Influence of the Alcoholic/Ethanolic Extract of Mangifera indica Residues on the Green Synthesis of FeO Nanoparticles and Their Application for the Remediation of Agricultural Soils
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Treatment of M. indica Husk
2.3. Obtaining Alcoholic Extract 96%/Absolute Ethanolic
2.4. Green Synthesis of Iron Oxide Nanoparticles with Alcoholic 96% and Absolute Ethanolic Solvent Extracts
2.5. Characterization of Iron Oxide Nanoparticles (FeO NP)
2.6. Removal of Cr-VI, Cd and Pb Present in Agricultural Soil
3. Results and Discussion
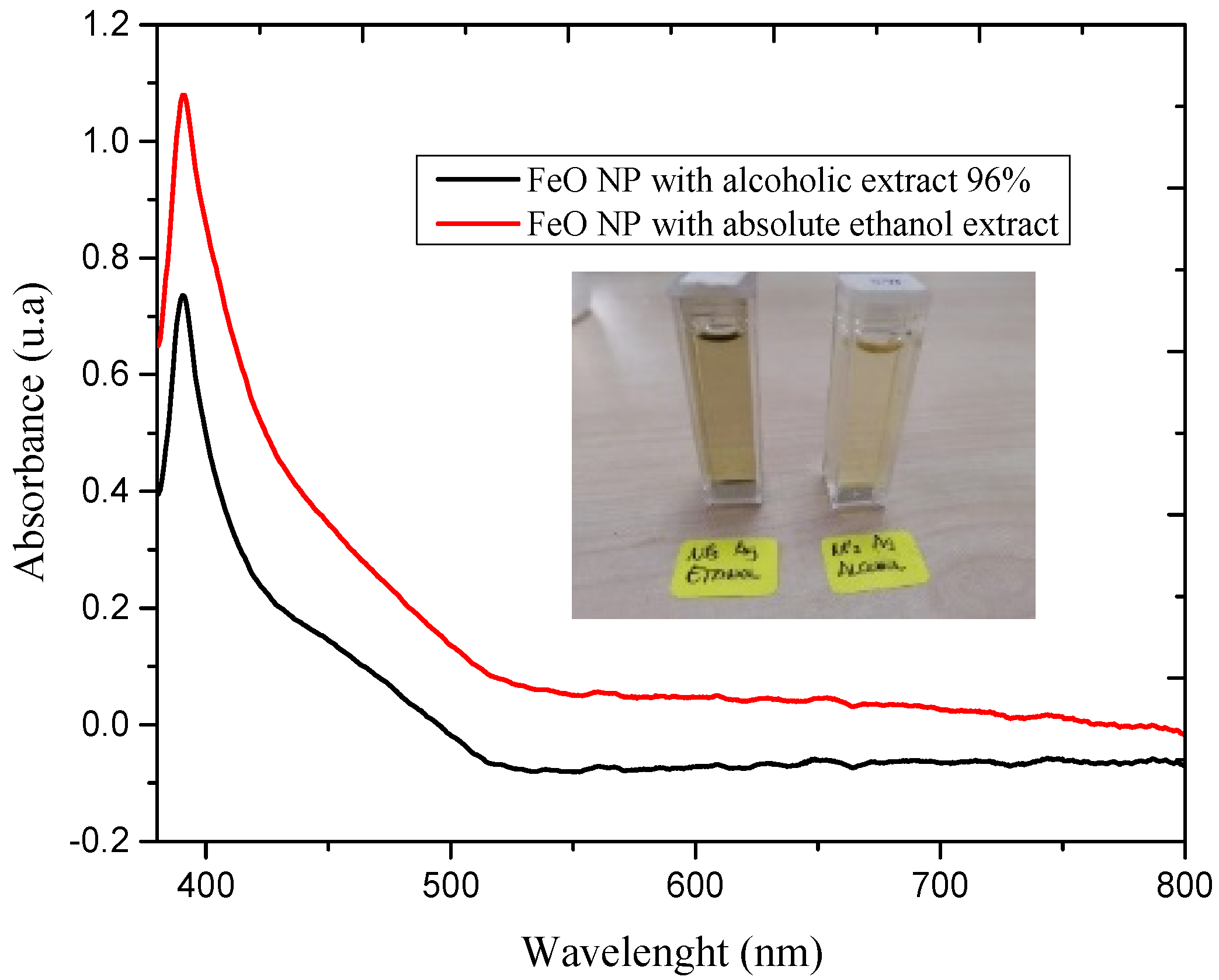
3.1. UV-Vis Spectrum Analysis
3.2. Fourier Transform Infrared Spectroscopy—FTIR
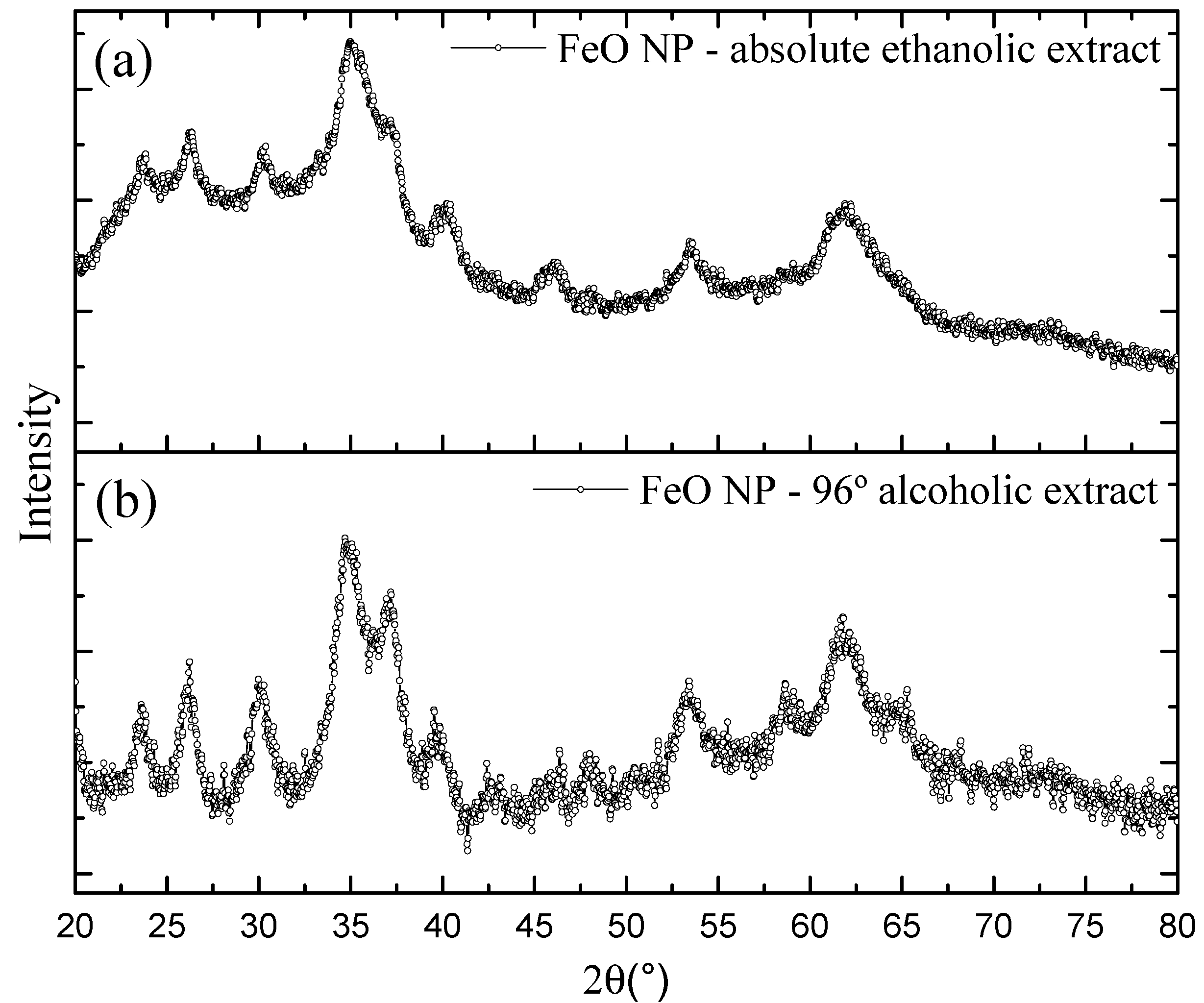
3.3. X-ray Diffraction—XRD
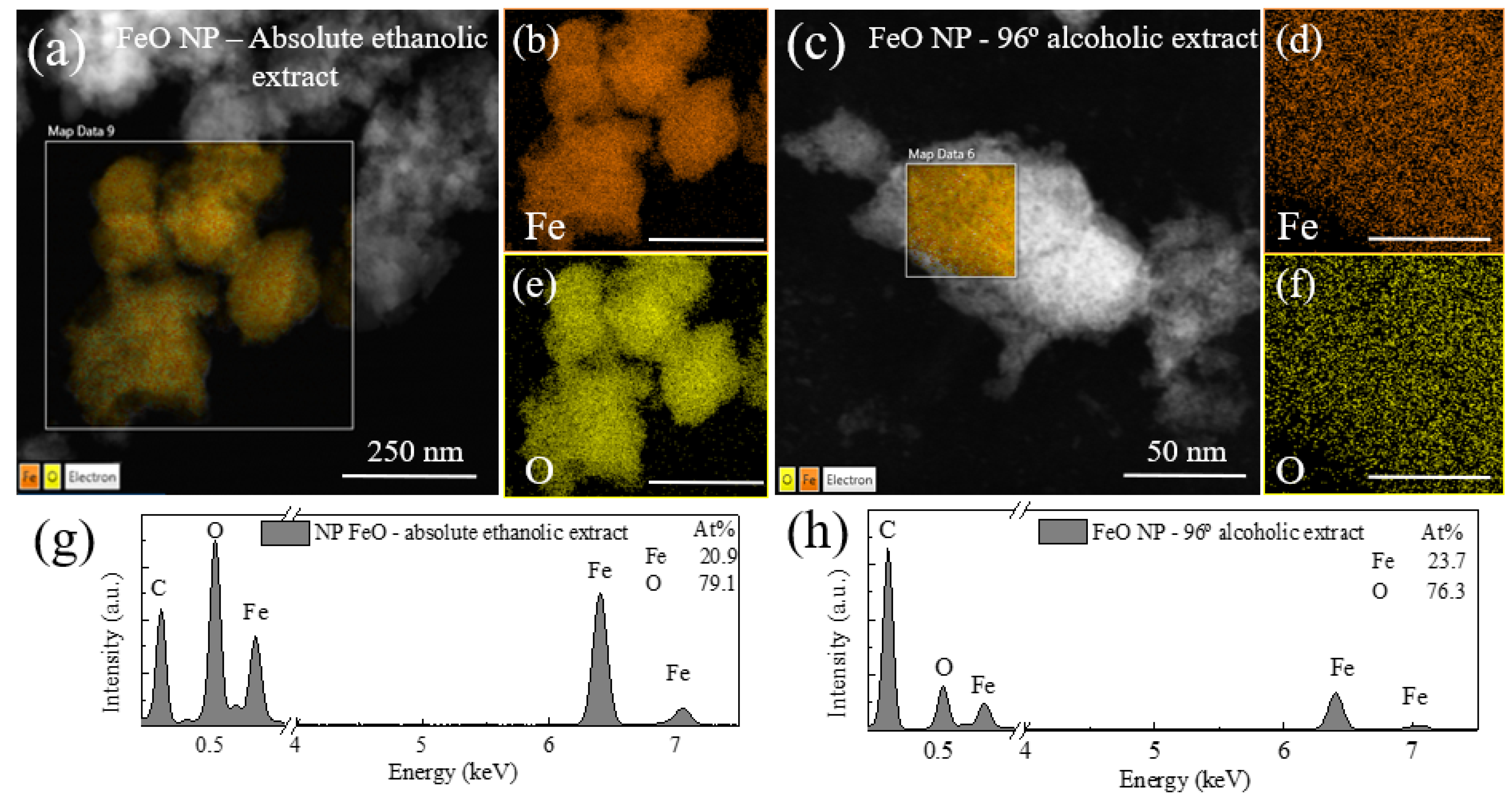
3.4. Elemental Composition
3.5. Transmission Electron Microscopy TEM/STEM
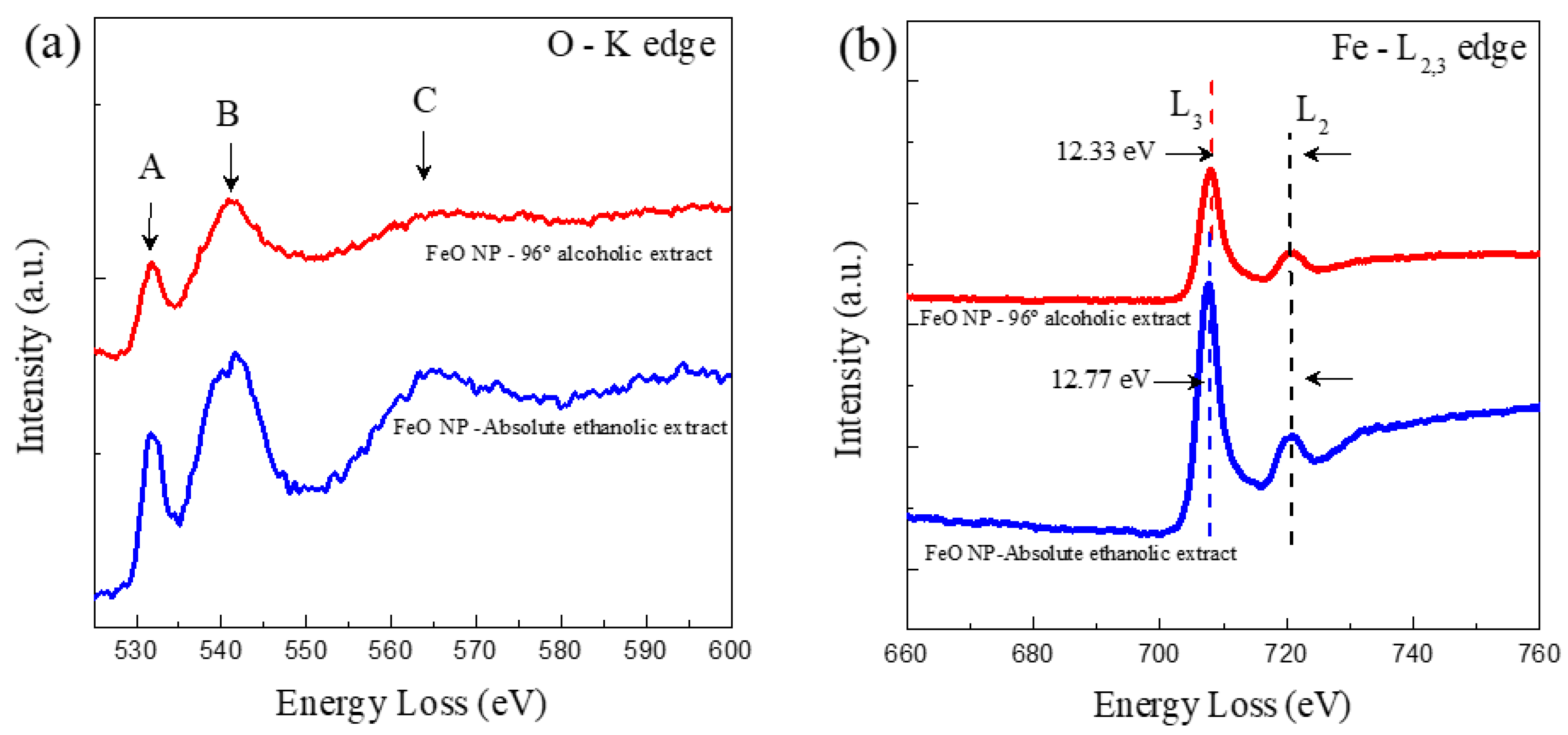
3.6. Characterization by Electron Energy Loss Spectroscopy (EELS)
3.7. Characterization by Potential Zeta
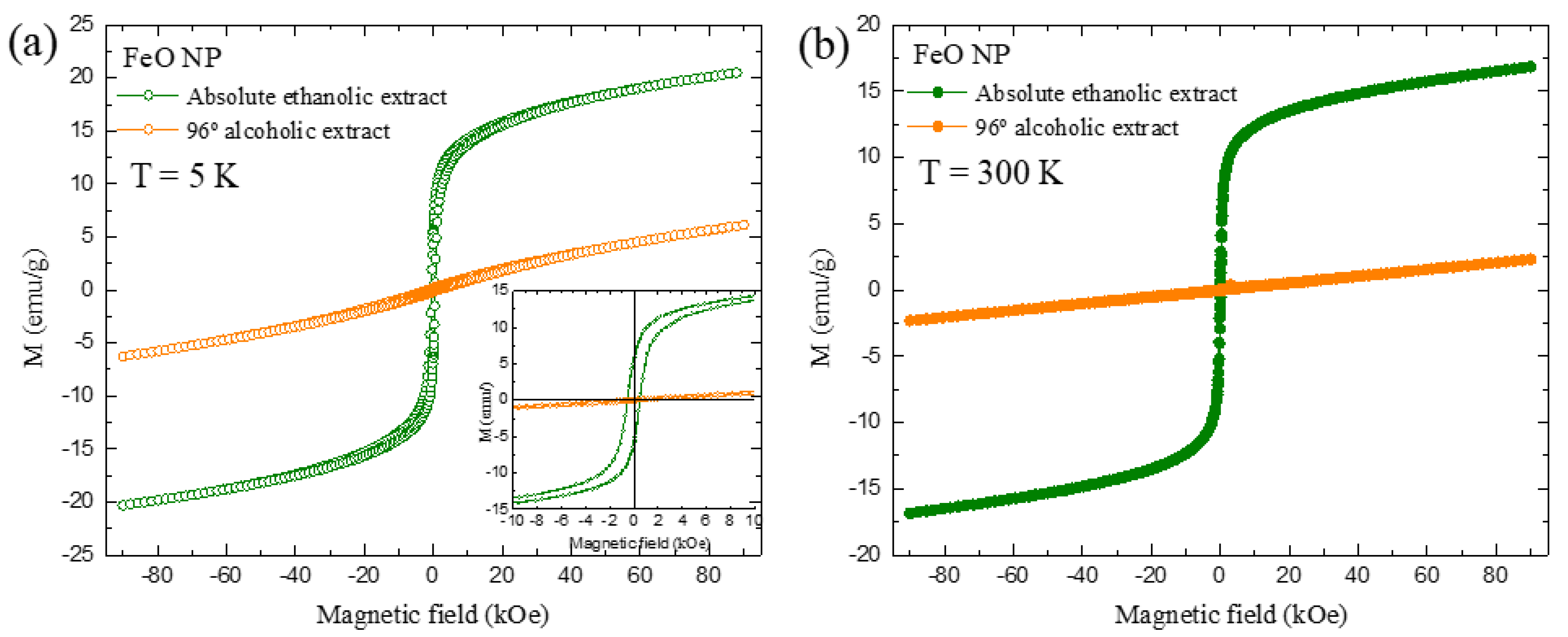
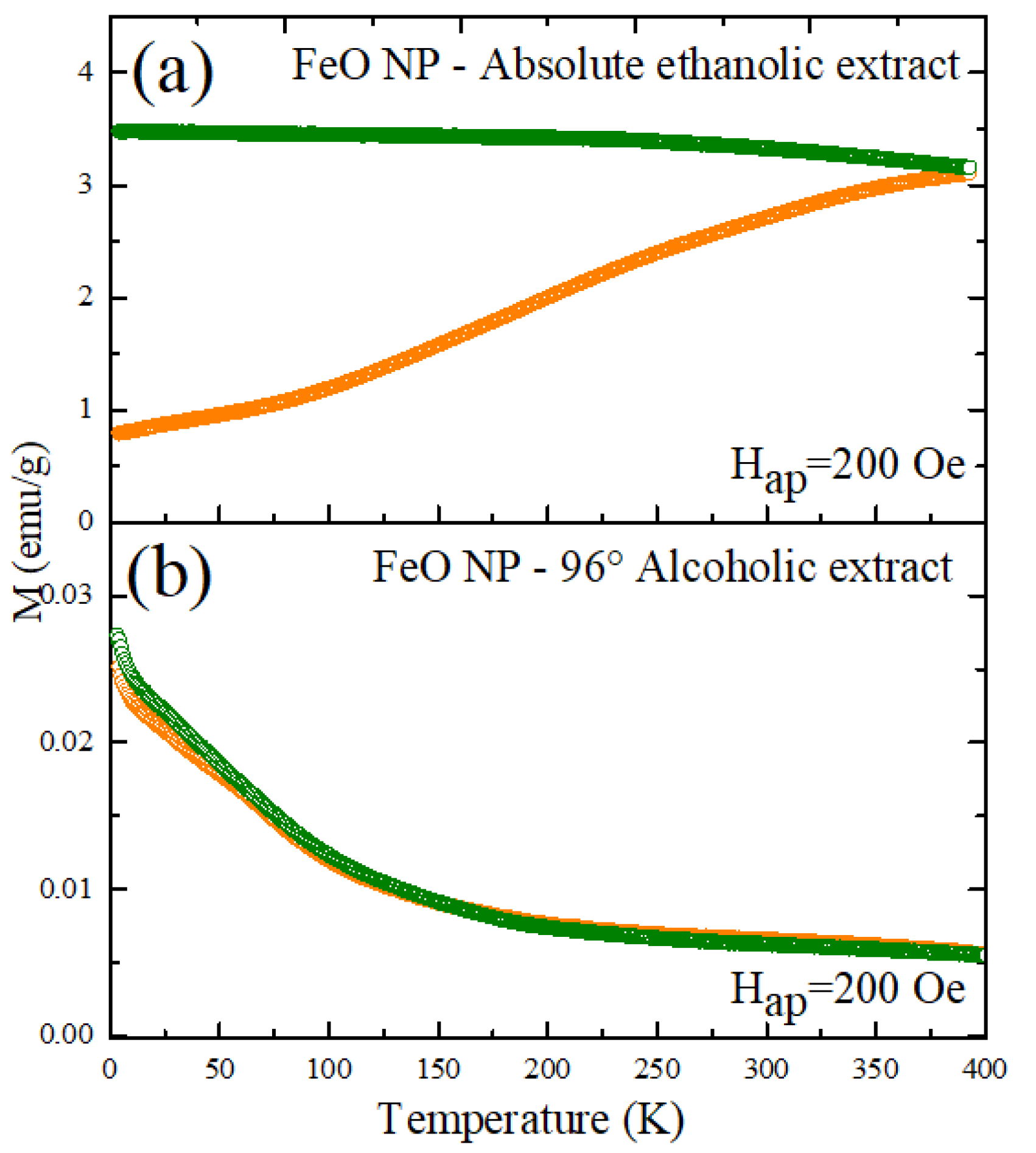
3.8. Magnetization Measurements (SPION)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Samadi, Z.; Yaghmaeian, K.; Mortazavi-Derazkola, S.; Khosravi, R.; Nabizadeh, R.; Alimohammadi, M. Facile green synthesis of zero-valent iron nanoparticles using barberry leaf extract (GnZVI@BLE) for photocatalytic reduction of hexavalent chromium. Bioorg. Chem. 2021, 114, 105051. [Google Scholar] [CrossRef] [PubMed]
- Patiño-Ruiz, D.; Sánchez-Botero, L.; Tejeda-Benitez, L.; Hinestroza, J.; Herrera, A. Green synthesis of iron oxide nanoparticles using Cymbopogon citratus extract and sodium carbonate salt: Nanotoxicological considerations for potential environmental applications. Environ. Nanotechnol. Monit. Manag. 2020, 14, 100377. [Google Scholar] [CrossRef]
- Ardakani, L.S.; Alimardani, V.; Tamaddon, A.M.; Amani, A.M.; Taghizadeh, S. Green synthesis of iron-based nanoparticles using Chlorophytum comosum leaf extract: Methyl orange dye degradation and antimicrobial properties. Heliyon 2021, 7, e06159. [Google Scholar] [CrossRef]
- Jiménez, E.E.G. Control de la superficie y el volumen en la nanoescala para la configuración y el diseño de nanodispositivos. Revista de la Academia Colombiana de Ciencias Exactas, Físicas y Naturales 2016, 40, 590–599. [Google Scholar] [CrossRef] [Green Version]
- Bibi, I.; Nazar, N.; Ata, S.; Sultan, M.; Ali, A.; Abbas, A.; Jilani, K.; Kamal, S.; Sarim, F.M.; Khan, M.I.; et al. Green synthesis of iron oxide nanoparticles using pomegranate seeds extract and photocatalytic activity evaluation for the degradation of textile dye. J. Mater. Res. Technol. 2019, 8, 6115–6124. [Google Scholar] [CrossRef]
- Waris, A.; Din, M.; Ali, A.; Afridi, S.; Baset, A.; Khan, A.U.; Ali, M. Green fabrication of Co and Co3O4 nanoparticles and their biomedical applications: A review. Open Life Sci. 2021, 16, 14–30. [Google Scholar] [CrossRef] [PubMed]
- Devatha, C.; Thalla, A.K.; Katte, S.Y. Green synthesis of iron nanoparticles using different leaf extracts for treatment of domestic waste water. J. Clean. Prod. 2016, 139, 1425–1435. [Google Scholar] [CrossRef]
- Zhu, F.; Ma, S.; Liu, T.; Deng, X. Green synthesis of nano zero-valent iron/Cu by green tea to remove hexavalent chromium from groundwater. J. Clean. Prod. 2018, 174, 184–190. [Google Scholar] [CrossRef]
- Brinch, A.; Hansen, S.F.; Hartmann, N.B.; Baun, A. EU Regulation of Nanobiocides: Challenges in Implementing the Biocidal Product Regulation (BPR). Nanomaterials 2016, 6, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calderón-Jiménez, B.; Johnson, M.E.; Montoro Bustos, A.R.; Murphy, K.E.; Winchester, M.R.; Vega Baudrit, J.R. Silver Nanoparticles: Technological Advances, Societal Impacts, and Metrological Challenges. Front. Chem. 2017, 5, 6. [Google Scholar] [CrossRef] [Green Version]
- Ramos, A.P.; Cruz, M.; Tovani, C.B.; Ciancaglini, P. Biomedical applications of nanotechnology. Biophys. Rev. 2017, 9, 79–89. [Google Scholar] [CrossRef]
- Fazlzadeh, M.; Rahmani, K.; Zarei, A.; Abdoallahzadeh, H.; Nasiri, F.; Khosravi, R. A novel green synthesis of zero valent iron nanoparticles (NZVI) using three plant extracts and their efficient application for removal of Cr(VI) from aqueous solutions. Adv. Powder Technol. 2017, 28, 122–130. [Google Scholar] [CrossRef]
- Rong, K.; Wang, J.; Zhang, Z.; Zhang, J. Green synthesis of iron nanoparticles using Korla fragrant pear peel extracts for the removal of aqueous Cr(VI). Ecol. Eng. 2020, 149, 105793. [Google Scholar] [CrossRef]
- Wei, Y.; Fang, Z.; Zheng, L.; Tsang, P.E. Biosynthesized iron nanoparticles in aqueous extracts of Eichhornia crassipes and its mechanism in the hexavalent chromium removal. Appl. Surf. Sci. 2017, 399, 322–329. [Google Scholar] [CrossRef]
- Carneiro, L.A.; Ward, R.J. Functionalization of paramagnetic nanoparticles for protein immobilization and purification. Anal. Biochem. 2018, 540–541, 45–51. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, W.; Yang, F.; Zhou, B.; Zeng, D.; Zhang, N.; Zhao, G.; Hao, S.; Zhang, X. Ru nanoparticles dispersed on magnetic yolk–shell nanoarchitectures with Fe3O4 core and sulfoacid-containing periodic mesoporous organosilica shell as bifunctional catalysts for direct conversion of cellulose to isosorbide. Nanoscale 2018, 10, 2199–2206. [Google Scholar] [CrossRef]
- Khatami, M.; Alijani, H.Q.; Fakheri, B.; Mobasseri, M.M.; Heydarpour, M.; Farahani, Z.K.; Khan, A.U. Super-paramagnetic iron oxide nanoparticles (SPIONs): Greener synthesis using Stevia plant and evaluation of its antioxidant properties. J. Clean. Prod. 2019, 208, 1171–1177. [Google Scholar] [CrossRef]
- Periakaruppan, R.; Chen, X.; Thangaraj, K.; Jeyaraj, A.; Nguyen, H.H.; Yu, Y.; Hu, S.; Lu, L.; Li, X. Utilization of tea resources with the production of superparamagnetic biogenic iron oxide nanoparticles and an assessment of their antioxidant activities. J. Clean. Prod. 2021, 278, 123962. [Google Scholar] [CrossRef]
- Zhang, P.; Hou, D.; O’Connor, D.; Li, X.; Pehkonen, S.; Varma, R.; Wang, X. Green and Size-Specific Synthesis of Stable Fe–Cu Oxides as Earth-Abundant Adsorbents for Malachite Green Removal. ACS Sustain. Chem. Eng. 2018, 6, 9229–9236. [Google Scholar] [CrossRef]
- Plachtová, P.; Medříková, Z.; Zbořil, R.; Tuček, J.; Varma, R.S.; Maršálek, B. Iron and Iron Oxide Nanoparticles Synthesized with Green Tea Extract: Differences in Ecotoxicological Profile and Ability to Degrade Malachite Green. ACS Sustain. Chem. Eng. 2018, 6, 8679–8687. [Google Scholar] [CrossRef]
- Sebastian, A.; Nangia, A.; Prasad, M. A green synthetic route to phenolics fabricated magnetite nanoparticles from coconut husk extract: Implications to treat metal contaminated water and heavy metal stress in Oryza sativa L. J. Clean. Prod. 2018, 174, 355–366. [Google Scholar] [CrossRef]
- Xiao, X.; Wang, Q.; Owens, G.; Chiellini, F.; Chen, Z. Reduced graphene oxide/iron nanoparticles used for the removal of Pb (II) by one step green synthesis. J. Colloid Interface Sci. 2019, 557, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Gautam, P.K.; Shivalkar, S.; Banerjee, S. Synthesis of M. oleifera leaf extract capped magnetic nanoparticles for effective lead [Pb (II)] removal from solution: Kinetics, isotherm and reusability study. J. Mol. Liq. 2020, 305, 112811. [Google Scholar] [CrossRef]
- Lakshminarayanan, S.; Shereen, M.F.; Niraimathi, K.L.; Brindha, P.; Arumugam, A. Author Correction: One-pot green synthesis of iron oxide nanoparticles from Bauhinia tomentosa: Characterization and application towards synthesis of 1, 3 diolein. Sci. Rep. 2021, 11, 8643. [Google Scholar] [CrossRef] [PubMed]
- Makarov, V.V.; Love, A.J.; Sinitsyna, O.V.; Makarova, S.S.; Yaminsky, I.V.; Taliansky, M.E.; Kalinina, N.O. “Green” Nanotechnologies: Synthesis of Metal Nanoparticles Using Plants. Acta Nat. 2014, 6, 35. [Google Scholar] [CrossRef] [Green Version]
- Sundeep, D.; Vijaya Kumar, T.; Rao, P.S.S.; Ravikumar, R.V.S.S.N.; Gopala Krishna, A. Green synthesis and characterization of Ag nanoparticles from Mangifera indica leaves for dental restoration and antibacterial applications. Prog. Biomater. 2017, 6, 57–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koshti, B.; Kshtriya, V.; Nardin, C.; Gour, N. Chemical Perspective of the Mechanism of Action of Antiamyloidogenic Compounds Using a Minimalistic Peptide as a Reductionist Model. ACS Chem. Neurosci. 2021, 12, 2851–2864. [Google Scholar] [CrossRef] [PubMed]
- Soliemanzadeh, A.; Fekri, M. The application of green tea extract to prepare bentonite-supported nanoscale zero-valent iron and its performance on removal of Cr(VI): Effect of relative parameters and soil experiments. Microporous Mesoporous Mater. 2017, 239, 60–69. [Google Scholar] [CrossRef]
- Francy, N.; Shanthakumar, S.; Chiampo, F.; Sekhar, Y.R. Remediation of Lead and Nickel Contaminated Soil Using Nanoscale Zero-Valent Iron (nZVI) Particles Synthesized Using Green Leaves: First Results. Processes 2020, 8, 1453. [Google Scholar] [CrossRef]
- Su, B.; Lin, J.; Owens, G.; Chen, Z. Impact of green synthesized iron oxide nanoparticles on the distribution and transformation of As species in contaminated soil. Environ. Pollut. 2020, 258, 113668. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Su, B.; Sun, M.; Chen, B.; Chen, Z. Biosynthesized iron oxide nanoparticles used for optimized removal of cadmium with response surface methodology. Sci. Total Environ. 2018, 627, 314–321. [Google Scholar] [CrossRef]
- Rana, A.; Kumari, N.; Tyagi, M.; Jagadevan, S. Leaf-extract mediated zero-valent iron for oxidation of Arsenic (III): Preparation, characterization and kinetics. Chem. Eng. J. 2018, 347, 91–100. [Google Scholar] [CrossRef]
- Wu, Z.; Su, X.; Lin, Z.; Khan, N.I.; Owens, G.; Chen, Z. Removal of As(V) by iron-based nanoparticles synthesized via the complexation of biomolecules in green tea extracts and an iron salt. Sci. Total Environ. 2021, 764, 142883. [Google Scholar] [CrossRef]
- Prabhakar, R.; Samadder, S.R. Jyotsana Aquatic and terrestrial weed mediated synthesis of iron nanoparticles for possible application in wastewater remediation. J. Clean. Prod. 2017, 168, 1201–1210. [Google Scholar] [CrossRef]
- Ameen, F.; Srinivasan, P.; Selvankumar, T.; Kamala-Kannan, S.; Al Nadhari, S.; Almansob, A.; Dawoud, T.; Govarthanan, M. Phytosynthesis of silver nanoparticles using Mangifera indica flower extract as bioreductant and their broad-spectrum antibacterial activity. Bioorg. Chem. 2019, 88, 102970. [Google Scholar] [CrossRef] [PubMed]
- Philip, D. Rapid green synthesis of spherical gold nanoparticles using Mangifera indica leaf. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2010, 77, 807–810. [Google Scholar] [CrossRef]
- Schulze-Kaysers, N.; Feuereisen, M.M.; Schieber, A. Phenolic compounds in edible species of the Anacardiaceae family—A review. RSC Adv. 2015, 5, 73301–73314. [Google Scholar] [CrossRef]
- Arularasu, M.; Devakumar, J.; Rajendran, T. An innovative approach for green synthesis of iron oxide nanoparticles: Characterization and its photocatalytic activity. Polyhedron 2018, 156, 279–290. [Google Scholar] [CrossRef]
- Rizvi, M.; Bhatia, T.; Gupta, R. Green & sustainable synthetic route of obtaining iron oxide nanoparticles using Hylocereus undantus (pitaya or dragon fruit). Mater. Today Proc. 2021, 1, 1–7. [Google Scholar] [CrossRef]
- Kumar, K.M.; Mandal, B.K.; Kumar, K.S.; Reddy, P.S.; Sreedhar, B. Biobased green method to synthesise palladium and iron nanoparticles using Terminalia chebula aqueous extract. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 102, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Asmat-Campos, D.; Abreu, A.C.; Romero-Cano, M.S.; Urquiaga-Zavaleta, J.; Contreras-Cáceres, R.; Delfín-Narciso, D.; Juárez-Cortijo, L.; Nazario-Naveda, R.; Rengifo-Penadillos, R.; Fernández, I. Unraveling the Active Biomolecules Responsible for the Sustainable Synthesis of Nanoscale Silver Particles through Nuclear Magnetic Resonance Metabolomics. ACS Sustain. Chem. Eng. 2020, 8, 17816–17827. [Google Scholar] [CrossRef]
- Devi, T.B.; Ahmaruzzaman, M.; Begum, S. A rapid, facile and green synthesis of Ag@AgCl nanoparticles for the effective reduction of 2,4-dinitrophenyl hydrazine. New J. Chem. 2016, 40, 1497–1506. [Google Scholar] [CrossRef]
- Demirezen, D.A.; Yıldız, Ş.Y.; Yılmaz, Ş.; Yılmaz, D.D. Green synthesis and characterization of iron oxide nanoparticles using Ficus carica (common fig) dried fruit extract. J. Biosci. Bioeng. 2019, 127, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, S.; Habib, F.; Darwish, N.; Shanableh, A. Iron sulfide nanoparticles prepared using date seed extract: Green synthesis, characterization and potential application for removal of ciprofloxacin and chromium. Powder Technol. 2021, 380, 219–228. [Google Scholar] [CrossRef]
- Maity, D.; Choo, S.-G.; Yi, J.; Ding, J.; Xue, J.M. Synthesis of magnetite nanoparticles via a solvent-free thermal decomposition route. J. Magn. Magn. Mater. 2009, 321, 1256–1259. [Google Scholar] [CrossRef]
- Petcharoen, K.; Sirivat, A. Synthesis and characterization of magnetite nanoparticles via the chemical co-precipitation method. Mater. Sci. Eng. B 2012, 177, 421–427. [Google Scholar] [CrossRef]
- Rajput, S.; Singh, L.P.; Pittman, C.U.; Mohan, D. Lead (Pb2+ ) and copper (Cu2+ ) remediation from water using superparamagnetic maghemite (γ-Fe2O3 ) nanoparticles synthesized by Flame Spray Pyrolysis (FSP). J. Colloid Interface Sci. 2017, 492, 176–190. [Google Scholar] [CrossRef] [PubMed]
- Karade, V.; Dongale, T.; Sahoo, S.; Kollu, P.; Chougale, A.; Patil, P. Effect of reaction time on structural and magnetic properties of green-synthesized magnetic nanoparticles. J. Phys. Chem. Solids 2018, 120, 161–166. [Google Scholar] [CrossRef]
- Yadav, V.K.; Fulekar, M. Biogenic synthesis of maghemite nanoparticles (γ-Fe2O3) using Tridax leaf extract and its application for removal of fly ash heavy metals (Pb, Cd). Mater. Today Proc. 2018, 5, 20704–20710. [Google Scholar] [CrossRef]
- Rajendran, A.; Alsawalha, M.; Alomayri, T. Biogenic synthesis of husked rice-shaped iron oxide nanoparticles using coconut pulp (Cocos nucifera L.) extract for photocatalytic degradation of Rhodamine B dye and their in vitro antibacterial and anticancer activity. J. Saudi Chem. Soc. 2021, 25, 101307. [Google Scholar] [CrossRef]
- Tovar, G.I.; Briceño, S.; Suarez, J.; Flores, S.; González, G. Biogenic synthesis of iron oxide nanoparticles using Moringa oleifera and chitosan and its evaluation on corn germination. Environ. Nanotechnol. Monit. Manag. 2020, 14, 100350. [Google Scholar] [CrossRef]
- Puca Pacheco, M.; Guerrero Aquino, M.; Tacuri Calanchi, E.; López Campos, R.G. Síntesis Y Caracterización de Nanopartículas Superparamagnéticas Obtenidas Por Precipitación en Microemulsión Inversa Para Aplicaciones Biomédicas Synthesis and Characterization of Superparamagnetic Nanoparticles Obtained by Precipitation in Inverse Microemulsion for Biomedical Applications. Rev. Soc. Quím. Perú 2013, 79, 99–106. [Google Scholar]
- Gholami, L.; Oskuee, R.K.; Tafaghodi, M.; Farkhani, A.R.; Darroudi, M. Green facile synthesis of low-toxic superparamagnetic iron oxide nanoparticles (SPIONs) and their cytotoxicity effects toward Neuro2A and HUVEC cell lines. Ceram. Int. 2018, 44, 9263–9268. [Google Scholar] [CrossRef]
- Ma, X.; Gong, A.; Chen, B.; Zheng, J.; Chen, T.; Shen, Z.; Wu, A. Exploring a new SPION-based MRI contrast agent with excellent water-dispersibility, high specificity to cancer cells and strong MR imaging efficacy. Colloids Surfaces B Biointerfaces 2015, 126, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhang, J.; You, C.; Song, Z.; Yu, B.; Shen, Y. Influences of different synthesis conditions on properties of Fe3O4 nanoparticles. Mater. Chem. Phys. 2009, 113, 46–52. [Google Scholar] [CrossRef]
- Ministerio del Ambiente -MINAM Estándares de Calidad Ambiental (ECA) para Suelo. Available online: http://www.minam.gob.pe/wp-content/uploads/2017/12/DS_011-2017-MINAM.pdf (accessed on 18 October 2021).
- Jia, Z.; Shu, Y.; Huang, R.; Liu, J.; Liu, L. Enhanced reactivity of nZVI embedded into supermacroporous cryogels for highly efficient Cr(VI) and total Cr removal from aqueous solution. Chemosphere 2018, 199, 232–242. [Google Scholar] [CrossRef]
- Wu, Z.; Su, X.; Lin, Z.; Owens, G.; Chen, Z. Mechanism of As(V) removal by green synthesized iron nanoparticles. J. Hazard. Mater. 2019, 379, 120811. [Google Scholar] [CrossRef]
- Yi, Y.; Tu, G.; Tsang, P.E.; Xiao, S.; Fang, Z. Green synthesis of iron-based nanoparticles from extracts of Nephrolepis auriculata and applications for Cr(VI) removal. Mater. Lett. 2019, 234, 388–391. [Google Scholar] [CrossRef]
- Biftu, W.K.; Ravindhranath, K.; Ramamoorty, M. New research trends in the processing and applications of iron-based nanoparticles as adsorbents in water remediation methods. Nanotechnol. Environ. Eng. 2020, 5, 12. [Google Scholar] [CrossRef]
- Hlongwane, G.; Sekoai, P.T.; Meyyappan, M.; Moothi, K. Simultaneous removal of pollutants from water using nanoparticles: A shift from single pollutant control to multiple pollutant control. Sci. Total Environ. 2019, 656, 808–833. [Google Scholar] [CrossRef]
- Mashkoor, F.; Nasar, A. Magsorbents: Potential candidates in wastewater treatment technology—A review on the removal of methylene blue dye. J. Magn. Magn. Mater. 2020, 500, 166408. [Google Scholar] [CrossRef]
- Giri, R.K.V.; Raju, L.S.; Nancharaiah, Y.V.; Pulimi, M.; Chandrasekaran, N.; Mukherjee, A. Anaerobic nano zero-valent iron granules for hexavalent chromium removal from aqueous solution. Environ. Technol. Innov. 2019, 16, 100495. [Google Scholar] [CrossRef]
- Yirsaw, B.D.; Megharaj, M.; Chen, Z.; Naidu, R. Reduction of hexavalent chromium by green synthesized nano zero valent iron and process optimization using response surface methodology. Environ. Technol. Innov. 2016, 5, 136–147. [Google Scholar] [CrossRef]
- Mitra, P.; Sarkar, D.; Chakrabarti, S.; Dutta, B.K. Reduction of hexa-valent chromium with zero-valent iron: Batch kinetic studies and rate model. Chem. Eng. J. 2011, 171, 54–60. [Google Scholar] [CrossRef]
- Chen, W.; Duan, L.; Wang, L.; Zhu, D. Adsorption of Hydroxyl- and Amino-Substituted Aromatics to Carbon Nanotubes. Environ. Sci. Technol. 2008, 42, 6862–6868. [Google Scholar] [CrossRef]
- Nassar, N.N. Rapid removal and recovery of Pb(II) from wastewater by magnetic nanoadsorbents. J. Hazard. Mater. 2010, 184, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Ngueagni, P.T.; Kumar, P.S.; Woumfo, E.D.; Abilarasu, A.; Joshiba, G.J.; Carolin, C.F.; Prasannamedha, G.; Fotsing, P.N.; Siewe, M. Effectiveness of a biogenic composite derived from cattle horn core/iron nanoparticles via wet chemical impregnation for cadmium (II) removal in aqueous solution. Chemosphere 2021, 272, 129806. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Barick, K.C.; Bahadur, D. Functional Oxide Nanomaterials and Nanocomposites for the Removal of Heavy Metals and Dyes. Nanomater. Nanotechnol. 2013, 3, 20. [Google Scholar] [CrossRef] [Green Version]
- Tu, B.; Chen, H.; Xue, S.; Deng, J.; Tao, H. Ultrafast and efficient removal of aqueous Cr(VI) using iron oxide nanoparticles supported on Bermuda grass-based activated carbon. J. Mol. Liq. 2021, 334, 116026. [Google Scholar] [CrossRef]
- Aragaw, T.A.; Bogale, F.M.; Aragaw, B.A. Iron-based nanoparticles in wastewater treatment: A review on synthesis methods, applications, and removal mechanisms. J. Saudi Chem. Soc. 2021, 25, 101280. [Google Scholar] [CrossRef]
- Jesitha, K.; Jaseela, C.; Harikumar, P. Nanotechnology enhanced phytoremediation and photocatalytic degradation techniques for remediation of soil pollutants. Nanomater. Soil Remediat. 2021, 463–499. [Google Scholar] [CrossRef]




| CHROMIUM | CADMIUM | LEAD | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Abs. | Initial ppm | Final ppm | Removal (%) | Abs. | Initial ppm | Final ppm | Removal (%) | Abs. | Initial ppm | Final ppm | Removal (%) |
| C3 | 0.0046 | 734.07 | 3.87 | 99.47 | 0.008 | 0.2511 | 0.0465 | 81.48 | 0.16 | 497.27 | 467.91 | 5.90 |
| C2 | 0.0045 | 3.815 | 99.48 | 0.0010 | 0.1488 | 40.74 | 0.18 | 526.63 | 0 | |||
| C1 | 1.17 | 734.07 | 0 | 0.013 | 0.3023 | 0 | 0.18 | 526.63 | 0 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bautista-Guzman, J.; Gomez-Morales, R.; Asmat-Campos, D.; Checca, N.R. Influence of the Alcoholic/Ethanolic Extract of Mangifera indica Residues on the Green Synthesis of FeO Nanoparticles and Their Application for the Remediation of Agricultural Soils. Molecules 2021, 26, 7633. https://doi.org/10.3390/molecules26247633
Bautista-Guzman J, Gomez-Morales R, Asmat-Campos D, Checca NR. Influence of the Alcoholic/Ethanolic Extract of Mangifera indica Residues on the Green Synthesis of FeO Nanoparticles and Their Application for the Remediation of Agricultural Soils. Molecules. 2021; 26(24):7633. https://doi.org/10.3390/molecules26247633
Chicago/Turabian StyleBautista-Guzman, Jhordi, Rosa Gomez-Morales, David Asmat-Campos, and Noemi Raquel Checca. 2021. "Influence of the Alcoholic/Ethanolic Extract of Mangifera indica Residues on the Green Synthesis of FeO Nanoparticles and Their Application for the Remediation of Agricultural Soils" Molecules 26, no. 24: 7633. https://doi.org/10.3390/molecules26247633
APA StyleBautista-Guzman, J., Gomez-Morales, R., Asmat-Campos, D., & Checca, N. R. (2021). Influence of the Alcoholic/Ethanolic Extract of Mangifera indica Residues on the Green Synthesis of FeO Nanoparticles and Their Application for the Remediation of Agricultural Soils. Molecules, 26(24), 7633. https://doi.org/10.3390/molecules26247633





