Reuse of Food Waste: The Chemical Composition and Health Properties of Pomelo (Citrus maxima) Cultivar Essential Oils †
Abstract
:1. Introduction
2. Results and Discussions
2.1. Composition of EOs and Their Blends
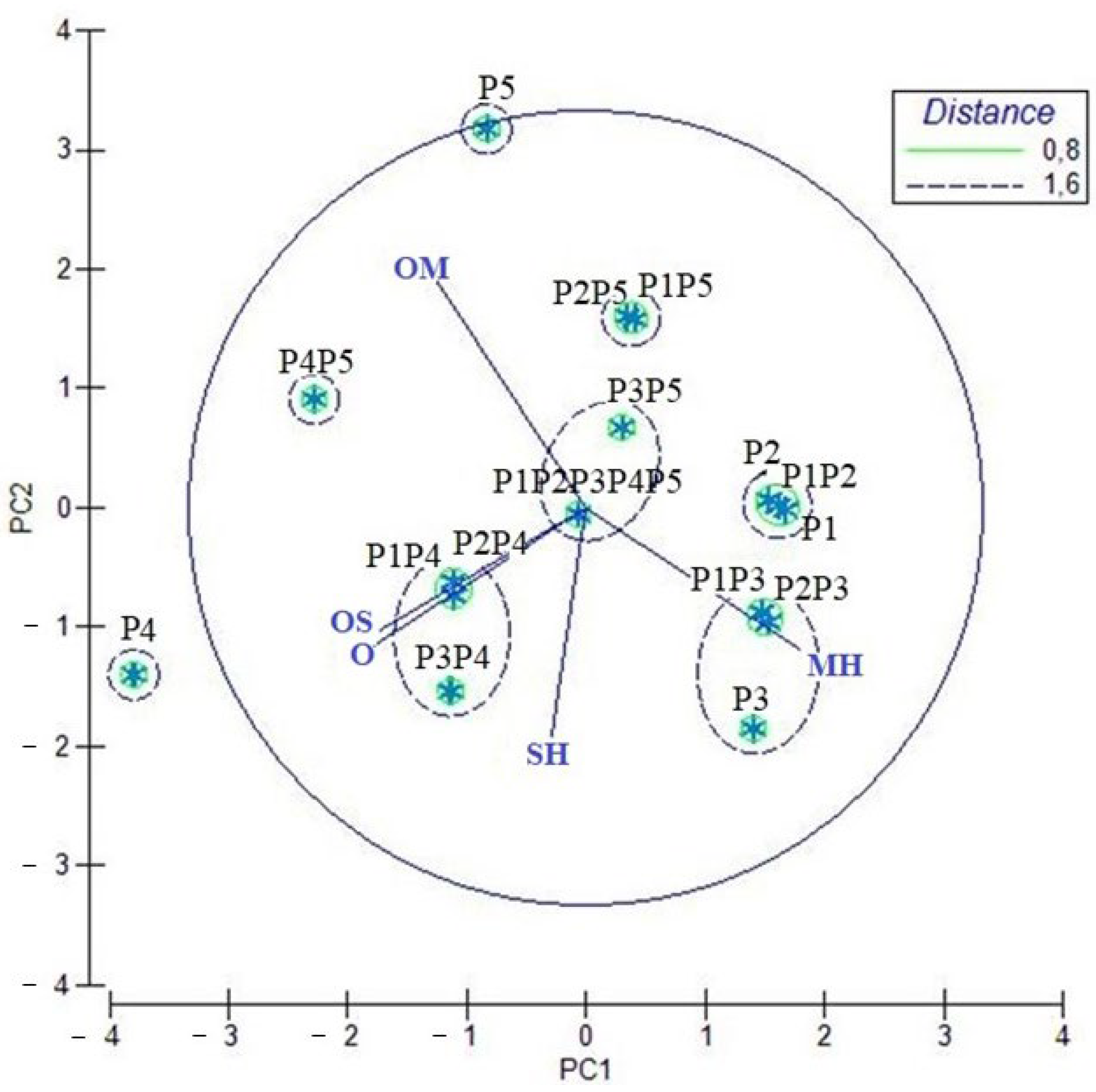
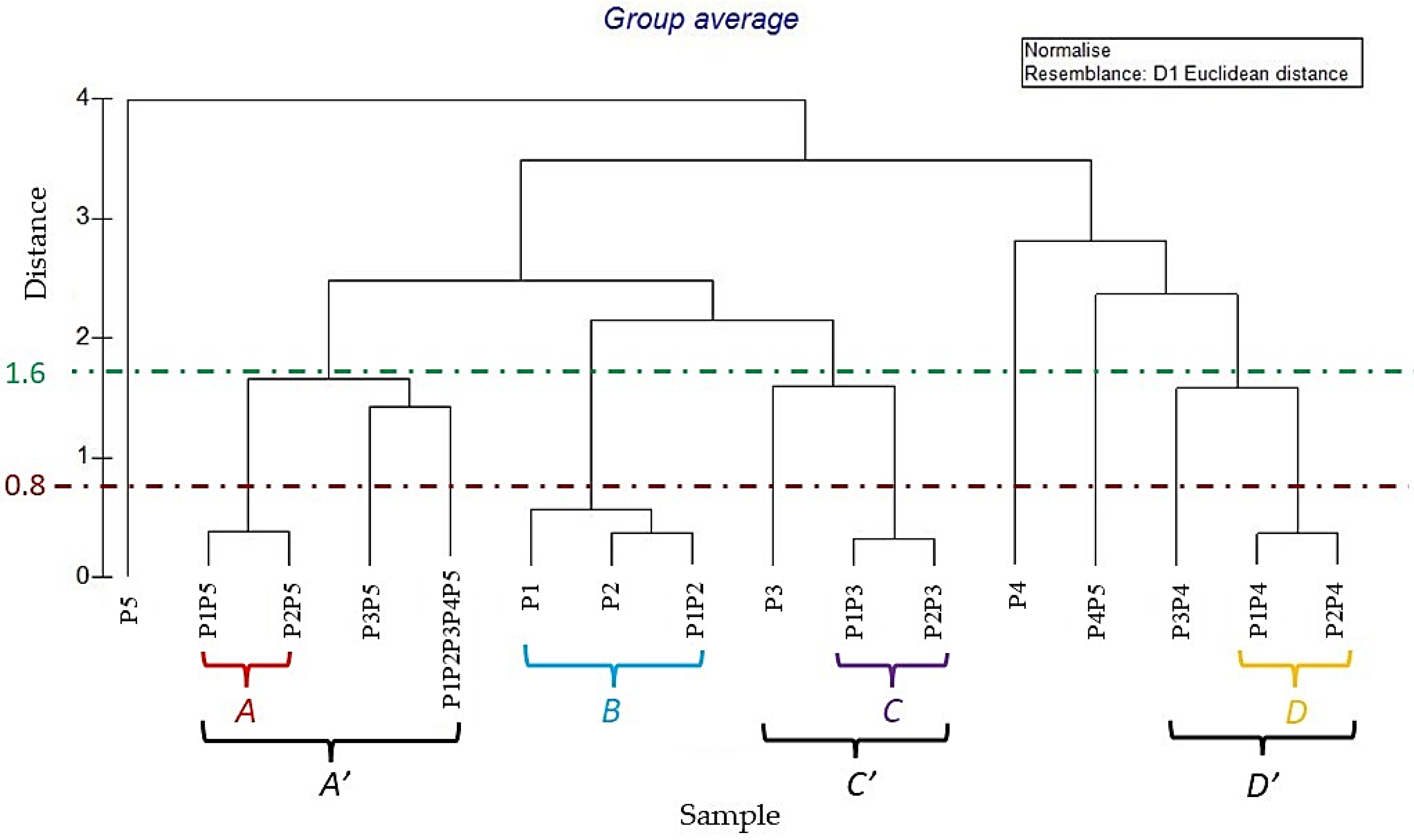
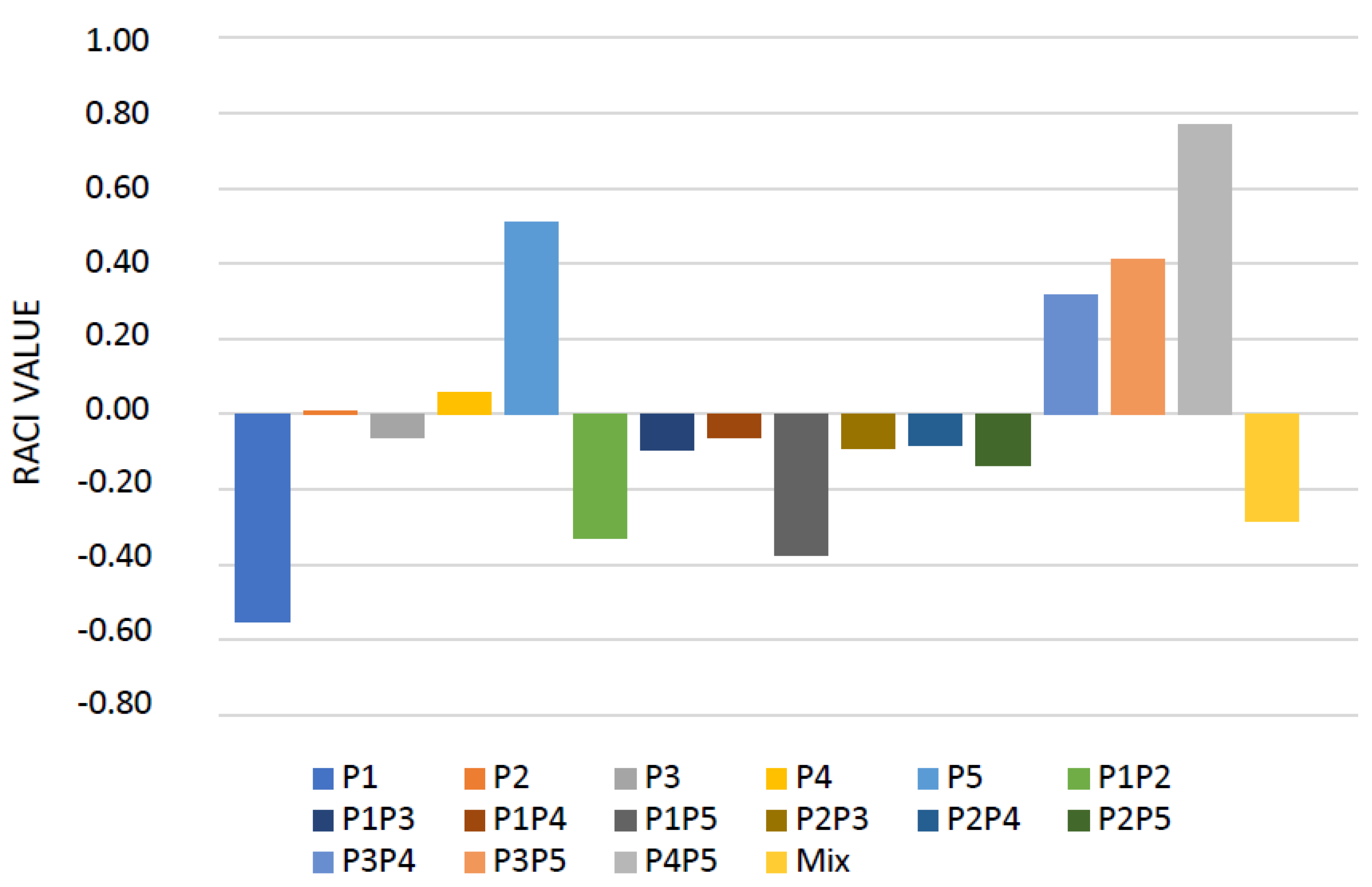
2.2. PCA and HCA Analyses of the EOs and Their Mixes
2.3. Radical Scavenging and Antioxidant Activities of Pomelo EOs and Their Blends
2.4. Inhibition of Enzymes Involved in Metabolic Syndrome and Obesity by Pomelo EOs and Their Blends
3. Materials and Methods
3.1. Plant Material
3.2. Essential Oil Extractions and Their Blend Preparation
3.3. Pomelo EOs' Volatile Profiles
3.4. Evaluation of Radical Scavenging Activity by ABTS and DPPH Assays
3.5. Ferric Reducing Ability Power (FRAP Assay)
3.6. β-Carotene Bleaching Test
3.7. Lipase-Inhibitory Assay
3.8. Carbohydrate-Hydrolysing Enzyme-Inhibition Assay
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- FAO. Citrus Fruit Fresh and Processed. Statistical Bulletin; Trade and Markets Division: Rome, Italy, 2016. [Google Scholar]
- USDA. Citrus Annual Expected Rebound in EU-28 Citrus Production May Reduce Imports for MY 2018/19; USDA Foreign Agricultural Service: Washington, DC, USA.
- USDA. Citrus: World Markets and Trade [WWW Document]. 2020. Available online: https://www.fas.usda.gov/data/citrus-world-markets-and-trade (accessed on 1 February 2022).
- Mahato, N.; Sinha, M.; Sharma, K.; Koteswararao, R.; Cho, M.H. Modern extraction and purification techniques for obtaining high purity food-grade bioactive compounds and value-added co-products from citrus wastes. Foods 2019, 8, 523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geraci, A.; Di Stefano, V.; Di Martino, E.; Schillaci, D.; Schicchi, R. Essential oil components of orange peels and antimicrobial activity. Nat. Prod. Res. 2017, 31, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Martín, M.A.; Fernandez, R.; Gutiérrez, M.C.; Siles, J.A. Thermophilic anaerobic digestion of pre-treated orange peel: Modelling of methane production. Process Saf. Environ. Protect. 2018, 117, 245–253. [Google Scholar] [CrossRef]
- Siles López, Á.J.; Li, Q.; Thompson, I.P. Biorefinery of waste orange peel. Crit. Rev. Biotechnol. 2010, 30, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Zema, D.A.; Calabrò, P.S.; Folino, A.; Tamburino, V.; Zappia, G.; Zimbone, S.M. Valorisation of citrus processing waste: A review. Waste Manag. 2018, 80, 252–273. [Google Scholar] [CrossRef] [PubMed]
- Teigiserova, D.A.; Hamelin, L.; Thomsen, M. Review of high-value food waste and food residues biorefineries with focus on unavoidable wastes from processing. Resour. Conserv. Recycl. 2019, 149, 413–426. [Google Scholar] [CrossRef]
- Oberoi, H.S.; Vadlani, P.V.; Nanjundaswamy, A.; Bansal, S.; Singh, S.; Kaur, S.; Babbar, N. Enhanced ethanol production from Kinnow Mandarin (Citrus reticulata) waste via a statistically optimized simultaneous saccharification and fermentation process. Bioresour. Technol. 2011, 102, 1593–1601. [Google Scholar] [CrossRef]
- Del Bravo, F.; Finizia, A.; Moriello, M.S.L.; Ronga, M. Report Ministero delle Politiche Agricole Alimentari e Forestali. La Competitività Della Filiera Agrumicola in Italia Aprile 2020. Available online: https://www.ismea.it/istituto-di-servizi-per-il-mercato-agricolo-alimentare (accessed on 12 April 2022).
- Baiamonte, G.; Domina, G.; Raimondo, F.M.; Bazan, G. Agricultural landscapes and biodiversity conservation: A case study in Sicily (Italy). Biodivers. Conserv. 2015, 24, 3201–3216. [Google Scholar] [CrossRef]
- Badalamenti, N.; Bruno, M.; Schicchi, R.; Geraci, A.; Leporini, M.; Gervasi, L.; Tundis, R.; Loizzo, M.R. Chemical Compositions and Antioxidant Activities of Essential Oils, and Their Combinations, Obtained from Flavedo By-Product of Seven Cultivars of Sicilian Citrus aurantium L. Molecules 2022, 27, 1580. [Google Scholar] [CrossRef]
- Khan, N.H.; Qian, C.J.; Perveen, N. Phytochemical screening, antimicrobial and antioxidant activity determination of citrus maxima peel. Pharm. Pharmacol. Int. J. 2018, 6, 279–285. [Google Scholar] [CrossRef]
- Russo, C.; Maugeri, A.; Lombardo, G.E.; Musumeci, L.; Barreca, D.; Rapisarda, A.; Cirmi, S.; Navarra, M. The second life of Citrus fruit waste: A valuable source of bioactive compounds. Molecules 2021, 26, 5991. [Google Scholar] [CrossRef] [PubMed]
- Nieto, G.; Fernández-López, J.; Pérez-Álvarez, J.A.; Peñalver, R.; Ros-Berruezo, G.; Viuda-Martos, M. Valorization of Citrus co-products: Recovery of bioactive compounds and application in meat and meat products. Plants 2021, 10, 1069. [Google Scholar] [CrossRef] [PubMed]
- Mamma, D.; Christakopoulos, P. Biotransformation of Citrus by-products into value added products. Waste Biomass Valoriz. 2014, 5, 529–549. [Google Scholar] [CrossRef]
- Sharma, K.; Mahato, N.; Cho, M.H.; Lee, Y.R. Converting Citrus wastes into value-added products: Economic and environmently friendly approaches. Nutrition 2017, 34, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Suntar, I.; Khan, H.; Patel, S.; Celano, R.; Rastrelli, L. An overview on Citrus aurantium L. Its functions as food ingredient and therapeutic agent. Oxidative Med. Cell. Longev. 2018, 2018, 7864269. [Google Scholar] [CrossRef] [Green Version]
- Leporini, M.; Tundis, R.; Sicari, V.; Loizzo, M.R. Citrus species: Modern functional food and nutraceutical-based product ingredient. It. J. Food Sci. 2021, 33, 63–107. [Google Scholar] [CrossRef]
- Scora, R.W. Biochemistry, taxonomy and evolution of modern cultivated Citrus. In Proceedings of the Sixth International Citrus Congress, Middle-East, Tel Aviv, Israel, 6–11 March 1988; pp. 277–289. [Google Scholar]
- Barrett, H.C.; Rhodes, R.A. A numerical taxonomic study of affinity relationships in cultivated Citrus and its close relatives. Syst. Bot. 1976, 1, 105–136. [Google Scholar] [CrossRef]
- Hynniewta, M.; Malik, S.M.; Rao, S.R. Karyological studies in ten species of Citrus (Linnaeus, 1753) (Rutaceae) of North-East India. Comp. Cytogen. 2011, 5, 277–287. [Google Scholar] [CrossRef] [Green Version]
- Federici, C.T.; Fang, D.Q.; Scora, R.W.; Roose, M.L. Phylogenetic relationships within the genus Citrus (Rutaceae) and related genera as revealed by RFLP and RAPD analysis. Theor. Appl. Genet. 1998, 96, 812–822. [Google Scholar] [CrossRef]
- Nicolosi, E.; Deng, Z.N.; Gentile, A.; Sl, M.; Continella, G.; Tribulato, E. Citrus phylogeny and genetic origin of important species as investigated by molecular markers. Theor. Appl. Genet. 2000, 100, 1155–1166. [Google Scholar] [CrossRef]
- Barkley, N.A.; Roose, M.L.; Krueger, R.R.; Federici, C.T. Assessing genetic diversity and population structure in a Citrus germplasm collection utilizing simple sequence repeat markers (SSRs). Theor. Appl. Genet. 2006, 112, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Lor, A.; Curk, F.; Snoussi-Trifa, H.; Morillon, G.R.; Ancillo, G. A nuclear phylogenetic analysis: SNPs, indels and SSRs deliver new insights into the relationships in the ‘true Citrus fruit trees’ group (Citrinae, Rutaceae) and the origin of cultivated species. Ann. Bot. 2013, 111, 1–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, G.A.; Prochnik, S.; Jenkins, J.; Salse, J.; Hellsten, U.; Murat, F.; Perrier, X.; Ruiz, M.; Scalabrin, S.; Terol, J.; et al. Sequencing of diverse mandarin, pummelo and orange genomes reveals complex history of admixture during citrus domestication. Nat. Biotech. 2014, 32, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.A.; Terol, J.; Ibanez, V.; Lopez-Garcia, A.; Perez-Roman, E.; Carles, B.; Domingo, C.; Tadeo, F.R.; Carbonell-Caballero, J.; Alonso, R.; et al. Genomics of the origin and evolution of Citrus. Nature 2018, 554, 311–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Curk, F.; Ancillo, G.; Ollitrault, F.; Perrier, X.; Jacquemoud-Collet, J.P.; Garcia-Lor, A.; Navarro, L.; Ollitrault, P.; Fang, D.D. Nuclear species-diagnostic SNP markers mined from 454 amplicon sequencing reveal admixture genomic structure of modern Citrus varieties. PLoS ONE 2015, 10, e0125628. [Google Scholar] [CrossRef] [Green Version]
- Ollitrault, P.; Curk, F.; Krueger, R. Citrus taxonomy. In The Genus Citrus; Talon, M., Caruso, M., Gmitter, J.F.G., Eds.; Elsevier WP: Duxford, UK, 2020; pp. 57–81. ISBN 978-0-12-812217-4. [Google Scholar]
- Sun, H.; Ni, H.; Yang, Y.; Chen, F.; Cai, H.; Xiao, A. Sensory evaluation and gas chromatography-mass spectrometry (GC-MS) analysis of the volatile extracts of pummelo (Citrus maxima) peel. Flavour Frag. J. 2014, 29, 305–312. [Google Scholar] [CrossRef]
- Ou, M.C.; Liu, Y.H.; Sun, Y.W.; Chan, C.F. The composition, antioxidant and antibacterial activities of cold-pressed and distilled essential oils of Citrus paradisi and Citrus grandis (L.) Osbeck. Evid. Based Complement. Alternat. Med. 2015, 2015, 804091. [Google Scholar] [CrossRef] [Green Version]
- Njoroge, S.M.; Koaze, H.; Karanja, P.N.; Sawamura, M. Volatile constituents of redblush grapefruit (Citrus paradisi) and pummelo (Citrus grandis) peel essential oils from Kenya. J. Agric. Food Chem. 2005, 53, 9790–9794. [Google Scholar] [CrossRef]
- Hosseini, B.; Saedisomeolia, A.; Allman-Farinelli, M. Association Between Antioxidant Intake/Status and Obesity: A Systematic Review of Observational Studies. Biol. Trace Elem. Res. 2017, 175, 287–297. [Google Scholar] [CrossRef]
- Abdali, D.; Samson, S.E.; Grover, A.K. How effective are antioxidant supplements in obesity and diabetes? Med. Princ. Pract. 2015, 24, 201–215. [Google Scholar] [CrossRef]
- Tao, N.G.; Liu, Y.J. Chemical composition and antimicrobial activity of the essential oil from the peel of shatian pummelo (Citrus grandis Osbeck). Int. J. Food. Prop. 2012, 15, 709–716. [Google Scholar] [CrossRef]
- Tuan, N.T.; Dang, L.N.; Cam Huong, B.T.; Thai Danh, L. One step extraction of essential oils and pectin from pomelo (Citrus grandis) peels. Chem. Eng. Process Process Intensif. 2019, 142, 107550. [Google Scholar] [CrossRef]
- Lan-Phi, N.T.; Vy, T.T. Chemical composition, antioxidant and antibacterial activities of peel’s essential oils of different pomelo varieties in the south of Vietnam. Int. Food Res. J. 2015, 22, 2426–2431. [Google Scholar]
- Singh, P.; Shukla, R.; Prakash, B.; Kumar, A.; Singh, S.; Mishra, P.K.; Dubey, N.K. Chemical profile, antifungal, antiaflatoxigenic and antioxidant activity of Citrus maxima Burm. and Citrus sinensis (L.) Osbeck essential oils and their cyclic monoterpene, DL-limonene. Food Chem. Toxicol. 2010, 48, 1734–1740. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Cheng, Y.; Zhang, T.; Li, Y.; Han, L.; Liang, G. Characterization of Oxygenated Heterocyclic Compounds and in vitro Antioxidant Activity of Pomelo Essential Oil. Drug Des. Dev. Ther. 2021, 15, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Oboh, G.; Olasehinde, T.A.; Ademosun, A.O. Inhibition of enzymes linked to type-2 diabetes and hypertension by essential oils from peels of orange and lemon. Int. J. Food Prop. 2017, 20, 586–594. [Google Scholar] [CrossRef] [Green Version]
- Itoh, K.; Matsukawa, T.; Murata, K.; Deguchi, T.; Yamagami, M.; Tomohiro, N.; Murata, K.; Kajiyama, S.; Endo, Y.; Matsuda, H.; et al. Effective Utilization of Citrus unshiu Plant Waste Extracts with Lipase Inhibitory Activities. J. Plant Stud. 2021, 10, 1–7. [Google Scholar] [CrossRef]
- Leporini, M.; Tundis, M.; Sicari, V.; Pellicanò, T.M.; Dugay, A.; Deguin, B.; Loizzo, M.R. Impact of extraction processes on phytochemicals content and biological activity of Citrus × clementina Hort. Ex Tan. leaves: New opportunity for under-utilized food by-products. Food Res. Int. 2020, 127, 108742. [Google Scholar] [CrossRef]
- Leporini, M.; Loizzo, M.R.; Sicari, V.; Pellicanò, T.M.; Reitano, A.; Dugay, A.; Deguin, B.; Tundis, R. Citrus × Clementina Hort. Juice Enriched with Its By-Products (Peels and Leaves): Chemical Composition, In Vitro Bioactivity, and Impact of Processing. Antioxidants 2020, 9, 298. [Google Scholar] [CrossRef] [Green Version]
- Dang, N.; Pham, N.; Bui, A.; Thuy, D.; Minh, C.; Tien Dat, N. Chemical Composition and α-Glucosidase Inhibitory Activity of Vietnamese Citrus Peels Essential Oils. J. Chem. 2016, 2016, 6787952. [Google Scholar] [CrossRef] [Green Version]
- Murali, R.; Saravanan, R. Antidiabetic effect of d-limonene, a monoterpene in streptozotocin-induced diabetic rats. Biomed. Prev. Nutr. 2012, 2, 269–275. [Google Scholar] [CrossRef]
- More, T.A.; Kulkarni, B.R.; Nalawade, M.L.; Arvindekar, A.U. Antidiabetic activity of linalool and limonene in streptozotocin-induced diabetic rat: A combinatorial therapy approach. Int. J. Pharm. Pharmaceut. Sci. 2014, 6, 159–163. [Google Scholar]
- Capetti, F.; Cagliero, C.; Marengo, A.; Bicchi, C.; Rubiolo, P.; Sgorbini, B. Bio-Guided Fractionation Driven by In Vitro α-Amylase Inhibition Assays of Essential Oils Bearing Specialized Metabolites with Potential Hypoglycemic Activity. Plants 2020, 9, 1242. [Google Scholar] [CrossRef] [PubMed]
- Basile, S.; Badalamenti, N.; Riccobono, O.; Guarino, S.; Ilardi, V.; Bruno, M.; Peri, E. Chemical composition and evaluation of insecticidal activity of Calendula incana subsp. maritima and Laserpitium siler subsp. siculum essential oils against stored products pests. Molecules 2022, 27, 588. [Google Scholar] [CrossRef]
- Council of Europe (EDQM). European Pharmacopoeia, 6th ed.; EDQM: Strasbourg, France, 2008. [Google Scholar]
- Catinella, G.; Badalamenti, N.; Ilardi, V.; Rosselli, S.; De Martino, L.; Bruno, M. The essential oil compositions of three Teucrium taxa growing wild in Sicily: HCA and PCA analyses. Molecules 2021, 26, 643. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Loizzo, M.R.; Tundis, R.; Leporini, M.; D’Urso, G.; Gagliano Candela, R.; Falco, T.; Piacente, S.; Bruno, M.; Sottile, F. Almond (Prunus dulcis cv. Casteltermini) Skin Confectionery By-Products: New Opportunity for the Development of a Functional Blackberry (Rubus ulmifolius Schott) Jam. Antioxidants 2021, 10, 1218. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.R.; Menichini, F. Natural products as α-amylase and α-glucosidase inhibitors and their hypoglycaemic potential in the treatment of diabetes: An update. Mini Rev. Med. Chem. 2010, 10, 315–331. [Google Scholar] [CrossRef] [PubMed]
- Todorovic, V.; Milenkovic, M.; Vidovic, B.; Todorovic, Z.; Sobajic, S. Correlation between antimicrobial, antioxidant activity, and polyphenols of alkalized/nonalkalized cocoa powders. J. Food Sci. 2017, 82, 1020–1027. [Google Scholar] [CrossRef]
- Caesar, L.K.; Cech, N.B. Synergy and antagonism in natural product extracts: When 1 + 1 does not equal 2. Nat. Prod. Rep. 2019, 36, 869–888. [Google Scholar] [CrossRef] [Green Version]

| Content (%) C | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No. | Compounds | LRIexp A | LRIexp B | P1 | P2 | P3 | P4 | P5 | Ident. D | Sign. E |
| 1 | α-Pinene | 937 | 1008 | 1.92 ± 0.19 d | 2.97 ± 0.25 c | 4.03 ± 0.53 a | 3.23 ± 0.36 b | 1.80 ± 0.14 e | 1, 2, 3 | ** |
| 2 | Camphene | 948 | 1069 | 0.01 ± 0.01 b | 0.07 ± 0.05 a b | 0.19 ± 01 a | - | - | 1, 2, 3 | ** |
| 3 | β-Pinene | 979 | 1102 | 4.52 ± 0.56 d | 8.23 ± 0.84 c | 9.83 ± 1.02 b | 13.99 ± 1.08 a | 1.00 ± 0.95 e | 1, 2, 3 | ** |
| 4 | β-Myrcene | 988 | 1162 | 5.31 ± 0.61 b | 5.38 ± 0.71 b | 5.91 ± 0.62 a | - | 4.72 ± 0.37 c | 1, 2, 3 | ** |
| 5 | Octanal | 1005 | 1265 | - | - | - | 3.53 ± 0.28 a | - | 1, 2 | ** |
| 6 | α-Phellandrene | 1009 | 1178 | 0.08 ± 0.07 d | 0.72 ± 0.12 b | 1.55 ± 0.21 a | - | 0.22 ± 0.11 c | 1, 2 | ** |
| 7 | 4-Carene | 1013 | 1157 | - | - | - | 0.28 ± 0.05 b | 7.90 ± 0.89 a | 1, 2 | ** |
| 8 | β-Phellandrene | 1017 | 1197 | 1.25 ± 0.21 c | 2.61 ± 0.18 b | 3.21 ± 0.32 a | - | 0.07 ± 0.05 d | 1, 2 | ** |
| 9 | o-Cymene | 1021 | 1266 | - | - | - | - | 0.66 ± 0.09 a | 1,2 | ** |
| 10 | Limonene | 1028 | 1206 | 71.13 ± 2.7 a | 62.73 ± 1.9 b | 58.66 ± 1.7 c | 32.42 ± 1.2 d | 21.72 ± 1.1 e | 1, 2, 3 | ** |
| 11 | β-cis-Ocimene | 1040 | 1234 | - | - | - | - | 1.67 ± 0.25 a | 1, 2 | ** |
| 12 | γ-Terpinene | 1047 | 1246 | 1.19 ± 0.21 b | 0.76 ± 0.02 c | - | - | 4.57 ± 0.52 a | 1, 2 | ** |
| 13 | 1-Octanol | 1071 | 1549 | - | - | - | 1.13 ± 0.09 a | - | 1, 2 | ** |
| 14 | cis-Linalool oxide | 1073 | 1435 | 1.51 ± 0.21 b | 5.51 ± 0.61 a | - | 0.62 ± 0.07 c | 0.19 ± 0.0 d | 1, 2 | ** |
| 15 | β-Linalool | 1084 | 1552 | 2.05 ± 0.36 c | 2.16 ± 0.11 c | 2.15 ± 0.18 c | 3.28 ± 0.24 b | 19.58 ± 1.3 a | 1, 2, 3 | ** |
| 16 | α-Terpinolene | 1088 | 1287 | - | - | 0.52 ± 0.09 b | - | 0.87 ± 0.10 a | 1, 2 | ** |
| 17 | trans-Linalool oxide | 1095 | 1473 | 0.79 ± 0.08 b | 2.27 ± 0.31 a | - | 0.34 ± 0.02 c | 0.13 ± 0.0 d | 1, 2 | ** |
| 18 | Heptyl acetate | 1098 | 1377 | - | - | - | 0.20 ± 0.01 a | - | 1, 2 | ** |
| 19 | neo-allo-Ocimene | 1136 | 1354 | - | - | - | - | 0.71 ± 0.08 a | 1, 2 | ** |
| 20 | β-Citronellal | 1138 | 1490 | - | - | - | 0.73 ± 0.09 a | - | 1, 2 | ** |
| 21 | Nonanal | 1143 | 1374 | - | - | 0.30 ± 0.02 b | 1.26 ± 0.12 a | - | 1, 2 | ** |
| 22 | trans-3-Pinanone | 1165 | 1532 | - | - | - | 0.04 ± 0.0 a | - | 1, 2 | ** |
| 23 | 1-Nonanol | 1179 | 1645 | - | - | - | 1.24 ± 0.17 a | - | 1, 2 | ** |
| 24 | 4-Terpineol | 1184 | 1597 | 0.44 ± 0.05 d | 0.91 ± 0.10 c | 1.76 ± 0.18 a | 1.56 ± 0.21 b | - | 1, 2, 3 | ** |
| 25 | α-Terpineol | 1196 | 1695 | 1.06 ± 0.11 e | 2.22 ± 0.26 c | 3.05 ± 0.35 b | 1.84 ± 0.18 d | 6.20 ± 0.75 a | 1, 2 | ** |
| 26 | Decanal | 1205 | 1485 | - | - | - | 0.99 ± 0.10 a | - | 1, 2 | ** |
| 27 | cis-Geraniol | 1228 | 1806 | 0.84 ± 0.09 e | 0.63 ± 0.08 d | 1.35 ± 0.15 c | 2.11 ± 0.25 b | 4.62 ± 0.51 a | 1, 2 | ** |
| 28 | β-Citral | 1241 | 1678 | 1.75 ± 0.75 b | 0.36 ± 0.02 d | 1.20 ± 0.18 c | 3.68 ± 0.43 a | - | 1, 2 | ** |
| 29 | trans-Geraniol | 1252 | 1843 | 0.49 ± 0.05 d | 0.66 ± 0.05 c | 0.44 ± 0.0 d | 3.57 ± 0.37 b | 4.19 ± 0.51 a | 1, 2 | ** |
| 30 | 1-Decanol | 1267 | 1760 | - | - | 0.78 ± 0.09 a | - | - | 1, 2 | ** |
| 31 | α-Citral | 1272 | 1723 | 1.53 ± 0.21 b | - | 1.06 ± 0.18 c | 8.50 ± 0.76 a | - | 1, 2 | ** |
| 32 | Linalyl propionate | 1314 | 1678 | - | - | - | - | 0.16 ± 0.0 a | 1, 2 | ** |
| 33 | Neryl acetate | 1365 | 1722 | - | - | - | - | 4.64 ± 0.4 b | 1, 2 | ** |
| 34 | Geranyl acetate | 1383 | 1751 | - | - | - | - | 4.70 ± 0.3 b | 1, 2 | ** |
| 35 | Caryophyllene | 1421 | 1583 | - | 0.34 ± 0.02 b | 0.95 ± 1.01 a | 0.53 ± 0.05 a,b | - | 1, 2, 3 | ** |
| 36 | α-Bergamotene | 1438 | 1565 | - | - | - | 0.79 ± 0.09 a | - | 1, 2 | ** |
| 37 | β-Bisabolene | 1507 | 1728 | - | - | 1.27 ± 0.15 a | - | - | 1, 2 | ** |
| 38 | Epiglobulol | 1571 | 2047 | - | - | - | 0.15 ± 0.0 a | - | 1, 2 | ** |
| 39 | trans-Nerolidol | 1573 | 2053 | - | 0.50 ± 0.08 a | - | - | - | 1, 2 | ** |
| 40 | Nerolidyl acetate | 1680 | 2272 | - | - | - | 0.08 ± 0.02 a | - | 1, 2 | ** |
| 41 | cis-Farnesol | 1687 | 2339 | 0.88 ± 0.09 a | - | - | - | - | 1, 2 | ** |
| 42 | Nootkatone | 1806 | 2505 | - | - | - | 4.45 ± 0.52 a | - | 1, 2 | ** |
| Monoterpene hydrocarbons | 85.41 | 83.47 | 83.90 | 49.92 | 45.91 | |||||
| Oxygenated monoterpenes | 10.46 | 14.72 | 11.01 | 26.27 | 44.41 | |||||
| Sesquiterpene hydrocarbons | - | 0.34 | 2.22 | 1.32 | - | |||||
| Oxygenated sesquiterpenes | 0.88 | 0.50 | - | 4.68 | - | |||||
| Others | - | - | 1.08 | 8.35 | - | |||||
| Total | 96.75 | 99.03 | 98.21 | 90.54 | 98.67 | |||||
| Compounds | P1P2 | P1P3 | P1P4 | P1P5 | P2P3 | P2P4 | P2P5 | P3P4 | P3P5 | P4P5 | Mix ^ | Sign. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| α-Pinene | 2.49 ± 0.21 d,e | 2.98 ± 0.30 b,c | 2.58 ± 0.26 d | 1.90 ± 0.21 f | 3.50 ± 0.34 a | 3.11 ± 0.29 b | 2.38 ± 0.20 e | 3.64 ± 0.31 a | 2.93 ± 0.27 b,c | 2.53 ± 0.23 d,e | 2.80 ± 0.25 c | ** |
| Camphene | 0.07 ± 0.0 b | 0.10 ± 0.00 a b | - | - | 0.13 ± 0.01 a | 0.03 ± 0.00 c | 0.02 ± 0.00 c | 0.11 ± 0.01 a,b | 0.09 ± 0.00 a,b | - | 0.05 ± 0.00 c | ** |
| β-Pinene | 6.38 ± 0.61 e | 7.18 ± 0.73 d | 9.25 ± 0.93 c | 2.75 ± 0.2 h | 9.03 ± 0.89 c | 11.10 ± 1.12 b | 4.63 ± 0.38 g | 11.90 ± 1.02 a | 5.43 ± 0.53 f | 7.50 ± 0.61 d | 7.53 ± 0.68 d | ** |
| β-Myrcene | 5.35 ± 0.49 bc | 5.60 ± 0.52 a | 2.66 ± 0.24 f | 5.00 ± 0.46 c | 5.65 ± 0.51 a | 2.70 ± 0.23 f | 5.07 ± 0.46 c | 2.95 ± 0.21 e | 5.33 ± 0.47 b,c | 2.35 ± 0.21 g | 4.28 ± 0.38 d | ** |
| Octanal | - | - | 1.72 ± 0.18 a,b | - | - | 1.67 ± 0.54 c | - | 1.78 ± 0.19 a,b | - | 1.82 ± 0.20 a | 0.70 ± 0.09 d | ** |
| α-Phellandrene | 0.40 ± 0.03 d | 0.83 ± 0.07 b | 0.05 ± 0.00 f | 0.15 ± 0.00 e | 1.13 ± 0.09 a | 0.35 ± 0.02 d | 0.48 ± 0.01 c,d | 0.78 ± 0.05 b,c | 0.88 ± 0.09 b | 0.10 ± 0.02 e,f | 0.53 ± 0.04 c | ** |
| 4-Carene | - | - | 0.15 ± 0.00 d | 3.95 ± 0.42 b | - | 0.15 ± 0.000 d | 3.99 ± 0.41 a,b | 0.13 ± 0.0 d | 4.00 ± 0.35 a,b | 4.11 ± 0.43 a | 1.63 ± 0.14 c | ** |
| β-Phellandrene | 1.93 ± 0.18 b,c | 2.23 ± 0.24 b | 0.63 ± 0.57 f | 0.65 ± 0.59 f | 2.90 ± 0.18 a | 1.30 ± 0.15 e | 1.35 ± 0.14 d,e | 1.60 ± 0.18 c | 1.65 ± 0.18 c | 0.03 ± 0.00 g | 1.43 ± 0.12 d | ** |
| o-Cymene | - | - | - | 0.31 ± 0.02 a,b | - | - | 0.29 ± 0.02 b | - | 0.30 ± 0.03 a,b | 0.35 ± 0.03 a | 0.13 ± 0.01 c | ** |
| D-Limonene | 66.93 ± 5.23 a | 64.90 ± 5.12 b | 51.78 ± 4.83 d | 46.43 ± 3.41 ef | 60.70 ± 5.02 c | 47.58 ± 3.56 e | 42.23 ± 3.24 g | 45.55 ± 3.41 f | 40.20 ± 3.07 h | 27.08 ± 1.89 i | 49.33 ± 3.63 d,e | ** |
| β-cis-Ocimene | - | - | - | 0.81 ± 0.08 a | - | - | 0.83 ± 0.09 a | - | 0.81 ± 0.08 a | 0.82 ± 0.07 a | 0.33 ± 0.01 b | ** |
| γ-Terpinene | 0.98 ± 0.11 d | 0.61 ± 0.05 e | 0.60 ± 0.06 e | 2.88 ± 0.24 a | 0.42 ± 0.03 f | 0.38 ± 0.02 f | 2.68 ± 0.31 a,b | - | 2.28 ± 0.26 b | 2.31 ± 0.29 b | 1.30 ± 0.14 c | ** |
| 1-Octanol | - | - | 0.54 ± 0.04 a | - | - | 0.55 ± 0.05 a | - | 0.54 ± 0.05 a | - | 0.50 ± 0.04 a | 0.23 ± 0.01 b | ** |
| cis-Linalool oxide | 3.50 ± 0.29 a | 0.75 ± 0.06 f | 1.08 ± 0.12 e | 0.85 ± 0.08 f | 2.75 ± 0.28 c | 3.08 ± 0.32 b | 2.85 ± 0.25 c | 0.30 ± 0.02 g,h | 0.10 ± 0.00 h | 0.40 ± 0.02 g | 1.58 ± 0.13 d | ** |
| β-Linalool | 2.10 ± 0.18 e | 2.10 ± 0.15 e | 2.68 ± 0.25 d | 10.83 ± 1.05 b | 2.15 ± 0.24 e | 2.73 ± 0.29 d | 10.88 ± 1.07 b | 2.73 ± 0.31 d | 10.88 ± 1.04 b | 11.43 ± 1.12 a | 5.85 ± 0.61 c | ** |
| α-Terpinolen | - | 0.22 ± 0.00 c | - | 0.43 ± 0.05 b,c | 0.20 ± 0.01 c | - | 0.44 ± 0.03 b,c | 0.23 ± 0.01 c | 0.70 ± 0.05 a | 0.44 ± 0.02 b,c | 0.28 ± 0.01 c | ** |
| trans-Linalool oxide | 1.53 ± 0.14 a | 0.40 ± 0.05 e | 0.58 ± 0.07 d,e | 0.45 ± 0.04 e | 1.13 ± 0.15 c | 1.30 ± 0.18 b,c | 1.20 ± 0.17 c | 0.18 ± 0.15 f,g | 0.08 ± 0.00 g | 0.23 ± 0.01 f | 0.70 ± 0.05 d | ** |
| Heptyl acetate | - | - | 0.11 ± 0.00 a | - | - | 0.08 ± 0.00 a,b | - | 0.10 ± 0.01 a | - | 0.09 ± 0.00 a | 0.06 ± 0.00 b | ** |
| Neo-allo-ocimene | - | - | - | 0.36 ± 0.03 a | - | - | 0.35 ± 0.02 a | - | 0.33 ± 0.03 a | 0.33 ± 0.02 a | 0.18 ± 0.01 b | ** |
| β-Citronellal | - | - | 0.39 ± 0.04 a | - | - | 0.36 ± 0.03 a | - | 0.38 ± 0.04 a | - | 0.37 ± 0.05 a | 0.13 ± 0.00 b | ** |
| Nonanal | - | 0.15 ± 0.00 e | 0.65 ± 0.08 c | - | 0.12 ± 0.00 e | 0.69 ± 0.08 b,c | - | 0.78 ± 0.06 a | 0.13 ± 0.00 e | 0.64 ± 0.05 c | 0.29 ± 0.01 d | ** |
| trans-3-Pinanone | - | - | 0.03 ± 0.00 a | - | - | 0.03 ± 0.00 a | - | 0.02 ± 0.00 a | - | 0.01 ± 0.00 a | 0.01 ± 0.00 a | ** |
| 1-Nonanol | - | - | 0.63 ± 0.05 a | - | - | 0.58 ± 0.04 b | - | 0.61 ± 0.05 a,b | - | 0.59 ± 0.06 b | 0.27 ± 0.01 c | ** |
| 4-Terpineol | 0.68 f | 1.10 ± 0.15 c,d | 1.00 ± 0.12 d | 0.23 ± 0.19 g | 1.33 ± 0.21 b,c | 1.23 ± 0.23 c | 0.45 ± 0.05 f,g | 1.65 ± 0.22 a | 0.88 ± 0.09 e | 0.78 ± 0.07 e,f | 0.93 ± 0.09 d,e | ** |
| α-Terpineol | 1.65 f | 2.05 ± 0.21 ef | 1.45 ± 0.15 g | 3.63 ± 0.34 c | 2.63 ± 0.25 d,e | 2.03 ± 0.19 e,f | 4.20 ± 0.34 a,b | 2.45 ± 0.23 e | 4.63 ± 0.38 a | 4.03 ± 0.31 b | 2.88 ± 0.29 d | ** |
| Decanal | - | - | 0.45 ± 0.04 a | - | - | 0.46 ± 0.04 a | - | 0.45 ± 0.05 a | - | 0.40 ± 0.03 a,b | 0.24 ± 0.01 b | ** |
| cis-Geraniol | 0.73 f | 1.10 ± 0.11 e | 1.48 ± 0.15 d | 2.73 ± 0.23 b | 1.00 ± 0.09 e | 1.38 ± 0.12 d,e | 2.63 ± 0.21 b | 1.73 ± 0.15 c,d | 2.98 ± 0.32 a,b | 3.38 ± 0.36 a | 1.90 ± 0.18 c | ** |
| β-Citral | 1.05 f | 1.48 ± 0.15 d,e | 2.73 ± 0.28 a | 0.88 ± 0.08 f,g | 0.78 ± 0.06 g | 2.03 ± 0.16 c,d | 0.18 ± 0.01 i | 2.45 ± 0.23 b | 0.60 ± 0.03 h | 1.85 ± 0.12 d | 1.40 ± 0.11 e | ** |
| trans-Geraniol | 0.58 f | 0.48 ± 0.02 g | 2.03 ± 0.21 d | 2.35 ± 0.24 c | 0.55 ± 0.04 f,g | 2.13 ± 0.18 c,d | 2.43 ± 0.22 b | 2.00 ± 0.19 d | 2.33 ± 0.23 bc | 3.88 ± 0.28 a | 1.88 ± 0.18 e | ** |
| 1-Decanol | - | 0.40 ± 0.03 a | - | - | 0.38 ± 0.02 a,b | - | - | 0.42 ± 0.03 a | 0.34 ± 0.02 b | - | 0.37 ± 0.04 a,b | ** |
| α-Citral | 0.78 f | 1.30 ± 0.11 e | 5.03 ± 0.48 a | 0.78 ± 0.05 f | 0.53 ± 0.04 g | 4.25 ± 0.41 c | - | 4.78 ± 0.45 b | 0.53 ± 0.04 g | 4.25 ± 0.39 c | 2.23 ± 0.21 d | ** |
| Linalyl propionate | - | - | - | 0.11 ± 0.01 a | - | - | 0.08 ± 0.00 a,b | - | 0.10 ± 0.00 a | 0.07 ± 0.00 a,b | 0.03 ± 0.00 b | ** |
| Neryl acetate | - | - | - | 2.32 ± 0.25 a | - | - | 2.28 ± 0.22 a | - | 2.31 ± 0.24 a | 2.32 ± 0.23 a | 0.93 ± 0.08 b | ** |
| Geranyl acetate | - | - | - | 2.34 ± 0.21 a | - | - | 2.35 ± 0.25 a | - | 2.31 ± 0.22 a | 2.36 ± 0.24 a | 0.99 ± 0.09 b | ** |
| Caryophyllene | 0.18 e | 0.51 ± 0.04 b | 0.28 ± 0.02 d | - | 0.65 ± 0.06 a,b | 0.43 ± 0.04 c | 0.18 ± 0.01 e | 0.75 ± 0.08 a | 0.48 ± 0.05 bc | 0.28 ± 0.03 d | 0.38 ± 0.04 cd | ** |
| α-Bergamotene | - | - | 0.39 ± 0.04 a | - | - | 0.36 ± 0.03 a | - | 0.36 ± 0.03 a | - | 0.38 ± 0.04 a | 0.17 ± 0.01 b | ** |
| β-Bisabolene | - | 0.63 ± 0.07 a | - | - | 0.61 ± 0.06 a | - | - | 0.60 ± 0.05 a | 0.63 ± 0.06 a | - | 0.24 ± 0.02 b | ** |
| Epiglobulol | - | - | 0.11a | - | - | 0.11a | - | 0.09 ab | - | 0.08 a,b | 0.04 b | ** |
| trans-Nerolidol | 0.29 ± 0.03 a | - | - | - | 0.28 ± 0.03 a | 0.28 ± 0.02 a | 0.26 ± 0.02 a | - | - | - | 0.10 ± 0.00 b | ** |
| Nerolidyl acetate | - | - | 0.07 ± 0.00 a | - | - | 0.06 ± 0.00 a | - | 0.03 ± 0.00 a | - | 0.03 ± 0.00 a | 0.02 ± 0.00 a | ** |
| cis-Farnesol | 0.39 ± 0.02 ab | 0.44 ± 0.03 a | 0.48 ± 0.05 a | 0.46 ± 0.05 a | - | - | - | - | - | - | 0.18 ± 0.00 b | ** |
| Nootkatone | - | - | 2.28 ± 0.23 a | - | - | 2.20 ± 0.21 a,b | - | 2.18 ± 0.22 b | - | 2.22 ± 0.23 a | 0.87 ± 0.09 c | ** |
| Monoterpene hydrocarbons | 84.53 | 84.65 | 67.70 | 65.62 | 83.66 | 66.70 | 64.74 | 66.89 | 64.93 | 47.95 | 69.80 | |
| Oxygenated monoterpenes | 12.60 | 10.76 | 18.48 | 27.50 | 12.85 | 20.55 | 29.53 | 18.67 | 27.73 | 35.36 | 21.44 | |
| Sesquiterpene hydrocarbons | 0.18 | 1.14 | 0.67 | - | 1.26 | 0.79 | 0.18 | 1.71 | 1.11 | 0.66 | 0.79 | |
| Oxygenated sesquiterpenes | 0.68 | 0.44 | 2.94 | 0.46 | 0.28 | 2.65 | 0.26 | 2.22 | - | 2.33 | 1.21 | |
| Others | - | 0.55 | 4.10 | - | 0.50 | 4.02 | - | 4.68 | 0.47 | 4.04 | 2.16 |
| ABTS (IC50 μg/mL) | DPPH (IC50 μg/mL) | FRAP μM Fe2+/g | β-Carotene Bleaching Test (IC50 μg/mL) | |
|---|---|---|---|---|
| C. maxima EO | ||||
| P1 | 22.24 ± 2.07 a | 27.23 ± 2.02 b | 65.76 ± 3.27 g | 42.24 ± 2.66 j |
| P2 | 31.82 ± 2.12 h | 35.32 ± 2.13 h | 58.59 ± 4.09 i | 32.22 ± 245 d |
| P3 | 25.45 ± 1.66 c | 32.27 ± 2.64 g | 65.29 ± 3.12 g | 40.66 ± 2.80 i |
| P4 | 32.56 ± 2.20 i | 35.26 ± 2.23 h | 64.67 ± 3.82 h | 25.56 ± 2.38 b |
| P5 | 30.42 ± 2.45 g | 38.14 ± 2.92 i | 56.26 ± 3.63 j | 46.23 ± 2.73 l |
| EO blends (1:1 v/v) | ||||
| P1P2 | 28.12 ± 2.23 e | 32.25 ± 1.81 g | 66.28 ± 3.12 f | 26.28 ± 2.11 c |
| P1P3 | 29.01 ± 2.45 f | 35.12 ± 2.27 h | 69.56 ± 3.02 d | 21.87 ± 2.28 a |
| P1P4 | 35.23 ± 2.12 l | 29.89 ± 2.78 d | 65.23 ± 3.11 k | 40.33 ± 2.87 i |
| P1P5 | 31.24 ± 2.43 h | 31.71 ± 2.26 f | 69.12 ± 3.09 d | 35.23 ± 2.03 f |
| P2P3 | 33.26 ± 2.56 j | 35.24 ± 2.95 h | 47.11 ± 3.04 l | 39.34 ± 3.02 h |
| P2P4 | 31.12 ± 2.24 h | 28.76 ± 2.35 c | 68.89 ± 3.78 d,e | 34.22 ± 2.94 e |
| P2P5 | 26.92 ± 2.45 d | 28.12 ± 2.31 c | 71.32 ± 3.09 c | 38.76 ± 2.07 g |
| P3P4 | 32.31 ± 2.65 i | 32.25 ± 2.24 g | 72.33 ± 3.12 b | 32.72 ± 2.32 d |
| P3P5 | 34.24 ± 2.78 k | 30.12 ± 2.20 e | 65.35 ± 3.20 g | 45.16 ± 2.94 k |
| P4P5 | 35.27 ± 2.68 l | 29.22 ± 2.14 d | 72.78 ± 3.23 a | 48.51 ± 2.83 m |
| Mix ^ | 24.28 ± 2.71 b | 26.82 ± 2.98 a | 68.06 ± 3.54 e | 45.41 ± 3.34 k |
| Sign. | ** | ** | ** | ** |
| Lipase | α-Amylase | α-Glucosidase | |
|---|---|---|---|
| C. maxima EO | |||
| P1 | 26.32 ± 2.32 d | 35.28 ± 2.13 j | 28.78 ± 2.24 f |
| P2 | 34.12 ± 2.14 j | 34.22 ± 1.89 i | 25.67 ± 2.31 c |
| P3 | 37.26 ± 2.53 l | 27.56 ± 2.82 e | 45.23 ± 2.21 k |
| P4 | 23.22 ± 2.02 a | 25.23 ± 2.05 d | 48.69 ± 2.78 l |
| P5 | 24.23 ± 2.12 b | 39.86 ± 2.54 m | 54.56 ± 3.45 m |
| EO blends (1:1 v/v) | |||
| P1P2 | 28.21 ± 2.12 e | 38.18 ± 2.21 l | 24.01 ± 1.88 b |
| P1P3 | 33.87 ± 2.90 i | 36.19 ± 2.11 k | 27.87 ± 1.72 e |
| P1P4 | 39.12 ± 2.61 m | 21.43 ± 2.05 a | 28.23 ± 2.02 f |
| P1P5 | 30.95 ± 2.23 g | 22.46 ± 2.87 b | 37.26 ± 2.34 j |
| P2P3 | 29.12 ± 2.45 f | 30.22 ± 2.18 g | 29.12 ± 2.62 g |
| P2P4 | 25.22 ± 2.46 c | 24.71 ± 2.19 c | 26.55 ± 2.76 d |
| P2P5 | 26.76 ± 2.69 d | 28.55 ± 2.23 f | 21.67 ± 1.98 a |
| P3P4 | 28.24 ± 2.08 e | 31.05 ± 2.94 h | 32.05 ± 2.03 h |
| P3P5 | 35.06 ± 2.12 k | 36.45 ± 2.98 k | 27.85 ± 2.28 e |
| P4P5 | 31.12 ± 2.05 h | 30.23 ± 2.75 g | 34.67 ± 2.65 i |
| Mix ^ | 30.12 ± 2.52 g | 25.67 ± 2.05 d | 28.12 ± 2.50 f |
| Sign. | ** | ** | ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badalamenti, N.; Bruno, M.; Schicchi, R.; Geraci, A.; Leporini, M.; Tundis, R.; Loizzo, M.R. Reuse of Food Waste: The Chemical Composition and Health Properties of Pomelo (Citrus maxima) Cultivar Essential Oils. Molecules 2022, 27, 3273. https://doi.org/10.3390/molecules27103273
Badalamenti N, Bruno M, Schicchi R, Geraci A, Leporini M, Tundis R, Loizzo MR. Reuse of Food Waste: The Chemical Composition and Health Properties of Pomelo (Citrus maxima) Cultivar Essential Oils. Molecules. 2022; 27(10):3273. https://doi.org/10.3390/molecules27103273
Chicago/Turabian StyleBadalamenti, Natale, Maurizio Bruno, Rosario Schicchi, Anna Geraci, Mariarosaria Leporini, Rosa Tundis, and Monica Rosa Loizzo. 2022. "Reuse of Food Waste: The Chemical Composition and Health Properties of Pomelo (Citrus maxima) Cultivar Essential Oils" Molecules 27, no. 10: 3273. https://doi.org/10.3390/molecules27103273
APA StyleBadalamenti, N., Bruno, M., Schicchi, R., Geraci, A., Leporini, M., Tundis, R., & Loizzo, M. R. (2022). Reuse of Food Waste: The Chemical Composition and Health Properties of Pomelo (Citrus maxima) Cultivar Essential Oils. Molecules, 27(10), 3273. https://doi.org/10.3390/molecules27103273











