Extraction Optimization of Astragaloside IV by Response Surface Methodology and Evaluation of Its Stability during Sterilization and Storage
Abstract
1. Introduction
2. Materials and Methods
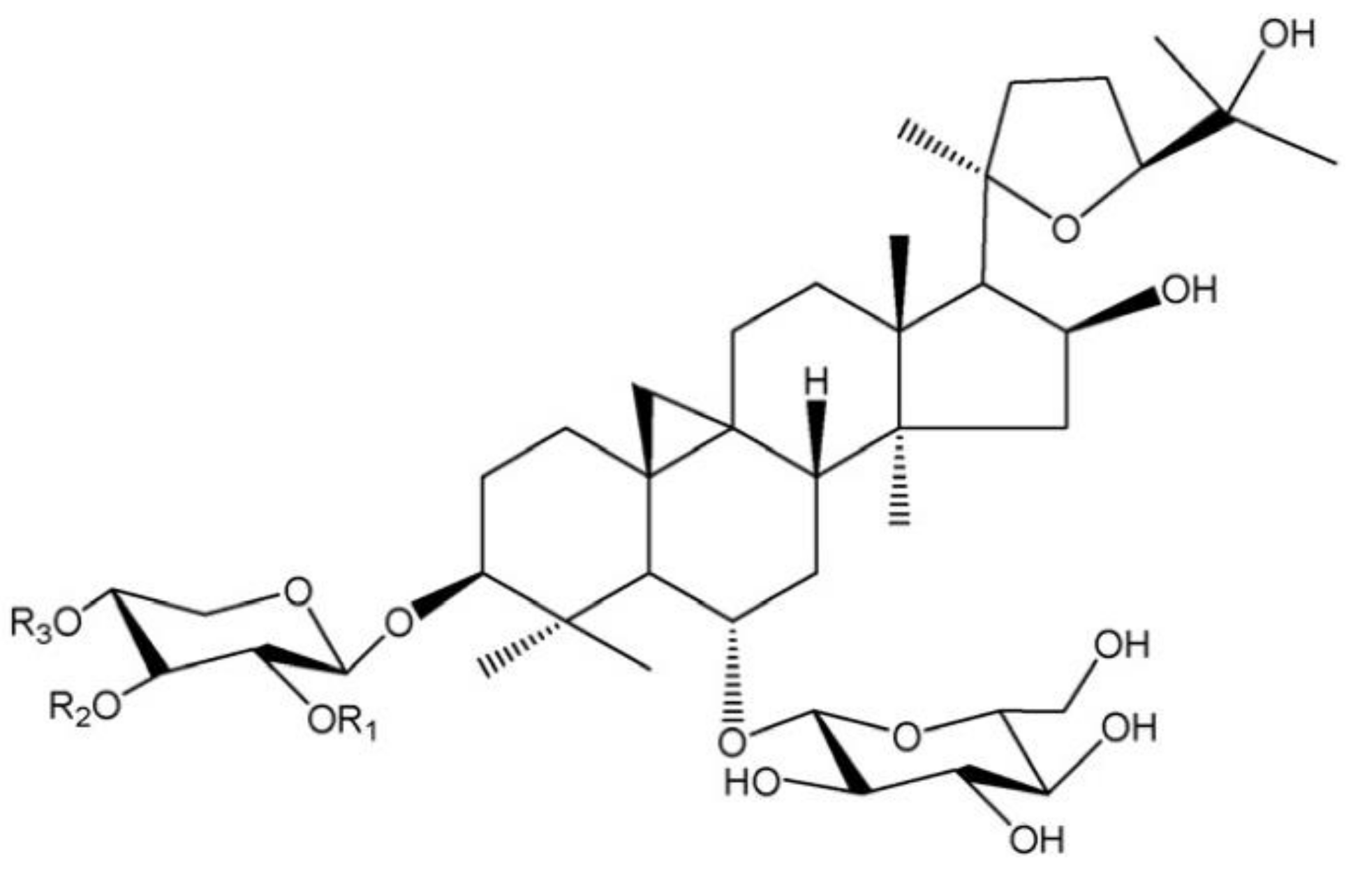
2.1. Materials
2.2. Quantitation of Astragaloside I, Astragaloside II, and Astragaloside IV
2.3. Quantitative Validation Calibration Curves
2.3.1. Calibration Curves, Limits of Detection (LOD) and Quantification (LOQ)
2.3.2. Precision and Accuracy
2.4. Single-Factor Experiments of the Extraction Process
2.4.1. Ammonia Concentration
2.4.2. Solid–Liquid Ratio
2.4.3. Soaking Time
2.4.4. Extraction Time
2.4.5. Extraction Temperature
2.4.6. Extraction Times
2.5. Response Surface Method to Optimize Extraction Conditions
2.6. The Retention Rate of the Astragaloside IV Under Different Sterilization Conditions
2.7. The Retention Rate of Astragaloside IV in Different pH Value Solutions Stored at 4 or 25 °C
2.8. Statistical Analysis
3. Results and Discussion
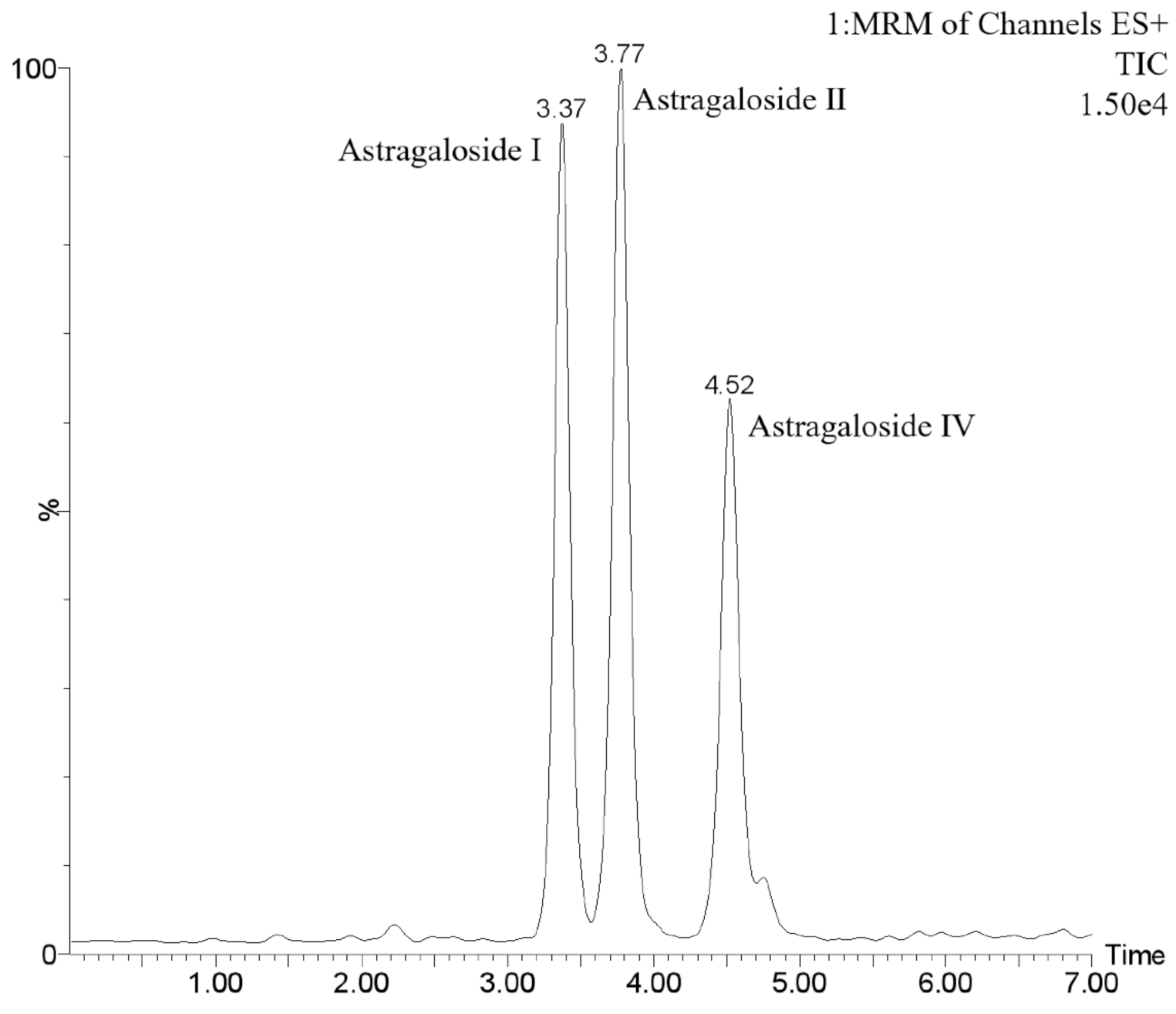
3.1. Identification of Astragaloside I, Astragaloside II, and Astragaloside IV by UPLC–MS/MS
Quantitative Validation
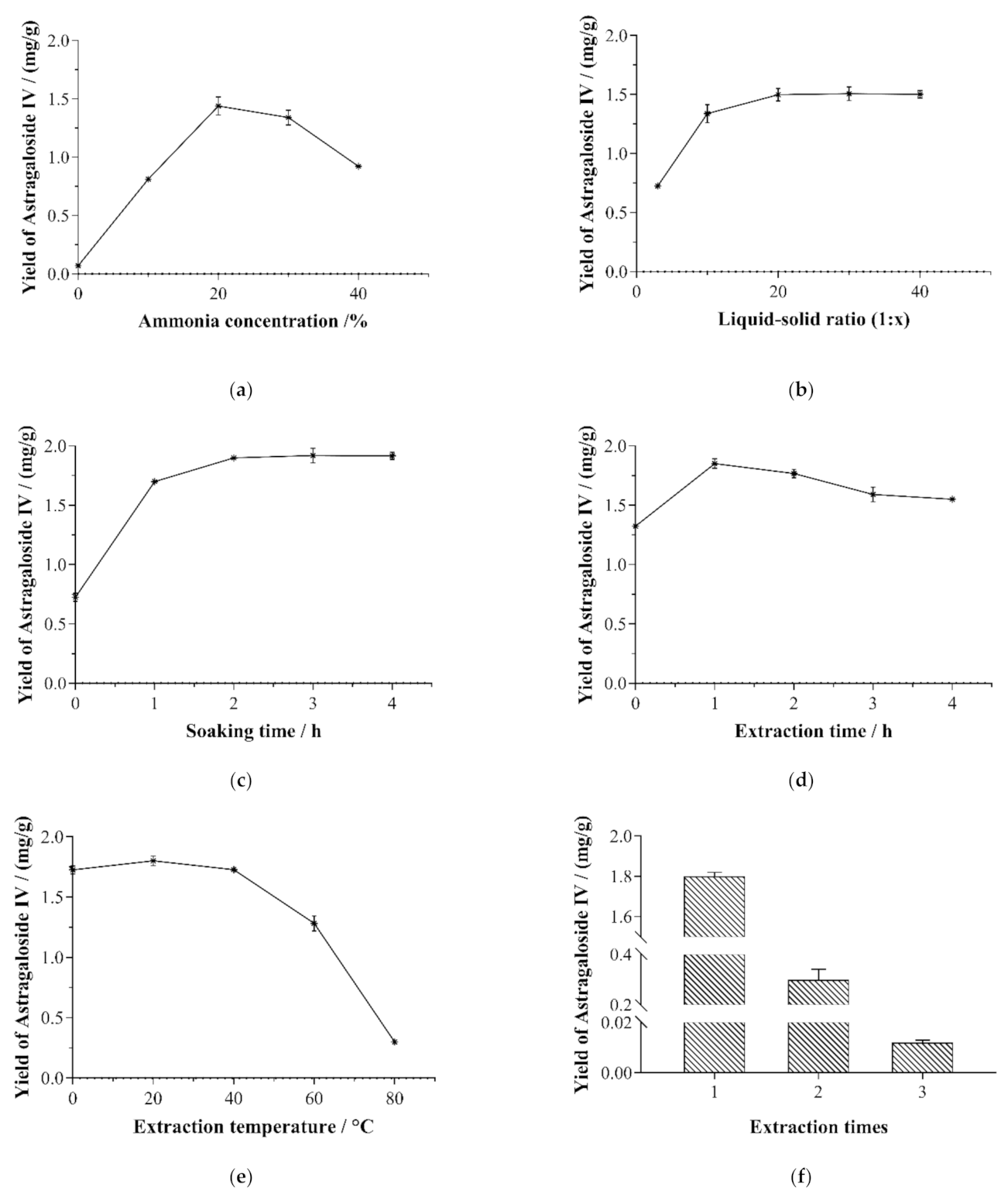
3.2. Single-Factor Experiments
3.2.1. Ammonia Concentration
3.2.2. Solid–Liquid Ratio
3.2.3. Soaking Time
3.2.4. Extraction Time
3.2.5. Extraction Temperature
3.2.6. Extraction Times
3.3. Response Surface Methodology
3.3.1. Response Surface Experimental Design and Response
3.3.2. The ANOVA of Response Surface Methodology
3.3.3. The Impact of Different Factors on the Yield of Astragaloside IV
3.3.4. Optimization and Validation Procedures
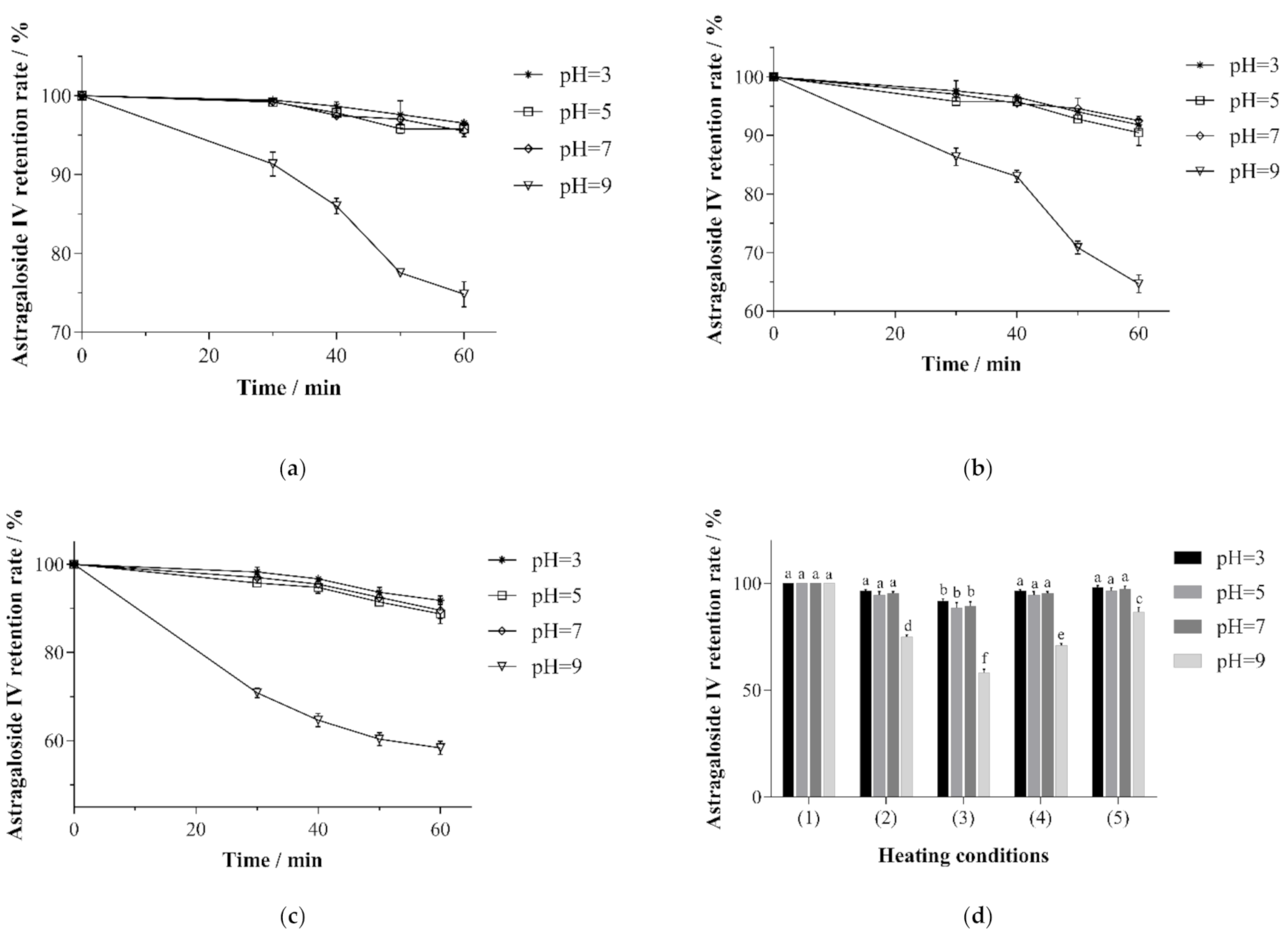
3.4. The Changes in the Astragaloside IV Retention Rate Under Different Sterilization Conditions
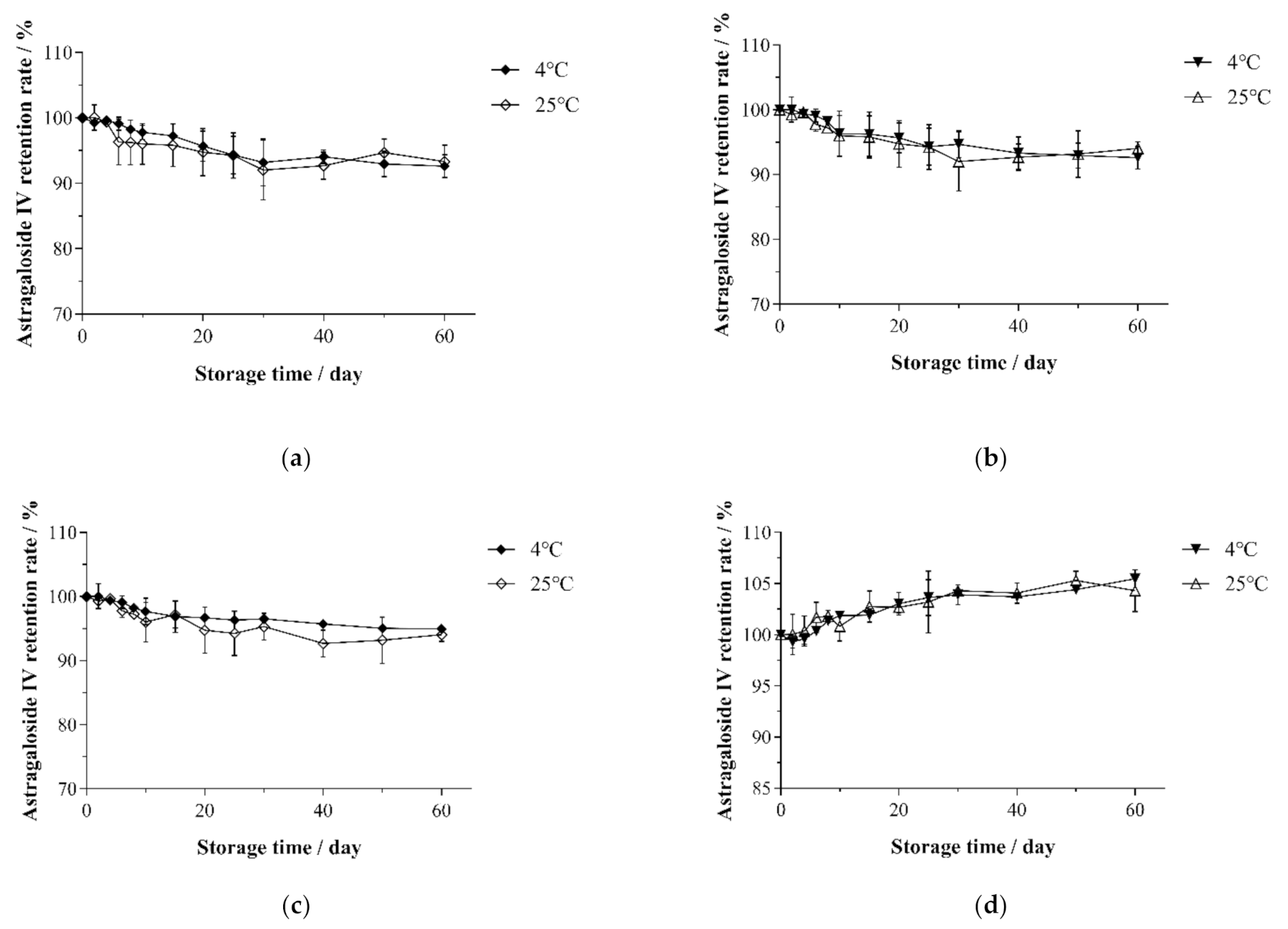
3.5. Changes in the Astragaloside IV Retention Rate under Different Storage Conditions
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Sample Availability
Appendix A
| Treatment Number | Ammonia Concentration (A) (%, v/v) | Solid–Liquid Ratio (B) (mL/g) | Soaking Time (C) (min) | Extraction Time (D) (min) | Content of Astragaloside IV (mg/g) | Astragaloside Ⅰ (mg/g) | Astragaloside Ⅱ (mg/g) |
|---|---|---|---|---|---|---|---|
| 1 | 25 | 10 | 90 | 0 | 2.012 ± 0.073 | Not detected | Not detected |
| 2 | 25 | 20 | 120 | 30 | 2.301 ± 0.011 | Not detected | Not detected |
| 3 | 25 | 10 | 60 | 30 | 1.904 ± 0.023 | Not detected | Not detected |
| 4 | 25 | 15 | 120 | 0 | 2.235 ± 0.084 | Not detected | Not detected |
| 5 | 30 | 15 | 60 | 30 | 0.701 ± 0.036 | 0.107 ± 0.005 | 0.142 ± 0.010 |
| 6 | 25 | 20 | 90 | 0 | 2.049 ± 0.042 | Not detected | Not detected |
| 7 | 25 | 20 | 90 | 60 | 2.283 ± 0.086 | Not detected | Not detected |
| 8 | 25 | 15 | 90 | 30 | 2.215 ± 0.061 | Not detected | Not detected |
| 9 | 30 | 15 | 120 | 30 | 1.436 ± 0.021 | Not detected | 0.086 ± 0.002 |
| 10 | 30 | 20 | 90 | 30 | 0.938 ± 0.025 | 0.067 ± 0.004 | 0.103 ± 0.003 |
| 11 | 20 | 15 | 60 | 30 | 1.473 ± 0.077 | Not detected | 0.079 ± 0.002 |
| 12 | 25 | 15 | 90 | 30 | 2.157 ± 0.113 | Not detected | Not detected |
| 13 | 30 | 15 | 90 | 0 | 0.851 ± 0.032 | 0.076 ± 0.003 | 0.129 ± 0.004 |
| 14 | 30 | 10 | 90 | 30 | 0.940 ± 0.023 | 0.071 ± 0.002 | 0.116 ± 0.003 |
| 15 | 25 | 15 | 60 | 60 | 1.981 ± 0.049 | Not detected | Not detected |
| 16 | 20 | 20 | 90 | 30 | 1.702 ± 0.026 | Not detected | Not detected |
| 17 | 25 | 20 | 60 | 30 | 2.063 ± 0.036 | Not detected | Not detected |
| 18 | 25 | 10 | 120 | 30 | 2.602 ± 0.072 | Not detected | Not detected |
| 19 | 25 | 15 | 90 | 30 | 2.151 ± 0.034 | Not detected | Not detected |
| 20 | 25 | 15 | 90 | 30 | 2.220 ± 0.065 | Not detected | Not detected |
| 21 | 20 | 15 | 90 | 0 | 1.566 ± 0.073 | Not detected | 0.067 ± 0.002 |
| 22 | 20 | 15 | 90 | 60 | 1.757 ± 0.029 | Not detected | Not detected |
| 23 | 20 | 15 | 120 | 30 | 1.861 ± 0.016 | Not detected | Not detected |
| 24 | 25 | 15 | 90 | 30 | 2.205 ± 0.035 | Not detected | Not detected |
| 25 | 25 | 10 | 90 | 60 | 2.102 ± 0.039 | Not detected | Not detected |
| 26 | 30 | 15 | 90 | 60 | 0.882 ± 0.026 | 0.069 ± 0.003 | 0.125 ± 0.002 |
| 27 | 25 | 15 | 120 | 60 | 2.590 ± 0.108 | Not detected | Not detected |
| 28 | 20 | 10 | 90 | 30 | 1.660 ± 0.025 | Not detected | 0.062 ± 0.002 |
| 29 | 25 | 15 | 60 | 0 | 1.801 ± 0.049 | Not detected | Not detected |
References
- Joung, J.Y.; Lee, J.S.; Cho, J.H.; Lee, D.S.; Ahn, Y.C.; Son, C. The Efficacy and Safety of Myelophil, an Ethanol Extract Mixture of Astragali Radix and Salviae Radix, for Chronic Fatigue Syndrome: A Randomized Clinical Trial. Front. Pharmacol. 2019, 10, 991–1003. [Google Scholar] [CrossRef]
- Turck, D.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Kearney, J.; Maciuk, A. Safety of a botanical extract derived from Panax notoginseng and Astragalus membranaceus (AstraGin (TM)) as a novel food pursuant to Regulation (EU) 2015/2283. Efsa J. 2020, 18, 1–16. [Google Scholar]
- Butkute, B.; Benetis, R.; Padarauskas, A.; Ceseviciene, J.; Dagilyte, A.; Taujenis, L.; Rodovicius, H.; Lemeziene, N.; Lemeziene, N. Young herbaceous legumes–a natural reserve of bioactive compounds and antioxidants for healthy food and supplements. J. Appl. Botany Food Q./Angewandte Botanik 2017, 90, 346–353. [Google Scholar]
- Korany, M.A.; Moneeb, M.S.; Asaad, A.M.; El-Sebakhy, N.A.; El-Banna, A.A. Analysis of astragalosides I, II and IV in some Egyptian Astragalus species and Astragalus dietary supplements using high-performance liquid chromatography/evaporative light scattering detector and non-parametric regression. Phytochem. Anal. 2020, 31, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Wenjing, Z.; Biwang, L.; Meng, Z.; Shanshan, W.; Nan, Q. Preparation technology and efficacy of the anti-haze Qingfei drink. Sci. Technol. Food Ind. 2020, 15, 43–52. [Google Scholar]
- Guo, Z.; Lou, Y.; Kong, M.; Luo, Q.; Liu, Z.; Wu, J. A Systematic Review of Phytochemistry, Pharmacology and Pharmacokinetics on Astragali Radix: Implications for Astragali Radix as a Personalized Medicine. Int. J. Mol. Sci. 2019, 20, 1463. [Google Scholar] [CrossRef]
- Graziani, V.; Scognamiglio, M.; Esposito, A.; Fiorentino, A.; D’Abrosca, B. Chemical diversity and biological activities of the saponins isolated from Astragalus genus: Focus on Astragaloside IV. Phytochem. Rev. 2019, 1133–1166. [Google Scholar] [CrossRef]
- Qi, Y.; Gao, F.; Hou, L.; Wan, C. Anti-Inflammatory and Immunostimulatory Activities of Astragalosides. Am. J. Chin. Med. 2017, 45, 1157–1167. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Lu, Q.; Li, Q.; Ji, Y.; Chen, W.; Xue, X. Astragaloside IV inhibits breast cancer cell invasion by suppressing Vav3 mediated Rac1/MAPK signaling. Int. Immunopharmacol. 2017, 42, 195–202. [Google Scholar] [CrossRef]
- Chan, W.S.; Durairajan, S.S.K.; Lu, J.H.; Wang, Y.; Xie, L.X.; Kum, W.F.; Koo, I.; Yung, K.K.L.; Li, M. Neuroprotective effects of Astragaloside IV in 6-hydroxydopamine-treated primary nigral cell culture. Neurochem. Int. 2009, 55, 414–422. [Google Scholar] [CrossRef]
- Yang, H.; Zhao, Z.; Gao, S.; Lei, J.; Jiang, Y. Extraction of astragaloside in Astragali broken-cell pieces by accelerated solvent extraction. Lishizhen Medicine and Materia Medica Research 2017, 28, 310–312. [Google Scholar]
- Liu, M.; Zhou, J.; Zhang, Q.-W.; Liu, F. Amic solution hydrolysis for improving content of astragaloside IV in extract of Radix Astragali. China J. Chin. Mater. Medica 2008, 33, 635–643. [Google Scholar]
- Chu, C.; Liu, E.H.; Qi, L.W.; Li, P. Transformation of astragalosides from Radix Astragali under acidic, neutral, and alkaline extraction conditions monitored by LC-ESI-TOF/MS. Chin. J. Nat. Med. 2014, 12, 314–320. [Google Scholar] [CrossRef]
- Qi, L.-W.; Cao, J.; Li, P.; Wang, Y.-X. Rapid and sensitive quantitation of major constituents in Danggui Buxue Tang by ultra-fast HPLC–TOF/MS. J. Pharm. Biomed. Anal. 2009, 49, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.; Wang, Y.; Liu, Z.; Wang, S.; Dong, F.; Wang, Z.; Qiao, Y.; Zhang, J. The chemical transformations for Radix Astragali via different alkaline wash conditions by quantitative and qualitative analyses. J. Pharm. Biomed. Anal. 2020, 185, 113164. [Google Scholar] [CrossRef]
- Zheng, N.; Shi, Y.; Ji, Y.; Lu, W. Stability and transformation of astragaloside Ⅳ, Ⅲ and Ⅰ. Cent. S Pharm. 2014, 22, 1062–1065. [Google Scholar]
- Feng, X.; Huang, K.; Wei, G.; Huang, Y. Changes of Astragalosides and Astragaloside Ⅳ Contents in Acid and Alkali Solution. Trad. Chin. Drug Res. Clin. Plarmacol. 2010, 21, 533–537. [Google Scholar]
- Linhares, M.D.F.D.; Filho, E.G.A.; Silva, L.M.A.; Fonteles, T.V.; Wurlitzer, N.J.; de Brito, E.S.; Fernandes, F.A.; Rodrigues, S. Thermal and non-thermal processing effect on açai juice composition. Food Res. Int. 2020, 136, 109506. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Cui, X.; Zhang, Z.; Chen, L.; Zhang, Y.; Wang, C.; Yang, X.; Qu, Y.; Xiong, Y. Optimisation of Ethanol-Reflux Extraction of Saponins from Steamed Panax notoginseng by Response Surface Methodology and Evaluation of Hematopoiesis Effect. Molecules 2018, 23, 1206. [Google Scholar] [CrossRef]
- Fang, J.-L.; Luo, Y.; Yuan, K.; Guo, Y.; Jin, S.-H. Preparation and evaluation of an encapsulated anthocyanin complex for enhancing the stability of anthocyanin. LWT 2020, 117, 108543. [Google Scholar] [CrossRef]
- Du, K.; Li, J.; Guo, X.; Li, Y.; Chang, Y. New Method for Determination of Astragaloside Ⅳ in Astragali Radix. Chin. J. Exp. Trad. Med. Formulae 2020, 26, 132–137. [Google Scholar]
- Shaw, L.-H.; Lin, L.-C.; Tsai, T.-H. HPLC–MS/MS Analysis of a Traditional Chinese Medical Formulation of Bu-Yang-Huan-Wu-Tang and Its Pharmacokinetics after Oral Administration to Rats. PLoS ONE 2012, 7, e43848. [Google Scholar] [CrossRef] [PubMed]
- Tamjidi, F.; Shahedi, M.; Varshosaz, J.; Nasirpour, A. Stability of astaxanthin-loaded nanostructured lipid carriers as affected by pH, ionic strength, heat treatment, simulated gastric juice and freeze–thawing. J. Food Sci. Technol. 2017, 54, 3132–3141. [Google Scholar] [CrossRef]
- Ghavi, P.P. The extraction process optimization of antioxidant polysaccharides from Marshmallow (Althaea officinalis L.) roots. Int. J. Biol. Macromol. 2015, 75, 51–57. [Google Scholar] [CrossRef]
- Chen, Y.; Xie, M.-Y.; Gong, X.-F. Microwave-assisted extraction used for the isolation of total triterpenoid saponins from Ganoderma atrum. J. Food Eng. 2007, 81, 162–170. [Google Scholar] [CrossRef]
- Vongsangnak, W.; Gua, J.; Chauvatcharin, S.; Zhong, J.-J. Towards efficient extraction of notoginseng saponins from cultured cells of Panax notoginseng. Biochem. Eng. J. 2004, 18, 115–120. [Google Scholar] [CrossRef]
- Du, K.-Z.; Li, J.; Guo, X.; Li, Y.; Chang, Y.-X. Quantitative Analysis of Phenolic Acids and Flavonoids in Cuscuta chinensis Lam. by Synchronous Ultrasonic-Assisted Extraction with Response Surface Methodology. J. Anal. Methods Chem. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Zhang, X.-F.; Yang, J.-L.; Chen, J.; Shi, Y.-P. Optimization of a Decoction Process for an Herbal Formula Using a Response Surface Methodology. J. AOAC Int. 2017, 100, 1776–1784. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wang, M.; Zhang, Y.; Zhou, J.; Xu, L.; Xia, G.; Yan, Z.; Shen, Y.; Yang, H. Efficient and clean preparation of rare prosaikogenin D by enzymatic hydrolysis of saikosaponin B2 and response surface methodology optimization. Enzym. Microb. Technol. 2020, 142, 109690. [Google Scholar] [CrossRef]
- Yan, M.-M.; Liu, W.; Fu, Y.-J.; Zu, Y.-G.; Chen, C.-Y.; Luo, M. Optimisation of the microwave-assisted extraction process for four main astragalosides in Radix Astragali. Food Chem. 2010, 119, 1663–1670. [Google Scholar] [CrossRef]


| The Factors | Level | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| Ammonia concentration (A) (%, v/v) | 20 | 25 | 30 |
| Solid–liquid ratio (B) (mL/g) | 10:1 | 15:1 | 20:1 |
| Soaking time (C) (min) | 60 | 90 | 120 |
| Extraction time (D) (min) | 0 | 30 | 60 |
| Qualitative Ion Pair | Quantitative Ion Pair | Cone Hole Voltage/V | Collision Energy/eV | |
|---|---|---|---|---|
| Astragaloside I | 891.5 > 891.5 | 891.5 > 891.5 | 13 | 10 |
| Astragaloside II | 849.4 > 849.4 | 849.4 > 849.4 | 14 | 9 |
| 827.5 > 827.5 | 14 | 10 | ||
| 827.5 > 143 | 14 | 11 | ||
| Astragaloside IV | 785.4 > 473.39 | 785.4 > 143.11 | 14 | 12 |
| 785.4 > 143.11 | 14 | 10 |
| Parameters | Substances | ||
|---|---|---|---|
| Astragaloside I | Astragaloside II | Astragaloside IV | |
| Calibration curve | y = 400.12x + 1309.5 | y = 655.11x + 1353.9 | y = 1047.7x + 197.46 |
| Test range (μg/mL) | 0.083–2.67 | 0.042–1.33 | 0.026–0.83 |
| r2 | 0.9991 | 0.9935 | 0.9997 |
| LOD (μg) | 0.006 | 0.003 | 0.002 |
| LOQ (μg) | 0.024 | 0.015 | 0.009 |
| Intra-day RSD (%) (n = 5) | 1.5 | 3.7 | 1.4 |
| Inter-day RSD (%) (n = 5) | 1.8 | 1.2 | 1.1 |
| Recoveries (%) | 94.1 ± 4.3 | 98.4 ± 5.8 | 96.5 ± 1.9 |
| Content of Astragaloside IV | Sum of Squares | Degrees of Freedom | Mean Square | F-Value | p-value |
|---|---|---|---|---|---|
| Model | 7.71 | 14 | 0.55 | 135.37 | <0.0001 |
| A | 1.52 | 1 | 1.52 | 373.33 | <0.0001 |
| B | 8.00 × 10−4 | 1 | 8.00 × 10−4 | 0.20 | 0.6642 |
| C | 0.80 | 1 | 0.80 | 196.77 | <0.0001 |
| D | 0.099 | 1 | 0.099 | 24.42 | 0.0002 |
| AB | 6.25 × 10−4 | 1 | 6.25 × 10−4 | 0.15 | 0.7010 |
| AC | 0.029 | 1 | 0.029 | 7.10 | 0.0185 |
| AD | 6.40 × 10−3 | 1 | 6.40 × 10−3 | 1.57 | 0.2304 |
| BC | 0.053 | 1 | 0.053 | 13.00 | 0.0029 |
| BD | 5.77 × 10−3 | 1 | 5.77 × 10−3 | 1.42 | 0.2533 |
| CD | 8.1 × 10−3 | 1 | 8.1 × 10−3 | 1.99 | 0.1801 |
| A2 | 4.81 | 1 | 4.81 | 1181.04 | <0.0001 |
| B2 | 9.87 × 10−4 | 1 | 9.87 × 10−4 | 0.24 | 0.6301 |
| C2 | 9.33 × 10−3 | 1 | 9.33 × 10−3 | 2.29 | 0.1523 |
| D2 | 0.031 | 1 | 0.031 | 7.50 | 0.0160 |
| Residual | 0.057 | 14 | 4.07 × 10−3 | ||
| Lack of Fit | 0.052 | 10 | 5.25 × 10−3 | 4.64 | 0.0761 |
| Pure Error | 4.52 × 10−3 | 4 | 1.13 × 10−3 | ||
| Cor Total | 7.77 | 28 | |||
| R-Squared | 0.9927 | ||||
| Adj R-Squared | 0.9853 | ||||
| C.V. % | 3.52 | ||||
| Adequate Precision | 41.691 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, L.; Wei, K.; Jiang, J.; Zhang, L. Extraction Optimization of Astragaloside IV by Response Surface Methodology and Evaluation of Its Stability during Sterilization and Storage. Molecules 2021, 26, 2400. https://doi.org/10.3390/molecules26082400
Xu L, Wei K, Jiang J, Zhang L. Extraction Optimization of Astragaloside IV by Response Surface Methodology and Evaluation of Its Stability during Sterilization and Storage. Molecules. 2021; 26(8):2400. https://doi.org/10.3390/molecules26082400
Chicago/Turabian StyleXu, Lin, Kongjiong Wei, Jiaolong Jiang, and Lianfu Zhang. 2021. "Extraction Optimization of Astragaloside IV by Response Surface Methodology and Evaluation of Its Stability during Sterilization and Storage" Molecules 26, no. 8: 2400. https://doi.org/10.3390/molecules26082400
APA StyleXu, L., Wei, K., Jiang, J., & Zhang, L. (2021). Extraction Optimization of Astragaloside IV by Response Surface Methodology and Evaluation of Its Stability during Sterilization and Storage. Molecules, 26(8), 2400. https://doi.org/10.3390/molecules26082400






