Electrosprayed Shrimp and Mushroom Nanochitins on Cellulose Tissue for Skin Contact Application
Abstract
:1. Introduction
2. Results
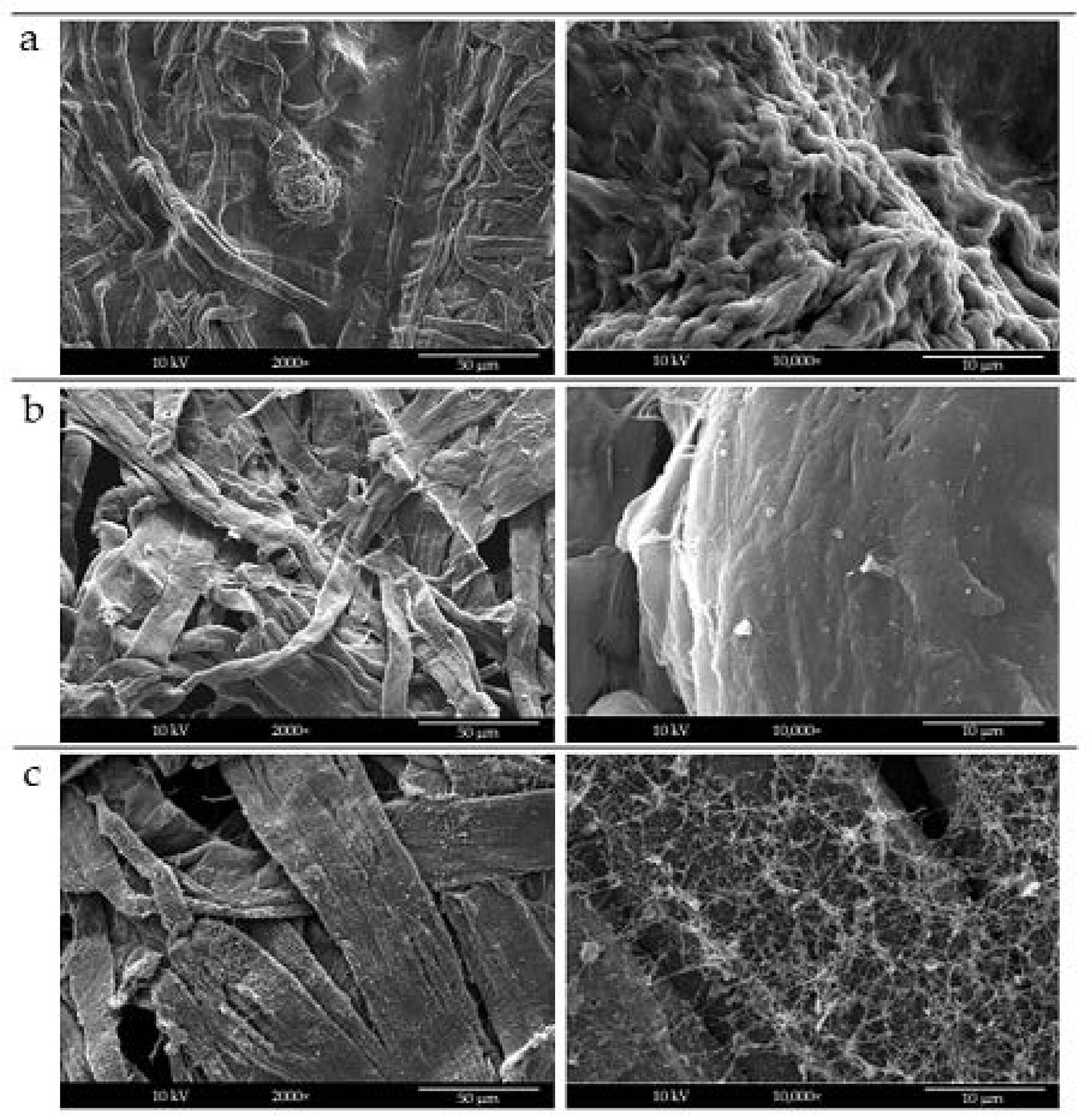
2.1. Morphological Characterization
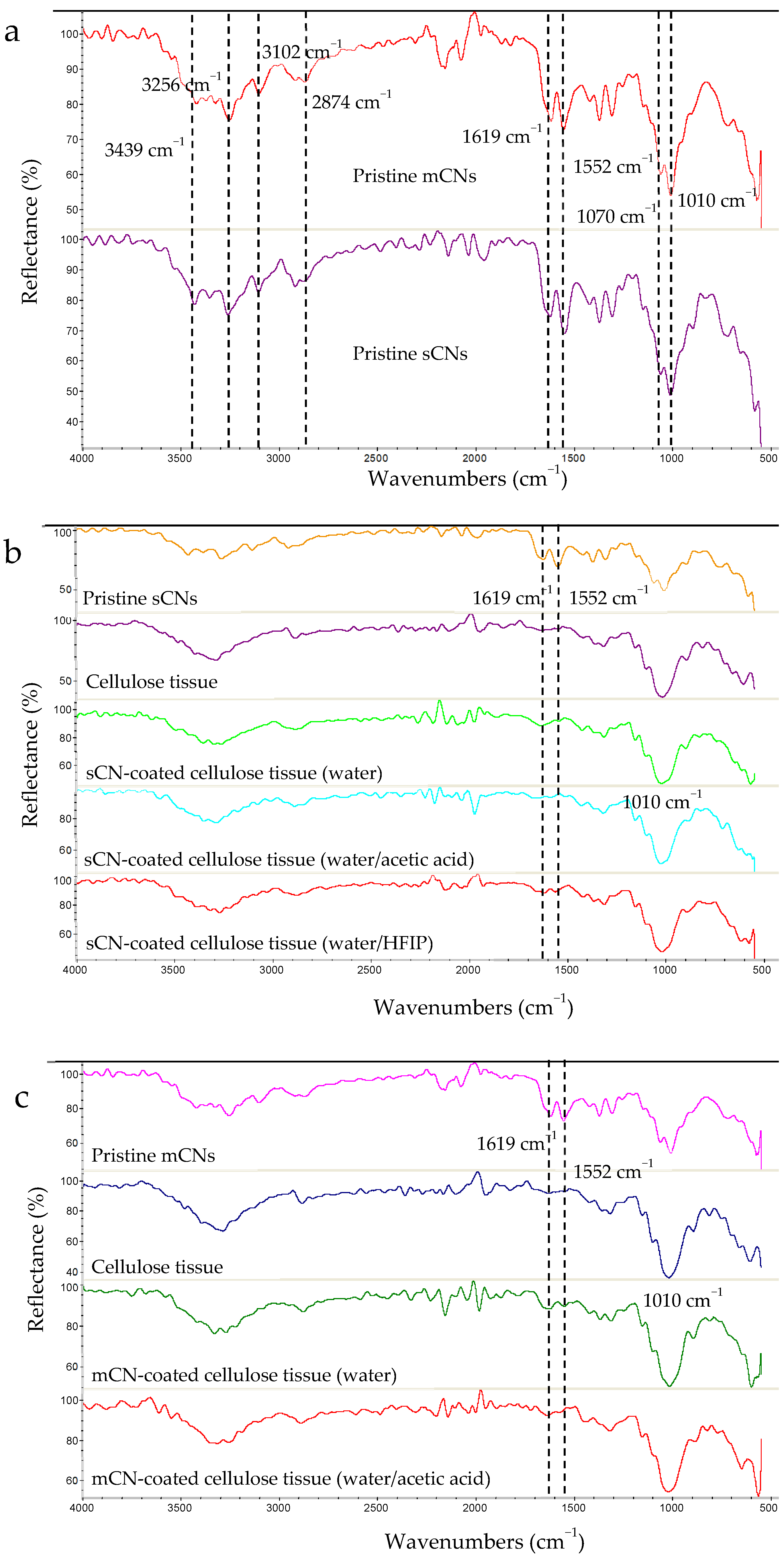
2.2. Chemical Structure Characterization
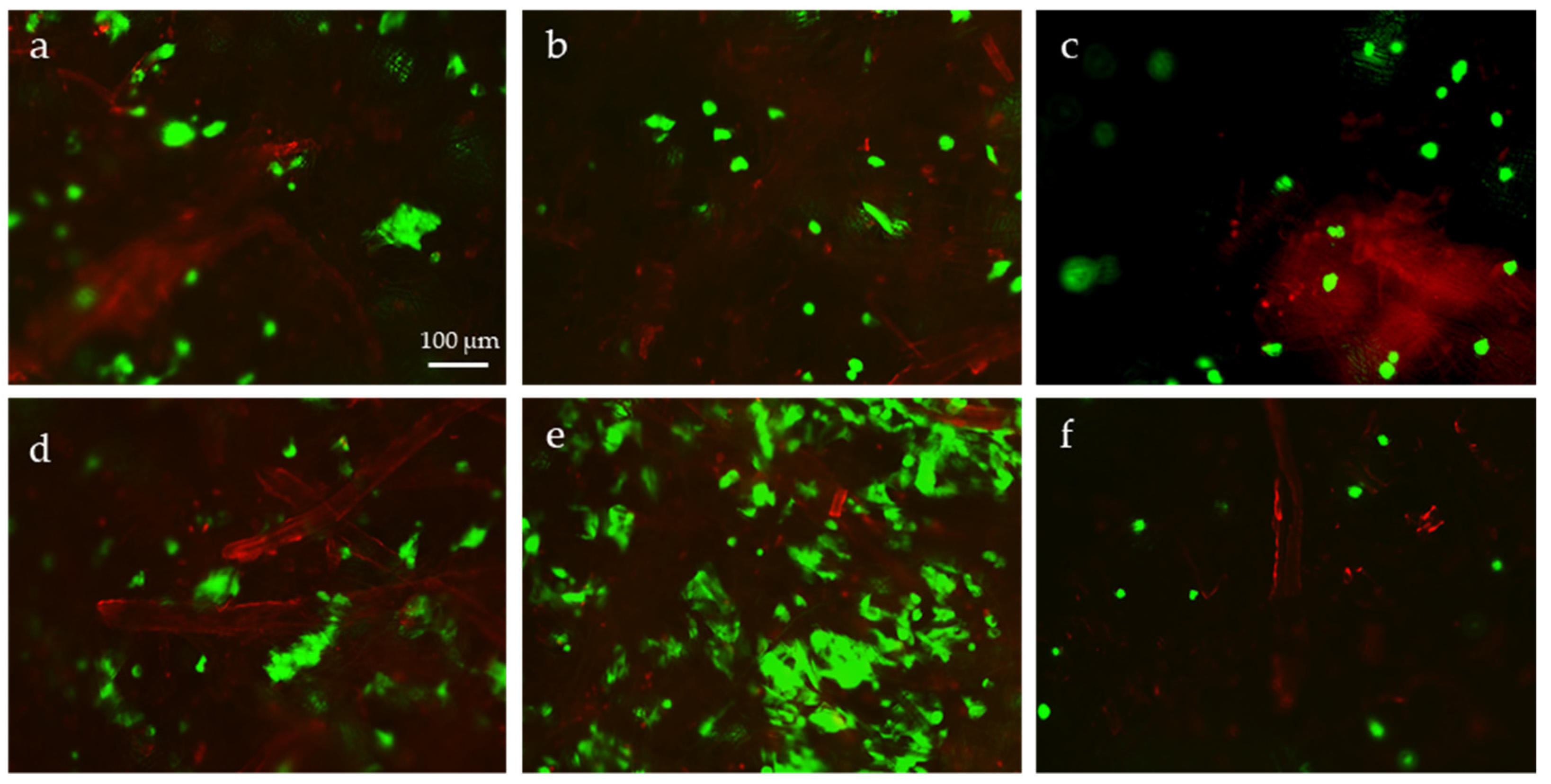
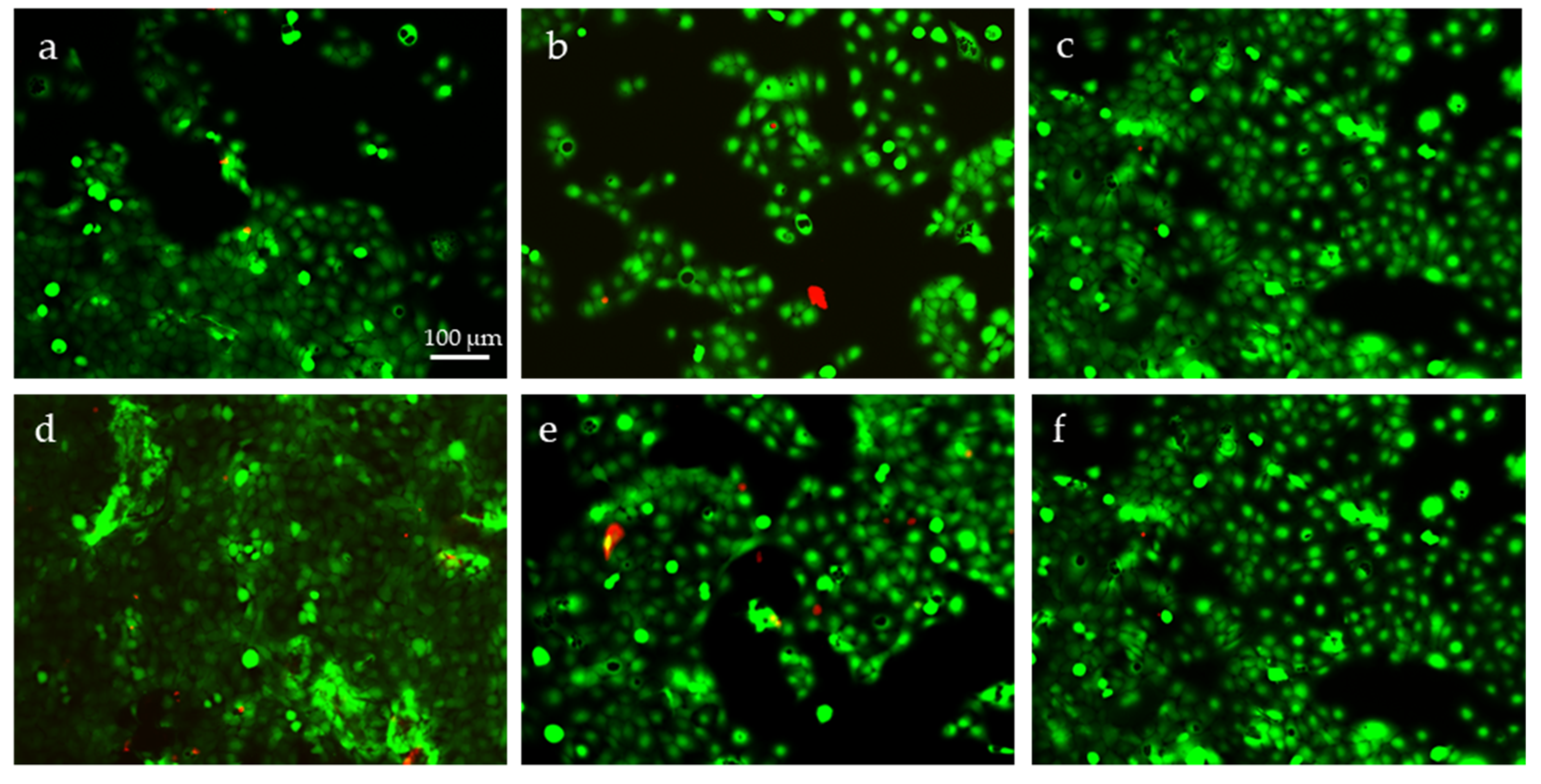
2.3. Cytotoxicity Evaluation
2.4. Evaluation of Cell Metabolic Activity
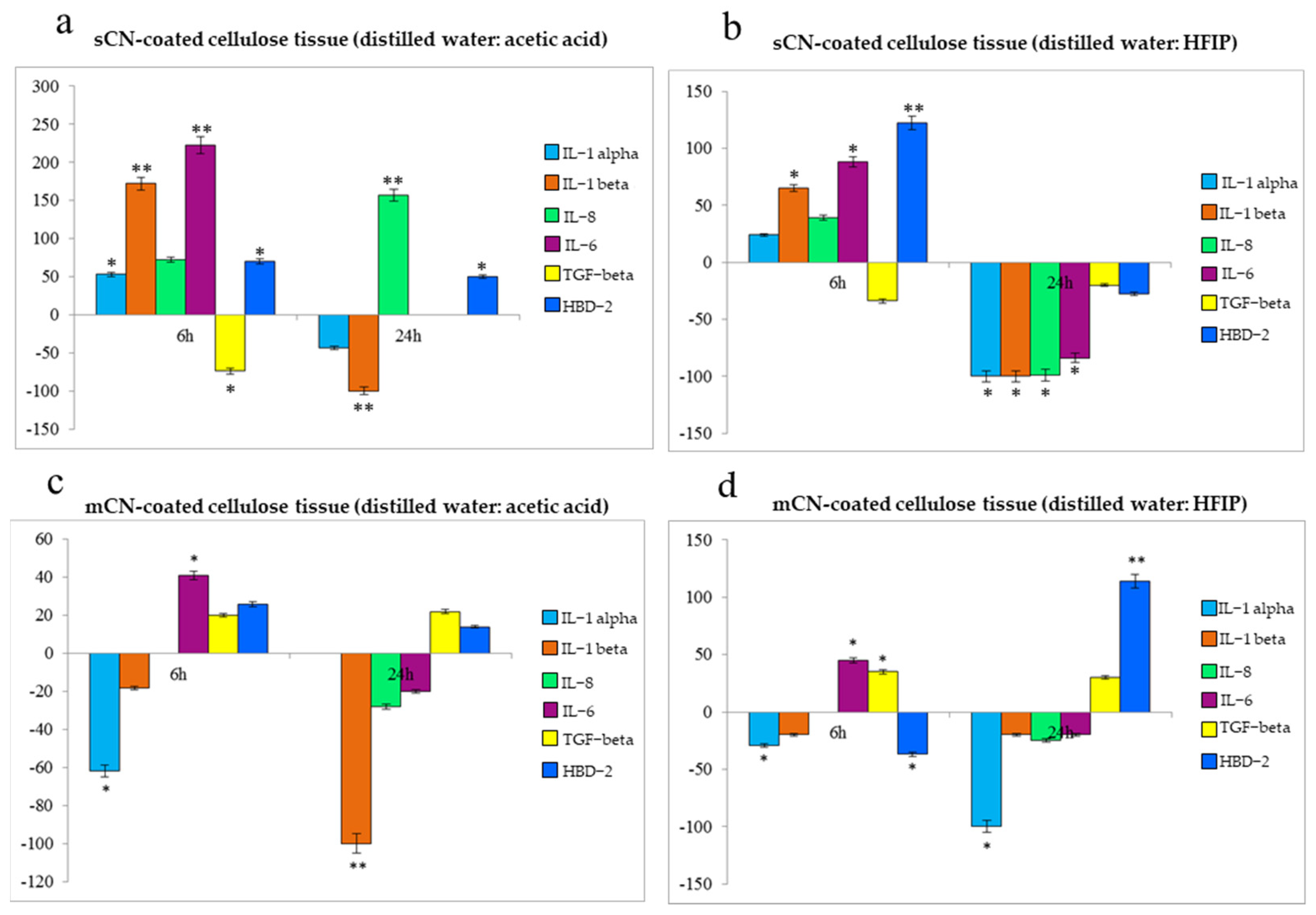
2.5. Anti-Inflammatory and Immune Response of HaCaT Cells
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation of CN Solutions and Electrospray Protocols
4.3. Morphological Characterization of CNs
4.4. Chemical Structure Characterization
4.5. Cytotoxicity Evaluation with Epidermal Cells
4.6. Cell Metabolic Activity Evaluation
4.7. Immune Response by Epidermal Cells
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Danti, S.; Trombi, L.; Fusco, A.; Azimi, B.; Lazzeri, A.; Morganti, P.; Coltelli, M.B.; Donnarumma, G. Chitin nanofibrils and nanolignin as functional agents in skin regeneration. Int. J. Mol. Sci. 2019, 20, 2669. [Google Scholar] [CrossRef] [Green Version]
- Morganti, P. Circular Economy: A New Horizon for Bio–Nanocomposites from Waste Materials. Int. J. Biotechnol. Wellness Ind. 2016, 5, 121–127. [Google Scholar] [CrossRef]
- Hoell, I.A.; Vaaje-Kolstad, G.; Eijsink, V.G.H. Structure and function of enzymes acting on chitin and chitosan. Biotechnol. Genet. Eng. Rev. 2010, 27, 331–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thangadurai, T.D.; Manjubaashini, N.; Thomas, S.; Maria, H.J. Nanostructured Materials; Springer International Publishing: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Coltelli, M.B.; Aliotta, L.; Vannozzi, A.; Morganti, P.; Panariello, L.; Danti, S.; Neri, S.; Fernandez-Avila, C.; Fusco, A.; Donnarumma, G.; et al. Properties and Skin Compatibility of Films Based on Poly (Lactic Acid) (PLA) Bionanocomposites Incorporating Chitin Nanofibrils (CN). J. Funct. Biomater. 2020, 11, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teno, J.; Pardo-Figuerez, M.; Hummel, N.; Bonin, V.; Fusco, A.; Ricci, C.; Donnarumma, G.; Coltelli, M.B.; Danti, S.; Lagaron, J.M. Preliminary Studies on an Innovative Bioactive Skin Soluble Beauty Mask Made by Combining Electrospinning and Dry Powder Impregnation. Cosmetics 2020, 7, 96. [Google Scholar] [CrossRef]
- Roy, J.C.; Salaün, F.; Giraud, S.; Ferri, A.; Chen, G.; Guan, J. Solubility of Chitin: Solvents, Solution Behaviors and Their Related Mechanisms. In Solubility of Polysaccharides; Xu, Z., Ed.; InTech: Rijeka, Croatia, 2017. [Google Scholar] [CrossRef] [Green Version]
- Kavadiya, S.; Biswas, P. Electrospray deposition of biomolecules: Applications, challenges, and recommendations. J. Aerosol. Sci. 2018, 125, 182–207. [Google Scholar] [CrossRef]
- Azimi, B.; Thomas, L.; Fusco, A.; Kalaoglu-Altan, O.I.; Basnett, P.; Cinelli, P.; De Clerck, K.; Roy, I.; Donnarumma, G.; Coltelli, M.B.; et al. Electrosprayed Chitin Nanofibril/Electrospun Polyhydroxyalkanoate Fiber Mesh as Functional Nonwoven for Skin Application. J. Funct. Biomater. 2020, 11, 62. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.N.; Clasen, C.; Van den Mooter, G. Pharmaceutical Applications of Electrospraying. J. Pharm. Sci. 2016, 105, 2601–2620. [Google Scholar] [CrossRef] [Green Version]
- Khezri, K.; Saeedi, M.; Maleki Dizaj, S. Application of nanoparticles in percutaneous delivery of active ingredients in cosmetic preparations. Biomed. Pharm. 2018, 106, 1499–1505. [Google Scholar] [CrossRef]
- Pereira, L. Seaweeds as Source of Bioactive Substances and Skin Care Therapy—Cosmeceuticals, Algotheraphy, and Thalassotherapy. Cosmetics 2018, 5, 68. [Google Scholar] [CrossRef] [Green Version]
- Reddy Desam, N.; Al-Rajab, A.J. The Importance of Natural Products in Cosmetics. In Bioactive Natural Products for Pharmaceutical Applications; Pal, D., Nayak, A.K., Eds.; Springer: New York, NY, USA, 2021; pp. 643–685. [Google Scholar]
- Azimi, B.; Milazzo, M.; Danti, S. Cellulose-Based Fibrous Materials From Bacteria to Repair Tympanic Membrane Perforations. Front. Bioeng. Biotechnol. 2021, 9, 474. [Google Scholar] [CrossRef]
- Gregory, D.A.; Tripathi, L.; Frickera, A.T.R.; Asare, E.; Orlando, I.; Raghavendran, V.; Roy, I. Bacterial cellulose: A smart biomaterial with diverse applications. Mater. Sci. Eng. R Rep. 2021, 145, 100623. [Google Scholar] [CrossRef]
- Portela, R.; Leal, C.R.; Almeida, P.L.; Sobral, R.G. Bacterial cellulose: A versatile biopolymer for wound dressing applications. Microb. Biotechnol. 2019, 12, 586–610. [Google Scholar] [CrossRef]
- Qiu, Y.; Qiub, L.; Cui, J.; Wei, Q. Bacterial cellulose and bacterial cellulose-vaccarin membranes for wound healing. Mater. Sci. Eng. C 2016, 59, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Coltelli, M.B.; Danti, S.; De Clerck, K.; Lazzeri, A.; Morganti, P. Pullulan for Advanced Sustainable Body- and Skin-Contact Applications. J. Funct. Biomater. 2020, 11, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azimi, B.; Maleki, H.; Zavagna, L.; De la Ossa, J.G.; Linari, S.; Lazzeri, A.; Danti, S. Bio-Based Electrospun Fibers for Wound Healing. J. Funct. Biomater. 2020, 11, 67. [Google Scholar] [CrossRef] [PubMed]
- Azimi, B.; Milazzo, M.; Lazzeri, A.; Berrettini, S.; Uddin, M.J.; Qin, Z.; Buehler, M.J.; Danti, S. Electrospinning Piezoelectric Fibers for Biocompatible Devices. Adv. Healthc. Mater. 2020, 9, 1901287. [Google Scholar] [CrossRef] [PubMed]
- Panariello, L.; Vannozzi, A.; Morganti, P.; Coltelli, M.B.; Lazzeri, A. Biobased and Eco-Compatible Beauty Films Coated with Chitin Nanofibrils, Nanolignin and Vitamin E. Cosmetics 2021, 8, 27. [Google Scholar] [CrossRef]
- Fan, Y.; Fukuzumi, H.; Saito, T.; Isogai, A. Comparative characterization of aqueous dispersions and cast films of different chitin nanowhiskers/nanofibers. Int. J. Biol. Macromol. 2012, 50, 69–76. [Google Scholar] [CrossRef]
- Satam, C.C.; Irvin, C.W.; Lang, A.W.; Jallorina, J.C.R.; Shofner, M.L.; Reynolds, J.R.; Meredith, J.C. Spray-Coated Multilayer Cellulose Nanocrystal—Chitin Nanofiber Films for Barrier Applications. ACS Sustain. Chem. Eng. 2018, 6, 10637–10644. [Google Scholar] [CrossRef]
- Yu, Z.; Ji, Y.; Bourg, V.; Bilgen, M.; Meredith, J.C. Chitin-and cellulose-based sustainable barrier materials: A review. Emergent Mater. 2020, 3, 919–936. [Google Scholar] [CrossRef]
- Morganti, P.; Del Ciotto, P.; Stoller, M.; Chianese, A. Antibacterial and anti-inflammatory green nanocomposites. Chem. Eng. Trans. 2016, 47, 61–66. [Google Scholar]
- Obisesan, K.A.; Neri, S.; Bugnicourt, E.; Campos, I.; Rodriguez-Turienzo, L. Determination and Quantification of the Distribution of CN-NL Nanoparticles Encapsulating Glycyrrhetic Acid on Novel Textile Surfaces with Hyperspectral Imaging. J. Funct. Biomater. 2020, 11, 32. [Google Scholar] [CrossRef]
- Gaskell, S.J. Electrospray: Principles and Practice. J. Mass Spectrom 1997, 32, 677–688. [Google Scholar] [CrossRef]
- Smeets, A.; Clasen, C.; Van Den Mooter, G. Electrospraying of polymer solutions: Study of formulation and process parameters. Eur. J. Pharm. Biopharm. 2017, 119, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Faramarzi, A.R.; Barzin, J.; Mobedi, H. Effect of solution and apparatus parameters on the morphology and size of electrosprayed PLGA microparticles. Fibers Polym. 2016, 17, 1806–1819. [Google Scholar] [CrossRef]
- Geng, X.; Kwon, O.H.; Jang, J. Electrospinning of chitosan dissolved in concentrated acetic acid solution. Biomaterials 2005, 26, 5427–5432. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Cooper, A.; Kapetanovic, A.; Fang, Z.; Zhang, M.; Rolandi, M. A facile bottom-up route to self-assembled biogenic chitin nanofibers. Soft Matter 2010, 6, 5298–5301. [Google Scholar] [CrossRef]
- Min, B.M.; Lee, S.W.; Lim, J.N.; You, Y. Chitin and chitosan nanofibers: Electrospinning of chitin and deacetylation of chitin nanofibers. Polymer 2004, 45, 7137–7142. [Google Scholar] [CrossRef]
- Hickey, R.J.; Pelling, A.E. Cellulose biomaterials for tissue engineering. Front. Bioeng. Biotechnol. 2019, 7, 45. [Google Scholar] [CrossRef] [Green Version]
- Dutta, S.D.; Patel, D.K.; Lim, K.T. Functional cellulose-based hydrogels as extracellular matrices for tissue engineering. J. Biol. Eng. 2019, 13, 55. [Google Scholar] [CrossRef] [PubMed]
- Pang, M.; Huang, Y.; Meng, F.; Zhuang, Y.; Liu, H.; Du, M.; Ma, Q.; Wang, Q.; Chen, Z.; Chene, L.; et al. Application of bacterial cellulose in skin and bone tissue engineering. Eur. Polym. J. 2020, 122, 109365. [Google Scholar] [CrossRef]
- Lv, X.G.; Yang, J.X.; Feng, C.; Li, Z.; Chen, S.; Xie, M.K.; Huang, J.W.; Li, H.B.; Wang, H.P.; Xu, Y.M. Bacterial Cellulose-Based Biomimetic Nanofibrous Scaffold withMuscle Cells for Hollow Organ Tissue Engineering. ACS Biomater. Sci. Eng. 2016, 2, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Cooney, D.O.; Nagerl, A.; Hines, A.L. Solvent regeneration of activated carbon. Water Res. 1983, 17, 403–410. [Google Scholar] [CrossRef]
- Alvarez, F.J. The Effect of chitin size, shape, source and purification method on immune recognition. Molecules 2014, 19, 4433–4451. [Google Scholar] [CrossRef]


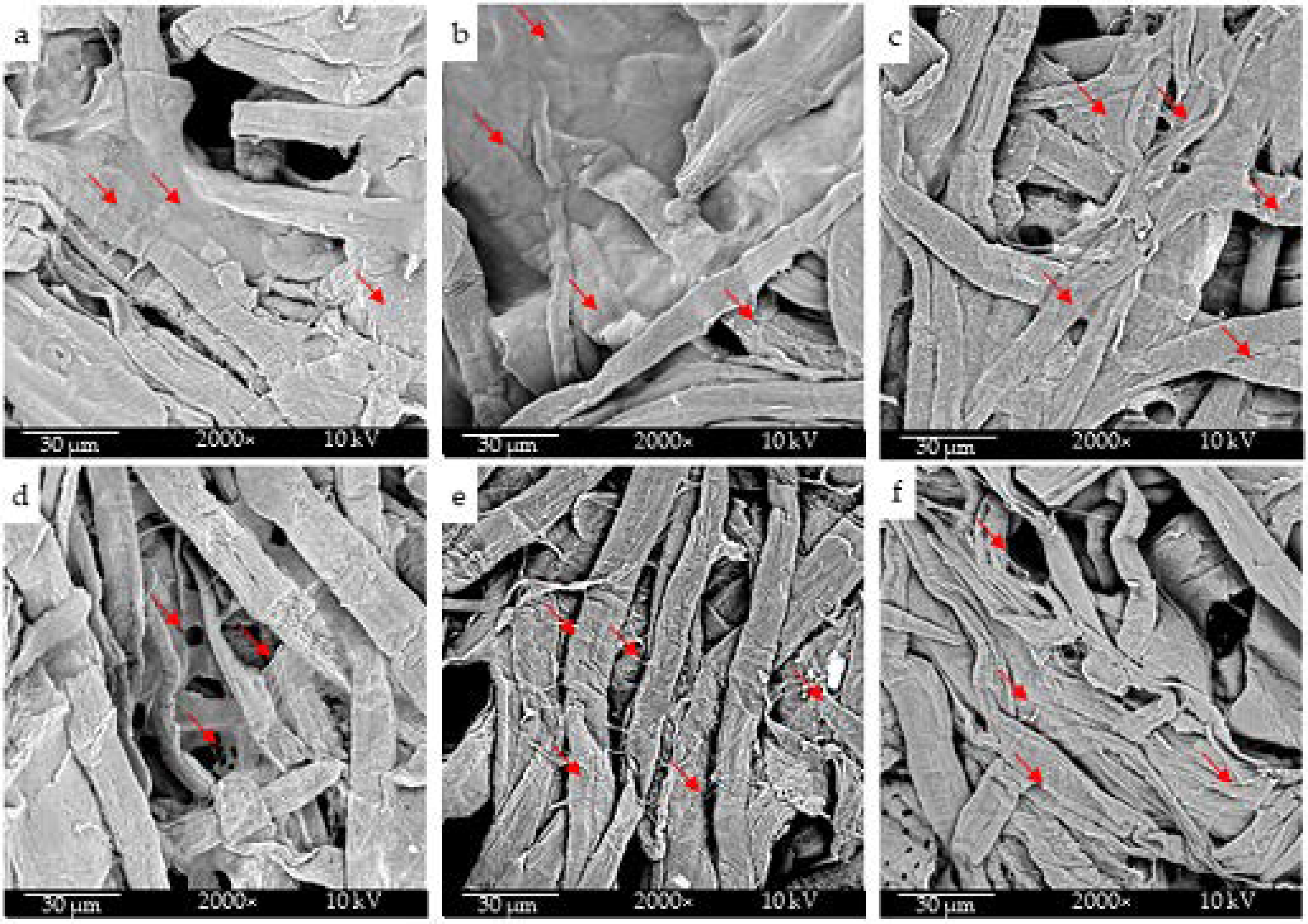
| Cellulose Tissue Sample | Electrospray Solvent(s) | % ABRED |
|---|---|---|
| sCN-coated | distilled water | 73% |
| sCN-coated | distilled water/acetic acid | 79% |
| sCN-coated | distilled water/HFIP | 98% |
| mCN-coated | distilled water | 95% |
| mCN-coated | distilled water/acetic acid | 88% |
| Pristine | none | 71% |
| Gene | Primer Sequence (Forward, Reverse) | Conditions | Base Pairs |
|---|---|---|---|
| IL-1α | 5′-CATGTCAAATTTCACTGCTTCATCC-3′ 5′-GTCTCTGAATCAGAAATCCTTCTATC-3′ | 5 s at 95 °C, 8 s at 55 °C, 1 s at 72 °C for 45 cycles | 421 |
| IL-1β | 5′-GCATCCAGCTACGAATCTCC-3′ 5′-CCACATTCAGCACAGGACTC-3′ | 5 s at 95 °C, 14 s at 58 °C, 28 s at 72 °C for 40 cycles | 708 |
| TNF-α | 5′-CAGAGGGAAGAGTTCCCCAG -3′ 5′-CCTTGGTCTGGTAGGAGACG-3′ | 5 s at 95 °C, 6 s at 57 °C, 13 s at 72 °C for 40 cycles | 324 |
| IL-6 | 5′-ATGAACTCCTTCTCCACAAGCGC-3′ 5′-GAAGAGCCCTCAGGCTGGACTG-3′ | 5 s at 95 °C, 13 s at 56 °C, 25 s at 72 °C for 40 cycles | 628 |
| IL-8 | 5-ATGACTTCCAAGCTGGCCGTG-3′ 5-TGAATTCTCAGCCCTCTTCAAAAACTTCTC-3′ | 5 s at 94 °C, 6 s at 55 °C, 12 s at 72 °C for 40 cycles | 297 |
| TGF-β | 5′-CCGACTACTACGCCAAGGAGGTCAC-3′ 5′-AGGCCGGTTCATGCCATGAATGGTG-3′ | 5 s at 94 °C, 9 s at 60 °C, 18 s at 72 °C for 40 cycles | 439 |
| HBD-2 | 5′-GGATCCATGGGTATAGGCGATCCTGTTA-3′ 5′-AAGCTTCTCTGATGAGGGAGCCCTTTCT-3′ | 5 s at 94 °C, 6 s at 63 °C, 10 s at 72 °C for 50 cycles | 198 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azimi, B.; Ricci, C.; Fusco, A.; Zavagna, L.; Linari, S.; Donnarumma, G.; Hadrich, A.; Cinelli, P.; Coltelli, M.-B.; Danti, S.; et al. Electrosprayed Shrimp and Mushroom Nanochitins on Cellulose Tissue for Skin Contact Application. Molecules 2021, 26, 4374. https://doi.org/10.3390/molecules26144374
Azimi B, Ricci C, Fusco A, Zavagna L, Linari S, Donnarumma G, Hadrich A, Cinelli P, Coltelli M-B, Danti S, et al. Electrosprayed Shrimp and Mushroom Nanochitins on Cellulose Tissue for Skin Contact Application. Molecules. 2021; 26(14):4374. https://doi.org/10.3390/molecules26144374
Chicago/Turabian StyleAzimi, Bahareh, Claudio Ricci, Alessandra Fusco, Lorenzo Zavagna, Stefano Linari, Giovanna Donnarumma, Ahdi Hadrich, Patrizia Cinelli, Maria-Beatrice Coltelli, Serena Danti, and et al. 2021. "Electrosprayed Shrimp and Mushroom Nanochitins on Cellulose Tissue for Skin Contact Application" Molecules 26, no. 14: 4374. https://doi.org/10.3390/molecules26144374
APA StyleAzimi, B., Ricci, C., Fusco, A., Zavagna, L., Linari, S., Donnarumma, G., Hadrich, A., Cinelli, P., Coltelli, M.-B., Danti, S., & Lazzeri, A. (2021). Electrosprayed Shrimp and Mushroom Nanochitins on Cellulose Tissue for Skin Contact Application. Molecules, 26(14), 4374. https://doi.org/10.3390/molecules26144374