A Multi-Objective Approach for Drug Repurposing in Preeclampsia
Abstract
1. Introduction
- Target based approach. The disease target is known and this information is used for drug repurposing. These methods usually use a variety of data sources, i.e., protein-protein interaction and drug-target interaction networks.
- Drugs-Drugs similarity based approach: The similarity between drugs is explored (through a wide variety of information like gene expression profile, possible targets, etc.) to identify new candidates.
2. Results and Discussion
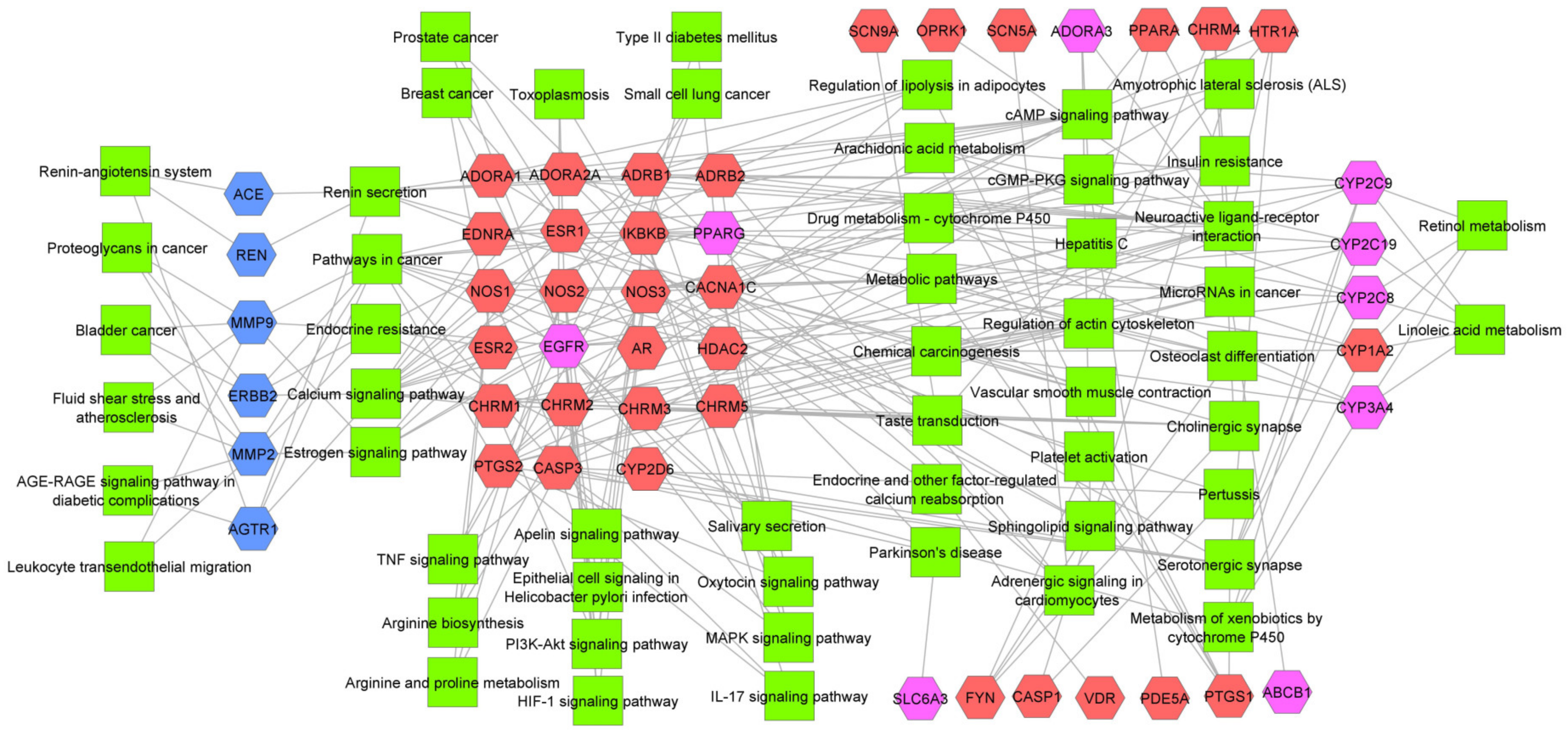
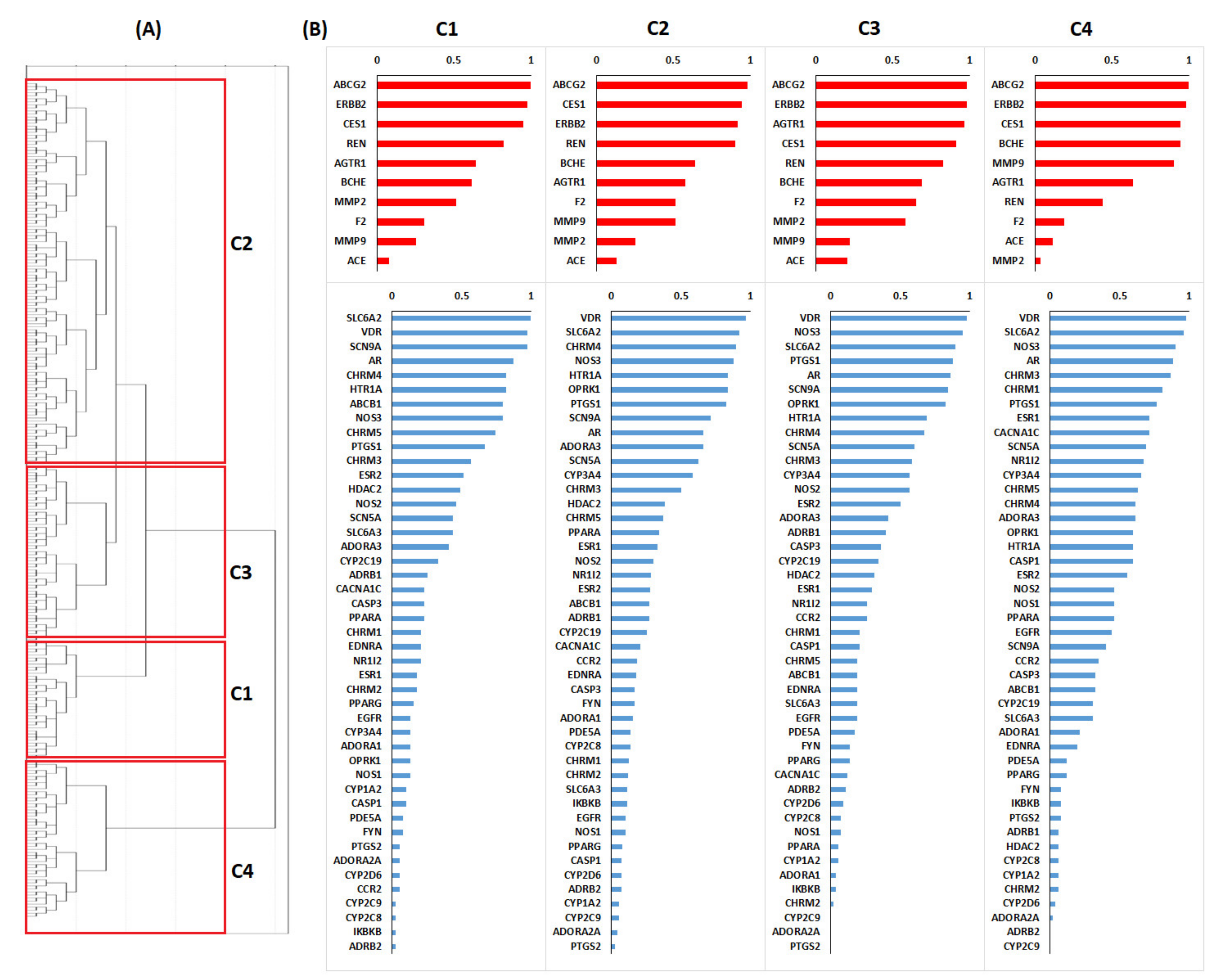
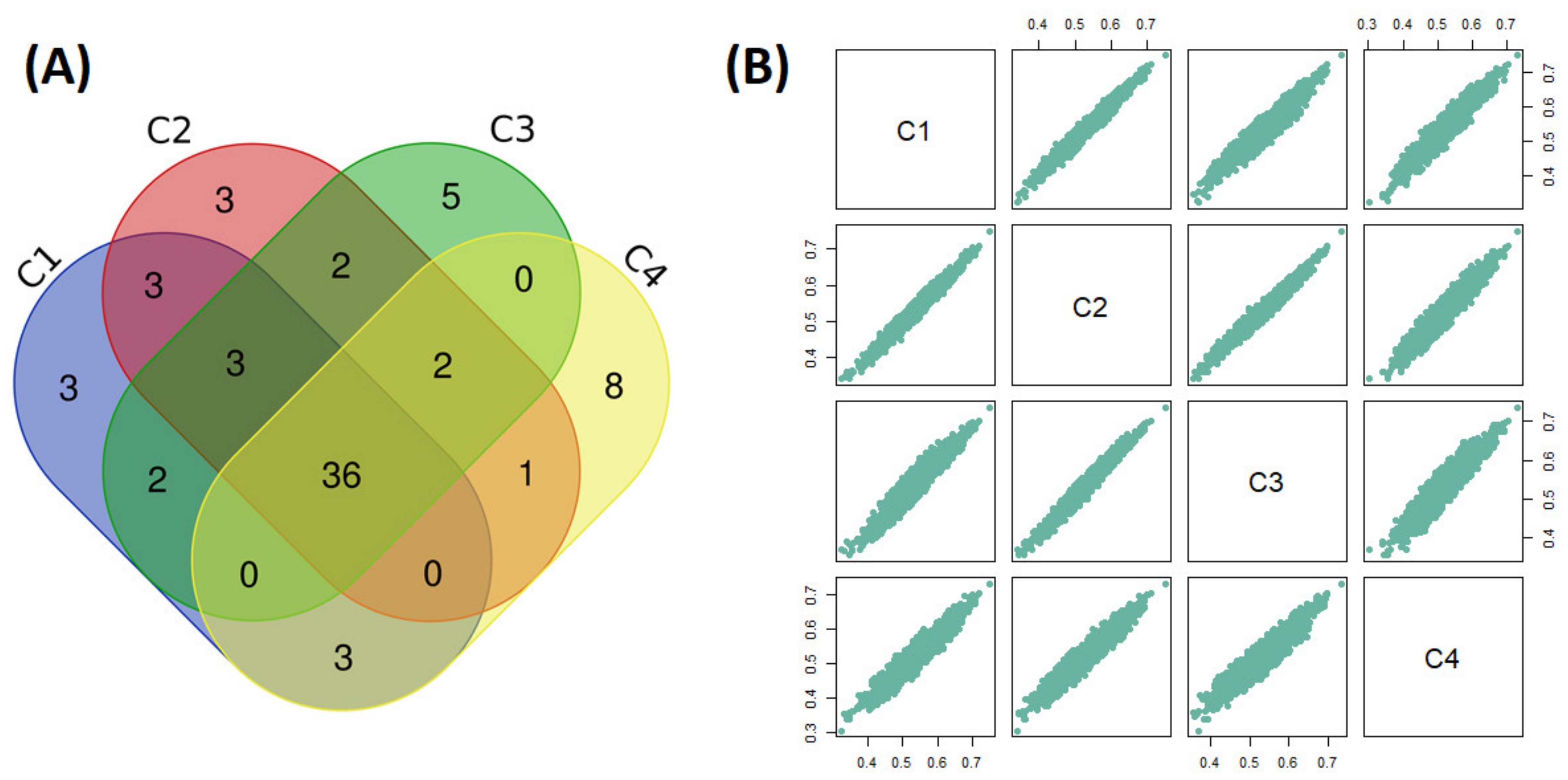
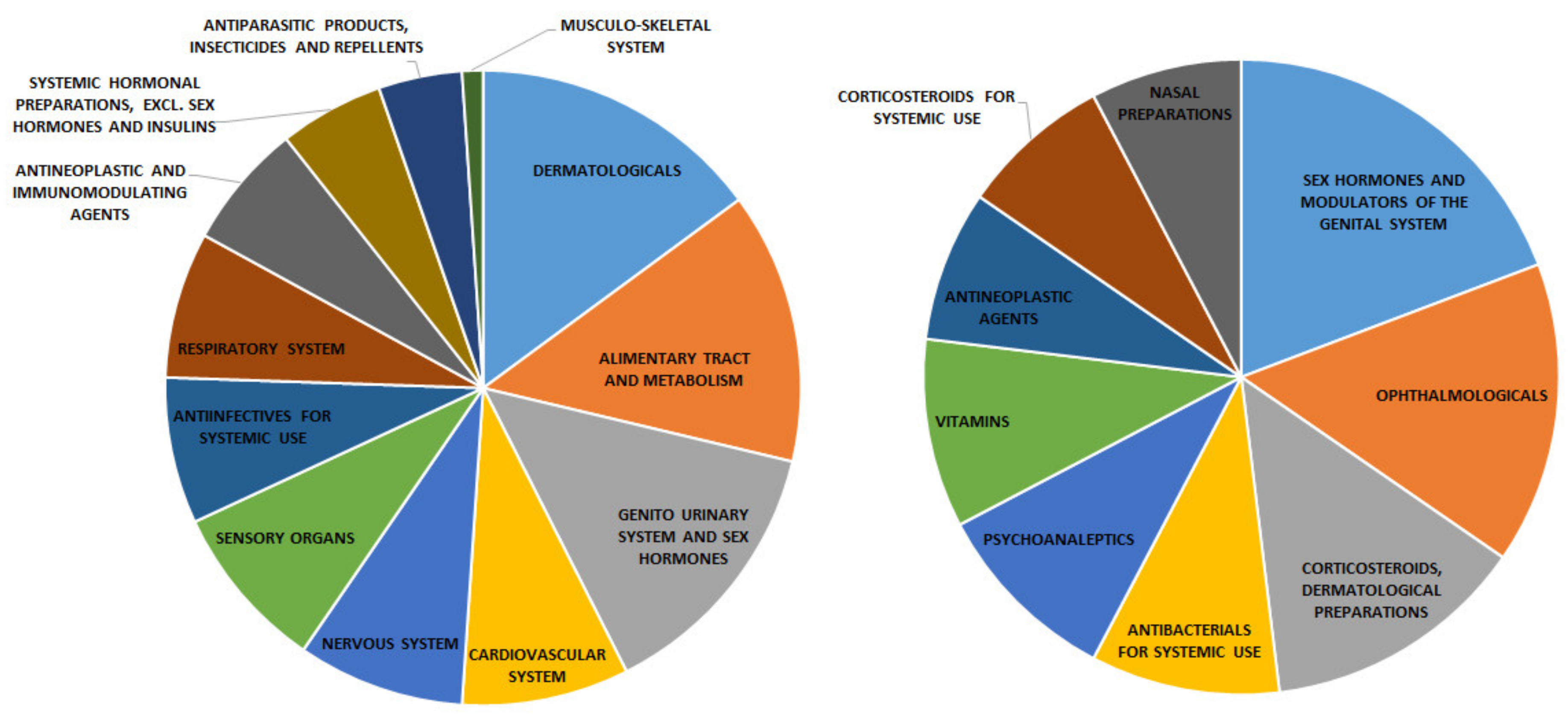
2.1. Targets Analysis
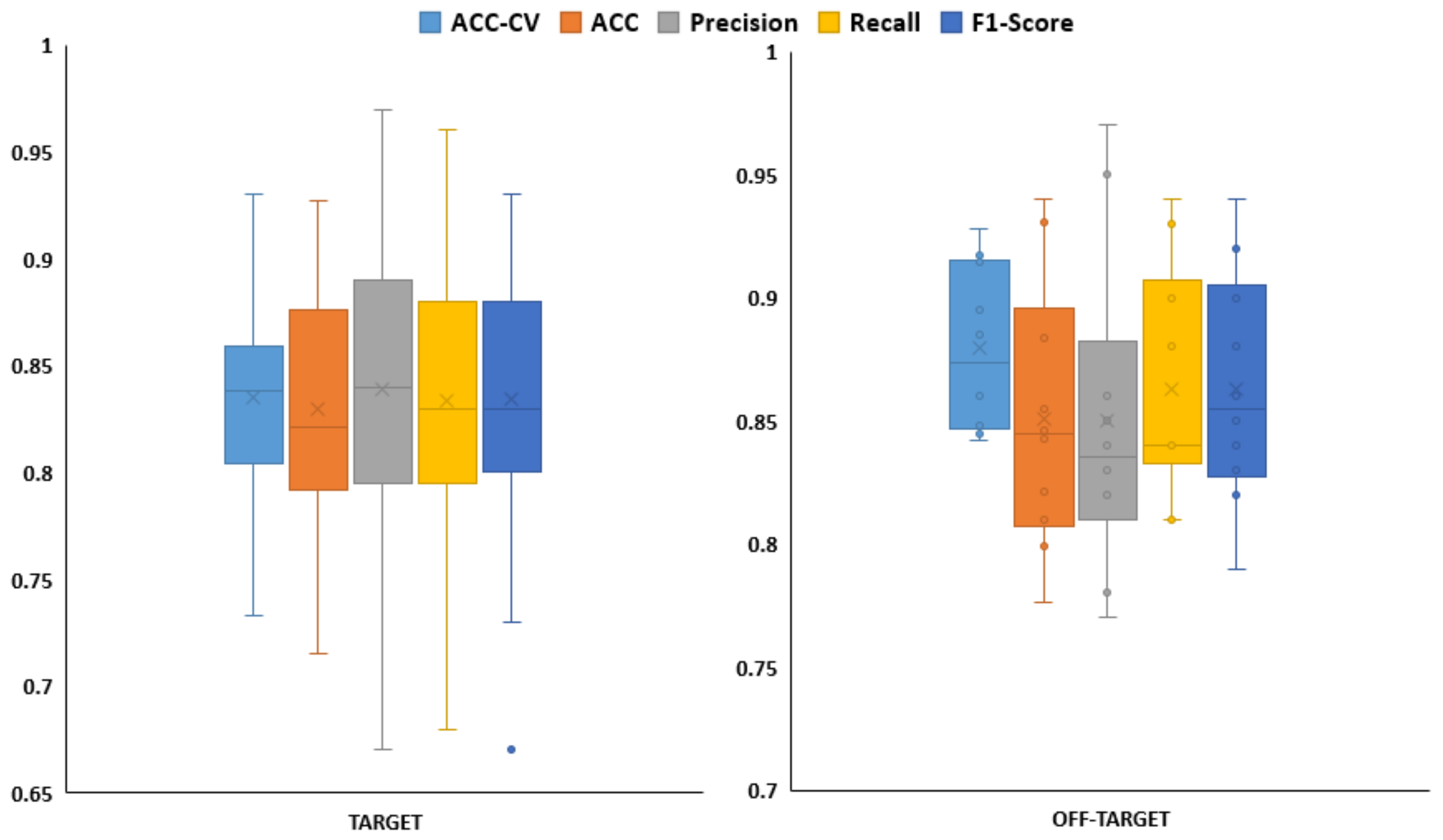
2.2. Models and Early Recognition Ability
2.3. Drug Repurposing
3. Materials and Methods
3.1. Candidate Drugs for Targtes and Off-Targtes Definition
- (1)
- All reported interactions with (IC50, Ki, EC50, GI50) were extracted from the ChEMBL database version 27.
- (2)
- All extracted interactions were labeled as active if interaction values are lower than 10 μM and as inactive otherwise.
- (3)
- If more than one report (active or inactive) was available for the same compound-target interaction, then the final criterion (active or inactive) was assigned considering 75% agreement to occur among reports. That is, if at least 75% of the reports did not agree on the same criterion, then the relevant compound was discarded.
3.2. Data Curation and Model Construction
3.3. Virtual Screening Dataset and Procedures
- and , being:
- NP and NO are the number of targets for preeclampsia and off-targets, respectively.
- and are the average probabilities of compound “i” that are active against the targets of preeclampsia and off-targets, respectively.
- and are the probabilities of the compound “i” that are active against target “j” in the preeclampsia and off-targets target lists, respectively.
3.4. Enrishment Analysis
3.5. Targets Relevance and Global Desirability of Repurposed Drugs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Duhig, K.; Vandermolen, B.; Shennan, A. Recent advances in the diagnosis and management of pre-eclampsia. F1000Research 2018, 7, 242. [Google Scholar] [CrossRef] [PubMed]
- Rolnik, D.L.; Wright, D.; Poon, L.C.; O’Gorman, N.; Syngelaki, A.; de Paco Matallana, C.; Akolekar, R.; Cicero, S.; Janga, D.; Singh, M.; et al. Aspirin versus Placebo in Pregnancies at High Risk for Preterm Preeclampsia. N. Engl. J. Med. 2017, 377, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Vigil-De Gracia, P.; Ludmir, J. The use of magnesium sulfate for women with severe preeclampsia or eclampsia diagnosed during the postpartum period. J. Matern. Neonatal Med. 2015, 28, 2207–2209. [Google Scholar] [CrossRef] [PubMed]
- Mirzakhani, H.; Litonjua, A.A.; McElrath, T.F.; O’Connor, G.; Lee-Parritz, A.; Iverson, R.; Macones, G.; Strunk, R.C.; Bacharier, L.B.; Zeiger, R.; et al. Early pregnancy vitamin D status and risk of preeclampsia. J. Clin. Investig. 2016, 126, 4702–4715. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Díaz, N.C.; Gamboa-Delgado, E.M.; Domínguez-Urrego, C.L.; Vesga-Varela, A.L.; Serrano-Gómez, S.E.; Quintero-Lesmes, D.C. Vitamin D and risk of preeclampsia: A systematic review and meta-analysis. Biomedica 2018, 38, 43–53. [Google Scholar] [CrossRef]
- Stocks, G. Preeclampsia. Eur. J. Anaesthesiol. 2014, 31, 183–189. [Google Scholar] [CrossRef][Green Version]
- Witcher, P.M. Preeclampsia: Acute Complications and Management Priorities. AACN Adv. Crit. Care 2018, 29, 316–326. [Google Scholar] [CrossRef]
- Scaffidi, J.; Mol, B.; Keelan, J. The pregnant women as a drug orphan: A global survey of registered clinical trials of pharmacological interventions in pregnancy. BJOG An Int. J. Obstet. Gynaecol. 2017, 124, 132–140. [Google Scholar] [CrossRef]
- Goldstein, J.A.; Bastarache, L.A.; Denny, J.C.; Pulley, J.M.; Aronoff, D.M. PregOMICS-Leveraging systems biology and bioinformatics for drug repurposing in maternal-child health. Am. J. Reprod. Immunol. 2018, 80, e12971. [Google Scholar] [CrossRef]
- Lotfi Shahreza, M.; Ghadiri, N.; Mousavi, S.R.; Varshosaz, J.; Green, J.R. A review of network-based approaches to drug repositioning. Brief. Bioinform. 2018, 19, 878–892. [Google Scholar] [CrossRef]
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; Doig, A.; Guilliams, T.; Latimer, J.; McNamee, C.; et al. Drug repurposing: Progress, challenges and recommendations. Nat. Rev. Drug Discov. 2018, 18, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Lv, N.; Li, Y.; Liu, T.; Sun, Y.; Chu, X. Identification and characterization of methylation-mediated transcriptional dysregulation dictate methylation roles in preeclampsia. Hum. Genomics 2020, 14, 5. [Google Scholar] [CrossRef] [PubMed]
- Tejera, E.; Bernardes, J.; Rebelo, I. Preeclampsia: A bioinformatics approach through protein-protein interaction networks analysis. BMC Syst. Biol. 2012, 6, 97. [Google Scholar] [CrossRef] [PubMed]
- Tejera, E.; Cruz-Monteagudo, M.; Burgos, G.; Sánchez, M.-E.; Sánchez-Rodríguez, A.; Pérez-Castillo, Y.; Borges, F.; Cordeiro, M.N.D.S.; Paz-y-Miño, C.; Rebelo, I. Consensus strategy in genes prioritization and combined bioinformatics analysis for preeclampsia pathogenesis. BMC Med. Genomics 2017, 10, 50. [Google Scholar] [CrossRef] [PubMed]
- Vaiman, D.; Calicchio, R.; Miralles, F. Landscape of transcriptional deregulations in the preeclamptic placenta. PLoS ONE 2013, 8, e65498. [Google Scholar] [CrossRef]
- Leavey, K.; Bainbridge, S.A.; Cox, B.J. Large scale aggregate microarray analysis reveals three distinct molecular subclasses of human preeclampsia. PLoS ONE 2015, 10, e0116508. [Google Scholar] [CrossRef]
- Song, Y.; Liu, J.; Huang, S.; Zhang, L. Analysis of differentially expressed genes in placental tissues of preeclampsia patients using microarray combined with the Connectivity Map database. Placenta 2013, 34, 1190–1195. [Google Scholar] [CrossRef]
- Kakigano, A.; Tomimatsu, T.; Mimura, K.; Kanayama, T.; Fujita, S.; Minato, K.; Kumasawa, K.; Taniguchi, Y.; Kanagawa, T.; Endo, M.; et al. Drug Repositioning for Preeclampsia Therapeutics by In Vitro Screening: Phosphodiesterase-5 Inhibitor Vardenafil Restores Endothelial Dysfunction via Induction of Placental Growth Factor. Reprod. Sci. 2015, 22, 1272–1280. [Google Scholar] [CrossRef]
- Sun, M.; Fan, Y.; Hou, Y.; Fan, Y. Preeclampsia and maternal risk of breast cancer: A meta-analysis of cohort studies. J. Matern. Neonatal Med. 2018, 31, 2484–2491. [Google Scholar] [CrossRef]
- Shibuya, M. Involvement of Flt-1 (VEGF receptor-1) in cancer and preeclampsia. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2011, 87, 167–178. [Google Scholar] [CrossRef]
- Todros, T.; Verdiglione, P.; Ogge, G.; Paladini, D.; Vergani, P.; Cardaropoli, S. Low incidence of hypertensive disorders of pregnancy in women treated with spiramycin for toxoplasma infection. Br. J. Clin. Pharmacol. 2006, 61, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Alshareef, S.A.; Nasr, A.M.; Adam, I. Toxoplasma gondii infection and pre-eclampsia among Sudanese women. Trans. R. Soc. Trop. Med. Hyg. 2018, 112, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Ambia, A.M.; Seasely, A.R.; Macias, D.A.; Nelson, D.B.; Wells, C.E.; McIntire, D.D.; Cunningham, F.G. The impact of baseline proteinuria in pregnant women with pregestational diabetes mellitus. Am. J. Obstet. Gynecol. MFM 2020, 2, 100072. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, N.; Gonzalez, J.M.; Rosenstein, M.G. Does the 1-step method of gestational diabetes mellitus screening improve pregnancy outcomes? Am. J. Obstet. Gynecol. MFM 2020, 2, 100199. [Google Scholar] [CrossRef] [PubMed]
- Lan, K.-C.; Lai, Y.-J.; Cheng, H.-H.; Tsai, N.-C.; Su, Y.-T.; Tsai, C.-C.; Hsu, T.-Y. Levels of sex steroid hormones and their receptors in women with preeclampsia. Reprod. Biol. Endocrinol. 2020, 18, 12. [Google Scholar] [CrossRef]
- Naghshineh, E.; Sheikhaliyan, S. Effect of vitamin D supplementation in the reduce risk of preeclampsia in nulliparous women. Adv. Biomed. Res. 2016, 5, 7. [Google Scholar] [CrossRef]
- Tarca, A.L.; Romero, R.; Erez, O.; Gudicha, D.W.; Than, N.G.; Benshalom-Tirosh, N.; Pacora, P.; Hsu, C.D.; Chaiworapongsa, T.; Hassan, S.S.; et al. Maternal whole blood mRNA signatures identify women at risk of early preeclampsia: A longitudinal study. J. Matern. Neonatal Med. 2020. [Google Scholar] [CrossRef]
- Soobryan, N.; Murugesan, S.; Pandiyan, A.; Moodley, J.; Mackraj, I. Angiogenic Dysregulation in Pregnancy-Related Hypertension—A Role for Metformin. Reprod. Sci. 2018, 1531–1539. [Google Scholar] [CrossRef]
- Saleh, L.; Verdonk, K.; Visser, W.; van den Meiracker, A.H.; Danser, A.H.J. The emerging role of endothelin-1 in the pathogenesis of pre-eclampsia. Ther. Adv. Cardiovasc. Dis. 2016, 10, 282–293. [Google Scholar] [CrossRef]
- Dong, T.; Sato, S.; Lyu, J.; Imachi, H.; Kobayashi, T.; Fukunaga, K.; Saheki, T.; Iwama, H.; Zhang, G.; Murao, K. Treatment with 2-methoxyestradiol increases endothelial nitric oxide synthase activity via scavenger receptor class BI in human umbilical vein endothelial cells. Mol. Hum. Reprod. 2020, 26, 441–451. [Google Scholar] [CrossRef]
- Cleophas, M.; Joosten, L.; Stamp, L.; Dalbeth, N.; Woodward, O.; Merriman, T. ABCG2 polymorphisms in gout: Insights into disease susceptibility and treatment approaches. Pharmgenomics. Pers. Med. 2017, 10, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Moreno Santillan, A.A.; Briones Garduño, J.C.; Diaz de Leon Ponce, M.A. Uric Acid in Pregnancy: New Concepts. In Contributions to Nephrology; Treviño-Becerra, A., Iseki, K., Eds.; Karger Publishers: Basel, Switzerland, 2018; Volume 192, pp. 110–115. [Google Scholar]
- Lye, P.; Bloise, E.; Nadeem, L.; Peng, C.; Gibb, W.; Ortiga-Carvalho, T.M.; Lye, S.J.; Matthews, S.G. Breast Cancer Resistance Protein (BCRP/ABCG2) Inhibits Extra Villous Trophoblast Migration: The Impact of Bacterial and Viral Infection. Cells 2019, 8, 1150. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.; Geng, M.; Zhang, R.; Liu, W.; Wang, J.; Li, J.; Lin, Y.; Liu, S.; Wang, Z.; Guo, H.; et al. Value of ABCG2 Q141K and Q126X genotyping in predicting risk of preeclampsia in Chinese Han women population. Pregnancy Hypertens. 2019, 17, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Valente, F.M.; De Andrade, D.O.; Cosenso-Martin, L.N.; Cesarino, C.B.; Guimarães, S.M.; Guimarães, V.B.; Lacchini, R.; Tanus-Santos, J.E.; Yugar-Toledo, J.C.; Vilela-Martin, J.F. Plasma levels of matrix metalloproteinase-9 are elevated in individuals with hypertensive crisis. BMC Cardiovasc. Disord. 2020, 20, 132. [Google Scholar] [CrossRef] [PubMed]
- Onal, I.K.; Altun, B.; Onal, E.D.; Kirkpantur, A.; Gul Oz, S.; Turgan, C. Serum levels of MMP-9 and TIMP-1 in primary hypertension and effect of antihypertensive treatment. Eur. J. Intern. Med. 2009, 20, 369–372. [Google Scholar] [CrossRef]
- Hamutoğlu, R.; Bulut, H.E.; Kaloğlu, C.; Önder, O.; Dağdeviren, T.; Aydemir, M.N.; Korkmaz, E.M. The regulation of trophoblast invasion and decidual reaction by matrix metalloproteinase-2, metalloproteinase-7, and metalloproteinase-9 expressions in the rat endometrium. Reprod. Med. Biol. 2020, 19, 385–397. [Google Scholar] [CrossRef]
- Chen, J.; Khalil, R.A. Matrix Metalloproteinases in Normal Pregnancy and Preeclampsia. In Progress in Molecular Biology and Translational Science; Elsevier B.V.: Amsterdam, The Netherlands, 2017; Volume 148, pp. 87–165. [Google Scholar]
- Yu, W.Y.; Hill, S.T.; Chan, E.R.; Pink, J.J.; Cooper, K.; Leachman, S.; Lund, A.W.; Kulkarni, R.; Bordeaux, J.S. Computational drug repositioning identifies statins as a modifier of prognostic genetic expression signatures and metastatic behavior in melanoma. J. Invest. Dermatol. 2021, in press. [Google Scholar] [CrossRef]
- Gimenez, N.; Tripathi, R.; Giró, A.; Rosich, L.; López-Guerra, M.; López-Oreja, I.; Playa-Albinyana, H.; Arenas, F.; Mas, J.M.; Pérez-Galán, P.; et al. Systems biology drug screening identifies statins as enhancers of current therapies in chronic lymphocytic leukemia. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Rahimi, R.; Nikfar, S.; Abdollahi, M. Meta-analysis finds use of inhaled corticosteroids during pregnancy safe: A systematic meta-analysis review. Hum. Exp. Toxicol. 2006, 25, 447–452. [Google Scholar] [CrossRef]
- Vatti, R.R.; Teuber, S.S. Asthma and pregnancy. Clin. Rev. Allergy Immunol. 2012, 43, 45–56. [Google Scholar] [CrossRef]
- Murphy, V.E.; Namazy, J.A.; Powell, H.; Schatz, M.; Chambers, C.; Attia, J.; Gibson, P.G. A meta-analysis of adverse perinatal outcomes in women with asthma. BJOG An Int. J. Obstet. Gynaecol. 2011, 118, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Martel, M.J.; Rey, É.; Beauchesne, M.F.; Perreault, S.; Forget, A.; Maghni, K.; Lefebvre, G.; Blais, L. Use of short-acting β2-agonists during pregnancy and the risk of pregnancy-induced hypertension. J. Allergy Clin. Immunol. 2007, 119, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Katz, O.; Sheiner, E. Asthma and pregnancy: A review of twto decades. Expert Rev. Respir. Med. 2008, 2, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Mao, M.; Liu, S.; Xu, S.; Yang, J. A comparative study of bolus norepinephrine, phenylephrine, and ephedrine for the treatment of maternal hypotension in parturients with preeclampsia during cesarean delivery under spinal anesthesia. Med. Sci. Monit. 2019, 25, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Ngene, N.C.; Moodley, J. Pre-eclampsia with severe features: Management of antihypertensive therapy in the postpartum period. Pan Afr. Med. J. 2020, 36, 1–15. [Google Scholar] [CrossRef]
- De Alwis, N.; Beard, S.; Mangwiro, Y.T.; Binder, N.K.; Kaitu’u-Lino, T.J.; Brownfoot, F.C.; Tong, S.; Hannan, N.J. Pravastatin as the statin of choice for reducing pre-eclampsia-associated endothelial dysfunction. Pregnancy Hypertens. 2020, 20, 83–91. [Google Scholar] [CrossRef]
- Rana, S.; Rajakumar, A.; Geahchan, C.; Salahuddin, S.; Cerdeira, A.S.; Burke, S.D.; George, E.M.; Granger, J.P.; Karumanchi, S.A. Ouabain inhibits placental sFlt1 production by repressing HSP27-dependent HIF-1α pathway. FASEB J. 2014, 28, 4324–4334. [Google Scholar] [CrossRef]
- El-Shourbagy, M.A.A.; El-Refaie, T.A.; Sayed, K.K.A.; Wahba, K.A.H.; El-Din, A.S.S.; Fathy, M.M. Impact of seroconversion and antichlamydial treatment on the rate of pre-eclampsia among Egyptian primigravidae. Int. J. Gynecol. Obstet. 2011, 113, 137–140. [Google Scholar] [CrossRef]
- Nascimento, R.A.; Possomato-Vieira, J.S.; Gonçalves-Rizzi, V.H.; Bonacio, G.F.; Rizzi, E.; Dias-Junior, C.A. Hypertension, augmented activity of matrix metalloproteinases-2 and -9 and angiogenic imbalance in hypertensive pregnancy are attenuated by doxycycline. Eur. J. Pharmacol. 2018, 840, 60–69. [Google Scholar] [CrossRef]
- Rustveld, L.O.; Kelsey, S.F.; Sharma, R. Association Between Maternal Infections and Preeclampsia: A Systematic Review of Epidemiologic Studies. Matern. Child Health J. 2008, 12, 223–242. [Google Scholar] [CrossRef]
- Tooke, L.; Riemer, L.; Matjila, M.; Harrison, M. Antiretrovirals causing severe pre-eclampsia. Pregnancy Hypertens. 2016, 6, 266–268. [Google Scholar] [CrossRef] [PubMed]
- Saums, M.K.; King, C.C.; Adams, J.C.; Sheth, A.N.; Badell, M.L.; Young, M.; Yee, L.M.; Chadwick, E.G.; Jamieson, D.J.; Haddad, L.B. Combination Antiretroviral Therapy and Hypertensive Disorders of Pregnancy. Obstet. Gynecol. 2019, 134, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Hastie, R.; Ye, L.; Hannan, N.J.; Brownfoot, F.C.; Cannon, P.; Nguyen, V.; Tong, S.; Kaitu’u-Lino, T.J. Disulfiram inhibits placental soluble FMS-like tyrosine kinase-1 and soluble endoglin secretion independent of the proteasome. Pregnancy Hypertens. 2018, 14, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.K.; Nevo, O. 2-Methoxyestradiol regulates VEGFR-2 and sFlt-1 expression in human placenta. Placenta 2015, 36, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.; Jaiswar, S.P.; Deo, S.; Shankhwar, P. Association of 2-Methoxyestradiol (2ME) Plasma Levels with Clinical Severity Indices and Biomarkers of Preeclampsia. J. Obstet. Gynecol. India 2019, 69, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Berkane, N.; Liere, P.; Lefevre, G.; Alfaidy, N.; Nahed, R.A.; Vincent, J.; Oudinet, J.-P.; Pianos, A.; Cambourg, A.; Rozenberg, P.; et al. Abnormal steroidogenesis and aromatase activity in preeclampsia. Placenta 2018, 69, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Thadhani, R.; Stampfer, M.J.; Chasan-Taber, L.; Willett, W.C.; Curhan, G.C. A prospective study of pregravid oral contraceptive use and risk of hypertensive disorders of pregnancy. Contraception 1999, 60, 145–150. [Google Scholar] [CrossRef]
- Farley, K.E.; Huber, L.R.B.; Warren-Findlow, J.; Ersek, J.L. The association between contraceptive use at the time of conception and hypertensive disorders during pregnancy: A Retrospective Cohort Study of Prams Participants. Matern. Child Health J. 2014, 18, 1779–1785. [Google Scholar] [CrossRef]
- Magnussen, E.B.; Vatten, L.J.; Lund-Nilsen, T.I.; Salvesen, K.Å.; Smith, G.D.; Romundstad, P.R. Prepregnancy cardiovascular risk factors as predictors of pre-eclampsia: Population based cohort study. Br. Med. J. 2007, 335, 978–981. [Google Scholar] [CrossRef]
- Liao, Q.P.; Buhimschi, I.A.; Saade, G.; Chwalisz, K.; Garfield, R.E. Regulation of vascular adaptation during pregnancy and post-partum: Effects of nitric oxide inhibition and steroid hormones. Hum. Reprod. 1996, 11, 2777–2784. [Google Scholar] [CrossRef][Green Version]
- Lorzadeh, N.; Kazemirad, Y.; Kazemirad, N. Investigating the preventive effect of vitamins C and e on preeclampsia in nulliparous pregnant women. J. Perinat. Med. 2020, 48, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Meng, W.Y.; Huang, W.T.; Zhang, J.; Jiao, M.Y.; Jin, L.; Jin, L. Relationship between serum vitamin E concentration in first trimester and the risk of developing hypertension disorders complicating pregnancy. Beijing Da Xue Xue Bao. 2020, 52, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Kasture, V.; Kale, A.; Randhir, K.; Sundrani, D.; Joshi, S. Effect of maternal omega-3 fatty acids and vitamin E supplementation on placental apoptotic markers in rat model of early and late onset preeclampsia. Life Sci. 2019, 239, 117038. [Google Scholar] [CrossRef] [PubMed]
- Valdimarsdottir, R.; Wikström, A.-K.; Kallak, T.K.; Elenis, E.; Axelsson, O.; Preissl, H.; Ubhayasekera, S.J.K.A.; Bergquist, J.; Poromaa, I.S. Pregnancy outcome in women with polycystic ovary syndrome in relation to second-trimester testosterone levels. Reprod. Biomed. Online 2021, 42, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Keya, S.L.; Khanam, N.N.; Chowdhury, A.A.; Ripon, R.; Tasnim, T.; Sharmin, A. Relationship between Free Testosterone and Preeclampsia. Mymensingh Med. J. 2019, 28, 574–581. [Google Scholar] [PubMed]
- Ibrahim, Z.M.; Kishk, E.A.; Elzamlout, M.S.; Elshahat, A.M.; Taha, O.T. Fetal gender, serum human chorionic gonadotropin, and testosterone in women with preeclampsia. Hypertens. Pregnancy 2020, 39, 302–307. [Google Scholar] [CrossRef]
- Kumar, S.; Gordon, G.H.; Abbott, D.H.; Mishra, J.S. Androgens in maternal vascular and placental function: Implications for preeclampsia pathogenesis. Reproduction 2018, 156, R155–R167. [Google Scholar] [CrossRef]
- Källén, B.A.J. Antiobesity drugs in early pregnancy and congenital malformations in the offspring. Obes. Res. Clin. Pract. 2014, 8, e571–e576. [Google Scholar] [CrossRef]
- Källén, B.; Nilsson, E.; Olausson, P.O. Maternal use of loperamide in early pregnancy and delivery outcome. Acta Paediatr. Int. J. Paediatr. 2008, 97, 541–545. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2018, 46, D1074–D1082. [Google Scholar] [CrossRef]
- Bento, A.P.; Gaulton, A.; Hersey, A.; Bellis, L.J.; Chambers, J.; Davies, M.; Krüger, F.A.; Light, Y.; Mak, L.; McGlinchey, S.; et al. The ChEMBL bioactivity database: An update. Nucleic Acids Res. 2014, 42, D1083–D1090. [Google Scholar] [CrossRef] [PubMed]
- RDKit, Open-Source Cheminformatics. 2018. Available online: http://www.rdkit.org (accessed on 1 November 2020).
- Le, T.T.; Fu, W.; Moore, J.H. Scaling tree-based automated machine learning to biomedical big data with a feature set selector. Bioinformatics 2020, 36, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Truchon, J.-F.; Bayly, C.I. Evaluating virtual screening methods: Good and bad metrics for the “early recognition” problem. J. Chem. Inf. Model. 2007, 47, 488–508. [Google Scholar] [CrossRef]
- Kirchmair, J.; Markt, P.; Distinto, S.; Wolber, G.; Langer, T. Evaluation of the performance of 3D virtual screening protocols: RMSD comparisons, enrichment assessments, and decoy selection—What can we learn from earlier mistakes? J. Comput. Aided. Mol. Des. 2008, 22, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Kaimal, V.; Bardes, E.E.; Tabar, S.C.; Jegga, A.G.; Aronow, B.J. ToppCluster: A multiple gene list feature analyzer for comparative enrichment clustering and network-based dissection of biological systems. Nucleic Acids Res. 2010, 38, W96–W102. [Google Scholar] [CrossRef]
- Kim, S.; Chen, J.; Cheng, T.; Gindulyte, A.; He, J.; He, S.; Li, Q.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B.; et al. PubChem 2019 update: Improved access to chemical data. Nucleic Acids Res. 2019, 47, D1102–D1109. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tejera, E.; Pérez-Castillo, Y.; Chamorro, A.; Cabrera-Andrade, A.; Sanchez, M.E. A Multi-Objective Approach for Drug Repurposing in Preeclampsia. Molecules 2021, 26, 777. https://doi.org/10.3390/molecules26040777
Tejera E, Pérez-Castillo Y, Chamorro A, Cabrera-Andrade A, Sanchez ME. A Multi-Objective Approach for Drug Repurposing in Preeclampsia. Molecules. 2021; 26(4):777. https://doi.org/10.3390/molecules26040777
Chicago/Turabian StyleTejera, Eduardo, Yunierkis Pérez-Castillo, Andrea Chamorro, Alejandro Cabrera-Andrade, and Maria Eugenia Sanchez. 2021. "A Multi-Objective Approach for Drug Repurposing in Preeclampsia" Molecules 26, no. 4: 777. https://doi.org/10.3390/molecules26040777
APA StyleTejera, E., Pérez-Castillo, Y., Chamorro, A., Cabrera-Andrade, A., & Sanchez, M. E. (2021). A Multi-Objective Approach for Drug Repurposing in Preeclampsia. Molecules, 26(4), 777. https://doi.org/10.3390/molecules26040777






