Anti-Oxidant and Anti-Inflammatory Effects of Lipopolysaccharide from Rhodobacter sphaeroides against Ethanol-Induced Liver and Kidney Toxicity in Experimental Rats
Abstract
1. Introduction
2. Results and Discussion
2.1. Effect of LPS-RS on Serum Levels of Liver Enzymes and Kidney Function Markers
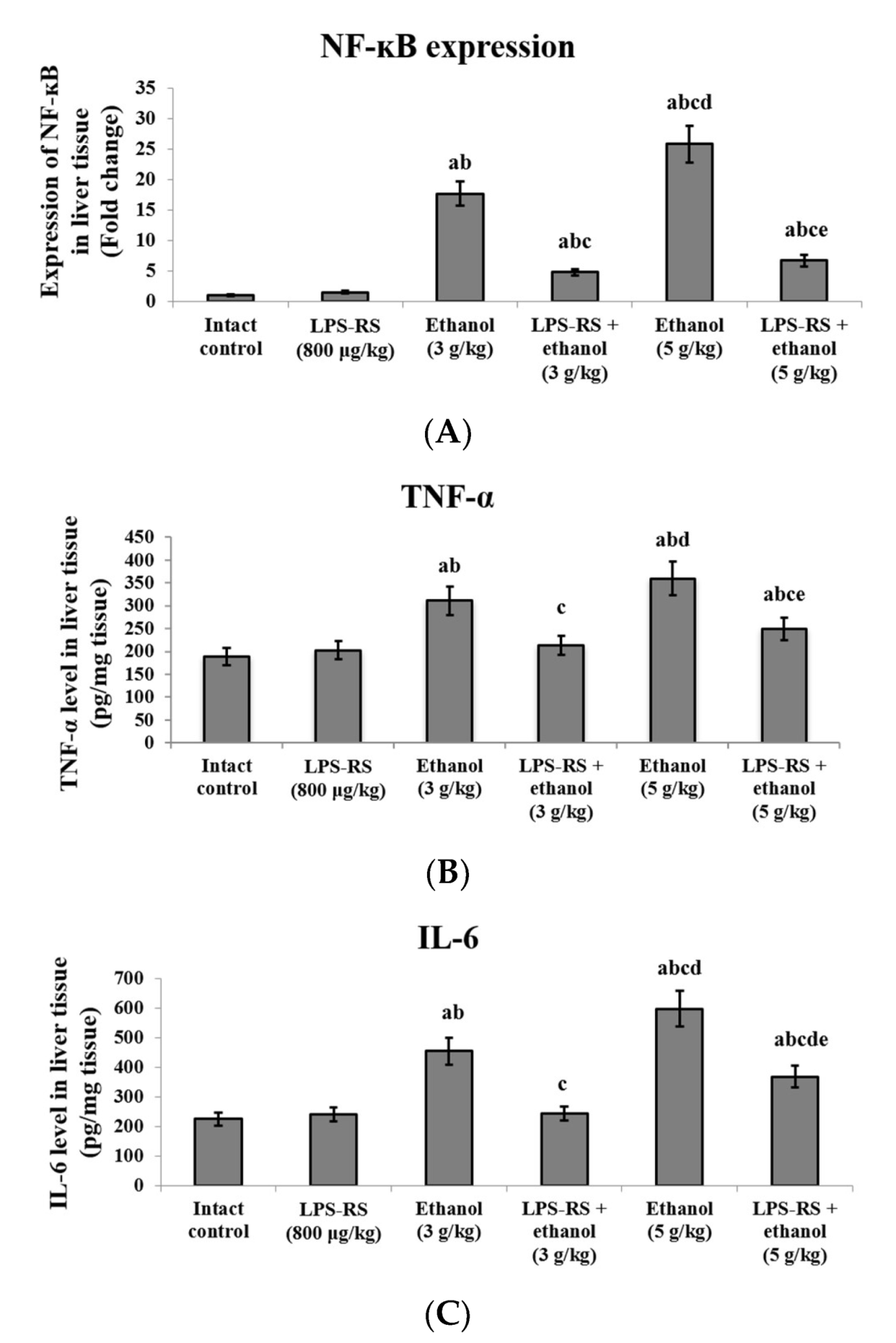
2.2. Effect of LPS-RS on Oxidative Stress and Inflammatory Markers in the Liver Tissue
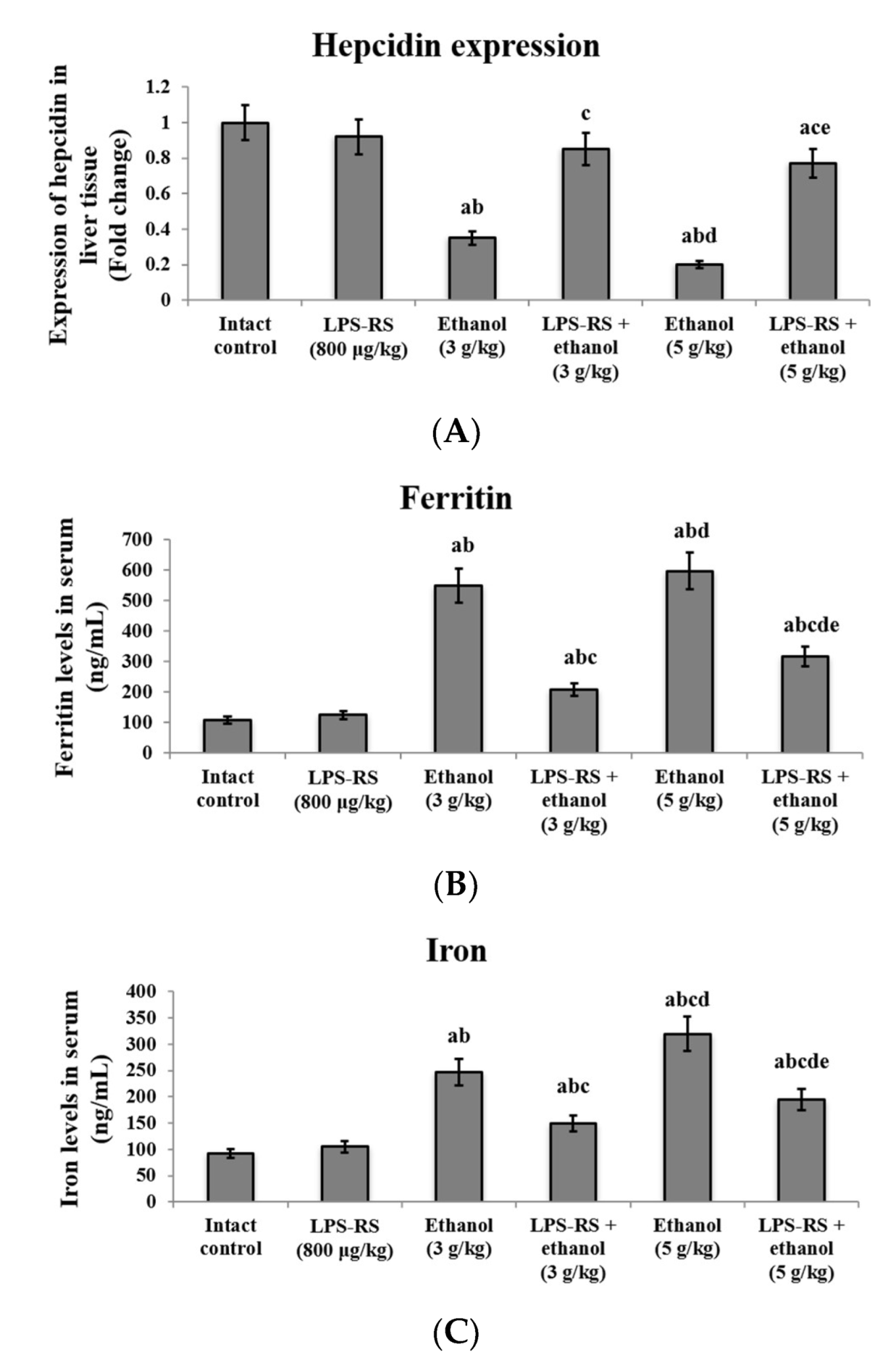
2.3. Effect of LPS-RS on the Expression of Hepcidin in the Liver Tissue and Serum Levels of Iron and Ferritin
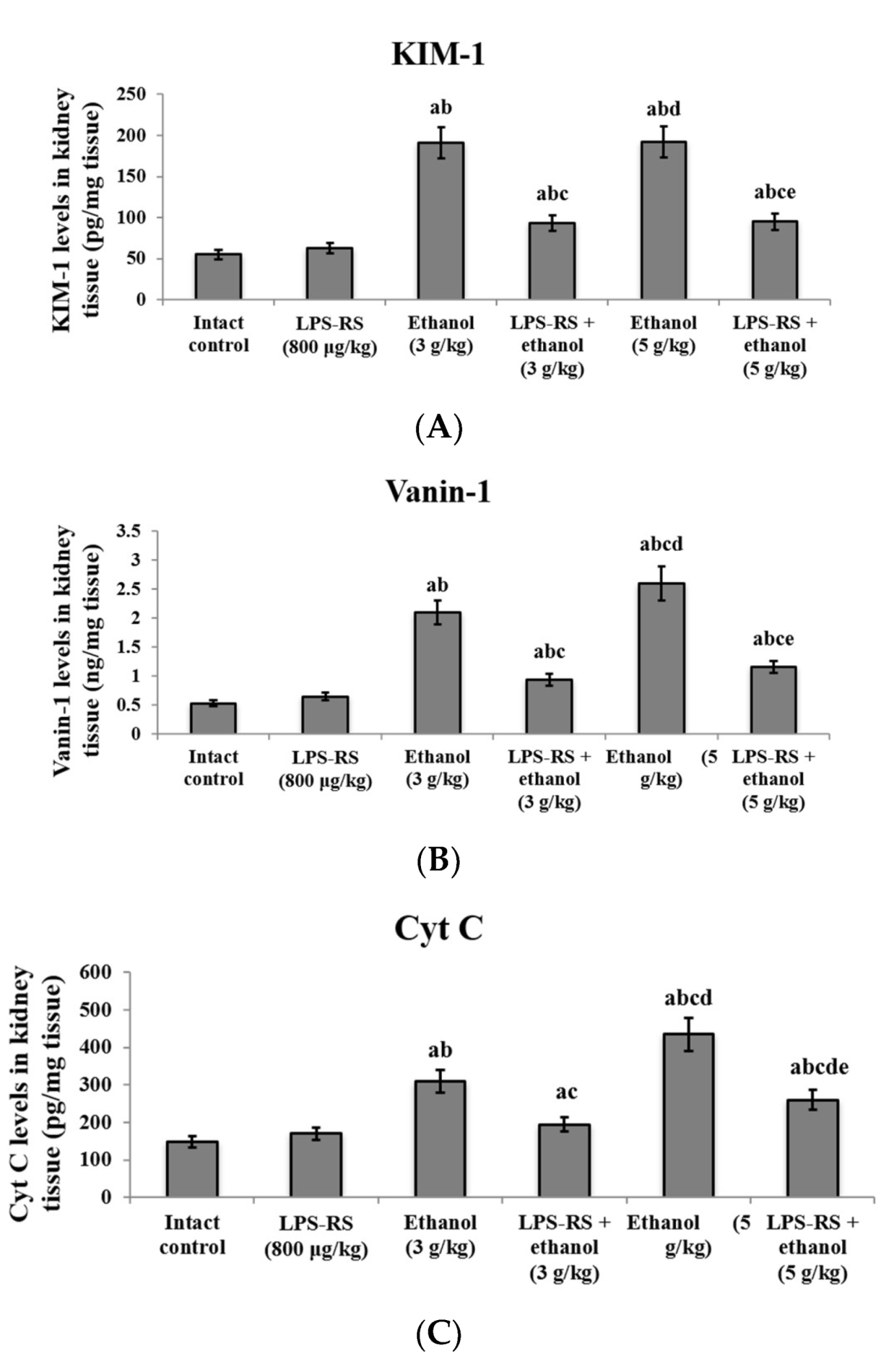
2.4. Effect of LPS-RS on Markers of Inflammation and Kidney Injury in the Renal Tissue
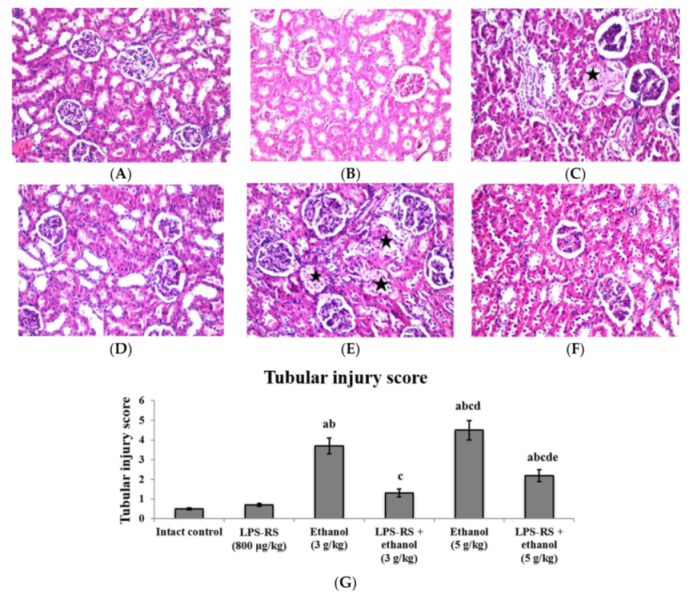
2.5. Histopathological Examination of the Liver and Kidney Tissues
3. Materials and Methods
3.1. Experimental Animals
3.2. Study Design
3.3. Collection of Samples
3.4. Determination of Liver and Kidney Function Markers in Serum
3.5. Assessment of Markers of Oxidative Stress and Inflammation in the Liver Tissue
3.6. Quantitative Assessment of the Expression of Nuclear Factor Kappa B (NF-κB) and Hepcidin by Quantitative Real-Time PCR
3.7. Determination of Markers of Renal Inflammation and Injury in the Kidney Tissue
3.8. Histopathological Examination of the Liver and the Kidney
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Osna, N.A.; Donohue, T.M., Jr.; Kharbanda, K.K. Alcoholic liver disease: Pathogenesis and current management. Alcohol Res. 2017, 38, 147–161. [Google Scholar]
- Cederbaum, A.I. Alcohol metabolism. Clin. Liver Dis. 2012, 16, 667–865. [Google Scholar] [CrossRef]
- Galicia-Moreno, M.; Gutiérrez-Reyes, G. The role of oxidative stress in the development of alcoholic liver disease. Rev. Gastroenterol. Mex. 2014, 79, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Ceni, E.; Mello, T.; Galli, A. Pathogenesis of alcoholic liver disease: Role of oxidative metabolism. World J. Gastroenterol. 2014, 20, 17756–17772. [Google Scholar] [CrossRef]
- Souli, A.; Sebai, H.; Chehimi, L.; Rtibi, K.; Tounsi, H.; Boubaker, S.; Sakly, M.; El-Benna, J.; Amri, M. Hepatoprotective effect of carob against acute ethanol-induced oxidative stress in rat. Toxicol. Ind. Health 2015, 31, 802–810. [Google Scholar] [CrossRef]
- Lin, H.Z.; Yang, S.Q.; Zeldin, G.; Diehl, A.M. Chronic ethanol consumption induces the production of tumor necrosis factor-alpha and related cytokines in liver and adipose tissue. Alcohol. Clin. Exp. Res. 1998, 22, 231S–237S. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Hong, M.; Tan, H.Y.; Wang, N.; Feng, Y. Insights into the Role and Interdependence of Oxidative Stress and Inflammation in Liver Diseases. Oxid. Med. Cell. Longev. 2016, 2016, 4234061. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.J.; Zhou, Z.; Parker, R.; Gao, B. Targeting inflammation for the treatment of alcoholic liver disease. Pharmacol. Ther. 2017, 180, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Radicheva, M.P.; Andonova, A.N.; Milcheva, H.T.; Ivanova, N.G.; Kyuchukova, S.G.; Nikolova, M.S.; Platikanova, A.S. Serum Markers of Iron Metabolism in Chronic Liver Diseases. Open Access Maced. J. Med. Sci. 2018, 6, 1010–1016. [Google Scholar] [CrossRef] [PubMed]
- Bridle, K.; Cheung, T.K.; Murphy, T.; Walters, M.; Anderson, G.; Crawford, D.G.; Fletcher, L.M. Hepcidin is down-regulated in alcoholic liver injury: Implications for the pathogenesis of alcoholic liver disease. Alcohol. Clin. Exp. Res. 2006, 30, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Bloomer, S.A.; Brown, K.E. Hepcidin and iron metabolism in experimental liver injury. Am. J. Pathol. 2021, 191, 1165–1179. [Google Scholar] [CrossRef] [PubMed]
- Harrison-Findik, D.D.; Schafer, D.; Klein, E.; Timchenko, N.A.; Kulaksiz, H.; Clemens, D.; Fein, E.; Andriopoulos, B.; Pantopoulos, K.; Gollan, J. Alcohol metabolism-mediated oxidative stress down-regulates hepcidin transcription and leads to increased duodenal iron transporter expression. J. Biol. Chem. 2006, 281, 22974–22982. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, T.; Saito, H.; Hosoki, Y.; Inoue, M.; Miyoshi, S.; Suzuki, Y.; Fujimoto, Y.; Kohgo, Y. Hepcidin is down-regulated in alcohol loading. Alcohol. Clin. Exp. Res. 2007, 31, S2–S8. [Google Scholar] [CrossRef] [PubMed]
- Mehta, K.J.; Farnaud, S.J.; Sharp, P.A. Iron and liver fibrosis: Mechanistic and clinical aspects. World J. Gastroenterol. 2019, 25, 521–538. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A.H.; do Vale, G.T.; da Silva, C.B.P.; Awata, W.M.C.; Pinheiro, L.C.; Tirapelli, C.R. Ethanol and cyclophosphamide induce similar nephrotoxic effects: Possible role for Nox4 and superoxide. Can. J. Physiol. Pharmacol. 2021, 99, 744–751. [Google Scholar] [CrossRef]
- Das Kumar, S.; Vasudevan, D.M. Alcohol induced effects on kidney. Indian J. Clin. Biochem. 2008, 23, 4–9. [Google Scholar] [CrossRef]
- Varga, Z.V.; Matyas, C.; Paloczi, J.; Pacher, P. Alcohol Misuse and Kidney Injury: Epidemiological Evidence and Potential Mechanisms. Alcohol Res. 2017, 38, 283–288. [Google Scholar] [PubMed]
- Vallance, T.M.; Ravishankar, D.; Albadawi, D.A.I.; Layfield, H.; Sheard, J.; Vaiyapuri, R.; Dash, P.; Patel, K.; Widera, D.; Vaiyapuri, S. Effect of ultrapure lipopolysaccharides derived from diverse bacterial species on the modulation of platelet activation. Sci. Rep. 2019, 9, 18258. [Google Scholar] [CrossRef]
- Hyun, J.; Han, J.; Lee, C.; Yoon, M.; Jung, Y. Pathophysiological Aspects of Alcohol Metabolism in the Liver. Int. J. Mol. Sci. 2021, 22, 5717. [Google Scholar] [CrossRef]
- Lu, Y.; Zhuge, J.; Wang, X.; Bai, J.; Cederbaum, A.I. Cytochrome P450 2E1 contributes to ethanol-induced fatty liver in mice. Hepatology 2008, 47, 1483–1494. [Google Scholar] [CrossRef]
- Thurman, R.G.; Bradford, B.U.; Iimuro, Y.; Frankenberg, M.V.; Knecht, K.T.; Connor, H.D.; Adachi, Y.; Wall, C.; Arteel, G.E.; Raleigh, J.A.; et al. Mechanisms of alcohol-induced hepatotoxicity: Studies in rats. Front. Biosci. 1999, 4, e42–e46. [Google Scholar] [CrossRef][Green Version]
- Ambade, A.; Mandrekar, P. Oxidative stress and inflammation: Essential partners in alcoholic liver disease. Int. J. Hepatol. 2012, 2012, 853175. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.D. Endotoxin and Kupffer cell activation in alcoholic liver disease. Alcohol Res. Health 2003, 27, 300–306. [Google Scholar]
- Wu, D.; Cederbaum, A.I. Oxidative stress and alcoholic liver disease. Semin. Liver Dis. 2009, 29, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yang, F.; Wei, F.; Ren, X. The role of toll-like receptor 4 in tumor microenvironment. Oncotarget 2017, 8, 66656–66667. [Google Scholar] [CrossRef]
- McKernan, K.; Varghese, M.; Patel, R.; Singer, K. Role of TLR4 in the induction of inflammatory changes in adipocytes and macrophages. Adipocyte 2020, 9, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Altavilla, D.; Marini, H.; Seminara, P.; Squadrito, G.; Minutoli, L.; Passaniti, M.; Bitto, A.; Calapai, G.; Calò, M.; Caputi, A.P.; et al. Protective effects of antioxidant raxofelast in alcohol-induced liver disease in mice. Pharmacology 2005, 74, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.-M.; Chen, H.; Li, Y. Protective effect of bicyclol on liver injury induced by hepatic warm ischemia/reperfusion in rats. Hepatol. Res. 2009, 39, 833–842. [Google Scholar] [CrossRef]
- Deng, S.; Yu, K.; WU, Q.; Li, Y.; Zhang, X.; Zhang, B.; Liu, G.; Liu, y.; Lian, Z. Toll-Like Receptor 4 Reduces Oxidative Injury via Glutathione Activity in Sheep. Oxid. Med. Cell. Longev. 2016, 2016, 9151290. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, S.; Agrawal-Rajput, R. Lipopolysaccharide from Rhodobacter sphaeroides Attenuates Microglia-Mediated Inflammation and Phagocytosis and Directs Regulatory T Cell Response. Int. J. Inflam. 2015, 2015, 361326. [Google Scholar] [CrossRef][Green Version]
- Abdelmageed, M.E.; El-Awady, M.S.; Abdelrahim, M.; Suddek, G.M. LPS-RS attenuation of lipopolysaccharide-induced acute lung injury involves NF-κB inhibition. Can. J. Physiol. Pharmacol. 2016, 94, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Jurga, A.M.; Rojewska, E.; Makuch, W.; Mika, J. Lipopolysaccharide from Rhodobacter sphaeroides (TLR4 antagonist) attenuates hypersensitivity and modulates nociceptive factors. Pharm. Biol. 2018, 56, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Schürmann, M.; Greiner, J.F.W.; Volland-Thurn, V.; Oppel, F.; Kaltschmidt, C.; Sudhoff, H.; Kaltschmidt, B. Stem Cell-Induced Inflammation in Cholesteatoma is Inhibited by the TLR4 Antagonist LPS-RS. Cells 2020, 9, 199. [Google Scholar] [CrossRef] [PubMed]
- Pagani, A.; Nai, A.; Silvestri, L.; Camaschella, C. Hepcidin and anemia: A tight relationship. Front. Physiol. 2019, 10, 1294. [Google Scholar] [CrossRef]
- Harrison-Findik, D.D. Is the iron regulatory hormone hepcidin a risk factor for alcoholic liver disease? World J. Gastroenterol. 2009, 15, 1186–1193. [Google Scholar] [CrossRef]
- Harrison-Findik, D.D.; Klein, E.; Crist, C.; Evans, J.; Timchenko, N.; Gollan, J. Iron-mediated regulation of liver hepcidin expression in rats and mice is abolished by alcohol. Hepatology 2007, 46, 1979–1985. [Google Scholar] [CrossRef]
- Zmijewski, E.; Lu, S.; Harrison-Findik, D.D. TLR4 signaling and the inhibition of liver hepcidin expression by alcohol. World J. Gastroenterol. 2014, 20, 12161–12170. [Google Scholar] [CrossRef]
- Vela, D. Low hepcidin in liver fibrosis and cirrhosis; a tale of progressive disorder and a case for a new biochemical marker. Mol. Med. 2018, 24, 5. [Google Scholar] [CrossRef]
- Ribot-Hernández, I.; Martín-González, C.; Vera-Delgado, V.; González-Navarrete, L.; de Armas-González, J.F.; Viña-Rodríguez, J.; Sánchez-Pérez, M.J.; Rodríguez-Gaspar, M.; González-Reimers, E. Prognostic value of serum iron, ferritin, and transferrin in chronic alcoholic liver disease. Biol. Trace Elem. Res. 2020, 195, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.N.; Eubanks, S.K.; Schaffer, K.J.; Zhou, C.Y.; Linder, M.C. Secretion of ferritin by rat hepatoma cells and its regulation by inflammatory cytokines and iron. Blood 1997, 90, 4979–4986. [Google Scholar] [CrossRef] [PubMed]
- Costa Matos, L.; Batista, P.; Monteiro, N.; Ribeiro, J.; Cipriano, M.A.; Henriques, P.; Girão, F.; Carvalho, A. Iron stores assessment in alcoholic liver disease. Scand. J. Gastroenterol. 2013, 48, 712–718. [Google Scholar] [CrossRef]
- Milic, S.; Mikolasevic, I.; Orlic, L.; Devcic, E.; Starcevic-Cizmarevic, N.; Stimac, D.; Kapovic, M.; Ristic, S. The role of iron and iron overload in chronic liver disease. Med. Sci. Monit. 2016, 22, 2144–2151. [Google Scholar] [CrossRef] [PubMed]
- Philippe, M.A.; Ruddell, R.G.; Ramm, G.A. Role of iron in hepatic fibrosis: One piece in the puzzle. World J. Gastroenterol. 2007, 13, 4746–4754. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Knovich, M.A.; Coffman, L.G.; Torti, F.M.; Torti, S.V. Serum ferritin: Past, present and future. Biochim. Biophys. Acta 2010, 1800, 760–769. [Google Scholar] [CrossRef]
- Ruddell, R.G.; Hoang-Le, D.; Barwood, J.M.; Rutherford, P.S.; Piva, T.J.; Watters, D.J.; Santambrogio, P.; Arosio, P.; Ramm, G.A. Ferritin functions as a proinflammatory cytokine via iron-independent protein kinase C zeta/nuclear factor kappaB-regulated signaling in rat hepatic stellate cells. Hepatology 2009, 49, 887–900. [Google Scholar] [CrossRef]
- Harris, P.S.; Roy, S.R.; Coughlan, C.; Orlicky, D.J.; Liang, Y.; Shearn, C.T.; Roede, J.R.; Fritz, K.S. Chronic ethanol consumption induces mitochondrial protein acetylation and oxidative stress in the kidney. Redox Biol. 2015, 6, 33–40. [Google Scholar] [CrossRef]
- Song, N.; Thaiss, F.; Guo, L. NFκB and Kidney Injury. Front. Immunol. 2019, 10, 815. [Google Scholar] [CrossRef]
- Jha, A.K.; Gairola, S.; Kundu, S.; Doye, P.; Syed, A.M.; Ram, C.; Murty, U.S.; Naidu, V.G.M.; Sahu, B.D. Toll-like receptor 4: An attractive therapeutic target for acute kidney injury. Life Sci. 2021, 271, 119155. [Google Scholar] [CrossRef]
- Liu, M.; Zen, K. Toll-like receptors regulate the development and progression of renal diseases. Kidney Dis. 2021, 7, 14–23. [Google Scholar] [CrossRef]
- Zhang, B.; Ramesh, G.; Uematsu, S.; Akira, S.; Reeves, W.B. TLR4 signaling mediates inflammation and tissue injury in nephrotoxicity. J. Am. Soc. Nephrol. 2008, 19, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yan, L.; Zhu, Q.; Shao, F. Puerarin attenuates cisplatin-induced rat nephrotoxicity: The involvement of TLR4/NF-κB signaling pathway. PLoS ONE 2017, 12, e0171612. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Miao, L.; Yu, H.; Han, Z.; Sun, H. Ethanol extract of illicium henryi attenuates LPS-induced acute kidney injury in mice via regulating inflammation and oxi-dative stress. Nutrients 2019, 11, 1412. [Google Scholar] [CrossRef]
- Song, J.; Yu, J.; Prayogo, G.W.; Cao, W.; Wu, Y.; Jia, Z.; Zhang, A. Understanding kidney injury molecule 1: A novel immune factor in kidney pathophysiology. Am. J. Transl. Res. 2019, 11, 1219–1229. [Google Scholar] [PubMed]
- Gardiner, L.; Akintola, A.; Chen, G.; Catania, J.M.; Vaidya, V.; Burghardt, R.C.; Bonventre, J.V.; Trzeciakowski, J.; Parrish, A.R. Structural equation modeling highlights the potential of Kim-1 as a biomarker for chronic kidney disease. Am. J. Nephrol. 2012, 35, 152–163. [Google Scholar] [CrossRef]
- Tuttle, K.R.; Alicic, R.Z. Glycemic variability and KIM-1-induced inflammation in the diabetic kidney. Diabetes 2021, 70, 1617–1619. [Google Scholar] [CrossRef]
- Tian, L.; Shao, X.; Xie, Y.; Wang, Q.; Che, X.; Zhang, M.; Xu, W.; Xu, Y.; Mou, S.; Ni, Z. Kidney injury molecule-1 is elevated in nephropathy and mediates macrophage activation via the mapk signalling pathway. Cell. Physiol. Biochem. 2017, 41, 769–783. [Google Scholar] [CrossRef]
- Van Diepen, J.A.; Jansen, P.A.; Ballak, D.B.; Hijmans, A.; Rutjes, F.P.; Tack, C.J.; Netea, M.G.; Schalkwijk, J.; Stienstra, R. Genetic and pharmacological inhibition of vanin-1 activity in animal models of type 2 diabetes. Sci. Rep. 2016, 6, 21906. [Google Scholar] [CrossRef]
- Boersma, Y.L.; Newman, J.; Adams, T.E.; Cowieson, N.; Krippner, G.; Bozaoglu, K.; Peat, T.S. The structure of vanin 1: A key enzyme linking metabolic disease and inflammation. Acta Crystallogr. D Biol. Crystallogr. 2014, 70, 3320–3329. [Google Scholar] [CrossRef] [PubMed]
- Bartucci, R.; Salvati, A.; Olinga, P.; Boersma, Y.L. Vanin 1: Its physiological function and role in diseases. Int. J. Mol. Sci. 2019, 20, 3891. [Google Scholar] [CrossRef]
- Hong, F.; Wu, N.; Ge, Y.; Zhou, Y.; Shen, T.; Qiang, Q.; Zhang, Q.; Chen, M.; Wang, Y.; Wang, L.; et al. Nanosized titanium dioxide resulted in the activation of TGF-β/Smads/p38MAPK pathway in renal inflammation and fibration of mice. J. Biomed. Mater. Res. A 2016, 104, 1452–1461. [Google Scholar] [CrossRef] [PubMed]
- Eleftheriadis, T.; Pissas, G.; Liakopoulos, V.; Stefanidis, I. Cytochrome c as a potentially clinical useful marker of mitochondrial and cellular damage. Front. Immunol. 2016, 7, 279. [Google Scholar] [CrossRef] [PubMed]
- Small, D.M.; Gobe, G.C. Cytochrome c: Potential as a noninvasive biomarker of drug-induced acute kidney injury. Expert Opin. Drug Metab. Toxicol. 2012, 8, 655–664. [Google Scholar] [CrossRef]
- Wenzel, T.J.; Bajwa, E.; Klegeris, A. Cytochrome c can be released into extracellular space and modulate functions of human astrocytes in a toll-like receptor 4-dependent manner. Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 129400. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Liu, X.; Zhu, X.; Qu, Z.; Gong, Z.; Li, J.; Xiao, L.; Yang, Y.; Liu, H.; Sun, L.; et al. The role of TLR4 on PGC-1α-mediated oxidative stress in tubular cell in diabetic kidney disease. Oxid. Med. Cell. Longev. 2018, 2018, 6296802. [Google Scholar] [CrossRef]
- Antushevich, A.E.; Antushevich, A.E.; Grebenyuk, A.N.; Khalyutin, D.A.; Yartseva, A.A. Experimental modeling of alcohol-induced liver cirrhosis in rats. Bull. Exp. Biol. Med. 2018, 164, 404–407. [Google Scholar] [CrossRef]
- Fields, C.T.; Chassaing, B.; Castillo-Ruiz, A.; Osan, R.; Gewirtz, A.T.; de Vries, G.J. Effects of gut-derived endotoxin on anxiety-like and repetitive behaviors in male and female mice. Biol. Sex Differ. 2018, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Rezaee-Khorasany, A.; Razavi, B.M.; Taghiabadi, E.; Tabatabaei Yazdi, A.; Hosseinzadeh, H. Effect of crocin, an active saffron constituent, on ethanol toxicity in the rat: Histopathological and biochemical studies. Iran. J. Basic Med. Sci. 2020, 23, 51–62. [Google Scholar] [CrossRef]
- Najafi, M.; Shirazi, A.; Motevaseli, E.; Geraily, G.; Amini, P.; Shabeeb, D.; Eleojo Musa, A. Evaluating the expression of NOX2 and NOX4 signaling pathways in rats’ lung tissues following local chest irradiation; Modulatory effect of melatonin. Int. J. Mol. Cell. Med. 2018, 7, 220–225. [Google Scholar] [CrossRef]
- Peyssonnaux, C.; Zinkernagel, A.S.; Schuepbach, R.A.; Rankin, E.; Vaulont, S.; Haase, V.H.; Nizet, V.; Johnson, R.S. Regulation of iron homeostasis by the hypoxia-inducible transcription factors (HIFs). J. Clin. Investig. 2007, 117, 1926–1932. [Google Scholar] [CrossRef]
- Mehanna, E.T.; El-Sayed, N.M.; Ibrahim, A.K.; Ahmed, S.A.; Abo-Elmatty, D.M. Isolated compounds from Cuscuta pedicellata ameliorate oxidative stress and upregulate expression of some energy regulatory genes in high fat diet induced obesity in rats. Biomed. Pharmacother. 2018, 108, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Goodman, Z.D. Grading and staging systems for inflammation and fibrosis in chronic liver diseases. J. Hepatol. 2007, 47, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Terada, Y.; Inoue, K.; Matsumoto, T.; Ishihara, M.; Hamada, K.; Shimamura, Y.; Ogata, K.; Inoue, K.; Taniguchi, Y.; Horino, T.; et al. 5-Aminolevulinic acid protects against cisplatin-induced nephrotoxicity without compromising the anticancer efficiency of cisplatin in rats in vitro and in vivo. PLoS ONE 2013, 8, e80850. [Google Scholar] [CrossRef] [PubMed]



| ALT (U\L) | AST (U\L) | ALP (U\L) | BUN (mg\dL) | Creatinine (mg\dL) | |
|---|---|---|---|---|---|
| Intact control | 42.0 ± 4.7 | 51.8 ± 5.3 | 53.0 ± 6.9 | 19.6 ± 2.1 | 0.9 ± 0.2 |
| LPS-RS (800 μg/kg) | 45.5 ± 5.2 | 57.7 ± 6.9 | 55.9 ± 6.6 | 21.0 ± 2.5 | 1.1 ± 0.2 |
| Ethanol (3 g/kg) | 105.0 ± 15.5 ab | 225.6 ± 22.2 ab | 185.0 ± 19.1 ab | 27.9 ± 1.9 ab | 1.6 ± 0.3 ab |
| LPS-RS + ethanol (3 g/kg) | 55.0 ± 6.0 abc | 92.3 ± 9.5 abc | 56.6 ± 6.0 c | 26.6 ± 2.4 ab | 1.1 ± 0.3 c |
| Ethanol (5 g/kg) | 162.4 ± 17.1 abcd | 265 ± 27.7 abcd | 248.7 ± 25.8 abcd | 28.3 ± 2.1 ab | 1.9 ± 0.2 abd |
| LPS-RS + ethanol (5 g/kg) | 99.0 ± 11.2 abde | 204.4 ± 21.9 abde | 115.6 ± 12.6 abcde | 26.7 ± 1.3 ab | 1.4 ± 0.3 abe |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mehanna, E.T.; Ali, A.-S.A.; El-Shaarawy, F.; Mesbah, N.M.; Abo-Elmatty, D.M.; Aborehab, N.M. Anti-Oxidant and Anti-Inflammatory Effects of Lipopolysaccharide from Rhodobacter sphaeroides against Ethanol-Induced Liver and Kidney Toxicity in Experimental Rats. Molecules 2021, 26, 7437. https://doi.org/10.3390/molecules26247437
Mehanna ET, Ali A-SA, El-Shaarawy F, Mesbah NM, Abo-Elmatty DM, Aborehab NM. Anti-Oxidant and Anti-Inflammatory Effects of Lipopolysaccharide from Rhodobacter sphaeroides against Ethanol-Induced Liver and Kidney Toxicity in Experimental Rats. Molecules. 2021; 26(24):7437. https://doi.org/10.3390/molecules26247437
Chicago/Turabian StyleMehanna, Eman T., Al-Shimaa A. Ali, Fatma El-Shaarawy, Noha M. Mesbah, Dina M. Abo-Elmatty, and Nora M. Aborehab. 2021. "Anti-Oxidant and Anti-Inflammatory Effects of Lipopolysaccharide from Rhodobacter sphaeroides against Ethanol-Induced Liver and Kidney Toxicity in Experimental Rats" Molecules 26, no. 24: 7437. https://doi.org/10.3390/molecules26247437
APA StyleMehanna, E. T., Ali, A.-S. A., El-Shaarawy, F., Mesbah, N. M., Abo-Elmatty, D. M., & Aborehab, N. M. (2021). Anti-Oxidant and Anti-Inflammatory Effects of Lipopolysaccharide from Rhodobacter sphaeroides against Ethanol-Induced Liver and Kidney Toxicity in Experimental Rats. Molecules, 26(24), 7437. https://doi.org/10.3390/molecules26247437






