Synergistic Effect of Mandarin Peels and Hesperidin with Sodium Nitrite against Some Food Pathogen Microbes
Abstract
1. Introduction
2. Results and Discussion
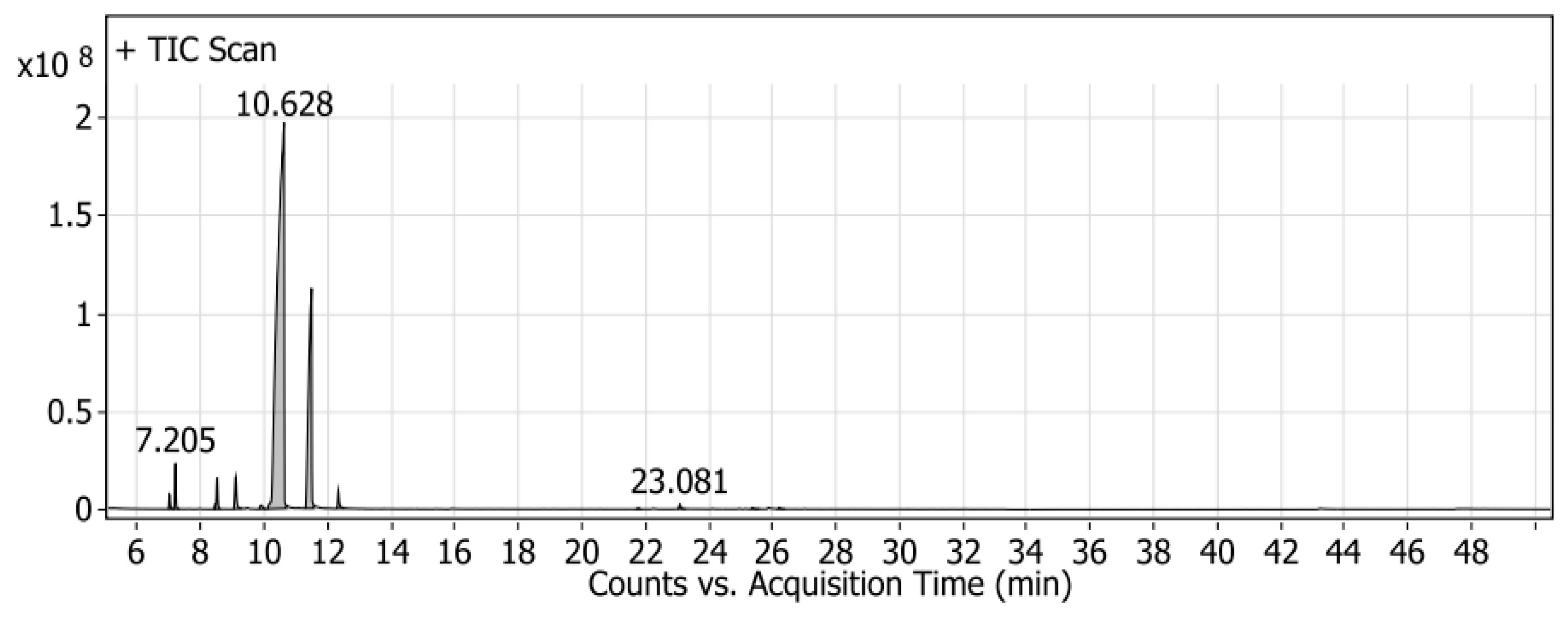
2.1. Identification of Mandarin Oil by GC/MS
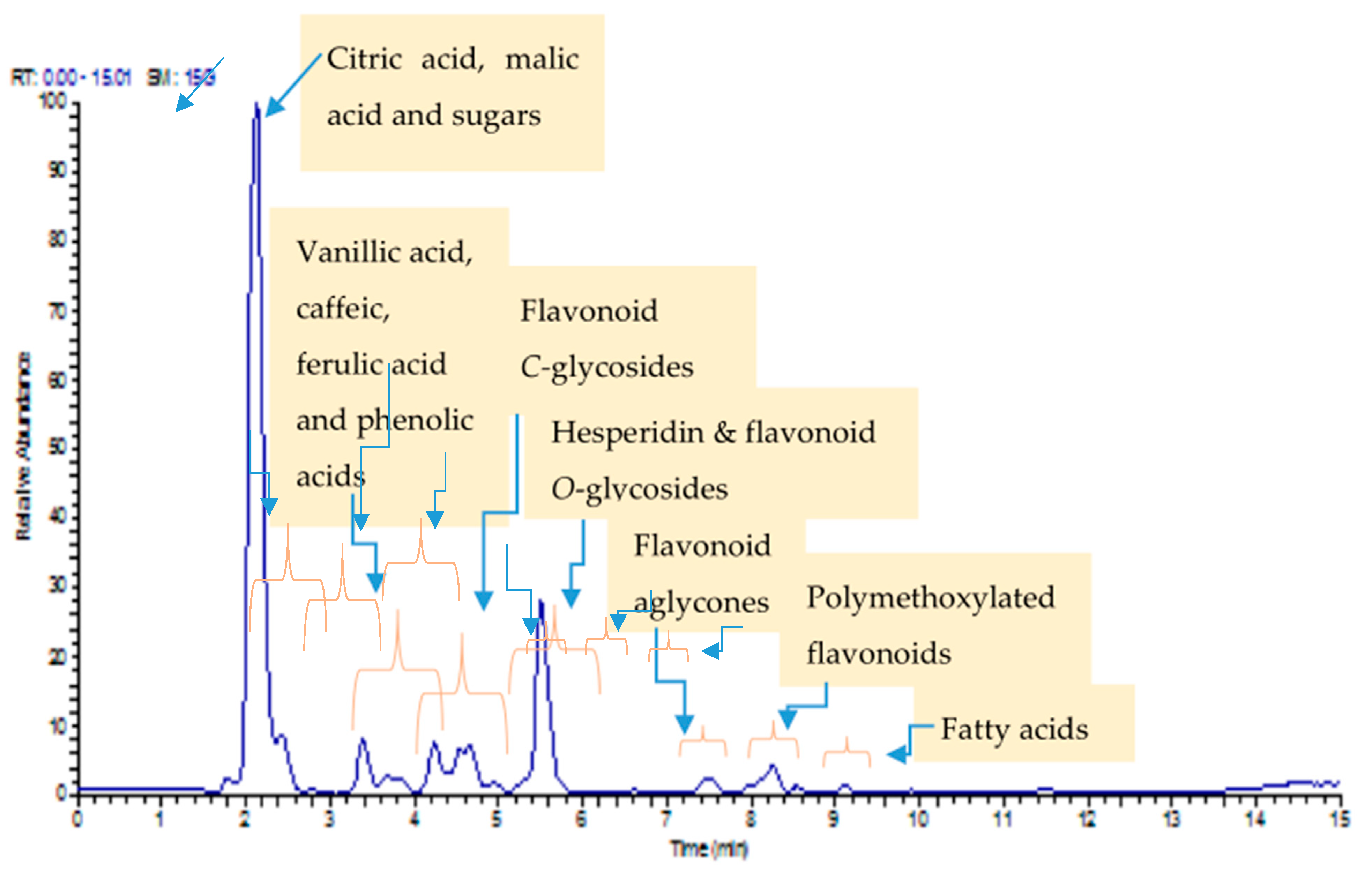
2.2. Identification of Mandarin Peel Chemical Constituents by UPLC/MS/MS
2.3. Antibacterial Activity of NaNO2, Mandarin Peel Extract, and Hesperidin
2.3.1. Minimum Inhibitory Concentration and Synergy Interactions of Mandarin Peel Extract with NaNO2
2.3.2. Minimum Inhibitory Concentration and Synergy Interactions of Hesperidin
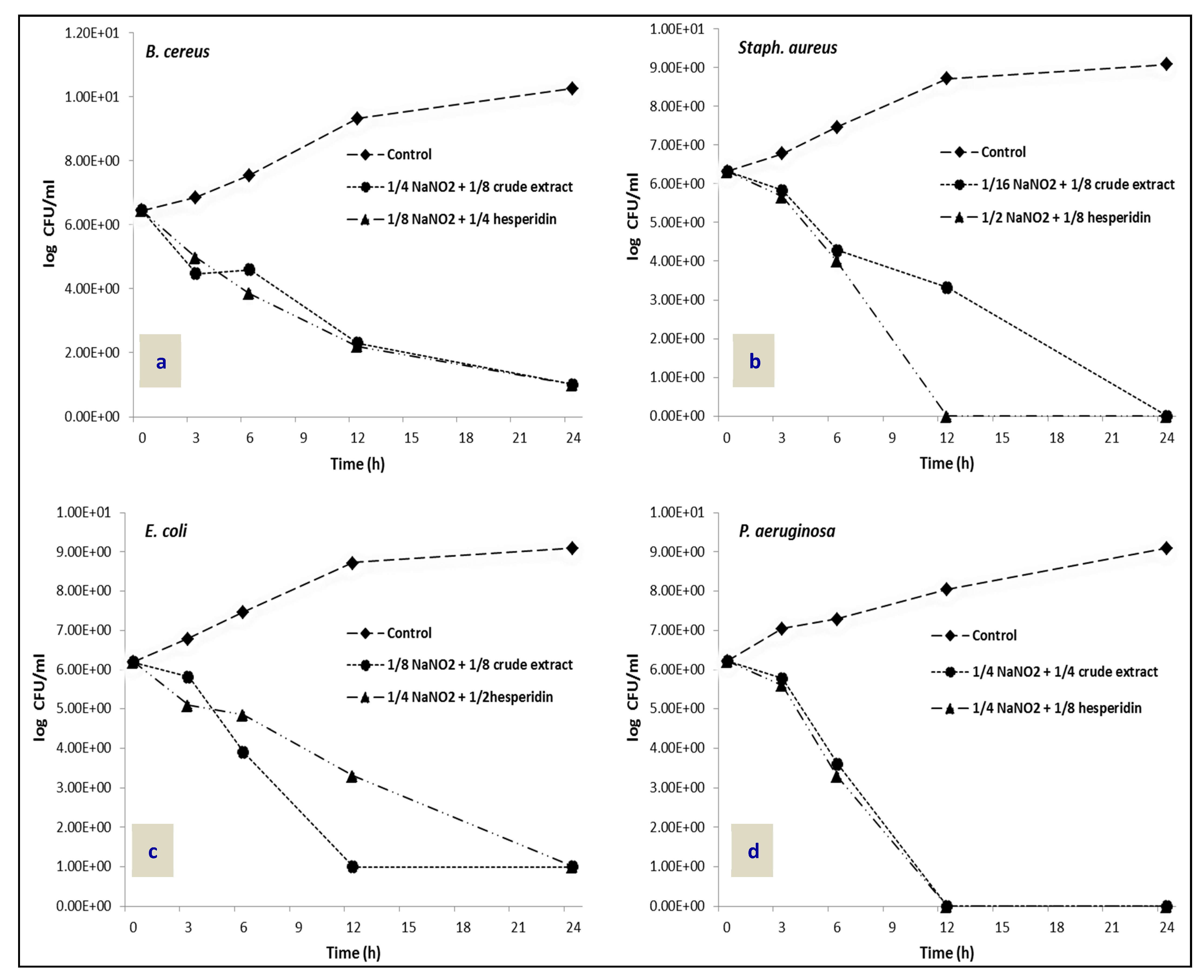
2.4. Time–Kill Assay
3. Materials and Methods
3.1. Plant Materials and Extraction
3.2. Gas Chromatography-Mass Spectrometry Analysis (GC/MS)
3.3. Ultra-Performance Liquid Chromatography-Mass Spectrometry Analysis (UPLC/MS/MS)
3.4. Isolation and Identification of Hesperidin
3.5. Nuclear Magnetic Resonance (NMR Analysis)
3.6. Antibacterial Activity Assay
3.6.1. Tested Bacteria Strains
3.6.2. Disc Diffusion Technique
3.7. Determination of Minimum Inhibitory Concentration (MIC)
3.8. Checkerboard Assay
3.9. Time–Kill Assay
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Helal, E.G.E.; Mustafa, R.A.A.; Mohamed, A.; El-Gamal, M.S. Adverse effects of two kinds of food additive mixtures (flavor enhancer, food preservative or food coloring agent) on physiological parameters in young male albino rats. Egypt. J. Hosp. Med. 2017, 67, 344–351. [Google Scholar] [CrossRef]
- Embaby, M.A.; El-Raey, M.A.; Zaineldain, M.; Almaghrabi, O.; Marrez, D.A. Synergistic effect and efflux pump inhibitory activity of Ficus nitida phenolic extract with tetracycline against some pathogenic bacteria. Toxin Rev. 2019, 1–11. [Google Scholar] [CrossRef]
- Marrez, D.A.; Sultan, Y.Y.; Embaby, M.A. Biological activity of the cyanobacterium Oscillatoria brevis extracts as a source of nutraceutical and bio-preservative agents. Int. J. Pharmacol. 2017, 13, 1010–1019. [Google Scholar] [CrossRef]
- Carocho, M.; Barreiro, M.F.; Morales, P.; Ferreira, I.C.F.R. Adding molecules to food, pros and cons: A review on synthetic and natural food additives. Compr. Rev. Food Sci. Food Saf. 2014, 13, 377–399. [Google Scholar] [CrossRef]
- Gyawali, R.; Ibrahim, S.A. Natural products as antimicrobial agents. Food Control 2014, 46, 412–429. [Google Scholar] [CrossRef]
- Xue, J.; Davidson, P.M.; Zhong, Q. Thymol nanoemulsified by whey protein-maltodextrin conjugates: The enhanced emulsifying capacity and antilisterial properties in milk by propylene glycol. J. Agric. Food Chem. 2013, 61, 12720–12726. [Google Scholar] [CrossRef] [PubMed]
- Tortora, F.; Notariale, R.; Maresca, V.; Good, K.V.; Sorbo, S.; Basile, A.; Piscopo, M.; Manna, C. Phenol-rich Feijoa sellowiana (pineapple guava) extracts protect human red blood cells from mercury-induced cellular toxicity. Antioxidants 2019, 8, 220. [Google Scholar] [CrossRef] [PubMed]
- Ultee, A.; Bennik, M.H.J.; Moezelaar, R. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl. Environ. Microbiol. 2002, 68, 1561–1568. [Google Scholar] [CrossRef]
- Piscopo, M.; Tenore, G.C.; Notariale, R.; Maresca, V.; Maisto, M.; De Ruberto, F.; Heydari, M.; Sorbo, S.; Basile, A. Antimicrobial and antioxidant activity of proteins from Feijoa sellowiana Berg. fruit before and after in vitro gastrointestinal digestion. Nat. Prod. Res. 2020, 34, 2607–2611. [Google Scholar] [CrossRef]
- Attia, G.H.; Alyami, H.S.; Orabi, M.A.A.; Gaara, A.H.; El Raey, M.A. Antimicrobial Activity of Silver and Zinc Nanoparticles Mediated by Eggplant Green Calyx. Int. J. Pharmacol. 2020, 16, 236–243. [Google Scholar] [CrossRef]
- Rios, J.-L.; Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol. 2005, 100, 80–84. [Google Scholar] [CrossRef]
- de la Rosa, J.D.P.; Ruiz-Palomino, P.; Arriola-Guevara, E.; García-Fajardo, J.; Sandoval, G.; Guatemala-Morales, G.M. A green process for the extraction and purification of hesperidin from mexican lime peel (Citrus aurantifolia Swingle) that is extendible to the citrus genus. Processes 2018, 6, 266. [Google Scholar] [CrossRef]
- Umagiliyage, A.L.; Becerra-Mora, N.; Kohli, P.; Fisher, D.J.; Choudhary, R. Antimicrobial efficacy of liposomes containing d-limonene and its effect on the storage life of blueberries. Postharvest Biol. Technol. 2017, 128, 130–137. [Google Scholar] [CrossRef]
- Iranshahi, M.; Rezaee, R.; Parhiz, H.; Roohbakhsh, A.; Soltani, F. Protective effects of flavonoids against microbes and toxins: The cases of hesperidin and hesperetin. Life Sci. 2015, 137, 125–132. [Google Scholar] [CrossRef]
- El Dib, R.A.; Soliman, H.S.M.; Hussein, M.H.; Attia, H.G. Two new flavonoids and biological activity of Astragalus abyssinicus (Hochst.) Steud. ex A. Rich. Aerial Parts. Drug Res. 2015, 65, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Gokoglu, N. Novel natural food preservatives and applications in seafood preservation: A review. J. Sci. Food Agric. 2019, 99, 2068–2077. [Google Scholar] [CrossRef] [PubMed]
- Dugo, P.; Bonaccorsi, I.; Ragonese, C.; Russo, M.; Donato, P.; Santi, L.; Mondello, L. Analytical characterization of mandarin (Citrus deliciosa Ten.) essential oil. Flavour Fragr. J. 2011, 26, 34–46. [Google Scholar] [CrossRef]
- Hąc-Wydro, K.; Flasiński, M.; Romańczuk, K. Essential oils as food eco-preservatives: Model system studies on the effect of temperature on limonene antibacterial activity. Food Chem. 2017, 235, 127–135. [Google Scholar] [CrossRef]
- Wang, M.; Carver, J.J.; Phelan, V.V.; Sanchez, L.M.; Garg, N.; Peng, Y.; Nguyen, D.D.; Watrous, J.; Kapono, C.A.; Luzzatto-Knaan, T.; et al. Sharing and community curation of mass spectrometry data with Global Natural Products Social Molecular Networking. Nat. Biotechnol. 2016, 34, 828–837. [Google Scholar] [CrossRef]
- Sobeh, M.; Hassan, S.A.; El Raey, M.A.; Khalil, W.A.; Hassan, M.A.E.; Wink, M. Polyphenolics from Albizia harveyi exhibit antioxidant activities and counteract oxidative damage and ultra-structural changes of cryopreserved bull semen. Molecules 2017, 22, 1993. [Google Scholar] [CrossRef]
- Emam, M.; Abdel-Haleem, D.R.; Salem, M.M.; Abdel-Hafez, L.J.; Latif, R.R.; Farag, S.M.; Sobeh, M.; El Raey, M.A. Phytochemical Profiling of Lavandula coronopifolia Poir. Aerial Parts Extract and Its Larvicidal, Antibacterial, and Antibiofilm Activity Against Pseudomonas aeruginosa. Molecules 2021, 26, 1710. [Google Scholar] [CrossRef]
- Taamalli, A.; Arráez-Román, D.; Abaza, L.; Iswaldi, I.; Fernández-Gutiérrez, A.; Zarrouk, M.; Segura-Carretero, A. LC-MS-based metabolite profiling of methanolic extracts from the medicinal and aromatic species Mentha pulegium and Origanum majorana. Phytochem. Anal. 2015, 26, 320–330. [Google Scholar] [CrossRef] [PubMed]
- Fang, N.; Yu, S.; Prior, R.L. LC/MS/MS characterization of phenolic constituents in dried plums. J. Agric. Food Chem. 2002, 50, 3579–3585. [Google Scholar] [CrossRef] [PubMed]
- El Raey, M.A.; El-Hagrassi, A.M.; Osman, A.F.; Darwish, K.M.; Emam, M. Acalypha wilkesiana flowers: Phenolic profiling, cytotoxic activity of their biosynthesized silver nanoparticles and molecular docking study for its constituents as Topoisomerase-I inhibitors. Biocatal. Agric. Biotechnol. 2019, 20, 101243. [Google Scholar] [CrossRef]
- González-Domínguez, R.; Urpi-Sarda, M.; Jáuregui, O.; Needs, P.W.; Kroon, P.A.; Andrés-Lacueva, C. Quantitative dietary fingerprinting (QDF)—A novel tool for comprehensive dietary assessment based on urinary nutrimetabolomics. J. Agric. Food Chem. 2019, 68, 1851–1861. [Google Scholar] [CrossRef] [PubMed]
- Fayek, N.M.; Farag, M.A.; Monem, A.R.A.; Moussa, M.Y.; Abd-Elwahab, S.M.; El-Tanbouly, N.D. Comparative metabolite profiling of four citrus peel cultivars via ultra-performance liquid chromatography coupled with quadrupole-time-of-flight-mass spectrometry and multivariate data analyses. J. Chromatogr. Sci. 2009, 57, 349–360. [Google Scholar] [CrossRef]
- Hegazi, N.M.; Sobeh, M.; Rezq, S.; El-Raey, M.A.; Dmirieh, M.; El-Shazly, A.M.; Mahmoud, M.F.; Wink, M. Characterization of phenolic compounds from Eugenia supra-axillaris leaf extract using HPLC-PDA-MS/MS and its antioxidant, anti-inflammatory, antipyretic and pain killing activities in vivo. Sci. Rep. 2019, 9, 1–12. [Google Scholar]
- Mencherini, T.; Campone, L.; Piccinelli, A.L.; Garcia Mesa, M.; Sánchez, D.M.; Aquino, R.P.; Rastrelli, L. HPLC-PDA-MS and NMR characterization of a hydroalcoholic extract of Citrus aurantium L. var. amara peel with antiedematogenic activity. J. Agric. Food Chem. 2013, 61, 1686–1693. [Google Scholar] [CrossRef]
- Ahmed, R.; Elkhrisy, E.; EL-kashak, W.A.; El Raey, M.; Nassar, M.; Aboutabl, E.S. Structural Characterization of Polyphenolics in Livistona chinensis Using HPLC-PDA-MS. J. Adv. Pharm. Res. 2019, 3, 23–29. [Google Scholar] [CrossRef]
- Szajwaj, B.; Moldoch, J.; Masullo, M.; Piacente, S.; Oleszek, W.; Stochmal, A. Amides and esters of phenylpropenoic acids from the aerial parts of Trifolium pallidum. Nat. Prod. Commun. 2011, 6, 1293–1296. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Z.; Zeng, S.L.; Wu, Y.; Zheng, G.D.; Chu, C.; Yin, Q.; Chen, B.Z.; Li, P.; Lu, X.; Liu, E.H. Cultivar differentiation of Citri Reticulatae Pericarpium by a combination of hierarchical three-step filtering metabolomics analysis, DNA barcoding and electronic nose. Anal. Chim. Acta 2019, 1056, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Fahmi, A.A.; El Raey, M.A.; Ibrahim, A.Y.; Abdelfattah, M.A.O.; Abdelmageed, A.M.; Sobeh, M. A sulfated polyphenols-rich extract from Sabal yapa exhibits antitumor activities in Ehrlich ascites carcinoma. Saudi J. Biol. Sci. 2021, 28, 3117–3125. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G.-D.; Sun, C.-F.; Pu, J.-W.; Chen, J.; Jiang, X.-Y.; Zou, S.-M. Two myostatin genes exhibit divergent and conserved functions in grass carp (Ctenopharyngodon idellus). Gen. Com. Endocrinol. 2015, 214, 68–76. [Google Scholar] [CrossRef]
- Zhang, B.; Schmoyer, D.; Kirov, S.; Snoddy, J. GOTree Machine (GOTM): A web-based platform for interpreting sets of interesting genes using Gene Ontology hierarchies. BMC Bioinform. 2004, 5, 1–8. [Google Scholar]
- Janek, T.; Krasowska, A.; Czyżnikowska, Ż.; Łukaszewicz, M. Trehalose lipid biosurfactant reduces adhesion of microbial pathogens to polystyrene and silicone surfaces: An experimental and computational approach. Front. Microbiol. 2018, 9, 2441. [Google Scholar] [CrossRef]
- Pratiwi, F.; Tinata, J.K.; Prakasa, A.W.; Hartini, E.; Isworo, S. Citric acid compounds of tangerines peel extract (Citrus reticulata) as potential materials teeth whitening. J. Phys. Conf. Ser. 2017, 824, 12071. [Google Scholar] [CrossRef]
- Braïek, O.B.; Smaoui, S. Chemistry, Safety, and Challenges of the Use of Organic Acids and Their Derivative Salts in Meat Preservation. J. Food Qual. 2021, 2021, 1–20. [Google Scholar] [CrossRef]
- Coban, H.B. Organic acids as antimicrobial food agents: Applications and microbial productions. Bioprocess Biosyst. Eng. 2020, 43, 569–591. [Google Scholar] [CrossRef]
- Nićiforović, N.; Abramovič, H. Sinapic acid and its derivatives: Natural sources and bioactivity. Compr. Rev. Food Sci. Food Saf. 2014, 13, 34–51. [Google Scholar] [CrossRef]
- Sova, M. Antioxidant and antimicrobial activities of cinnamic acid derivatives. Mini Rev. Med. Chem. 2012, 12, 749–767. [Google Scholar] [CrossRef]
- Barreca, D.; Mandalari, G.; Calderaro, A.; Smeriglio, A.; Trombetta, D.; Felice, M.R.; Gattuso, G. Citrus flavones: An update on sources, biological functions, and health promoting properties. Plants 2020, 9, 288. [Google Scholar] [CrossRef] [PubMed]
- Ke, Z.; Yang, Y.; Tan, S.; Zhou, Z. Characterization of polymethoxylated flavonoids in the peels of Chinese wild mandarin (Citrus reticulata Blanco) by UPLC-Q-TOF-MS/MS. Food Anal. Methods 2017, 10, 1328–1338. [Google Scholar] [CrossRef]
- Attia, G.H.; Moemen, Y.S.; Youns, M.; Ibrahim, A.M.; Abdou, R.; El Raey, M.A. Antiviral zinc oxide nanoparticles mediated by hesperidin and in silico comparison study between antiviral phenolics as anti-SARS-CoV-2. Colloids Surf. B Biointerfaces 2021, 203, 111724. [Google Scholar] [CrossRef]
- Espina, L.; Gelaw, T.K.; de Lamo-Castellví, S.; Pagán, R.; García-Gonzalo, D. Mechanism of bacterial inactivation by (+)-limonene and its potential use in food preservation combined processes. PLoS ONE 2013, 8, e56769. [Google Scholar] [CrossRef] [PubMed]
- Abuelsaad, A.S.A.; Mohamed, I.; Allam, G.; Al-Solumani, A.A. Antimicrobial and immunomodulating activities of hesperidin and ellagic acid against diarrheic Aeromonas hydrophila in a murine model. Life Sci. 2013, 93, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Eliuz, E. Antimicrobial activity of citric acid against Escherichia coli, Staphylococcus aureus and Candida albicans as a sanitizer agent. Eurasian J. For. Sci. 2020, 8, 295–301. [Google Scholar] [CrossRef]
- Kjer, J.; Debbab, A.; Aly, A.H.; Proksch, P. Methods for isolation of marine-derived endophytic fungi and their bioactive secondary products. Nat. Protoc. 2010, 5, 479–490. [Google Scholar] [CrossRef]
- Ammar, N.M.; Hassan, H.A.; Mohammed, M.A.; Serag, A.; Abd El-Alim, S.H.; Elmotasem, H.; El Raey, M.; El Gendy, A.N.; Sobeh, M.; Abdel-Hamid, A.H. Metabolomic profiling to reveal the therapeutic potency of Posidonia oceanica nanoparticles in diabetic rats. RSC Adv. 2021, 11, 8398–8410. [Google Scholar] [CrossRef]
- Osman, S.M.; El Kashak, W.A.; Wink, M.; El Raey, M.A. New isorhamnetin derivatives from Salsola imbricata Forssk. leaves with distinct anti-inflammatory activity. Pharmacogn. Mag. 2016, 12, S47. [Google Scholar]
- Bauer, R.A. Social Indicators; MIT Press: Cambridge, MA, USA, 1966. [Google Scholar]
- Marrez, D.A.; Abdelhamid, A.E.; Darwesh, O.M. Eco-friendly cellulose acetate green synthesized silver nano-composite as antibacterial packaging system for food safety. Food Packag. Shelf Life 2019, 20, 100302. [Google Scholar] [CrossRef]
- Andrews, G.; Issakidis, C.; Carter, G. Shortfall in mental health service utilisation. Br. J. Psychiatry 2001, 179, 417–425. [Google Scholar] [CrossRef] [PubMed]
- White, M.; Hu, W. The sachs-wolfe effect. arXiv 1996, arXiv:astro-ph/9609105. Available online: https://arxiv.org/abs/astro-ph/9609105 (accessed on 16 September 1996).
- Tallarida, R.J. Drug synergism: Its detection and applications. J. Pharmacol. Exp. Ther. 2001, 298, 865–872. [Google Scholar] [PubMed]
- Bansal, T.; Alaniz, R.C.; Wood, T.K.; Jayaraman, A. The bacterial signal indole increases epithelial-cell tight-junction resistance and attenuates indicators of inflammation. Proc. Natl. Acad. Sci. USA 2010, 107, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Tomaino, A.; Rich, G.T.; Curto, R.L.; Arcoraci, T.; Martorana, M.; Bisignano, C.; Saija, A.; Parker, M.L.; Waldron, K.W.; et al. Polyphenol and nutrient release from skin of almonds during simulated human digestion. Food Chem. 2010, 122, 1083–1088. [Google Scholar] [CrossRef]
- Petersen, P.; Axler, S.; Ribet, K.A. Riemannian Geometry; Springer: New York, NY, USA, 2006; Volume 171. [Google Scholar]
| Peak | RT | Name | Formula | Area | Area Sum % |
|---|---|---|---|---|---|
| 1 | 7.016 | l-Phellandrene | C10H16 | 18,336,447 | 0.47 |
| 2 | 7.205 | α-(-)-pinene | C10H16 | 53,171,439.3 | 1.37 |
| 3 | 8.453 | 3-Carene | C10H16 | 5,287,840.96 | 0.14 |
| 4 | 8.519 | 2-.β-pinene | C10H16 | 44,154,069.3 | 1.13 |
| 5 | 9.096 | β-myrcene | C10H16 | 59,501,702.7 | 1.53 |
| 6 | 9.9 | 2-Carene | C10H16 | 11,948,052.3 | 0.31 |
| 7 | 10.628 | D-Limonene | C10H16 | 2,928,075,732 | 75.21 |
| 8 | 11.488 | ɤ-Terpinene | C10H16 | 725,626,383 | 18.64 |
| 9 | 12.33 | α-Terpinolene | C10H16 | 30,695,286.9 | 0.79 |
| 10 | 21.767 | α-ylangene | C15H24 | 2,130,866.43 | 0.05 |
| 11 | 23.081 | Caryophyllene | C15H24 | 8,035,838.48 | 0.21 |
| 12 | 25.369 | 1H-Cycloprop[e]azulene, 1a,2,3,5,6,7,7a,7b-octahydro-1,1,4,7-tetramethyl-, [1aR-(1a.alpha.,7.alpha.,7a.beta.,7b.alpha.)]- | C15H24 | 3,360,910.92 | 0.09 |
| 13 | 26.211 | β-copaene | C15H24 | 3,051,328.78 | 0.08 |
| No | RT [Min] | Metabolite Identification | Chemical Formula | [M − H]− | Ref. | |
|---|---|---|---|---|---|---|
| Measured | Fragmentation | |||||
| 1 | 2.03 | Trehalose | C12H22O11 | 341.1092 | 179.0552 | [19] |
| 2 | 2.08 | Citric acid | C7H11O6 | 191.0552 | 173.0445 | [19] |
| 3 | 2.15 | Hexose | C6H12O6 | 179.0550 | 161.0443 | [19] |
| 4 | 2.16 | Malic acid | C4H6O5 | 133.0128 | 115.0022 | [20] |
| 6 | 3.21 | Tryptophan | C11H12N2O2 | 203.0818 | 186.0546, 159.0915, 142.0650, 116.0491 | [21] |
| 7 | 3.39 | Vanillic acid hexoside | C14H18O9 | 329.0879 | 167.0337 | [22] |
| 8 | 3.41 | Caffeic acid | C9H8O4 | 179.0551 | 134.9866 | [23] |
| 9 | 4.25 | Feruloyl quinic acid | C17H20O9 | 367.1033 | 193.0497, 191.0185 | [24] |
| 10 | 4.29 | Sinapic acid hexouronide | C17H20O11 | 399.0932 | 223.0462, 193.0497 | [25] |
| 11 | 4.46 | Apigenin-di-C-hexoside (vicenin 2) | C27H30O15 | 593.1360 | 503.1203, 473.1094, 383.0774, 353.0667 | [26] |
| 12 | 4.48 | Sinapic acid hexoside | C17H22O10 | 385.1854 | 223.1331 | [27] |
| 13 | 4.73 | Meranzin hydrate | C15H18O5 | 277.1080 | 259.0951, 233.1181, 215.1074, 189.9480, 87.0070 | [28] |
| 14 | 4.75 | Methoxyluteolin di-C-hexoside | C30H38O16 | 623.1752 | 533.1318, 503.1197, 413.6878, 383.0773 | [26] |
| 15 | 4.87 | Syringic acid | C9 H10 O5 | 197.0446 | 169.0130, 125.0227 | [22] |
| 16 | 4.89 | Orientin | C21H20O11 | 447.0935 | 357.0616, 327.0511 | [19] |
| 17 | 5.28 | Vitexin | C21H20O10 | 431.0985 | 341.0668, 311.0562 | [29] |
| 18 | 5.47 | Naringenin-7-O-rutinoside | C27H32O14 | 579.1774 | 271.0612 | [19] |
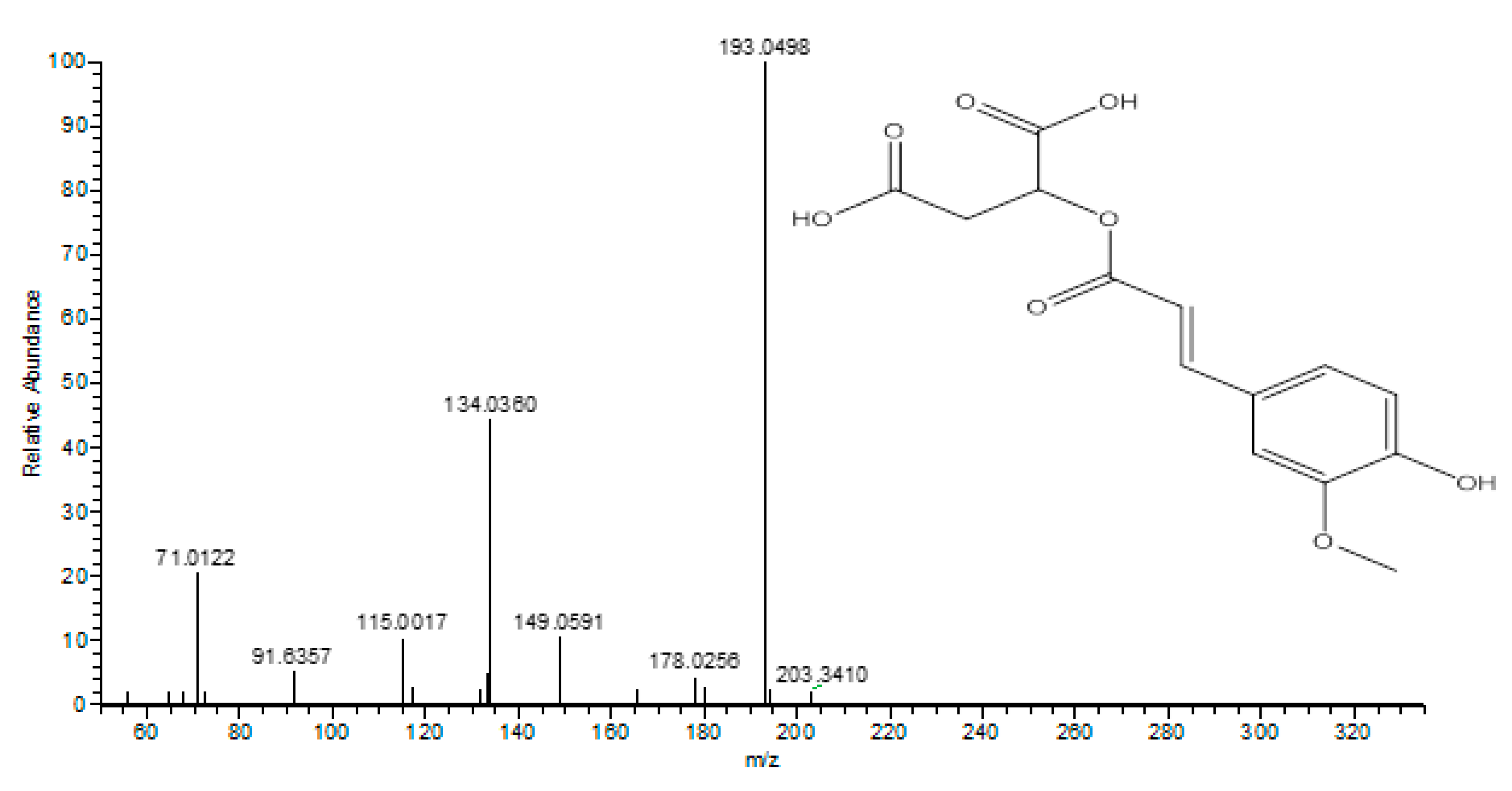
| 19 | 5.48 | Feruloyl-O-malic acid ester | C14H13O8 | 309.0616 | 193.0498, 134.0360, 115.0017 | [30] |
| 20 | 5.55 | Hesperidin | C28H34O15 | 609.1814 | 475.2522, 430.9161, 367.2440, 301.0651 | [31] |
| 21 | 5.52 | Isosakuranetin-7-O-neohesperidoside | C28H34O14 | 593.1551 | 285.0405 | [26] |
| 22 | 5.59 | Diosmetin-C-hexoside | C22H22O11 | 461.1085 | 371.0770, 341.0664, 298.0481 | [32] |
| 23 | 5.68 | Naringenin-7-O-Hexoside | C21H22O10 | 433.1142 | 271.0614 | [26] |
| 24 | 5.79 | Rhiofolin | C27H29O14 | 577.0258 | 269.0456 | [33] |
| 25 | 5.88 | Isorhamnetin-3-O-hexoside | C22H22O12 | 477.1044 | 315.0505, 314.0432, 300.0292 | [34] |
| 26 | 7.41 | Diosmetin | C16H12O6 | 299.0550 | 284.0326 | [19] |
| 27 | 7.51 | hesperitin | C16H14O6 | 301.0618 | 286.0382 | [19] |
| 28 | 8.04 | Dihydroxy trimethoxy flavone | C18H15O7 | 343.0823 | 328.0589, 313.0355, 298.0126, 285.0407 | [26] |
| 29 | 8.10 | Dihydroxy dimethoxy flavone | C17H13O7 | 329.0328 | 314.0423, 300.509, 299.0182 | [26] |
| 30 | 8.23 | Dihydroxy tetramethoxy flavone | C19H18O8 | 373.0932 | 358.0694, 343.0458, 328.0222, 300.0268 | [26] |
| 31 | 8.25 | Dihydroxy trimethoxy flavone | C18H15O7 | 343.0819 | 328.0589, 313.0355, 298.0126, 285.0407 | [26] |
| 32 | 9.12 | Hydroxy-hexadecanoic acid | C16H31O3 | 271.1914 | 253.1805, 209.1901 | [26] |
| 33 | 10.25 | Linolenic acid | C18H30O2 | 277.2170 | 233.1541, 205.1590, 59.0121 | [31] |
| Bacteria | Inhibition Zone, mm (Mean ± S.E) | |||
|---|---|---|---|---|
| Negative Control | NaNO2 1 mg mL−1 | Mandarin Peel Extract 10 mg mL−1 | Hesperidin 10 mg mL−1 | |
| B. cereus | 0 | 15.2 ± 1.04 a | 10.2 ± 0.81 b | 8.8 ± 0.76 c |
| Staph. aureus | 0 | 17.2 ± 0.76 a | 9.3 ± 0.58 b | 9.8 ± 1.25 b |
| E. coli | 0 | 11.8 ± 0.76 c | 16.7 ± 2.46 a | 15.8 ± 0.86 b |
| P. aeruginosa | 0 | 19.3 ± 1.04 a | 13.0 ± 0.50 b | 10.8 ± 0.86 c |
| Bacteria | MICA mg mL−1 | MICB mg mL−1 | FICA | FICB | FIC Index | Interaction |
|---|---|---|---|---|---|---|
| B. cereus | 1.67 | 0.93 | 0.25 | 0.12 | 0.37 | S |
| Staph. aureus | 0.92 | 1.13 | 0.06 | 0.12 | 0.18 | S |
| E. coli | 4.76 | 0.77 | 0.13 | 0.12 | 0.25 | S |
| P. aeruginosa | 0.67 | 1.03 | 0.25 | 0.25 | 0.5 | S |
| Bacteria | MICA mg mL−1 | MICB mg mL−1 | FICA | FICB | FIC Index | Interaction |
|---|---|---|---|---|---|---|
| B. cereus | 1.67 | 1.33 | 0.13 | 0.25 | 0.37 | S |
| Staph. aureus | 0.92 | 1.53 | 0.5 | 0.13 | 0.63 | A |
| E. coli | 4.76 | 1.13 | 0.25 | 0.5 | 0.75 | A |
| P. aeruginosa | 0.67 | 1.27 | 0.25 | 0.13 | 0.37 | S |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Attia, G.H.; Marrez, D.A.; Mohammed, M.A.; Albarqi, H.A.; Ibrahim, A.M.; Raey, M.A.E. Synergistic Effect of Mandarin Peels and Hesperidin with Sodium Nitrite against Some Food Pathogen Microbes. Molecules 2021, 26, 3186. https://doi.org/10.3390/molecules26113186
Attia GH, Marrez DA, Mohammed MA, Albarqi HA, Ibrahim AM, Raey MAE. Synergistic Effect of Mandarin Peels and Hesperidin with Sodium Nitrite against Some Food Pathogen Microbes. Molecules. 2021; 26(11):3186. https://doi.org/10.3390/molecules26113186
Chicago/Turabian StyleAttia, Gouda H., Diaa A. Marrez, Mona A. Mohammed, Hassan A. Albarqi, Ammar M. Ibrahim, and Mohamed A. El Raey. 2021. "Synergistic Effect of Mandarin Peels and Hesperidin with Sodium Nitrite against Some Food Pathogen Microbes" Molecules 26, no. 11: 3186. https://doi.org/10.3390/molecules26113186
APA StyleAttia, G. H., Marrez, D. A., Mohammed, M. A., Albarqi, H. A., Ibrahim, A. M., & Raey, M. A. E. (2021). Synergistic Effect of Mandarin Peels and Hesperidin with Sodium Nitrite against Some Food Pathogen Microbes. Molecules, 26(11), 3186. https://doi.org/10.3390/molecules26113186





