Tuning the Charge Transport in Nickel Salicylaldimine Polymers by the Ligand Structure
Abstract
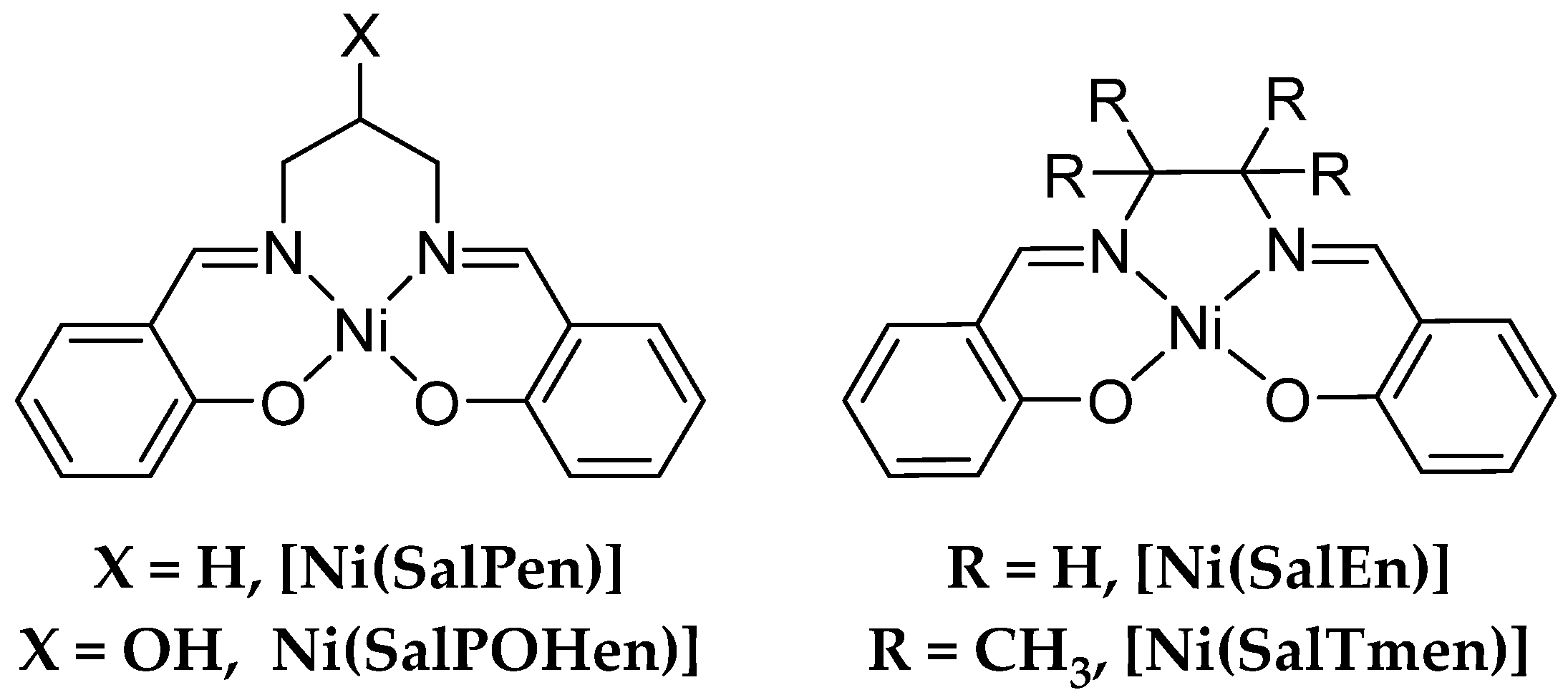
1. Introduction
2. Results and Discussion
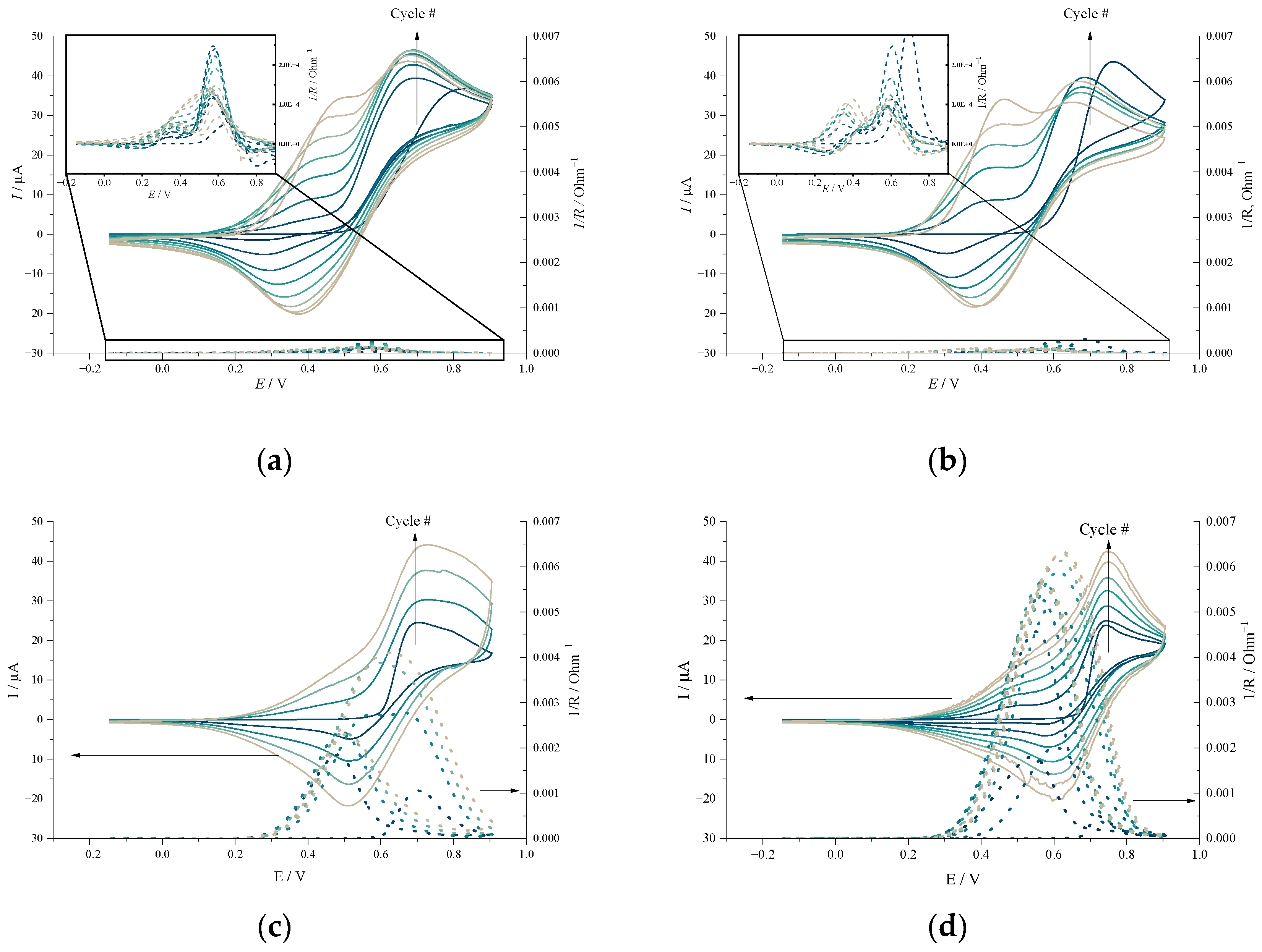
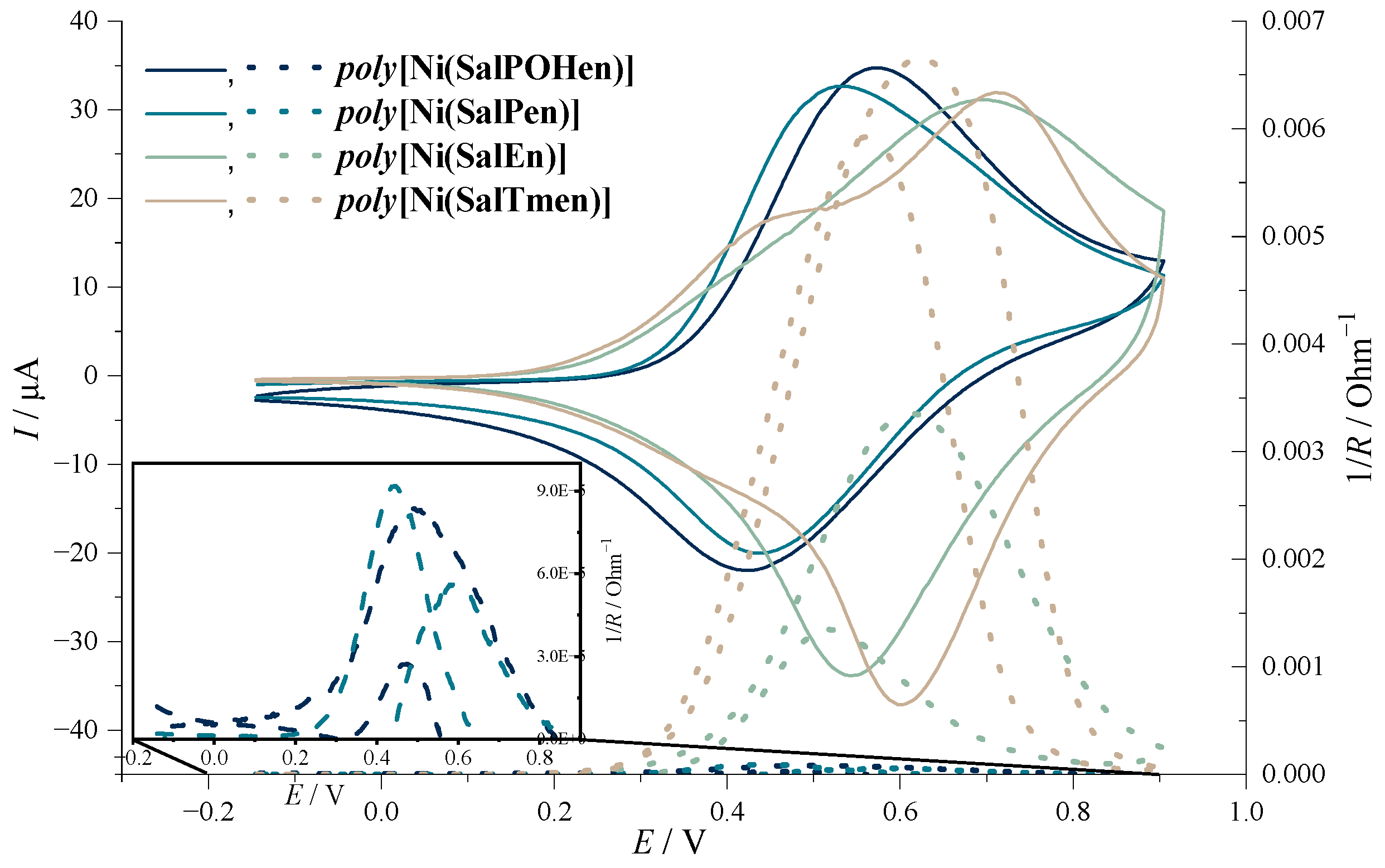
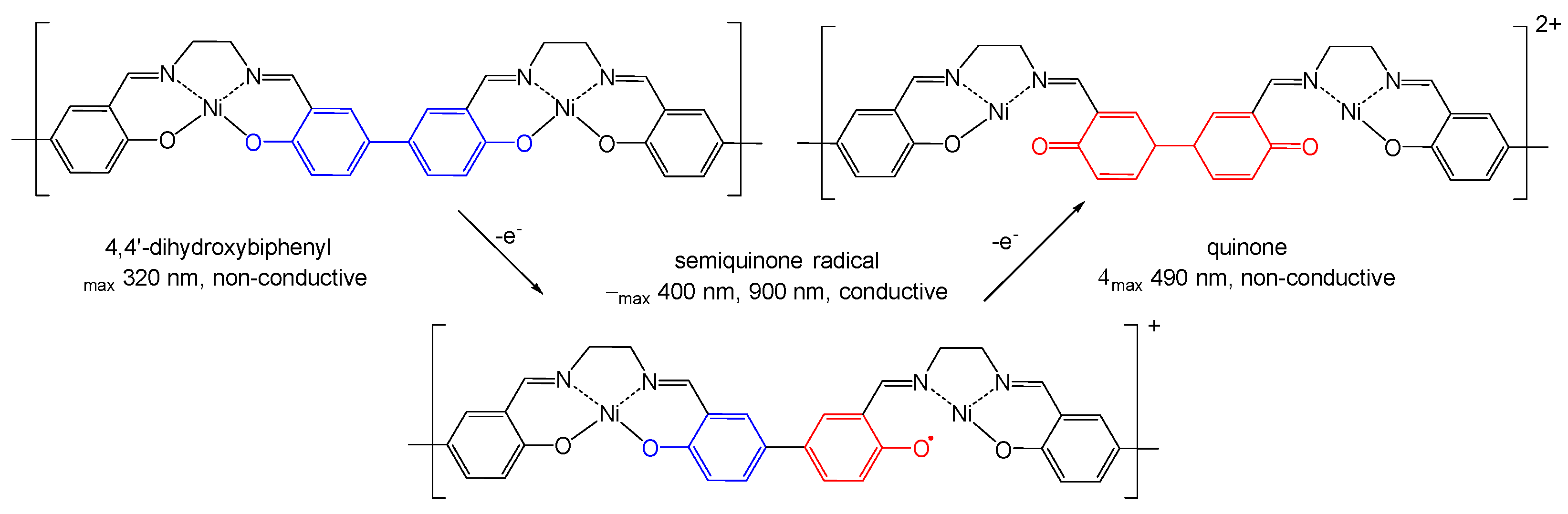
2.1. Electrochemical Studies
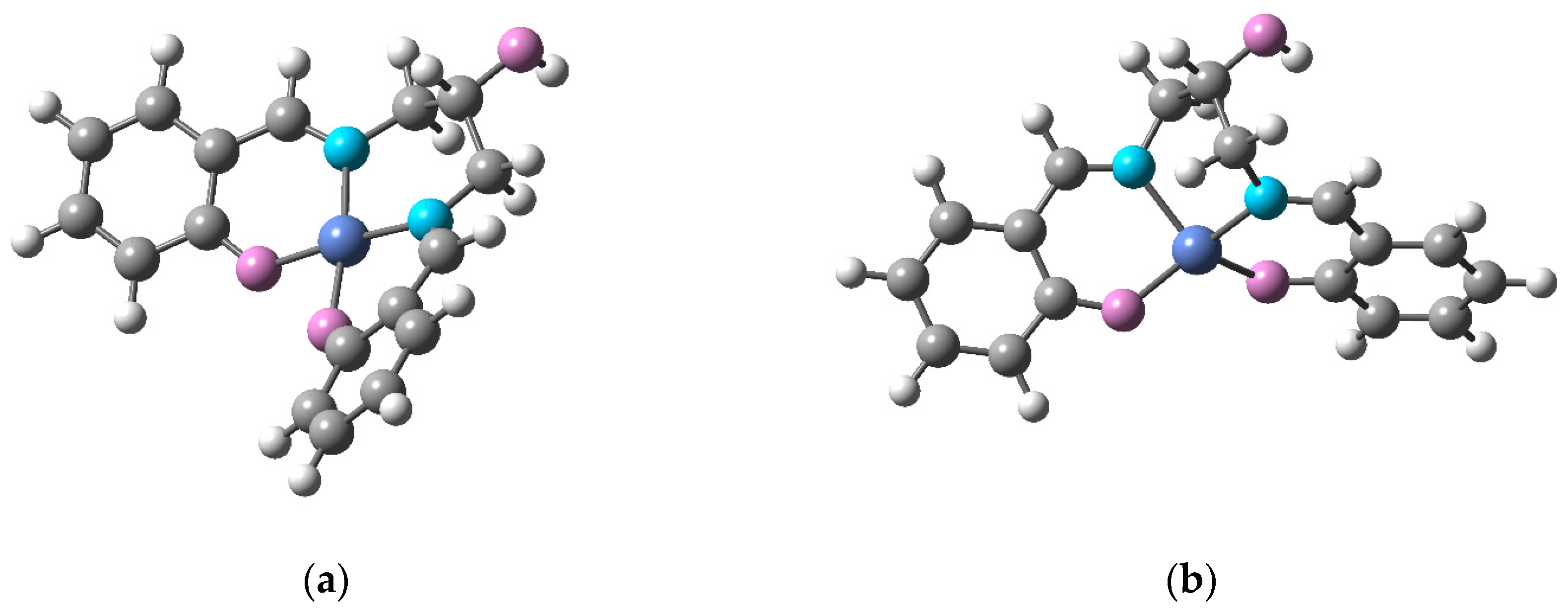
2.2. Crystal Structure
2.3. UV-Vis Spectroscopy
2.4. DFT Calculations
3. Conclusions
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, X.; Li, J.; Deng, F.; Kang, F. Enhanced electrochemical performance of nitrogen-doped graphene and poly[Ni(salen)] composite electrodes for supercapacitors. Ionics 2018, 24, 3143–3153. [Google Scholar] [CrossRef]
- Deng, F.; Li, X.; Ding, F.; Niu, B.; Li, J. Pseudocapacitive Energy Storage in Schiff Base Polymer with Salphen-Type Ligands. J. Phys. Chem. C 2018, 122, 5325–5333. [Google Scholar] [CrossRef]
- Alekseeva, E.V.; Chepurnaya, I.A.; Malev, V.V.; Timonov, A.M.; Levin, O.V. Polymeric nickel complexes with salen-type ligands for modification of supercapacitor electrodes: Impedance studies of charge transfer and storage properties. Electrochim. Acta 2017, 225, 378–391. [Google Scholar] [CrossRef]
- Zhu, Z.; Lu, J.; Li, X.; Xu, G.; Chen, C.; Li, J. Effects of Potential Modes on Performances of Electrodeposited Poly[Ni(salen)]/MWCNTs Composite as Supercapacitor Electrode Material. Electrochemistry 2016, 84, 427–431. [Google Scholar] [CrossRef]
- Yan, G.; Li, J.; Zhang, Y.; Gao, F.; Kang, F. Electrochemical Polymerization and Energy Storage for Poly[Ni(salen)] as Supercapacitor Electrode Material. J. Phys. Chem. C 2014, 118, 9911–9917. [Google Scholar] [CrossRef]
- Eliseeva, S.N.; Alekseeva, E.V.; Vereshchagin, A.A.; Volkov, A.I.; Vlasov, P.S.; Konev, A.S.; Levin, O.V. Nickel-Salen Type Polymers as Cathode Materials for Rechargeable Lithium Batteries. Macromol. Chem. Phys. 2017, 218, 1700361. [Google Scholar] [CrossRef]
- Vereshchagin, A.A.; Lukyanov, D.A.; Kulikov, I.R.; Panjwani, N.A.; Alekseeva, E.A.; Behrends, J.; Levin, O.V. The Fast and the Capacious: A [Ni(Salen)]-TEMPO Redox-Conducting Polymer for Organic Batteries. Batter. Supercaps 2020, 4, 336–346. [Google Scholar] [CrossRef]
- Kulikov, I.; Panjwani, N.A.; Vereshchagin, A.A.; Spallek, D.; Lukianov, D.A.; Alekseeva, E.V.; Levin, O.V.; Behrends, J. Spins at work: Probing charging and discharging of organic radical batteries by electron paramagnetic resonance spectroscopy. Energy Environ. Sci. 2022, 15, 3275–3290. [Google Scholar] [CrossRef]
- Vereshchagin, A.A.; Vlasov, P.S.; Konev, A.S.; Yang, P.; Grechishnikova, G.A.; Levin, O.V. Novel highly conductive cathode material based on stable-radical organic framework and polymerized nickel complex for electrochemical energy storage devices. Electrochim. Acta 2019, 295, 1075–1084. [Google Scholar] [CrossRef]
- O’Meara, C.; Karushev, M.P.; Polozhentceva, I.A.; Dharmasena, S.; Cho, H.; Yurkovich, B.J.; Kogan, S.; Kim, J.-H. Nickel–Salen-Type Polymer as Conducting Agent and Binder for Carbon-Free Cathodes in Lithium-Ion Batteries. ACS Appl. Mater. Interfaces 2019, 11, 525–533. [Google Scholar] [CrossRef]
- Konev, A.S.; Kayumov, M.Y.; Karushev, M.P.; Novoselova, Y.V.; Lukyanov, D.A.; Alekseeva, E.V.; Levin, O.V. Polymeric Metal Salen-Type Complexes as Catalysts for Photoelectrocatalytic Hydrogen Peroxide Production. ChemElectroChem 2018, 5, 3138–3142. [Google Scholar] [CrossRef]
- Petrov, A.; Lukyanov, D.; Kopytko, O.; Novoselova, J.; Alekseeva, E.; Levin, O. Inversion of the Photogalvanic Effect of Conductive Polymers by Porphyrin Dopants. Catalysts 2021, 11, 729. [Google Scholar] [CrossRef]
- Łępicka, K. Pieta, P., Shkurenko, A., Borowicz, P., Majewska, M., Rosenkranz, M., Avdoshenko, S., Popov, A.A., Kutner, W. Spectroelectrochemical Approaches to Mechanistic Aspects of Charge Transport in meso-Nickel(II) Schiff Base Electrochromic Polymer. J. Phys. Chem. C 2017, 121, 16710–16720. [Google Scholar] [CrossRef]
- Nunes, M.; Araújo, M.; Fonseca, J.; Moura, C.; Hillman, R.; Freire, C. High-Performance Electrochromic Devices Based on Poly[Ni(salen)]-Type Polymer Films. ACS Appl. Mater. Interfaces 2016, 8, 14231–14243. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.F.; Olean-Oliveira, A.; David-Parra, D.N.; Teixeira, M.F. A chemiresistor sensor based on a cobalt(salen) metallopolymer for dissolved molecular oxygen. Talanta 2018, 190, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Martins, T.S.; Bott-Neto, J.L.; Raymundo-Pereira, P.A.; Ticianelli, E.A.; Machado, S.A.S. An electrochemical furosemide sensor based on pencil graphite surface modified with polymer film Ni-salen and Ni(OH)2/C nanoparticles. Sens. Actuators B Chem. 2018, 276, 378–387. [Google Scholar] [CrossRef]
- Raymundo-Pereira, P.A.; Teixeira, M.F.; Fatibello-Filho, O.; Dockal, E.R.; Bonifácio, V.G.; Marcolino-Junior, L.H. Electrochemical sensor for ranitidine determination based on carbon paste electrode modified with oxovanadium (IV) salen complex. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 4081–4085. [Google Scholar] [CrossRef]
- Beletskii, E.; Fedorova, A.; Lukyanov, D.; Kalnin, A.; Ershov, V.; Danilov, S.; Spiridonova, D.; Alekseeva, E.; Levin, O. Switchable resistance conducting-polymer layer for Li-ion battery overcharge protection. J. Power Sources 2021, 490, 229548. [Google Scholar] [CrossRef]
- Beletskii, E.V.; Alekseeva, E.V.; Levin, O.V. Variable-resistance materials for lithium-ion batteries. Russ. Chem. Rev. 2022, 91, RCR5030. [Google Scholar] [CrossRef]
- Beletskii, E.V.; Volosatova, Y.A.; Eliseeva, S.N.; Levin, O.V. The Effect of Electrode Potential on the Conductivity of Polymer Complexes of Nickel with Salen Ligands. Russ. J. Electrochem. 2019, 55, 339–345. [Google Scholar] [CrossRef]
- Chepurnaya, I.A.; Karushev, M.P.; Alekseeva, E.V.; Lukyanov, D.A.; Levin, O.V. Redox-conducting polymers based on metal-salen complexes for energy storage applications. Pure Appl. Chem. 2020, 92, 1239–1258. [Google Scholar] [CrossRef]
- Vereschagin, A.A.; Sizov, V.V.; Vlasov, P.S.; Alekseeva, E.V.; Konev, A.S.; Levin, O.V. Water-stable [Ni(salen)]-type electrode material based on phenylazosubstituted salicylic aldehyde imine ligand. New J. Chem. 2017, 41, 13918–13928. [Google Scholar] [CrossRef]
- Audebert, P.; Capdevielle, P.; Maumy, M. Redox and Conducting Polymers Based on Salen-Type Metal Units—Electrochemical Study and Some Characteristics. New J. Chem. 1992, 16, 697–703. [Google Scholar]
- Yankin, A.N.; Lukyanov, D.A.; Beletskii, E.V.; Bakulina, O.Y.; Vlasov, P.S.; Levin, O.V. Aryl-Aryl Coupling of Salicylic Aldehydes through Oxidative CH-activation in Nickel Salen Derivatives. ChemistrySelect 2019, 4, 8886–8890. [Google Scholar] [CrossRef]
- Karushev, M.P.; Alekseeva, E.V.; Polozhentseva, J.A. Semiconductor Properties of Polymer Films Based on Nickel Complex with Salen-type Ligand. Phys. Solid State 2022, 1, 62. [Google Scholar]
- Dmitrieva, E.; Rosenkranz, M.; Danilova, J.S.; Smirnova, E.A.; Karushev, M.P.; Chepurnaya, I.A.; Timonov, A.M. Radical formation in polymeric nickel complexes with N2O2 Schiff base ligands: An in situ ESR and UV–vis–NIR spectroelectrochemical study. Electrochim. Acta 2018, 283, 1742–1752. [Google Scholar] [CrossRef]
- Malev, V.; Levin, O.; Timonov, A. Quasi-equilibrium voltammetric curves resulting from the existence of two immobile charge carriers within electroactive polymer films. Electrochim. Acta 2013, 108, 313–320. [Google Scholar] [CrossRef]
- Heinze, J.; Frontana-Uribe, B.A.; Ludwigs, S. Electrochemistry of Conducting Polymers—Persistent Models and New Concepts. Chem. Rev. 2010, 110, 4724–4771. [Google Scholar] [CrossRef]
- Ofer, D.; Crooks, R.M.; Wrighton, M.S. Potential dependence of the conductivity of highly oxidized polythiophenes, polypyrroles, and polyaniline: Finite windows of high conductivity. J. Am. Chem. Soc. 1990, 112, 7869–7879. [Google Scholar] [CrossRef]
- Shimazaki, Y.; Stack, T.D.P.; Storr, T. Detailed evaluation of the geometric and electronic structures of one-electron oxidized group 10 (Ni, Pd, and Pt) metal(II)-(disalicylidene)diamine complexes. Inorg. Chem. 2009, 48, 8383–8392. [Google Scholar] [CrossRef]
- D’Alessandro, D.M.; Keene, F.R. Current trends and future challenges in the experimental, theoretical and computational analysis of intervalence charge transfer (IVCT) transitions. Chem. Soc. Rev. 2006, 35, 424–440. [Google Scholar] [CrossRef] [PubMed]
- Drew, M.G.B.; Prasad, R.N.; Sharma, R.P. Structures of (N,N’-trimethylenedisalicylideneaminato)nickel(II) (1) and (N,N’-trimethylenedisalicylideneaminato)copper(II) (2). Acta Crystallogr. Sect. C Cryst. Struct. Commun. 1985, 41, 1755–1758. [Google Scholar] [CrossRef]
- Lutz, M. [N,N′-Ethylenebis(salicylideneiminato)]nickel(II) dimethylformamide solvate. Acta Crystallogr. Sect. E Struct. Rep. Online 2003, 59, m950–m952. [Google Scholar] [CrossRef]
- Santos, I.; Vilas-Boas, M.; Piedade, M.F.; Freire, C.; Duarte, M.T.; de Castro, B. Electrochemical and X-ray studies of nickel(II) Schiff base complexes derived from salicylaldehyde: Structural effects of bridge substituents on the stabilisation of the +3 oxidation state. Polyhedron 2000, 19, 655–664. [Google Scholar] [CrossRef]
- Bosnich, B. An interpretation of the circular dichroism and electronic spectra of salicylaldimine complexes of square-coplanar diamagnetic nickel(II). J. Am. Chem. Soc. 1968, 90, 627–632. [Google Scholar] [CrossRef]
- Vilas-Boas, M.; Freire, C.; de Castro, B.; Christensen, A.P.A.; Hillman, A.R. New Insights into the Structure and Properties of Electroactive Polymer Films Derived from [Ni(salen)]. Inorg. Chem. 1997, 36, 4919–4929. [Google Scholar] [CrossRef]
- Clarke, R.M.; Herasymchuk, K.; Storr, T. Electronic structure elucidation in oxidized metal–salen complexes. Coord. Chem. Rev. 2017, 352, 67–82. [Google Scholar] [CrossRef]
- Sizov, V.V.; Novozhilova, M.V.; Alekseeva, E.V.; Karushev, M.P.; Timonov, A.M.; Eliseeva, S.N.; Vanin, A.A.; Malev, V.V.; Levin, O.V. Redox transformations in electroactive polymer films derived from complexes of nickel with SalEn-type ligands: Computational, EQCM, and spectroelectrochemical study. J. Solid State Electrochem. 2014, 19, 453–468. [Google Scholar] [CrossRef]
- Rao, P.S.; Hayon, E. Ionization constants and spectral characteristics of some semiquinone radicals in aqueous solution. J. Phys. Chem. 1973, 77, 2274–2276. [Google Scholar] [CrossRef]
- Simpson, S.H.; Richardson, R.M.; Hanna, S. Influence of dye molecules on the birefringence of liquid crystal mixtures at near infrared frequencies. J. Chem. Phys. 2007, 127, 104901. [Google Scholar] [CrossRef]
- Atakol, O.; Durmus, S.; Durmus, Z.; Arici, C.; Çiçek, B. Investigations on Some Hetero-Trinuclear Complexes of Nickel(Ii) and Copper(Ii). Synth. React. Inorg. Met. Nano-Metal Chem. 2001, 31, 1689–1704. [Google Scholar] [CrossRef]
- Dieng, M.; Elhadji, T.I.; Gaye, M.; Sall, A.S..; Barry, A.H. Synthesis, crystal structures and spectroscopic properties of a trinuclear [Cu-3(HL)(2)(NO3)(2)](H2O)(CH2CH2OH) complex and a [Mn(HL)(CH3COO)](n) polymer with H3L=N,N ‘-(2-hydroxypropane-1,3-diyl)-bis-(salicylaldimine). Acta Chim. Slov. 2006, 53, 417–423. [Google Scholar]
- Apraksin, R.; Volosatova, Y.; Volkov, A.; Vlasov, P.; Lukyanov, D.; Kulikov, I.; Eliseeva, S.; Levin, O. Electrochemical synthesis and characterization of poly [Ni(CH3Osalen)] with immobilized poly(styrenesulfonate) anion dopants. Electrochim. Acta 2021, 368, 137637. [Google Scholar] [CrossRef]
- Yanai, T.; Tew, D.; Handy, N.C. A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104–154119. [Google Scholar] [CrossRef]
- Martin, R.L. Natural transition orbitals. J. Chem. Phys. 2003, 118, 4775–4777. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16 Rev. A.03; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef]
- Dennington, R.D.; TKeith, A.; Millam, J.M. GaussView; Version 5; Semichem Inc.: Shawnee Mission, KS, USA, 2009. [Google Scholar]
- Palatinus, L.; Gervais, C. SUPERFLIP–a computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J. Appl. Cryst. 2007, 40, 786–790. [Google Scholar] [CrossRef]
- Palatinus, L.; van der Lee, A. Symmetry determination following structure solution in P1. J. Appl. Cryst. 2008, 41, 975–984. [Google Scholar] [CrossRef]
- Palatinus, L.; Prathapa, S.J.; van Smaalen, S. EDMA: A computer program for topological analysis of discrete electron densities. J. Appl. Cryst. 2012, 45, 575–580. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, 71, 3–8. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
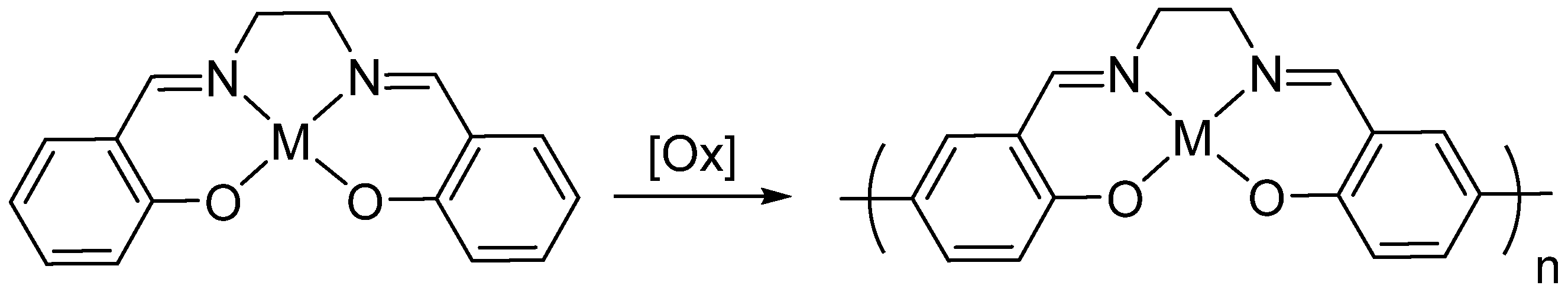
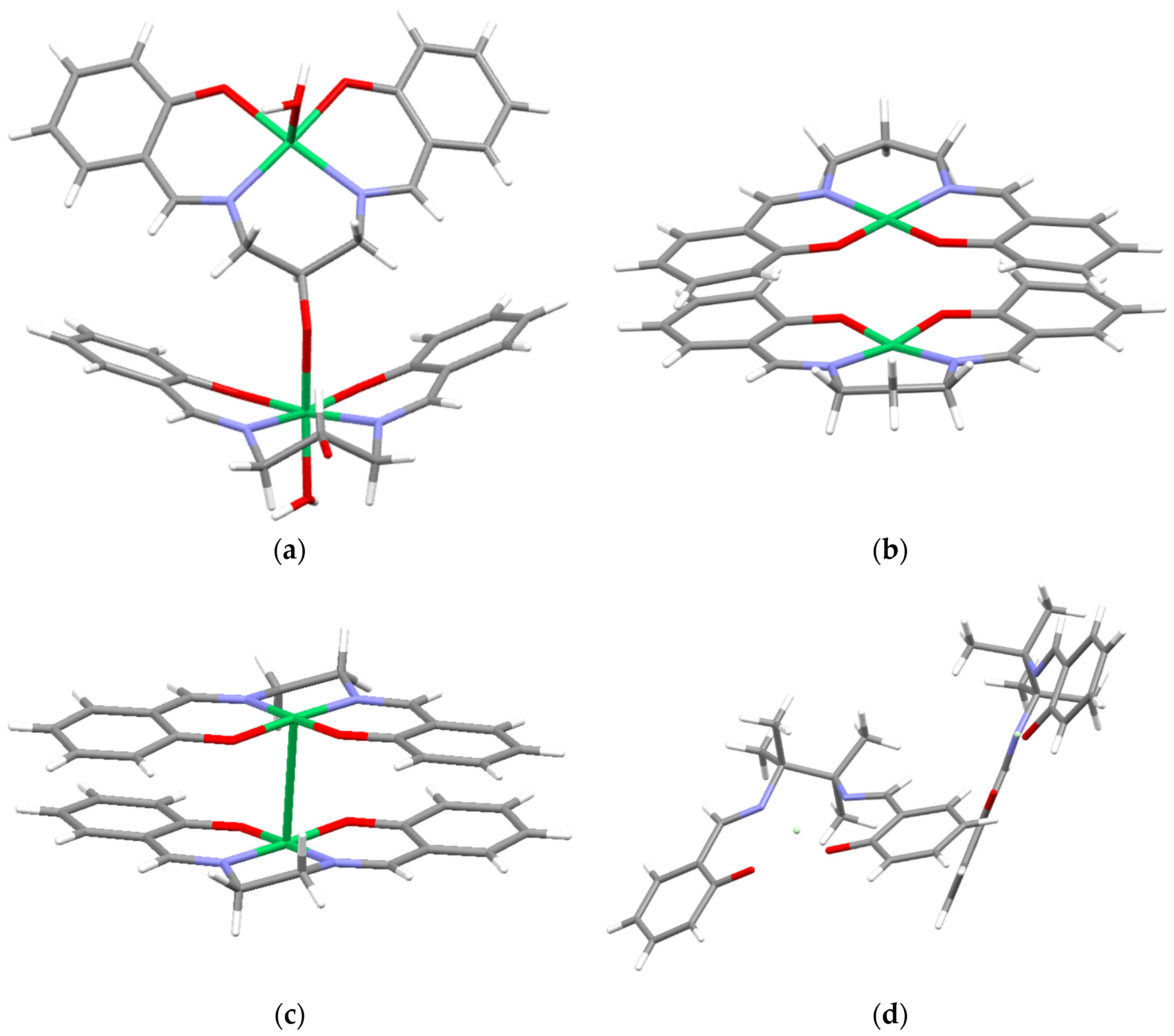
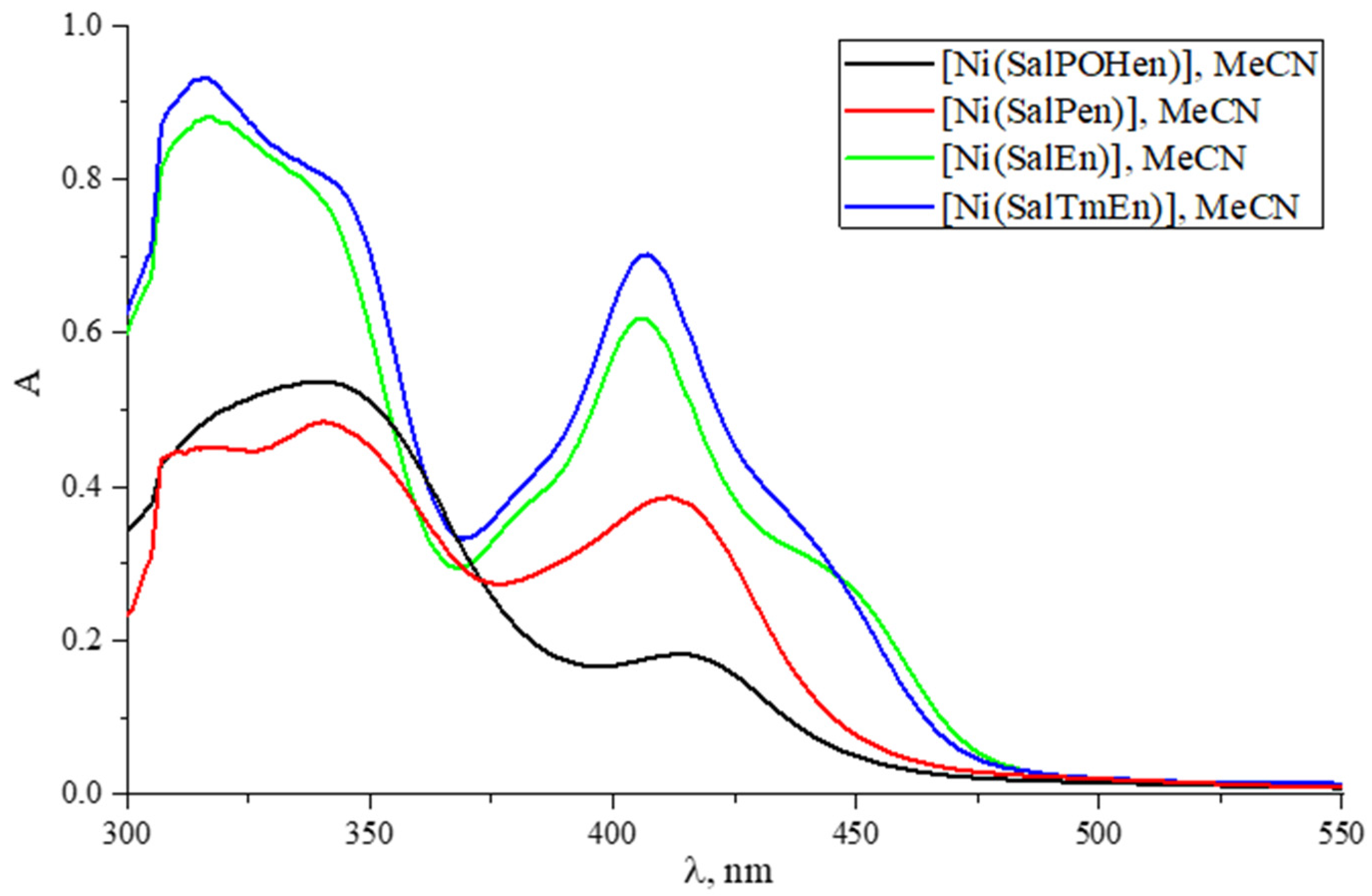
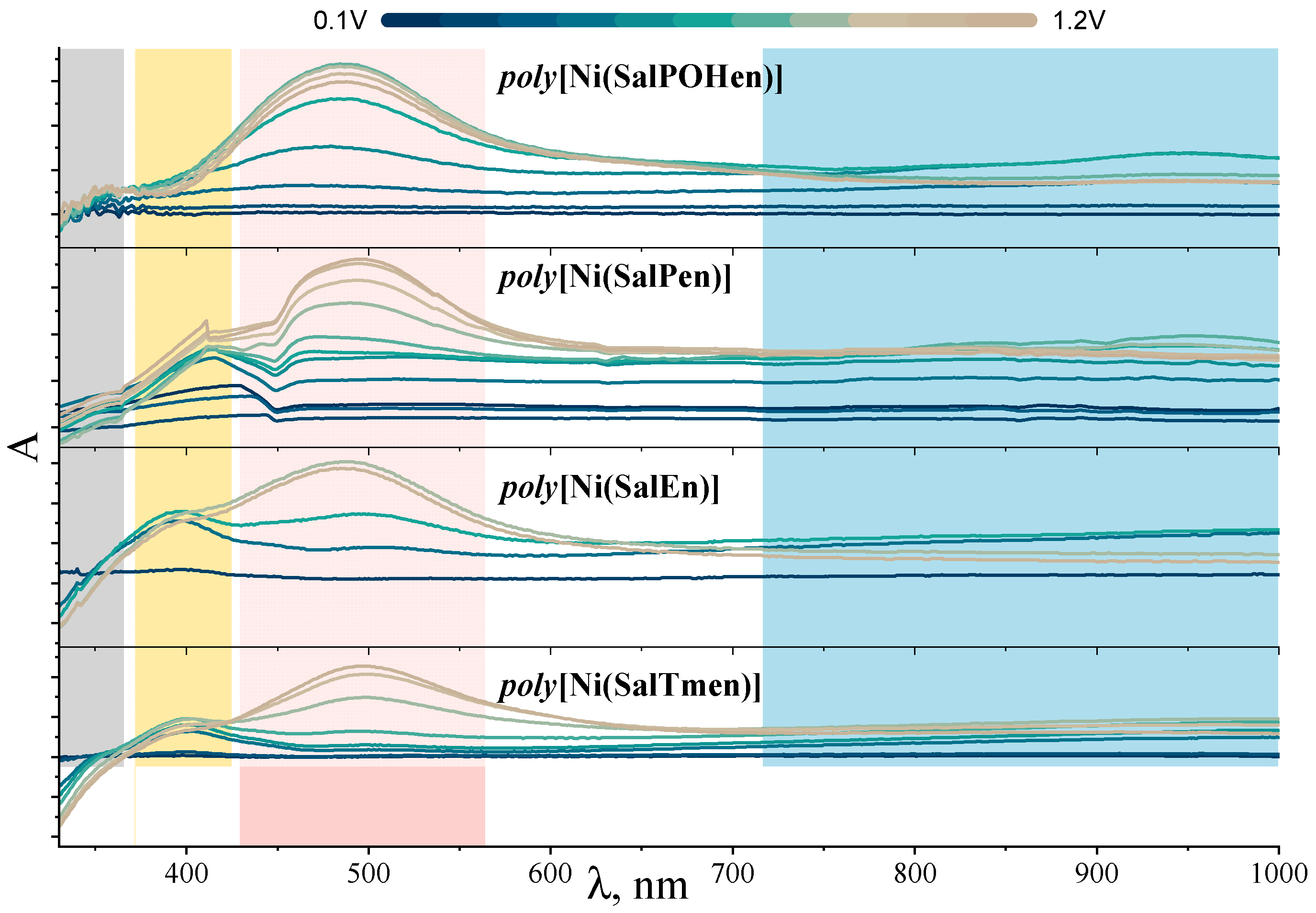
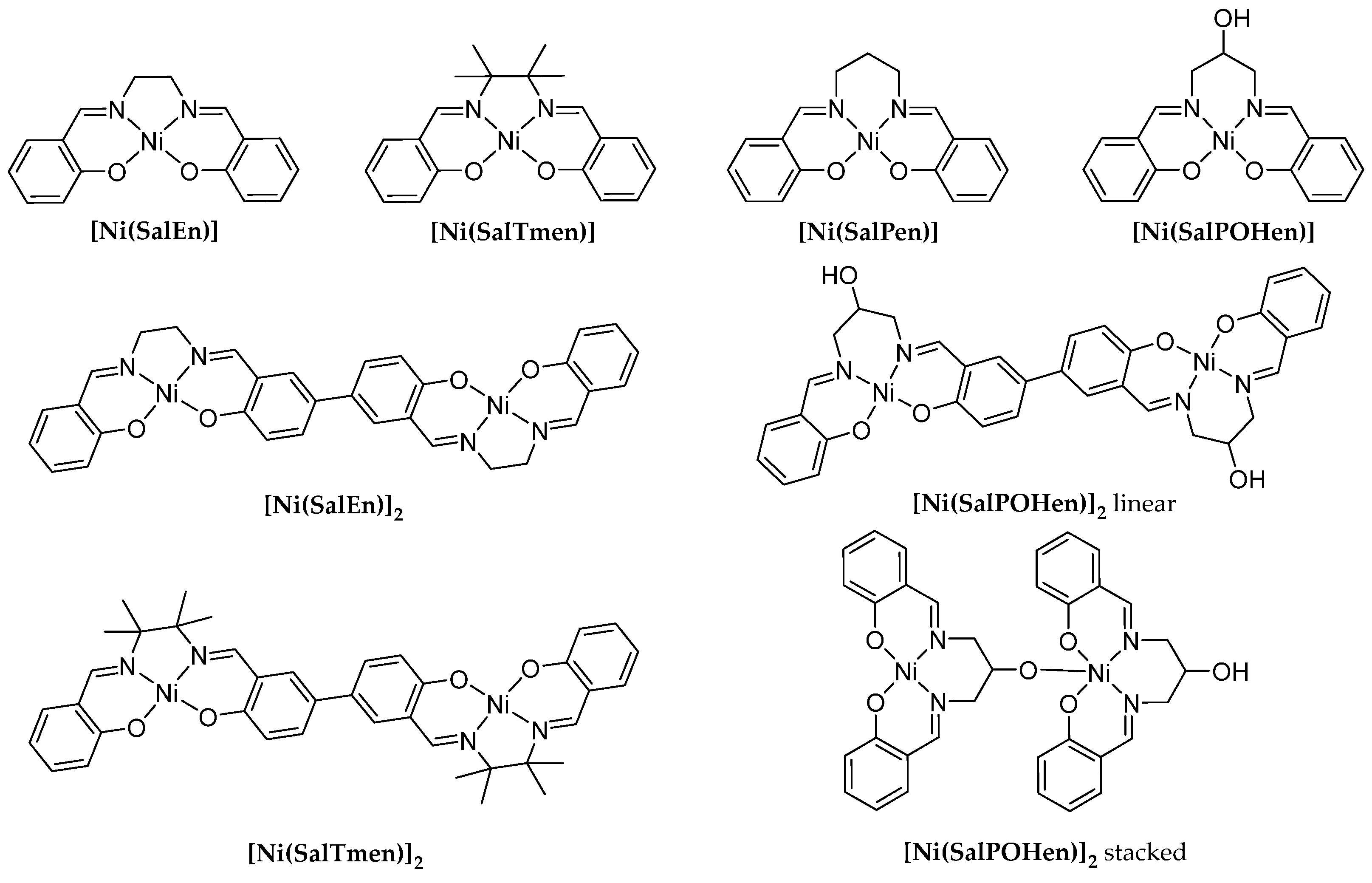
| [Ni(SalEn)] | [Ni(SalTmen)] | [Ni(SalPen)] | [Ni(SalPOHen)] | ||
|---|---|---|---|---|---|
| Low-Spin | Low-Spin | Low-Spin | High-Spin | Low-Spin | High-Spin |
| 347 (0.16) | 347 (0.17) | 345 (0.16) | 346 (0.17) | ||
| 276 (0.14) | 275 (0.12) | ||||
| 250 (0.21) | 249 (0.21) | 248 (0.15) | 250 (0.20) | 249 (0.18) | 248 (0.40) |
| 238 (0.16) | 239 (0.17) | ||||
| 233 (0.31) | 233 (0.22) | ||||
| 230 (0.13) | |||||
| 229 (0.38) | 227 (0.54) | 228 (0.53) | 228 (0.13) | 227 (0.43) | 227 (0.23) |
| [Ni(SalEn)]20 | [Ni(SalTmen)]20 | [Ni(SalPen)]20 | [Ni(SalPOHen)]20 | ||
|---|---|---|---|---|---|
| Low-Spin | Low-Spin | Low-Spin | High-Spin | Low-Spin | High-Spin |
| 379 (0.16) | 374 (0.17) | ||||
| 354 (0.22) | 352 (0.22) | 350 (0.21) | 350 (0.11) | 352 (0.22) | |
| 286 (0.19) | 284 (0.29) | 292 (0.12) | 291 (0.13) | ||
| 265 (0.82) | 265 (0.80) | 264 (0.76) | 257 (0.36) | 263 (0.74) | |
| 259 (0.17) | 256 (0.12) | ||||
| 257 (0.47) | 257 (0.77) | 253 (0.15) | 252 (0.43) | 253 (0.19) | |
| 254 (0.47) | 253 (0.31) | 250 (0.25) | 250 (0.55) | ||
| 248 (1.04) | 247 (0.71) | 249 (1.95) | 248 (0.29) | 250 (1.45) | |
| 241 (0.10) | 246 (0.39) | 248 (0.10) | 244 (0.22) | ||
| 241 (0.16) | |||||
| 236 (0.42) | 235 (0.57) | 235 (0.15) | 239 (0.31) | 236 (0.16) | |
| [Ni(SalEn)]22+ | [Ni(SalTmen)]22+ | [Ni(SalPen)]22+ | [Ni(SalPOHen)]22+ |
|---|---|---|---|
| 1354 (0.61) | 1355 (0.59) | 1393 (0.39) | 1371 (0.39) |
| 661 (0.23) | 654 (0.24) | ||
| 475 (0.84) | 480 (0.78) | 462 (1.26) | 462 (1.25) |
| 444 (0.20) | 446 (0.28) | 432 (0.12) | 431 (0.12) |
| 351 (0.10) | 356 (0.29) | 356 (0.26) | |
| 342 (0.29) | 344 (0.28) | ||
| 338 (0.13) | 335 (0.13) | 333 (0.13) | 335 (0.13) |
| 263 (0.20) | |||
| 260 (0.57) | 260 (0.35) | 256 (0.50) | 258 (0.47) |
| 245 (0.11) | 256 (0.33) | 249 (0.13) | 253 (0.12) |
| 241 (0.18) | 243 (0.17) | 244 (0.30) | |
| 237 (0.17) | 235 (0.15) | 241 (0.13) |
| [Ni(SalTmen)]22+ | [Ni(SalPOHen)]22+ |
|---|---|
| 1355 (0.59) | 1371 (0.39) |
| 1235 (0.00) | 1278 (0.00) |
| 687 * (0.00) | |
| 654 (0.24) | 675 (0.03) |
| 672 (0.00) | |
| 634 (0.08) | |
| 639 (0.00) | 630 (0.00) |
| 557 * (0.00) | 585 * (0.00) |
| 538 * (0.00) | |
| 527 * (0.00) | 510 * (0.00) |
| 492 * (0.00) | 500 * (0.00) |
| 480 (0.78) | 462 (1.25) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lukyanov, D.A.; Sizov, V.V.; Volkov, A.I.; Beletskii, E.V.; Yankin, A.N.; Alekseeva, E.V.; Levin, O.V. Tuning the Charge Transport in Nickel Salicylaldimine Polymers by the Ligand Structure. Molecules 2022, 27, 8798. https://doi.org/10.3390/molecules27248798
Lukyanov DA, Sizov VV, Volkov AI, Beletskii EV, Yankin AN, Alekseeva EV, Levin OV. Tuning the Charge Transport in Nickel Salicylaldimine Polymers by the Ligand Structure. Molecules. 2022; 27(24):8798. https://doi.org/10.3390/molecules27248798
Chicago/Turabian StyleLukyanov, Daniil A., Vladimir V. Sizov, Alexey I. Volkov, Evgenii V. Beletskii, Andrey N. Yankin, Elena V. Alekseeva, and Oleg V. Levin. 2022. "Tuning the Charge Transport in Nickel Salicylaldimine Polymers by the Ligand Structure" Molecules 27, no. 24: 8798. https://doi.org/10.3390/molecules27248798
APA StyleLukyanov, D. A., Sizov, V. V., Volkov, A. I., Beletskii, E. V., Yankin, A. N., Alekseeva, E. V., & Levin, O. V. (2022). Tuning the Charge Transport in Nickel Salicylaldimine Polymers by the Ligand Structure. Molecules, 27(24), 8798. https://doi.org/10.3390/molecules27248798









