In Melting Points We Trust: A Review on the Misguiding Characterization of Multicomponent Reactions Adducts and Intermediates
Abstract
1. Introduction
2. “Melting Point-Only” Characterization and Its Consequences to the MCRs Arena
3. Examples of Contrasting Mps for MCRs Adducts and Intermediates
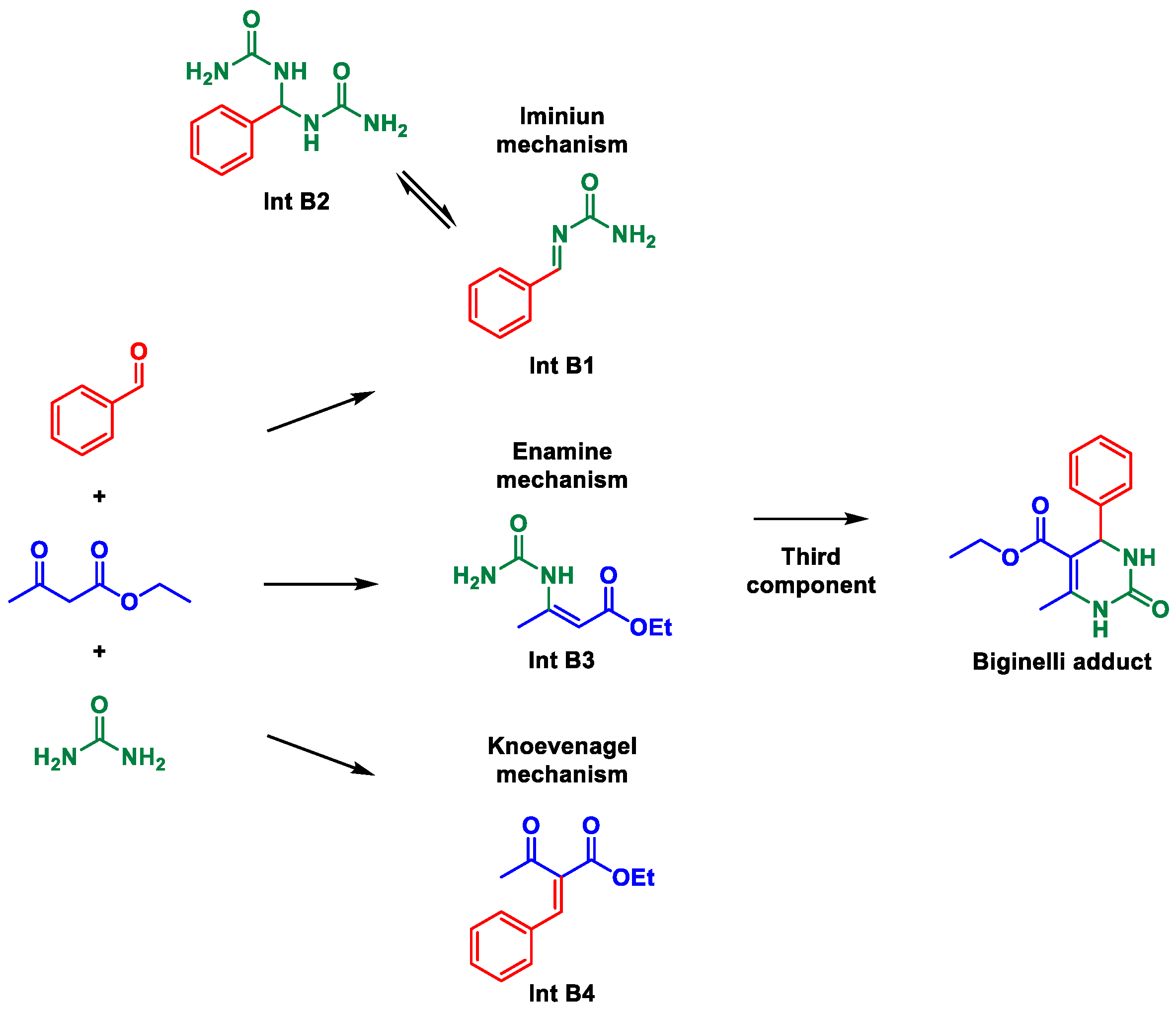
4. Mechanistic Implications
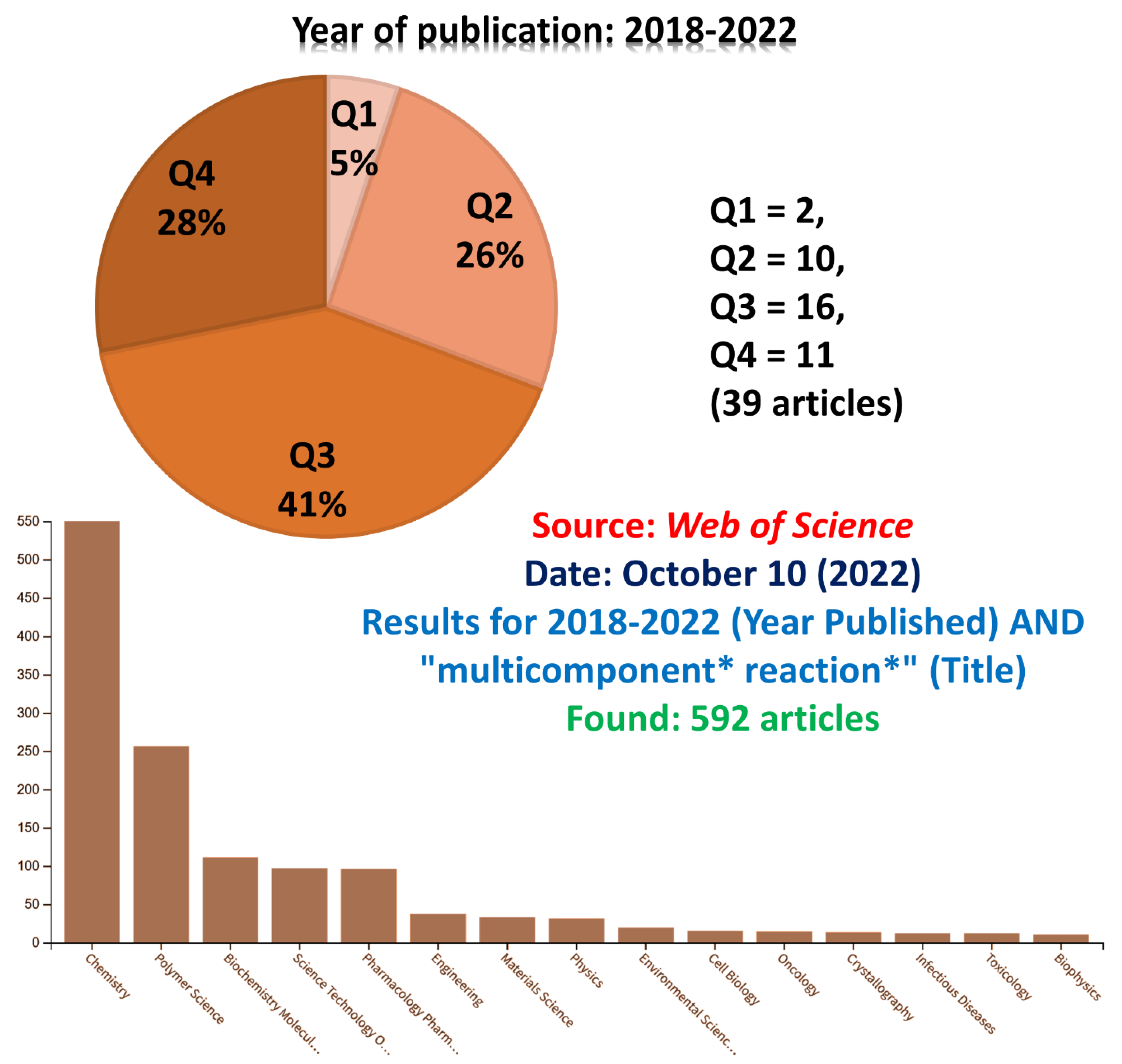
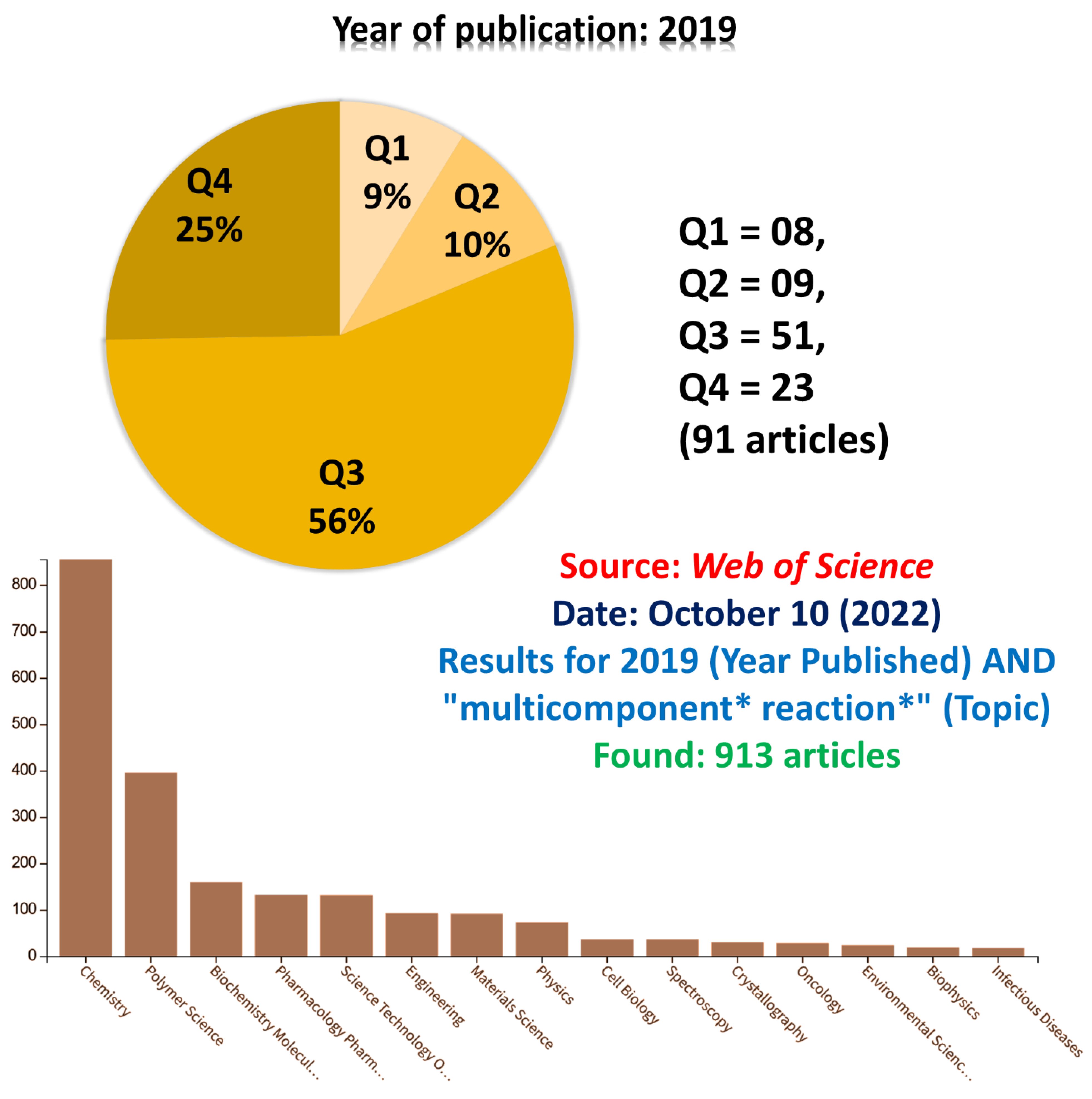
5. A General View of Recent Years
6. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Younus, H.A.; Al-Rashida, M.; Hameed, A.; Uroos, M.; Salar, U.; Rana, S.; Khan, K.M. Multicomponent reactions (MCR) in medicinal chemistry: A patent review (2010–2020). Expert Opin. Ther. Pat. 2021, 31, 267–289. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Cobo, A.A.; Franz, A.K. Recent advances in organocatalytic asymmetric multicomponent cascade reactions for enantioselective synthesis of spirooxindoles. Org. Chem. Front. 2021, 8, 4315–4348. [Google Scholar] [CrossRef]
- Anantha, I.S.S.; Kerru, N.; Maddila, S.; Jonnalagadda, S.B. Recent Progresses in the Multicomponent Synthesis of Dihydropyridines by Applying Sustainable Catalysts Under Green Conditions. Front. Chem. 2021, 9, 800236. [Google Scholar] [CrossRef] [PubMed]
- Preeti; Singh, K.N. Metal-free multicomponent reactions: A benign access to monocyclic six-membered N-heterocycles. Org. Biomol. Chem. 2021, 19, 2622–2657. [Google Scholar] [CrossRef]
- Liu, C.H.; Huang, W.B.; Zhang, J.H.; Rao, Z.H.; Gu, Y.L.; Jerome, F. Formaldehyde in multicomponent reactions. Green Chem. 2021, 23, 1447–1465. [Google Scholar] [CrossRef]
- John, S.E.; Gulati, S.; Shankaraiah, N. Recent advances in multi-component reactions and their mechanistic insights: A triennium review. Org. Chem. Front. 2021, 8, 4237–4287. [Google Scholar] [CrossRef]
- Ghosh, S.; Biswas, K. Metal-free multicomponent approach for the synthesis of propargylamine: A review. RSC Adv. 2021, 11, 2047–2065. [Google Scholar] [CrossRef]
- Fairoosa, J.; Neetha, M.; Anilkumar, G. Recent developments and perspectives in the copper-catalyzed multicomponent synthesis of heterocycles. RSC Adv. 2021, 11, 3452–3469. [Google Scholar] [CrossRef]
- Das, K.K.; Manna, S.; Panda, S. Transition metal catalyzed asymmetric multicomponent reactions of unsaturated compounds using organoboron reagents. Chem. Commun. 2021, 57, 441–459. [Google Scholar]
- Rodrigues, M.O.; Eberlin, M.N.; Neto, B.A.D. How and Why to Investigate Multicomponent Reactions Mechanisms? A Critical Review. Chem. Rec. 2021, 21, 2762–2781. [Google Scholar] [CrossRef]
- Neto, B.A.D.; Eberlin, M.N.; Sherwood, J. Solvent Screening Is Not Solvent Effect: A Review on the Most Neglected Aspect of Multicomponent Reactions. Eur. J. Org. Chem. 2022, 2022, e202200172. [Google Scholar] [CrossRef]
- Neto, B.A.D.; Rocha, R.O.; Rodrigues, M.O. Catalytic Approaches to Multicomponent Reactions: A Critical Review and Perspectives on the Roles of Catalysis. Molecules 2022, 27, 132. [Google Scholar] [CrossRef] [PubMed]
- Neto, B.A.D.; Rocha, R.O.; Lapis, A.A.M. What do we know about the ionic liquid effect in catalyzed multicomponent reactions?: A critical review. Curr. Opin. Green Sustain. Chem. 2022, 35, 100608. [Google Scholar] [CrossRef]
- Banfi, L.; Basso, A.; Lambruschini, C.; Moni, L.; Riva, R. The 100 facets of the Passerini reaction. Chem. Sci. 2021, 12, 15445–15472. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, M.A.; Mirhosseini-Eshkevari, B.; Tavakoli, M.; Zamani, F. Metal-organic frameworks: Advanced tools for multicomponent reactions. Green Chem. 2020, 22, 7265–7300. [Google Scholar] [CrossRef]
- Marinescu, M. Biginelli Reaction Mediated Synthesis of Antimicrobial Pyrimidine Derivatives and Their Therapeutic Properties. Molecules 2021, 26, 6022. [Google Scholar] [CrossRef]
- Machado, I.V.; dos Santos, J.R.N.; Januario, M.A.P.; Correea, A.G. Greener organic synthetic methods: Sonochemistry and heterogeneous catalysis promoted multicomponent reactions. Ultrason. Sonochem. 2021, 78, 105704. [Google Scholar] [CrossRef]
- Kar, S.; Sanderson, H.; Roy, K.; Benfenati, E.; Leszczynski, J. Green Chemistry in the Synthesis of Pharmaceuticals. Chem. Rev. 2022, 122, 3637–3710. [Google Scholar] [CrossRef]
- Luo, J.; Chen, G.S.; Chen, S.J.; Li, Z.D.; Liu, Y.L. Catalytic Enantioselective Isocyanide-Based Reactions: Beyond Passerini and Ugi Multicomponent Reactions. Chem. Eur. J. 2021, 27, 6598–6619. [Google Scholar] [CrossRef]
- Imtiaz, S.; War, J.A.; Banoo, S.; Khan, S. α-Aminoazoles/azines: Key reaction partners for multicomponent reactions. RSC Adv. 2021, 11, 11083–11165. [Google Scholar] [CrossRef]
- Domling, A.; Wang, W.; Wang, K. Chemistry and Biology Of Multicomponent Reactions. Chem. Rev. 2012, 112, 3083–3135. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, M.; Villacampa, M.; Menendez, J.C. Multicomponent mechanochemical synthesis. Chem. Sci. 2018, 9, 2042–2064. [Google Scholar] [CrossRef] [PubMed]
- Nenajdenko, V.G. Access to molecular complexity. Multicomponent reactions involving five or more components. Russ. Chem. Rev. 2020, 89, 1274–1336. [Google Scholar] [CrossRef]
- Yalkowsky, S.H.; Alantary, D. Estimation of Melting Points of Organics. J. Pharm. Sci. 2018, 107, 1211–1227. [Google Scholar] [CrossRef] [PubMed]
- Young, J.C.O.C. True Melting Point Determination. Chem. Educ. 2013, 18, 203–208. [Google Scholar]
- Allen, E. The melting point of impure organic compounds. J. Chem. Educ. 1942, 19, 278–281. [Google Scholar] [CrossRef]
- Cammenga, H.K.; Epple, M. Basic Principles of Thermoanalytical Techniques and Their Applications in Preparative Chemistry. Angew. Chem. Int. Ed. 1995, 34, 1171–1187. [Google Scholar] [CrossRef]
- Giron, D. Investigations of polymorphism and pseudo-polymorphism in pharmaceuticals by combined thermoanalytical techniques. J. Therm. Anal. Calorim. 2001, 64, 37–60. [Google Scholar] [CrossRef]
- Clark, J.B.; Hastie, J.W.; Kihlborg, L.H.E.; Metselaar, R.; Thackeray, M.M. Definitions of Terms Relating to Phase-Transitions of the Solid-State. Pure Appl. Chem. 1994, 66, 577–594. [Google Scholar] [CrossRef]
- Galwey, A.K. Melting and thermal decompositions of solids—An appraisal of mechanistic interpretations of thermal processes in crystals. J. Therm. Anal. Calorim. 2007, 87, 601–615. [Google Scholar] [CrossRef]
- Glass, B.D.; Brown, M.E. Thermoanalytical methods applied to medicine. Pure Appl. Chem. 2009, 81, 1881–1888. [Google Scholar] [CrossRef]
- Kerč, J.; Srčič, S. Thermal analysis of glassy pharmaceuticals. Thermochim. Acta 1995, 248, 81–95. [Google Scholar] [CrossRef]
- de Moura, A.; Gaglieri, C.; Alarcon, R.T.; Ferreira, L.T.; Vecchi, R.; Sanches, M.L.R.; de Oliveira, R.C.; Venturini, J.; da Silva, L.C.; Caires, F. A New Curcuminoids-Coumarin Derivative: Mechanochemical Synthesis, Characterization and Evaluation of Its In Vitro Cytotoxicity and Antimicrobial Properties. Chemistryselect 2021, 6, 11352–11361. [Google Scholar] [CrossRef]
- Belenguer, A.M.; Lampronti, G.I.; Michalchuk, A.A.L.; Emmerling, F.; Sanders, J.K.M. Quantitative reversible one pot interconversion of three crystalline polymorphs by ball mill grinding. Crystengcomm 2022, 24, 4256–4261. [Google Scholar] [CrossRef]
- Kamali, N.; Gniado, K.; McArdle, P.; Erxleben, A. Application of Ball Milling for Highly Selective Mechanochemical Polymorph Transformations. Org. Proc. Res. Dev. 2018, 22, 796–802. [Google Scholar] [CrossRef]
- Chen, F.; Fan, J.; Chen, X.; Li, Y.; Liang, C.; Ren, S.; Xue, R. Polymorph control of 5-fluorouracil during a ball milling process. CrystEngComm 2021, 23, 8027–8032. [Google Scholar] [CrossRef]
- Souza, R.Y.; Bataglion, G.A.; Ferreira, D.A.C.; Gatto, C.C.; Eberlin, M.N.; Neto, B.A.D. Insights on the Petasis Borono-Mannich multicomponent reaction mechanism. RSC Adv. 2015, 5, 76337–76341. [Google Scholar] [CrossRef]
- Fedorova, O.V.; Valova, M.S.; Titova, Y.A.; Ovchinnikova, I.G.; Grishakov, A.N.; Uimin, M.A.; Mysik, A.A.; Ermakov, A.E.; Rusinov, G.L.; Charushin, V.N. Catalytic effect of nanosized metal oxides in the Biginelli reaction. Kin. Catal. 2011, 52, 226–233. [Google Scholar] [CrossRef]
- Akhaja, T.N.; Raval, J.P. 1,3-dihydro-2H-indol-2-ones derivatives: Design, Synthesis, in vitro antibacterial, antifungal and antitubercular study. Eur. J. Med. Chem. 2011, 46, 5573–5579. [Google Scholar] [CrossRef]
- Makaev, F.; Styngach, E.; Muntyanu, V.; Pogrebnoi, S.; Rybkovskaya, Z.; Barba, A. New catalysts of Biginelli reaction. Russ. J. Org. Chem. 2007, 43, 1512–1515. [Google Scholar] [CrossRef]
- Munoz-Muniz, O.; Juaristi, E. An enantioselective approach to the Biginelli dihydropyrimidinone condensation reaction using CeCl3 and InCl3 in the presence of chiral ligands. Arkivoc 2003, 2003, 16–26. [Google Scholar] [CrossRef]
- Singh, K.; Arora, D.; Singh, S. Dowex-promoted general synthesis of N,N’-disubstituted-4-aryl-3,4-dihydropyrimidinones using a solvent-free Biginelli condensation protocol. Tetrahedron Lett. 2006, 47, 4205–4207. [Google Scholar] [CrossRef]
- Bose, A.K.; Manhas, M.S.; Pednekar, S.; Ganguly, S.N.; Dang, H.; He, W.; Mandadi, A. Large scale Biginelli reaction via water-based biphasic media: A green chemistry strategy. Tetrahedron Lett. 2005, 46, 1901–1903. [Google Scholar] [CrossRef]
- Chen, X.F.; Peng, Y.Q. Chloroferrate(III) ionic liquid: Efficient and recyclable catalyst for solvent-free synthesis of 3,4-dihydropyrimidin-2(1H)-ones. Catal. Lett. 2008, 122, 310–313. [Google Scholar] [CrossRef]
- Kumar, A.; Maurya, R.A. Synthesis of 3,4-dihydropyrimidin-2(1H)-ones using Ziegler–Natta catalyst system under solvent free conditions. J. Mol. Catal. A Chem. 2007, 272, 53–56. [Google Scholar] [CrossRef]
- Kumar, A.; Maurya, R.A. An efficient bakers’ yeast catalyzed synthesis of 3,4-dihydropyrimidin-2-(1H)-ones. Tetrahedron Lett. 2007, 48, 4569–4571. [Google Scholar] [CrossRef]
- Ramalingan, C.; Kwak, Y.-W. Tetrachlorosilane catalyzed multicomponent one-step fusion of biopertinent pyrimidine heterocycles. Tetrahedron 2008, 64, 5023–5031. [Google Scholar] [CrossRef]
- Singh, K.; Arora, D.; Singh, S. A highly regio- and chemoselective addition of carbon nucleophiles to pyrimidinones. A new route to C4 elaborated Biginelli compounds. Tetrahedron Lett. 2007, 48, 1349–1352. [Google Scholar] [CrossRef]
- Kappe, C.O. A reexamination of the mechanism of the Biginelli dihydropyrimidine synthesis. Support for an N-acyliminium ion intermediate. J. Org. Chem. 1997, 62, 7201–7204. [Google Scholar] [CrossRef]
- Boumoud, T.; Boumoud, B.; Rhouati, S.; Belfaitah, A.; Debache, A.; Mosset, P. An Efficient and Recycling Catalyst for the One-Pot Three-Component Synthesis of Substituted 3,4-Dihydropyrimidin-2(1H)-ones. E-J. Chem. 2008, 5, 688–695. [Google Scholar] [CrossRef]
- Bigdeli, M.A.; Gholami, G.; Sheikhhosseini, E. P-Dodecylbenzenesulfonic acid (DBSA), a Brønsted acid-surfactant catalyst for Biginelli reaction in water and under solvent free conditions. Chin. Chem. Lett. 2011, 22, 903–906. [Google Scholar] [CrossRef]
- Shirini, F.; Zolfigolb, M.A.; Mollarazia, E. Solvent-Free Synthesis of 3,4-Dihydropyrimidin-2(1H)-Ones Using Trichloroisocyanuric Acid. Lett. Org. Chem. 2005, 2, 718–720. [Google Scholar] [CrossRef]
- Ahmed, B.; Khan, R.A.; Habibullah; Keshari, M. An improved synthesis of Biginelli-type compounds via phase-transfer catalysis. Tetrahedron Lett. 2009, 50, 2889–2892. [Google Scholar] [CrossRef]
- Russowsky, D.; Lopes, F.A.; da Silva, V.S.S.; Canto, K.F.S.; D’Oca, M.G.M.; Godoi, M.N. Multicomponent Biginelli’s synthesis of 3,4-dihydropyrimidin-2(1H)-ones promoted by SnCl2.2H2O. J. Braz. Chem. Soc. 2004, 15, 165–169. [Google Scholar] [CrossRef]
- Ghosh, R.; Maiti, S.; Chakraborty, A. In(OTf)3-catalysed one-pot synthesis of 3,4-dihydropyrimidin-2(lH)-ones. J. Mol. Catal. A Chem. 2004, 217, 47–50. [Google Scholar] [CrossRef]
- Mahdavinia, G.H.; Sepehrian, H. MCM-41 anchored sulfonic acid (MCM-41-R-SO3H): A mild, reusable and highly efficient heterogeneous catalyst for the Biginelli reaction. Chin. Chem. Lett. 2008, 19, 1435–1439. [Google Scholar] [CrossRef]
- Pasunooti, K.K.; Chai, H.; Jensen, C.N.; Gorityala, B.K.; Wang, S.; Liu, X.-W. A microwave-assisted, copper-catalyzed three-component synthesis of dihydropyrimidinones under mild conditions. Tetrahedron Lett. 2011, 52, 80–84. [Google Scholar] [CrossRef]
- Gholap, A.R.; Venkatesan, K.; Daniel, T.; Lahoti, R.J.; Srinivasan, K.V. Ionic liquid promoted novel and efficient one pot synthesis of 3,4-dihydropyrimidin-2-(1H)-ones at ambient temperature under ultrasound irradiation. Green Chem. 2004, 6, 147–150. [Google Scholar] [CrossRef]
- Ranu, B.C.; Hajra, A.; Jana, U. Indium(III) chloride-catalyzed one-pot synthesis of dihydropyrimidinones by a three-component coupling of 1,3-dicarbonyl compounds, aldehydes, and urea: An improved procedure for the Biginelli reaction. J. Org. Chem. 2000, 65, 6270–6272. [Google Scholar] [CrossRef]
- Fu, R.; Yang, Y.; Ma, X.; Sun, Y.; Li, J.; Gao, H.; Hu, H.; Zeng, X.; Yi, J. An Efficient, Eco-friendly and Sustainable One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones Directly from Alcohols Catalyzed by Heteropolyanion-Based Ionic Liquids. Molecules 2017, 22, 1531. [Google Scholar] [CrossRef]
- Liu, C.J.; Wang, J.D. Copper(II) sulfamate: An efficient catalyst for the one-pot synthesis of 3,4-dihydropyrimidine-2(1H)-ones and thiones. Molecules 2009, 14, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Adibi, H.; Samimi, H.A.; Beygzadeh, M. Iron(III) trifluoroacetate and trifluoromethanesulfonate: Recyclable Lewis acid catalysts for one-pot synthesis of 3,4-dihydropyrimidinones or their sulfur analogues and 1,4-dihydropyridines via solvent-free Biginelli and Hantzsch condensation protocols. Catal. Commun. 2007, 8, 2119–2124. [Google Scholar] [CrossRef]
- Cepanec, I.; Litvić, M.; Filipan-Litvić, M.; Grüngold, I. Antimony(III) chloride-catalysed Biginelli reaction: A versatile method for the synthesis of dihydropyrimidinones through a different reaction mechanism. Tetrahedron 2007, 63, 11822–11827. [Google Scholar] [CrossRef]
- Dadhania, A.N.; Patel, V.K.; Raval, D.K. A Convenient and Efficient Protocol for the One Pot Synthesis of 3,4-Dihydropyrimidin-2-(1H)-ones Catalyzed by Ionic Liquids under Ultrasound Irradiation. J. Braz. Chem. Soc. 2011, 22, 511–516. [Google Scholar] [CrossRef][Green Version]
- Garima; Srivastava, V.P.; Yadav, L.D.S. Biginelli reaction starting directly from alcohols. Tetrahedron Lett. 2010, 51, 6436–6438. [Google Scholar] [CrossRef]
- Chandak, H.S.; Lad, N.P.; Upare, P.P. Recyclable Amberlyst-70 as a Catalyst for Biginelli Reaction: An Efficient One-Pot Green Protocol for the Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones. Catal. Lett. 2009, 131, 469–473. [Google Scholar] [CrossRef]
- Bandgar, B.P.; Kamble, V.T.; Bavikar, S.N.; Dhavane, A. Sodium Tetrafluoroborate as a New and Highly Efficient Catalyst for One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-Ones and Thiones. J. Chin. Chem. Soc. 2007, 54, 263–266. [Google Scholar] [CrossRef]
- Saxena, I.; Borah, D.C.; Sarma, J.C. Three component condensations catalyzed by iodine–alumina for the synthesis of substituted 3,4-dihydropyrimidin-2(1H)-ones under microwave irradiation and solvent-free conditions. Tetrahedron Lett. 2005, 46, 1159–1160. [Google Scholar] [CrossRef]
- Ghassamipour, S.; Sardarian, A.R. One-Pot Synthesis of Dihydropyrimidinones by Dodecylphosphonic Acid as Solid Bronsted Acid Catalyst under Solvent-Free Conditions via Biginelli Condensation. J. Iran. Chem. Soc. 2010, 7, 237–242. [Google Scholar] [CrossRef]
- Yu, Y.; Liu, D.; Liu, C.; Luo, G. One-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones using chloroacetic acid as catalyst. Bioorg. Med. Chem. Lett. 2007, 17, 3508–3510. [Google Scholar] [CrossRef]
- Shanmugam, P.; Annie, G.; Perumal, P.T. Synthesis of novel 3,4-dihydropyrimidinones on water soluble solid support catalyzed by indium triflate. J. Heterocycl. Chem. 2003, 40, 879–883. [Google Scholar] [CrossRef]
- Kato, T.; Chiba, T.; Sasaki, M. Biginelli Reaction using Acetoacetamide Derivatives. Yakugaku Zasshi 1981, 101, 182–185. [Google Scholar] [CrossRef][Green Version]
- Singh, V.K.; Pandey, V.K. Synthesis, antibacterial and antifungal activity of some new pyrido quinazolones. Indian J. Chem. Sect. B Org. Chem. Incl. Med. Chem. 2006, 45, 2745–2748. [Google Scholar] [CrossRef]
- Petersen, S.; Polyurethans, V. Low-molecular conversion products of diisocyanates. Ann. Der Chem. Und Pharm. 1949, 562, 205–229. [Google Scholar] [CrossRef]
- Pandey, V.K.; Mukesh; Kumar, A.; Trivedi, N. An investigation leading to preparation of tetrahydro-quinazoline derivatives involving ureidoalkylation and α-amidoalkylation reactions. Indian J. Chem. Sect. B Org. Chem. Incl. Med. Chem. 2008, 47B, 1910–1914. [Google Scholar] [CrossRef]
- Pandey, V.K.; Tusi, S. Synthesis of 6-(1H-benzimidazol-2-yl)-4-aralkyl-3,4-dihydro-1H-quinazolin-2-ones involving ureidoalkylation reaction. Indian J. Heterocycl. Chem. 2008, 18, 83–84. [Google Scholar]
- Muthusubramanian, L.; Mitra, R.B. Convenient Synthesis of 1-Acetyl-2,2-Dimethyl-3-Arylcyclopropanes. Org. Prep. Proced. Int. 2008, 40, 311–315. [Google Scholar] [CrossRef]
- Yi, W.-B.; Cai, C. Perfluoroalkylated pyridine catalyzed Knoevenagel condensation: An important complement of fluorous catalysis without fluorous solvent. Catal. Commun. 2008, 9, 1291–1296. [Google Scholar] [CrossRef]
- Yi, W.-B.; Yin, Y.-Q.; Cai, C. Ytterbium Perfluorooctanesulfonate-Catalyzed Knoevenagel Condensation in a Fluorotjs Biphasic System. Org. Prep. Proced. Int. 2007, 39, 71–75. [Google Scholar] [CrossRef]
- De Oliveira, F.S.; De Oliveira, P.M.; Farias, L.M.; Brinkerhoff, R.C.; Sobrinho, R.; Treptow, T.M.; D’Oca, C.R.M.; Marinho, M.A.G.; Hort, M.A.; Horn, A.P.; et al. Synthesis and antitumoral activity of novel analogues monastrol-fatty acids against glioma cells. Med. Chem. Commun. 2018, 9, 1282–1288. [Google Scholar] [CrossRef]
- Alvim, H.G.O.; da Silva Junior, E.N.; Neto, B.A.D. What do we know about multicomponent reactions? Mechanisms and trends for the Biginelli, Hantzsch, Mannich, Passerini and Ugi MCRs. RSC Adv. 2014, 4, 54282–54299. [Google Scholar] [CrossRef]
- Santos, V.G.; Godoi, M.N.; Regiani, T.; Gama, F.H.S.; Coelho, M.B.; de Souza, R.O.M.A.; Eberlin, M.N.; Garden, S.J. The Multicomponent Hantzsch Reaction: Comprehensive Mass Spectrometry Monitoring Using Charge-Tagged Reagents. Chem. Eur. J. 2014, 20, 12808–12816. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.R.; Idhayadhulla, A.; Nasser, A.J.A.; Selvin, J. Synthesis and antimicrobial activity of a new series 1,4-dihydropyridine derivatives. J. Serb. Chem. Soc. 2011, 76, 1–11. [Google Scholar] [CrossRef]
- Mandal, P.K.; Misra, A.K. HClO4-SiO2 Catalyzed Multicomponent Reactions for the Synthesis of Privileged Heterocyclic Structures. Lett. Org. Chem. 2006, 3, 848–853. [Google Scholar] [CrossRef]
- Bagley, M.; Lubinu, M. Microwave-Assisted Oxidative Aromatization of Hantzsch 1,4-Dihydro pyridines using Manganese Dioxide. Synthesis 2006, 2006, 1283–1288. [Google Scholar] [CrossRef]
- Patil, D.R.; Dalal, D.S. One-Pot, Solvent Free Synthesis of Hantzsch 1, 4-Dihydropyridines Using β-Cyclodextrin as a Supramolecular Catalyst. Lett. Org. Chem. 2011, 8, 477–483. [Google Scholar] [CrossRef]
- Reddy, B.P.; Rajesh, K.; Vijayakumar, V. Ionic Liquid [EMIM]OAc under Ultrasonic Irradiation towards Synthesis of 1,4-DHP’s. J. Chin. Chem. Soc. 2011, 58, 384–388. [Google Scholar] [CrossRef]
- Salehi, H.; Guo, Q.X. Synthesis of Substituted 1,4-Dihydropyridines in Water Using Phase-Transfer Catalyst Under Microwave Irradiation. Synth. Commun. 2004, 34, 4349–4357. [Google Scholar] [CrossRef]
- Suresh; Kumar, D.; Sandhu, J.S. New Efficient Protocol for the Production of Hantzsch 1,4-Dihydropyridines Using RuCl3. Synth. Commun. 2009, 39, 1957–1965. [Google Scholar] [CrossRef]
- Sabitha, G.; Arundhathi, K.; Sudhakar, K.; Sastry, B.S.; Yadav, J.S. CeCl3·7H2O-Catalyzed One-Pot Synthesis of Hantzsch 1,4-Dihydropyridines at Room Temperature. Synth. Commun. 2009, 39, 2843–2851. [Google Scholar] [CrossRef]
- Ming, L.; Guo, W.S.; Wen, L.R.; Li, Y.F.; Yang, H.Z. One-pot synthesis of Biginelli and Hantzsch products catalyzed by non-toxic ionic liquid (BMImSac) and structural determination of two products. J. Mol. Catal. A Chem. 2006, 258, 133–138. [Google Scholar]
- Yadav, D.K.; Patel, R.; Srivastava, V.P.; Watal, G.; Yadav, L.D.S. LiBr as an Efficient Catalyst for One-pot Synthesis of Hantzsch 1,4-Dihydropyridines under Mild Conditions. Chin. J. Chem. 2011, 29, 118–122. [Google Scholar] [CrossRef]
- Loev, B.; Goodman, M.M.; Snader, K.M.; Tedeschi, R.; Macko, E. Hantzsch-type dihydropyridine hypotensive agents. J. Med. Chem. 1974, 17, 956–965. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani-Choghamarani, A.; Zolfigol, M.A.; Hajjami, M.; Goudarziafshar, H.; Nikoorazm, M.; Yousefi, S.; Tahmasbi, B. Nano aluminium nitride as a solid source of ammonia for the preparation of hantzsch 1,4-dihydropyridines and bis-(1,4-dihydropyridines) in water via one pot multicomponent reaction. J. Braz. Chem. Soc. 2011, 22, 525–531. [Google Scholar] [CrossRef]
- Das Sharma, S.; Hazarika, P.; Konwar, D. A simple, green and one-pot four-component synthesis of 1,4-dihydropyridines and their aromatization. Catal. Commun. 2008, 9, 709–714. [Google Scholar] [CrossRef]
- Debache, A.; Ghalem, W.; Boulcina, R.; Belfaitah, A.; Rhouati, S.; Carboni, B. An efficient one-step synthesis of 1,4-dihydropyridines via a triphenylphosphine-catalyzed three-component Hantzsch reaction under mild conditions. Tetrahedron Lett. 2009, 50, 5248–5250. [Google Scholar] [CrossRef]
- Gopalakrishnan, M.; Sureshkumar, P.; Kanagarajan, V.; Thanusu, J.; Govindaraju, R. A simplified green chemistry approaches to organic synthesis in solid media. Activated fly ash, an industrial waste (pollutant) as an efficient and novel catalyst for some selected organic reactions in solvent-free conditions under microwave irradiation. Arkivoc 2006, 2006, 130–141. [Google Scholar] [CrossRef]
- Puri, S.; Parmar, A.; Kaur, B.; Kumar, H. Ultrasound Promoted Cu(Clo4)2 Catalyzed Rapid Synthesis Of Substituted 1,2,3,4- Tetrahydropyrimidine-2-Ones & Hantzsch 1,4-Dihydropyridines In Dry Media. Heterocycl. Commun. 2009, 15, 51–55. [Google Scholar]
- Saha, M.; Roy, S.; Kumar Chaudhuri, S.; Bhar, S. Microwave-assisted ammonium formate-mediated synthesis of Hanstzch dihydropyridines under solvent-free conditions—A green protocol. Green Chem. Lett. Rev. 2008, 1, 99–102. [Google Scholar] [CrossRef]
- Singh, H.; Sarin, R.; Singh, K. One Carbon Unit Transfer to Enamines through Oxazolidines and Tetrahydro-2H-1,3-oxazine. Heterocycles 1986, 24, 3039–3042. [Google Scholar] [CrossRef]
- Angeles-Beltran, D.; Lomas-Romero, L.; Lara-Corona, V.H.; Gonzalez-Zamora, E.; Negron-Silva, G. Sulfated zirconia-catalyzed synthesis of 3,4-dihydropyrimidin-2(1H)-ones (DHPMs) under solventless conditions: Competitive multicomponent Biginelli vs. Hantzsch reactions. Molecules 2006, 11, 731–738. [Google Scholar] [CrossRef]
- Lei, M.; Ma, L.; Hu, L. Thiamine Hydrochloride–Catalyzed One-Pot Synthesis of 1,4-Dihydropyridine Derivatives Under Solvent-Free Conditions. Synth. Commun. 2011, 41, 1969–1976. [Google Scholar] [CrossRef]
- Kumar, R.S.; Idhayadhulla, A.; Nasser, A.J.A.; Selvin, J. Synthesis and anticoagulant activity of a new series of 1,4-dihydropyridine derivatives. Eur. J. Med. Chem. 2011, 46, 804–810. [Google Scholar] [CrossRef]
- Wang, S.X.; Li, Z.Y.; Zhang, J.C.; Li, J.T. The solvent-free synthesis of 1,4-dihydropyridines under ultrasound irradiation without catalyst. Ultrason. Sonochem. 2008, 15, 677–680. [Google Scholar] [CrossRef] [PubMed]
- Salazar, R.; Navarrete-Encina, P.A.; Camargo, C.; Squella, J.A.; Núñez-Vergara, L.J. Oxidation of 4-(3-Indolyl)- and 4-(5-Indolyl)-1,4-dihydropyridines in Aprotic and Protic Media: Reactivity toward Alkylperoxyl Radicals. J. Electrochem. Soc. 2008, 155, 103–108. [Google Scholar] [CrossRef][Green Version]
- Khrustalev, D.P.; Suleimenova, A.A.; Fazylov, S.D.; Gazaliev, A.M.; Ayapbergenov, K.A. Synthesis of Some 1,4-Dihydropyridines under the Microwave Irradiation. Russ. J. Gen. Chem. 2010, 80, 376–377. [Google Scholar] [CrossRef]
- Kloek, J.A.; Leschinsky, K.L. An Improved Method for the Synthesis of Stabilized Primary Enamines and Imines. J. Org. Chem. 1978, 43, 1460–1462. [Google Scholar] [CrossRef]
- Davood, A.; Nematollahi, A.R.; Iman, M.; Shafiee, A. Synthesis and docking studies of new 1,4-dihydropyridines containing 4-(5)-Chloro-2-ethyl-5-(4)-imidazolyl substituent as novel calcium channel agonist. Arch Pharm. Res. 2009, 32, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhang, Q.; Xu, J. A Convenient and Effective Method for Synthesizing β-Amino-α,β-Unsaturated Esters and Ketones. Synth. Commun. 2006, 34, 909–916. [Google Scholar] [CrossRef]
- Heravi, M.M.; Zakeri, M.; Mohammadi, N. Guanidine hydrochloride: An active and simple catalyst for Mannich type reaction in solvent-free condition. Chin. Chem. Lett. 2011, 22, 797–800. [Google Scholar] [CrossRef]
- Luo, H.-T.; Kang, Y.-R.; Nie, H.-Y.; Yang, L.-M. Sulfamic Acid as a Cost-Effective and Recyclable Catalyst for β-Amino Carbonyl Compounds Synthesis. J. Chin. Chem. Soc. 2009, 56, 186–195. [Google Scholar] [CrossRef]
- Ying, A.-G.; Wang, L.-M.; Deng, H.-X.; Chen, J.-H.; Chen, X.-Z.; Ye, W.-D. Green and efficient aza-Michael additions of aromatic amines to α,β-unsaturated ketones catalyzed by DBU based task-specific ionic liquids without solvent. Arkivoc 2009, 2009, 288–298. [Google Scholar] [CrossRef]
- Li, J.; Peng, Y.; Song, G. Mannich reaction catalyzed by carboxyl-functionalized ionic liquid in aqueous media. Catal. Lett. 2005, 102, 159–162. [Google Scholar] [CrossRef]
- Barluenga, J.; Cuervo, H.; Olano, B.; Fustero, S.; Gotor, V. Synthesis ofN-Aryl-substituted 2-Aminoalkyl Ketones and 1,3-Alkanediamines. Synthesis 1986, 1986, 469–473. [Google Scholar] [CrossRef]
- Shen, W.; Wang, L.-M.; Tian, H. Quaternary ammonium salt gemini surfactants containing perfluoroalkyl tails catalyzed one-pot Mannich reactions in aqueous media. J. Fluor. Chem. 2008, 129, 267–273. [Google Scholar] [CrossRef]
- Minami, T.; Takimoto, F.; Agawa, T. Reactions of N-sulfinylarylamines with carbonyl compounds and a nitrile in the presence of copper. J. Org. Chem. 2002, 41, 3811–3813. [Google Scholar] [CrossRef]
- Ollevier, T.; Nadeau, E. Bismuth triflate-catalyzed three-component mannich-type reaction. J. Org. Chem. 2004, 69, 9292–9295. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, N.G.; Basalaeva, L.I. Synthesis of Unsymmetrical -Arylaminoketones. Russ. J. Gen. Chem. 2004, 74, 926–932. [Google Scholar] [CrossRef]
- Nemati, F.; Bigdeli, M.A.; Mahdavinia, G.H.; Kiani, H. 2,4,6-Trichloro[1,3,5]triazine (TCT)-catalyzed one-pot Mannich-type reaction: Three component synthesis of β-amino carbonyl compounds. Green Chem. Lett. Rev. 2010, 3, 89–92. [Google Scholar] [CrossRef]
- Li, H.; Zeng, H.-Y.; Shao, H.-W. Bismuth(III) chloride-catalyzed one-pot Mannich reaction: Three-component synthesis of β-amino carbonyl compounds. Tetrahedron Lett. 2009, 50, 6858–6860. [Google Scholar] [CrossRef]
- Wang, R.; Li, B.-G.; Huang, T.-K.; Shi, L.; Lu, X.-X. NbCl5-Catalyzed one-pot Mannich-type reaction: Three component synthesis of β-amino carbonyl compounds. Tetrahedron Lett. 2007, 48, 2071–2073. [Google Scholar] [CrossRef]
- Li, Z.; Ma, X.; Liu, J.; Feng, X.; Tian, G.; Zhu, A. Silica-supported aluminum chloride: A recyclable and reusable catalyst for one-pot three-component Mannich-type reactions. J. Mol. Catal. A Chem. 2007, 272, 132–135. [Google Scholar] [CrossRef]
- Kidwai, M.; Anwar, J. Cerium Chloride (CeCl3·7H2O) as a highly efficient catalyst for one-pot Three-Component Mannich reaction. J. Braz. Chem. Soc. 2010, 21, 2175–2179. [Google Scholar] [CrossRef]
- Bigdeli, M.A.; Nemati, F.; Mahdavinia, G.H. HClO4–SiO2 catalyzed stereoselective synthesis of β-amino ketones via a direct Mannich-type reaction. Tetrahedron Lett. 2007, 48, 6801–6804. [Google Scholar] [CrossRef]
- Dong, F.; Zhenghao, F.; Zuliang, L. Functionalized ionic liquid as the recyclable catalyst for Mannich-type reaction in aqueous media. Catal. Commun. 2009, 10, 1267–1270. [Google Scholar] [CrossRef]
- Roth, H.J.S.E.; George, H.; Assadi, F. Photochemical reactions of Mannich bases of aromatic amines. Tetrahedron Lett. 1968, 9, 3433–3436. [Google Scholar] [CrossRef]
- Pilli, R.A.; Russowsky, D. Secondary Mannich bases via trimethylsilyl trifluoromethanesulphonate promoted addition of silyl enol ethers to Schiff bases. J. Chem. Soc. Chem. Commun. 1987, 14, 1053–1054. [Google Scholar] [CrossRef]
- Chimenti, F.; Fioravanti, R.; Bolasco, A.; Chimenti, P.; Secci, D.; Rossi, F.; Yanez, M.; Orallo, F.; Ortuso, F.; Alcaro, S. Chalcones: A valid scaffold for monoamine oxidases inhibitors. J. Med. Chem. 2009, 52, 2818–2824. [Google Scholar] [CrossRef]
- Mansour, E.S.M.E.; El-Sadany, S.K.; Kassem, A.A.; Maksoud, H.A. Aminolysis of para-substituted benzalacetophenones. J. Chem. Eng. Data 2002, 34, 368–370. [Google Scholar] [CrossRef]
- Mandel, H.G.; Hill, A.J. The Conversion of Formamides into Formamidines1. J. Am. Chem. Soc. 1954, 76, 3978–3982. [Google Scholar] [CrossRef]
- Begum, N.A.; Roy, N.; Laskar, R.A.; Roy, K. Mosquito larvicidal studies of some chalcone analogues and their derived products: Structure–activity relationship analysis. Med. Chem. Res. 2010, 20, 184–191. [Google Scholar] [CrossRef]
- Hayat, F.; Salahuddin, A.; Umar, S.; Azam, A. Synthesis, characterization, antiamoebic activity and cytotoxicity of novel series of pyrazoline derivatives bearing quinoline tail. Eur. J. Med. Chem. 2010, 45, 4669–4675. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Zhu, S.; Jin, G.; Li, Z. Strong phenyl–perfluorophenyl π–π stacking and C–H⋯F–C hydrogen bonding interactions in the crystals of the corresponding aromatic aldimines. Tetrahedron Lett. 2005, 46, 2713–2716. [Google Scholar] [CrossRef]
- Deng, G.; Ren, T. Indium Trichloride Catalyzes Aldol-Condensations of Aldehydes and Ketones. Synth. Commun. 2006, 33, 2995–3001. [Google Scholar] [CrossRef]
- Kumar, A.; Akanksha. Zirconium chloride catalyzed efficient synthesis of 1,3-diaryl-2-propenones in solvent free conditions via aldol condensation. J. Mol. Catal. A Chem. 2007, 274, 212–216. [Google Scholar] [CrossRef]
- Montes-Avila, J.; Diaz-Camacho, S.P.; Sicairos-Felix, J.; Delgado-Vargas, F.; Rivero, I.A. Solution-phase parallel synthesis of substituted chalcones and their antiparasitary activity against Giardia lamblia. Bioorg. Med. Chem. 2009, 17, 6780–6785. [Google Scholar] [CrossRef]
- Neumann, W.P. Metallorganische Molekülverbindungen, I. Komplexe von Äthern und Aminen mit Organoaluminium-amiden R2Al-NR’2. Ann. Der Chem. Und Pharm. 1963, 667, 1–11. [Google Scholar] [CrossRef]
- Kakati, D.; Sarma, J.C. Microwave assisted solvent free synthesis of 1,3-diphenylpropenones. Chem. Cent. J. 2011, 5, 8. [Google Scholar] [CrossRef]
- Narender, T.; Papi Reddy, K. A simple and highly efficient method for the synthesis of chalcones by using borontrifluoride-etherate. Tetrahedron Lett. 2007, 48, 3177–3180. [Google Scholar] [CrossRef]
- Ranu, B.C.; Jana, R. Catalysis by ionic liquid. A green protocol for the stereoselective debromination of vicinal-dibromides by [pmIm]BF4 under microwave irradiation. J. Org. Chem. 2005, 70, 8621–8624. [Google Scholar] [CrossRef]
- Thiot, C.; Mioskowski, C.; Wagner, A. Sequential Hiyama Coupling/Narasaka Acylation Reaction of (E)-1,2-Disilylethene: Rapid Assembly of α,β-Unsaturated Carbonyl Motifs. Eur. J. Org. Chem. 2009, 2009, 3219–3227. [Google Scholar] [CrossRef]
- Wu, X.F.; Neumann, H.; Spannenberg, A.; Schulz, T.; Jiao, H.; Beller, M. Development of a general palladium-catalyzed carbonylative Heck reaction of aryl halides. J. Am. Chem. Soc. 2010, 132, 14596–14602. [Google Scholar] [CrossRef] [PubMed]
- Mahesh, M.; Murphy, J.A.; Wessel, H.P. Novel deoxygenation reaction of epoxides by indium. J. Org. Chem. 2005, 70, 4118–4123. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.; Liu, R.; Meng, Q.; Zhao, Y.; Zhou, Y.; Huang, J. The reaction of ketones with benzaldehyde catalyzed by TiCl4·2THF. J. Mol. Catal. A Chem. 2005, 225, 239–243. [Google Scholar] [CrossRef]
- Kryshtal, G.V.; Zhdankina, G.M.; Zlotin, S.G. Tetraalkylammonium and 1,3-Dialkylimidazolium Salts with Fluorinated Anions as Recoverable Phase-Transfer Catalysts in Solid Base-Promoted Cross-Aldol Condensations. Eur. J. Org. Chem. 2005, 2005, 2822–2827. [Google Scholar] [CrossRef]
- Sikdar, A.P.; Chetri, A.B.; Baishya, G.; Das, P.J. Solid Phase Oxidation of Alcohols and Benzyl Halides to Carbonyls Using Bromate Exchange Resin. Synth. Commun. 2003, 33, 3147–3151. [Google Scholar] [CrossRef]
- Hasegawa, A.; Naganawa, Y.; Fushimi, M.; Ishihara, K.; Yamamoto, H. Design of Bronsted acid-assisted chiral Bronsted acid catalyst bearing a bis(triflyl)methyl group for a Mannich-type reaction. Org. Lett. 2006, 8, 3175–3178. [Google Scholar] [CrossRef]
- Silva, V.D.; Stambuk, B.U.; Nascimento, M.D.G. Efficient chemoselective biohydrogenation of 1,3-diaryl-2-propen-1-ones catalyzed by Saccharomyces cerevisiae yeasts in biphasic system. J. Mol. Catal. B-Enzym. 2010, 63, 157–163. [Google Scholar] [CrossRef]
- Fang, F.; Li, Y.A.; Tian, S.K. Stereoselective Olefination of N-Sulfonyl Imines with Stabilized Phosphonium Ylides for the Synthesis of Electron-Deficient Alkenes. Eur. J. Org. Chem. 2011, 2011, 1084–1091. [Google Scholar] [CrossRef]
- Wadsworth, W.S.; Emmons, W.D. The Utility of Phosphonate Carbanions in Olefin Synthesis. J. Am. Chem. Soc. 2002, 83, 1733–1738. [Google Scholar] [CrossRef]
- Kanagarajan, V.; Thanusu, J.; Gopalakrishnan, M. Synthesis and in vitro microbiological evaluation of an array of biolabile 2-morpholino-N-(4,6-diarylpyrimidin-2-yl)acetamides. Eur. J. Med. Chem. 2010, 45, 1583–1589. [Google Scholar] [CrossRef] [PubMed]
- Łukasiewicz, A. A study of certain reactions of trihalogenoacetic acids: The reaction of trichloro- and tribromoacetic acids with schiff bases. Tetrahedron 1964, 20, 1–12. [Google Scholar] [CrossRef]
- Blanco-Ania, D.; Hermkens, P.H.H.; Sliedregt, L.A.J.M.; Scheeren, H.W.; Rutjes, F.P.J.T. Synthesis of cucurbitine derivatives: Facile straightforward approach to methyl 3-amino-4-aryl-1-methylpyrrolydine-3-carboxylates. Tetrahedron 2009, 65, 5393–5401. [Google Scholar] [CrossRef]
- Margerum, J.D.; Sousa, J.A. Spectroscopic Studies of Substituted Benzalanilines. Appl. Spectrosc. 2016, 19, 91–97. [Google Scholar] [CrossRef]
- Stacy, G.W.; Day, R.I.; Morath, R.J. Schiff Bases and Related Substances. II. Reactions of Thiols with N-Benzylideneaniline and N-Benzylideneanthranilic Acid. J. Am. Chem. Soc. 2002, 77, 3869–3873. [Google Scholar] [CrossRef]
- Liu, W.; Hu, G.; Gu, M. The probability of publishing in first-quartile journals. Scientometrics 2016, 106, 1273–1276. [Google Scholar] [CrossRef]
- Miranda, R.; Garcia-Carpintero, E. Comparison of the share of documents and citations from different quartile journals in 25 research areas. Scientometrics 2019, 121, 479–501. [Google Scholar] [CrossRef]


 | |||||
|---|---|---|---|---|---|
| Adduct | Reference | Int B1 (Ref.) | Int B2 (Ref.) | Int B3 (Ref.) | Int B4 (Ref.) |
| 225–226 ([38]) | |||||
| 218 | [39] | ||||
| 213–214 | [40] | ||||
| 211–212 | [41] | ||||
| 210–211 | [42] | ||||
| 210 | [43] | ||||
| 209–211 | [44] | ||||
| 209–210 | [45,46] | ||||
| 208–209 | [47] | ||||
| 207 | [48] | ||||
| 206–207 | [49,50] | ||||
| 204–207 | [51] | 202–205 ([41]) | |||
| 204–206 | [38,52,53] | ||||
| 204–205 | [54] | ||||
| 204 | [55] | ||||
| 202–205 | [56] | ||||
| 202–204 | [57] | ||||
| 202–203 | [58,59,60] | ||||
| 201–204 | [61] | ||||
| 201–203 | [62,63] | ||||
| 201–202 | [64,65] | ||||
| 200–201 | [66] | ||||
| 200–202 | [67] | ||||
| 199–201 | [68] | ||||
| 198–200 | [69,70] | ||||
| 198–199 | [71] | ||||
| 194 ([72]) | |||||
| 178 ([73]) | |||||
| 168–170 ([74]) | |||||
| 157–158 ([75]) | 158–160 ([63]) | ||||
| 154 ([76]) | |||||
| 78–80 ([77]) | |||||
| 44–45 ([78,79]) | |||||
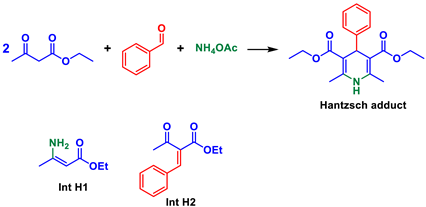 | |||
|---|---|---|---|
| Adduct | Reference | Int H1 (Ref.) | Int H2 (Ref.) |
| 253 | [83] | ||
| 160 | [84] | ||
| 158–160 | [85,86,87,88,89] | ||
| 158 | [90] | ||
| 157–159 | [91,92,93] | ||
| 157–158 | [94,95] | ||
| 156–158 | [96] | ||
| 156–157 | [97] | ||
| 156 | [98,99,100] | ||
| 154–155 | [101] | ||
| 153–155 | [102] | ||
| 153 | [103] | ||
| 152–154 | [104] | ||
| 150–153 | [105] | ||
| 145–146 | [106] | ||
| 78–80 ([77]) | |||
| 44–45 ([78,79]) | |||
| 33–35 ([107]) | |||
| 32–35 ([108]) | |||
| 30–32 ([109]) | |||
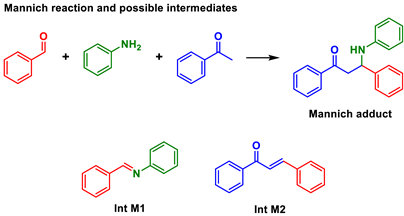 | |||
|---|---|---|---|
| Adduct | Reference | Int M1 (Ref.) | Int M2 (Ref.) |
| 176–179 | [110] | ||
| 174–176 | [111] | ||
| 173–175 | [112] | ||
| 172–174 | [113,114] | ||
| 172–173 | [115] | ||
| 171–172 | [116] | ||
| 170–172 | [117] | ||
| 170–171 | [118,119] | ||
| 169–170 | [120,121,122] | ||
| 168–170 | [123,124] | ||
| 168–169 | [125] | ||
| 167 | [126] | ||
| 163–165 | [127] | 163–164 ([128]) | |
| 161 | [129] | ||
| 140–141 ([130]) | |||
| 58 ([131]) | |||
| 57 ([132]) | |||
| 51–56 ([133]) | 56–58 ([134,135,136]) | ||
| 56 ([137]) | 56–57 ([138,139]) 56 ([140]) 55–57 ([141,142]) 55–56 ([143]) 54–56 ([144]) 54–55 ([145]) 54 ([146]) | ||
| 52 ([147]) | 53–54 ([148,149]) 52–53 ([150]) 52 ([151]) | ||
| 51–52 ([152,153,154]) | |||
| 48–50 ([155]) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neto, B.A.D.; Beck, P.S.; Sorto, J.E.P.; Eberlin, M.N. In Melting Points We Trust: A Review on the Misguiding Characterization of Multicomponent Reactions Adducts and Intermediates. Molecules 2022, 27, 7552. https://doi.org/10.3390/molecules27217552
Neto BAD, Beck PS, Sorto JEP, Eberlin MN. In Melting Points We Trust: A Review on the Misguiding Characterization of Multicomponent Reactions Adducts and Intermediates. Molecules. 2022; 27(21):7552. https://doi.org/10.3390/molecules27217552
Chicago/Turabian StyleNeto, Brenno A. D., Pedro S. Beck, Jenny E. P. Sorto, and Marcos N. Eberlin. 2022. "In Melting Points We Trust: A Review on the Misguiding Characterization of Multicomponent Reactions Adducts and Intermediates" Molecules 27, no. 21: 7552. https://doi.org/10.3390/molecules27217552
APA StyleNeto, B. A. D., Beck, P. S., Sorto, J. E. P., & Eberlin, M. N. (2022). In Melting Points We Trust: A Review on the Misguiding Characterization of Multicomponent Reactions Adducts and Intermediates. Molecules, 27(21), 7552. https://doi.org/10.3390/molecules27217552








