Bioactivity Performance of Pure Mg after Plasma Electrolytic Oxidation in Silicate-Based Solutions
Abstract
1. Introduction
2. Results and Discussion
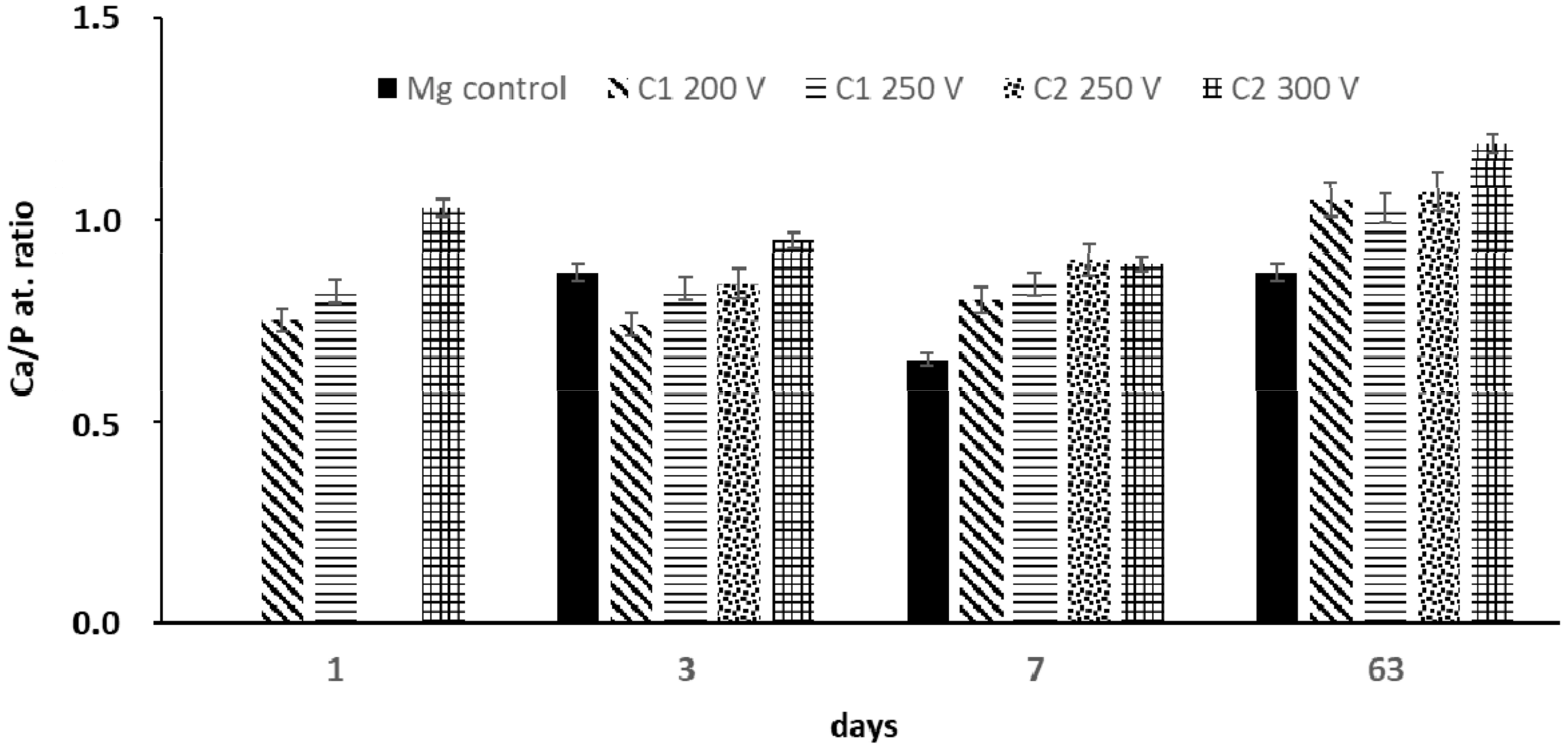
2.1. Coatings Characterization
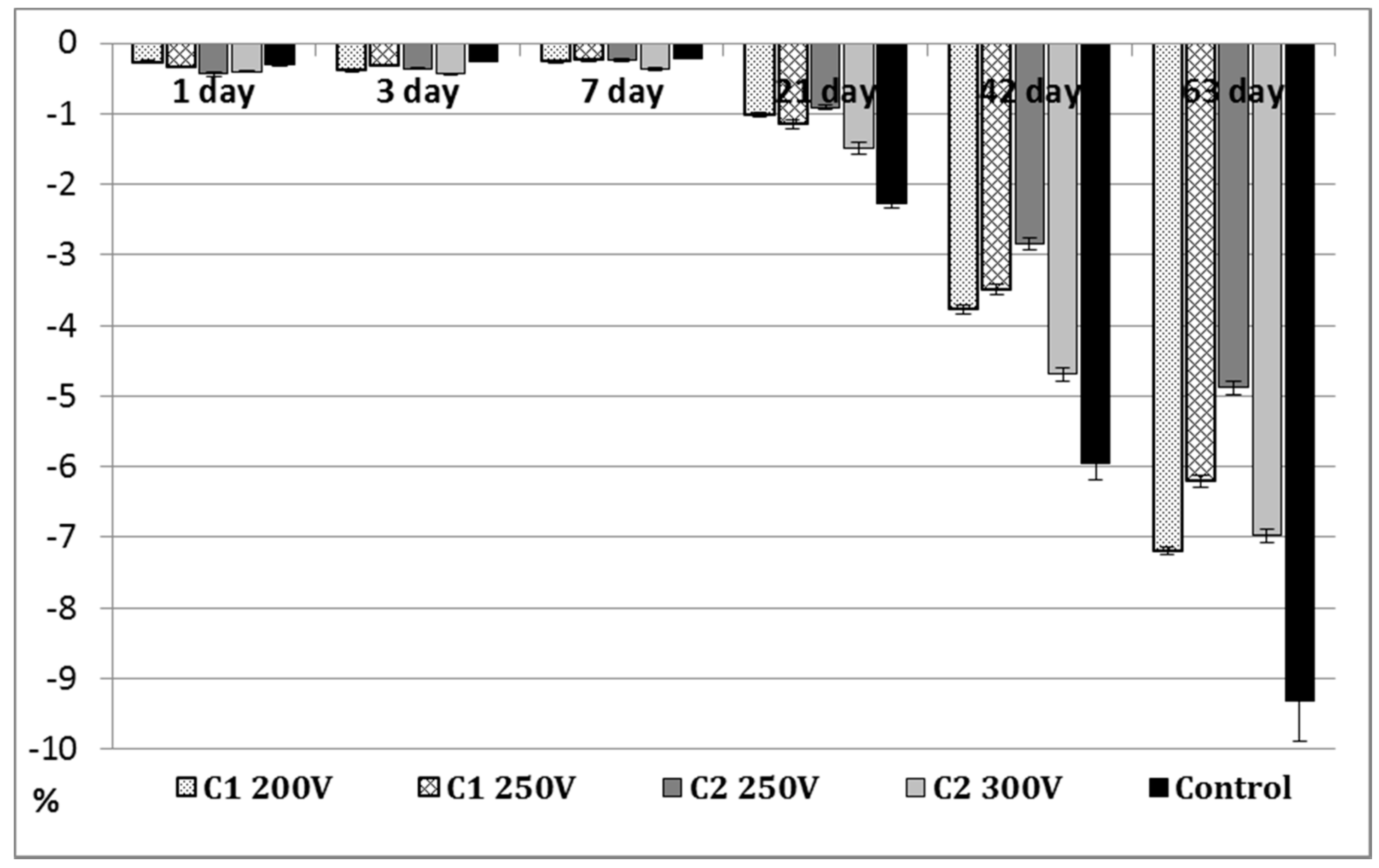
2.2. Long-Term Immersion Test in SBF
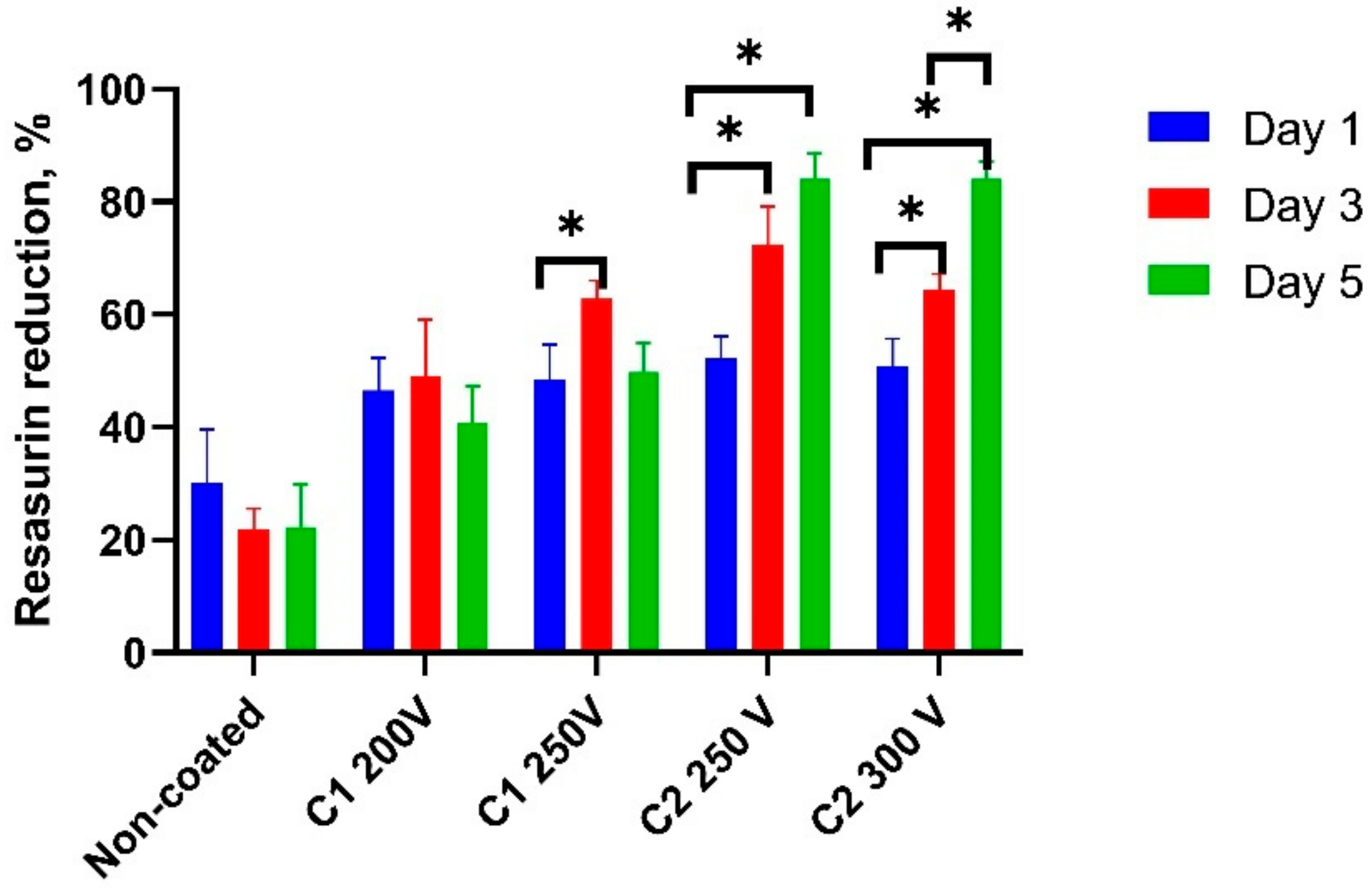
2.3. Cell Viability
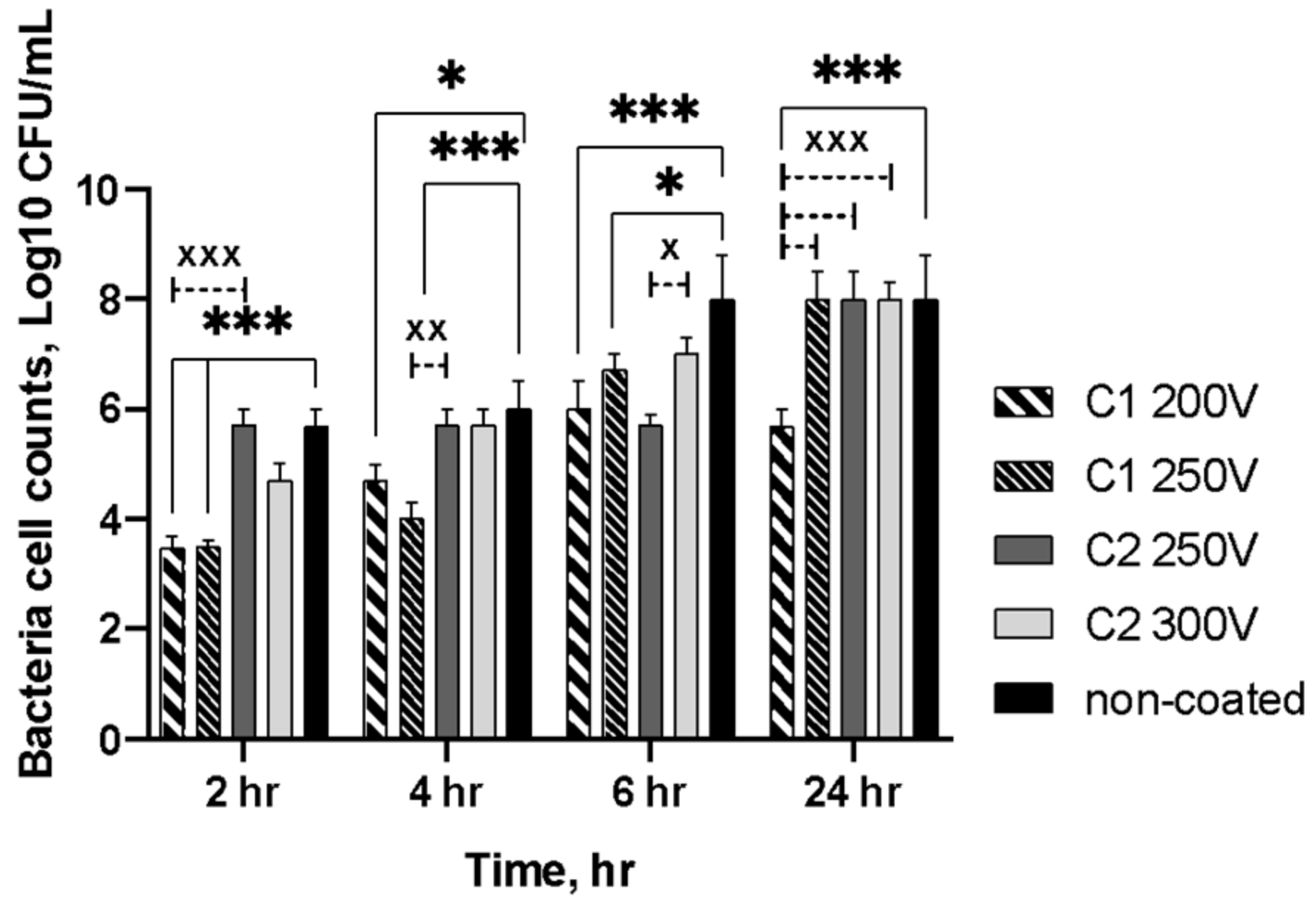
2.4. Antibacterial Properties
3. Materials and Methods
3.1. Materials
3.2. Plasma Electrolytic Oxidation (PEO)
3.3. Surface Analysis
3.3.1. SEM and EDX Analysis
3.3.2. Surface Roughness
3.3.3. Contact Angle Measurement
3.3.4. Photoluminescence Analysis
3.4. Long–Term Immersion Test in SBF
3.5. Cell Culture
3.6. In Vitro Antimicrobial Activity Assay
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability:
References
- Li, J.; Jansen, J.A.; Walboomers, X.F.; van den Beucken, J.J. Mechanical Aspects of Dental Implants and Osseointegration: A Narrative Review. J. Mech. Behav. Biomed. Mater. 2020, 103, 103574. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hady Gepreel, M.; Niinomi, M. Biocompatibility of Ti-alloys for Long-term Implantation. J. Mech. Behav. Biomed. Mater. 2013, 20, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Fage, S.W.; Muris, J.; Jakobsen, S.S.; Thyssen, J.P. Titanium: A Review on Exposure, Release, Penetration, Allergy, Epidemiology, and Clinical Reactivity. Contact Dermatitis 2016, 74, 323–345. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Xu, J.K.; Hopkins, C.; Chow, D.H.K.; Qin, L. Biodegradable Magnesium-Based Implants in Orthopedics—A General Review and Perspectives. Adv. Sci. 2020, 7, 1902443. [Google Scholar] [CrossRef]
- Kamrani, S.; Fleck, C. Biodegradable Magnesium Alloys as Temporary Orthopaedic Implants: A Review. BioMetals 2019, 32, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Heise, S.; Virtanen, S.; Boccaccini, A.R. Tackling Mg alloy Corrosion by Natural Polymer Coatings—A review. J. Biomed. Mater. Res. Part A 2016, 104, 2628–2641. [Google Scholar] [CrossRef]
- Ali, M.; Elsherif, M.; Salih, A.E.; Ul-Hamid, A.; Hussein, M.; Park, S.; Yetisen, A.K.; Butt, H. Surface modification and cytotoxicity of Mg-based bio-alloys: An overview of recent advances. J. Alloys Compd. 2020, 825, 154140. [Google Scholar] [CrossRef]
- Agarwal, S.; Curtin, J.; Duffy, B.; Jaiswal, S. Biodegradable Magnesium Alloys for Orthopaedic Applications: A Review on Corrosion, Biocompatibility and Surface Modifications. Mater. Sci. Eng. C 2016, 68, 948–963. [Google Scholar] [CrossRef]
- Pogorielov, M.; Husak, E.; Solodivnik, A.; Zhdanov, S. Magnesium-Based Biodegradable Alloys: Degradation, Application, and Alloying Elements. Interv. Med. Appl. Sci. 2017, 9, 27–38. [Google Scholar] [CrossRef]
- Wei, S.; Ma, J.X.; Xu, L.; Gu, X.S.; Ma, X.L. Biodegradable Materials for Bone Defect Repair. Mil. Med. Res. 2020, 7, 1–25. [Google Scholar] [CrossRef]
- Höhlinger, M.; Christa, D.; Zimmermann, V.; Heise, S.; Boccaccini, A.R.; Virtanen, S. Influence of Proteins on The Corrosion Behavior of a Chitosan-Bioactive Glass Coated Magnesium Alloy. Mater. Sci. Eng. C 2019, 100, 706–714. [Google Scholar] [CrossRef]
- Gray-Munro, J.E.; Seguin, C.; Strong, M. Influence of Surface Modification on the In Vitro Corrosion Rate of Magnesium Alloy Az31. J. Biomed. Mater. Res. Part A 2009, 91, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.L.; Yan, Y.H.; Wan, T.; Yang, H. Poly(L-Lactic Acid)/Hydroxyapatite/Collagen Composite Coatings on AZ31 Magnesium Alloy for Biomedical Application. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2013, 227, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Kunjukunju, S.; Roy, A.; Ramanathan, M.; Lee, B.; Candiello, J.E.; Kumta, P.N. A Layer-By-Layer Approach to Natural Polymer-Derived Bioactive Coatings on Magnesium Alloys. Acta Biomaterialia 2013, 9, 8690–8703. [Google Scholar] [CrossRef]
- Pina, S.; Oliveira, J.M.; Reis, R.L. Natural-Based Nanocomposites for Bone Tissue Engineering and Regenerative Medicine: A Review. Adv. Mater. 2015, 27, 1143–1169. [Google Scholar] [CrossRef] [PubMed]
- Xiong, P.; Jia, Z.; Zhou, W. Osteogenic and pH Stimuli-Responsive Self-Healing Coating on Biomedical Mg-1Ca Alloy. Acta Biomaterialia 2019, 92, 336–350. [Google Scholar] [CrossRef]
- Kyrylenko, S.; Warchoł, F.; Oleshko, O. Effects of The Sources of Calcium and Phosphorus on The Structural and Functional Properties of Ceramic Coatings on Titanium Dental Implants Produced by Plasma Electrolytic Oxidation. Mater. Sci. Eng. C 2021, 119, 111607. [Google Scholar] [CrossRef]
- Echeverry-Rendon, M.; Duque, V.; Quintero, D.; Harmsen, M.C.; Echeverria, F. Novel Coatings Obtained by Plasma Electrolytic Oxidation to Improve the Corrosion Resistance of Magnesium-based Biodegradable Implants. Surf. Coat. Technol. 2018, 354, 28–37. [Google Scholar] [CrossRef]
- Dong, K.; Song, Y.; Shan, D.; Han, E.H. Corrosion Behavior of a Self-Sealing Pore Micro-Arc Oxidation Film on AM60 Magnesium Alloy. Corros. Sci. 2015, 100, 275–283. [Google Scholar] [CrossRef]
- Peng, F.; Wang, D.; Tian, Y.; Cao, H.; Qiao, Y.; Liu, X. Sealing the Pores of PEO Coating with Mg-Al Layered Double Hydroxide: Enhanced Corrosion Resistance, Cytocompatibility and Drug Delivery Ability. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Koo, B.H.; Jung, Y.G.; Lee, J.H.; Choi, D. Effect of K2zrf6 Concentration on The Two-Step Peo Coating Prepared on AZ91 Mg Alloy In Alkaline Silicate Solution. Materials 2020, 13, 499. [Google Scholar] [CrossRef]
- Bigham, A.; Saudi, A.; Rafienia, M.; Rahmati, S.; Bakhtiyari, H.; Salahshouri, F.; Sattary, M.; Hassanzadeh-Tabrizi, S.A. Electrophoretically Deposited Mesoporous Magnesium Silicate with Ordered Nanopores as An Antibiotic-Loaded Coating On Surface-Modified Titanium. Mater. Sci. Eng. C 2019, 96, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Wierzbicka, E.; Pillado, B.; Mohedano, M.; Arrabal, R.; Matykina, E. Calcium Doped Flash-PEO Coatings for Corrosion Protection of Mg Alloy. Metals 2020, 10, 916. [Google Scholar] [CrossRef]
- Wang, C.; Duoling, X.; Shujun, L.; Chen, Y.; Xiliu, Z.; Yi, H.; Dongsheng, Y. Effect of Pore Size on the Physicochemical Properties and Osteogenesis of Ti6Al4V Porous Scaffolds with Bionic Structure. ACS Omega 2020, 5, 28684–28692. [Google Scholar] [CrossRef] [PubMed]
- Brennan, C.M.; Eichholz, K.F.; Hoey, D.A. The effect of pore size within fibrous scaffolds fabricated using melt electrowriting on human bone marrow stem cell osteogenesis. Biomed. Mater. 2019, 14, 065016. [Google Scholar] [CrossRef] [PubMed]
- Verhorstert, K.W.J.; Guler, Z.; De Boer, L.; Riool, M.; Roovers, J.P.W.R.; Zaat, S.A.J. In Vitro Bacterial Adhesion and Biofilm Formation on Fully Absorbable Poly-4-hydroxybutyrate and Nonabsorbable Polypropylene Pelvic Floor Implants. ACS Appl. Mater. Interfaces 2020, 12, 53646–53653. [Google Scholar] [CrossRef]
- Tian, P.; Liu, X.; Ding, C. In Vitro Degradation Behavior and Cytocompatibility of Biodegradable AZ31 Alloy With PEO/HT Composite Coating. Colloids Surf. B Biointerfaces 2015, 128, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Santos-Coquillat, A.; Martínez-Campos, E.; Vargas-Alfredo, N.; Arrabal, R.; Rodríguez-Hernández, J.; Matykina, E. Hierarchical Functionalized Polymeric-Ceramic Coatings on Mg-Ca Alloys for Biodegradable Implant Applications. Macromol. Biosci. 2019, 19, 1900179. [Google Scholar] [CrossRef]
- Aziz, B.K.; Karim, M.A.H. Efficient Catalytic Photodegradation of Methylene Blue From Medical Lab Wastewater Using MgO Nanoparticles Synthesized by Direct Precipitation Method. React. Kinet. Mech. Catal. 2019, 128, 1127–1139. [Google Scholar] [CrossRef]
- Janet, C.M.; Viswanathan, B.; Viswanath, R.P.; Varadarajan, T.K. Characterization and Photoluminescence Properties of MgO Microtubes Synthesized From Hydromagnesite Flowers. J. Phys. Chem. C 2007, 111, 10267–10272. [Google Scholar] [CrossRef]
- El-Shaer, A.; Abdelfatah, M.; Mahmoud, K.R.; Momay, S.; Eraky, M.R. Correlation Between Photoluminescence and Positron Annihilation Lifetime Spectroscopy to Characterize Defects in Calcined MgO Nanoparticles as A First Step to Explain Antibacterial Activity. J. Alloys Compd. 2020, 817, 152799. [Google Scholar] [CrossRef]
- Sellaiyan, S.; Uedono, A.; Sivaji, K.; Janet Priscilla, S.; Sivasankari, J.; Selvalakshmi, T. Vacancy Defects and Defect Clusters in Alkali Metal Ion-Doped MgO Nanocrystallites Studied by Positron Annihilation and Photoluminescence Spectroscopy. Appl. Phys. A Mater. Sci. Process. 2016, 122, 1–9. [Google Scholar] [CrossRef]
- Soma, H.; Uchino, T. Blue and Orange Photoluminescence and Surface Band-Gap Narrowing in Lithium-Doped MgO Microcrystals. J. Phys. Chem. C. 2017, 121, 1884–1892. [Google Scholar] [CrossRef]
- Li, X. Pressure-induced photoluminescence of MgO. J. Phys. Condens. Matter. 2018, 30, 194002. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Atrens, A. Understanding Magnesium Corrosion. A Framework for Improved Alloy Performance. Adv. Eng. Mater. 2003, 5, 837–858. [Google Scholar] [CrossRef]
- Yang, J.; Lu, X.; Blawert, C.; Di, S.; Zheludkevich, M.L. Microstructure and Corrosion Behavior of Ca/P Coatings Prepared On Magnesium By Plasma Electrolytic Oxidation. Surf Coatings Technol. 2017, 319, 359–369. [Google Scholar] [CrossRef]
- Sakiewicz, P.; Piotrowski, K.; Bajorek, A.; Młynarek, K.; Babilas, R.; Simka, W. Surface Modification of Biomedical MgCa4.5 and MgCa4.5Gd0.5 Alloys by Micro-Arc Oxidation. Materials 2021, 14, 1360. [Google Scholar] [CrossRef]
- Habibovic, P.; Barrère, F.; Van Blitterswijk, C.A.; De Groot, K.; Layrolle, P. Biomimetic Hydroxyapatite Coating on Metal Implants. J. Am. Ceram. Soc. 2002, 85, 517–522. [Google Scholar] [CrossRef]
- Wang, Y.; Ouyang, Y.; Peng, X.; Mao, L.; Yuan, G.; Jiang, Y.; He, Y. Effects of Degradable Mg-Nd-Zn-Zr alloy on Osteoblastic Cell Function. Int. J. Immunopathol. Pharmacol. 2012, 25, 597–606. [Google Scholar] [CrossRef]
- Niu, J.; Yuan, G.; Liao, Y.; Mao, L.; Zhang, J.; Wang, Y.; Ding, W. Enhanced Biocorrosion Resistance and Biocompatibility of Degradable Mg-Nd-Zn-Zr Alloy by Brushite Coating. Mater. Sci. Eng. C 2013, 33, 4833–4841. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Wang, C.; Zhou, J.J.; Xue, J.; Li, W. Influence of Surface Properties on Adhesion Forces and Attachment of Streptococcus Mutans to Zirconia In Vitro. Biomed. Res. Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Sousa, C.; Teixeira, P.; Oliveira, R. Influence of Surface Properties on the Adhesion of Staphylococcus Epidermidis to Acrylic and Silicone. Int. J. Biomater. 2009, 2009, 1–9. [Google Scholar] [CrossRef]
- Katsikogianni, M.; Missirlis, Y.F.; Harris, L.; Douglas, J. Concise Review of Mechanisms of Bacterial Adhesion to Biomaterials and of Techniques Used In Estimating Bacteria-Material Interactions. Eur. Cells Mater. 2004, 8, 37–57. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Catt, D.M.; Gregory, R.L. Streptococcus Mutons Surface A-Enolase Binds Salivary Mucin MG2 and Human Plasminogen. Infect. Immun. 2004, 72, 6748–6752. [Google Scholar] [CrossRef] [PubMed]
- Myakinin, A.; Turlybekuly, A.; Pogrebnjak, A.; Mirek, A.; Bechelany, M.; Liubchak, I.; Oleshko, O.; Husak, Y.; Korniienko, V.; Leśniak-Ziółkowska, K.; et al. In vitro evaluation of electrochemically bioactivated Ti6Al4V 3D porous scaffolds. Mater. Sci. Eng. C 2021, 121, 111870. [Google Scholar] [CrossRef]

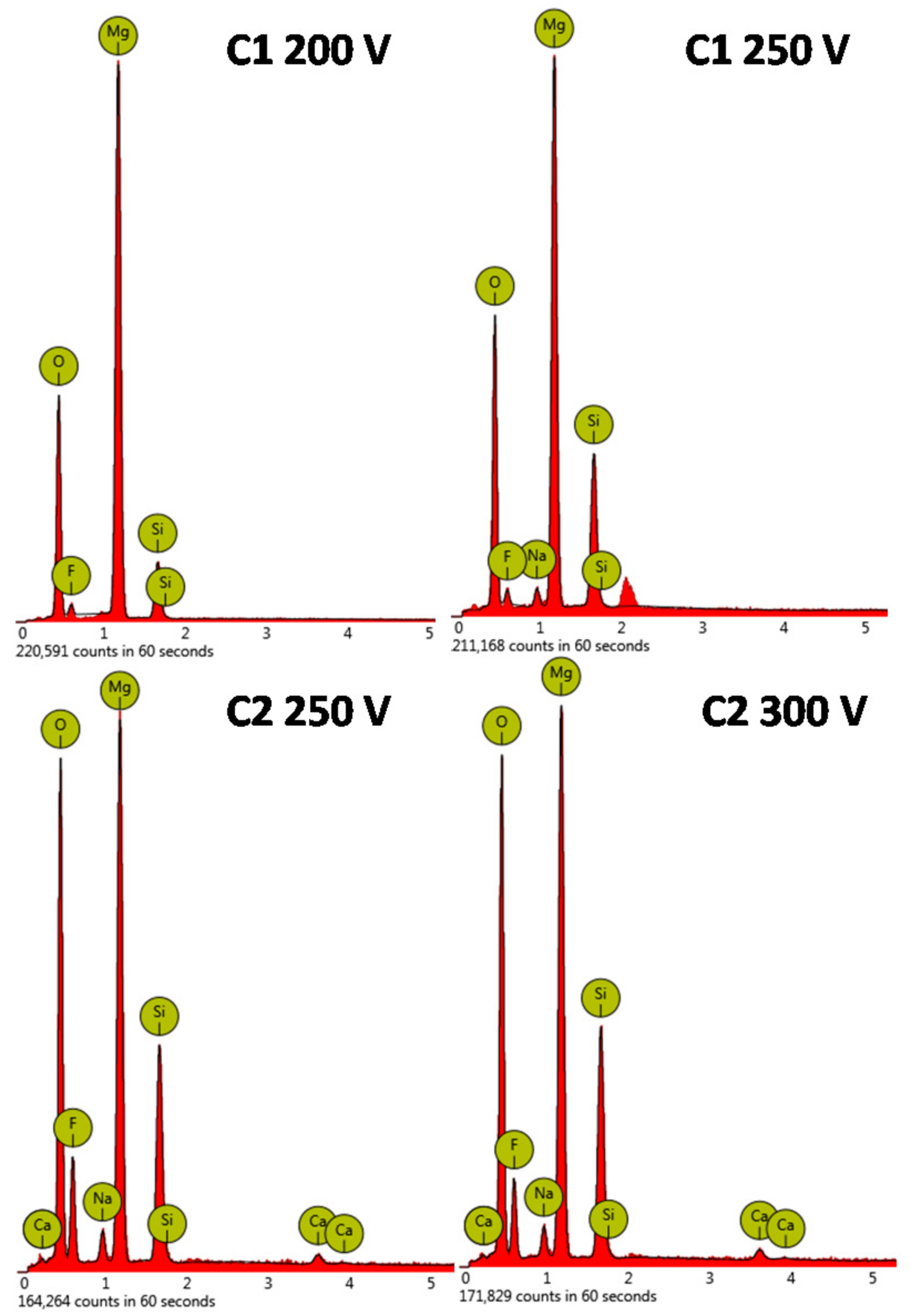

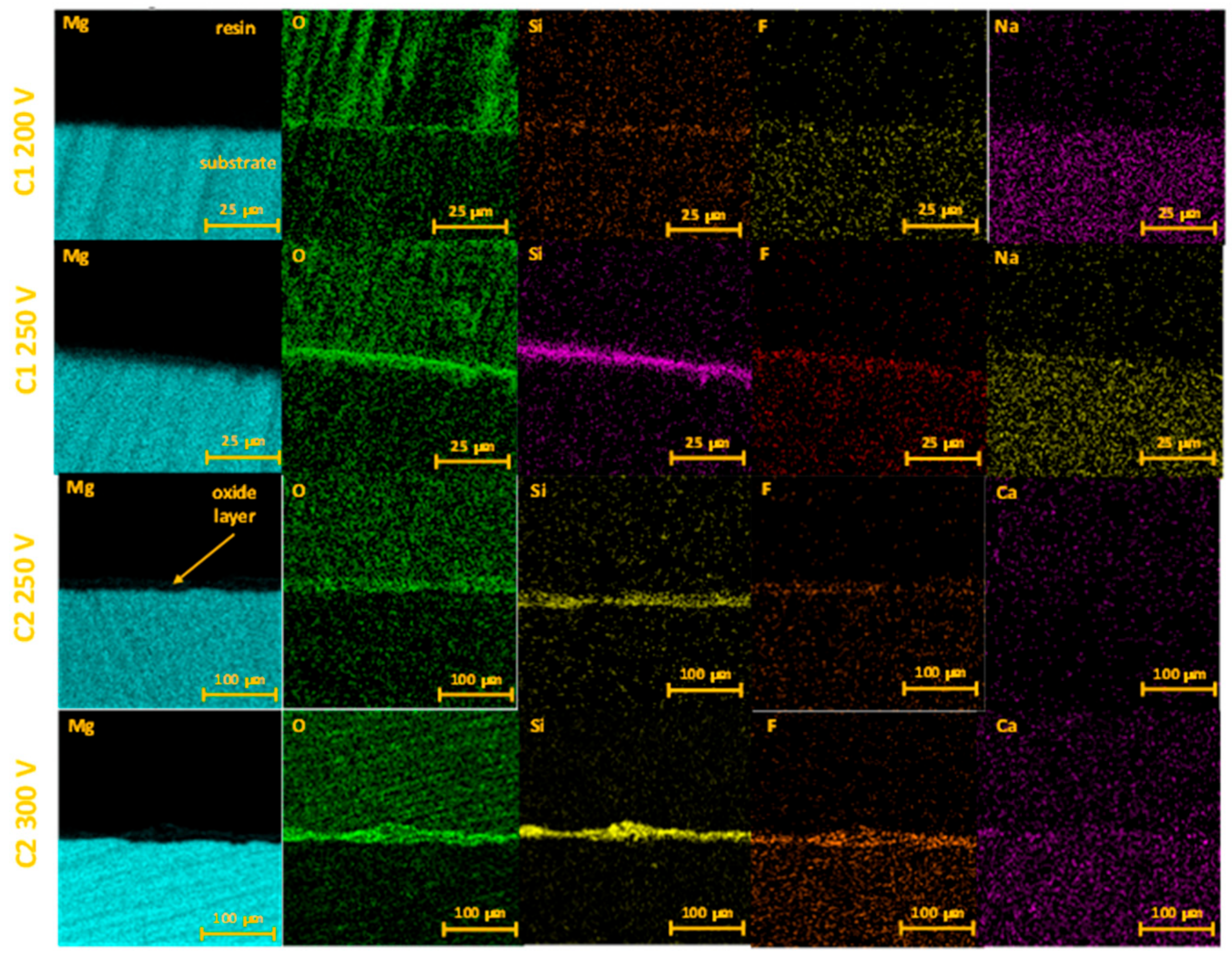
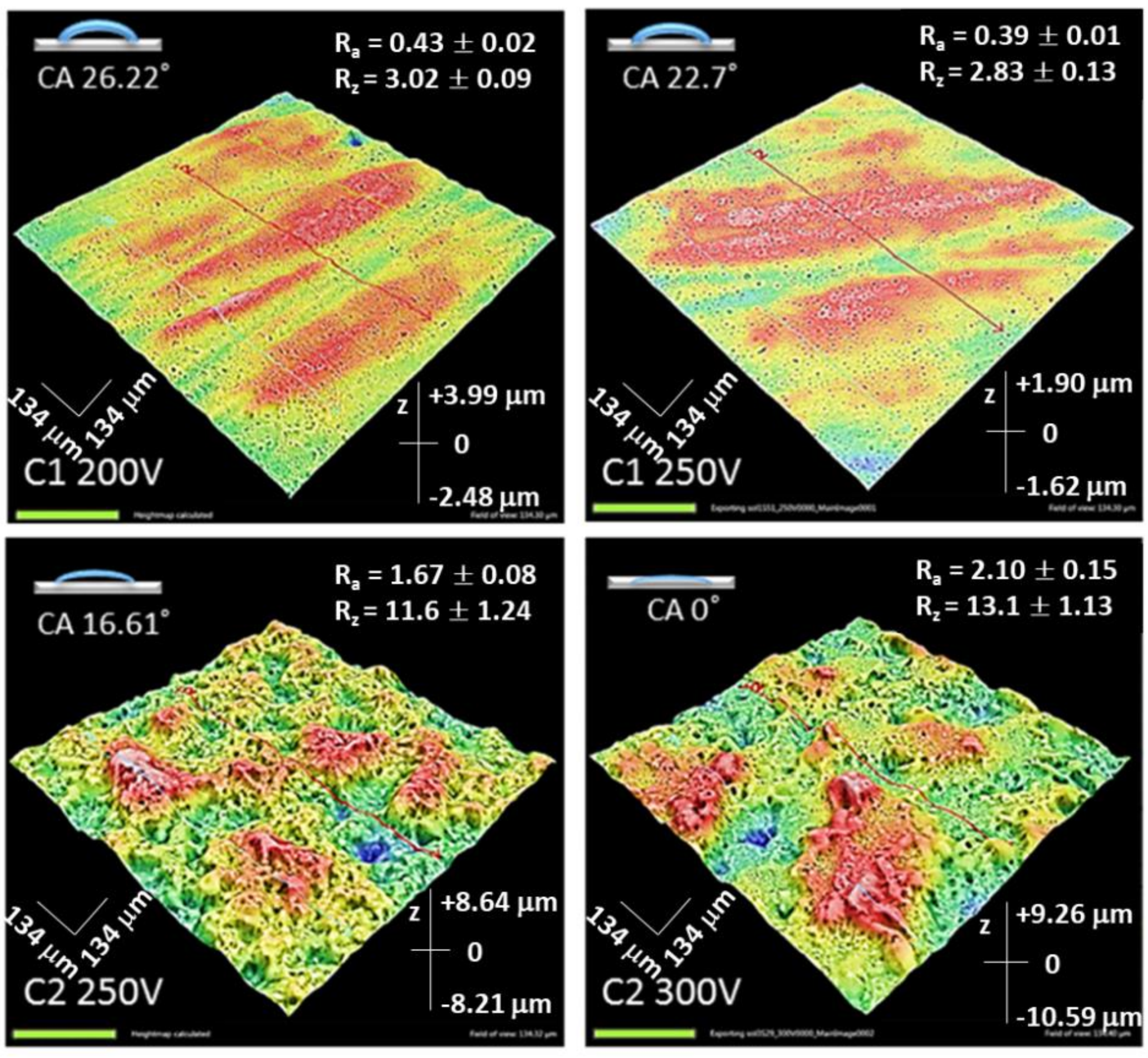
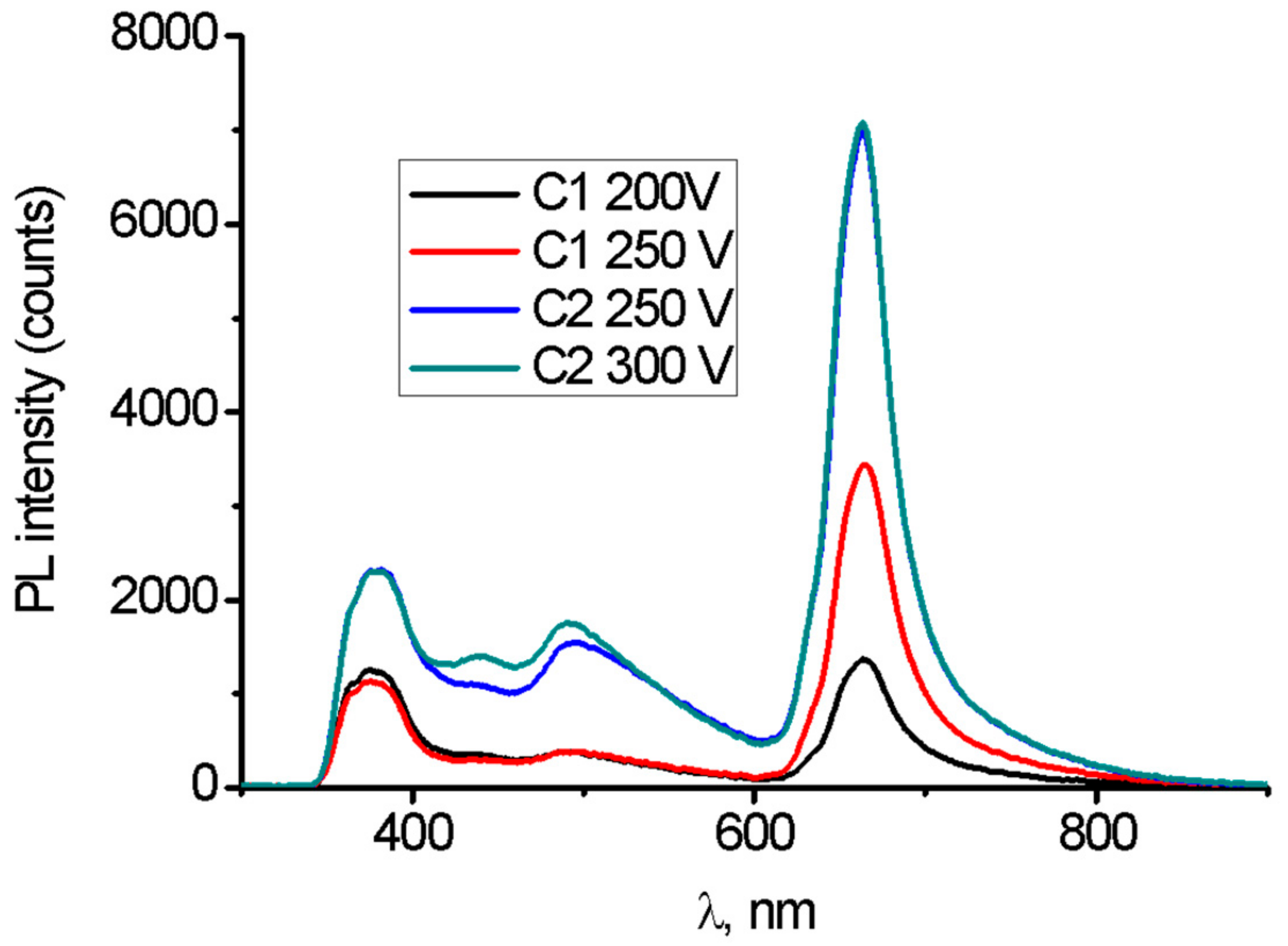


| Sample | Anodizing Voltage, V | Detected Elements | Si/Mg at. Ratio | F/Mg at. Ratio | Ca/Mg at. Ratio |
|---|---|---|---|---|---|
| C1 | 200 | O, Mg, F, Si | 0.26 ± 0,02 | 0.06 ± 0.01 | - |
| 250 | O, Mg, F, Si, Na | 0.52 ± 0.05 | 0.08 ± 0.01 | - | |
| C2 | 250 | O, Mg, F, Si, Na, Ca | 0.54 ± 0.04 | 0.43 ± 0.03 | 0.03 ± 0.01 |
| 300 | O, Mg, F, Si, Na, Ca | 0.60 ± 0.05 | 0.31 ± 0.02 | 0.03 ± 0.01 |
| Electrolyte Label | Concentration of Electrolyte Components; g dm−3 | Anodizing Voltage | |||
|---|---|---|---|---|---|
| Na2SiO3 | NaF | NaOH | Ca(OH)2 | ||
| C1 | 10 | 5 | 10 | 200, 250 | |
| C2 | 10 | 5 | 10 | 250, 300 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Husak, Y.; Michalska, J.; Oleshko, O.; Korniienko, V.; Grundsteins, K.; Dryhval, B.; Altundal, S.; Mishchenko, O.; Viter, R.; Pogorielov, M.; et al. Bioactivity Performance of Pure Mg after Plasma Electrolytic Oxidation in Silicate-Based Solutions. Molecules 2021, 26, 2094. https://doi.org/10.3390/molecules26072094
Husak Y, Michalska J, Oleshko O, Korniienko V, Grundsteins K, Dryhval B, Altundal S, Mishchenko O, Viter R, Pogorielov M, et al. Bioactivity Performance of Pure Mg after Plasma Electrolytic Oxidation in Silicate-Based Solutions. Molecules. 2021; 26(7):2094. https://doi.org/10.3390/molecules26072094
Chicago/Turabian StyleHusak, Yevheniia, Joanna Michalska, Oleksandr Oleshko, Viktoriia Korniienko, Karlis Grundsteins, Bohdan Dryhval, Sahin Altundal, Oleg Mishchenko, Roman Viter, Maksym Pogorielov, and et al. 2021. "Bioactivity Performance of Pure Mg after Plasma Electrolytic Oxidation in Silicate-Based Solutions" Molecules 26, no. 7: 2094. https://doi.org/10.3390/molecules26072094
APA StyleHusak, Y., Michalska, J., Oleshko, O., Korniienko, V., Grundsteins, K., Dryhval, B., Altundal, S., Mishchenko, O., Viter, R., Pogorielov, M., & Simka, W. (2021). Bioactivity Performance of Pure Mg after Plasma Electrolytic Oxidation in Silicate-Based Solutions. Molecules, 26(7), 2094. https://doi.org/10.3390/molecules26072094







