposomal paclitaxel formulations. J. Control. Release 2012, 163, 322–334. [Google Scholar] [CrossRef]
- Nurul, A.G.; Sarbon, N.M. Effects of pH on functional, rheological and structural properties of eel (Monopterus sp.) skin gelatin compared to bovine gelatin. Int. Food Res. J. 2015, 22, 572–583. [Google Scholar]
- Nanda, J.S.; Lorsch, J.R. Labeling a Protein with Fluorophores Using NHS Ester Derivitization. Methods Enzym. 2014, 536, 87–94. [Google Scholar] [CrossRef]
- Geyik, C.; Ciftci, M.; Demir, B.; Guler, B.; Ozkaya, A.B.; Gumus, Z.P.; Barlas, F.B.; Demirkol, D.O.; Coskunol, H.; Timur, S.; et al. Controlled release of anticancer drug Paclitaxel using nano-structured amphiphilic star-hyperbranched block copolymers. Polym. Chem. 2015, 6, 5470–5477. [Google Scholar] [CrossRef]
- Cacua, K.; Ordoñez, F.; Zapata, J.C.; Herrera, B.; Pabón, E.; Buitrago-Sierra, R. Surfactant concentration and pH effects on the zeta potential values of alumina nanofluids to inspect stability. Colloids Surfaces A Physicochem. Eng. Asp. 2019, 583, 123960. [Google Scholar] [CrossRef]
- Mohanraj, V.J.; Barnes, T.J.; Prestidge, C.A. Silica nanoparticle coated liposomes: A new type of hybrid nanocapsule for proteins. Int. J. Pharm. 2010, 392, 285–293. [Google Scholar] [CrossRef]
- Hillson, P.J. Gelatin as a Protective Colloid. Nature 1962, 195, 1298–1299. [Google Scholar] [CrossRef]
- Mekuria, S.L.; Debele, T.A.; Chou, H.-Y.; Tsai, H.-C. IL-6 Antibody and RGD Peptide Conjugated Poly(amidoamine) Dendrimer for Targeted Drug Delivery of HeLa Cells. J. Phys. Chem. B. 2016, 120, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Bellotti, E.; Cascone, M.G.; Barbani, N.; Rossin, D.; Rastaldo, R.; Giachino, C.; Cristallini, C. Targeting Cancer Cells Overexpressing Folate Receptors with New Terpolymer-Based Nanocapsules: Toward a Novel Targeted DNA Delivery System for Cancer Therapy. Biomedicines 2021, 9, 1275. [Google Scholar] [CrossRef]

| PEGylated Liposomes | GCL-1 | GCL-2 | |
|---|---|---|---|
| Lipid 1 | HSPC 50 mg | HSPC 50 mg | HSPC 50 mg |
| Lipid 2 | DSPE-PEG2k 14 mg | Stearylamine 3.23 mg | DSPE-NHS 10.4 mg |
| Cholesterol | 17.2 mg | 15.8 mg | 15.8 mg |
| PTX | 4 mg | 4 mg | 4 mg |
| Molar ratio (Lipid 1:Lipid 2:Chol) | 57:4:39 | 55:10:35 | 55:10:35 |
| PEGylated Liposomes | GCL-1 | GCL-2 | |
|---|---|---|---|
| Size (nm) | 113.3 ± 1.7 | 115.7 ± 5.2 | 105.0 ± 3.1 |
| Polydispersity index | 0.241 | 0.250 | 0.250 |
| Zeta potential (mV) | 0.72 ± 0.14 | 21.7 ± 2.9 | −28.1 ± 0.5 |
| Encapsulation efficiency (%) | 82.5 ± 2.5 | 92.0 ± 5 | 92.7 ± 5.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Battogtokh, G.; Joo, Y.; Abuzar, S.M.; Park, H.; Hwang, S.-J. Gelatin Coating for the Improvement of Stability and Cell Uptake of Hydrophobic Drug-Containing Liposomes. Molecules 2022, 27, 1041. https://doi.org/10.3390/molecules27031041
Battogtokh G, Joo Y, Abuzar SM, Park H, Hwang S-J. Gelatin Coating for the Improvement of Stability and Cell Uptake of Hydrophobic Drug-Containing Liposomes. Molecules. 2022; 27(3):1041. https://doi.org/10.3390/molecules27031041
Chicago/Turabian StyleBattogtokh, Gantumur, Yechan Joo, Sharif Md Abuzar, Heejun Park, and Sung-Joo Hwang. 2022. "Gelatin Coating for the Improvement of Stability and Cell Uptake of Hydrophobic Drug-Containing Liposomes" Molecules 27, no. 3: 1041. https://doi.org/10.3390/molecules27031041
APA StyleBattogtokh, G., Joo, Y., Abuzar, S. M., Park, H., & Hwang, S.-J. (2022). Gelatin Coating for the Improvement of Stability and Cell Uptake of Hydrophobic Drug-Containing Liposomes. Molecules, 27(3), 1041. https://doi.org/10.3390/molecules27031041








