Optimization of Two Eco-Friendly Extractions of Black Medick (Medicago lupulina L.) Phenols and Their Antioxidant, Cosmeceutical, α-Glucosidase and α-Amylase Inhibitory Properties
Abstract
1. Introduction
2. Results and Discussion
2.1. Extraction of Phenolics from M. Lupulina
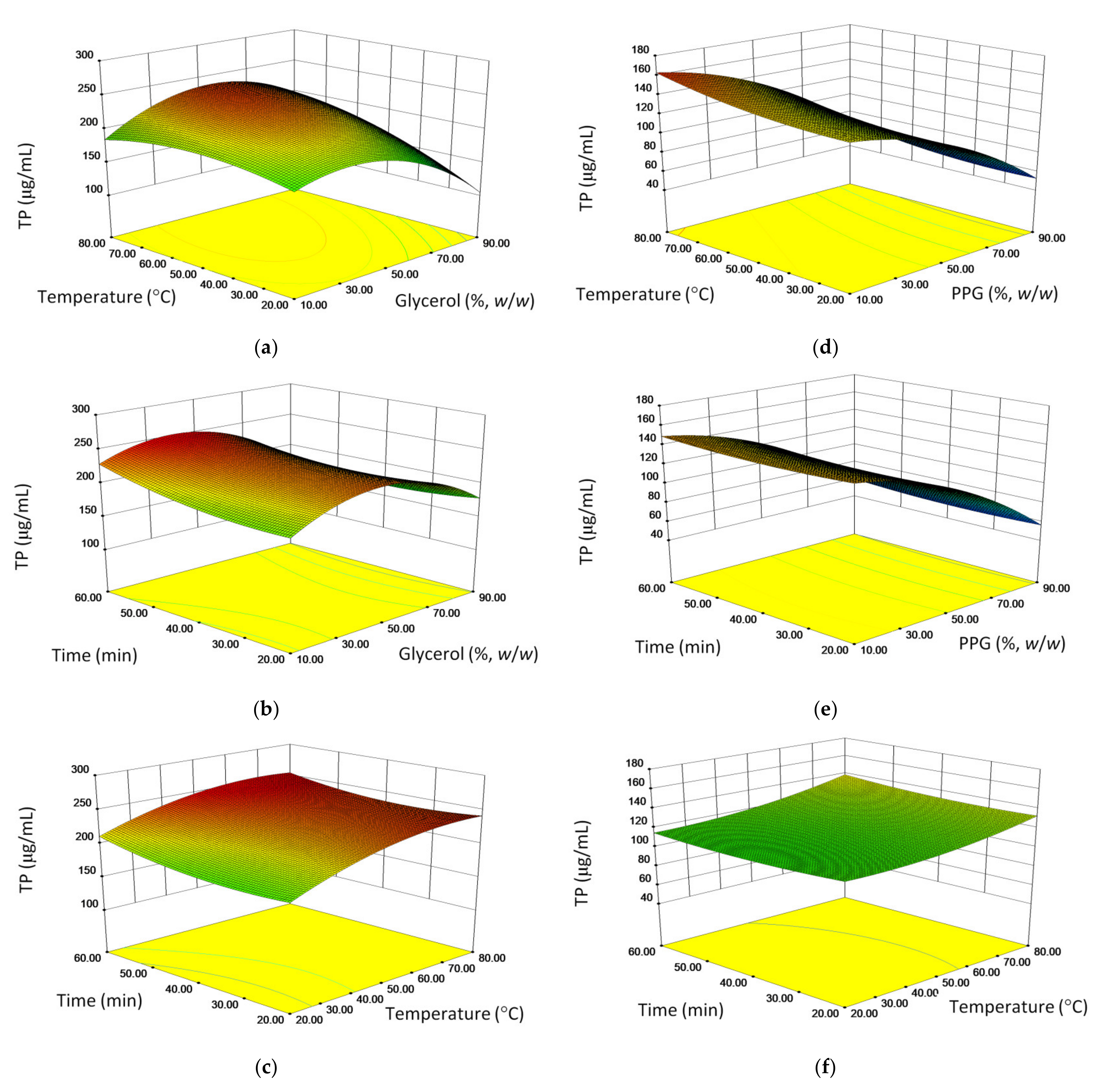
2.2. Optimization of Extraction Parameters and Model Validation
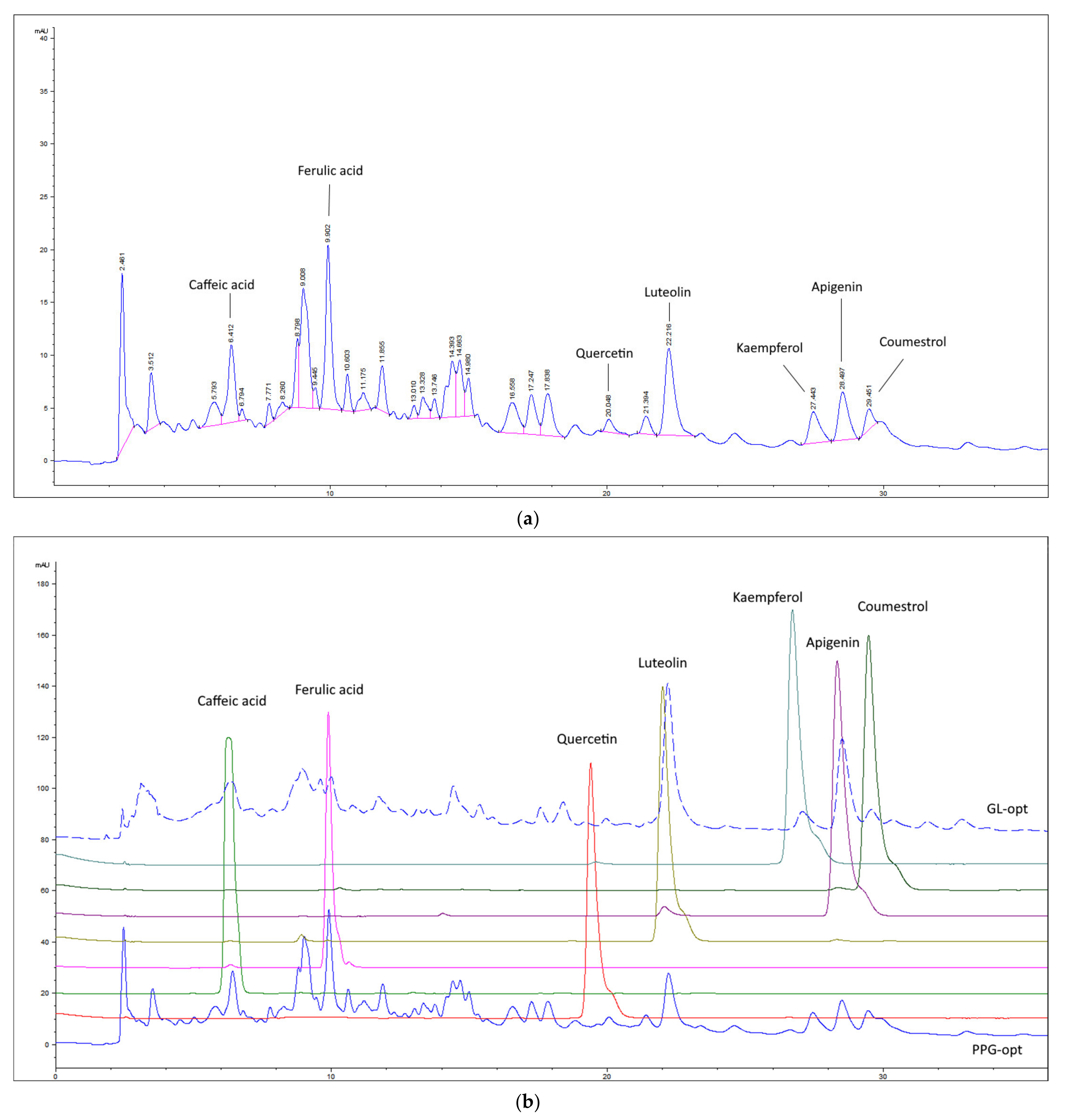
2.3. Chemical Composition of the Extracts
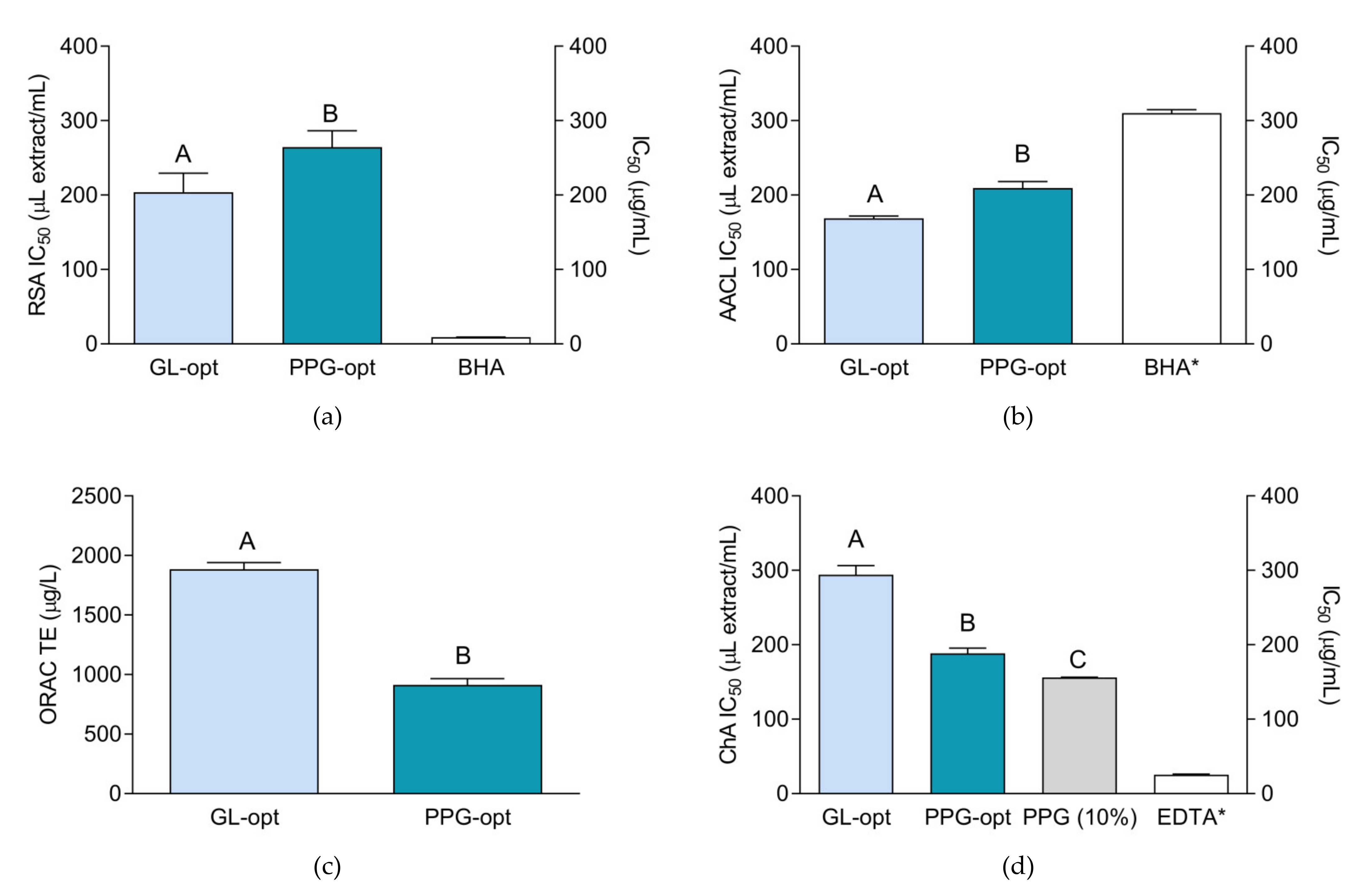
2.4. Antioxidant Activity of the Extracts
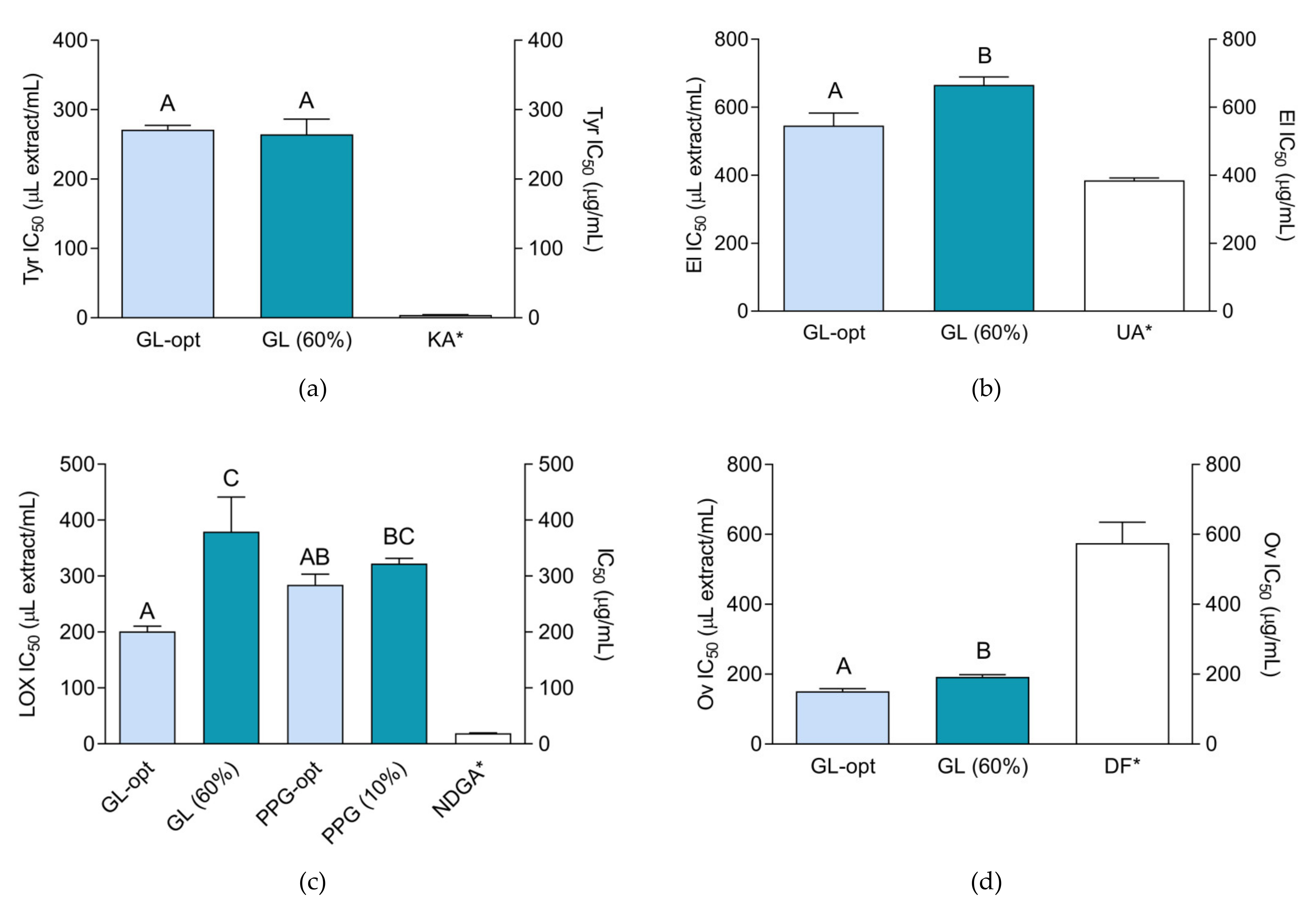
2.5. Tyrosinase-, Elastase-, Lipoxygenase and Coagulation Inhibiting Activity
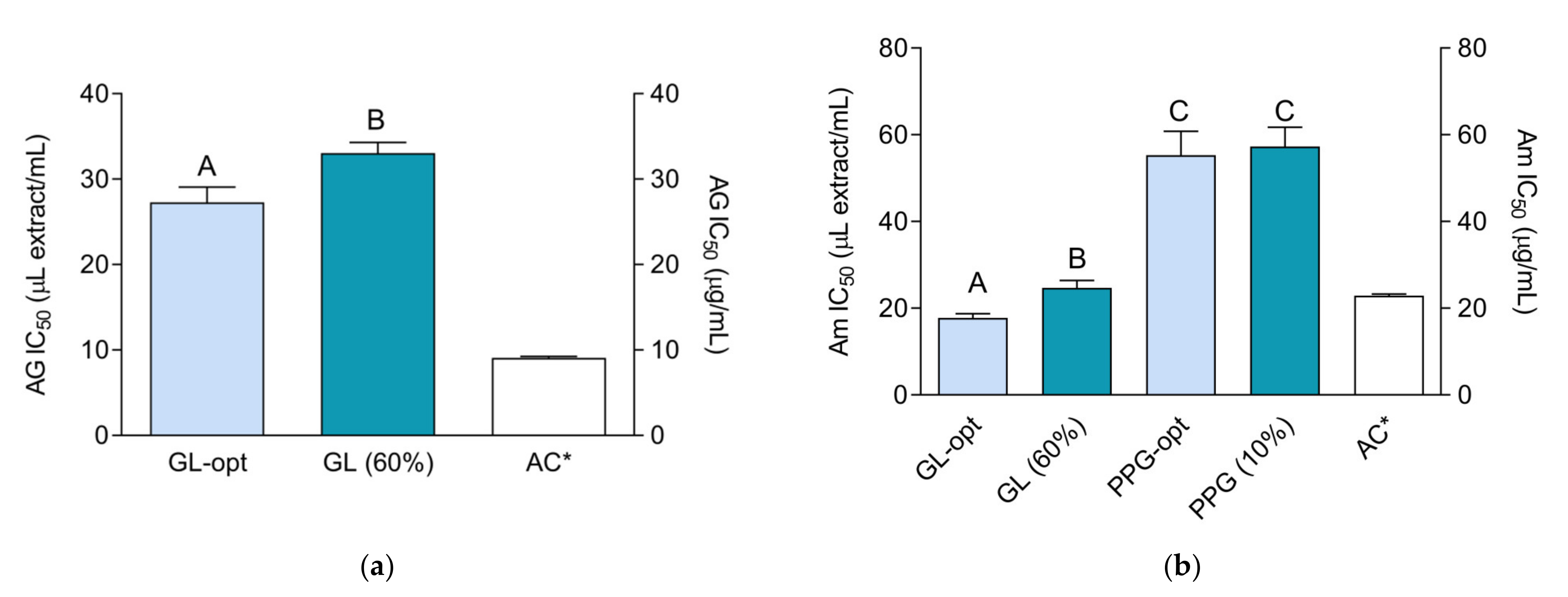
2.6. α-Glucosidase- and α-Amylase-Inhibiting Activity
3. Materials and Methods
3.1. Plant Materials and Chemicals
3.2. Preparation of Extracts
3.3. Experimental Design
3.4. Total Phenol Content
3.5. Total Flavonoid Content
3.6. Total Phenolic Acid Content
3.7. HPLC Analysis of Phenolic Constituents
3.8. Free Radical Scavenging Activity
3.9. Fe2+ Chelating Activity
3.10. Antioxidant Activity in β-Carotene-Linoleic Acid Assay
3.11. ORAC
3.12. Tyrosinase Inhibitory Activity
3.13. Elastase Inhibitory Activity
3.14. Lipoxygenase Inhibitory Activity
3.15. Inhibition of Heat-Induced Protein Coagulation
3.16. α-Glucosidase Inhibition Assay
3.17. α-amylase Inhibition Assay
3.18. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Appendix A
References
- Nicoletti, M. Nutraceuticals and Botanicals: Overview and Perspectives. Int. J. Food Sci. Nutr. 2012, 63, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Xiao, M.; Zhao, J.; Li, Z.; Xing, B.; Li, X.; Kong, M.; Li, L.; Zhang, Q.; Liu, Y.; et al. An Overview of Plant Phenolic Compounds and Their Importance in Human Nutrition and Management of Type 2 Diabetes. Molecules 2016, 21, 1374. [Google Scholar] [CrossRef] [PubMed]
- Zaid, A.N.; Al Ramahi, R. Depigmentation and Anti-Aging Treatment by Natural Molecules. Curr. Pharm. Des. 2019, 25, 2292–2312. [Google Scholar] [CrossRef]
- Azab, A.; Nassar, A.; Azab, A.N. Anti-Inflammatory Activity of Natural Products. Molecules 2016, 21, 1321. [Google Scholar] [CrossRef]
- Panzella, L.; Moccia, F.; Nasti, R.; Marzorati, S.; Verotta, L.; Napolitano, A. Bioactive Phenolic Compounds from Agri-Food Wastes: An Update on Green and Sustainable Extraction Methodologies. Front. Nutr. 2020, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Matos, A.L.; Bruno, D.F.; Ambrósio, A.F.; Santos, P.F. The Benefits of Flavonoids in Diabetic Retinopathy. Nutrients 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Vinayagam, R.; Jayachandran, M.; Xu, B. Antidiabetic Effects of Simple Phenolic Acids: A Comprehensive Review. Phytother. Res. PTR 2016, 30, 184–199. [Google Scholar] [CrossRef]
- De Lima Cherubim, D.J.; Buzanello Martins, C.V.; Oliveira Fariña, L.; da Silva de Lucca, R.A. Polyphenols as Natural Antioxidants in Cosmetics Applications. J. Cosmet. Dermatol. 2019. [Google Scholar] [CrossRef]
- Hwang, J.A.; Park, N.H.; Na, Y.J.; Lee, H.K.; Lee, J.H.; Kim, Y.J.; Lee, C.S. Coumestrol Down-Regulates Melanin Production in Melan-a Murine Melanocytes through Degradation of Tyrosinase. Biol. Pharm. Bull. 2017, 40, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Nagula, R.L.; Wairkar, S. Recent Advances in Topical Delivery of Flavonoids: A Review. J. Control. Release Soc. 2019, 296, 190–201. [Google Scholar] [CrossRef]
- Pandey, A.; Jatana, G.K.; Sonthalia, S. Cosmeceuticals. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Mlakar, A.; Batna, A.; Dudda, A.; Spiteller, G. Iron (II) Ions Induced Oxidation of Ascorbic Acid and Glucose. Free Radic. Res. 1996, 25, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.A.; Katiyar, S.K. Skin Photoprotection by Natural Polyphenols: Anti-Inflammatory, Antioxidant and DNA Repair Mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Jabs, H.-U. Elastase—Ziel Einer Neuen Anti-Aging Strategie Bei Hautalterung, Elastizitätsverlust Und Faltenbildung. Ästhetische Dermatol. 2014, 2012, 2–4. [Google Scholar]
- Mashima, R.; Okuyama, T. The Role of Lipoxygenases in Pathophysiology; New Insights and Future Perspectives. Redox Biol. 2015, 6, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Chatterjee, P.; Dey, P.; Bhattacharya, S. Evaluation of in Vitro Anti-Inflammatory Activity of Coffee against the Denaturation of Protein. Asian Pac. J. Trop. Biomed. 2012, 2, S178–S180. [Google Scholar] [CrossRef]
- Wolfson, A.; Dlugy, C.; Shotland, Y. Glycerol as a Green Solvent for High Product Yields and Selectivities. Environ. Chem. Lett. 2007, 5, 67–71. [Google Scholar] [CrossRef]
- Becker, L.C.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; et al. Safety Assessment of Glycerin as Used in Cosmetics. Int. J. Toxicol. 2019, 38, 6S–22S. [Google Scholar] [CrossRef] [PubMed]
- Ciganović, P.; Jakimiuk, K.; Tomczyk, M.; Zovko Končić, M. Glycerolic Licorice Extracts as Active Cosmeceutical Ingredients: Extraction Optimization, Chemical Characterization, and Biological Activity. Antioxidants 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Momchev, P.; Ciganović, P.; Jug, M.; Marguí, E.; Jablan, J.; Zovko Končić, M. Comparison of Maceration and Ultrasonication for Green Extraction of Phenolic Acids from Echinacea Purpurea Aerial Parts. Molecules 2020, 25, 5142. [Google Scholar] [CrossRef] [PubMed]
- Katsampa, P.; Valsamedou, E.; Grigorakis, S.; Makris, D.P. A Green Ultrasound-Assisted Extraction Process for the Recovery of Antioxidant Polyphenols and Pigments from Onion Solid Wastes Using Box–Behnken Experimental Design and Kinetics. Ind. Crops Prod. 2015, 77, 535–543. [Google Scholar] [CrossRef]
- Santos, J.; Capela, E.; Boal-Palheiros, I.; Coutinho, J.; Freire, M.; Ventura, S. Aqueous Biphasic Systems in the Separation of Food Colorants. Biochem. Mol. Biol. Educ. 2017, 46, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Kawai, F. Microbial Degradation of Polyethers. Appl. Microbiol. Biotechnol. 2002, 58, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Fiume, M.M.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.; Marks, J.G.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; et al. Safety Assessment of Propylene Glycol, Tripropylene Glycol, and PPGs as Used in Cosmetics. Int. J. Toxicol. 2012, 31, 245S–260S. [Google Scholar] [CrossRef]
- Xu, K.; Xu, P.; Wang, Y. Aqueous Biphasic Systems Formed by Hydrophilic and Hydrophobic Deep Eutectic Solvents for the Partitioning of Dyes. Talanta 2020, 213. [Google Scholar] [CrossRef]
- Kicel, A.; Olszewska, M.A. Evaluation of Antioxidant Activity, and Quantitative Estimation of Flavonoids, Saponins and Phenols in Crude Extract and Dry Fractions of Medicago Lupulina Aerial Parts. Nat. Prod. Commun. 2015, 10, 483–486. [Google Scholar] [PubMed]
- Turkington, R.; Cavers, P. The Biology of Canadian Weeds. 33. Medicago Lupulina, L. Can. J. Plant. Sci. 1979, 59, 99–110. [Google Scholar] [CrossRef]
- Somme, R. The Structure of a Galactomannan from Medicago Lupulina, L. Identification of Oligosaccharides from a Partial Acid Hydrolysate. Acta Chem. Scand. 1968, 22, 870–876. [Google Scholar] [CrossRef]
- Butkutė, B.; Padarauskas, A.; Cesevičienė, J.; Pavilonis, A.; Taujenis, L.; Lemežienė, N. Perennial Legumes as a Source of Ingredients for Healthy Food: Proximate, Mineral and Phytoestrogen Composition and Antibacterial Activity. J. Food Sci. Technol. 2017, 54, 2661–2669. [Google Scholar] [CrossRef]
- Górski, P.M.; Jurzysta, M.; Burda, S.; Oleszek, W.A.; Płoszyński, M. Studies on Medicago Lupulina Saponins. 2. Isolation, Chemical Characterization and Biological Activity of Saponins from M. Lupulina Tops. Acta Soc. Bot. Pol. 2014, 53, 527–533. [Google Scholar] [CrossRef][Green Version]
- Baloch, N.; Nabi, S.; Al-Kahraman, Y.M.S.A. In Vitro Antimicrobial, Insecticidal, Antitumor Activities and Their Phytochemical Estimation of Methanolic Extract and Its Fractions of Medicago Lupulina Leaves. World Appl. Sci. J. 2013, 23, 500–506. [Google Scholar]
- Tao, Y.; Zhang, Z.; Sun, D.-W. Kinetic Modeling of Ultrasound-Assisted Extraction of Phenolic Compounds from Grape Marc: Influence of Acoustic Energy Density and Temperature. Ultrason. Sonochem. 2014, 21, 1461–1469. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.B.; Tiwari, B.K.; Gangopadhyay, N.; O’Donnell, C.P.; Brunton, N.P.; Rai, D.K. Ultrasonic Extraction of Steroidal Alkaloids from Potato Peel Waste. Ultrason. Sonochem. 2014, 21, 1470–1476. [Google Scholar] [CrossRef]
- Marszalek, M.; Wolszczak, M. Radiolysis of Berberine or Palmatine in Aqueous Solution. Radiat. Phys. Chem. 2011, 80, 94–99. [Google Scholar] [CrossRef]
- Suslick, K.S.; Price, G.J. Applications of Ultrasound to Materials Chemistry. Annu. Rev. Mater. Sci. 1999, 29, 295–326. [Google Scholar] [CrossRef]
- Laurie, V.F.; Waterhouse, A.L. Oxidation of Glycerol in the Presence of Hydrogen Peroxide and Iron in Model Solutions and Wine. Potential Effects on Wine Color. J. Agric. Food Chem. 2006, 54, 4668–4673. [Google Scholar] [CrossRef] [PubMed]
- Laurie, V.F.; Waterhouse, A.L. Glyceraldehyde Bridging between Flavanols and Malvidin-3-Glucoside in Model Solutions. J. Agric. Food Chem. 2006, 54, 9105–9111. [Google Scholar] [CrossRef] [PubMed]
- Leopoldini, M.; Chiodo, S.G.; Russo, N.; Toscano, M. Detailed Investigation of the OH Radical Quenching by Natural Antioxidant Caffeic Acid Studied by Quantum Mechanical Models. J. Chem. Theory Comput. 2011, 7, 4218–4233. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zheng, S.; Li, L.; Jiang, H. Metabolism of Flavonoids in Human: A Comprehensive Review. Curr. Drug Metab. 2014, 15, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Fumić, B.; Jug, M.; Končić, M.Z. Multi-Response Optimization of Ultrasound-Assisted Extraction of Bioactive Components from Medicago Sativa, L. Croat. Chem. Acta 2017, 90, 481–491. [Google Scholar] [CrossRef]
- Kuiper, G.G.; Carlsson, B.; Grandien, K.; Enmark, E.; Häggblad, J.; Nilsson, S.; Gustafsson, J.A. Comparison of the Ligand Binding Specificity and Transcript Tissue Distribution of Estrogen Receptors Alpha and Beta. Endocrinology 1997, 138, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Pokorny, J.; Yanishlieva, N.; Gordon, M.H. Antioxidants in Food: Practical Applications; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Benaiges, A.; Marcet, P.; Armengol, R.; Betes, C.; Gironés, E. Study of the Refirming Effect of a Plant Complex. Int. J. Cosmet. Sci. 1998, 20, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Seddighfar, M.; Mirghazanfari, S.M.; Dadpay, M. Analgesic and Anti-Inflammatory Properties of Hydroalcoholic Extracts of Malva Sylvestris, Carum Carvi or Medicago Sativa, and Their Combination in a Rat Model. J. Integr. Med. 2020, 18, 181–188. [Google Scholar] [CrossRef]
- Hong, Y.-H.; Chao, W.-W.; Chen, M.-L.; Lin, B.-F. Ethyl Acetate Extracts of Alfalfa (Medicago Sativa L.) Sprouts Inhibit Lipopolysaccharide-Induced Inflammation in Vitro and in Vivo. J. Biomed. Sci. 2009, 16, 64. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-H.; Lee, J.Y.; Lee, J.E.; Jung, Y.W.; Jeong, W.; Hong, S.S.; Cho, Y.-R.; Choi, C.W. Skin-Related Properties and Constituents from the Aerial Parts Extract of Persicaria Senticosa. Oxid. Med. Cell. Longev. 2020, 2020, 6627752. [Google Scholar] [CrossRef] [PubMed]
- Krenn, L.; Wollenweber, E.; Steyrleuthner, K.; Görick, C.; Melzig, M.F. Contribution of Methylated Exudate Flavonoids to the Anti-Inflammatory Activity of Grindelia Robusta. Fitoterapia 2009, 80, 267–269. [Google Scholar] [CrossRef] [PubMed]
- Searle, T.; Al-Niaimi, F.; Ali, F.R. The Top 10 Cosmeceuticals for Facial Hyperpigmentation. Dermatol. Ther. 2020, 33, e14095. [Google Scholar] [CrossRef] [PubMed]
- Yuk, H.J.; Lee, J.H.; Curtis-Long, M.J.; Lee, J.W.; Kim, Y.S.; Ryu, H.W.; Park, C.G.; Jeong, T.-S.; Park, K.H. The Most Abundant Polyphenol of Soy Leaves, Coumestrol, Displays Potent α-Glucosidase Inhibitory Activity. Food Chem. 2011, 126, 1057–1063. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, G.; Pan, J.; Wang, Y. α-Glucosidase Inhibition by Luteolin: Kinetics, Interaction and Molecular Docking. Int. J. Biol. Macromol. 2014, 64, 213–223. [Google Scholar] [CrossRef]
- Bora, K.S.; Sharma, A. Phytochemical and Pharmacological Potential of Medicago Sativa: A Review. Pharm. Biol. 2011, 49, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Oboh, G.; Agunloye, O.M.; Adefegha, S.A.; Akinyemi, A.J.; Ademiluyi, A.O. Caffeic and Chlorogenic Acids Inhibit Key Enzymes Linked to Type 2 Diabetes (in Vitro): A Comparative Study. J. Basic Clin. Physiol. Pharmacol. 2015, 26. [Google Scholar] [CrossRef] [PubMed]
- Adisakwattana, S.; Ruengsamran, T.; Kampa, P.; Sompong, W. In Vitro Inhibitory Effects of Plant-Based Foods and Their Combinations on Intestinal α-Glucosidase and Pancreatic α-Amylase. BMC Complement. Altern. Med. 2012, 12, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1999; Volume 299, pp. 152–178. ISBN 978-0-12-182200-2. [Google Scholar]
- Kumazawa, S.; Hamasaka, T.; Nakayama, T. Antioxidant Activity of Propolis of Various Geographic Origins. Food Chem. 2004, 84, 329–339. [Google Scholar] [CrossRef]
- Nicolle, C.; Carnat, A.; Fraisse, D.; Lamaison, J.-L.; Rock, E.; Michel, H.; Amouroux, P.; Rémésy, C. Characterisation and Variation of Antioxidant Micronutrients in Lettuce (Lactuca Sativa Folium). J. Sci. Food Agric. 2004, 84, 2061–2069. [Google Scholar] [CrossRef]
- Bljajić, K.; Petlevski, R.; Vujić, L.; Čačić, A.; Šoštarić, N.; Jablan, J.; Saraiva de Carvalho, I.; Zovko Končić, M. Chemical Composition, Antioxidant and α-Glucosidase-Inhibiting Activities of the Aqueous and Hydroethanolic Extracts of Vaccinium Myrtillus Leaves. Molecules 2017, 22, 703. [Google Scholar] [CrossRef] [PubMed]
- Rajić, Z.; Končić, M.; Miloloža, K.; Perković, I.; Butula, I.; Bucar, F.; Zorc, B. Primaquine-NSAID Twin Drugs: Synthesis, Radical Scavenging, Antioxidant and Fe2+ Chelating Activity. Acta Pharm. 2010, 60. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gillespie, K.M.; Chae, J.M.; Ainsworth, E.A. Rapid Measurement of Total Antioxidant Capacity in Plants. Nat. Protoc. 2007, 2, 867–870. [Google Scholar] [CrossRef]
- Masuda, T.; Yamashita, D.; Takeda, Y.; Yonemori, S. Screening for Tyrosinase Inhibitors among Extracts of Seashore Plants and Identification of Potent Inhibitors from Garcinia Subelliptica. Biosci. Biotechnol. Biochem. 2005, 69, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Bose, B.; Choudhury, H.; Tandon, P.; Kumaria, S. Studies on Secondary Metabolite Profiling, Anti-Inflammatory Potential, in Vitro Photoprotective and Skin-Aging Related Enzyme Inhibitory Activities of Malaxis Acuminata, a Threatened Orchid of Nutraceutical Importance. J. Photochem. Photobiol. B 2017, 173, 686–695. [Google Scholar] [CrossRef]
- Chekir, S.; Debbabi, M.; Regazzetti, A.; Dargère, D.; Laprévote, O.; Ben Jannet, H.; Gharbi, R. Design, Synthesis and Biological Evaluation of Novel 1,2,3-Triazole Linked Coumarinopyrazole Conjugates as Potent Anticholinesterase, Anti-5-Lipoxygenase, Anti-Tyrosinase and Anti-Cancer Agents. Bioorganic Chem. 2018, 80, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Apostolidis, E.; Kwon, Y.-I.; Shetty, K. Inhibitory Potential of Herb, Fruit, and Fungal-Enriched Cheese against Key Enzymes Linked to Type 2 Diabetes and Hypertension. Innov. Food Sci. Emerg. Technol. 2007, 8, 46–54. [Google Scholar] [CrossRef]
| Independent Variables | Code | Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| Glycerol/PPG Concentration (%, w/w) | X1 | 10 | 50 | 90 |
| Temperature (°C) | X2 | 20 | 50 | 80 |
| Time (min) | X3 | 20 | 40 | 60 |
| Standard Order | Run in GL | Run in PPG | X1 (%. w/w) | X2 (°C) | X3 (min) | TP in GL | TP in PPG |
|---|---|---|---|---|---|---|---|
| μg/mL | μL/mL | ||||||
| 1 | 8 | 5 | 10 | 20 | 40 | 184.52 | 140.09 |
| 2 | 4 | 13 | 90 | 20 | 40 | 98.86 | 48.07 |
| 3 | 16 | 14 | 10 | 80 | 40 | 190.61 | 167.71 |
| 4 | 11 | 1 | 90 | 80 | 40 | 197.03 | 68.09 |
| 5 | 12 | 10 | 10 | 50 | 20 | 184.52 | 146.72 |
| 6 | 17 | 16 | 90 | 50 | 20 | 177.69 | 62.59 |
| 7 | 5 | 6 | 10 | 50 | 60 | 230.16 | 143.21 |
| 8 | 3 | 8 | 90 | 50 | 60 | 188.23 | 60.61 |
| 9 | 7 | 12 | 50 | 20 | 20 | 194.49 | 113.99 |
| 10 | 10 | 4 | 50 | 80 | 20 | 244.87 | 128.40 |
| 11 | 1 | 2 | 50 | 20 | 60 | 208.56 | 119.95 |
| 12 | 6 | 3 | 50 | 80 | 60 | 247.77 | 140.20 |
| 13 | 14 | 17 | 50 | 50 | 40 | 216.15 | 127.72 |
| 14 | 2 | 9 | 50 | 50 | 40 | 248.12 | 106.67 |
| 15 | 15 | 7 | 50 | 50 | 40 | 234.51 | 127.66 |
| 16 | 13 | 15 | 50 | 50 | 40 | 220.61 | 124.60 |
| 17 | 9 | 11 | 50 | 50 | 40 | 228.19 | 96.12 |
| Response | Unit | The Equation Coefficients: a×X12 + b × X22 + c × X32 + d × X1 × X2 + e × X1 × X3 + f × X2 × X3 + g × X1 + h × X2 + i × X3 + j | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | i | j | ||
| TP in GL | (μg/mL) | −45.3 * | −16.5 * | 10.9 | 23.0* | −8.8 | −2.8 | −16.0 * | 24.2* | 9.1 | 229.5 |
| TP in PPG | (mL/mL) | −16.5 * | 5.9 | 3.2 | −1.9 | 0.4 | 1.5 | −44.8 * | 10.3 * | 1.5 | 116.6 |
| TP-GL r2 = 0.9545; r2adj = 0.8959; r2pr = 0.7008 | TP-PPG r2 = 0.9483; r2adj = 0.8817; r2pr = 0.7942 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Source | SS | df | MS | F Value | p-Value | SS | df | MS | F Value | p-Value |
| Model | 20240 | 9 | 2248 | 16.3 | 0.0007 | 18218 | 9 | 2024 | 14.25 | 0.0010 |
| Lack of Fit | 335.0 | 3 | 111.7 | 0.708 | 0.5955 | 166.3 | 3 | 55.41 | 0.2677 | 0.8462 |
| Pure Error | 630.8 | 4 | 157.7 | 828.0 | 4 | 207.0 | ||||
| Extract Name | Optimized Response | Aim of the Optimization | X1 °C | X2 % | X3 W | Predicted | Observed | RD (%) |
|---|---|---|---|---|---|---|---|---|
| GL-opt | TP (μg/mL) | maximized | 45 | 70 | 60 | 255.9 | 256.0± 10.5A | +1.62 |
| PPG-opt | TP (μg/mL) | minimized | 10 | 80 | 60 | 168.4 | 162.5± 1.8B | −3.50 |
| Extract | TF (μg/mL) | TPA (μg/mL) | Api (μg/mL) | Kae (μg/mL) | Lut (μg/mL) | Que (μg/mL) | CA (μg/mL) | FA (μg/mL) | Cou (μg/mL) |
|---|---|---|---|---|---|---|---|---|---|
| GL-opt | 94.62 ± 5.5A | 41.01 ± 3.03A | 5.07 | 5.58 | 13.39 | 5.50 | 3.88 | 4.22 | 3.42 |
| PPG-opt | 77.96 ± 4.28B | 40.25 ± 1.77A | 1.44 | 5.34 | 4.80 | 5.45 | 2.42 | 6.98 | 1.52 |
| Standard | Equation | r2 | LOD (μg/mL) | LOQ (μg/mL) |
|---|---|---|---|---|
| Apigenin | y = 3755.16 x − 27.75 | 0.9998 | 0.028 | 0.085 |
| Kaempferol | y = 2802.76 x − 21.62 | 0.9998 | 0.025 | 0.078 |
| Luteolin | y = 2787.17 x + 46.60 | 0.9998 | 0.025 | 0.076 |
| Quercetin | y = 2200.20 x − 36.75 | 0.9998 | 0.027 | 0.083 |
| Caffeic acid | y = 5335.00 x − 19.45 | 0.9999 | 0.012 | 0.041 |
| Ferulic acid | y = 5045.04 x − 45.10 | 0.9998 | 0.026 | 0.088 |
| Coumestrol | y = 2759.54 x − 44.60 | 0.9997 | 0.076 | 0.2298 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakupović, L.; Kalvarešin, M.; Bukovina, K.; Poljak, V.; Vujić, L.; Zovko Končić, M. Optimization of Two Eco-Friendly Extractions of Black Medick (Medicago lupulina L.) Phenols and Their Antioxidant, Cosmeceutical, α-Glucosidase and α-Amylase Inhibitory Properties. Molecules 2021, 26, 1610. https://doi.org/10.3390/molecules26061610
Jakupović L, Kalvarešin M, Bukovina K, Poljak V, Vujić L, Zovko Končić M. Optimization of Two Eco-Friendly Extractions of Black Medick (Medicago lupulina L.) Phenols and Their Antioxidant, Cosmeceutical, α-Glucosidase and α-Amylase Inhibitory Properties. Molecules. 2021; 26(6):1610. https://doi.org/10.3390/molecules26061610
Chicago/Turabian StyleJakupović, Lejsa, Marko Kalvarešin, Karla Bukovina, Valentina Poljak, Lovorka Vujić, and Marijana Zovko Končić. 2021. "Optimization of Two Eco-Friendly Extractions of Black Medick (Medicago lupulina L.) Phenols and Their Antioxidant, Cosmeceutical, α-Glucosidase and α-Amylase Inhibitory Properties" Molecules 26, no. 6: 1610. https://doi.org/10.3390/molecules26061610
APA StyleJakupović, L., Kalvarešin, M., Bukovina, K., Poljak, V., Vujić, L., & Zovko Končić, M. (2021). Optimization of Two Eco-Friendly Extractions of Black Medick (Medicago lupulina L.) Phenols and Their Antioxidant, Cosmeceutical, α-Glucosidase and α-Amylase Inhibitory Properties. Molecules, 26(6), 1610. https://doi.org/10.3390/molecules26061610








