A Promiscuous Halogenase for the Derivatization of Flavonoids
Abstract
:1. Introduction
2. Results and Discussion
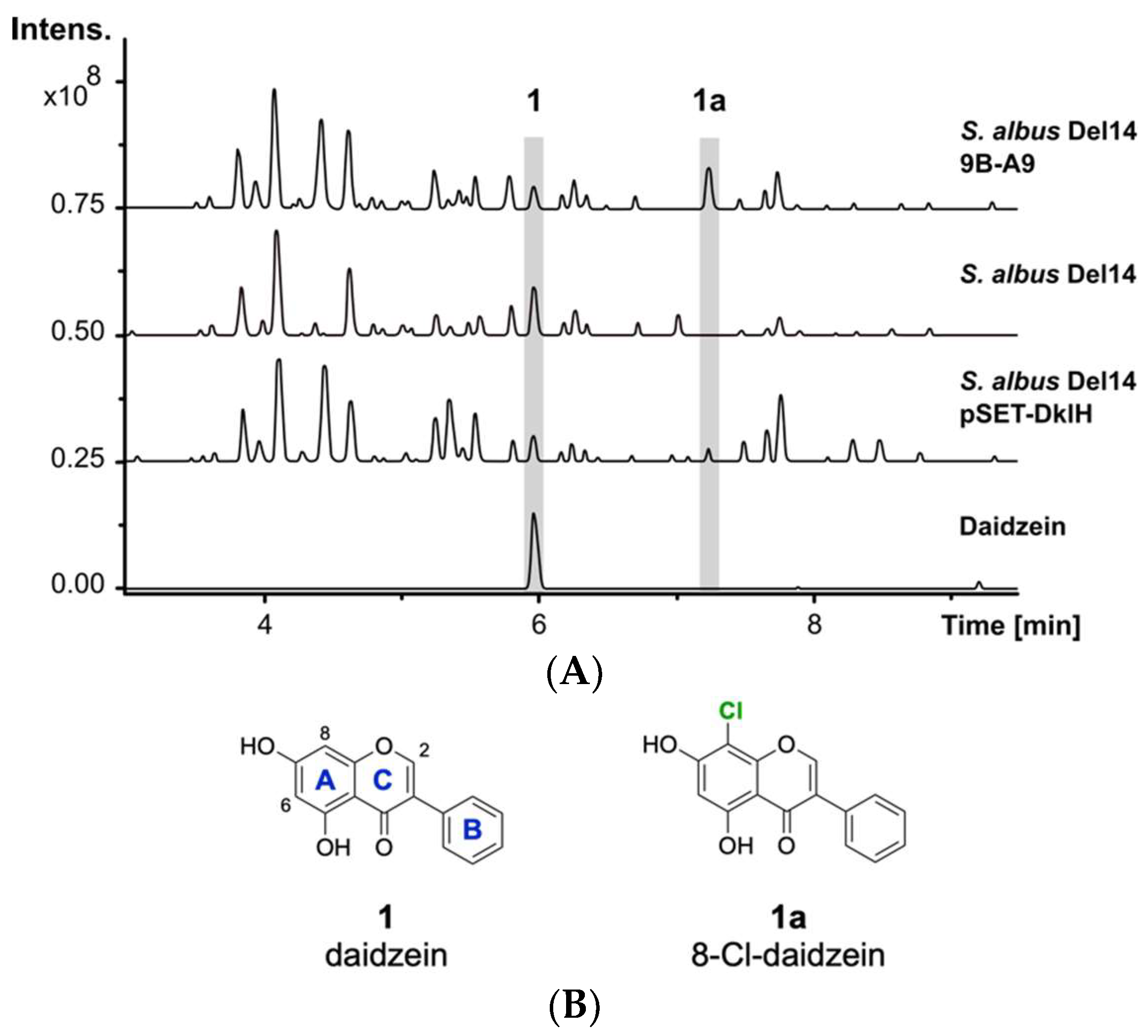
2.1. Identification of the Halogenase DklH through Heterologous Expression
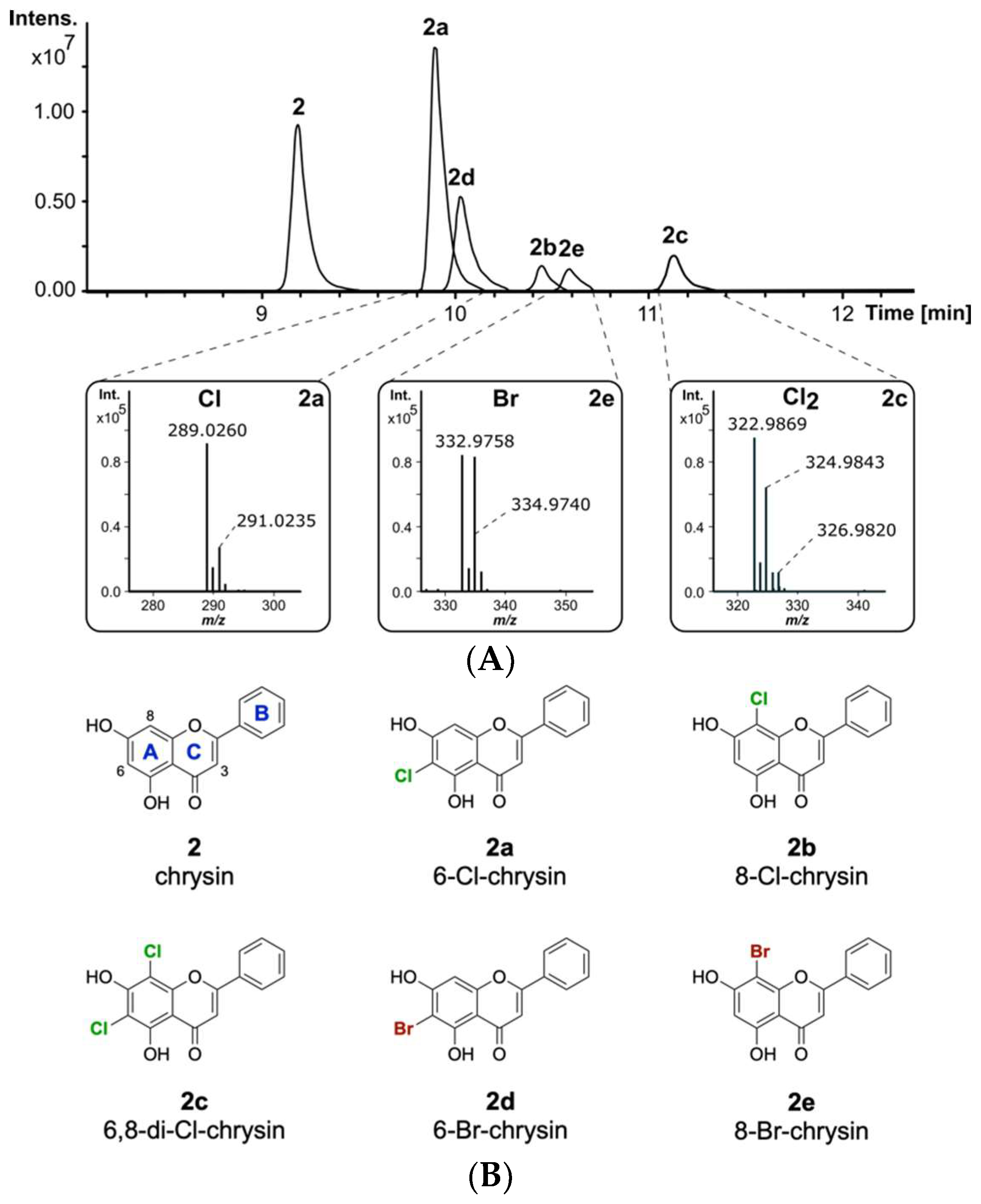
2.2. Halogenation Pattern of the Flavone Chrysin
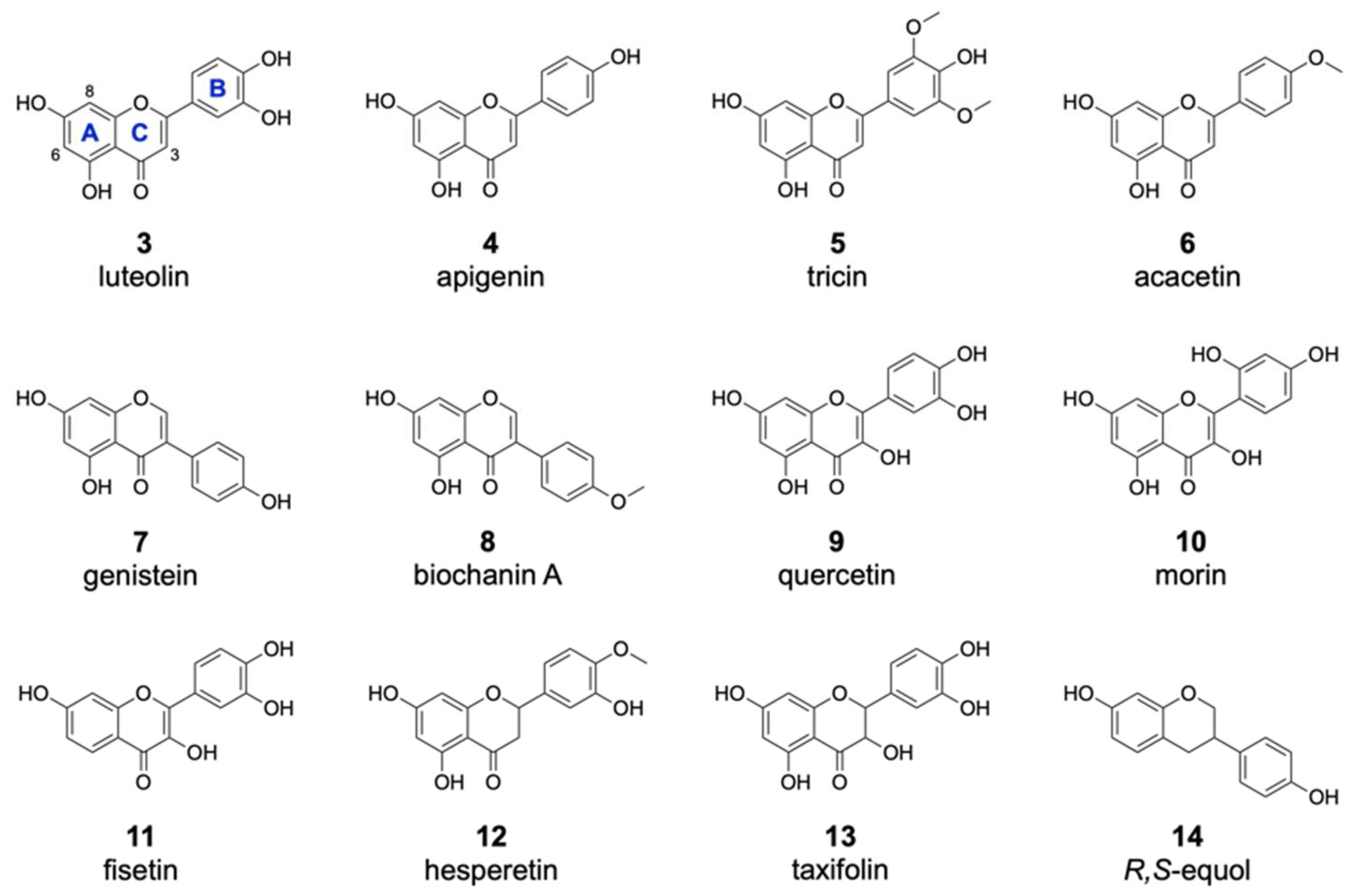
2.3. Analysis of the Substrate Specificity of the Halogenase DklH
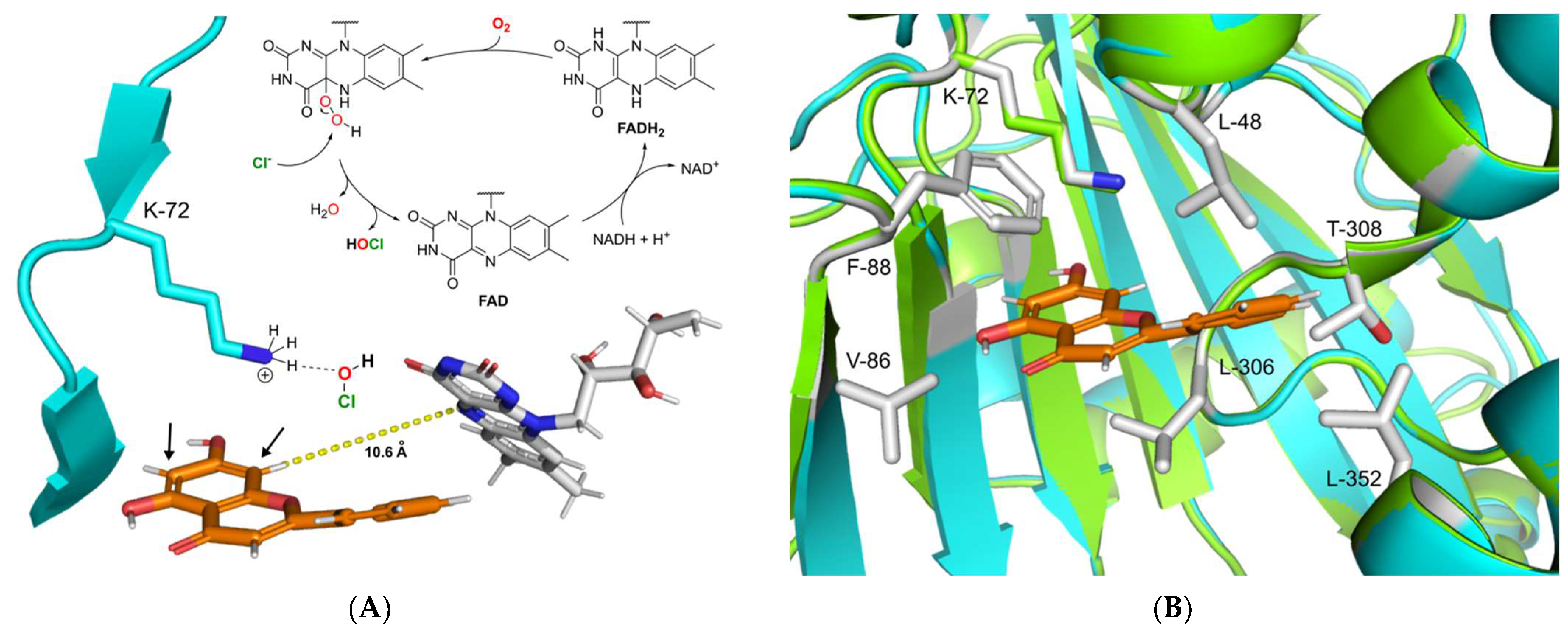
2.4. Proposed Mechanism of DklH and Docking Analysis of the Substrate Chrysin
3. Materials and Methods
3.1. General Experimental Procedures
3.2. DNA Isolation and Manipulation
3.3. Cultivation, Feeding Experiment and Metabolite Extraction
3.4. Mass Spectrometry (MS) and Metabolite Analysis
3.5. Purification
3.6. Nuclear Magnetic Resonance (NMR) Spectroscopy
3.7. Bioinformatics Analysis and Homology Modelling
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Liu, J.; Volk, K.J.; Lee, M.S.; Pucci, M.; Handwerger, S. Binding Studies of Vancomycin to the Cytoplasmic Peptidoglycan Precursors by Affinity Capillary Electrophoresis. Anal. Chem. 1994, 66, 2412–2416. [Google Scholar] [CrossRef] [PubMed]
- Spížek, J.; Řezanka, T. Lincomycin, Clindamycin and Their Applications. Appl. Microbiol. Biot. 2004, 64, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Sathya, A.; Prabhu, T.; Ramalingam, S. Structural, Biological and Pharmaceutical Importance of Antibiotic Agent Chloramphenicol. Heliyon 2020, 6, e03433. [Google Scholar] [CrossRef] [PubMed]
- Kasanah, N.; Triyanto, T. Bioactivities of Halometabolites from Marine Actinobacteria. Biomolecules 2019, 9, 225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pommerehne, K.; Walisko, J.; Ebersbach, A.; Krull, R. The Antitumor Antibiotic Rebeccamycin—Challenges and Advanced Approaches in Production Processes. Appl. Microbiol. Biot. 2019, 103, 3627–3636. [Google Scholar] [CrossRef]
- Hernandes, M.Z.; Cavalcanti, S.M.T.; Moreira, D.R.M.; Junior, W.F.d.A.; Leite, A.C.L. Halogen Atoms in the Modern Medicinal Chemistry: Hints for the Drug Design. Curr. Drug. Targets 2010, 11, 303–314. [Google Scholar] [CrossRef]
- Jiang, S.; Zhang, L.; Cui, D.; Yao, Z.; Gao, B.; Lin, J.; Wei, D. The Important Role of Halogen Bond in Substrate Selectivity of Enzymatic Catalysis. Sci. Rep.-UK 2016, 6, 34750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Z.; Yang, Z.; Liu, Y.; Lu, Y.; Chen, K.; Zhu, W. Halogen Bond: Its Role beyond Drug–Target Binding Affinity for Drug Discovery and Development. J. Chem. Inf. Model. 2014, 54, 69–78. [Google Scholar] [CrossRef]
- Bernini, R.; Pasqualetti, M.; Provenzano, G.; Tempesta, S. Ecofriendly Synthesis of Halogenated Flavonoids and Evaluation of Their Antifungal Activity. New J. Chem. 2015, 39, 2980–2987. [Google Scholar] [CrossRef]
- Dias, T.A.; Duarte, C.L.; Lima, C.F.; Proença, M.F.; Pereira-Wilson, C. Superior Anticancer Activity of Halogenated Chalcones and Flavonols over the Natural Flavonol Quercetin. Eur. J. Med. Chem. 2013, 65, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Senderowicz, A.M. Flavopiridol: The First Cyclin-Dependent Kinase Inhibitor in Human Clinical Trials. Investig. New Drug 1999, 17, 313–320. [Google Scholar] [CrossRef]
- Deep, A.; Marwaha, R.K.; Marwaha, M.G.; Jyoti; Nandal, R.; Sharma, A.K. Flavopiridol as Cyclin Dependent Kinase (CDK) Inhibitor: A Review. N. J. Chem. 2018, 42, 18500–18507. [Google Scholar] [CrossRef]
- Mendez, L.; Henriquez, G.; Sirimulla, S.; Narayan, M. Looking Back, Looking Forward at Halogen Bonding in Drug Discovery. Molecules 2017, 22, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. The Halogen Bond. Chem. Rev. 2016, 116, 2478–2601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiebel, J.; Chang, A.; Lu, H.; Baxter, M.V.; Tonge, P.J.; Kisker, C. Staphylococcus Aureus FabI: Inhibition, Substrate Recognition, and Potential Implications for In Vivo Essentiality. Structure 2012, 20, 802–813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bamber, A.I.; Neal, T.J. An Assessment of Triclosan Susceptibility in Methicillin-Resistant and Methicillin-Sensitive Staphylococcus Aureus. J. Hosp. Infect. 1999, 41, 107–109. [Google Scholar] [CrossRef]
- Bovicelli, P.; Bernini, R.; Antonioletti, R.; Mincione, E. Selective Halogenation of Flavanones. Tetrahedron Lett. 2002, 43, 5563–5567. [Google Scholar] [CrossRef]
- Podgoršek, A.; Zupan, M.; Iskra, J. Oxidative Halogenation with “Green” Oxidants: Oxygen and Hydrogen Peroxide. Angew. Chem. Int. Ed. 2009, 48, 8424–8450. [Google Scholar] [CrossRef]
- Lu, K.; Chu, J.; Wang, H.; Fu, X.; Quan, D.; Ding, H.; Yao, Q.; Yu, P. Regioselective Iodination of Flavonoids by N-Iodosuccinimide under Neutral Conditions. Tetrahedron Lett. 2013, 54, 6345–6348. [Google Scholar] [CrossRef]
- Glenn, W.S.; Nims, E.; O’Connor, S.E. Reengineering a Tryptophan Halogenase To Preferentially Chlorinate a Direct Alkaloid Precursor. J. Am. Chem. Soc. 2011, 133, 19346–19349. [Google Scholar] [CrossRef]
- Frese, M.; Sewald, N. Enzymatic Halogenation of Tryptophan on a Gram Scale. Angew. Chem. Int. Ed. 2015, 54, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Andorfer, M.C.; Belsare, K.D.; Girlich, A.M.; Lewis, J.C. Aromatic Halogenation by Using Bifunctional Flavin Reductase–Halogenase Fusion Enzymes. Chembiochem 2017, 18, 2099–2103. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A.; van Pelt, S. er Enzyme Immobilisation in Biocatalysis: Why, What and How. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheldon, R.A.; Brady, D. Broadening the Scope of Biocatalysis in Sustainable Organic Synthesis. ChemSusChem 2019, 12, 2859–2881. [Google Scholar] [CrossRef] [PubMed]
- Ludewig, H.; Molyneux, S.; Ferrinho, S.; Guo, K.; Lynch, R.; Gkotsi, D.S.; Goss, R.J. Halogenases: Structures and Functions. Curr. Opin. Struc. Biol. 2020, 65, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Yeh, E.; Blasiak, L.C.; Koglin, A.; Drennan, C.L.; Walsh, C.T. Chlorination by a Long-Lived Intermediate in the Mechanism of Flavin-Dependent Halogenases. Biochemistry 2007, 46, 1284–1292. [Google Scholar] [CrossRef]
- Pée, K.H.V.; Patallo, E.P. Flavin-Dependent Halogenases Involved in Secondary Metabolism in Bacteria. Appl. Microbiol. Biot. 2006, 70, 631–641. [Google Scholar] [CrossRef]
- Bitto, E.; Huang, Y.; Bingman, C.A.; Singh, S.; Thorson, J.S.; Phillips, G.N. The Structure of Flavin-dependent Tryptophan 7-halogenase RebH. Proteins Struct. Funct. Bioinform. 2008, 70, 289–293. [Google Scholar] [CrossRef]
- Menon, B.R.K.; Brandenburger, E.; Sharif, H.H.; Klemstein, U.; Shepherd, S.A.; Greaney, M.F.; Micklefield, J. RadH: A Versatile Halogenase for Integration into Synthetic Pathways. Angew. Chem.-Ger. Ed. 2017, 129, 12003–12007. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Pflanzliche Polyphenole: Chemische Eigenschaften, Biologische Aktivität Und Synthese. Angew. Chem.-Ger. Ed. 2011, 123, 610–646. [Google Scholar] [CrossRef]
- Betts, J.W.; Sharili, A.S.; Phee, L.M.; Wareham, D.W. In Vitro Activity of Epigallocatechin Gallate and Quercetin Alone and in Combination versus Clinical Isolates of Methicillin-Resistant Staphylococcus Aureus. J. Nat. Prod. 2015, 78, 2145–2148. [Google Scholar] [CrossRef]
- Zizkova, P.; Stefek, M.; Rackova, L.; Prnova, M.; Horakova, L. Novel Quercetin Derivatives: From Redox Properties to Promising Treatment of Oxidative Stress Related Diseases. Chem.-Biol. Interact. 2017, 265, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, O.; Fontanes, V.; Raychaudhuri, S.; Loo, R.; Loo, J.; Arumugaswami, V.; Sun, R.; Dasgupta, A.; French, S.W. The Heat Shock Protein Inhibitor Quercetin Attenuates Hepatitis C Virus Production. Hepatology 2009, 50, 1756–1764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cushnie, T.P.T.; Lamb, A.J. Antimicrobial Activity of Flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, J.; Jez, J.M.; Bowman, M.E.; Dixon, R.A.; Noel, J.P. Structure of Chalcone Synthase and the. Molecular Basis of Plant Polyketide Biosynthesis. Nat. Am. Inc. 1999, 6, 775–784. [Google Scholar]
- Myronovskyi, M.; Rosenkränzer, B.; Nadmid, S.; Pujic, P.; Normand, P.; Luzhetskyy, A. Generation of a Cluster-Free Streptomyces Albus Chassis Strains for Improved Heterologous Expression of Secondary Metabolite Clusters. Metab. Eng. 2018, 49, 316–324. [Google Scholar] [CrossRef]
- Buckingham, J. Dictionary of Natural Products; CRC Press/Taylor and Fracis Group: London, UK, 1993. [Google Scholar]
- Khattab, A.I.; Babiker, E.H.; Saeed, H.A. Streptomyces: Isolation, Optimization of Culture Conditions and Extraction of Secondary Metabolites. Int. Curr. Pharm. J. 2016, 5, 27–32. [Google Scholar] [CrossRef] [Green Version]
- Weber, T.; Blin, K.; Duddela, S.; Krug, D.; Kim, H.U.; Bruccoleri, R.; Lee, S.Y.; Fischbach, M.A.; Müller, R.; Wohlleben, W.; et al. AntiSMASH 3.0—A Comprehensive Resource for the Genome Mining of Biosynthetic Gene Clusters. Nucleic Acids Res. 2015, 43, W237–W243. [Google Scholar] [CrossRef] [Green Version]
- Medema, M.H.; Blin, K.; Cimermancic, P.; de Jager, V.; Zakrzewski, P.; Fischbach, M.A.; Weber, T.; Takano, E.; Breitling, R. AntiSMASH: Rapid Identification, Annotation and Analysis of Secondary Metabolite Biosynthesis Gene Clusters in Bacterial and Fungal Genome Sequences. Nucleic Acids Res. 2011, 39, W339–W346. [Google Scholar] [CrossRef]
- Siegl, T.; Tokovenko, B.; Myronovskyi, M.; Luzhetskyy, A. Design, Construction and Characterisation of a Synthetic Promoter Library for Fine-Tuned Gene Expression in Actinomycetes. Metab. Eng. 2013, 19, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, D.; Freitas, M.; Lima, J.L.F.C.; Fernandes, E. Proinflammatory Pathways: The Modulation by Flavonoids. Med. Res. Rev. 2015, 35, 877–936. [Google Scholar] [CrossRef]
- Park, H.; Dao, T.T.; Kim, H.P. Synthesis and Inhibition of PGE2 Production of 6,8-Disubstituted Chrysin Derivatives. Eur. J. Med. Chem. 2005, 40, 943–948. [Google Scholar] [CrossRef]
- Marzec, E.; Świtalska, M.; Winiewska-Szajewska, M.; Wójcik, J.; Wietrzyk, J.; Maciejewska, A.M.; Poznański, J.; Mieczkowski, A. The Halogenation of Natural Flavonoids, Baicalein and Chrysin, Enhances Their Affinity to Human Protein Kinase CK2. IUBMB Life 2020, 72, 1250–1261. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Meng, W.-D.; Xu, Y.-Y.; Cao, J.-G.; Qing, F.-L. Synthesis and Anticancer Effect of Chrysin Derivatives. Bioorg. Med. Chem. Lett. 2003, 13, 881–884. [Google Scholar] [CrossRef]
- Yaipakdee, P.; Robertson, L.W. Enzymatic Halogenation of Flavanones and Flavones. Phytochemistry 2001, 57, 341–347. [Google Scholar] [CrossRef]
- Schnepel, C.; Minges, H.; Frese, M.; Sewald, N. A High-Throughput Fluorescence Assay to Determine the Activity of Tryptophan Halogenases. Angew. Chem. Int. Ed. 2016, 55, 14159–14163. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Zhang, Z.; Wang, Y. PEG (300)–PdCl2 Promoted Efficient and Convenient Suzuki–Miyaura Coupling of Aryl Chlorides with Arylboronic Acids. Tetrahedron 2006, 62, 9359–9364. [Google Scholar] [CrossRef]
- Unversucht, S.; Hollmann, F.; Schmid, A.; van Pée, K. FADH2-Dependence of Tryptophan 7-Halogenase. Adv. Synth. Catal. 2005, 347, 1163–1167. [Google Scholar] [CrossRef]
- Sanchez, C.; Butovich, I.A.; Brana, A.F.; Rohr, J.; Méndez, C.; Salas, J.A. The Biosynthetic Gene Cluster for the Antitumor Rebeccamycin: Characterization and Generation of Indolocarbazole Derivatives. Chem. Biol. 2002, 9, 519–531. [Google Scholar] [CrossRef] [Green Version]
- Latimer, R.; Podzelinska, K.; Soares, A.; Bhattacharya, A.; Vining, L.C.; Jia, Z.; Zechel, D.L. Expression, Purification and Preliminary Diffraction Studies of CmlS. Acta Cryst. Sect. F Struct. Biol. Cryst. Commun. 2009, 65, 260–263. [Google Scholar] [CrossRef]
- Payne, J.T.; Andorfer, M.C.; Lewis, J.C. Regioselective Arene Halogenation Using the FAD-Dependent Halogenase RebH. Angew. Chem. Int. Ed. 2013, 52, 5271–5274. [Google Scholar] [CrossRef] [Green Version]
- Yeh, E.; Cole, L.J.; Barr, E.W.; Bollinger, J.M.; Ballou, D.P.; Walsh, C.T. Flavin Redox Chemistry Precedes Substrate Chlorination during the Reaction of the Flavin-Dependent Halogenase RebH. Biochemistry 2006, 45, 7904–7912. [Google Scholar] [CrossRef]
- Dong, C.; Flecks, S.; Unversucht, S.; Haupt, C.; Pée, K.H.V.; Naismith, J.H. Structural Biology: Tryptophan 7-Halogenase (PrnA) Structure Suggests a Mechanism for Regioselective Chlorination. Science 2005, 309, 2216–2219. [Google Scholar] [CrossRef]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology Modelling of Protein Structures and Complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef] [Green Version]
- Buedenbender, S.; Rachid, S.; Müller, R.; Schulz, G.E. Structure and Action of the Myxobacterial Chondrochloren Halogenase CndH: A New Variant of FAD-Dependent Halogenases. J. Mol. Biol. 2009, 385, 520–530. [Google Scholar] [CrossRef]
- Zhang, Y.; Skolnick, J. TM-Align: A Protein Structure Alignment Algorithm Based on TM-Score. Nucleic Acids Res. 2005, 33, 2302–2309. [Google Scholar] [CrossRef]
- Molecular Operating Enviroment (MOE), 2019.01; Chemical Computing Group ULC: Montreal, QC, Canada, 2021.
- Green, M.R.; Sambrook, J. Molecular Cloning: A Laboratory Manual, 4th ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2012. [Google Scholar]
- Rebets, Y.; Kormanec, J.; Luzhetskyy, A.; Bernaerts, K.; Anné, J. Metagenomics, Methods and Protocols. Methods Mol. Biol. 2016, 1539, 99–144. [Google Scholar] [CrossRef]
- Flett, F.; Mersinias, V.; Smith, C.P. Mersinias, V.; Smith, C.P. High Efficiency Intergeneric Conjugal Transfer of Plasmid DNA from Escherichia coli to Methyl DNA-restricting Streptomycetes. FEMS Microbiol. Lett. 1997, 155, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Kieser, T.; Buttner, M.J.; Carter, K.F.; Hopwood, D.A. Practical Streptomyces Genetics; John Innes Foundation: Norwich, UK, 2000. [Google Scholar]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An Integrated and Extendable Desktop Software Platform for the Organization and Analysis of Sequence Data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Wootton, J.C.; Gertz, E.M.; Agarwala, R.; Morgulis, A.; Schäffer, A.A.; Yu, Y. Protein Database Searches Using Compositionally Adjusted Substitution Matrices. FEBS J. 2005, 272, 5101–5109. [Google Scholar] [CrossRef]
- Notredame, C.; Higgins, D.G.; Heringa, J. T-Coffee: A Novel Method for Fast and Accurate Multiple Sequence Alignment. J. Mol. Biol. 2000, 302, 205–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schrödinger, L.L.C. The PyMOL Molecular Graphics System; Version 1.8; Schrödinger, L.L.C.: New York, NY, USA, 2015. [Google Scholar]
| Subclass | Flavonoid | Halide | Product | Yield * [%] | |
|---|---|---|---|---|---|
| Flavone | Chrysin (2) | Cl− | 6-Chlorochrysin | (2a) | 60.8 |
| 8-Chlorochrysin | (2b) | 4.0 | |||
| 6,8-Dichlorochrysin | (2c) | 10.3 | |||
| Br− | Bromochrysin | (2d/e) | 27.1/3.7 | ||
| Luteolin (3) | Cl− | Chloroluteolin | (3a) | 12.3 | |
| Dichloroluteolin | (3b) | 86.6 | |||
| Br− | Bromoluteolin | (3c/d) | 1.0/7.4 | ||
| Apigenin (4) | Cl− | Chloroapigenin | (4a) | 76.7 | |
| Dichloroapigenin | (4b) | 16.3 | |||
| Br− | Bromoapigenin | (4c/d) | 2.5/21.1 | ||
| Tricin (5) | Cl− | Chlorotricin | (5a) | 62.5 | |
| Br− | Bromotricin | (5b) | 78.8 | ||
| Acacetin (6) | Cl− | Chloroacacetin | (6a) | 30.1 | |
| Dichloroacacetin | (6b) | 40.7 | |||
| Br− | Bromoacacetin | (6c/d) | 9.8/n.d. | ||
| Isoflavone | Daidzein (1) | Cl− | 8-Chlorodaidzein | (1a) | 24.8 |
| Br− | Bromodaidzein | (1b) | 39.7 | ||
| Genistein (7) | Cl− | Chlorogenistein | (7a/b) | 5.9/52.2 | |
| Dichlorogenistein | (7c) | 28.3 | |||
| Br− | n.d. | n.d. | n.d. | ||
| Biochanin A (8) | Cl− | Chlorobiochanin A | (8a/b) | 5.9/22.3 | |
| Dichlorobiochanin A | (8c) | 25.8 | |||
| Br− | Bromobiochanin A | (8d/e) | 3.7/5.1 | ||
| Flavonol | Quercetin (9) | Cl− | Chloroquercetin | (9a) | 35.0 |
| Br− | - | - | - | ||
| Morin (10) | Cl− | Chloromorin | (10a) | 72.2 | |
| Br− | - | - | - | ||
| Fisetin (11) | Cl− | Chlorofisetin | (11a) | 48.2 | |
| Br− | - | - | - | ||
| Flavanone | Hesperetin (12) | Cl− | Chlorohesperetin | (12a/b) | 0.9/46.7 |
| Dichlorohesperetin | (12c) | 43.3 | |||
| Br− | Bromohesperetin | (12d) | 2.4/1.4 | ||
| Flavanonol | Taxifolin (13) | Cl− | Chlorotaxifolin | (13a) | 15.4 |
| Br− | - | - | - | ||
| Isoflavandiol | R,S-Equol (14) | Cl− | R,S-Chloroequol | (14a/b) | 10.7/3.6 |
| Br− | R,S-Bromoequol | (14c/d) | 14.9/10.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolling, D.; Stierhof, M.; Lasch, C.; Myronovskyi, M.; Luzhetskyy, A. A Promiscuous Halogenase for the Derivatization of Flavonoids. Molecules 2021, 26, 6220. https://doi.org/10.3390/molecules26206220
Kolling D, Stierhof M, Lasch C, Myronovskyi M, Luzhetskyy A. A Promiscuous Halogenase for the Derivatization of Flavonoids. Molecules. 2021; 26(20):6220. https://doi.org/10.3390/molecules26206220
Chicago/Turabian StyleKolling, Dominik, Marc Stierhof, Constanze Lasch, Maksym Myronovskyi, and Andriy Luzhetskyy. 2021. "A Promiscuous Halogenase for the Derivatization of Flavonoids" Molecules 26, no. 20: 6220. https://doi.org/10.3390/molecules26206220
APA StyleKolling, D., Stierhof, M., Lasch, C., Myronovskyi, M., & Luzhetskyy, A. (2021). A Promiscuous Halogenase for the Derivatization of Flavonoids. Molecules, 26(20), 6220. https://doi.org/10.3390/molecules26206220






