Abstract
This paper presents the results of the synthesis of samarium-doped bismuth ferrite (BFO) nanoparticles by the solution combustion method. The dependence of BFO properties on the amount of the samarium (Sm) in the composition was studied. The synthesized nanocomposites were characterized by scanning electron microscopy SEM), X-ray diffractometry (XRD), Raman, Electron Diffuse Reflectance Spectroscopy (EDRS) and Electron Magnetic Resonance (EMR). The photocatalytic (PC) measurements showed the absence of a strict correlation between the PC activity and the crystallite size and band gap. An increase in the PC activity of BFO samples with 10 and 15% doping was observed and it was concluded that in controlling the PC properties in doped BFO, the processes of interfacial polarization at the boundaries of the morphotropic phase transition are of decisive importance. It was supposed that the internal electric field formed at these boundaries contributes to the efficient separation of photogenerated charge carriers.
Keywords:
BFO; bismuth ferrite; morphotropic phase boundary; nanocomposite; photocatalysis; polarization; Sm 1. Introduction
The topic of the photocatalytic (PC) decomposition of organic water pollutants continues to be one of the hottest topics. Perovskite materials and ferrites have generated strong research interest in recent decades as they are among the most promising materials for photocatalytic degradation of pollutants as well as hydrogen generation due to their unique properties and the possibility of their controlled customization [1,2,3,4,5,6]. It has recently been shown that the combination of perovskite materials with ferrites and an S-type heterojunction made it possible to significantly enhance the photocatalytic properties [7]. Bismuth ferrite (BiFeO3, BFO) is a unique material that combines the properties of perovskite materials and ferrites [8]. One of the important advantages of this material is the smaller band gap (2.2 eV) compared to the traditional TiO2 photocatalyst [9,10]. This is important for improving the efficiency of solar energy use. BFO has been synthesized using various methods, and is used in the decomposition and mineralization of pollutants [11,12]. One of the synthesis methods that allow the synthesis of phase-pure BFO with a highly developed surface is solution combustion [13,14,15,16]. In our previous work, pure-phase BFO was synthesized by solution combustion method and its photocatalytic activity was studied [17]. The photocatalytic activity of BFO is highly dependent on particle size, morphology, crystallinity, and surface chemistry. Doping is known to be one of the main strategies for controlling material properties [18]. Doping of BFO, especially with rare earth elements, shows a significant improvement in the photocatalytic properties of the material [19,20,21,22]. In all of the mentioned studies, an increase in the photocatalytic activity of a doped material is reduced to the appearance of new energy states in the band gap, a narrowing of the band gap, and a decrease in particle size, leading to more efficient separation of photogenerated charge carriers. However, it has been shown that, depending on the amount of dopant, in BFO doped with rare earth elements Sm, Nd, Ho, and Er, a structural phase transition from the rhombohedral phase to the orthorhombic phase can be observed [23,24,25]. At the same time, at certain doping values, the rhombohedral and orthorhombic phases coexist, forming a morphotropic phase boundary (MPB), leading to a sharp increase in the permittivity and piezoelectric coefficient [26]. This indicates that the presence of MPB in the material increases the role of polarization processes, which form locally internal electric fields, which will affect the behavior of photogenerated charge carriers. It should also be noted that the unique properties of BFO and materials based on it open up broad prospects for application in various magnetoelectric devices, spintronics, sensor technology, and magnetic memory [27,28,29,30].
In this work, the phase composition, morphology, structure, and catalytic properties in the photodegradation of MO of synthesized BFO and doped with samarium BFO as a function of samarium concentration are presented, and the formation of a morphotropic phase boundary in these systems and its role in photocatalytic activity are discussed.
2. Results
The first step in the synthesis process is the formation of a coordination complex of metal ions with glycine since the latter is a complexing agent:
Bi(NO3)3·5H2O + Fe(NO3)3·9H2O + Sm(NO3)3·6H2O +NH2CH2COOH →
Bi/Sm[Fe(NH2CH2COO)](NO3)3·H2O + NO2 + H2O
Bi/Sm[Fe(NH2CH2COO)](NO3)3·H2O + NO2 + H2O
Nitrate ions in the complex itself provide the combustion process with the necessary oxygen. The complex, when heated, decomposes to oxides of bismuth, iron, and samarium:
Bi/Sm[Fe(NH2CH2COO)](NO3)3·H2O → Bi2O3 + Sm2O3 + Fe2O3 + NO2 + H2O + CO2
Bi, Fe and Sm oxides combine to form Bi1−xSmxFeO3.
Bi2O3 + Fe2O3 + Sm2O3 → Bi1−xSmxFeO3
During self-combustion, non-stoichiometric reactions between Bi2O3, Fe2O3, and Sm2O3 occur in some parts due to temperature unevenness throughout the gel. Thus, in this case, BiFeO3 is always accompanied by the formation of a small number of side phases, such as Bi2Fe4O9, Bi25FeO39, etc. Another important reason for the formation of secondary phases is due to the narrow temperature range in which BiFeO3 crystallizes. Below is the complete stoichiometric reaction. In the case of combustion of the glycine-nitrate complex, mainly N2, CO2, and H2O are emitted in the form of gaseous products:
6Bi(NO3)3·5H2O + 6Fe(NO3)3·9H2O + 6Sm(NO3)3·6H2O + 32NH2CH2COOH =
6(Bi/Sm)FeO3 + 43N2 + 200H2O + 64CO2
6(Bi/Sm)FeO3 + 43N2 + 200H2O + 64CO2
To study the morphology of the obtained samples, images were obtained by a scanning electron microscope (Figure 1 and Figure 2).

Figure 1.
SEM images of Bi1−xSmxFeO3 (x = 0.00—1, 0.05—2, 0.10—3, 0.15—4, 0.20—5).

Figure 2.
SEM images of Bi1−xSmxFeO3 (x = 0.00—1, 0.05—2, 0.10—3, 0.15—4, 0.20—5).
As can be seen from Figure 1, the samples have a highly porous structure. The interstices have an irregular shape, and their size varies in the submicron range. Cavities in this case are created due to the rapid release of many combustion gases. The nanoparticles are in agglomerates that are densely packed and have a uniform morphology. This morphology of the samples makes it almost impossible to analyze the shape or size of the grains. Higher resolution images (Figure 2) show that with increasing Sm concentration, the surface becomes less homogeneous and more developed, which indicates the presence of point structural defects on the surface caused by the substitution of Bi atoms (1.03 Å) in the BiFeO3 crystal structure into Sm atoms (0.958 Å), the ionic radius of which is smaller [31].
Surface inhomogeneity plays a significant role in processes on the surface. Foreign atoms form dipole moments on the surface, which can differ greatly in magnitude and direction from the core [32,33]. We believe that this effect should further increase the photocatalytic activity of the material since photocatalysis is a surface process. The sizes of agglomerates upon alloying from 5 to 10% decrease noticeably and, at the same time, porosity increases. With a further increase in the Sm concentration from 15 to 20%, particle enlargement and the formation of massive agglomerates up to 10 µm in size are observed.
This effect can, in our opinion, be explained by two factors:
- (1)
- As mentioned above, the ionic radius of Sm is smaller than that of Bi. Therefore, an increase in % substitution will reduce the size of crystallites, and, consequently, nanoparticles. As the particle size decreases, the contribution of van der Waals interactions between individual particles becomes significant. Due to these interactions, individual nanoparticles are agglomerated.
- (2)
- When Bi atoms are replaced by Sm atoms, a crystal lattice stress is created, and in this case, an excess dipole moment is accumulated on the surface of the nanoparticles, and the surface has an excess of free energy. However, as is known, from a thermodynamic point of view, the material will be the most stable when the Gibbs free energy is minimal. Therefore, under equilibrium conditions, the shape of the crystal tends to be one in which the value of the surface energy of the crystal is minimal. Since one of the main ways to reduce the surface energy of nanoparticles is to reduce the total surface area, agglomeration is observed.
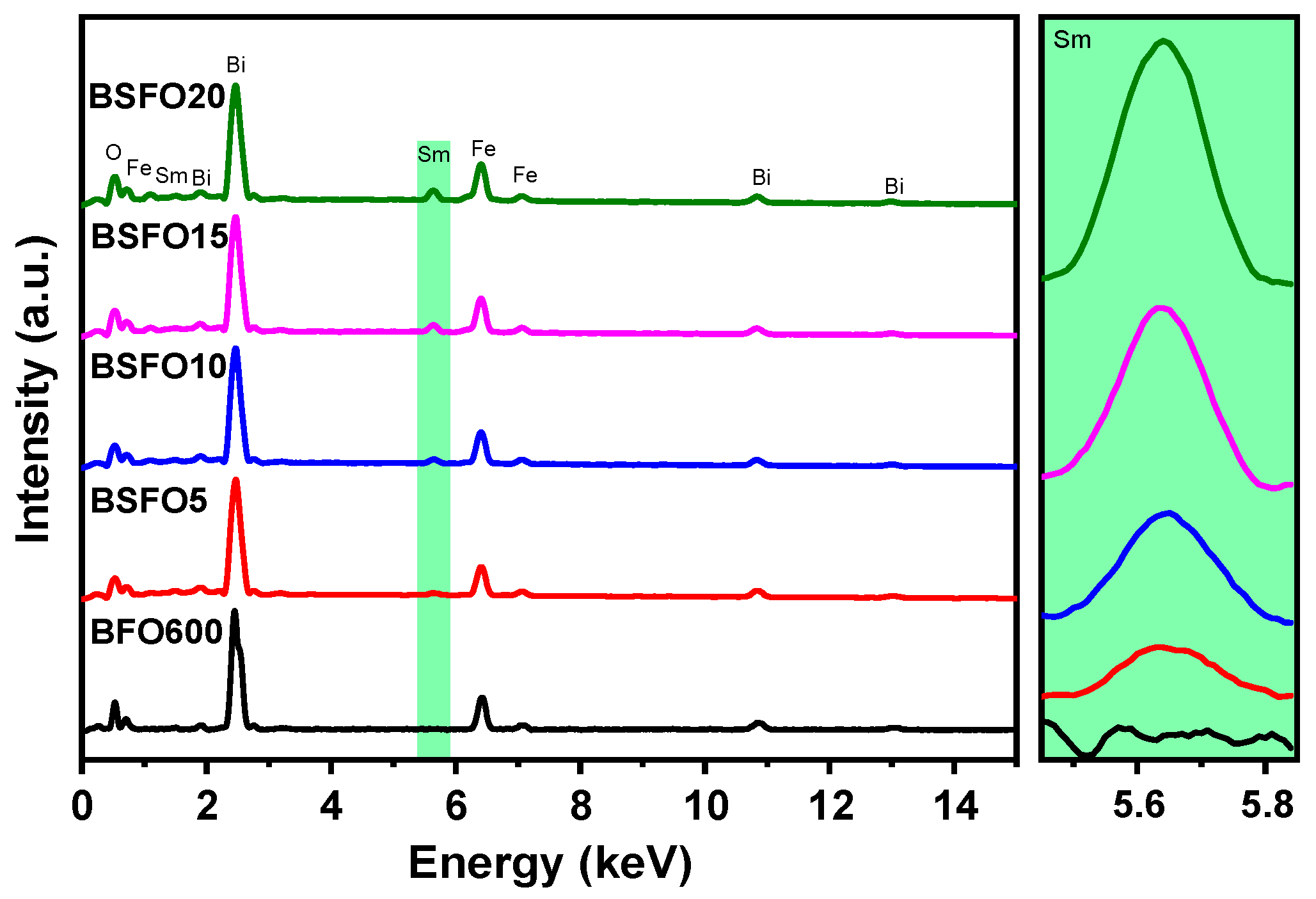
To confirm the presence of Sm atoms in the synthesized materials and the absence of additional impurities, we performed the elemental analysis of the samples. The analysis was carried out by a scanning electron microscope over the total area of the images at 250 × 250 µm. Figure 3 shows the energy-dispersive spectra of the samples. It can be seen from the spectra that the samples contain only Bi, Fe, O, and Sm atoms. The inset to the right of Figure 3 shows that the Sm concentration increases logically from 5 to 20% doping. To confirm the assumptions associated with structural changes in the sample during doping and the data of scanning electron microscopy, X-ray diffraction, and X-ray phase analysis were carried out.
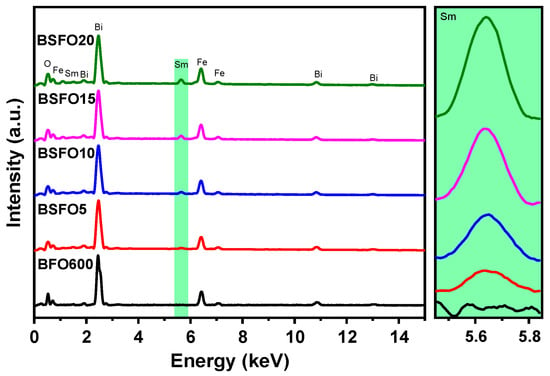
Figure 3.
Energy-dispersive spectra of Bi1−xSmxFeO3 samples (x = 0.00, 0.05, 0.10, 0.15, 0.20).
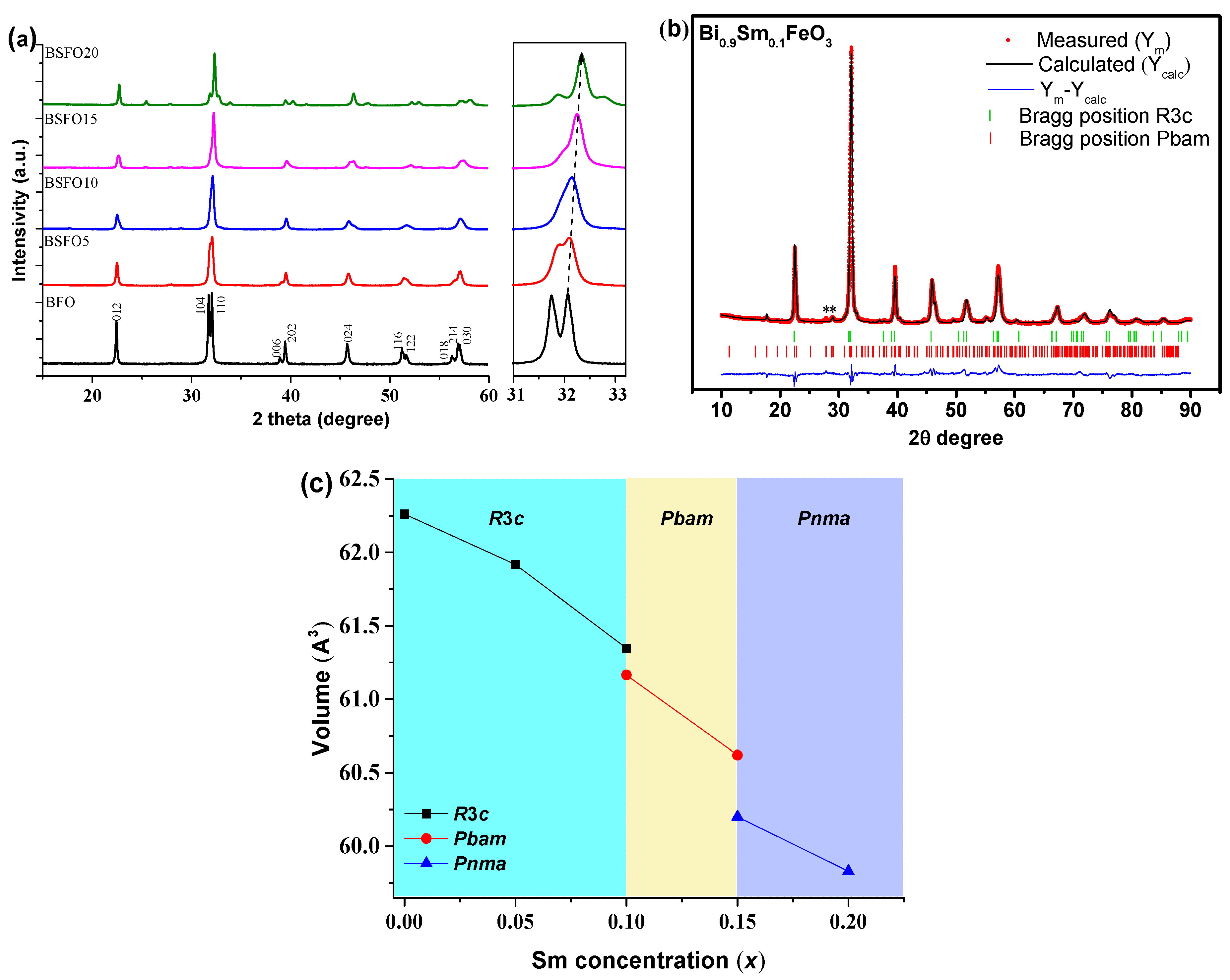
Figure 4 shows X-ray diffraction patterns of nanoparticle samples containing various Sm doping concentrations. The XRD patterns of the Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20) powders calcined at 600 °C for 30 min are shown in Figure 4. Phase analysis showed that in almost all samples, a small amount of impurities (less than 1% of phases), such as Bi2Fe4O9 and Bi25FeO39, can be observed [34,35,36]. In the case of doped BSFO formulations in the concentration range 0.05 ≤ x ≤ 0.20, the diffraction peaks are shifted towards higher values, which indicates lattice distortions (Figure 4a) because of the smaller ionic radius of samarium.
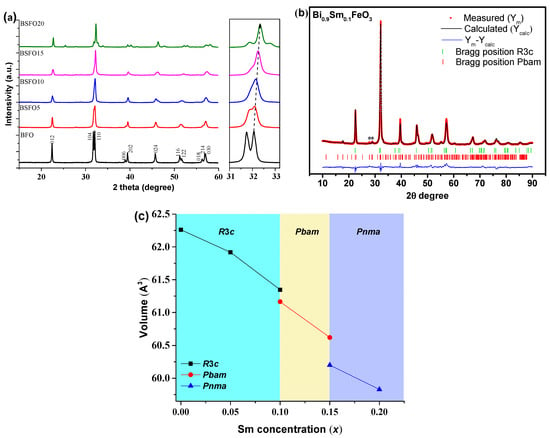
Figure 4.
(a) Results of diffraction analysis of Bi1−xSmxFeO3 samples (x = 0.00, 0.05, 0.10, 0.15, 0.20), (b) refined X-ray pattern by the Rietveld method of the Bi0.9Sm0.1FeO3 sample; (c) unit cell volume depending on x.
The compositions of Bi1−xSmxFeO3 (x = 0.00; 0.05) crystallize in a rhombohedral structure with the space symmetry group R3c. When the samarium concentration is increased to 10%, the crystal structure of the BSFO is described by a two-phase model: along with the rhombohedral phase R3c, the antipolar orthorhombic Pbam phase is formed in the ratio ~1:1 (Table 1).

Table 1.
Structural parameters were obtained from the Rietveld refinement of X-ray diffraction patterns of Bi1−xSmxFeO3 samples with x = 0.00, 0.05, 0.10, 0.15, and 0.20.
The Pbam space group suggests the antipolar nature of the displacement of ions in the A- and B-positions of perovskite [22,26]. It should be noted that in a few works [37,38,39,40,41], it is reported that a 10% substitution of Bi3+ for Sm3+ does not lead to any structural changes that are possible at a higher concentration. However, refinement of the XRD patterns of this composition by the Rietveld method indicates the presence of a new phase of Pbam. Sample BSFO15 (Bi0.85Sm0,15FeO3) is also described by a two-phase model however in this case the R3c phase is absent and a new Pnma phase appears. In the case of BSFO20 (Bi0.8Sm0.2FeO3), the structure is completely described by a single-phase model with the Pnma space group. The average crystallite size, calculated by Scherrer’s formula, is 56, 40, 35, 34 and 30 nm for x = 0.00, 0.05, 0.10, 0.15 and 0.20, respectively. An increase in Sm content would result in a unit cell volume contraction, because the ionic radius of Sm3+ is smaller than that of Bi3+ [42] (Figure 3). With increasing samarium fraction, the size of crystallites decreases.
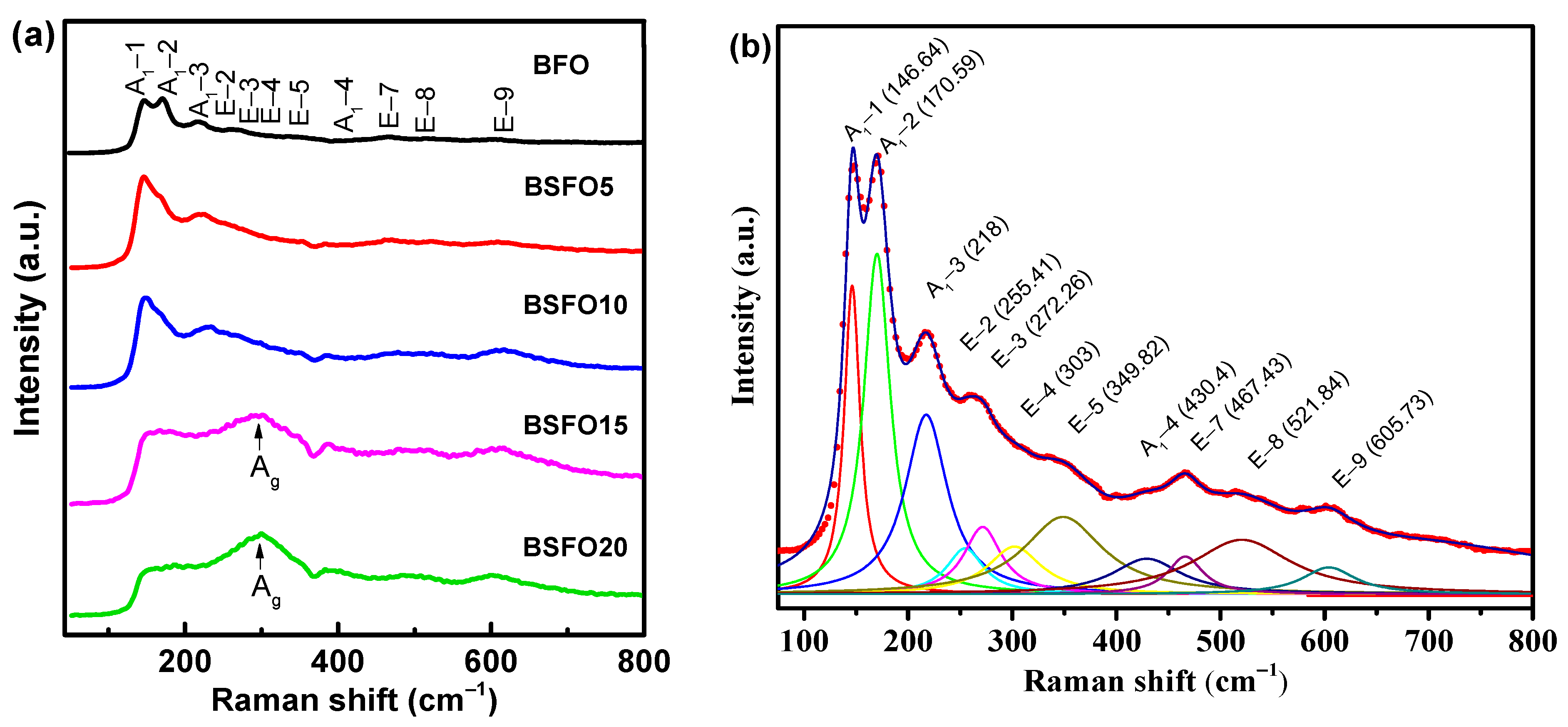
Raman spectra for BSFO, taken at room temperature, are shown in Figure 5.
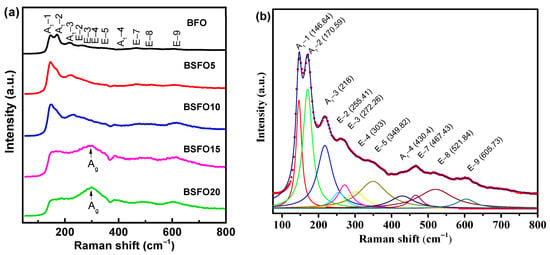
Figure 5.
(a) Raman spectra of Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20). (b) Lorentz-fitted Raman spectrum of pure BFO.
For rhombohedral BFO with space group R3c, thirteen (4A1 + 9E) active modes are expected [43,44]. For composition x = 0.00, eleven Raman modes were observed in total: 146.64 cm−1 (A1-1), 170.59 cm−1 (A1-2), 218 cm−1 (A1-3), 430.4 cm−1 (A1-4), 255.41 cm−1 (E-2), 272.26 cm−1 (E-3), 303 cm−1 (E-4), 349.82 cm−1 (E-5), 467.43 cm−1 (E-7), 521.84 cm−1 (E-8), 605.73 cm−1 (E-9) (see Figure 5b). At 10% Sm substitution, the most intense mode (A1-1) shifts to 149 cm−1. At a Sm concentration of more than 10%, the Raman spectra undergo significant changes, indicating structural distortions. The mode (149 cm−1) disappears completely when the Sm concentration reaches x ≥ 0.15. The broadening of the Raman modes with an increase in the Sm concentration corresponds to a decrease in the average crystallite size, which is in agreement with the XRD data. For x = 0.15–0.20, a broad peak is observed at 300 cm−1. This is due to the Ag mode, which arises due to the vibration of Sm–O bonds in an orthorhombic unit cell [45]. The 260 cm−1 mode (E-mode) is associated with the Fe-O covalent bond and the corresponding Fe-O-Fe angle. Modes exceeding 600 cm−1 refer to second-order Raman scattering, which is associated with electron–phonon interaction in BiFeO3.
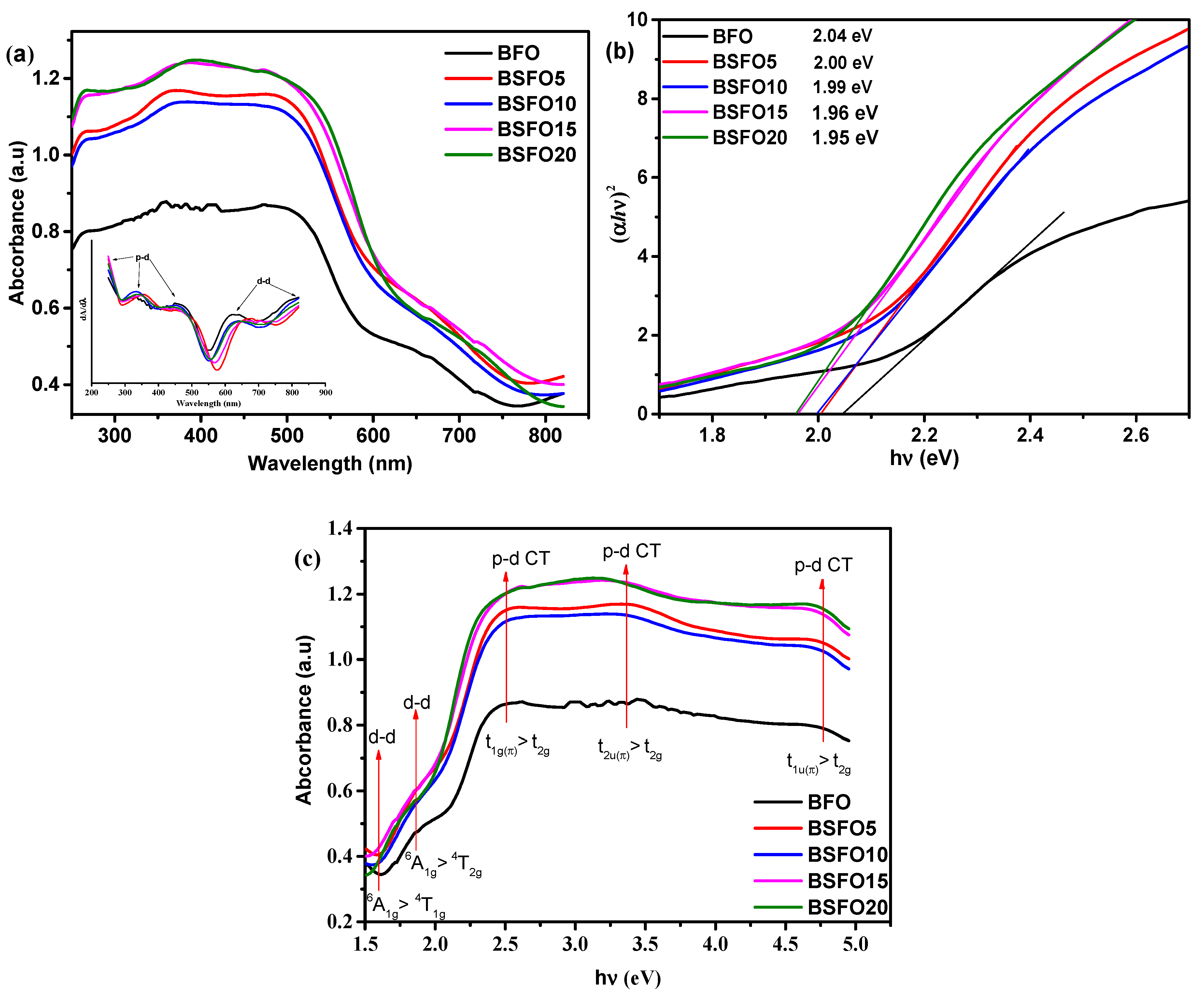
To study the optical characteristics of light absorption of photocatalysts Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20), diffuse reflectance spectra (DRS) of UV-Visible light were obtained. The results are shown in Figure 6.
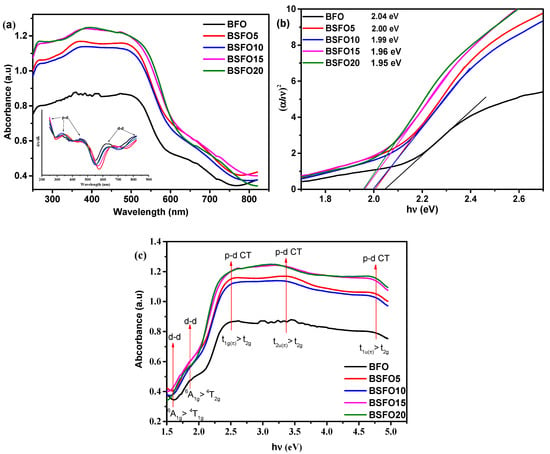
Figure 6.
(a) UV–Vis absorption spectra; (b) plot of (αhν)2 versus energy (hν) to determine the band gap energy. Lines indicate the direct extrapolation (DE) technique; (c) absorption vs. photon energy curves for Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20).
All samples exhibit strong light absorption in both the UV and visible region, demonstrating that the synthesized samples can exhibit photocatalytic activity under UV-Visible illumination. The edge of the absorption band of BFO nanoparticles is located near 600 nm, as pointed out by other authors [46]. Compared to pure BFO, samples doped with Sm show increased absorption capacity, especially in the visible light region. The intensity of absorption in this case gradually increases, with an increase in the content of the dopant. The spectra show five transitions, which are in the range from 1.5 to 4.5 eV. Similar results were also obtained by other authors [47,48]. From the DRS BFO spectrum, as shown in Figure 6, the shoulder centered at 1.6 and 2 eV corresponds to the 6A1g → 4T1g and 6A1g → 4T2g transitions, respectively, which arise due to d-d excitation of the crystal field of Fe3+ ions in BFO. These forbidden excitations appear due to the spin-orbit coupling, which weakens the spin selection rule [49,50]. Above 2 eV, absorption increases significantly, and peaks appear at ~2.5, ~3.3, and 4.5 eV, which can be attributed to p–d charge transfer (CT) excitations [49]. The peak of the CT transition centered at ~2.5 eV can be unambiguously attributed to the dipole forbidden t1g(π)→t2g p-d CT transition like other ferrites with FeO6 centers [51]. Intense bands near 3.3 and 4.5 eV are assigned to dipole allowed t2u(π)→t2g and t1u(π)→t2g p-d CT transitions in octahedral FeO6 centers, respectively [49]. For BFO compounds with samarium substitution, a slight redshift is observed at d-d and p-d CT transitions. This indicates that substitution in BFO increases the internal “chemical pressure”, which results from changes in the local environment in the FeO6 octahedra. This confirms the data of XRD spectra refinement by the Rietveld method, shown in Table 1, which indicates the decrease in the volume of the unit cell.
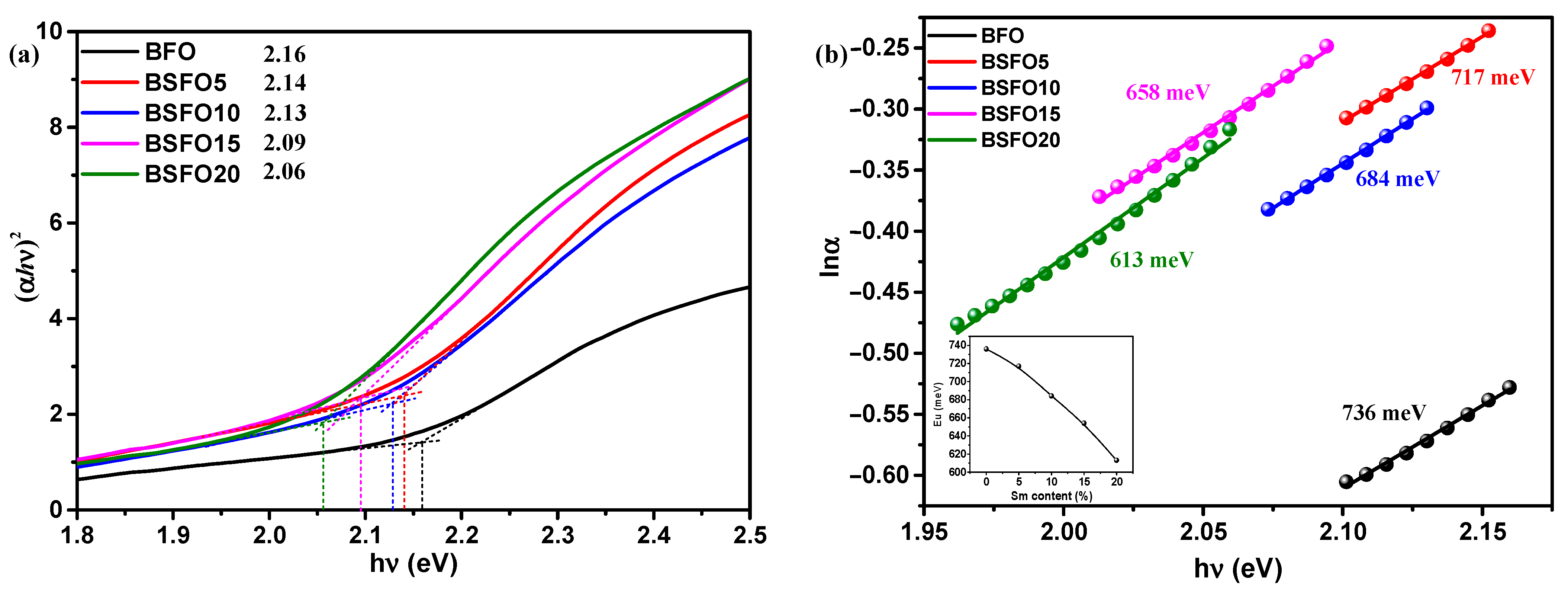
The optical band gap was calculated from the Kubelka–Munk plot (Figure 6b) plotted in the coordinates F((αhν)2) – (hν)), because BFO is a direct-gap semiconductor. The band gaps were 2.05, 2.00, 1.99, 1.96, and 1.95 eV for pure BFO, 5%, 10%, 15%, and 20% Sm doping, respectively. In this case, we applied the most common method for determining the band gap, the direct extrapolation (DE) method. The band gap is estimated by fitting a straight line to the linear part of the absorption spectrum. However, in [52], it is reported that this method is inaccurate. Proceeding from this, we estimated the value of the band gap using the “proper extrapolation” (PE) method, in which the point of intersection of linear approximations of the absorption edge and the baseline is taken as the band gap value. The results are shown in Figure 7a.
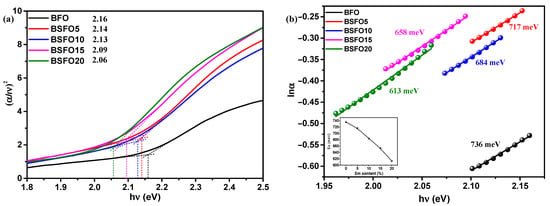
Figure 7.
(a) Plot of (αhν)2 versus energy (hν) to determine the band gap energy. Lines indicate the proper extrapolation (PE) technique; (b) lnα versus hν plots to determine the Urbach energy for Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20).
From a comparison of the two techniques for determining the band gap, it can be seen that the direct extrapolation gives slightly underestimated values. However, the nature of dependence remains the same, and the band gap decreases with increasing samarium content. A change in the band gap value can be caused by various reasons, such as structural distortions arising when Bi atoms are replaced by Sm, a change in the size of crystallites, and the presence of oxygen vacancies [53]. The creation of oxygen vacancies can be caused by valence nonstoichiometry of iron ions. In [8], we demonstrated that pure BFO synthesized by solution combustion is characterized by the presence of iron in +2 and +3 oxidation states. In addition, Sm doping creates localized states in the band gap near the valence band, which can also be regarded as a decrease in the band gap. The density of localized electronic states in synthesized systems can be estimated from the Urbach characteristic energy (Eu), which can be calculated from the rate of exponential decay of the “tail” in the absorption spectrum by equation α = αoexp(hν/Eu). The width of the Urbach tail is an indicator of the disorder in the material. Thus, Eu can be calculated from lnα and hν as the inverse slope of the curve (Figure 7b). The calculated value of Eu near the edge is 736 meV for the original BFO sample and decreases monotonically to 613 meV for the BSFO20 sample (see inset in Figure 7b). A decrease in Eu values indicates that Sm doping reduces the number of oxygen vacancies. All data on optical measurements are given in Table 2.

Table 2.
Optical properties of Bi1−xSmxFeO3 samples with x = 0.00, 0.5, 0.10, 0.15, and 0.20.
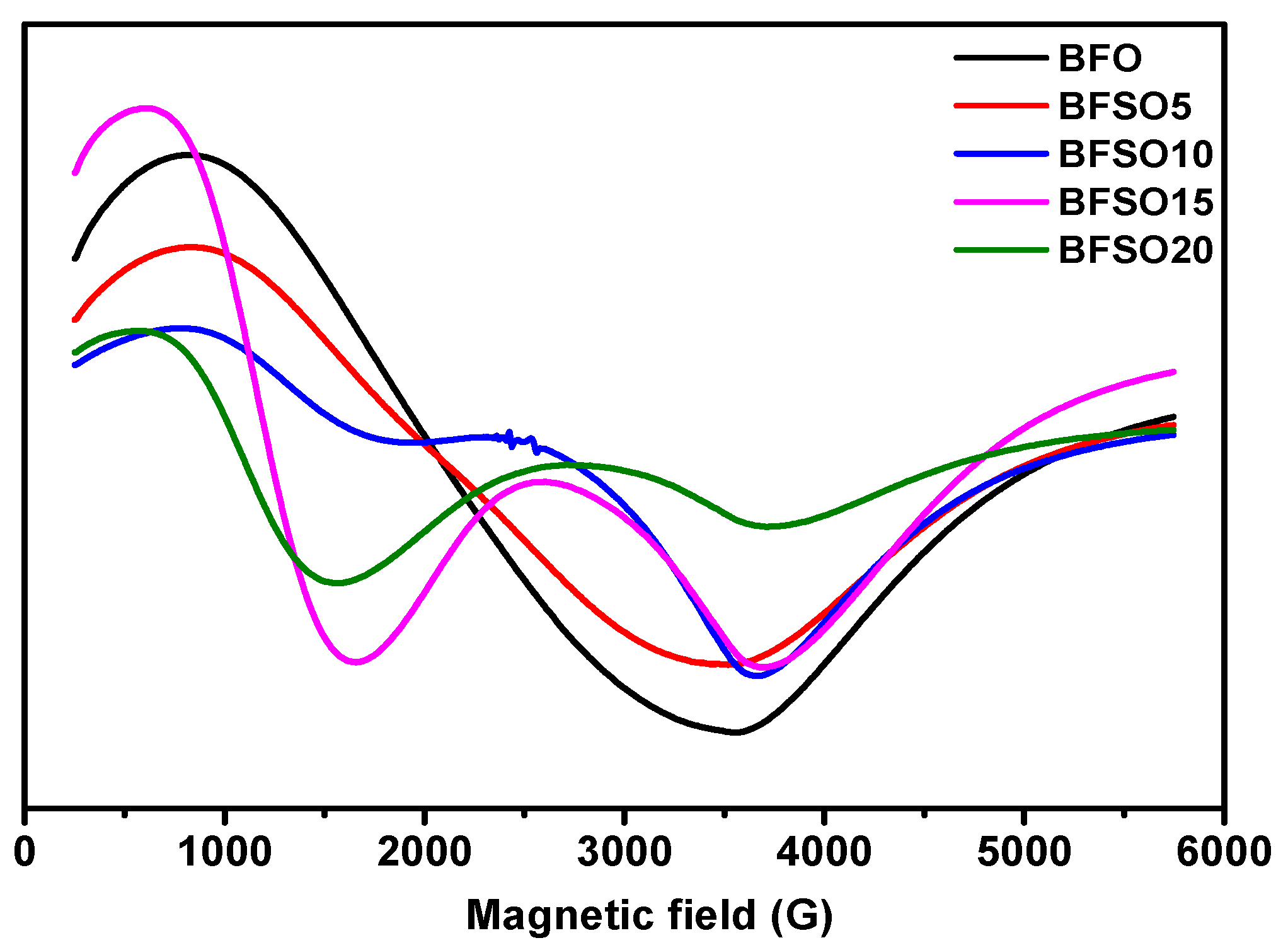
Electron magnetic resonance (EMR) is a useful tool for studying spin dynamics in ferromagnetism and antiferromagnetism [54]. Measurement of the magnetic moment provides information on local magnetic properties, the nature of spin–spin interactions, the distribution of the internal field, and spin-lattice correlations. In Figure 8 EMR spectra of BFO and Bi1−xSmxFeO3 with different concentration of samarium are presented.
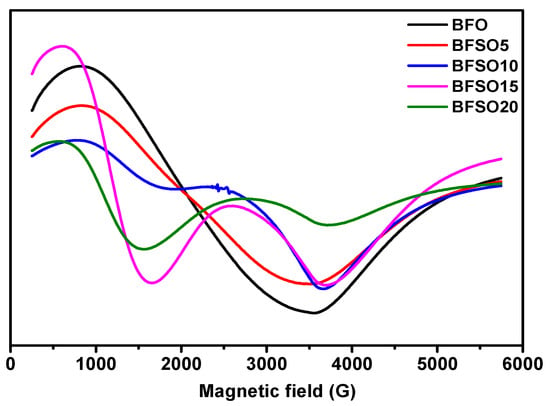
Figure 8.
EMR spectra of Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20).
As shown in Figure 8, Sm doping strongly affects the character of the EMR spectra. EMR spectra can be divided into two resonant regions, which are associated with the presence of various types of defects and magnetic anisotropy: the low-field (LF) and high-field (HF) resonance regions. As a rule, magnetic dipole interactions and superexchange interactions between magnetic ions through oxygen ions are two important factors that determine the values of the g factor and the width of the resonance line. Superexchange interactions usually increase when the distance between magnetic ions and oxygen ions decreases, and the corresponding bond angles are close to 180. Strong dipole interactions give a large resonant linewidth and g value, while strong superexchange interactions give a relatively small linewidth and g value.
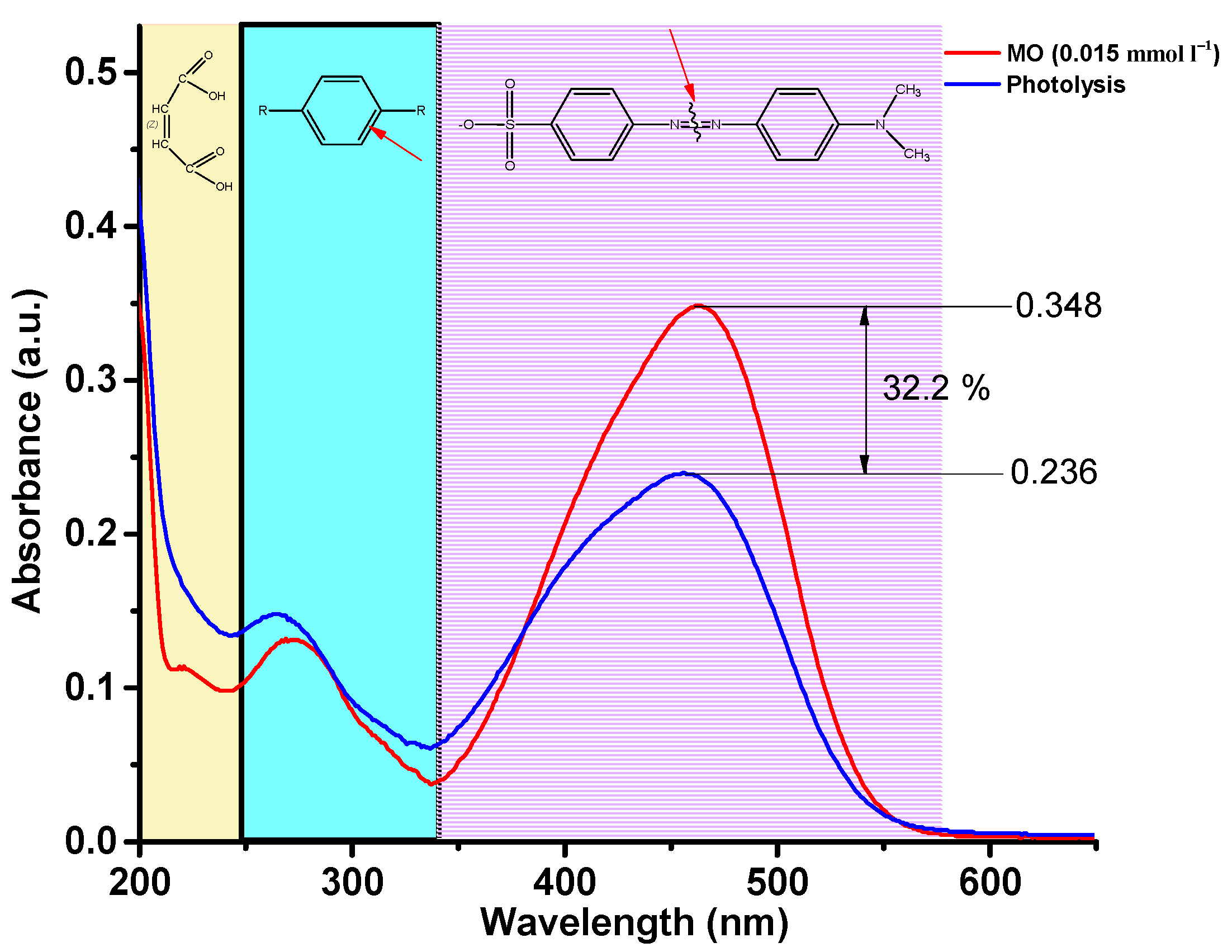
The EMR spectrum of pure BFO powder gives an almost isotropic signal with a g factor of 3.3280 and a width of ∆B = 277.0 mT. The EMR spectrum of BSFO5 is also a wide signal; however, it is slightly asymmetric and consists of a superposition of two signals with a total effective g-factor of 3.2847 and a width of ∆B = 271.0. The presence of a high-field isotropic signal is because the magnetic moments in the sample are randomly oriented with respect to the external magnetic field. For the samples with a high content of samarium (10, 15, 20%), EMR spectra are observed, consisting of a superposition of two signals, the intensity ratio of which depends on the Sm content in the samples. One of the signals is in the low-field resonance arm with g = 6.4934, which is characteristic of magnetically isolated high-spin Fe3+ ions (S = 5/2) in a medium with low symmetry (tetragonally coordinated Fe3+ ions), corresponding to Fe3+–Vo•• defective dipoles. Another signal is a high-field resonance with g = 2.118 associated with the presence of Fe3+ ions in the octahedral field because of superexchange interactions in the lattice [55], which can also be related to resonant absorption in the cycloidal spin structure and defects caused by uncompensated spins. The splitting of the resonant signal is obviously associated with a change in the magnetic medium for unpaired electrons in Fe ions, as well as the possible presence of a secondary magnetic phase. All obtained samples were studied in the process of photocatalytic decomposition of MO. To exclude the effect of MO photolysis under the action of UV-Vis irradiation, a blank experiment was carried out. Figure 9 shows the absorption spectra of the initial MO solution and the MO solution after 180 min of irradiation.
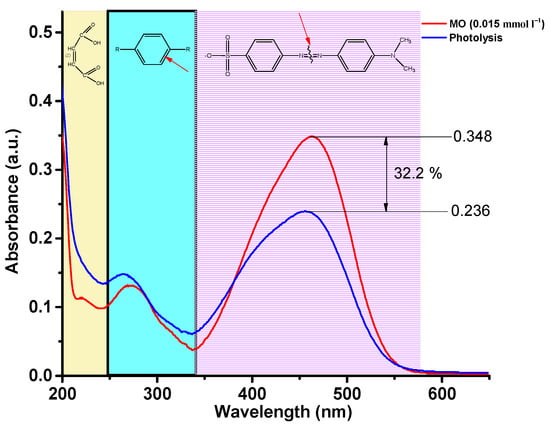
Figure 9.
Absorption spectra of MO solution over time during photolysis.
Three regions can be clearly distinguished on the spectrum of the initial MO solution, 340–580 nm, characterizing the presence of –N=N– bonds, 240–340 nm, characterizing the presence of aromatic rings, and 200–240 nm, corresponding to the absorption of unsaturated aliphatic acids. As can be seen, after three hours of irradiation, the concentration of the MO solution decreased by 32.2%. At the same time, it should be noted that the peaks at 200–240 and 240–340 nm increase, which indicates that the –N=N– bond of MO molecules is broken, and many intermediate aromatic and aliphatic molecules are formed. Figure 10 shows the absorption spectra of the MO solution over time in a photocatalytic experiment using BSFO5 as a photocatalyst.
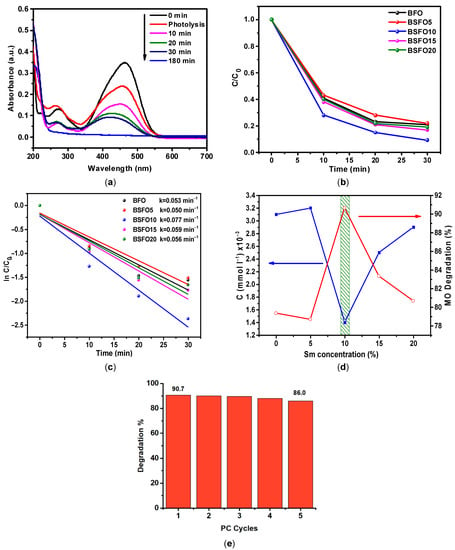
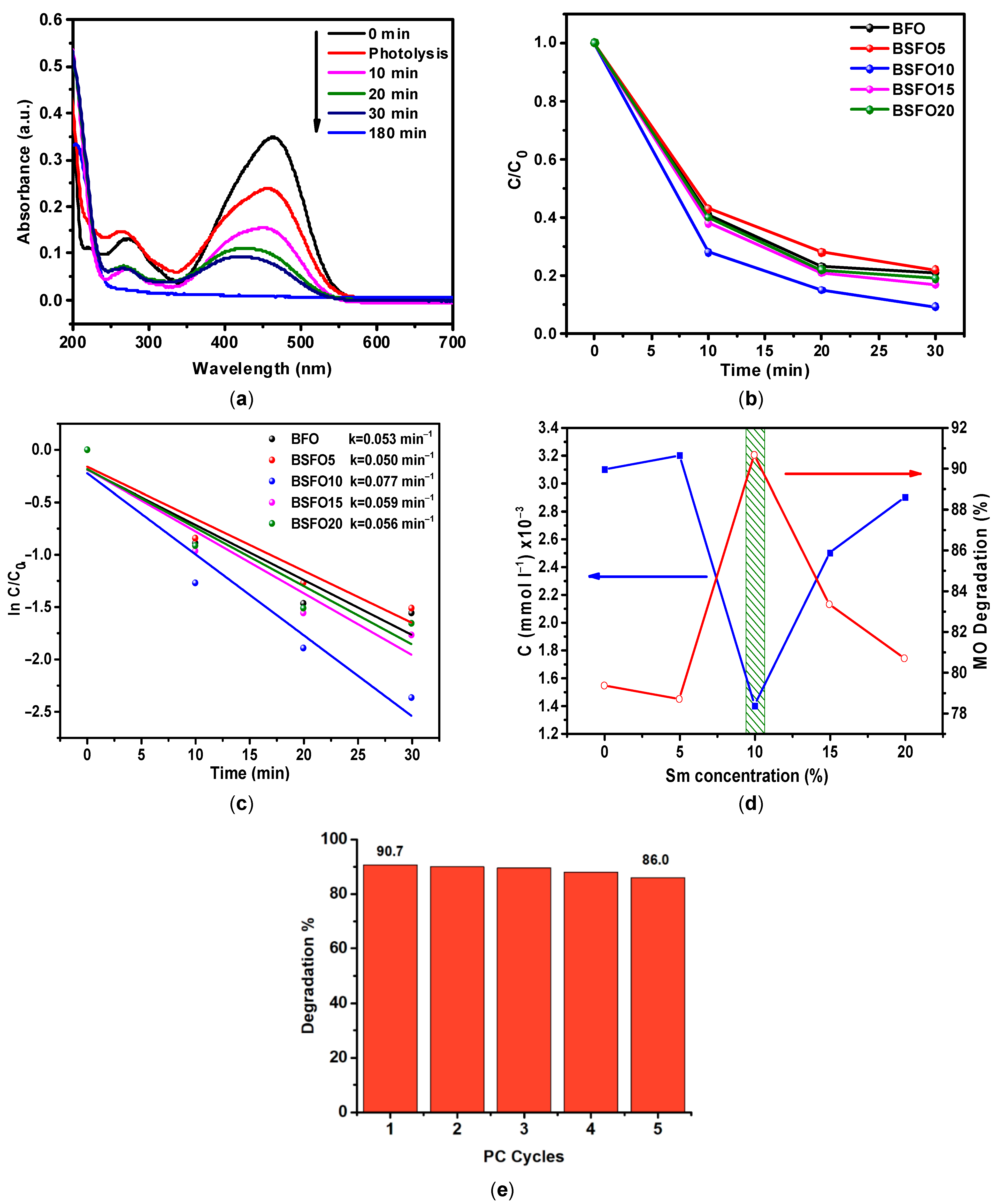
Figure 10.
(a) The absorption spectra of MO solution over time in a photocatalytic experiment for BSFO5. (b) C/C0 curve of MO degradation. (c) Pseudo-first order kinetics of the photocatalytic reactions. (d) Comparative photocatalytic activity of Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20). (e) Photocatalytic degradation of MO with the BSFO10 sample for five cycles.
It can be seen from the data that 78.2% of the dye decomposes during the first 30 min of the process. It is also seen that the peak at 240–340 nm, which characterizes the presence of aromatic compounds, decreases significantly, but the peak associated with the presence of unsaturated aliphatic compounds in the solution increases strongly. This indicates that highly oxidizing radicals are generated in the solution during the photocatalytic process, which effectively decomposes aromatic organic compounds. On the spectrum of the MO solution after 180 min, there is only a peak characterizing the presence of aliphatic compounds. Similar measurements were carried out with samples doped with Sm 10, 15, 20%, as well as pure BiFeO3. The data are presented in Figure 10b as a C/C0 ratio plot. It can be observed from the obtained results presented in Figure 10c that the process corresponds to pseudo first-order kinetics. For easier comprehension, the data are presented in Figure 10d. It can be seen from the data that the Bi/Sm substitution increases the photocatalytic activity of BFO. In this case, the sample with 10% Sm with a substitution of more than 90% in 30 min of the process exhibits the highest photocatalytic activity. It can also be seen that the photocatalytic activity of pure BFO and 5% Sm of the substituted sample is almost the same. The results once again confirm that the concentration of Sm3+ substitution can affect the photocatalytic activity of BFO and that there is an optimal doping concentration of Sm3+ ions. The stability and reusability of a photocatalyst is an important parameter for practical applications. To evaluate the stability and reusability of BSFO10 sample, photocatalyst was recycled for five runs, as depicted in Figure 10e. After five successive runs, the degradation efficiency could be largely maintained, indicating good stability.
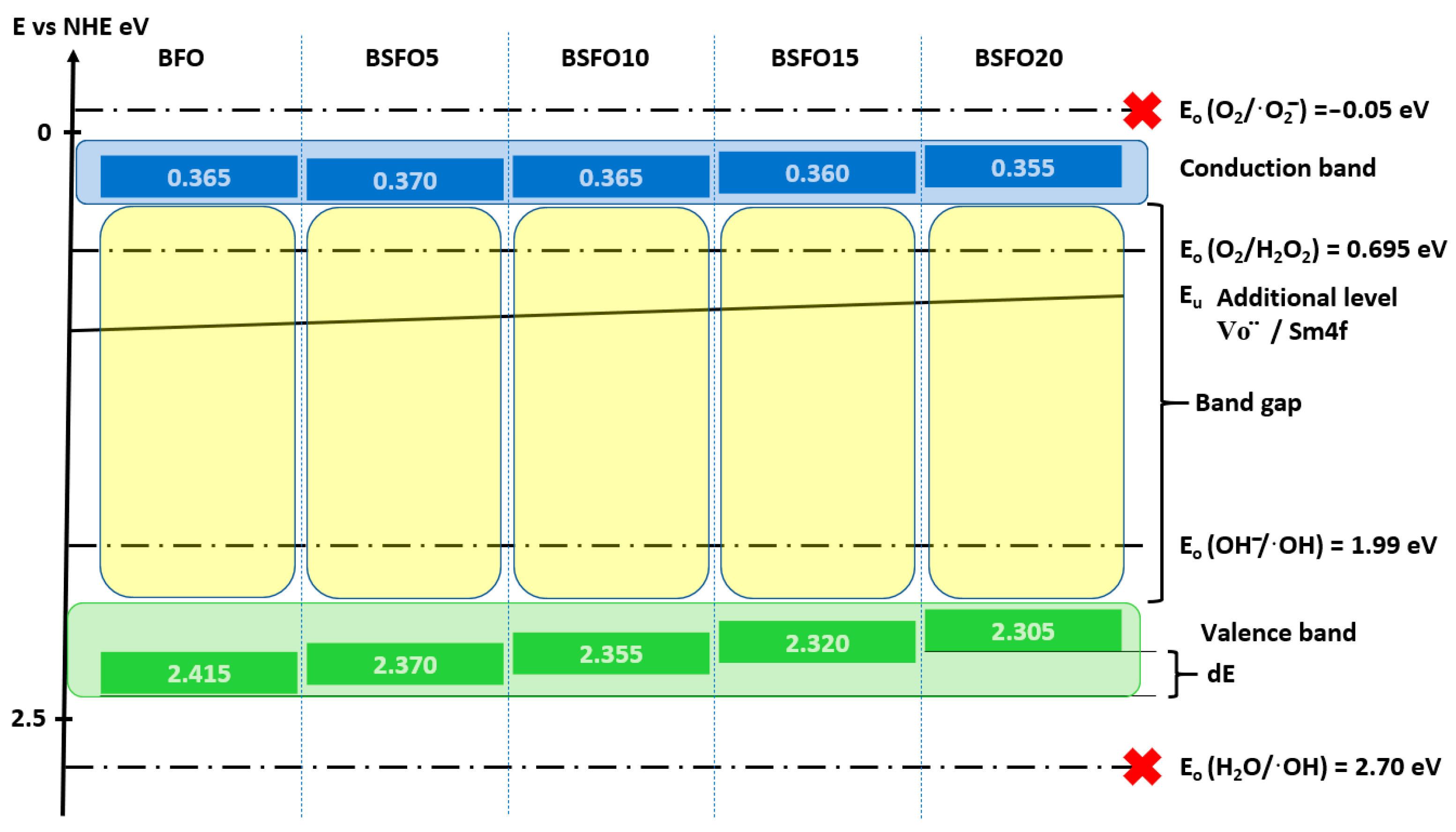
The formation of highly active oxidants is closely related to the energy of the conduction band (CB) and the valence band (VB) of the semiconductor. The CB and VB potentials of BiFeO3 were determined using Mulliken’s theory of electronegativity.
where EVB is the valence band potential, X is the electronegativity of the semiconductor, χ is the electronegativity of the element, Ee is the standard potential of the hydrogen electrode (~4.5 eV), EBG is the optical band gap, ECB is the conduction band potential.
EVB =X − Ee + 0.5EBG
ECB = EVB − EBG
The electronegativity of the semiconductor was calculated according to Equation (7):
and the electronegativity of a neutral atom according to Mulliken is the arithmetic mean of the affinity of an atom for an electron (EEA) and the first ionization potential (EIP):
Since the values of ionization potentials and electron affinities for most atoms are given in tables, Mulliken’s electronegativity becomes a parameter with absolute electronegativity values based only on measurable physical quantities. In a sense, Mulliken’s electronegativity is the electrochemical potential of an electron in a neutral atom. The layout of the energy levels is shown in Figure 11.
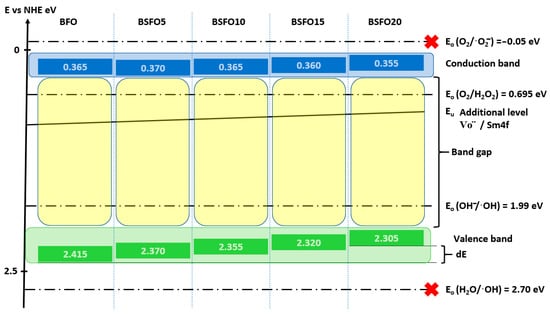
Figure 11.
Relative band position and possible photocatalysis mechanism for Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20) under UV-Vis light illumination.
Calculations showed that doping does not affect the energy position of the CB remaining at a level of ≈0.4 eV, while the top of the VB shifts monotonically from 2.415 eV for BFO to 2.305 eV for BSFO20. It is also seen that the position of the Urbach energy, caused by additional energy levels in the BG, shifts slightly to the bottom of the VB. This confirms the conclusion that the proportion of oxygen vacancies decreases with increasing doping. Based on these results, a possible mechanism for the photocatalytic reaction can be proposed. When BiFeO3 is irradiated with light with an energy comparable to or greater than the BG, an electron–hole pair (h+/e−) is generated (9).
BFO + hv → h+ + e−
The electrons in the BG of BiFeO3 are not able to reduce O2 to ·O2−, since the BG potential of all samples (≈0.4 eV vs. NHE) is more positive than the standard redox potential E0 (O2/·O2−) (−0.18 eV vs. NHE). However, the band gap potential of BiFeO3 is more negative than the standard redox potential E0 (O2/H2O2) (0.695 eV vs. NHE), so oxygen adsorbed on the semiconductor surface will react with two electrons to form H2O2 (10), which subsequently, when interacting with an electron, forms ·OH (11).
O2 + 2e− + 2H+ → H2O2
H2O2 + e− →·OH + OH−
The VB of BiFeO3 (≈2.4 eV relative to NHE) is more positive than the standard redox potential E0 (·OH/OH−) (1.99 eV relative to NHE), which indicates that holes localized in the VB can oxidize adsorbed OH− with the formation of ·OH (12).
OHad + h+ → ·OH
Thus, we can conclude that the main route of MO decomposition is oxidation by hydroxyl radicals. The discrepancies in the photocatalytic activity of Bi1−xSmxFeO3 (x = 0.00, 0.05, 0.10, 0.15, 0.20) can be explained by the following factors:
- Firstly, Sm doping reduces the band gap and increases optical absorption in the UV and visible light region. This means that since more charge carriers will be generated when irradiated with UV-visible light, the efficiency of photodegradation increases. However, in our case, with doping, the width of the band gap systematically decreases; however, the photocatalytic activity increases only up to x = 0.10, and then a noticeable decrease is observed. Therefore, other reasons need to be considered.
- Secondly, the Sm3+ substitution causes lattice deformation and, by changing the local environment of atoms, modifies the electronic structure. Since rare earth elements are known to be good electron acceptors [56], they will act as traps to capture excited electrons, which probably facilitates the separation of photogenerated electron–hole pairs and prolongs the lifetime of charge carriers [57], which ultimately enhances photocatalytic activity. However, excess amounts of Sm3+ dopant can act as recombination centers in BFO, resulting in low PC activity at high doping %.
- Thirdly, the rhombohedral R3c phase in BFO and BSFO5 is noncentrosymmetric (polar), has ferroelectric properties, and exhibits spontaneous polarization. The orthorhombic Pbam phase with antipolar Bi-O and Sm-O dipole moments is antiferroelectric, exhibiting weak spontaneous polarization, which is compensated within the unit cell. The orthorhombic Pnma phase in BSFO15 and BSFO20 is centrosymmetric, paraelectric, and nonpolar. In the absence of an internal electric field in these samples, photogenerated electrons and holes easily recombine, which should significantly reduce photocatalytic performance compared to BFO, BSFO5, and BSFO10.
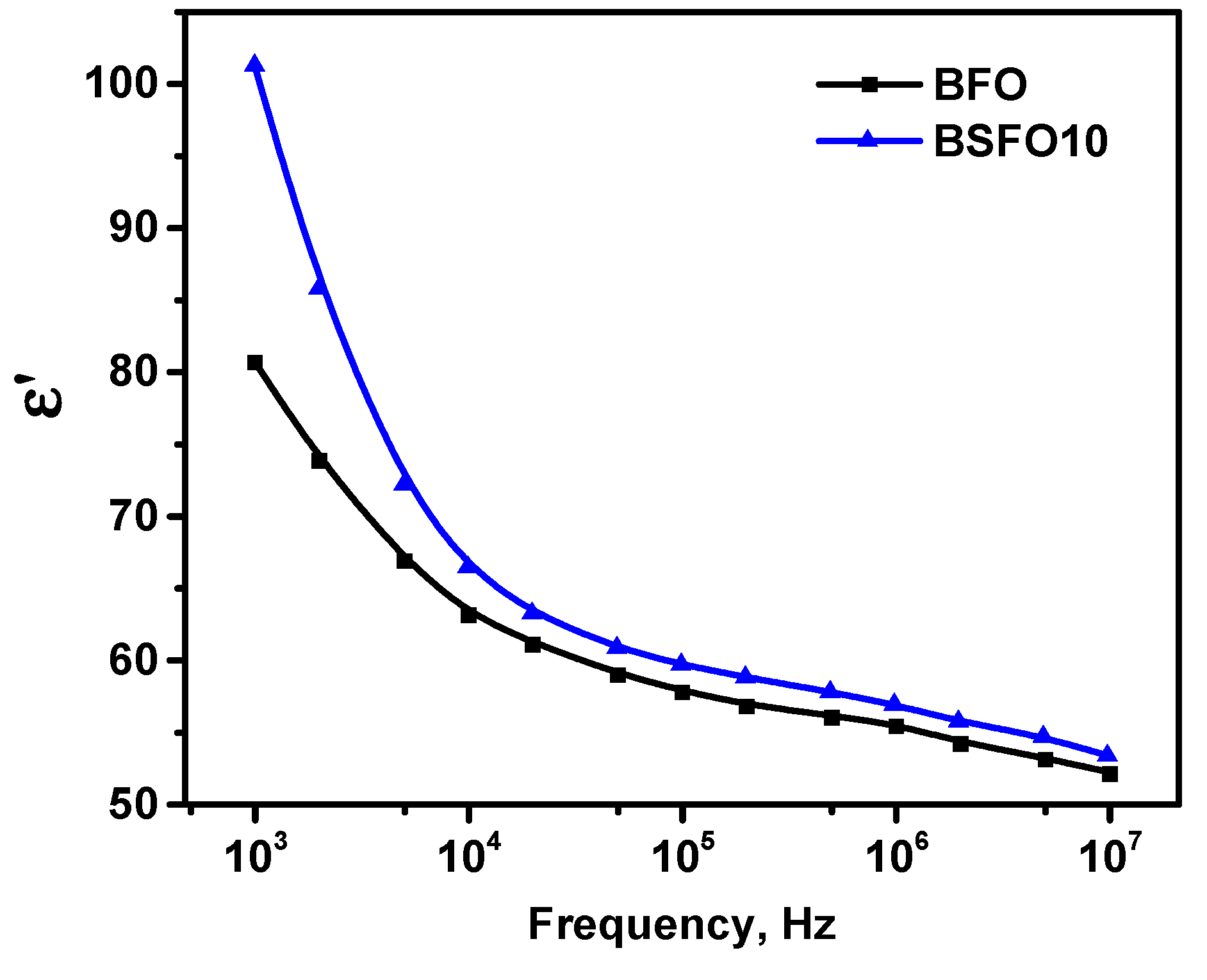
Thus, we confirm that the emerging internal electric field in polar noncentrosymmetric structures can separate photogenerated electrons and holes, reducing charge recombination. However, the fact that the PC activity of BSFO10, in which polar–antipolar phases coexist, is higher than that of BFO with a purely polar phase, seems illogical. To understand this effect, we performed dielectric measurements. It is known that material properties determined by polarization change, such as dielectric constant, can be enhanced in phase transition regions where there is a significant change in polarization [58]. Figure 12 shows the frequency dependences of the permittivity for pure BFO and BSFO10.
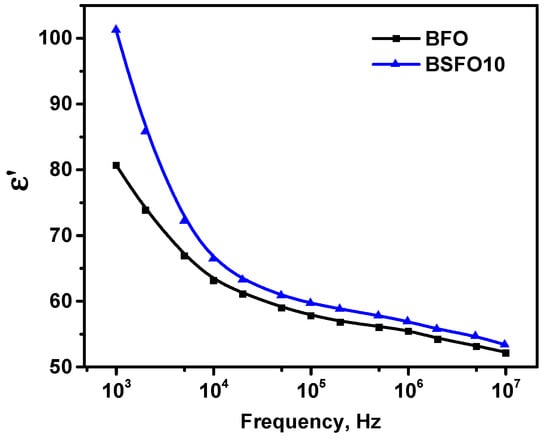
Figure 12.
Frequency dependence of permittivity for pure BFO and BSFO10.
For both samples, the permittivity decreases with increasing frequency, which can be explained by the effect of space charge relaxation based on polarization processes. The improvement in permittivity at x = 0.10 can be explained by the replacement of Bi3+ by smaller Sm3+, which provides an increase in the dipole moment. On the other hand, the contribution to the permittivity is made by space charges accumulating at the grain boundaries. Since at x = 0.10 the grain size decreases compared to pure BiFeO3, this leads to an increase in the grain boundaries. In addition to intergrain boundaries, the charges accumulated at the interphase boundaries of the MPB inside the grains contribute to the enhancement of the permittivity. Table 3 shows the comparative characteristics of the catalytic activity of some doped BFO catalysts.

Table 3.
Comparison of catalytic activity between some doped BFO catalysts.
From the presented data, it can be seen that our material is significantly superior in terms of its photocatalyst properties to photocatalysts of a similar composition from the works of other authors.
3. Materials and Methods
3.1. Synthesis of Bi1−xSmxFeO3 Nanoparticles
Nanoparticles of BiFeO3 were obtained using a new method of synthesis [8]. Analytically pure bismuth (Bi(NO3)3·5H2O), iron (Fe(NO3)3·9H2O), samarium (Sm(NO3)3·6H2O) nitrates and glycine were taken according to the stoichiometric ratio. Glycine played the role of fuels, which provided a platform for redox reactions between reagents during combustion. Metal nitrates are hygroscopic, and they tend to form a suspension mixture when mixed with glycine. The mixture was heated to ~300 °C for dehydration and initiation of the combustion process. Since metal nitrates also play the role of oxidizing agents, the combustion process can occur efficiently using the oxygen contained in the reactants themselves. As a result of the combustion process, ash was formed, and the yellow vapor was released. Then, the sample was heat-treated at temperatures of 600 °C for 30 min. The stoichiometry of the combustion reaction was calculated using the concepts of propellant chemistry [17], which shows a simple method for calculating the redox valences of the mixture components. According to these concepts, metals, carbon, and hydrogen are considered reducing elements with corresponding metal valences, +4 for carbon and +1 for hydrogen. Oxygen is considered to be an oxidizing agent with a valence of −2, and the valence of nitrogen is 0. The combustion reaction is highly exothermic, and the maximum combustion temperature is achieved when the equivalence ratio (φe is the oxidizer/fuel relation) is equal to unity. The equivalence relation was found according to [64]. In this work, we synthesized Bi1−xSmxFeO3 precisely at φe = 1. The resulting Bi1−xSmxFeO3 (x = 0, 0.05, 0.10, 0.15, 0.20) samples will be referred to below as BFO.
3.2. Characterizations
X-ray diffraction studies were performed using an Empyrean PANalytical X-ray diffractometer using the radiation of a copper anode with a nickel filter. Data processing was performed using the High Score Plus application program, included in the instrument software, and the diffraction database PDF-2. The morphology of the obtained samples was studied using an ASPEX EXpress scanning electron microscope equipped with an energy dispersive X-ray spectrometer (EDS). The morphology was studied in the mode of detection of secondary electrons. Raman spectra were measured by Ntegra Spectra (λ = 532 nm laser). The electron paramagnetic resonance spectra were recorded at room temperature using an EMXmicro spectrometer, Bruker, Germany, with an operating frequency of 9.8 GHz. Diffuse reflection spectra (DRS) in the coordinates F(R) = f(λ, nm), where F(R) is the Kubelka–Munk function, were performed on a Shimadzu UV-3600 spectrophotometer with an integrating sphere LISR-3100. To determine the width of the band gap of the obtained materials, we used the method of constructing the Kubelka–Munk curve by converting the optical absorption spectra using the values of the absorption coefficient (K) and the photon energy (hv) using the wavelengths: (hv = 1240/λ). The graph was plotted in coordinates (Khv)1/2 to (hv) and by extrapolating the curve to the zero value of the absorption coefficient, it was possible to find the energy value of the band gap.
3.3. Photocatalytic Measurements
The photocatalytic characteristics of the catalysts were evaluated by photodegradation of methyl orange (MO) in an aqueous solution (0.015 mmol/L). Photocatalytic experiments were carried out in a 100 mL quartz cell. As the light source, the 150 W high-pressure Xenon lamp with 410 nm cutoff filter was used. A constant cell temperature of 26 °C was maintained by air ventilation and monitored with a thermometer. For the photocatalytic reaction, 25 mg of the photocatalyst was added to 50 mL of an aqueous solution of MO (0.015 mmol). Before turning on the light, the cell was placed in the dark for 30 min to achieve adsorption equilibrium. Before the start of the experiment, the suspension was subjected to ultrasonic treatment to degas the photocatalyst. The whole process was carried out with magnetic stirring. Sampling (5 mL) was carried out every 10 min, large particles of the nanopowders were subjected to magnetic separation using a powerful neodymium magnet to prevent loss of the photocatalyst, and then centrifuged at 14,000 rpm for 3 min on an MR23i (Thermo Fisher Scientific, JOUAN, France) high-speed refrigeration centrifuge for deposition of ultrafine nanoparticles. The concentration of MO was measured using a Beckman Coulter DU730 series UV/Vis spectrophotometer at a constant temperature of 26 °C. After measurement, the solution was poured back into the cell and the process continued. For comparison, we also tested the MO solution under similar conditions, without a photocatalyst. The concentration of MO was determined by the Bouguer–Lambert–Beer law.
4. Conclusions
BFO nanoparticles doped with samarium in various amounts from 5 to 20% were synthesized using the solution combustion method. It was shown that an increase in the content of samarium leads to structural distortions. The sizes of crystallites and the unit cell volume decrease. As doping increases, the band gap also decreases. An analysis of the crystal structure showed that doping leads to phase transitions with the formation of morphotropic phase boundaries. Thus, at 10% doping, the polar rhombohedral R3c and antipolar orthorhombic Pbam phases coexist. At 15% doping, the system already consists of antipolar orthorhombic Pbam and nonpolar orthorhombic Pnma phases. The PC measurements showed the absence of a strict correlation between the PC activity and the crystallite size and band gap. A significant increase in the PC activity of samples with 10 and 15% doping enables us to conclude that in controlling the PC properties in doped BFO, the processes of interfacial polarization at the boundaries of the morphotropic phase transition are of decisive importance, since the internal electric field formed at these boundaries contributes to the efficient separation of photogenerated charge carriers.
Author Contributions
F.F.O.: project coordination and administration, formal analysis, validation. N.M.-R.A. and S.M.R.: conceptualization, methodology, and data collecting/analysis. D.S.S., R.K.M., E.H.I. and R.D.G.: original draft preparation, data analysis, interpretation, and software. A.S.A.: methodology, investigation and validation. Ș.Ț.: writing and editing, and funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
The reported study was funded by the Russian Science Foundation, Project No. 22-73-10091 in part of nanoparticle fabrication and characterization (SEM, EDS, Optical), by RFBR, project number 20-08-00242A in part of nanoparticle characterization (XRD, and dielectrical properties, EMR), and partial support of the state assignment FZNZ-2020-0002 in part of photocatalytic measurements. The research described in the paper was supported by the Internal Grant Agency of Brno University of Technology, grant No. FEKT-S-20-6352. Part of the work was carried out with the support of CEITEC Nano Research Infrastructure supported by MEYS CR (LM2018110).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Not available.
References
- Kumar, A.; Kumar, A.; Krishnan, V. Perovskite Oxide Based Materials for Energy and Environment-Oriented Photocatalysis. ACS Catal. 2020, 10, 10253–10315. [Google Scholar] [CrossRef]
- Wang, W.; Tadé, M.O.; Shao, Z. Research Progress of Perovskite Materials in Photocatalysis- and Photovoltaics-Related Energy Conversion and Environmental Treatment. Chem. Soc. Rev. 2015, 44, 5371–5408. [Google Scholar] [CrossRef] [PubMed]
- Irshad, M.; tul Ain, Q.; Zaman, M.; Aslam, M.Z.; Kousar, N.; Asim, M.; Rafique, M.; Siraj, K.; Tabish, A.N.; Usman, M.; et al. Photocatalysis and Perovskite Oxide-Based Materials: A Remedy for a Clean and Sustainable Future. RSC Adv. 2022, 12, 7009–7039. [Google Scholar] [CrossRef] [PubMed]
- Dandia, A.; Saini, P.; Sharma, R.; Parewa, V. Visible Light Driven Perovskite-Based Photocatalysts: A New Candidate for Green Organic Synthesis by Photochemical Protocol. Curr. Res. Green Sustain. Chem. 2020, 3, 100031. [Google Scholar] [CrossRef]
- Casbeer, E.; Sharma, V.K.; Li, X.Z. Synthesis and Photocatalytic Activity of Ferrites under Visible Light: A Review. Sep. Purif. Technol. 2012, 87, 1–14. [Google Scholar] [CrossRef]
- Tikhanova, S.M.; Lebedev, L.A.; Martinson, K.D.; Chebanenko, M.I.; Buryanenko, I.V.; Semenov, V.G.; Nevedomskiy, V.N.; Popkov, V.I. The Synthesis of Novel Heterojunction H-YbFeO3/o-YbFeO3 Photocatalyst with Enhanced Fenton-like Activity under Visible-Light. New J. Chem. 2021, 45, 1541–1550. [Google Scholar] [CrossRef]
- Zhang, K.; Li, D.; Tian, Q.; Cao, H.; Orudzhev, F.; Zvereva, I.A.; Xu, J.; Wang, C. Recyclable 0D/2D ZnFe2O4/Bi5FeTi3O15 S-Scheme Heterojunction with Bismuth Decoration for Enhanced Visible-Light-Driven Tetracycline Photodegradation. Ceram. Int. 2021, 47, 17109–17119. [Google Scholar] [CrossRef]
- Alikhanov, N.M.R.; Rabadanov, M.K.; Orudzhev, F.F.; Gadzhimagomedov, S.K.; Emirov, R.M.; Sadykov, S.A.; Kallaev, S.N.; Ramazanov, S.M.; Abdulvakhidov, K.G.; Sobola, D. Size-Dependent Structural Parameters, Optical, and Magnetic Properties of Facile Synthesized Pure-Phase BiFeO3. J. Mater. Sci. Mater. Electron. 2021, 32, 13323–13335. [Google Scholar] [CrossRef]
- Orudzhev, F.F.; Aliev, Z.M.; Gasanova, F.G.; Isaev, A.B.; Shabanov, N.S. Photoelectrocatalytic Oxidation of Phenol on TiO2 Nanotubes under Oxygen Pressure. Russ. J. Electrochem. 2015, 51, 1108–1114. [Google Scholar] [CrossRef]
- Magdalane, C.M.; Priyadharsini, G.M.A.; Kaviyarasu, K.; Jothi, A.I.; Simiyon, G.G. Synthesis and Characterization of TiO2 Doped Cobalt Ferrite Nanoparticles via Microwave Method: Investigation of Photocatalytic Performance of Congo Red Degradation Dye. Surf. Interfaces 2021, 25, 101296. [Google Scholar] [CrossRef]
- Zargazi, M.; Entezari, M.H. A Novel Synthesis of Forest like BiFeO3 Thin Film: Photo-Electrochemical Studies and Its Application as a Photocatalyst for Phenol Degradation. Appl. Surf. Sci. 2019, 483, 793–802. [Google Scholar] [CrossRef]
- Zargazi, M.; Entezari, M.H. BFO Thin Film on the Stainless Steel Mesh by Anodic EPD: A Visible Light Photocatalyst for Degradation of Rhodamin B. J. Photochem. Photobiol. A Chem. 2018, 365, 185–198. [Google Scholar] [CrossRef]
- Lomanova, N.A.; Tomkovich, M.V.; Sokolov, V.V.; Gusarov, V.V. Special Features of Formation of Nanocrystalline BiFeO3 via the Glycine-Nitrate Combustion Method. Russ. J. Gen. Chem. 2016, 86, 2256–2262. [Google Scholar] [CrossRef]
- Lomanova, N.A.; Tomkovich, M.V.; Sokolov, V.V.; Ugolkov, V.L.; Panchuk, V.V.; Semenov, V.G.; Pleshakov, I.V.; Volkov, M.P.; Gusarov, V.V. Thermal and Magnetic Behavior of BiFeO3 Nanoparticles Prepared by Glycine-Nitrate Combustion. J. Nanoparticle Res. 2018, 20, 17. [Google Scholar] [CrossRef]
- Zaboeva, E.A.; Izotova, S.G.; Popkov, V.I. Glycine-Nitrate Combustion Synthesis of CeFeO3-Based Nanocrystalline Powders. Russ. J. Appl. Chem. 2016, 89, 1228–1236. [Google Scholar] [CrossRef]
- Martinson, K.D.; Kondrashkova, I.S.; Popkov, V.I. Synthesis of EuFeO3 Nanocrystals by Glycine-Nitrate Combustion Method. Russ. J. Appl. Chem. 2017, 90, 1214–1218. [Google Scholar] [CrossRef]
- Orudzhev, F.; Alikhanov, N.; Rabadanov, M.; Ramazanov, S.; Isaev, A.; Gadzhimagomedov, S.; Aliyev, A.; Abdullaev, V. Synthesis and study of the properties of magnetically separable nanophotocatalyst BiFeO3. Chem. Probl. 2018, 4, 484–495. [Google Scholar] [CrossRef]
- Larosa, C.; Stura, E.; Eggenhöffner, R.; Nicolini, C. Optimization of Optical Properties of Polycarbonate Film with Thiol Gold-Nanoparticles. Materials 2009, 2, 1193–1204. [Google Scholar] [CrossRef]
- Guo, R.; Fang, L.; Dong, W.; Zheng, F.; Shen, M. Enhanced Photocatalytic Activity and Ferromagnetism in Gd Doped BiFeO3 Nanoparticles. J. Phys. Chem. C 2010, 114, 21390–21396. [Google Scholar] [CrossRef]
- Wu, C.; Wei, J.; Kong, F. Effect of Rare Earth Dopants on the Morphologies and Photocatalytic Activities of BiFeO3 Microcrystallites. J. Mater. Sci. Mater. Electron. 2012, 24, 1530–1535. [Google Scholar] [CrossRef]
- Kaur, M.; Yadav, K.L.; Uniyal, P. Investigations on Multiferroic, Optical and Photocatalytic Properties of Lanthanum Doped Bismuth Ferrite Nanoparticles. Adv. Mater. Lett. 2015, 6, 895–901. [Google Scholar] [CrossRef]
- Wei, J.; Liu, Y.; Bai, X.; Li, C.; Liu, Y.; Xu, Z.; Gemeiner, P.; Haumont, R.; Infante, I.C.; Dkhil, B. Crystal Structure, Leakage Conduction Mechanism Evolution and Enhanced Multiferroic Properties in Y-Doped BiFeO3 Ceramics. Ceram. Int. 2016, 42, 13395–13403. [Google Scholar] [CrossRef]
- Xu, B.; Wang, D.; Íñiguez, J.; Bellaiche, L. Finite-Temperature Properties of Rare-Earth-Substituted BiFeO3 Multiferroic Solid Solutions. Adv. Funct. Mater. 2015, 25, 552–558. [Google Scholar] [CrossRef]
- Singh, H.; Yadav, K.L. Structural, Dielectric, Vibrational and Magnetic Properties of Sm Doped BiFeO3 Multiferroic Ceramics Prepared by a Rapid Liquid Phase Sintering Method. Ceram. Int. 2015, 41, 9285–9295. [Google Scholar] [CrossRef]
- Xu, X.; Guoqiang, T.; Huijun, R.; Ao, X. Structural, Electric and Multiferroic Properties of Sm-Doped BiFeO3 Thin Films Prepared by the Sol–Gelprocess. Ceram. Int. 2013, 39, 6223–6228. [Google Scholar] [CrossRef]
- Fujino, S.; Murakami, M.; Anbusathaiah, V.; Lim, S.H.; Nagarajan, V.; Fennie, C.J.; Wuttig, M.; Salamanca-Riba, L.; Takeuchi, I. Combinatorial Discovery of a Lead-Free Morphotropic Phase Boundary in a Thin-Film Piezoelectric Perovskite. Appl. Phys. Lett. 2008, 92, 202904. [Google Scholar] [CrossRef]
- Catalan, G.; Scott, J.F. Physics and Applications of Bismuth Ferrite. Adv. Mater. 2009, 21, 2463–2485. [Google Scholar] [CrossRef]
- Eerenstein, W.; Mathur, N.D.; Scott, J.F. Multiferroic and Magnetoelectric Materials. Nature 2006, 442, 759–765. [Google Scholar] [CrossRef]
- Ramesh, R.; Spaldin, N.A. Multiferroics: Progress and Prospects in Thin Films. Nat. Mater. 2007, 6, 21–29. [Google Scholar] [CrossRef]
- Wu, J.; Fan, Z.; Xiao, D.; Zhu, J.; Wang, J. Multiferroic Bismuth Ferrite-Based Materials for Multifunctional Applications: Ceramic Bulks, Thin Films and Nanostructures. Prog. Mater. Sci. 2016, 84, 335–402. [Google Scholar] [CrossRef]
- Pattanayak, S.; Choudhary, R.N.P.; Das, P.R. Effect of Sm-Substitution on Structural, Electrical and Magnetic Properties of BiFeO3. Electron. Mater. Lett. 2014, 10, 165–172. [Google Scholar] [CrossRef]
- Kakekhani, A.; Ismail-Beigi, S.; Altman, E.I. Ferroelectrics: A Pathway to Switchable Surface Chemistry and Catalysis. Surf. Sci. 2016, 650, 302–316. [Google Scholar] [CrossRef]
- Georgescu, A.B.; Ismail-Beigi, S. Surface Piezoelectricity of (0001) Sapphire. Phys. Rev. Appl. 2019, 11, 64065. [Google Scholar] [CrossRef]
- Safi, R.; Shokrollahi, H. Physics, Chemistry and Synthesis Methods of Nanostructured Bismuth Ferrite (BiFeO3) as a Ferroelectro-Magnetic Material. Prog. Solid State Chem. 2012, 40, 6–15. [Google Scholar] [CrossRef]
- Morozov, M.I.; Lomanova, N.A.; Gusarov, V.V. Specific Features of BiFeO3 Formation in a Mixture of Bismuth(III) and Iron(III) Oxides. Russ. J. Gen. Chem. 2003, 73, 1676–1680. [Google Scholar] [CrossRef]
- Matjaz, V.; Anna-Karin, A.; Neil, A. Peculiarities of a Solid-State Synthesis of Multiferroic Polycrystalline BiFeO3. Chem. Mater. 2007, 19, 5431–5436. [Google Scholar] [CrossRef]
- Khomchenko, V.A.; Paixão, J.A.; Shvartsman, V.V.; Borisov, P.; Kleemann, W.; Karpinsky, D.V.; Kholkin, A.L. Effect of Sm Substitution on Ferroelectric and Magnetic Properties of BiFeO3. Scr. Mater. 2010, 62, 238–241. [Google Scholar] [CrossRef]
- Stojadinović, B.; Dohčević-Mitrović, Z.; Paunović, N.; Ilić, N.; Tasić, N.; Petronijević, I.; Popović, D.; Stojanović, B. Comparative Study of Structural and Electrical Properties of Pr and Ce Doped BiFeO3 Ceramics Synthesized by Auto-Combustion Method. J. Alloys Compd. 2016, 657, 866–872. [Google Scholar] [CrossRef]
- Hussain, A.; Xu, X.; Yuan, G.; Wang, Y.; Yang, Y.; Yin, J.; Liu, J.; Liu, Z. The Development of BiFeO3-Based Ceramics. Chin. Sci. Bull. 2014, 59, 5161–5169. [Google Scholar] [CrossRef]
- Deka, B.; Ravi, S.; Pamu, D. Evolution of Structural Transition, Grain Growth Inhibition and Collinear Antiferromagnetism in (Bi1−õSmx)FeO3 (x = 0 to 0.3) and Their Effects on Dielectric and Magnetic Properties. Ceram. Int. 2017, 43, 16580–16592. [Google Scholar] [CrossRef]
- Godara, P.; Agarwal, A.; Ahlawat, N.; Sanghi, S. Crystal Structure Refinement, Dielectric and Magnetic Properties of Sm Modified BiFeO3 Multiferroic. J. Mol. Struct. 2015, 1097, 207–213. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides. Acta Crystallogr. Sect. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Bielecki, J.; Svedlindh, P.; Tibebu, D.T.; Cai, S.; Eriksson, S.G.; Börjesson, L.; Knee, C.S. Structural and Magnetic Properties of Isovalently Substituted Multiferroic BiFeO3: Insights from Raman Spectroscopy. Phys. Rev. B Condens. Matter Mater. Phys. 2012, 86, 184422. [Google Scholar] [CrossRef]
- Fukumura, H.; Harima, H.; Kisoda, K.; Tamada, M.; Noguchi, Y.; Miyayama, M. Raman Scattering Study of Multiferroic BiFeO3 Single Crystal. J. Magn. Magn. Mater. 2007, 310, 367–369. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Shyam, P.; Apte, A.; Kumar, J.; Bhattacharyya, A.; Awasthi, A.M.; Kulkarni, S. Dynamics of Electron Density, Spin-Phonon Coupling, and Dielectric Properties of SmFeO3 Nanoparticles at the Spin-Reorientation Temperature: Role of Exchange Striction. Phys. Rev. B 2016, 93, 174117. [Google Scholar] [CrossRef]
- Wang, J.; Wei, Y.; Zhang, J.; Ji, L.; Huang, Y.; Chen, Z. Synthesis of Pure-Phase BiFeO3 Nanopowder by Nitric Acid-Assisted Gel. Mater. Lett. 2014, 124, 242–244. [Google Scholar] [CrossRef]
- Xu, X.S.; Brinzari, T.V.; Lee, S.; Chu, Y.H.; Martin, L.W.; Kumar, A.; McGill, S.; Rai, R.C.; Ramesh, R.; Gopalan, V.; et al. Optical Properties and Magnetochromism in Multiferroic BiFeO3. Phys. Rev. B Condens. Matter Mater. Phys. 2009, 79, 134425. [Google Scholar] [CrossRef]
- Ramirez, M.O.; Kumar, A.; Denev, S.A.; Podraza, N.J.; Xu, X.S.; Rai, R.C.; Chu, Y.H.; Seidel, J.; Martin, L.W.; Yang, S.Y.; et al. Magnon Sidebands and Spin-Charge Coupling in Bismuth Ferrite Probed by Nonlinear Optical Spectroscopy. Phys. Rev. B Condens. Matter Mater. Phys. 2009, 79, 224106. [Google Scholar] [CrossRef]
- Pisarev, R.V.; Moskvin, A.S.; Kalashnikova, A.M.; Rasing, T. Charge Transfer Transitions in Multiferroic BiFeO3 and Related Ferrite Insulators. Phys. Rev. B Condens. Matter Mater. Phys. 2009, 79, 235128. [Google Scholar] [CrossRef]
- Dionne, G.F. Magnetic Oxides; Springer: Berlin/Heidelberg, Germany, 2009; pp. 1–466. [Google Scholar] [CrossRef]
- Kumar, A.; Rai, R.C.; Podraza, N.J.; Denev, S.; Ramirez, M.; Chu, Y.H.; Martin, L.W.; Ihlefeld, J.; Heeg, T.; Schubert, J.; et al. Linear and Nonlinear Optical Properties of BiFeO3. Appl. Phys. Lett. 2008, 92, 121915. [Google Scholar] [CrossRef]
- Jubu, P.R.; Yam, F.K.; Igba, V.M.; Beh, K.P. Tauc-Plot Scale and Extrapolation Effect on Bandgap Estimation from UV–Vis–NIR Data—A Case Study of β-Ga2O3. J. Solid State Chem. 2020, 290, 121576. [Google Scholar] [CrossRef]
- Arora, M.; Kumar, M. Structural, Magnetic and Optical Properties of Ce Substituted BiFeO3 Nanoparticles. Ceram. Int. 2015, 4, 5705–5712. [Google Scholar] [CrossRef]
- Eichel, R.A. Characterization of Defect Structure in Acceptor-Modified Piezoelectric Ceramics by Multifrequency and Multipulse Electron Paramagnetic Resonance Spectroscopy. J. Am. Ceram. Soc. 2008, 91, 691–701. [Google Scholar] [CrossRef]
- Paul Blessington Selvadurai, A.; Pazhanivelu, V.; Jagadeeshwaran, C.; Murugaraj, R.; Mohammed Gazzali, P.M.; Chandrasekaran, G. An Analysis on Structural and Magnetic Properties of La1−xRExFeO3 (x = 0.0 and 0.5, RE = Nd, Sm and Gd) Nanoparticles. Appl. Phys. A 2016, 123, 13. [Google Scholar] [CrossRef]
- Krishna Reddy, J.; Srinivas, B.; Durga Kumari, V.; Subrahmanyam, M. Sm3+-Doped Bi2O3 Photocatalyst Prepared by Hydrothermal Synthesis. ChemCatChem 2009, 1, 492–496. [Google Scholar] [CrossRef]
- Choi, W.; Termin, A.; Hoffmann, M.R. The Role of Metal Ion Dopants in Quantum-Sized TiO2: Correlation between Photoreactivity and Charge Carrier Recombination Dynamics. J. Phys. Chem. 1994, 98, 13669–13679. [Google Scholar] [CrossRef]
- Damjanovic, D. A Morphotropic Phase Boundary System Based on Polarization Rotation and Polarization Extension. Appl. Phys. Lett. 2010, 97, 62906. [Google Scholar] [CrossRef]
- Hu, Z.; Chen, D.; Wang, S.; Zhang, N.; Qin, L.; Huang, Y. Facile Synthesis of Sm-Doped BiFeO3 Nanoparticles for Enhanced Visible Light Photocatalytic Performance. Mater. Sci. Eng. B 2017, 220, 1–12. [Google Scholar] [CrossRef]
- Zilabi, S.; Habibzadeh, S.; Gheytanzadeh, M.; Rahmani, M. Direct Sunlight Catalytic Decomposition of Organic Pollutants via Sm- and Ce-Doped BiFeO3 Nanopowder Synthesized by a Rapid Combustion Technique. Catal. Lett. 2021, 151, 3462–3476. [Google Scholar] [CrossRef]
- Gu, Y.; Zhou, Y.; Zhang, W.; Guo, C.; Zhang, X.; Zhao, J.; Zhang, Y.; Zheng, H. Optical and Magnetic Properties of Sm-Doped BiFeO3 Nanoparticles around the Morphotropic Phase Boundary Region. AIP Adv. 2021, 11, 45223. [Google Scholar] [CrossRef]
- Wang, S.; Chen, D.; Niu, F.; Zhang, N.; Qin, L.; Huang, Y. Pd Cocatalyst on Sm-Doped BiFeO3 Nanoparticles: Synergetic Effect of a Pd Cocatalyst and Samarium Doping on Photocatalysis. RSC Adv. 2016, 6, 34574–34587. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, D.; Niu, F.; Wang, S.; Qin, L.; Huang, Y. Enhanced Visible Light Photocatalytic Activity of Gd-Doped BiFeO3 Nanoparticles and Mechanism Insight. Sci. Rep. 2016, 6, 26467. [Google Scholar] [CrossRef] [PubMed]
- Patil, K.C.; Hegde, M.S.; Rattan, T.; Aruna, S.T. Chemistry of Nanocrystalline Oxide Materials; World Scientific: Singapore, 2008. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).