Supported TiO2 in Ceramic Materials for the Photocatalytic Degradation of Contaminants of Emerging Concern in Liquid Effluents: A Review
Abstract
1. Introduction
2. Contaminants of Emerging Concern in Wastewater
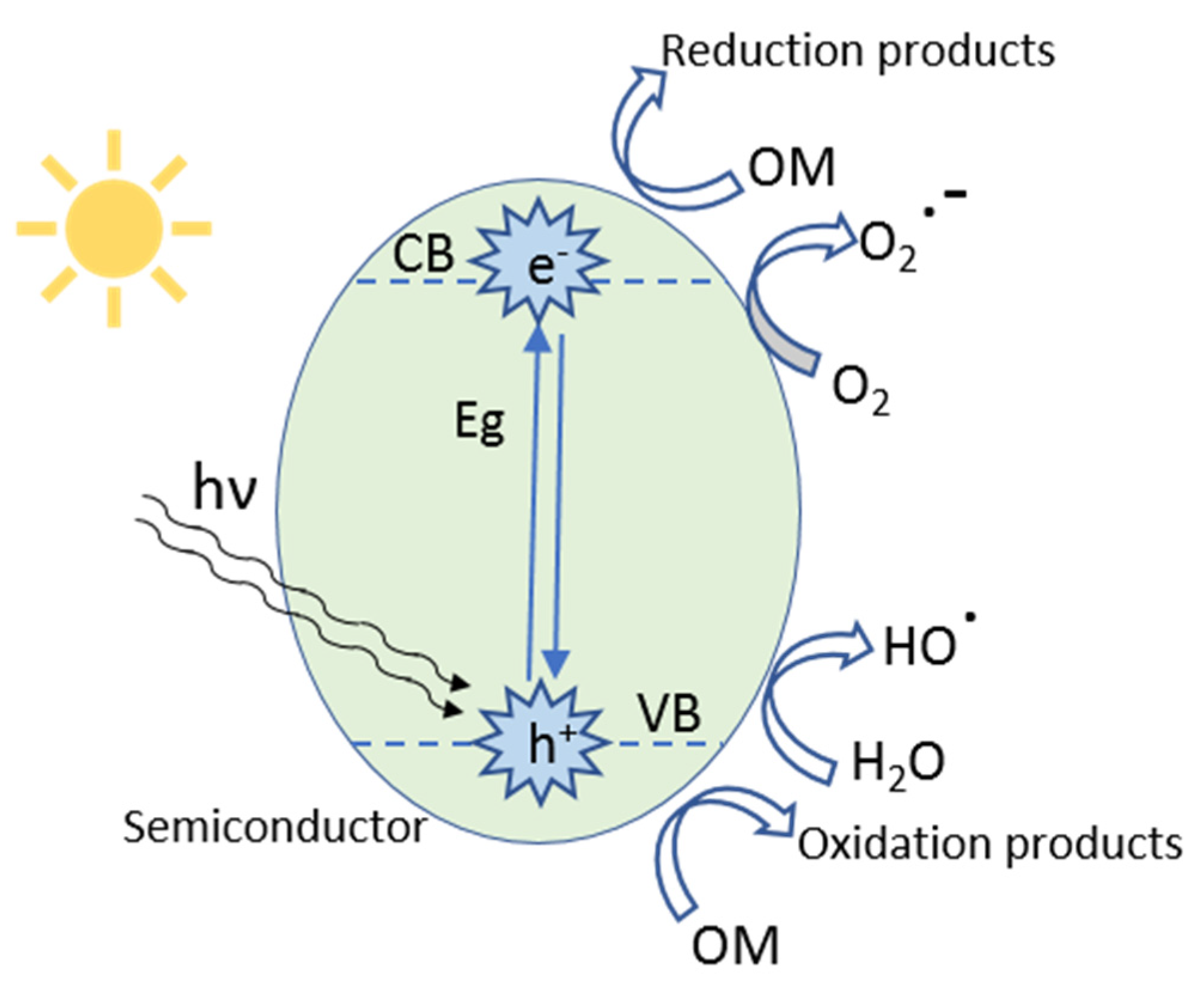
3. Heterogeneous Photocatalysis
3.1. Radiation Source
3.1.1. UV Radiation
3.1.2. Visible Radiation
3.1.3. Sunlight Radiation
3.2. Type of Catalysts
3.2.1. Powder Catalysts
3.2.2. Supported Catalysts
4. Photocatalysts Supported in Ceramic Materials
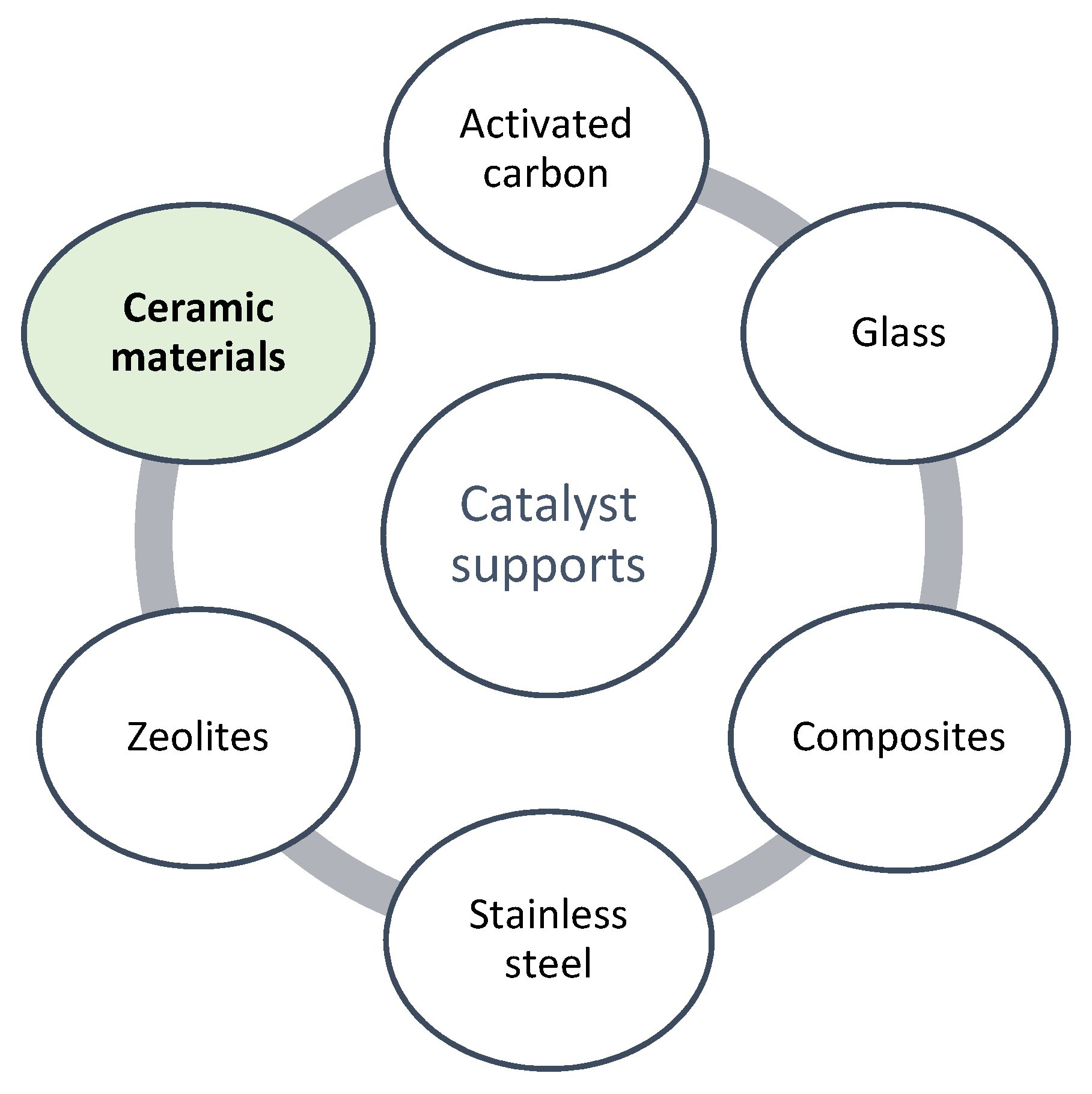
4.1. Ceramic Supports
4.2. Other Alternative Supports
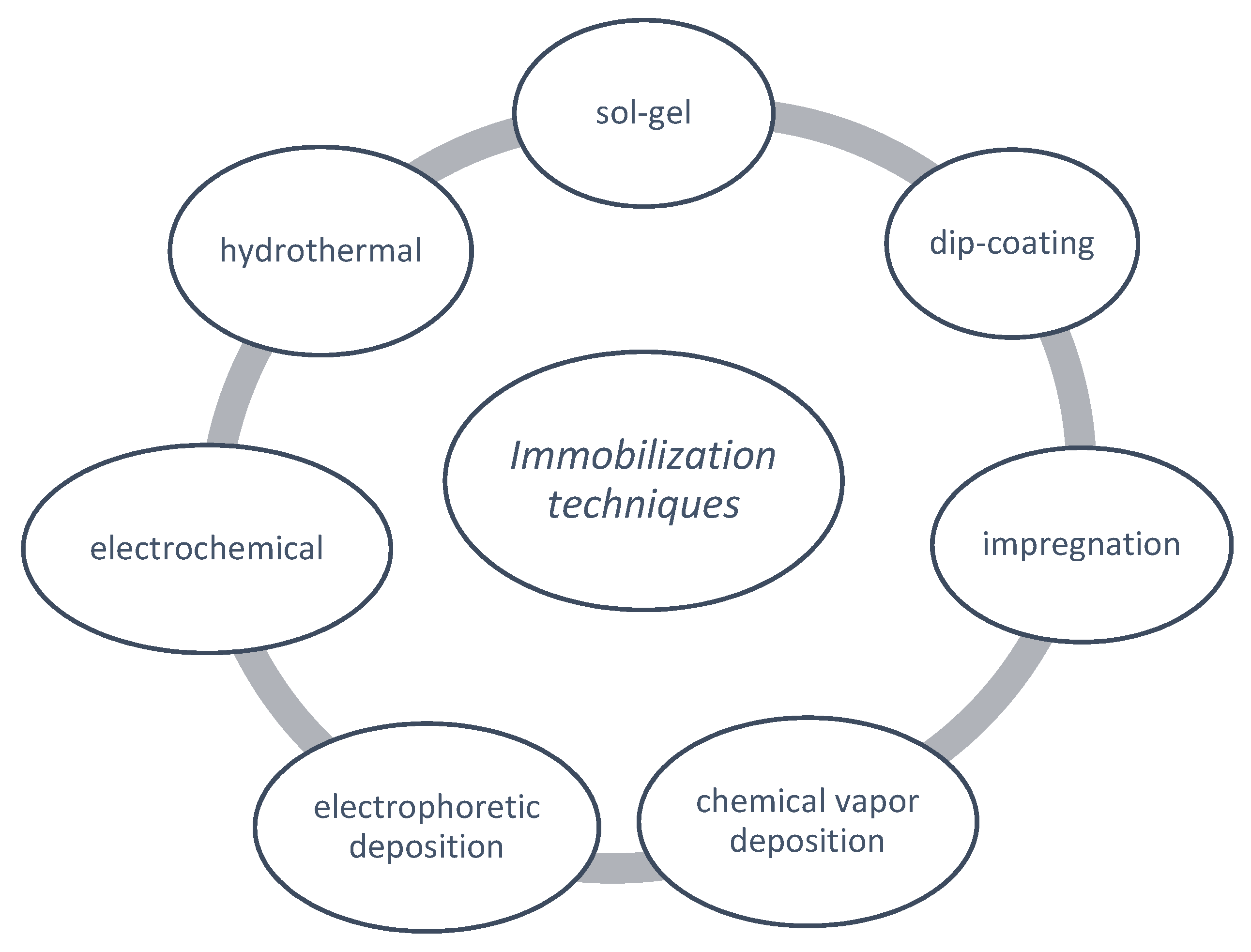
4.3. Immobilization Techniques of TiO2 onto Ceramic Materials
4.4. Catalyst Characterization
5. Photocatalysis with Supported Material
- (i)
- increasing light-harvesting capacity via defect engineering.
- (ii)
- enhancing charge separation via interface engineering.
- (iii)
- accelerating surface reaction.
5.1. Chemical Contaminants of Emerging Concern Removal
| Reference | Support | Incorporation Method | Pollutant | Operating Conditions | Efficiency (%) |
|---|---|---|---|---|---|
| [7] | Leca (doped with Cu) | Sol-gel | 200 ppm of phenol | UV light, catalyst amount (0.5 g L−1); aerated | 61 (2 h) |
| [27] | Lightweight aggregate-photocatalytic ceramsite sand | Vacuum ultrasonic method | 2 µL of benzene | UV light, catalyst amount (4 g L−1) | 100 (3.3 h) |
| [38] | Leca | Impregnation method | 0.05 M of ammonia | UV light, catalyst amount (0.5 g), 750 mL of solution, pH 11 | 85 (5 h) |
| [120] | Laponite | Hydrothermal method | 10 ppm of Bromacil and Alachlor; 5 ppm of Chlorotoluron, Sulfosulfuron, and Imazaquin | UV light as a radiation source, Catalyst amount 1 g L−1 of solution | 60 to 100 (1 h) |
| [135] | Perlite | Dip-coating | 1 mM of phenol | UV light, 11 g L−1 catalyst amount; aerated | 83.3 (4 h) |
| [141] | Porous ceramic | Sol-gel | 5 mg L−1 of Rhodamine B | UV light, catalyst charge 140 mg L−1 | 83 (2 h) |
| [156] | Reticulated Al2O3 ceramics | Dip-coating | 20 mg L−1 of RO16 azo dye (Dystar); | UV-C, catalyst amount (20 g); 500 mL dye solution | 100 (1.25 h) |
| [157] | Sepiolite (Sep) | Sol-gel | 2.0 × 10−5 mol L−1 of eosin dye | UV light, 1.0 g L−1 of the catalyst, neutral pH | 72 (2.5 h) |
| [158] | Ceramic material | Sol-gel | 5 mg L−1 of MB | UV-C, catalyst amount (0.4 g) inject air into the reactor | 60 to 70 (1 h) |
| [159] | Clay beads | Dip-coating | 25 mg L−1 of pesticide Monocrotophos | UV light, catalyst amount (26.8 g), 2 L of solution, pH 5 | 78 (7 h) |
5.2. Disinfection Capacity
5.3. Toxicity Assessment
5.4. Water Recovery
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Anjum, M.; Miandad, R.; Waqas, M.; Gehany, F.; Barakat, M. Remediation of wastewater using various nanomaterials. Arab. J. Chem. 2019, 12, 4897–4919. [Google Scholar] [CrossRef]
- Köck-Schulmeyer, M.; Villagrasa, M.; López de Alda, M.; Céspedes-Sánchez, R.; Ventura, F.; Barceló, D. Occurrence and Behavior of Pesticides in Wastewater Treatment Plants and Their Environmental Impact. Sci. Total Environ. 2013, 458–460, 466–476. [Google Scholar] [CrossRef] [PubMed]
- Vela, N.; Calín, M.; Yáñez-Gascón, M.J.; Garrido, I.; Pérez-Lucas, G.; Fenoll, J.; Navarro, S. Solar reclamation of wastewater effluent polluted with bisphenols, phthatalates and parabens by photocatalytic treatment with TiO2/Na2S2O8 at pilot plant scale. Chemosphere 2018, 212, 95–104. [Google Scholar] [CrossRef]
- Terzić, S.; Senta, I.; Ahel, M.; Gros, M.; Petrović, M.; Barcelo, D.; Müller, J.; Knepper, T.; Martí, I.; Ventura, F.; et al. Occurrence and Fate of Emerging Wastewater Contaminants in Western Balkan Region. Sci. Total Environ. 2008, 399, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R. Application of ozonation for pharmaceuticals and personal care products removal from water. Sci. Total Environ. 2017, 586, 265–283. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Kannan, K. Accumulation profiles of parabens and their metabolites in fish, black bear, and birds, including bald eagles and albatrosses. Environ. Int. 2016, 94, 546–553. [Google Scholar] [CrossRef]
- Sohrabi, S.; Akhlaghian, F. Light expanded clay aggregate (LECA) as a support for TiO2, Fe/TiO2, and Cu/TiO2 nanocrystalline photocatalysts: A comparative study on the structure, morphology, and activity. J. Iran. Chem. Soc. 2016, 13, 1785–1796. [Google Scholar] [CrossRef]
- Ibhadon, A.O.; Fitzpatrick, P. Heterogeneous Photocatalysis: Recent Advances and Applications. Catalysts 2013, 3, 189–218. [Google Scholar] [CrossRef]
- Fendrich, M.A.; Quaranta, A.; Orlandi, M.; Bettonte, M.; Miotello, A. Solar Concentration for Wastewaters Remediation: A Review of Materials and Technologies. Appl. Sci. 2018, 9, 118. [Google Scholar] [CrossRef]
- Jabri, A.H.; Feroz, S. Studies on the Effect of TiO2 Nano Photocatalysis in the Pretreatment of Seawater Reverse Osmosis Desalination. Int. J. Environ. Sci. Dev. 2015, 6, 543. [Google Scholar] [CrossRef]
- Chen, J.; Ollis, D.F.; Rulkens, W.H.; Bruning, H. Photocatalyzed oxidation of alcohols and organochlorides in the presence of native TiO2 and metallized TiO2 suspensions. Part (II): Photocatalytic mechanisms. Water. Res. 1999, 33, 669–676. [Google Scholar] [CrossRef]
- Xuzhuang, Y.; Yang, D.; Huaiyong, Z.; Jiangwen, L.; Martins, W.N.; Frost, R.; Daniel, L.; Yuenian, S. Mesoporous Structure with Size Controllable Anatase Attached on Silicate Layers for Efficient Photocatalysis. J. Phys. Chem. C 2009, 113, 8243–8248. [Google Scholar] [CrossRef]
- Semreen, M.H.; Shanableh, A.; Semerjian, L.; Alniss, H.; Mousa, M.; Bai, X.; Acharya, K. Simultaneous Determination of Pharmaceuticals by Solid-phase Extraction and Liquid Chromatography-Tandem Mass Spectrometry: A Case Study from Sharjah Sewage Treatment Plant. Molecules 2019, 24, 633. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.; Domingues, E.; Gmurek, M.; Quinta-Ferreira, R.M.; Martins, R.C. Advanced oxidation processes for recalcitrant compounds removal comparison with biofiltration by Corbicula fluminea. Energy Rep. 2020, 6, 666–671. [Google Scholar] [CrossRef]
- Gavrilescu, M.; Demnerová, K.; Aamand, J.; Agathos, S.; Fava, F. Emerging pollutants in the environment: Present and future challenges in biomonitoring, ecological risks and bioremediation. New. Biotechnol. 2015, 32, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Juliano, C.; Magrini, G.A. Cosmetic Ingredients as Emerging Pollutants of Environmental and Health Concern. A Mini-Review. Cosmetics 2017, 4, 11. [Google Scholar] [CrossRef]
- Baalbaki, Z.; Sultana, T.; Metcalfe, C.; Yargeau, V. Estimating removals of contaminants of emerging concern from wastewater treatment plants: The critical role of wastewater hydrodynamics. Chemosphere 2017, 178, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Alygizakis, N.A.; Urík, J.; Beretsou, V.; Kampouris, I.; Galani, A.; Oswaldova, M.; Berendonk, T.; Oswald, P.; Thomaidis, N.S.; Slobodnik, J.; et al. Evaluation of chemical and biological contaminants of emerging concern in treated wastewater intended for agricultural reuse. Environ. Int. 2020, 138, 105597. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.; Sun, J.; Fang, M.; Luo, S.; Tian, Y.; Dong, P.; Xu, B.; Zheng, C. Occurrence of antibiotics in the main rivers of Shenzhen, China: Association with antibiotic resistance genes and microbial community. Sci. Total Environ. 2019, 653, 334–341. [Google Scholar] [CrossRef]
- Chaves, M.D.J.S.; Barbosa, S.C.; Malinowski, M.D.M.; Volpato, D.; Castro, Í.B.; Franco, T.C.R.D.S.; Primel, E.G. Pharmaceuticals and Personal Care Products in a Brazilian Wetland of International Importance: Occurrence and Environmental Risk Assessment. Sci. Total Environ. 2020, 734, 139374. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.W.-P.; Lin, Y.-C.; Wang, Y.-H.; Guo, Y.L.; Lin, A.Y.-C. Occurrence of Emerging Contaminants in Aquaculture Waters: Cross-Contamination between Aquaculture Systems and Surrounding Waters. Water Air Soil Pollut. 2018, 229, 249. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, B.; Cagnetta, G.; Duan, L.; Yang, J.; Deng, S.; Huang, J.; Wang, Y.; Yu, G. Typical pharmaceuticals in major WWTPs in Beijing, China: Occurrence, load pattern and calculation reliability. Water Res. 2018, 140, 291–300. [Google Scholar] [CrossRef]
- Fatta-Kassinos, D.; Michael, C. Wastewater reuse applications and contaminants of emerging concern. Environ. Sci. Pollut. Res. 2013, 20, 3493–3495. [Google Scholar] [CrossRef] [PubMed]
- Heberling, M.T.; Nietch, C.T.; Thurston, H.W.; Elovitz, M.S.; Birkenhauer, K.H.; Panguluri, S.; Ramakrishnan, B.; Heiser, E.; Neyer, T. Comparing drinking water treatment costs to source water protection costs using time series analysis. Water Resour. Res. 2015, 51, 8741–8756. [Google Scholar] [CrossRef]
- Qureshi, T.; Bakhshpour, M.; Çetin, K.; Topçu, A.A.; Denizli, A. Wastewater Treatment. In Photocatalysts in Advanced Oxidation Processes for Wastewater Treatment; Fosso-Kankeu, E., Pandey, S., Ray, S.S., Eds.; John Wiley & Sons: Hoboken, NJ, USA; Scrivener Publishing: Beverly, MA, USA, 2020; pp. 33–64. [Google Scholar]
- Rueda-Marquez, J.J.; Levchuk, I.; Fernández Ibañez, P.; Sillanpää, M. A Critical Review on Application of Photocatalysis for Toxicity Reduction of Real Wastewaters. J. Clean. Prod. 2020, 258, 120694. [Google Scholar] [CrossRef]
- Zhao, D.; Wang, F.; Liu, P.; Yang, L.; Hu, S.; Zhang, W. Preparation, Physicochemical Properties, and Long-Term Performance of Photocatalytic Ceramsite Sand in Cementitious Materials. Appl. Sci. 2017, 7, 828. [Google Scholar] [CrossRef]
- Puma, L.G.; Bono, A.; Krishnaiah, D.; Collin, J.G. Preparation of Titanium Dioxide Photocatalyst Loaded onto Activated Carbon Support Using Chemical Vapor Deposition: A Review Paper. J. Hazard. Mater. 2008, 157, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.; Falaras, P.; Kontos, A.G.; Dunlop, P.; Hamilton, J.; Byrne, J.; O’Shea, K.; et al. A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl. Catal. B Environ. 2012, 125, 331–349. [Google Scholar] [CrossRef]
- Vargas, R.; Carvajal, D.; Madriz, L.; Scharifker, B.R. Chemical Kinetics in Solar to Chemical Energy Conversion: The Photoe-lectrochemical Oxygen Transfer Reaction. Energy Rep. 2020, 6, 2–12. [Google Scholar] [CrossRef]
- Van Gerven, T.; Mul, G.; Moulijn, J.; Stankiewicz, A.M. A review of intensification of photocatalytic processes. Chem. Eng. Process. Process. Intensif. 2007, 46, 781–789. [Google Scholar] [CrossRef]
- Silva, G.C.S.C. Synthesis, Spectroscopy and Characterization of Titanium Dioxide Based Photocatalysts for the Degradative Oxidation of Organic Pollutants. Ph.D. Thesis, University of Porto, Porto, Portugal, 2008. [Google Scholar]
- Koe, W.S.; Lee, J.W.; Chong, W.C.; Pang, Y.L.; Sim, L.C. An overview of photocatalytic degradation: Photocatalysts, mechanisms, and development of photocatalytic membrane. Environ. Sci. Pollut. Res. 2019, 27, 2522–2565. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, B.; Prieto-Mahaney, O.; Li, D.; Abe, R. What is Degussa (Evonik) P25? Crystalline composition analysis, reconstruction from isolated pure particles and photocatalytic activity test. J. Photochem. Photobiol. A Chem. 2010, 216, 179–182. [Google Scholar] [CrossRef]
- Beltrán, F.J.; Rey, A. Solar or UVA-Visible Photocatalytic Ozonation of Water Contaminants. Molecules 2017, 22, 1177. [Google Scholar] [CrossRef] [PubMed]
- Hanson, S.; Tikalsky, P. Influence of Ultraviolet Light on Photocatalytic TiO2 Materials. J. Mater. Civ. Eng. 2013, 25, 893–898. [Google Scholar] [CrossRef]
- Alaton, I.A.; Balcioglu, I.A.; Bahnemann, D.W. Advanced oxidation of a reactive dyebath effluent: Comparison of O3, H2O2/UV-C and TiO2/UV-A processes. Water Res. 2002, 36, 1143–1154. [Google Scholar] [CrossRef]
- Zendehzaban, M.; Sharifnia, S.; Hosseini, S.N. Photocatalytic degradation of ammonia by light expanded clay aggregate (LECA)-coating of TiO2 nanoparticles. Korean J. Chem. Eng. 2013, 30, 574–579. [Google Scholar] [CrossRef]
- Mohammadi, Z.; Sharifnia, S.; Shavisi, Y. Photocatalytic degradation of aqueous ammonia by using TiO2-ZnO/LECA hybrid photocatalyst. Mater. Chem. Phys. 2016, 184, 110–117. [Google Scholar] [CrossRef]
- Würtele, M.; Kolbe, T.; Lipsz, M.; Külberg, A.; Weyers, M.; Kneissl, M.; Jekel, M. Application of Gan-Based Ultraviolet-C Light Emitting Diodes–UV LEDs–for Water Disinfection. Water Res. 2011, 45, 1481–1489. [Google Scholar] [CrossRef]
- Chevremont, A.-C.; Farnet, A.-M.; Coulomb, B.; Boudenne, J.-L. Effect of coupled UV-A and UV-C LEDs on both microbiological and chemical pollution of urban wastewaters. Sci. Total Environ. 2012, 426, 304–310. [Google Scholar] [CrossRef]
- Gomes, J.F.; Leal, I.; Bednarczyk, K.; Gmurek, M.; Stelmachowski, M.; Zaleska-Medynska, A.; Quinta-Ferreira, M.E.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R.C. Detoxification of parabens using UV-A enhanced by noble metals—TiO2 supported catalysts. J. Environ. Chem. Eng. 2017, 5, 3065–3074. [Google Scholar] [CrossRef]
- Ferreira, L.C.; Fernandes, J.R.A.; Chueca, J.J.R.; Peres, J.A.; Lucas, M.S.; Tavares, P.B. Photocatalytic degradation of an agro-industrial wastewater model compound using a UV LEDs system: Kinetic study. J. Environ. Manag. 2020, 269, 110740. [Google Scholar] [CrossRef]
- Xiong, P.; Hu, J. Decomposition of acetaminophen (Ace) using TiO2/UVA/LED system. Catal. Today 2017, 282, 48–56. [Google Scholar] [CrossRef]
- Jallouli, N.; Elghniji, K.; Hentati, O.; Ribeiro, A.R.; Silva, A.; Ksibi, M. UV and solar photo-degradation of naproxen: TiO2 catalyst effect, reaction kinetics, products identification and toxicity assessment. J. Hazard. Mater. 2016, 304, 329–336. [Google Scholar] [CrossRef]
- Alvarez-Corena, J.R.; Bergendahl, J.A.; Hart, F.L. Advanced oxidation of five contaminants in water by UV/TiO2: Reaction kinetics and byproducts identification. J. Environ. Manag. 2016, 181, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, B.; Anvaripour, B.; Jorfi, S.; Jaafarzadeh, N. Enhanced Photocatalytic Degradation and Mineralization of Furfural Using UVC/TiO2/GAC Composite in Aqueous Solution. Int. J. Photoenergy 2016, 2016, 2782607. [Google Scholar] [CrossRef]
- Mehrabani-Zeinabad, M. Advanced Oxidative Processes for Treatment of Emerging Contaminants in Water. Ph.D. Thesis, University of Calgary, Calgary, AB, Canada, 2016. [Google Scholar]
- McMurray, T.A.; Dunlop, P.S.M.; Byrne, J.A. The photocatalytic degradation of atrazine on nanoparticulate TiO2 films. J. Photochem. Photobiol. A Chem. 2006, 182, 43–51. [Google Scholar] [CrossRef]
- Gomes, J.; Frasson, D.; Quinta-Ferreira, R.M.; Matos, A.; Martins, R.C. Removal of Enteric Pathogens from Real Wastewater Using Single and Catalytic Ozonation. Water 2019, 11, 127. [Google Scholar] [CrossRef]
- Durán-Álvarez, J.C.; Avella, E.; Ramírez-Zamora, R.M.; Zanella, R. Photocatalytic degradation of ciprofloxacin using mono- (Au, Ag and Cu) and bi- (Au–Ag and Au–Cu) metallic nanoparticles supported on TiO2 under UV-C and simulated sunlight. Catal. Today 2016, 266, 175–187. [Google Scholar] [CrossRef]
- Joseph, C.G.; Taufiq-Yap, Y.H.; Puma, G.L.; Sanmugam, K.; Quek, K.S. Photocatalytic degradation of cationic dye simulated wastewater using four radiation sources, UVA, UVB, UVC and solar lamp of identical power output. Desalin. Water Treat. 2015, 57, 7976–7987. [Google Scholar] [CrossRef]
- Čizmić, M.; Ljubas, D.; Rožman, M.; Ašperger, D.; Ćurković, L.; Babić, S. Photocatalytic Degradation of Azithromycin by Nanostructured TiO2 Film: Kinetics, Degradation Products, and Toxicity. Materials 2019, 12, 873. [Google Scholar] [CrossRef] [PubMed]
- Islam, T.; Dominguez, A.; Turley, R.S.; Kim, H.; Sultana, K.A.; Shuvo, M.; Alvarado-Tenorio, B.; Montes, M.O.; Lin, Y.; Gardea-Torresdey, J.; et al. Development of photocatalytic paint based on TiO2 and photopolymer resin for the degradation of organic pollutants in water. Sci. Total Environ. 2020, 704, 135406. [Google Scholar] [CrossRef]
- Varnagiris, S.; Urbonavicius, M.; Sakalauskaite, S.; Daugelavicius, R.; Pranevicius, L.; Lelis, M.; Milcius, D. Floating TiO2 photocatalyst for efficient inactivation of E. coli and decomposition of methylene blue solution. Sci. Total Environ. 2020, 720, 137600. [Google Scholar] [CrossRef]
- Ramírez, J.R.; Arellano, P.A.C.; Varia, C.J.; Martínez, S.S. Visible Light-Induced Photocatalytic Elimination of Organic Pollu-tants by TiO2: A Review. Curr. Org. Chem. 2015, 19, 540–555. [Google Scholar] [CrossRef]
- Martins, R.C.; Domingues, E.; Bosio, M.; Quina, M.J.; Gmurek, M.; Quinta-Ferreira, R.M.; Gomes, J. Effect of Different Radiation Sources and Noble Metal Doped onto TiO2 for Contaminants of Emerging Concern Removal. Water 2019, 11, 894. [Google Scholar] [CrossRef]
- Gusain, R.; Kumar, N.; Ray, S.S. Factors Influencing the Photocatalytic Activity of Photocatalysts in Wastewater Treatment. In Photocatalysts in Advanced Oxidation Processes for Wastewater Treatment; Fosso-Kankeu, E., Pandey, S., Ray, S.S., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2020; pp. 229–270. [Google Scholar]
- Kim, S.; Hwang, A.S.-J.; Choi, W. Visible Light Active Platinum-Ion-Doped TiO2Photocatalyst. J. Phys. Chem. B 2005, 109, 24260–24267. [Google Scholar] [CrossRef]
- Wang, J.; Tafen, D.N.; Lewis, J.P.; Hong, Z.; Manivannan, A.; Zhi, M.; Li, M.; Wu, N. Origin of Photocatalytic Activity of Nitro-gen-Doped TiO2 Nanobelts. J. Am. Chem. Soc. 2009, 131, 12290–12297. [Google Scholar] [CrossRef]
- Lee, H.; Jang, H.S.; Kim, N.Y.; Joo, J.B. Cu-doped TiO2 hollow nanostructures for the enhanced photocatalysis under visible light conditions. J. Ind. Eng. Chem. 2021, 99, 352–363. [Google Scholar] [CrossRef]
- Wang, T.; Li, B.; Wu, L.; Yin, Y.; Jiang, B.; Lou, J. Enhanced performance of TiO2/reduced graphene oxide doped by rare-earth ions for degrading phenol in seawater excited by weak visible light. Adv. Powder Technol. 2019, 30, 1920–1931. [Google Scholar] [CrossRef]
- Wang, Z.M.; Yang, G.; Biswas, P.; Bresser, W.; Boolchand, P. Processing of Iron-Doped Titania Powders in Flame Aerosol Reactors. Powder Technol. 2001, 114, 197–204. [Google Scholar] [CrossRef]
- Zheng, S.; Bai, C.; Gao, R. Preparation and Photocatalytic Property ofTiO2/Diatomite-Based Porous Ceramics Composite Materials. Int. J. Photoenergy 2012, 2012, 264186. [Google Scholar] [CrossRef]
- Lin, L.; Wang, H.; Luo, H.; Xu, P. Enhanced photocatalysis using side-glowing optical fibers coated with Fe-doped TiO2 nanocomposite thin films. J. Photochem. Photobiol. A Chem. 2015, 307–308, 88–98. [Google Scholar] [CrossRef]
- Vega, M.P.B.; Hinojosa-Reyes, M.; Hernández-Ramírez, A.; Mar, J.L.G.; Rodríguez-González, V.; Hinojosa-Reyes, L. Visible light photocatalytic activity of sol–gel Ni-doped TiO2 on p-arsanilic acid degradation. J. Sol Gel Sci. Technol. 2018, 85, 723–731. [Google Scholar] [CrossRef]
- Malato, S.; Blanco, J.; Alarcón, D.C.; Maldonado, M.I.; Fernandez-Ibañez, P.; Gernjak, W. Photocatalytic decontamination and disinfection of water with solar collectors. Catal. Today 2007, 122, 137–149. [Google Scholar] [CrossRef]
- Pirnie, M.; Engineers, C.; Linden, G.K.; Malley, P.J. Ultraviolet Disinfection Guidance Manual for the Final Long Term 2 en-Hanced Surface Water Treatment Rule; EPA 815-R-06-007; EPA-U.S. Environmental Protection Agency Office of Water: Washington, DC, USA, 2006; pp. 1–6.
- Bustos, N.; Cruz-Alcalde, A.; Iriel, A.; Cirelli, A.F.; Sans, C. Sunlight and UVC-254 irradiation induced photodegradation of organophosphorus pesticide dichlorvos in aqueous matrices. Sci. Total Environ. 2019, 649, 592–600. [Google Scholar] [CrossRef]
- Zhang, Q.; Du, R.; Tan, C.; Chen, P.; Yu, G.; Deng, S. Efficient degradation of typical pharmaceuticals in water using a novel TiO2/ONLH nano-photocatalyst under natural sunlight. J. Hazard. Mater. 2021, 403, 123582. [Google Scholar] [CrossRef]
- Karunakaran, C.; Senthilvelan, S. Semiconductor Catalysis of Solar Photooxidation. Curr. Sci. 2005, 6, 962–967. [Google Scholar]
- Pereira, J.H.; Vilar, V.J.; Borges, M.T.; González, O.; Esplugas, S.; Boaventura, R.A.R. Photocatalytic Degradation of Oxytetra-cycline Using TiO2 under Natural and Simulated Solar Radiation. Sol. Energy 2011, 85, 2732–2740. [Google Scholar] [CrossRef]
- Abdel-Maksoud, Y.; Imam, E.; Ramadan, A. TiO2 Solar Photocatalytic Reactor Systems: Selection of Reactor Design for Scale-up and Commercialization—Analytical Review. Catalysts 2016, 6, 138. [Google Scholar] [CrossRef]
- Jamil, T.S.; Ghaly, M.Y.; Fathy, N.A.; Abd el-Halim, T.A.; Österlund, L. Enhancement of TiO2 Behavior on Photocatalytic Oxi-dation of Mo Dye Using TiO2/AC under Visible Irradiation and Sunlight Radiation. Sep. Purif. Technol. 2012, 98, 270–279. [Google Scholar] [CrossRef]
- Rodríguez, S.M.; Gálvez, J.B.; Rubio, M.M.; Ibáñez, P.F.; Padilla, D.A.; Pereira, M.C.; Mendes, J.F.; Oliveira, C.J. Engineering of Solar Photocatalytic Collectors. Sol. Energy 2004, 77, 513–524. [Google Scholar] [CrossRef]
- McLoughlin, O.; Fernandez-Ibañez, P.; Gernjak, W.; Rodríguez, S.M.; Gill, L. Photocatalytic disinfection of water using low cost compound parabolic collectors. Sol. Energy 2004, 77, 625–633. [Google Scholar] [CrossRef]
- Saettone, E.; Paredes-Larroca, F.; Quino, J.; Ponce, S.; Eyzaguirre, R. Ultraviolet Concentration Factor of a Truncated Compound Parabolic Concentrator under Different Weather Conditions. Appl. Sol. Energy 2020, 56, 99–106. [Google Scholar] [CrossRef]
- Malato, S.; Blanco, J.; Vidal, A.; Richter, C. Photocatalysis with solar energy at a pilot-plant scale: An overview. Appl. Catal. B Environ. 2002, 37, 1–15. [Google Scholar] [CrossRef]
- Borowska, E.; Gomes, J.F.; Martins, R.C.; Quinta-Ferreira, R.M.; Horn, H.; Gmurek, M. Solar Photocatalytic Degradation of Sulfamethoxazole by TiO2 Modified with Noble Metals. Catalysts 2019, 9, 500. [Google Scholar] [CrossRef]
- Eufinger, K.; Poelman, D.; Poelman, H.; De Gryse, R.; Marin, G.B. TiO2 Thin Films for Photocatalytic Applications. Thin Solid Film. Process. Appl. 2008, 37, 189–227. [Google Scholar]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Kaur, A.; Umar, A.; Kansal, S.K. Heterogeneous photocatalytic studies of analgesic and non-steroidal anti-inflammatory drugs. Appl. Catal. A Gen. 2016, 510, 134–155. [Google Scholar] [CrossRef]
- Ngoepe, M.N.; Hato, J.M.; Modibane, D.K.; Hintsho-Mbita, C.N. Biogenic Synthesis of Metal Oxide Nanoparticle Semiconductors for Wastewater Treatment. In Photocatalysts in Advanced Oxidation Processes for Wastewater Treatment; Fosso-Kankeu, E., Pandey, S., Ray, S.S., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2020; pp. 3–31. [Google Scholar]
- Qin, X.; Jing, L.; Tian, G.; Qu, Y.; Feng, Y. Enhanced Photocatalytic Activity for Degrading Rhodamine B Solution of Com-mercial Degussa P25 TiO2 and Its Mechanisms. J. Hazard. Mater. 2009, 172, 1168–1174. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, M.; Chowdhury, M.; Parvin, M.S. Potential Nanomaterials and Their Applications in Modern Medicine: An Over-view. ARC J. Cancer Sci. 2016, 2, 25–33. [Google Scholar]
- Karnan, T.; Selvakumar, S.A.S. Biosynthesis of ZnO nanoparticles using rambutan (Nephelium lappaceum L.) peel extract and their photocatalytic activity on methyl orange dye. J. Mol. Struct. 2016, 1125, 358–365. [Google Scholar] [CrossRef]
- Hariharan, C. Photocatalytic degradation of organic contaminants in water by ZnO nanoparticles: Revisited. Appl. Catal. A Gen. 2006, 304, 55–61. [Google Scholar] [CrossRef]
- Varner, K.; Manager, T.O. State of the Science Literature Review: Nano Titanium Dioxide Environmental Matters; Scientific, Technical, Research, Engineering and Modeling Support (STREAMS) Final Report; EPA-U.S. Environmental Protection Agency: Washington, DC, USA, 2010; pp. 14–15.
- Abdel-Maksoud, Y.K.; Imam, E.; Ramadan, A.R. Sand supported TiO2 photocatalyst in a tray photo-reactor for the removal of emerging contaminants in wastewater. Catal. Today 2018, 313, 55–62. [Google Scholar] [CrossRef]
- Neppolian, B.; Choi, H.; Sakthivel, S.; Arabindoo, B.; Murugesan, V. Solar light induced and TiO2 assisted degradation of textile dye reactive blue 4. Chemosphere 2002, 46, 1173–1181. [Google Scholar] [CrossRef]
- Li, Z.X.; Liu, H.; Cheng, F.L.; Tong, J.H. Photocatalytic Oxidation Using a New Catalysts TiO2 Microspheresfor Water and Wastewater Treatment. Environ. Sci. Technol. 2003, 37, 3989–3994. [Google Scholar] [CrossRef] [PubMed]
- Bertolotti, F.; Vivani, A.; Moscheni, D.; Ferri, F.; Cervellino, A.; Masciocchi, N.; Guagliardi, A. Structure, Morphology, and Faceting of TiO2 Photocatalysts by the Debye Scattering Equation Method. The P25 and P90 Cases of Study. Nanomaterials 2020, 10, 743. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Park, S. Review TiO2 photocatalyst for water treatment applications. J. Ind. Eng. Chem. 2013, 19, 1761–1769. [Google Scholar] [CrossRef]
- Sakkas, V.A.; Arabatzis, M.I.; Konstantinou, K.I.; Dimou, D.A.; Albanis, T.A.; Falaras, P. Metolachlor photocatalytic degradation using TiO2 photocatalysts. Appl. Catal. B Environ. 2004, 49, 195–205. [Google Scholar] [CrossRef]
- Yin, S.; Zhang, Q.; Saito, F.; Sato, T. Preparation of Visible Light-Activated Titania Photocatalyst by Mechanochemical Method. Chem. Lett. 2003, 32, 358–359. [Google Scholar] [CrossRef]
- Canle, M.; Pérez, F.I.M.; Santabala, A.J. Photocatalyzed degradation/abatement of endocrine disruptors. Curr. Opin. Green Sustain. Chem. 2017, 6, 101–138. [Google Scholar] [CrossRef]
- Gao, B.; Yap, P.S.; Lim, T.M.; Lim, T. Adsorption-photocatalytic degradation of Acid Red 88 by supported TiO2: Effect of activated carbon support and aqueous anions. Chem. Eng. J. 2011, 171, 1098–1107. [Google Scholar] [CrossRef]
- Mallakpour, S.; Nikkhoo, E. Surface modification of nano-TiO2 with trimellitylimido-amino acid-based diacids for preventing aggregation of nanoparticles. Adv. Powder Technol. 2014, 25, 348–353. [Google Scholar] [CrossRef]
- Helali, S.; López, M.I.P.; Fernandez-Ibañez, P.; Ohtani, B.; Amano, F.; Malato, S.; Guillard, C. Solar photocatalysis: A green technology for E. coli contaminated water disinfection. Effect of concentration and different types of suspended catalyst. J. Photochem. Photobiol. A Chem. 2014, 276, 31–40. [Google Scholar] [CrossRef]
- Herrmann, J.-M. Heterogeneous photocatalysis: Fundamentals and applications to the removal of various types of aqueous pollutants. Catal. Today 1999, 53, 115–129. [Google Scholar] [CrossRef]
- Dong, H.; Zeng, G.; Tang, L.; Fan, C.; Zhang, C.; He, X.; He, Y. Review An overview on limitations of TiO2-based particles for photocatalytic degradation of organic pollutants and the corresponding countermeasures. Water Res. 2015, 79, 128–146. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, S.P.; Dalavi, D.K.; Hunge, Y.M. Disinfection of Water using Supported Titanium Dioxide Photocatalysts. Am. J. Appl. Sci. 2020, 13, 819–826. [Google Scholar] [CrossRef]
- Długosz, M.; Żmudzki, P.; Kwiecień, A.; Szczubiałka, K.; Krzek, J.; Nowakowska, M. Photocatalytic degradation of sulfa-methoxazole in aqueous solution using a floating TiO2-expanded perlite photocatalyst. J. Hazard. Mater. 2015, 298, 146–153. [Google Scholar] [CrossRef]
- Morjène, L.; Tasbihi, M.; Schwarze, M.; Schomäcker, R.; Aloulou, F.; Seffen, M. A composite of clay, cement, and wood as natural support material for the immobilization of commercial titania (P25, P90, PC500, C-TiO2) towards photocatalytic phenol degradation. Water Sci. Technol. 2020, 81, 1882–1893. [Google Scholar] [CrossRef]
- Otitoju, A.T.; Okoye, U.P.; Chen, G.; Li, Y.; Okoye, O.M.; Li, S. Advanced ceramic components: Materials, fabrication, and applications. J. Ind. Eng. Chem. 2020, 85, 34–65. [Google Scholar] [CrossRef]
- Pozzo, R.L.; Baltanás, M.; Cassano, A. Supported titanium oxide as photocatalyst in water decontamination: State of the art. Catal. Today 1997, 39, 219–231. [Google Scholar] [CrossRef]
- Shan, A.Y.; Ghazi, T.I.M.; Rashid, S.A. Immobilisation of titanium dioxide onto supporting materials in heterogeneous photocatalysis: A review. Appl. Catal. A Gen. 2010, 389, 1–8. [Google Scholar] [CrossRef]
- Tototzintle, M.J. Desarrollo de Nuevas Estrategias Basadas en Fotocatálisis Solar para la Regeneración de Aguas de una Industria Agro-Alimentari. Ph.D. Thesis, Universidad de Almería, Almería, Spain, 2015. [Google Scholar]
- Primo, A.; Corma, A.; García, H. Titania supported gold nanoparticles as photocatalyst. Phys. Chem. Chem. Phys. 2011, 13, 886–910. [Google Scholar] [CrossRef] [PubMed]
- Petala, A.; Frontistis, Z.; Antonopoulou, M.; Konstantinou, I.; Kondarides, D.; Mantzavinos, D. Kinetics of ethyl paraben degradation by simulated solar radiation in the presence of N-doped TiO2 catalysts. Water Res. 2015, 81, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Gopinath, K.P.; Madhav, N.V.; Krishnan, A.; Malolan, R.; Rangarajan, G. Present applications of titanium dioxide for the photocatalytic removal of pollutants from water: A review. J. Environ. Manag. 2020, 270, 110906. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, J.; Teixeira, V.; Azevedo, S.; Fernandes, F.; Neves, J. Development of Photocatalytic Ceramic Materials through the Deposition of TiO2 Nanoparticles Layers. J. Nano Res. 2012, 18–19, 165–176. [Google Scholar] [CrossRef]
- Phattepur, H.; Gowrishankar, B.S. Non-linear regression analysis of the kinetics of photocatalytic degradation of phenol using immobilised mesoporous TiO2 nanoparticles on glass beads. Indian Chem. Eng. 2020, 1–14. [Google Scholar] [CrossRef]
- Doremus, R.H. Alumina. In Ceramic and Glass Materials; Shackelford, J.F., Doremus, R.H., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 1–2. [Google Scholar]
- Mustafa, G.; Wyns, K.; Janssens, S.; Buekenhoudt, A.; Meynen, V. Evaluation of the fouling resistance of methyl grafted ceramic membranes for inorganic foulants and co-effects of organic foulants. Sep. Purif. Technol. 2018, 193, 29–37. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Ko, C.-C.; Chen, L.-H.; Huang, C.-T.; Tung, K.-L.; Liao, Y.-C. A simple coating method to prepare superhydrophobic layers on ceramic alumina for vacuum membrane distillation. Sep. Purif. Technol. 2018, 198, 79–86. [Google Scholar] [CrossRef]
- Yaqoob, S.; Ullah, F.; Mehmood, S.; Mahmood, T.; Ullah, M.; Khattak, A.; Zeb, A.M. Effect of Wastewater Treated with TiO2 Nanoparticles on Early Seedling Growth of Zea Mays L. J. Water Reuse Desalin. 2018, 8, 424–431. [Google Scholar] [CrossRef]
- Fahrenholtz, W.G. Clays. In Ceramic and Glass Materials; Shackelford, J.F., Doremus, R.H., Eds.; University of California: Berkeley, CA, USA, 2008; pp. 111–112. [Google Scholar]
- Mishra, A.; Mehta, A.; Basu, S. Clay supported TiO2 nanoparticles for photocatalytic degradation of environmental pollutants: A review. J. Environ. Chem. Eng. 2018, 6, 6088–6107. [Google Scholar] [CrossRef]
- Paul, B.; Martens, N.W.; Frost, L.R. Immobilised anatase on clay mineral particles as a photocatalyst for herbicides degradation. Appl. Clay Sci. 2012, 57, 49–54. [Google Scholar] [CrossRef]
- Szczepanik, B. Photocatalytic degradation of organic contaminants over clay-TiO2 nanocomposites: A review. Appl. Clay Sci. 2017, 141, 227–239. [Google Scholar] [CrossRef]
- Radeka, M.; Markov, S.; Lončar, E.; Rudić, O.; Vučetić, S.; Ranogajec, J. Photocatalytic effects of TiO2 mesoporous coating immobilized on clay roofing tiles. J. Eur. Ceram. Soc. 2014, 34, 127–136. [Google Scholar] [CrossRef]
- Mariquit, E.G.; Kurniawan, W.; Miyauchi, M.; Hinode, H. Effect of Addition of Surfactant to the Surface Hydrophilicity and Photocatalytic Activity of Immobilized Nano-TiO2 Thin Films. J. Chem. Eng. Jpn. 2015, 48, 856–861. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, J.; Parr, J. Preparation and properties of TiO2/fumed silica composite photocatalytic materials. Procedia Eng. 2012, 27, 448–456. [Google Scholar] [CrossRef]
- Bouazizi, A.; Breida, M.; Karim, A.; Achiou, B.; Ouammou, M.; Calvo, J.; Aaddane, A.; Khiat, K.; Younssi, S.A. Development of a new TiO2 ultrafiltration membrane on flat ceramic support made from natural bentonite and micronized phosphate and applied for dye removal. Ceram. Int. 2017, 43, 1479–1487. [Google Scholar] [CrossRef]
- Berger, T.; Regmi, C.; Schäfer, A.; Richards, B. Photocatalytic degradation of organic dye via atomic layer deposited TiO2 on ceramic membranes in single-pass flow-through operation. J. Membr. Sci. 2020, 604, 118015. [Google Scholar] [CrossRef]
- Pinato, K.; Suttiponparnit, K.; Panpa, W.; Jinawath, S.; Kashima, D.P. Photocatalytic activity of TiO2 coated porous silica beads on degradation of cumene hydroperoxide. Int. J. Appl. Ceram. Technol. 2018, 15, 1542–1549. [Google Scholar] [CrossRef]
- Castañeda-Juárez, M.; Martínez-Miranda, V.; Almazán-Sánchez, P.T.; Linares-Hernández, I.; Santoyo-Tepole, F.; Vázquez-Mejía, G. Synthesis of TiO2 catalysts doped with Cu, Fe, and Fe/Cu support on clinoptilolite zeolite by na electro-chemical-treatment method of the degradation of dicoflenac by heterogeneous photocatalysis. J. Photochem. Photobiol. A Chem. 2019, 380, 111834. [Google Scholar] [CrossRef]
- Miranda-García, N.; Suárez, S.; Sanchez, B.; Coronado, J.; Malato, S.; Maldonado, M.I. Photocatalytic degradation of emerging contaminants in municipal wastewater treatment plant effluents using immobilized TiO2 in a solar pilot plant. Appl. Catal. B Environ. 2011, 103, 294–301. [Google Scholar] [CrossRef]
- Binas, V.; Papadaki, D.; Maggos, T.; Katsanaki, A.; Kiriakidis, G. Study of innovative photocatalytic cement based coatings: The effect of supporting materials. Constr. Build. Mater. 2018, 168, 923–930. [Google Scholar] [CrossRef]
- Borges, M.E.; García, D.M.; Hernández, T.; Ruiz-Morales, J.C.; Esparza, P. Supported Photocatalyst for Removal of Emerging Contaminants from Wastewater in a Continuous Packed-Bed Photoreactor Configuration. Catalysts 2015, 5, 77–87. [Google Scholar] [CrossRef]
- Sillanpää, M. Photocatalytic Activities of Antimony: Advanced Water Treatment Advanced Oxidation Processes; Elsevier: Amsterdam, The Netherlands, 2020; pp. 170–173. [Google Scholar]
- Brinker, C.J.; Scherer, G.W. SOL-GEL SCIENCE: The Physics and Chemistry of Sol-Gel Processing; Academic Press Inc.: Boston, MA, USA; San Diego, CA, USA; New York, NY, USA, 1990; pp. 1–10. [Google Scholar]
- Belet, A.; Wolfs, C.; Mahy, J.G.; Poelman, D.; Vreuls, C.; Gillard, N.; Lambert, S.D. Sol-gel Syntheses of Photocatalysts for the Removal of Pharmaceutical Products in Water. Nanomaterials 2019, 9, 126. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.; Borghei, S.M.; Vossoughi, M.; Taghavinia, N. Immobilization of TiO2 on perlite granules for photocatalytic degradation of phenol. Appl. Catal. B Environ. 2007, 74, 53–62. [Google Scholar] [CrossRef]
- Liu, Y.; Tian, J.; Wei, L.; Wang, Q.; Wang, C.; Yang, C. A novel microwave-assisted impregnation method with water as the dispersion medium to synthesize modified g-C3N4/TiO2 heterojunction photocatalysts. Opt. Mater. 2020, 107, 110128. [Google Scholar] [CrossRef]
- Jones, A.C.; Chalker, P.R. Some recent developments in the chemical vapour deposition of electroceramic oxides. J. Phys. D Appl. Phys. 2003, 36, R53–R79. [Google Scholar] [CrossRef]
- Shimosaki, S.; Ogasawara, T.; Nagaoka, S.; Masaki, Y. Method for Forming Metal Oxide Coating Filmand Vapor Deposition Apparatus. U.S. Patent US2007/0054044A1, 8 March 2007. [Google Scholar]
- Fernández, A.; Lassaletta, G.; Jiménez, V.M.; Justo, A.; González-Elipe, A.R.; Herrmann, J.-M.; Tahiri, H.; Ait-Ichou, Y. Preparation and characterization of TiO2 photocatalysts supported on various rigid supports (glass, quartz and stainless steel). Comparative studies of photocatalytic activity in water purification. Appl. Catal. B Environ. 1995, 7, 49–63. [Google Scholar] [CrossRef]
- Schiemann, D.; Alphonse, P.; Taberna, P.L. Synthesis of High Surface Area TiO2 Coatings on Stainless Steel by Electro-phoretic Deposition. J. Mater. Res. 2013, 28, 2023–2030. [Google Scholar] [CrossRef]
- Scharnberg, A.R.A.; Loreto, C.A.; Wermuth, B.T.; Alves, K.A.; Arcaro, S.; Santos, M.A.P.; Rodriguez, L.A.A. Porous ceramic supported TiO2 nanoparticles: Enhanced photocatalytic activity for Rhodamine B degradation. Bol. Soc. Española Cerám. Vidr. 2020, 59, 230–238. [Google Scholar] [CrossRef]
- Guillaume, P.L.A.; Chelaru, A.M.; Visa, M.; Lassine, O. “Titanium Oxide-Clay” as Adsorbent and Photocatalysts for Wastewater Treatment. J. Membr. Sci. Technol. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Matos, M.J.M. Investigação de TiO2 Aplicado em Superfícies Cerâmicas para Exterior—Efeito Self-Cleaning. Master’s Thesis, Universidade de Porto, Porto, Portugal, 2012. [Google Scholar]
- Sciancalepore, C.; Bondioli, F. Durability of SiO2–TiO2 Photocatalytic Coatings on Ceramic Tiles. Int. J. Appl. Ceram. Technol. 2014, 12, 679–684. [Google Scholar] [CrossRef]
- Sun, H.; Wu, H.; Jin, Y.; Lv, Y.; Ju, G.; Chen, L.; Feng, Z.; Hu, Y. Photocatalytic titanium dioxide immobilized on an ultraviolet emitting ceramic substrate for water purification. Mater. Lett. 2019, 240, 100–102. [Google Scholar] [CrossRef]
- Najafidoust, A.; Allahyari, S.; Rahemi, N.; Tasbihi, M. Uniform coating of TiO2 nanoparticles using biotemplates for photocatalytic wastewater treatment. Ceram. Int. 2020, 46, 4707–4719. [Google Scholar] [CrossRef]
- Saleiro, G.; Cardoso, S.; Toledo, R.; Holanda, J. Avaliação Das Fases Cristalinas De Dióxido De Titânio Suportado Em Cerâmica Vermelha. Cerâmica 2010, 56, 162–167. [Google Scholar] [CrossRef]
- Manickam, K.; Muthusamy, V.; Manickam, S.; Senthil, T.; Periyasamy, G.; Shanmugam, S. Effect of annealing temperature on structural, morphological and optical properties of nanocrystalline TiO2 thin films synthesized by sol–gel dip coating method. Mater. Today Proc. 2020, 23, 68–72. [Google Scholar] [CrossRef]
- Bagheri, S.; Shameli, K.; Hamid, S.B.A. Synthesis and Characterization of Anatase Titanium Dioxide Nanoparticles Using Egg White Solution via Sol-Gel Method. J. Chem. 2012, 2013, 1–5. [Google Scholar] [CrossRef]
- Peikertova, P.; Rebilasová, S.; Gröplová, K.; Neuwirthová, L.; Kukutschová, J.; Matějka, V. Raman Study of Clay/TiO2 Composites. In Proceedings of the 3rd International Conference, Brno, Czech Republic, 21–23 September 2011. [Google Scholar]
- Yang, X.; Chen, Z.; Zhao, W.; Liu, C.; Qian, X.; Zhang, M.; Wei, G.; Khan, E.; Ng, Y.H.; Ok, Y.S. Recent advances in photodegradation of antibiotic residues in water. Chem. Eng. J. 2021, 405, 126806. [Google Scholar] [CrossRef]
- Roccaro, P. Treatment processes for municipal wastewater reclamation: The challenges of emerging contamina nts and direct potable reuse. Curr. Opin. Environ. Sci. Health 2018, 2, 46–54. [Google Scholar] [CrossRef]
- Malakootian, M.; Nasiri, A.; Amiri Gharaghani, M.A. Photocatalytic Degradation of Ciprofloxacin Antibiotic by TiO2 Nanoparticles Immobilized on a Glass Plate. Chem. Eng. Commun. 2020, 207, 56–72. [Google Scholar] [CrossRef]
- Belver, C.; Bedia, J.; Rodriguez, J. Zr-doped TiO2 supported on delaminated clay materials for solar photocatalytic treatment of emerging pollutants. J. Hazard. Mater. 2017, 322, 233–242. [Google Scholar] [CrossRef]
- Doorslaer, X.V.; Demeestere, K.; Heynderickx, P.; Van Langenhove, H.; Dewulf, J. UV-A and UV-C induced photolytic and photocatalytic degradation of aqueous ciprofloxacin and moxifloxacin: Reaction kinetics and role of adsorption. Appl. Catal. B Environ. 2011, 101, 540–547. [Google Scholar] [CrossRef]
- Yildiz, T.; Yatmaz, H.C.; Öztürk, K. Anatase TiO2 powder immobilized on reticulated Al2O3 ceramics as a photocatalyst for degradation of RO16 azo dye. Ceram. Int. 2020, 46, 8651–8657. [Google Scholar] [CrossRef]
- De Oliveira, W.V.; Morais, A.Í.S.; Honorio, L.; Trigueiro, P.; Almeida, L.; Garcia, R.R.P.; Viana, B.; Furtini, M.B.; Silva-Filho, E.C.; Osajima, J.A. TiO2 Immobilized on Fibrous Clay as Strategies to Photocatalytic Activity. Mater. Res. 2020, 23, 20190463. [Google Scholar] [CrossRef]
- Peters, M.F.R.; Santos, M.A.P.; Machado, C.T.; Lopez, R.A.D.; Machado, L.Ê.; Rodriguez, L.A.A. Photocatalytic Degradation of Methylene Blue Using TiO2 Supported in Ceramic Material. Eclética Química J. 2018, 43, 26–32. [Google Scholar]
- Sraw, A.; Kaur, T.; Pandey, Y.; Sobti, A.; Wanchoo, R.K.; Toor, A.P. Fixed bed recirculation type photocatalytic reactor with TiO2 immobilized clay beads for the degradation of pesticide polluted water. J. Environ. Chem. Eng. 2018, 6, 7035–7043. [Google Scholar] [CrossRef]
- Liga, M.V.; Maguire-Boyle, S.J.; Jafry, H.R.; Barron, A.R.; Li, Q. Silica Decorated TiO2 for Virus Inactivation in Drinking Water-Simple Synthesis Method and Mechanisms of Enhanced Inactivation Kinetics. Environ. Sci. Technol. 2013, 47, 6463–6470. [Google Scholar] [CrossRef]
- Teodoro, A.; Boncz, M.Á.; Paulo, P.L.; Machulek, A., Jr. Desinfecção De Água Cinza Por Fotocatálise Heterogênea. Eng. Sanit. Ambient. 2017, 22, 1017–1026. [Google Scholar] [CrossRef][Green Version]
- Foster, H.A.; Ditta, I.B.; Varghese, S.; Steele, A. Photocatalytic disinfection using titanium dioxide: Spectrum and mechanism of antimicrobial activity. Appl. Microbiol. Biotechnol. 2011, 90, 1847–1868. [Google Scholar] [CrossRef] [PubMed]
- Sirimahachai, U.; Phongpaichit, S.; Wongnawa, S. Evaluation of Bactericidal Activity of TiO2 Photocatalysts: A Comparative Study of Laboratory-Made and Commercial TiO2 Samples. Songklanakarin J. Sci. Technol. 2009, 31, 517–525. [Google Scholar]
- Devipriya, S.P.; Yesodharan, S.; Yesodharan, E.P. Solar Photocatalytic Removal of Chemical and Bacterial Pollutants from Water Using Pt/TiO2-Coated Ceramic Tiles. Int. J. Photoenergy 2012, 2012, 1–8. [Google Scholar] [CrossRef][Green Version]
- Machado, L.C.; Torchia, C.B.; Lago, R.M. Floating photocatalysts based on TiO2 supported on high surface area exfoliated vermiculite for water decontamination. Catal. Commun. 2006, 7, 538–541. [Google Scholar] [CrossRef]
- Zhao, Y.; Huang, G.; An, C.; Huang, J.; Xin, X.; Chen, X.; Hong, Y.; Song, P. Removal of Escherichia Coli from water using functionalized porous ceramic disk filter coated with Fe/TiO2 nano-composites. J. Water Process. Eng. 2020, 33, 101013. [Google Scholar] [CrossRef]
- Lei, S.; Guo, G.; Xiong, B.; Gong, W.; Mei, G. Disruption of bacterial cells by photocatalysis of montmorillonite supported titanium dioxide. J. Wuhan Univ. Technol. Sci. Ed. 2009, 24, 557–561. [Google Scholar] [CrossRef]
- Zan, L.; Fa, W.; Peng, T.; Gong, Z.-K. Photocatalysis effect of nanometer TiO2 and TiO2-coated ceramic plate on Hepatitis B virus. J. Photochem. Photobiol. B Biol. 2007, 86, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Reddy, L.P.V.; Kavitha, B.; Reddy, K.P.A.; Kim, K.H. TiO2-Based Photocatalytic Disinfection of Microbes in Aqueous Media: A Review. Environ. Res. 2017, 154, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Salih, F. Enhancement of solar inactivation of Escherichia coli by titanium dioxide photocatalytic oxidation. J. Appl. Microbiol. 2002, 92, 920–926. [Google Scholar] [CrossRef]
- Nassef, M.; Matsumoto, S.; Seki, M.; Kang, I.J.; Moroishi, J.; Shimasaki, Y.; Oshima, Y. Pharmaceuticals and Personal Care Products Toxicity to Japanese Medaka Fish (Oryzias latipes). J. Fac. Agric. Kyushu Univ. 2009, 54, 407–411. [Google Scholar] [CrossRef]
- Babu, D.S.; Srivastava, V.; Nidheesh, P.; Kumar, M.S. Detoxification of water and wastewater by advanced oxidation processes. Sci. Total Environ. 2019, 696, 133961. [Google Scholar] [CrossRef]
- Xing, X.; Du, Z.; Zhuang, J.; Wang, D. Removal of ciprofloxacin from water by nitrogen doped TiO2 immobilized on glass spheres: Rapid screening of degradation products. J. Photochem. Photobiol. A Chem. 2018, 359, 23–32. [Google Scholar] [CrossRef]
- Khataee, A.; Fathinia, M.; Joo, S. Simultaneous monitoring of photocatalysis of three pharmaceuticals by immobilized TiO2 nanoparticles: Chemometric assessment, intermediates identification and ecotoxicological evaluation. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 112, 33–45. [Google Scholar] [CrossRef]
- Alfred, M.O.; Omorogie, M.O.; Bodede, O.; Moodley, R.; Ogunlaja, A.; Adeyemi, O.G.; Günter, C.; Taubert, A.; Iermak, I.; Eckert, H. Solar-Active Clay-TiO2 Nanocomposites Prepared Via Biomass Assisted Synthesis: Efficient Removal of Ampicillin, Sulfamethoxazole and Artemether from Water. Chem. Eng. J. 2020, 398, 125544. [Google Scholar] [CrossRef]
- Iervolino, G.; Zammit, I.; Vaiano, V.; Rizzo, L. Limitations and Prospects for Wastewater Treatment by Uv and Visible-Light-Active Heterogeneous Photocatalysis: A Critical Review. Heterog. Photocatal. 2020, 225–264. [Google Scholar]
- Iqbal, M.; Bhatti, I.A. Re-Utilization Option of Industrial Wastewater Treated by Advanced Oxidation Process. Pak. J. Agric. Sci. 2014, 51, 1141–1147. [Google Scholar]
- Hong, P.-Y.; Julian, T.R.; Pype, M.-L.; Jiang, S.C.; Nelson, K.L.; Graham, D.; Pruden, A.; Manaia, C.M. Reusing Treated Wastewater: Consideration of the Safety Aspects Associated with Antibiotic-Resistant Bacteria and Antibiotic Resistance Genes. Water 2018, 10, 244. [Google Scholar] [CrossRef]
- Hussein, F.H. Effect of Photocatalytic Treatments on Physical and Biological Properties of Textile Dyeing Wastewater. Asian J. Chem. 2013, 25, 9387–9392. [Google Scholar] [CrossRef]
- Vajnhandl, S.; Valh, J.V. The status of water reuse in European textile sector. J. Environ. Manag. 2014, 141, 29–35. [Google Scholar] [CrossRef]
- Sousa, M.; Gonçalves, C.; Pereira, J.H.; Vilar, V.J.; Boaventura, R.A.; Alpendurada, M.F. Photolytic and TiO2-Assisted Photo-catalytic Oxidation of the Anxiolytic Drug Lorazepam (Lorenin® Pills) under Artificial Uv Light and Natural Sunlight: A Comparative and Comprehensive Study. Sol. Energy 2013, 87, 219–228. [Google Scholar] [CrossRef]
- Jimenez, M.; Maldonado, M.I.; Rodríguez, E.M.; Hernández-Ramírez, A.; Saggioro, E.; Carra, I.; Pérez, J.A.S. Supported TiO2 solar photocatalysis at semi-pilot scale: Degradation of pesticides found in citrus processing industry wastewater, reactivity and influence of photogenerated species. J. Chem. Technol. Biotechnol. 2014, 90, 149–157. [Google Scholar] [CrossRef]
- Luo, H.; Zeng, Y.; Cheng, Y.; He, D.; Pan, X. Recent Advances in Municipal Landfill Leachate: A Review Focusing on Its Char-acteristics, Treatment, and Toxicity Assessment. Sci. Total Environ. 2020, 703, 135468. [Google Scholar] [CrossRef]
- Maryam, B.; Büyükgüngör, H. Wastewater reclamation and reuse trends in Turkey: Opportunities and challenges. J. Water Process. Eng. 2019, 30, 100501. [Google Scholar] [CrossRef]
- Deviller, G.; Lundy, L.; Fatta-Kassinos, D. Recommendations to derive quality standards for chemical pollutants in reclaimed water intended for reuse in agricultural irrigation. Chemosphere 2020, 240, 124911. [Google Scholar] [CrossRef]
- Pekakis, P.A.; Xekoukoulotakis, N.; Mantzavinos, D. Treatment of textile dyehouse wastewater by TiO2 photocatalysis. Water Res. 2006, 40, 1276–1286. [Google Scholar] [CrossRef] [PubMed]
- Sirirerkratana, K.; Kemacheevakul, P.; Chuangchote, S. Color removal from wastewater by photocatalytic process using titanium dioxide-coated glass; ceramic tile; and stain less steel sheets. J. Clean. Prod. 2019, 215, 123–130. [Google Scholar] [CrossRef]
| Ceramic Support | Incorporation Method | Application | Reference |
|---|---|---|---|
| Leca | Deposition of TiO2 solution in Leca followed by calcination at 550 °C | Photocatalytic degradation of ammonia | [38] |
| Diatomite-based porous ceramics | Hydrolysis deposition method with TiCl4 as the precursor | Degradation of malachite green solution | [64] |
| Ceramic materials | Dip-coating by immersing ceramics for 30 min followed by drying at 85 °C for 30 min and then 150 °C for 5 min | Self-cleaning properties to ceramic surfaces | [112] |
| Clay nanocomposites | Hydrothermal synthesis (T = 100 °C and P = 5 atm) during 24 h | Advanced treatment of wastewater loaded with dye (MB) and heavy metal (cadmium) | [119] |
| Laponite | Hydrothermal method (T = 100, 150 and 200 °C) | Photocatalytic degradation of Herbicides | [120] |
| Ceramic membranes | Atomic layer deposition of TiO2 onto anodized aluminum oxide membranes | Photocatalytic degradation of MB | [126] |
| Porous ceramic material | Sol-gel method, titanium tetra-isopropoxide was used as TiO2 precursor | Degradation of Rhodamine B 2020 | [141] |
| Clay roofing tiles | Spray method | Inactivation of Pseudomonas Aeruginosa bacteria and degradation of p-chlorobenzoic acid the mesoporous coating | [142] |
| Glazed ceramic pieces | Spraying with P-25 followed by calcination at 450 °C | Degradation of methylene blue (MB) | [143] |
| Ceramic tiles | Sol-gel coating for SiO2/TiO2 preparation followed by deposition technique on ceramic tiles | Degradation of MB | [144] |
| Ceramic substrate (Ca2MgSi2O7: Ce3+ and kaolin) | TiO2 (P25, Macklin) powder was dispersed in 5 mL deionized water by sonication. Then the dispersion was dropped onto the surface of the Ca2MgSi2O7: Ce3+ ceramic substrate followed by calcination at 400 °C | Water purification | [145] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Danfá, S.; Martins, R.C.; Quina, M.J.; Gomes, J. Supported TiO2 in Ceramic Materials for the Photocatalytic Degradation of Contaminants of Emerging Concern in Liquid Effluents: A Review. Molecules 2021, 26, 5363. https://doi.org/10.3390/molecules26175363
Danfá S, Martins RC, Quina MJ, Gomes J. Supported TiO2 in Ceramic Materials for the Photocatalytic Degradation of Contaminants of Emerging Concern in Liquid Effluents: A Review. Molecules. 2021; 26(17):5363. https://doi.org/10.3390/molecules26175363
Chicago/Turabian StyleDanfá, Sadjo, Rui C. Martins, Margarida J. Quina, and João Gomes. 2021. "Supported TiO2 in Ceramic Materials for the Photocatalytic Degradation of Contaminants of Emerging Concern in Liquid Effluents: A Review" Molecules 26, no. 17: 5363. https://doi.org/10.3390/molecules26175363
APA StyleDanfá, S., Martins, R. C., Quina, M. J., & Gomes, J. (2021). Supported TiO2 in Ceramic Materials for the Photocatalytic Degradation of Contaminants of Emerging Concern in Liquid Effluents: A Review. Molecules, 26(17), 5363. https://doi.org/10.3390/molecules26175363









