Rubesanolides F and G: Two Novel Lactone-Type Norditerpenoids from Isodon rubescens
Abstract
:1. Introduction
2. Results and Discussion
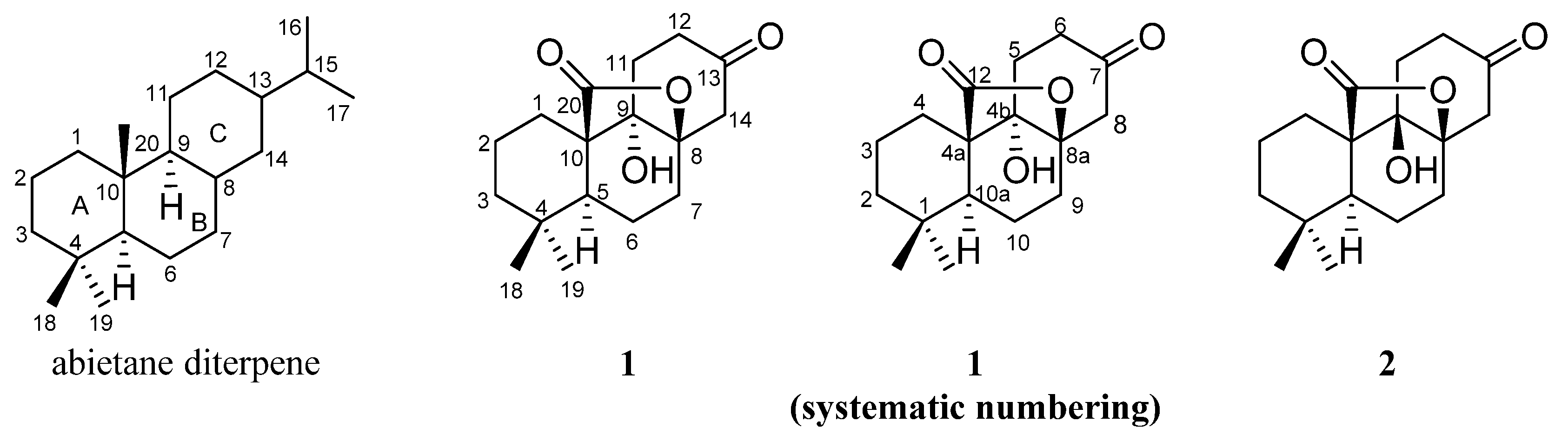
2.1. Structure Elucidation of Compounds
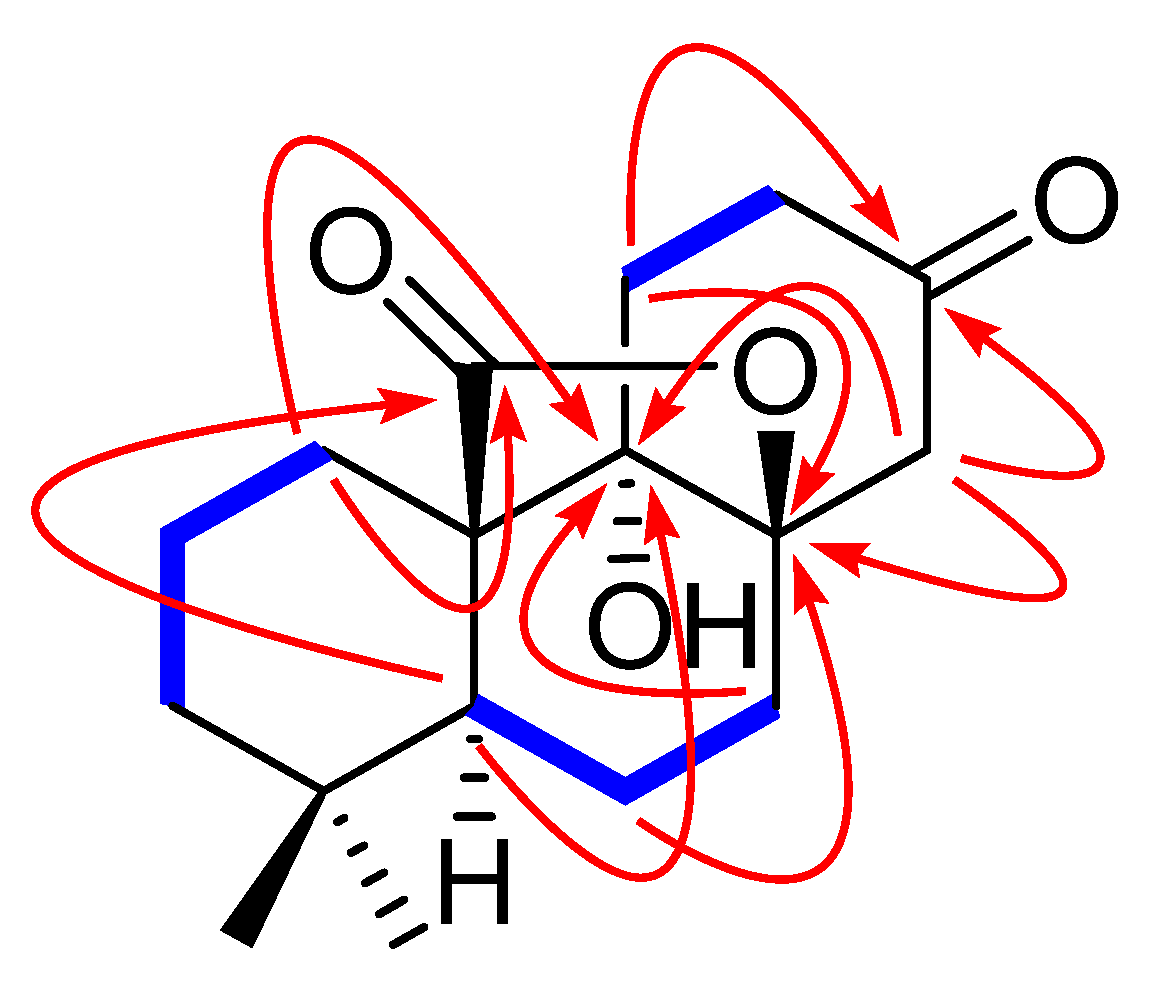
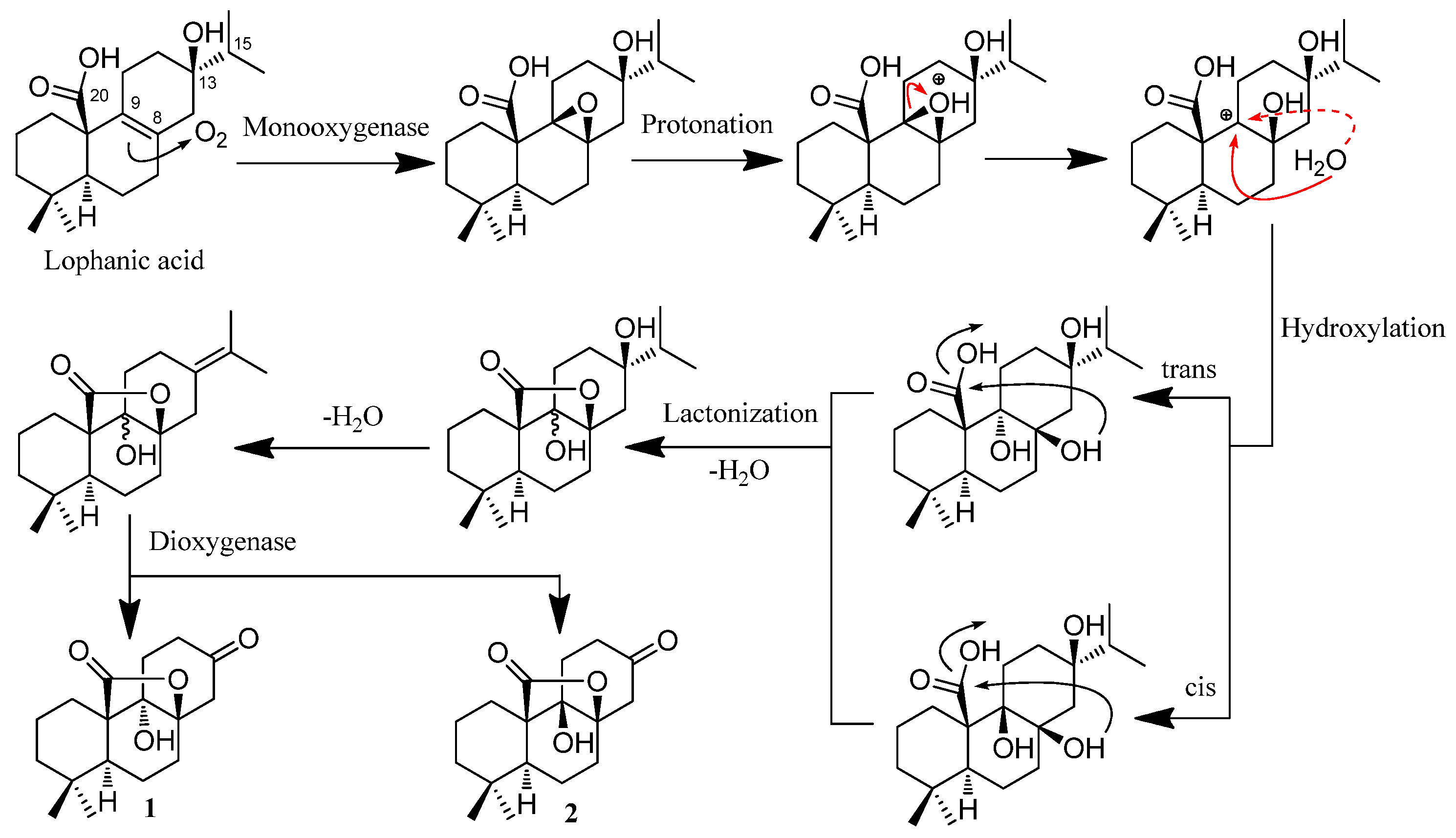
2.2. Hypothetical Biosynthetic Pathways of 1 and 2
2.3. Bioactivity Evaluation Against Cancer Cell Lines and Fungal and Bacterial Strains
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Characerization of Compounds 1 and 2
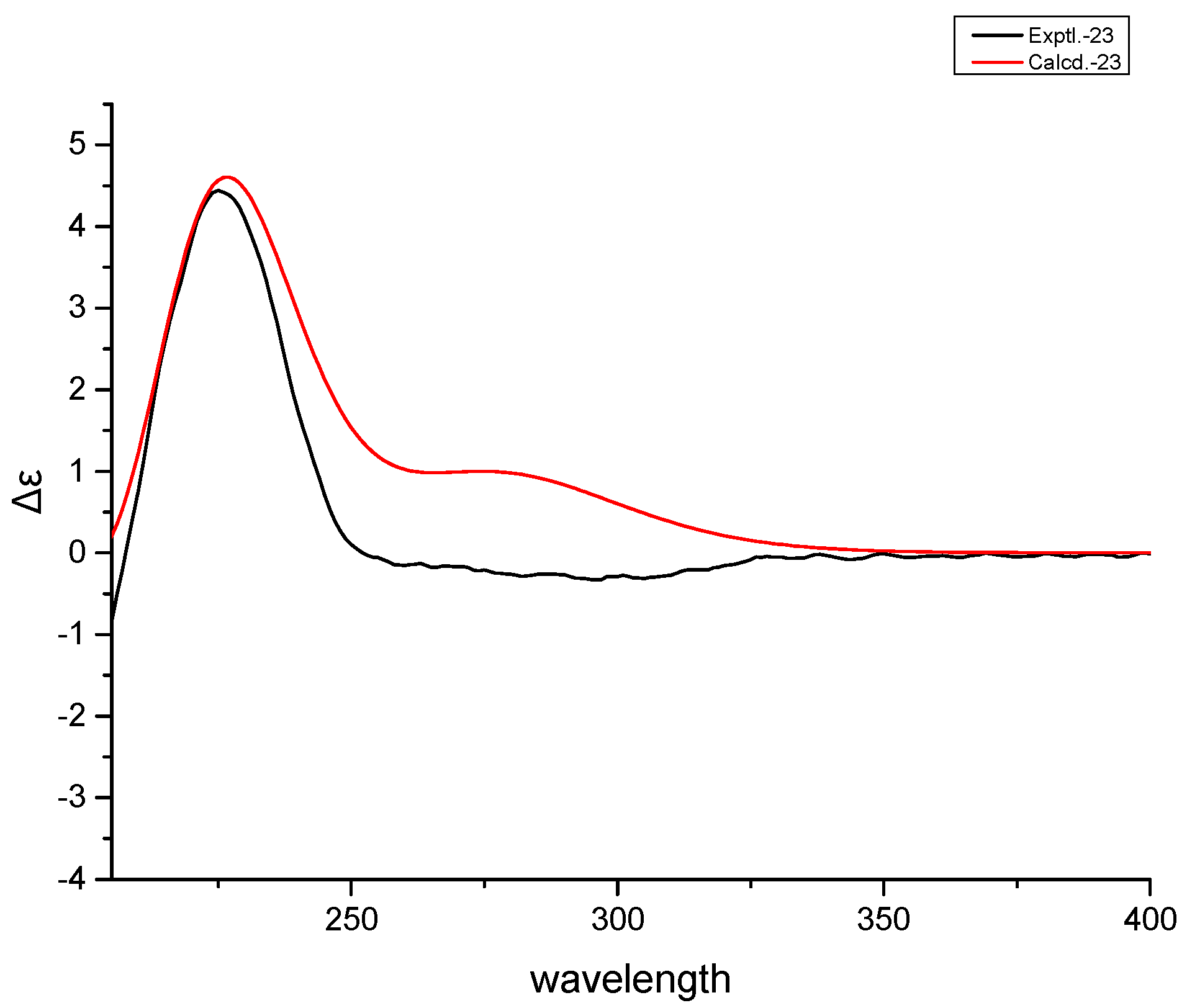
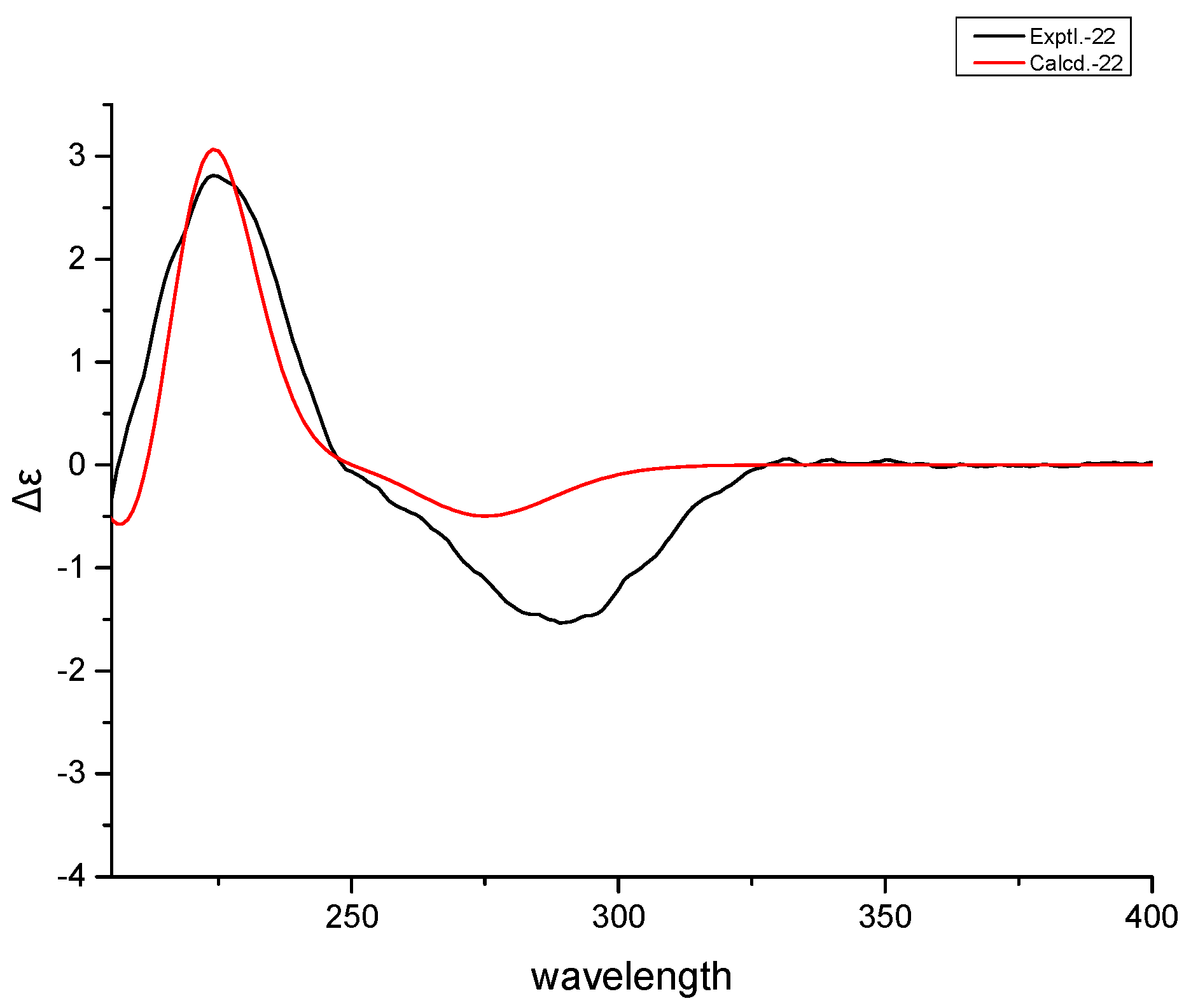
3.5. Theoretically Calculated ECD Spectra of 1 and 2
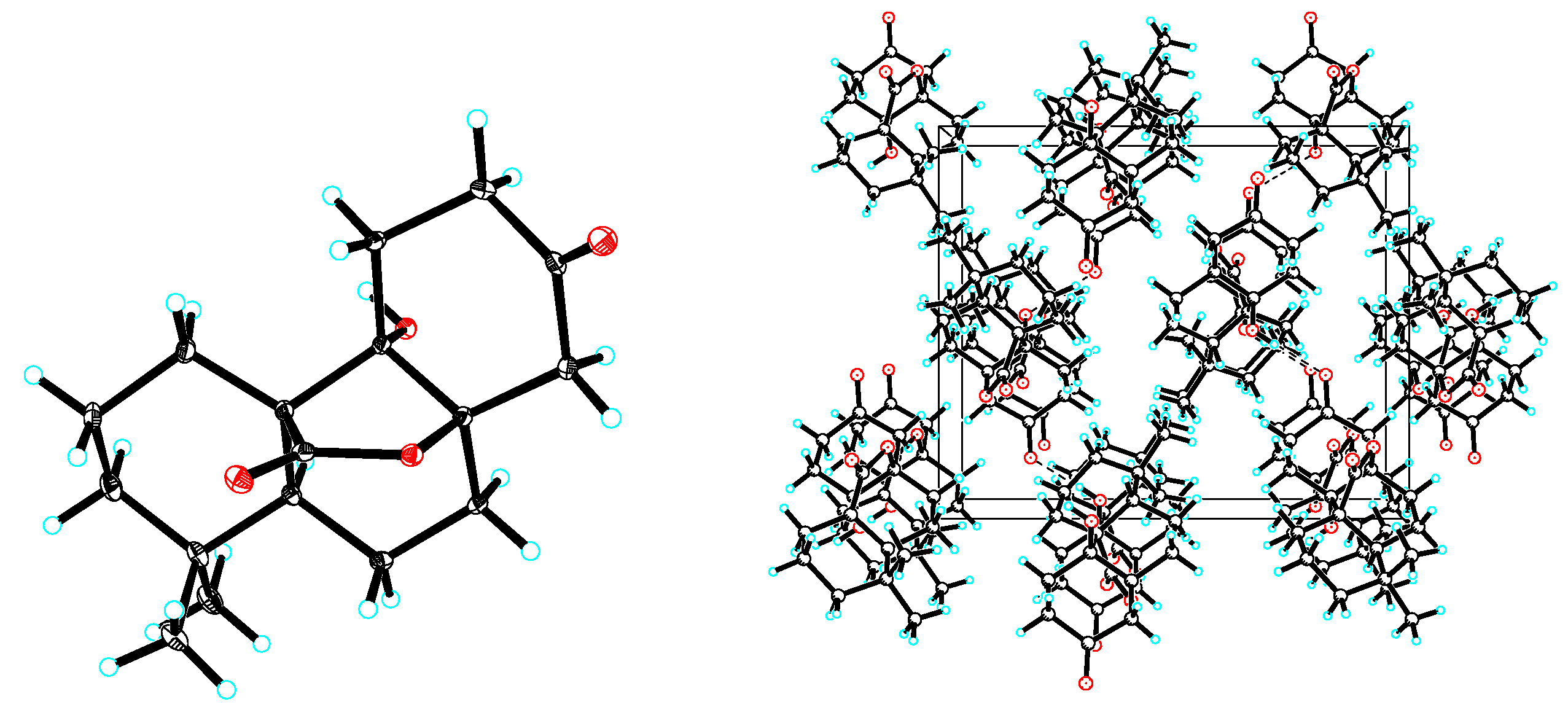
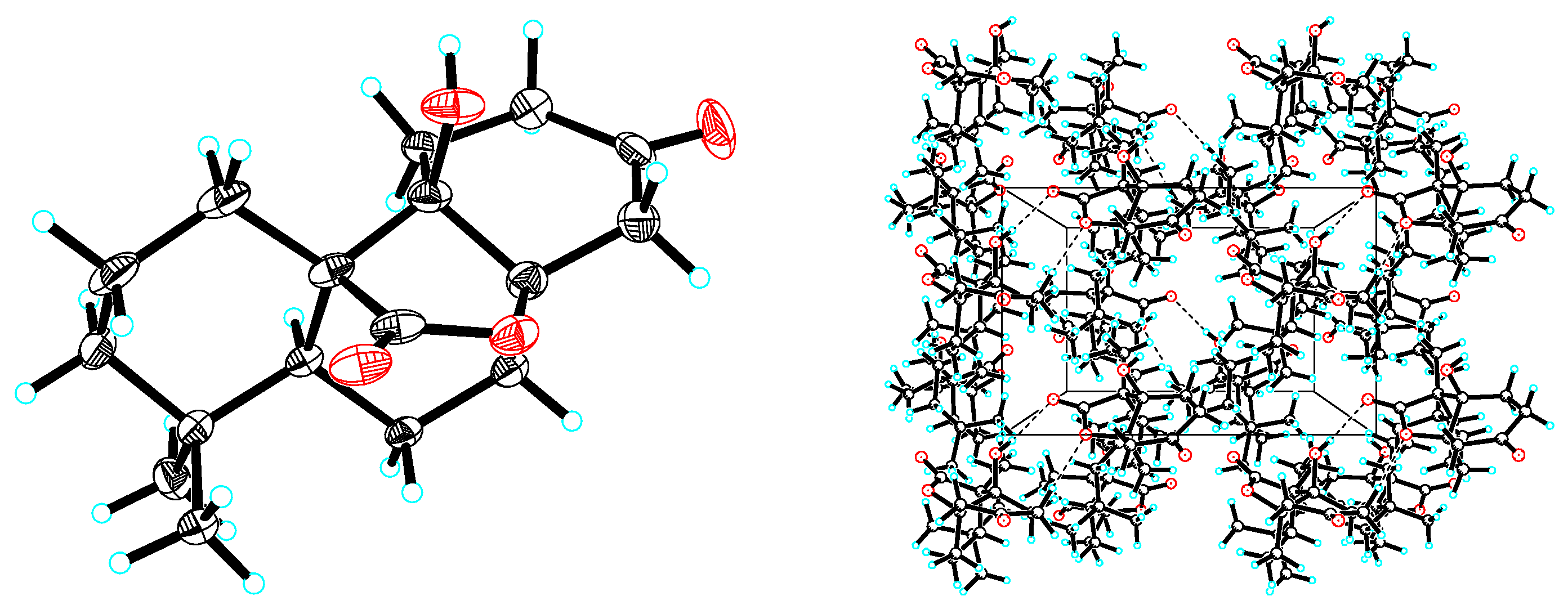
3.6. X-ray Crystallographic Analysis
3.7. Bioactivity Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- He, F.; Xiao, W.-L.; Pu, J.-X.; Wu, Y.-L.; Zhang, H.-B.; Li, X.-N.; Zhao, Y.; Yang, L.-B.; Chen, G.-Q.; Sun, H.-D. Cytotoxic ent-kaurane diterpenoids from Isodon sinuolata. Phytochemistry 2009, 70, 1462–1466. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wang, W.-G.; Sun, H.-D.; Pu, J.-X. Diterpenoids from Isodon species: An update. Nat. Prod. Rep. 2017, 34, 1090–1140. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.D.; Xu, Y.L.; Jiang, B. Chapter 1: Structural Classification of Diterpenoids in Isodon Plants. In Diterpenoids from Isodon Species; Science Press: Beijing, China, 2001; pp. 2–13. [Google Scholar]
- Dai, J.-M.; Hu, K.; Yan, B.-C.; Li, X.-R.; Li, X.-N.; Sun, H.-D.; Puno, P.-T. Ent-kaurane-based diterpenoids, dimers, and meroditerpenoids from Isodon xerophilus. J. Nat. Prod. 2020, 83, 3717–3725. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.-Y.; Wang, W.-G.; Tang, J.-W.; Liu, M.; Li, X.-R.; Hu, K.; Du, X.; Li, X.-N.; Zhang, H.-B.; Pu, J.-X.; et al. Structurally diverse diterpenoids from Isodon scoparius and their bioactivity. J. Nat. Prod. 2017, 80, 2026–2036. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.-D.; Huang, S.-X.; Han, Q. Diterpenoids from Isodon species and their biological activities. Nat. Prod. Rep. 2006, 23, 673–698. [Google Scholar] [CrossRef] [PubMed]
- Ndoile, M.M. Isodon Diterpenoids, Derivatives and Their Pharmacological Potentials—A Review. Tanz. J. Sci. 2020, 46, 171–189. Available online: https://www.ajol.info/index.php/tjs/article/view/194828 (accessed on 20 June 2021).
- Zou, J.; Pan, L.T.; Li, Q.J.; Zhao, J.H.; Pu, J.X.; Yao, P.; Gong, N.B.; Lu, Y.; Kondratyuk, T.P.; Pezzuto, J.M.; et al. Rubesanolides A and B: Diterpenoids from Isodon rubescens. Org. Lett. 2011, 13, 1406–1409. [Google Scholar] [CrossRef] [Green Version]
- Zou, J.; Pan, L.; Li, Q.; Pu, J.; Yao, P.; Zhu, M.; Banas, J.A.; Zhang, H.; Sun, H. Rubesanolides C–E: Abietane diterpenoids isolated from Isodon rubescens and evaluation of their anti-biofilm activity. Org. Biomol. Chem. 2012, 10, 5039–5044. [Google Scholar] [CrossRef] [Green Version]
- Flack, H.D.; Bernardinelli, G. The use of X-ray crystallography to determine absolute configuration. Chirality 2008, 20, 681–690. [Google Scholar] [CrossRef]
- Jiang, B.; Lu, Z.-Q.; Zhang, H.J.; Zhao, Q.-S.; Sun, H.-D. Diterpenoids from Isodon lophanthoides. Fitoterapia 2000, 71, 360–364. [Google Scholar] [CrossRef]
- Wang, Z.Q.; Xu, F.M.; Dong, H.Z.; Node, M.; Fuji, K. The chemical structure of lophanic acid. Nat. Prod. Res. Deve. 1995, 7, 24–28. Available online: https://www.semanticscholar.org/paper/THE-CHEMICAL-STRUCTURE-OF-LOPHANIC-ACID-Kaoru/2110c33fc94fe20f6c8d5a6efbb3d48b7b5b1562#paper-header (accessed on 20 June 2021).
- Liu, F.; Zhang, Y.; Sun, Q.Y.; Yang, F.M.; Gu, W.; Yang, J.; Niu, H.M.; Wang, Y.H.; Long, C.L. Diarylheptanoids and phenylphenalenones from Musa itinerans fruits. Phytochemistry 2014, 103, 171–177. [Google Scholar] [CrossRef]
- Tang, G.-H.; Zhang, Y.; Gu, Y.-C.; Li, S.-F.; Di, Y.-T.; Wang, Y.-H.; Yang, G.-X.; Zuo, G.-Y.; Li, S.-L.; He, H.-P.; et al. Trigoflavidols A–C, degraded diterpenoids with antimicrobial activity, from Trigonostemon flavidus. J. Nat. Prod. 2012, 75, 996–1000. [Google Scholar] [CrossRef] [PubMed]
| No. | 1 a | 2 b | ||
|---|---|---|---|---|
| δH, (J in Hz) c | δC | δH, (J in Hz) c | δC | |
| 1a | 1.41 (m) | 26.0 t | 1.21 (brtd, 13.9, 5.0) | 25.2 t |
| 1b | 1.90 (d, 13.1) | 2.11 (m) | ||
| 2a | 1.47 (m) | 19.0 t | 1.58 (m) | 18.0 t |
| 2b | 2.26 (m) | 2.08 (m) | ||
| 3a | 1.15 (brt, 13.4) | 42.1 t | 1.11 (brtd, 13.5, 3.0) | 41.3 t |
| 3b | 1.39 (m) | 1.48 (m) | ||
| 4 | – | 33.9 s | – | 33.8 s |
| 5 | 2.29 (dd, 13.0, 5.6) | 42.0 d | 1.41 (dd, 13.4, 4.7) | 44.4 d |
| 6a | 1.47 (m) | 20.8 t | 1.50 (m) | 19.8 t |
| 6b | 1.75 (dd, 13.7, 4.7) | 1.81 (m) | ||
| 7a | 1.64 (dd, 13.3, 5.2) | 30.9 t | 1.62 (m) | 31.6 t |
| 7b | 2.42 (dd, 13.4, 9.1) | 1.91 (dd, 15.5, 7.2) | ||
| 8 | – | 87.9 s | – | 84.3 s |
| 9 | – | 76.2 s | – | 80.5 s |
| 10 | – | 53.6 s | – | 52.8 s |
| 11a | 2.01 (brtd, 14.6, 2.0) | 32.4 t | 1.86 (d, 5.7) | 24.4 t |
| 11b | 2.20 (d, 14.5) | 2.15 (ddd, 13.7, 11.4, 6.2) | ||
| 12a | 2.34 (dd, 13.8, 2.1) | 37.0 t | 2.44 (dd, 16.6, 6.2) | 36.2 t |
| 12b | 3.00 (m) | 2.75 (m) | ||
| 13 | – | 207.4 s | – | 207.2 s |
| 14a | 2.82 (d, 14.3) | 46.4 t | 2.47 (d, 14.4) | 47.0 t |
| 14b | 2.80 (d, 14.3) | 3.28 (d, 14.4) | ||
| 18 | 0.87 (s) | 32.4 q | 0.89 (s) | 32.2 q |
| 19 | 1.02 (s) | 20.8 q | 0.90 (s) | 20.0 q |
| 20 | – | 179.0 s | – | 179.3 s |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, K.; Zou, J.; Wang, Y.-X.; Zhao, C.-L.; Ye, J.-H.; Zhang, J.-J.; Pan, L.-T.; Zhang, H.-J. Rubesanolides F and G: Two Novel Lactone-Type Norditerpenoids from Isodon rubescens. Molecules 2021, 26, 3865. https://doi.org/10.3390/molecules26133865
He K, Zou J, Wang Y-X, Zhao C-L, Ye J-H, Zhang J-J, Pan L-T, Zhang H-J. Rubesanolides F and G: Two Novel Lactone-Type Norditerpenoids from Isodon rubescens. Molecules. 2021; 26(13):3865. https://doi.org/10.3390/molecules26133865
Chicago/Turabian StyleHe, Kang, Juan Zou, Yu-Xue Wang, Chen-Liang Zhao, Jiang-Hai Ye, Jing-Jie Zhang, Lu-Tai Pan, and Hong-Jie Zhang. 2021. "Rubesanolides F and G: Two Novel Lactone-Type Norditerpenoids from Isodon rubescens" Molecules 26, no. 13: 3865. https://doi.org/10.3390/molecules26133865
APA StyleHe, K., Zou, J., Wang, Y.-X., Zhao, C.-L., Ye, J.-H., Zhang, J.-J., Pan, L.-T., & Zhang, H.-J. (2021). Rubesanolides F and G: Two Novel Lactone-Type Norditerpenoids from Isodon rubescens. Molecules, 26(13), 3865. https://doi.org/10.3390/molecules26133865






