An Overview on Graphene-Metal Oxide Semiconductor Nanocomposite: A Promising Platform for Visible Light Photocatalytic Activity for the Treatment of Various Pollutants in Aqueous Medium
Abstract
1. Introduction
2. Photocatalysis
2.1. Earth-Abundant Metal Oxide-GO/rGO Composites
2.1.1. rGO-WO3 Composites
2.1.2. rGO-Co3O4 Composites
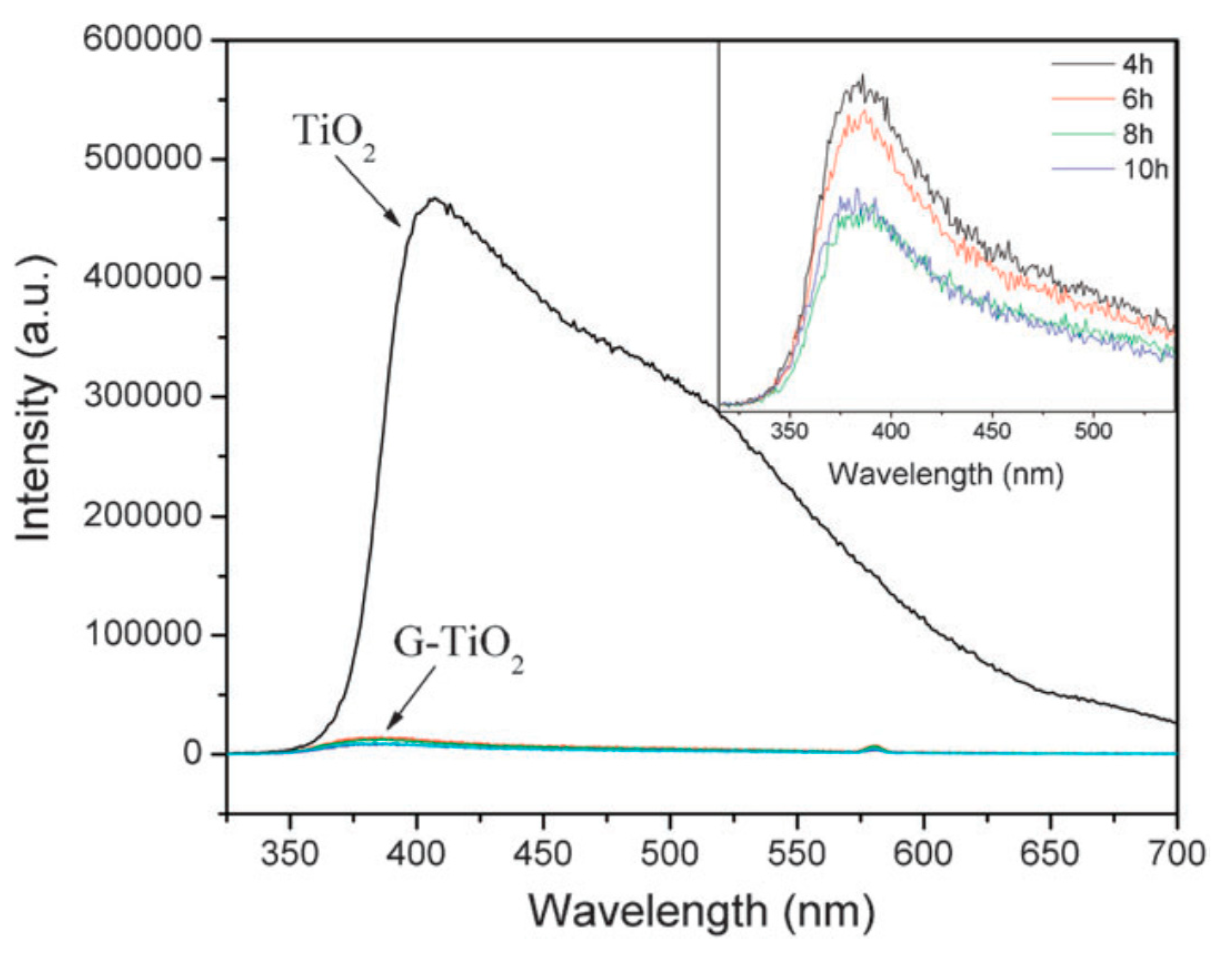
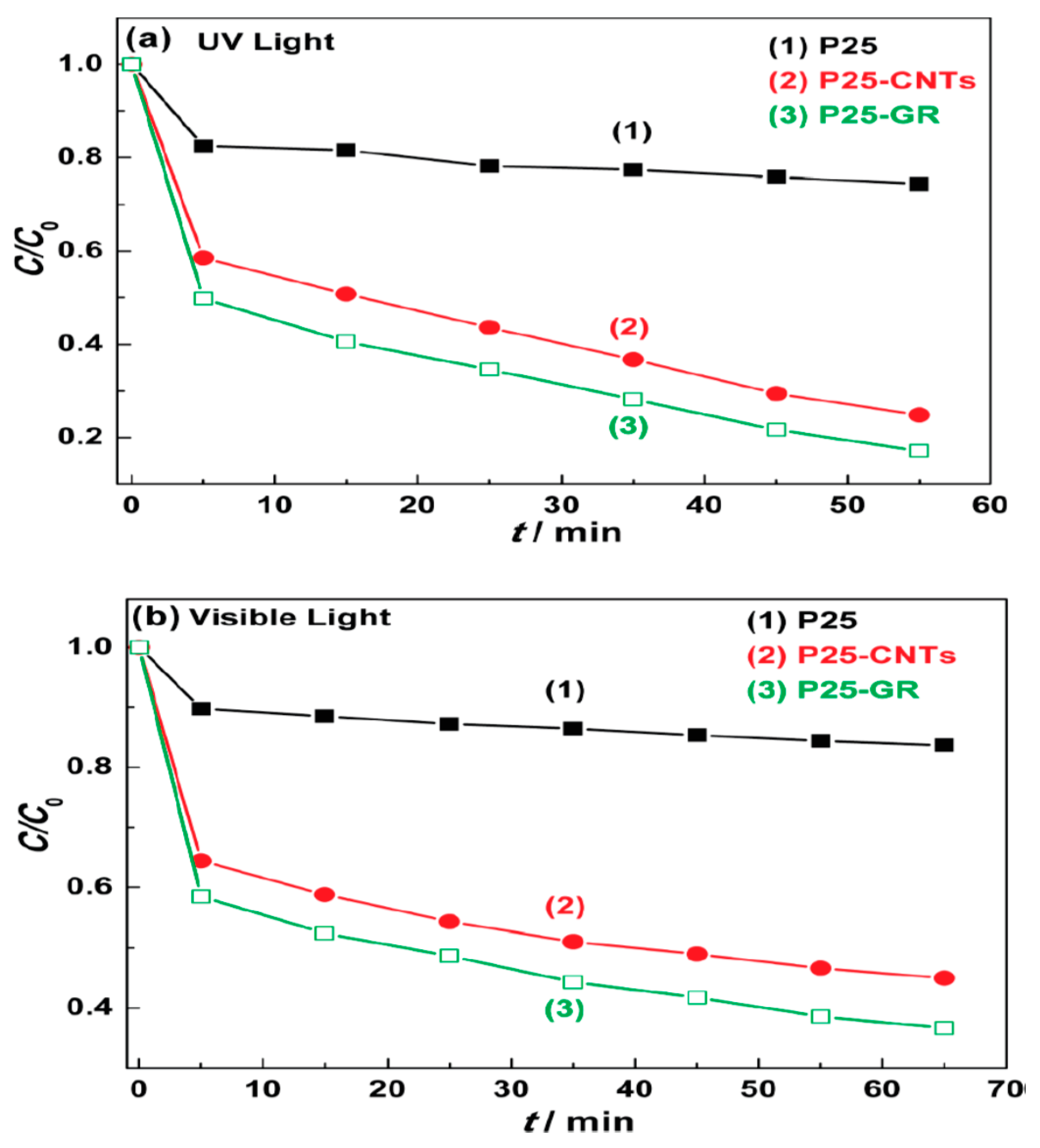
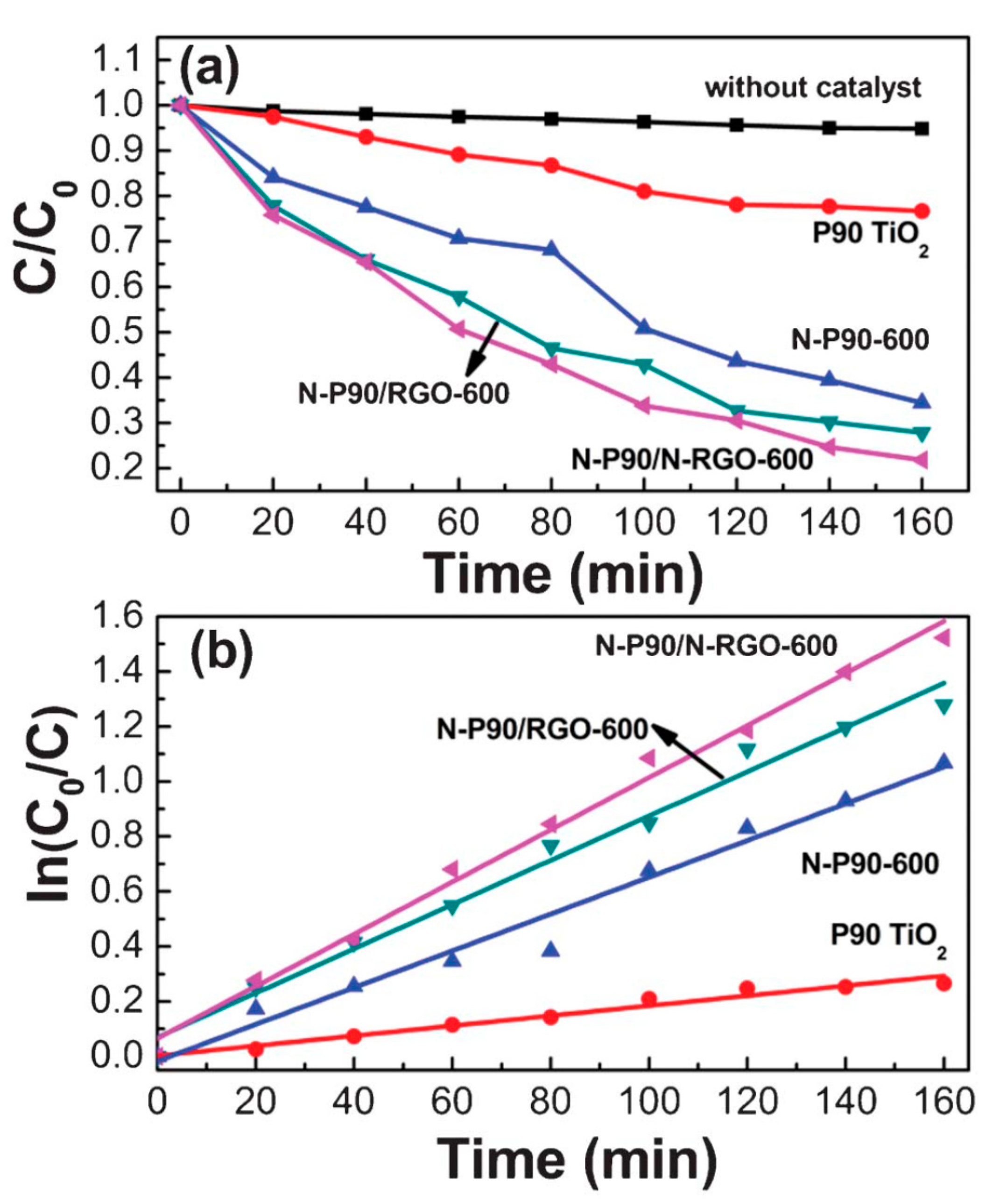
2.1.3. GO/rGO-TiO2 Composites
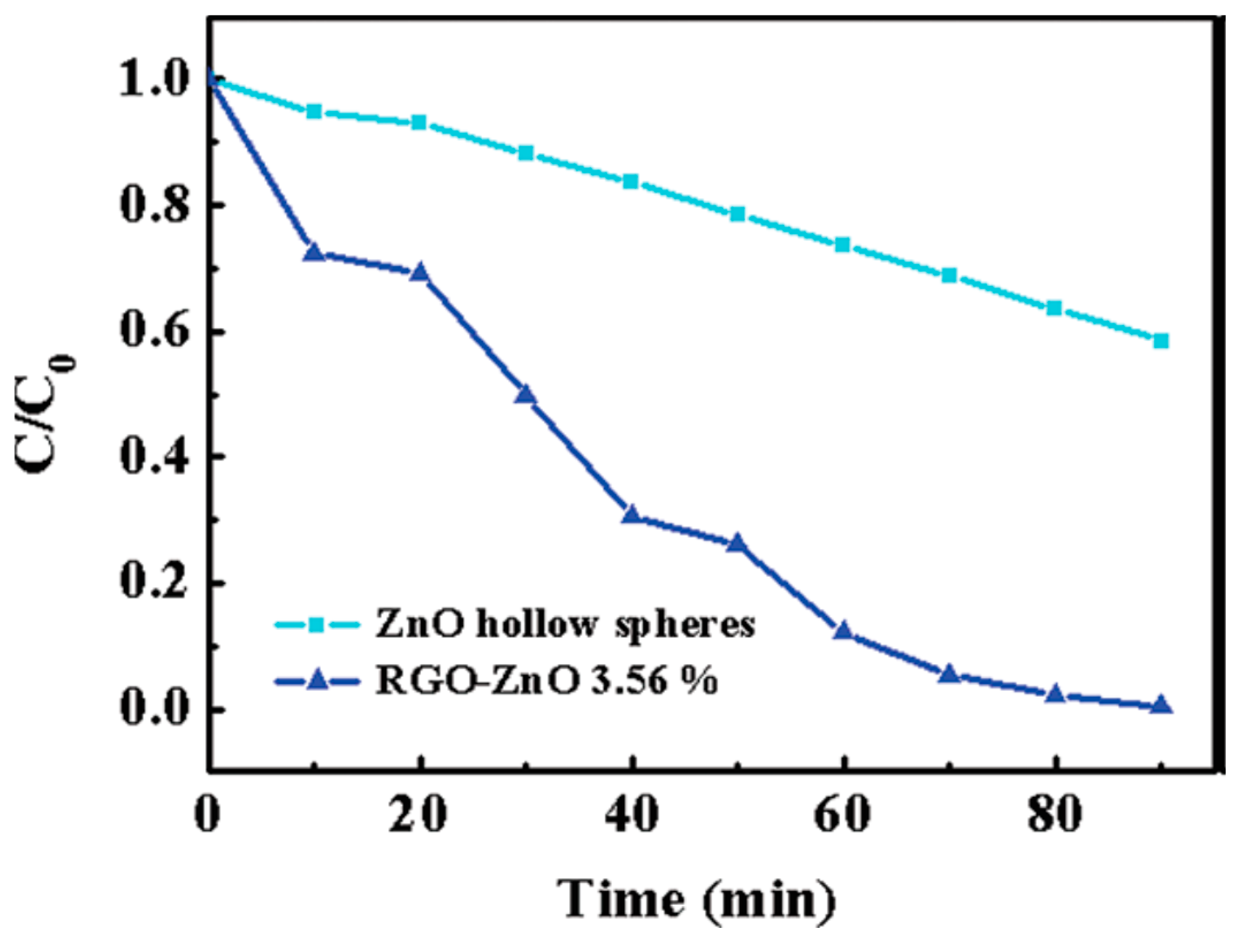
2.1.4. GO/rGO-ZnO Composite
2.2. Bimetal Oxide-GO/rGO Composites
2.2.1. GO/rGO-CoFe2O4 Composite
2.2.2. GO-rGO-ZnFe2O4 Composite
2.2.3. GO/rGO-NiFe2O4 and MnFe2O4 Composites
2.2.4. Other Composite Systems
3. Photocatalytic Evaluation
4. Perspectives and Challenges
Author Contributions
Funding
Conflicts of Interest
References
- Deng, X.; Lü, L.; Li, H.; Luo, F. The adsorption properties of Pb(II) and Cd(II) on functionalized graphene prepared by electrolysis method. J. Hazard. Mater. 2010, 183, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-T.; Chang, Y.; Wang, H.; Liu, G.; Chen, S.; Wang, Y.; Liu, Y.; Cao, A. Folding/aggregation of graphene oxide and its application in Cu2+ removal. J. Colloid Interface Sci. 2010, 351, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Qiu, H.; Si, Y.; Wang, W.; Gao, J. Fabrication of highly porous biodegradable monoliths strengthened by graphene oxide and their adsorption of metal ions. Carbon 2011, 49, 827–837. [Google Scholar] [CrossRef]
- Yeh, T.-F.; Syu, J.-M.; Cheng, C.; Chang, T.-H.; Teng, H. Graphite Oxide as a Photocatalyst for Hydrogen Production from Water. Adv. Funct. Mater. 2010, 20, 2255–2262. [Google Scholar] [CrossRef]
- Zhao, G.; Wen, T.; Chen, C.; Wang, X. Synthesis of graphene-based nanomaterials and their application in energy-related and environmental-related areas. RSC Adv. 2012, 2, 9286–9303. [Google Scholar] [CrossRef]
- Li, F.; Tian, X.; Yuhong, Z.; Jun, Y.; Aiwu, W.; Zhong, W. Preparation of WO3–reduced graphene oxide nanocomposites with Enhanced Photocatalytic Property. Ceram. Int. 2015, 41, 5903–5908. [Google Scholar]
- Lorestani, F.; Shahnavaz, Z.; Mn, P.; Alias, Y.; Manan, N.S.A. One-step hydrothermal green synthesis of silver nanoparticle-carbon nanotube reduced-graphene oxide composite and its application as hydrogen peroxide sensor. Sens. Actuators B-Chem. 2015, 208, 389–398. [Google Scholar] [CrossRef]
- Yu, B.; Xu, J.; Liu, J.-H.; Yang, S.-T.; Luo, J.; Zhou, Q.; Wan, J.; Liao, R.; Wang, H.; Liu, Y. Adsorption behavior of copper ions on graphene oxide–chitosan aerogel. J. Environ. Chem. Eng. 2013, 1, 1044–1050. [Google Scholar] [CrossRef]
- Fu, Y.; Wang, J.; Liu, Q.; Zeng, H. Water-dispersible magnetic nanoparticle–graphene oxide composites for selenium removal. Carbon 2014, 77, 710–721. [Google Scholar] [CrossRef]
- Li, B.; Liu, T.; Wang, Y.; Wang, Z. ZnO/graphene-oxide nanocomposite with remarkably enhanced visible-light-driven photocatalytic performance. J. Colloid Interface Sci. 2012, 377, 114–121. [Google Scholar] [CrossRef]
- Wu, S.; Wang, P.; Cai, Y.; Liang, D.; Ye, Y.; Tian, Z.; Liu, J.; Liang, C. Reduced graphene oxide anchored magnetic ZnFe2O4 nanoparticles with enhanced visible-light photocatalytic activity. RSC Adv. 2015, 5, 9069–9074. [Google Scholar] [CrossRef]
- Tan, L.-L.; Chai, S.-P.; Mohamed, A.R. Synthesis and Applications of Graphene-Based TiO2 Photocatalysts. Chem. Sus. Chem. 2012, 5, 1868–1882. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Du, Q.; Liu, T.; Peng, X.; Wang, J.; Sun, J.; Wang, Y.; Wu, S.; Wang, Z.; Xia, Y.; et al. Comparative study of methylene blue dye adsorption onto activated carbon, graphene oxide, and carbon nanotubes. Chem. Eng. Res. Des. 2013, 91, 361–368. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-Graphene Composite as a High Performance Photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Yu, J.C. Graphene-based photocatalytic composites. RSC Adv. 2011, 1, 1426–1434. [Google Scholar] [CrossRef]
- Du, J.; Lai, X.; Yang, N.; Zhai, J.; Kisailus, D.; Su, F.; Wang, D.; Jiang, L. Hierarchically Ordered Macro−Mesoporous TiO2−Graphene Composite Films: Improved Mass Transfer, Reduced Charge Recombination, and Their Enhanced Photocatalytic Activities. ACS Nano 2011, 5, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Choi, D.; Li, J.; Yang, Z.; Nie, Z.; Kou, R.; Hu, D.; Wang, C.; Saraf, L.V.; Zhang, J.; et al. Self-Assembled TiO2–Graphene Hybrid Nanostructures for Enhanced Li-Ion Insertion. ACS Nano 2009, 3, 907–914. [Google Scholar] [CrossRef]
- Wenyu, Z.; Faqian, S.; Ronn, G.; Yan, Z. Facile fabrication of RGO-WO3 composites for effective visible lightphotocatalytic degradation of sulfamethoxazole. Appl. Catal. B 2017, 207, 93–102. [Google Scholar]
- Hajishafiee, H.; Sangpour, P.; Tabrizi, N.S. Facile Synthesis and Photocatalytic Performance of WO3/rGO Nanocomposite for Degradation of 1-Naphthol. Nano 2015, 10, 1550072. [Google Scholar] [CrossRef]
- Hu, X.; Xu, P.; Gong, H.; Yin, G. Synthesis and characterization of WO3/Graphene nanocomposites for enhanced photocatalytic activities by one-step in-situ hydrothermal reaction. Materials 2018, 11, 147. [Google Scholar]
- Wang, W.; Levi, G.; Tade, M.O.; Li, Q. April. Evaluation of Photocatalytic Activity of Co3O4/Graphene Composite. In 2015 International Conference on Materials, Environmental and Biological Engineering; Atlantis Press: Paris, France, 2015; pp. 561–564. [Google Scholar]
- Pervaiz, E.; Liu, H.; Yang, M. Facile synthesis and enhanced photocatalytic activity of single-crystalline nanohybrids for the removal of organic pollutants. Nanotechnology 2017, 28, 105701. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Yang, Z.; Sun, H.; Wang, S. Hydrothermal synthesis of Co3O4–graphene for heterogeneous activation of peroxymonosulfate for decomposition of phenol. Ind. Eng. Chem. Res. 2012, 51, 14958–14965. [Google Scholar] [CrossRef]
- Štengl, V.; Bakardjieva, S.; Grygar, T.M.; Bludská, J.; Kormunda, M. TiO2-graphene oxide nanocomposite as advanced photocatalytic materials. Chem. Cent. J. 2013, 7, 41. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tang, Z.R.; Fu, X.; Xu, Y.J. TiO2−graphene nanocomposites for gas-phase photocatalytic degradation of volatile aromatic pollutant: Is TiO2−graphene truly different from other TiO2−carbon composite materials? ACS Nano 2010, 4, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Zhu, Y.; Yang, X.; Jiang, X.; Li, C. Preparation of graphene–TiO2 composites with enhanced photocatalytic activity. New J. Chem. 2011, 35, 353–359. [Google Scholar] [CrossRef]
- Anandan, S.; Narasinga Rao, T.; Sathish, M.; Rangappa, D.; Honma, I.; Miyauchi, M. Superhydrophilic graphene-loaded TiO2 thin film for self-cleaning applications. ACS Appl. Mater. Interfaces 2013, 5, 207–212. [Google Scholar] [CrossRef]
- Nguyen-Phan, T.D.; Pham, V.H.; Shin, E.W.; Pham, H.D.; Kim, S.; Chung, J.S.; Kim, E.J.; Hur, S.H. The role of graphene oxide content on the adsorption-enhanced photocatalysis of titanium dioxide/graphene oxide composites. Chem. Eng. J. 2011, 170, 226–232. [Google Scholar] [CrossRef]
- Min, Y.; Zhang, K.; Zhao, W.; Zheng, F.; Chen, Y.; Zhang, Y. Enhanced chemical interaction between TiO2 and graphene oxide for photocatalytic decolorization of methylene blue. Chem. Eng. J. 2012, 193–194, 203–210. [Google Scholar] [CrossRef]
- Pastrana-Martínez, L.M.; Morales-Torres, S.; Likodimos, V.; Figueiredo, J.L.; Faria, J.L.; Falaras, P.; Silva, A.M. Advanced nanostructured photocatalysts based on reduced graphene oxide–TiO2 composites for degradation of diphenhydramine pharmaceutical and methyl orange dye. Appl. Catal. B 2012, 123, 241–256. [Google Scholar] [CrossRef]
- Yin, X.; Zhang, H.; Xu, P.; Han, J.; Li, J.; He, M. Simultaneous N-doping of reduced graphene oxide and TiO2 in the composite for visible light photodegradation of methylene blue with enhanced performance. RSC Adv. 2013, 3, 18474–18481. [Google Scholar] [CrossRef]
- Chacko, D.K.; Madhavan, A.A.; Arun, T.A.; Thomas, S.; Anjusree, G.S.; Deepak, T.G.; Balakrishnan, A.; Subramanian, K.R.V.; Sivakumar, N.; Nair, S.V.; et al. Ultrafine TiO2 nanofibers for photocatalysis. RSC Adv. 2013, 3, 24858–24862. [Google Scholar] [CrossRef]
- Linley, S.; Liu, Y.; Ptacek, C.J.; Blowes, D.W.; Gu, F.X. Recyclable Graphene Oxide-Supported Titanium Dioxide Photocatalysts with Tunable Properties. ACS Appl. Mater. Interfaces 2014, 6, 4658–4668. [Google Scholar] [CrossRef] [PubMed]
- Maruthamani, D.; Divakar, D.; Kumaravel, M. Enhanced photocatalytic activity of TiO2 by reduced graphene oxide in mineralization of Rhodamine B dye. J. Ind. Eng. Chem. 2015, 30, 33–43. [Google Scholar] [CrossRef]
- Kim, C.H.; Kim, B.; Yang, K.S. TiO2 nanoparticles loaded on graphene/carbon composite nanofibers by electrospinning for increased photocatalysis. In Proceedings of the 2nd International Symposium on Physics and Technology of Sensors (ISPTS), Pune, India, 7–10 March 2015. [Google Scholar]
- Yang, Y.; Ren, L.; Zhang, C.; Huang, S.; Liu, T. Facile Fabrication of Functionalized Graphene Sheets (FGS)/ZnO Nanocomposites with Photocatalytic Property. ACS Appl. Mater. Interfaces 2011, 3, 2779–2785. [Google Scholar] [CrossRef]
- Azarang, M.; Shuhaimi, A.; Yousefi, R.; Moradi Golsheikh, A.; Sookhakian, M. Synthesis and characterization of ZnO NPs/reduced graphene oxide nanocomposite prepared in gelatin medium as highly efficient photo-degradation of MB. Ceram. Int. 2014, 40, 10217–10221. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Li, H.P.; Cui, X.L.; Lin, Y. Graphene/TiO2 nanocomposites: Synthesis, characterization and application in hydrogen evolution from water photocatalytic splitting. J. Mater. Chem. 2010, 20, 2801–2806. [Google Scholar] [CrossRef]
- Li, B.; Cao, H. ZnO@graphene composite with enhanced performance for the removal of dye from water. J. Mater. Chem. 2011, 21, 3346–3349. [Google Scholar] [CrossRef]
- Luo, Q.-P.; Yu, X.-Y.; Lei, B.-X.; Chen, H.-Y.; Kuang, D.-B.; Su, C.-Y. Reduced Graphene Oxide-Hierarchical ZnO Hollow Sphere Composites with Enhanced Photocurrent and Photocatalytic Activity. J. Phys. Chem. C 2012, 116, 8111–8117. [Google Scholar] [CrossRef]
- Xitao, W.; Rong, L.; Kang, W. Synthesis of ZnO@ZnS–Bi2S3 core–shell nanorod grown on reduced graphene oxide sheets and its enhanced photocatalytic performance. J. Mater. Chem. A 2014, 2, 8304–8313. [Google Scholar] [CrossRef]
- Yao, Y.; Yang, Z.; Zhang, D.; Peng, W.; Sun, H.; Wang, S. Magnetic CoFe2O4–Graphene Hybrids: Facile Synthesis, Characterization, and Catalytic Properties. Ind. Eng. Chem. Res. 2012, 51, 6044–6051. [Google Scholar] [CrossRef]
- Zhang, D.; Pu, X.; Gao, Y.; Su, C.; Li, H.; Li, H.; Hang, W. One-step combustion synthesis of CoFe2O4–graphene hybrid materials for photodegradation of methylene blue. Mater. Lett. 2013, 113, 179–181. [Google Scholar] [CrossRef]
- Shi, Y.; Zhou, K.; Wang, B.; Jiang, S.; Qian, X.; Gui, Z.; Yuen, R.K.; Hu, Y. Ternary graphene–CoFe2O4/CdS nanohybrids: Preparation and application as recyclable photocatalysts. J. Mater. Chem. A 2014, 2, 535–544. [Google Scholar] [CrossRef]
- He, G.; Ding, J.; Zhang, J.; Hao, Q.; Chen, H. One-Step Ball-Milling Preparation of Highly Photocatalytic Active CoFe2O4–Reduced Graphene Oxide Heterojunctions for Organic Dye Removal. Ind. Eng. Chem. Res. 2015, 54, 2862–2867. [Google Scholar] [CrossRef]
- Moitra, D.; Chandel, M.; Ghosh, B.K.; Jani, R.K.; Patra, M.K.; Vadera, S.R.; Ghosh, N.N. A simple ‘in situ’ co-precipitation method for the preparation of multifunctional CoFe2O4–reduced graphene oxide nanocomposites: Excellent microwave absorber and highly efficient magnetically separable recyclable photocatalyst for dye degradation. RSC Adv. 2016, 6, 76759–76772. [Google Scholar] [CrossRef]
- Suwanchawalit, C.; Somjit, V. Hydrothermal synthesis of magnetic CoFe2O4-graphene nanocomposite with enhanced photocatalytic performance. Dig. J. Nanomater. Biostruct. 2015, 10, 769–777. [Google Scholar]
- Haw, C.; Chiu, W.; Abdul Rahman, S.; Khiew, P.; Radiman, S.; Abdul Shukor, R.; Hamid, M.A.A.; Ghazali, N. The design of new magnetic-photocatalyst nanocomposites (CoFe2O4–TiO2) as smart nanomaterials for recyclable-photocatalysis applications. New J. Chem. 2016, 40, 1124–1136. [Google Scholar] [CrossRef]
- Chen, X.; Zhao, Q.; Li, X.; Wang, D. Enhanced photocatalytic activity of degrading short chain chlorinated paraffins over reduced graphene oxide/CoFe2O4/Ag nanocomposite. J. Colloid Interface Sci. 2016, 479, 89–97. [Google Scholar] [CrossRef]
- Devi, L.G.; Srinivas, M. Hydrothermal synthesis of reduced graphene oxide-CoFe2O4 heteroarchitecture for high visible light photocatalytic activity: Exploration of efficiency, stability and mechanistic pathways. J. Environ. Chem. Eng. 2017, 5, 3243–3255. [Google Scholar] [CrossRef]
- Fu, Y.; Wang, X. Magnetically Separable ZnFe2O4–Graphene Catalyst and its High Photocatalytic Performance under Visible Light Irradiation. Ind. Eng. Chem. Res. 2011, 50, 7210–7218. [Google Scholar] [CrossRef]
- Yang, D.; Feng, J.; Jiang, L.; Wu, X.; Sheng, L.; Jiang, Y.; Wei, T.; Fan, Z. Photocatalyst Interface Engineering: Spatially Confined Growth of ZnFe2O4 within Graphene Networks as Excellent Visible-Light-Driven Photocatalysts. Adv. Funct. Mater. 2015, 25, 7080–7087. [Google Scholar] [CrossRef]
- Khadgi, N.; Li, Y.; Upreti, A.R.; Zhang, C.; Zhang, W.; Wang, Y.; Wang, D. Enhanced Photocatalytic Degradation of 17α-Ethinylestradiol Exhibited by Multifunctional ZnFe2O4–Ag/rGO Nanocomposite Under Visible Light. Photochem. Photobiol. 2016, 92, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Jenita Rani, G.; Jothi Rajan, M.A.; Gnana kumar, G. Reduced graphene oxide/ZnFe2O4 nanocomposite as an efficient catalyst for the photocatalytic degradation of methylene blue dye. Res. Chem. Intermed. 2017, 43, 2669–2690. [Google Scholar] [CrossRef]
- Fu, Y.; Chen, H.; Sun, X.; Wang, X. Graphene-supported nickel ferrite: A magnetically separable photocatalyst with high activity under visible light. AIChE J. 2012, 58, 3298–3305. [Google Scholar] [CrossRef]
- Liu, S.-Q.; Xiao, B.; Feng, L.-R.; Zhou, S.-S.; Chen, Z.-G.; Liu, C.-B.; Chen, F.; Wu, Z.-Y.; Xu, N.; Oh, W.-C.; et al. Graphene oxide enhances the Fenton-like photocatalytic activity of nickel ferrite for degradation of dyes under visible light irradiation. Carbon 2013, 64, 197–206. [Google Scholar] [CrossRef]
- Zeng, J.; Song, T.; Lv, M.; Wang, T.; Qin, J.; Zeng, H. Plasmonic photocatalyst Au/g-C3N4/NiFe2O4 nanocomposites for enhanced visible-light-driven photocatalytic hydrogen evolution. RSC Adv. 2016, 6, 54964–54975. [Google Scholar] [CrossRef]
- Fu, Y.; Xiong, P.; Chen, H.; Sun, X.; Wang, X. High Photocatalytic Activity of Magnetically Separable Manganese Ferrite–Graphene Heteroarchitectures. Ind. Eng. Chem. Res. 2012, 51, 725–731. [Google Scholar] [CrossRef]
- Gao, E.; Wang, W.; Shang, M.; Xu, J. Synthesis and enhanced photocatalytic performance of graphene-Bi2WO6 composite. Phys. Chem. Chem. Phys. 2011, 13, 2887–2893. [Google Scholar] [CrossRef]
- Li, Z.; Shen, Y.; Yang, C.; Lei, Y.; Guan, Y.; Lin, Y.; Liu, D.; Nan, C.-W. Significant enhancement in the visible light photocatalytic properties of BiFeO3–graphene nanohybrids. J. Mater. Chem. A 2013, 1, 823–829. [Google Scholar] [CrossRef]
- Lee, S.; Amaranatha Reddy, D.; Kim, T.K. Well-wrapped reduced graphene oxide nanosheets on Nb3O7(OH) nanostructures as good electron collectors and transporters for efficient photocatalytic degradation of rhodamine B and phenol. RSC Adv. 2016, 6, 37180–37188. [Google Scholar] [CrossRef]
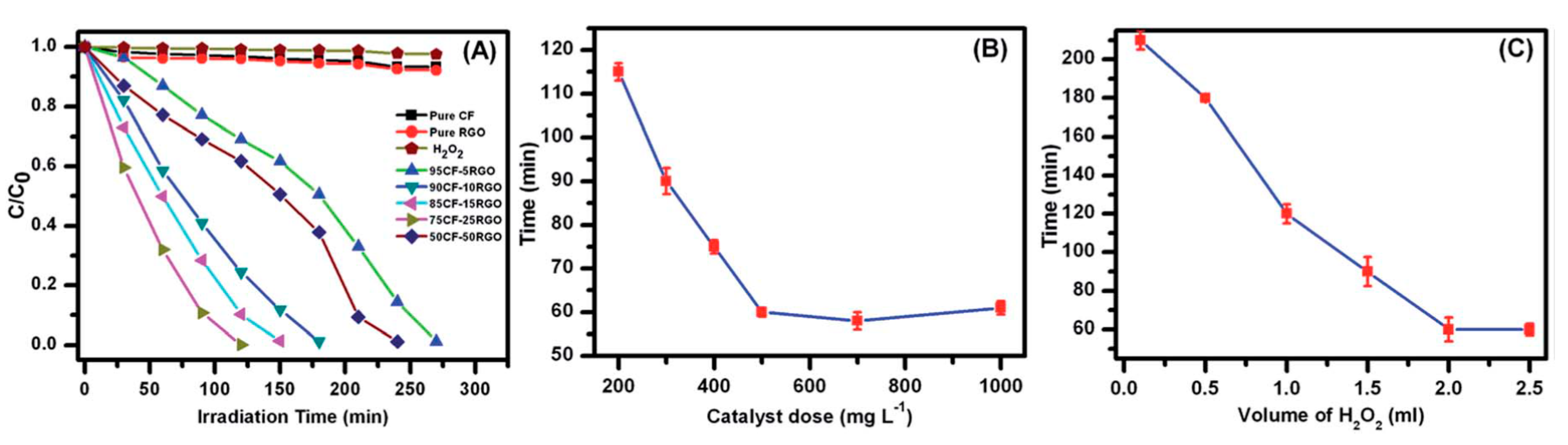
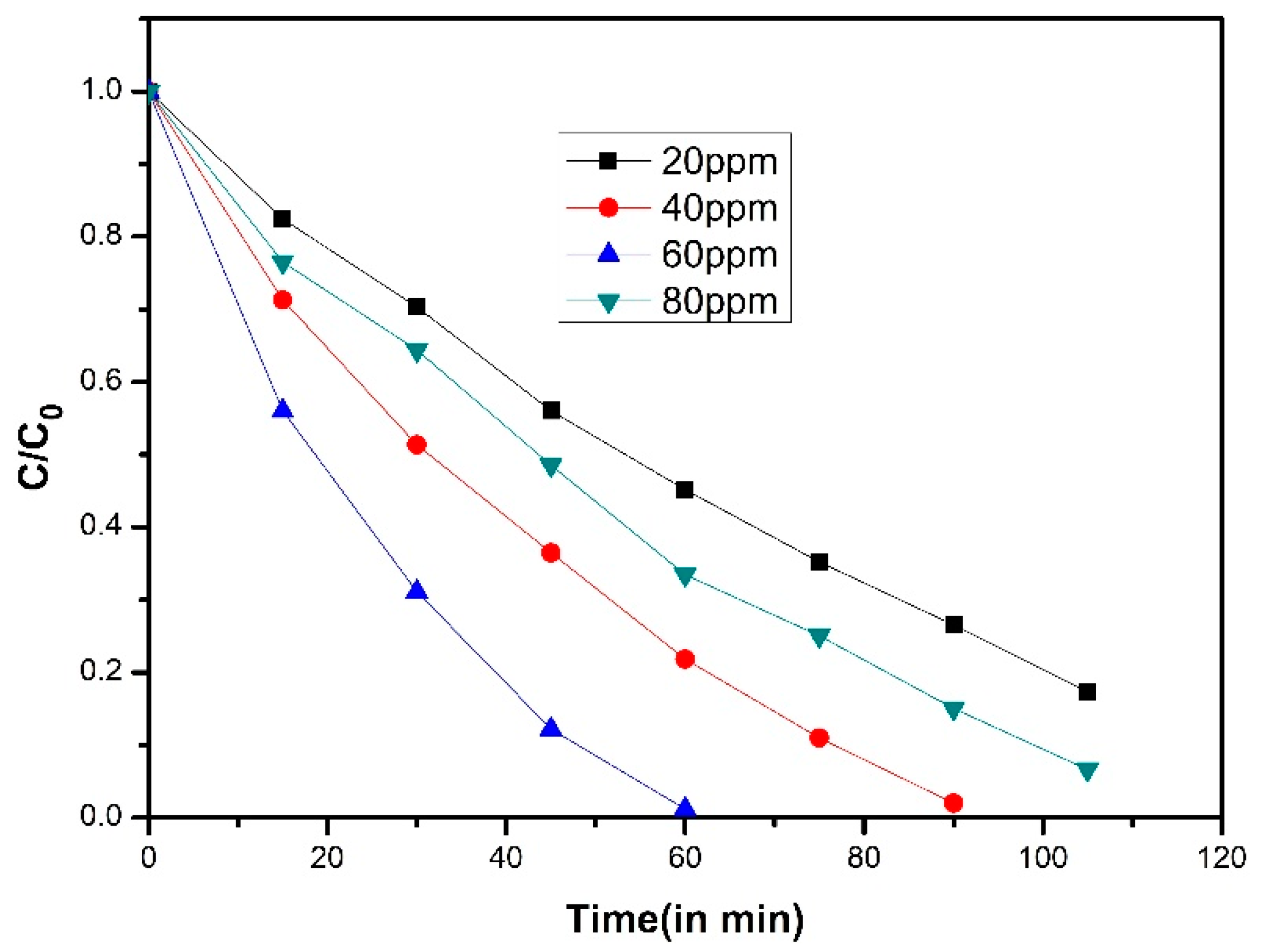
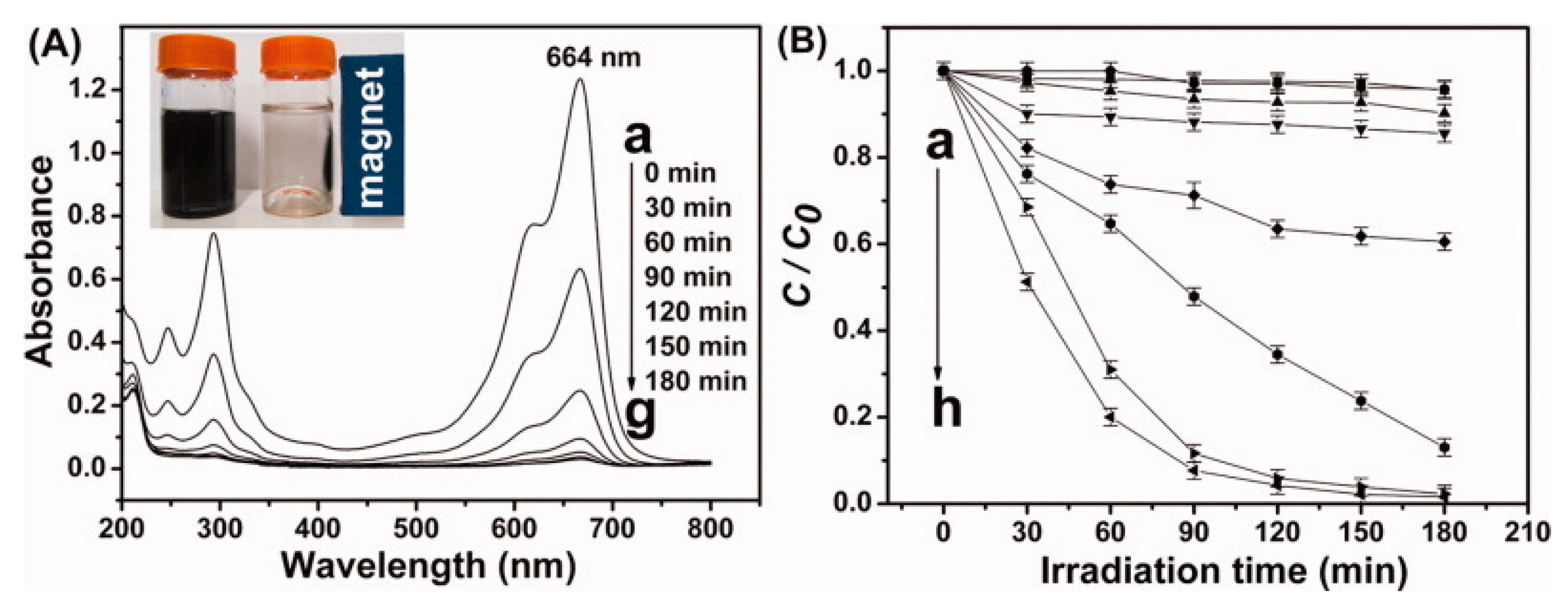
| Order | Photocatalyst | Preparation | References |
|---|---|---|---|
| A. Hydrothermal method for the synthesis of GO/rGO-NCs photocatalyst | |||
| 1 | rGO-WO3 | Na2WO4·2H2O and 0.05 g NaCl were dissolved in the above dispersion and kept stirring for 1 h. The pH was adjusted to 2 by using HCl solution. | [6] |
| 2 | rGO-WO3 | Preset amounts of Na2WO4·2H2O (100, 200, and 400 mg, respectively) were dissolved in 10 mL above GO suspension. 5 mL 35% HCl was added slowly. Transferred to autoclave heated at 140 °C for 8 h. | [18] |
| 3 | rGO_Co3O4 | GO dispersed into 24 mL of alcohol, sonicating for 60 min in an ultrasonic cleaner. Then, 0.2 M of Co(Ac)2 was added to the mixture followed by 1.2 mL of water, and continued to be stirred for 10 h at a temperature of 80 °C. The resulting solution was then transferred into a 40 mL autoclave for hydrothermal reaction at 150 °C for 3 h. | [21] |
| 4 | rGO_Co3O4 | GO dispersed in the Co (C2H3O2)2·4H2O. 10 mL with 28% ammonia solution were added to solution, and transferred into an autoclave for hydrothermal action at 180 °C for 12 h. | [23] |
| 5 | rGO/ZnFe2O4-Ag | The composite was synthesized by the co-precipitation of Zn (NO3)2·6H2O, Fe (NO3)3.9H2O, and AgNO3 in the presence of the GO powder. | [53] |
| 6 | GO-NiFe2O4 | GO in NiFe2O4 was dispersed in deionized water. Then, NiSO4H2O and FeCl36H2O (0.02 mol) were dissolved in 15 mL water. Transferred into autoclave and kept under high pressure. | [56] |
| 7 | GO-MnFe2O4 | GO and 60 mL of ethanol with sonication for 1 h and Mn(NO3)2 solution and Fe (NO3)3 9H2O were dissolved. The resulting mixture was transferred into a 100 mL Teflon-lined stainless-steel autoclave and heated to 180 °C for 20 h under autogenous pressure. | [58] |
| 8 | rGO-Bi2WO6 | GO by using Hammer method GO was reduced by ethylene glycol. Bi(NO3)3 5H2O was dispersed into 5 mL of 4 M nitric acid solution. Na2WO4 2H2O was dissolved in 5 mL of de-ionized water and then Na2WO4 was added dropwise to the solution. | [59] |
| B. Sol-gel method for the synthesis of GO/rGO-NCs photocatalysts | |||
| 9 | rGO-TiO2 | An aqueous solution of Ti(OH)4 was added into an aqueous suspension of GO. | [30] |
| 10 | rGO-ZnO | An aqueous solution of Zn (AcO)2·3H2O was added into an aqueous suspension of GO. | [39] |
| C. Solvothermal technique for the GO/rGO-NCs photocatalysts | |||
| 11 | GO/CoFe2O4/CdS | Gr–CoFe2O4 nanohybrids were sonicated in 60 mL of ethylene glycol for 10 min. The cadmium source containing 0.1431 g of Cd(NO3)24H2O and10 mL of ethylene glycol was added to the mixture containing Gr–CoFe2O4 nanohybrids. The mixture of 0.0348 g of thiourea, 0.0514 g of PVP, and 10 mL of ethyleneglycol was transferred into the above mixture. | [44] |
| 12 | rGO-ZnFe2O4 | GO dispersed in ZnOx(OH)y and FeOx solutions were put into a 50 mL autoclave. | [51] |
| D. Colloidal method for the synthesis of GO/rGO-NCs photocatalyst | |||
| 13 | rGO-ZnFe2O4 | C2H6O2 solution is containing 2M FeCl36H2O, and 1M ZnCl2 was gradually added. Then, 1MCH3COONa was introduced into the solution and magnetically stirred for 1 h. Then, transferred to autoclave heated at 1800 °C. | [54] |
| E. Thermal treatment for the synthesis of GO/rGO-NCs photocatalyst | |||
| 14 | rGO-WO3 | Na2WO4·2H2O was dissolved in 30 mL water. Then, nitric acid was added to the solution drop by drop until the precipitate was formed. Dried at 160 °C for 2 h and annealed at 500 °C for 5 h. | [19] |
| 15 | rGO-WO3 | Na2WO4·2H2O (0.5 g), H2C2O4 (1 g), and Na2SO4 (4 g) were added into subsequent solution and stirred for 3 h. The pH of the solution was maintained at 1.5 by adding 3M HCl and stirring was continued for 3 h. Then, transferred to autoclave maintained at 180 °C for 24 h. | [20] |
| 16 | FGS/ZnO | GO, Zn(NH3)4CO3, and poly(vinyl pyrrolidone) as an intermediate to combine zinc with carbon material | [36] |
| F. Ball-milling method for the synthesis of GO/rGO-NCs photocatalyst | |||
| 17 | rGO-CoFe2O4 | Co (NO3)2·6H2O and of Fe (NO3)3·9H2O were added to GO (2.5 wt%). The pH is maintained 10 | [45] |
| 18 | rGO-CoFe2O4 | [45] | |
| 19 | rGO-CoFe2O4 | [45] | |
| G. Liquid phase deposition method for the synthesis of GO/rGO-NCs photocatalyst | |||
| 20 | rGO-TiO2 | TiO2 powder (P25, Degussa) was dispersed in deionized water and subsequently added to the graphene oxide solution | [28] |
| H. Microwave irradiation method for the synthesis of GO/rGO-NCs photocatalyst | |||
| 21 | rGO-CoFe2O4 | (Co(NO3)2·6H2O and Fe(NO3)3 9H2O and glucose as oxidizer and fuel. GO, nitrates, and glucose were added in water for 30 min ultrasonic treatment. | [43] |
| 22 | rGO-CoFe2O4 | [43] | |
| 23 | rGO-CoFe2O4 | [43] | |
| 24 | rGO/CoFe2O4/Ag | GO, AgNO3, and CoFe2O4 were dissolved in deionized water and stirred for 2 h. Then, solution was further stirred for 2 h under the UV irradiation of a 22 W low-pressure mercury lamp. The product is washed with distilled water and ethanol in an oven at 60 °C for 12 h. | [49] |
| I. In situ co-precipitation method for the synthesis of GO/rGO-NCs photocatalyst | |||
| 25 | rGO-ZnO | GO dispersed in aqueous solution containing Zn(CH3COO)2, DMSO, and H2O | [40] |
| J. Annealing NH3 atmosphere method for the synthesis of GO/rGO-NCs photocatalyst | |||
| 26 | rGO/N-TiO2 | GO and 300 mg of P90 TiO2 was added and stirred for 3 h. GO and P90 TiO2 and a few drops of tetrabutyltitanate were added. | [31] |
| Order | Pollutants | Photocatalyst | Light Source | Reactor | Mass of Catalyst (mg) | Concentration (ppm) | Irradiation Time (min) | Conversion (%) | Mol. Wt. | Photon Flux (mW cm−2) | Quantum Yield (%) | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A. Photocatalytic performances of GO-rGO semiconductor composites for dye degradation | ||||||||||||
| A-1. MB | ||||||||||||
| 1 | MB | rGO-WO3 | light source was a 150 W xenon lamp. | 20 °C self-made Lab Solar gas photocatalysis system with external light irradiation | 50 | 7 | 120 | 100 | 2278.4 | NA | NA | [6] |
| 2 | MB | rGO-WO3 | One 300 W PLS-SXE 300 xenon lamp | equipped with a λ < 400 nm cut-off filter | 20 | 10 | 70 | 95 | 2247.4 | NA | NA | [20] |
| 3 | MB | rGO/N-TiO2 | Two 20 W black-lights with 352 nm (UV) and 545 nm (Visible) | NA | 10 | 8.8 | 60 | 80 (UV) and 95 (Visible) | 2123.6 | NA | NA | [28] |
| 4 | MB | rGO/N-TiO2 | one 500 W Xenon lamp > 400 nm | Quartz cell | 50 | 8.8 | 160 | 100 | 2137.6 | NA | NA | [31] |
| 5 | MB | rGO-ZnO | one 300 W Xe lamp with 420 nm | NA | 80 | 18 | 70 | 100 | 2125.2 | NA | NA | [39] |
| 6 | MB | rGO-ZnO | one 500 W mercury lamp | NA | 20 | 10 | 90 | 100 | 2125.2 | NA | NA | [40] |
| 7 | MB | GO/CoFe2O4/CdS | one 40 W daylight lamp | NA | 25 | 20 | 180 | 100 | 2422.8 | NA | NA | [44] |
| 8 | MB | rGO-CoFe2O4 | one 800 W Xe lamp | NA | 10 | 20 | 180 | 100 | 2278.4 | NA | NA | [45] |
| 9 | MB | rGO-CoFe2O4 | A 100 W reading lamp | installed glass cut-off filter | 25 | 10 | 75 | 90 | 2278.4 | NA | NA | [46] |
| 10 | MB | rGO-ZnFe2O4 | one 500 W xenon lamp > 420 nm | Glass reactor (100 mL) | 50 | 10 | 90 | 61 | 2284.7 | NA | NA | [51] |
| 11 | MB | rGO-ZnFe2O4 | one 530 W lamp with >400 nm | Pyrex glass tube (100 mL) | 25 | 10 | 120 | 100 | 2284.7 | NA | NA | [54] |
| 12 | MB | NiFe2O4-GO | One 300 W UV-visible lamp | Quartz glass (100 mL) | 100 | 20 | 600 | 90 | 2280.2 | NA | NA | [56] |
| 13 | MB | MnFe2O4-GO | one 500 W mercury and xenon lamp | Glass tube (100 mL) | 25 | 20 | 360 | 98 | 2274 | NA | NA | [58] |
| A-2. MO | ||||||||||||
| 14 | MO | rGO_Co3O4 | One 100 W Xenon lamp | NA | 10 | 30 | 180 | 80 | 2287 | NA | NA | [21] |
| 15 | MO | rGO-TiO2 | one 150 W medium-pressure mercury vapor lamp with >350 nm | quartz cylindrical reactor (7.5 mL) | 100 | 500 | 30 | 100 | 2123.6 | 6 | NA | [30] |
| 16 | MO | rGO-CoFe2O4 | one 800 W Xe lamp | NA | 10 | 20 | 180 | 25 | 2278.4 | NA | NA | [45] |
| 17 | MO | rGO-CoFe2O4 | A 100 W reading lamp | installed glass cut-off filter | 25 | 10 | 75 | 60 | 2278.4 | NA | NA | [46] |
| A-3. RhB | ||||||||||||
| 18 | RhB 6G | FGS/ZnO | Two 100 and 250 W high-pressure mercury lamps with 300 nm | Pyrex glass tube (1000 mL) | 10 | 10 | 100 | 100 | 2125.2 | NA | NA | [36] |
| 19 | RhB | rGO-CoFe2O4 | one 800 W Xe lamp | NA | 10 | 20 | 180 | 75 | 2278.4 | NA | NA | [45] |
| 20 | RhB | rGO-CoFe2O4 | A 100 W reading lamp | installed glass cut-off filter | 25 | 10 | 75 | 90 | 2278.4 | NA | NA | [46] |
| 21 | RhB | rGO-Bi2WO6 | one 500 W Xe lamp with 420 nm | Installed glass cut-off filter to visible-light irradiation glass tube (500 mL) | 100 | 0.355 | 15 | 98 | 2740.8 | NA | NA | [59] |
| B. Photocatalytic performances of GO-rGO semiconductor composites for organic pollutants degradation | ||||||||||||
| 22 | sulfamethoxazole | rGO-WO3 | 200 W Xe arc lamp with specific ranges 420–630 nm | 1.5 AM solar simulator | 10 | 20 | 180 | 100 | 2247 | NA | NA | [18] |
| 23 | 1-Naphthol | rGO-WO3 | One Xe lamp 570 W | cylindrical Pyrex reactor of 7 cm diameter and 15 cm height | 50 | 150 | 120 | 100 | 2247 | NA | NA | [19] |
| 24 | Chain chlorinated paraffin’s | RGO/CoFe2O4/Ag | One 500 W xenon lamp with 400 nm | in situ quartz reaction cell | 10 | NA | 720 | 90 | 2386.2 | NA | NA | [49] |
| 25 | 17 α-ethinylestradiol | rGO/ZnFe2O4-Ag | One 300 W Xe-lamp | NA | 100 | 2 | 240 | 100 | 2382 | NA | NA | [53] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mandal, S.; Mallapur, S.; Reddy, M.; Singh, J.K.; Lee, D.-E.; Park, T. An Overview on Graphene-Metal Oxide Semiconductor Nanocomposite: A Promising Platform for Visible Light Photocatalytic Activity for the Treatment of Various Pollutants in Aqueous Medium. Molecules 2020, 25, 5380. https://doi.org/10.3390/molecules25225380
Mandal S, Mallapur S, Reddy M, Singh JK, Lee D-E, Park T. An Overview on Graphene-Metal Oxide Semiconductor Nanocomposite: A Promising Platform for Visible Light Photocatalytic Activity for the Treatment of Various Pollutants in Aqueous Medium. Molecules. 2020; 25(22):5380. https://doi.org/10.3390/molecules25225380
Chicago/Turabian StyleMandal, Soumen, Srinivas Mallapur, Madhusudana Reddy, Jitendra Kumar Singh, Dong-Eun Lee, and Taejoon Park. 2020. "An Overview on Graphene-Metal Oxide Semiconductor Nanocomposite: A Promising Platform for Visible Light Photocatalytic Activity for the Treatment of Various Pollutants in Aqueous Medium" Molecules 25, no. 22: 5380. https://doi.org/10.3390/molecules25225380
APA StyleMandal, S., Mallapur, S., Reddy, M., Singh, J. K., Lee, D.-E., & Park, T. (2020). An Overview on Graphene-Metal Oxide Semiconductor Nanocomposite: A Promising Platform for Visible Light Photocatalytic Activity for the Treatment of Various Pollutants in Aqueous Medium. Molecules, 25(22), 5380. https://doi.org/10.3390/molecules25225380










