Mineral Composition of Cereal and Cereal-Free Dry Dog Foods versus Nutritional Guidelines
Abstract
1. Introduction
2. Results
2.1. Basic Composition
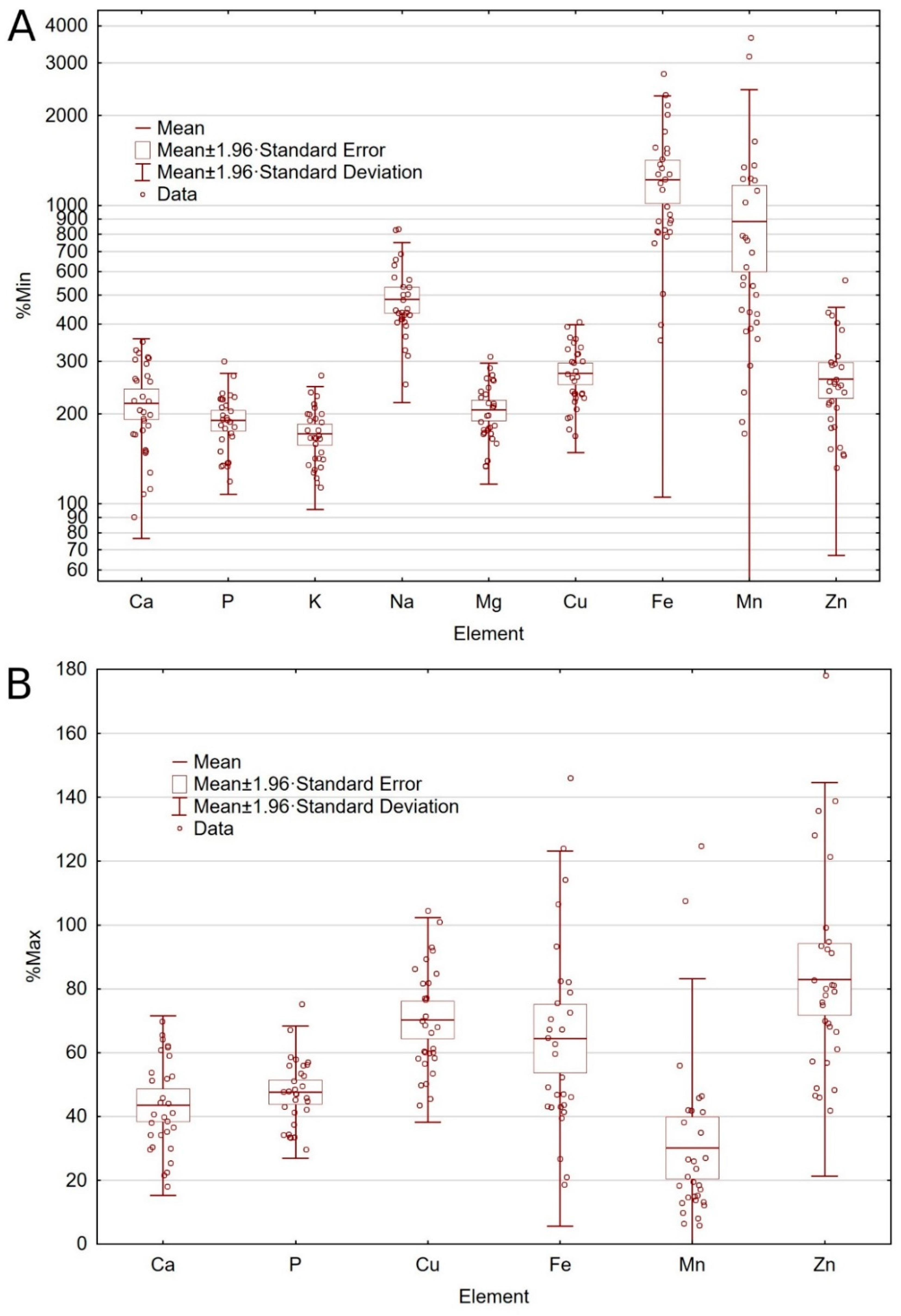
2.2. Macroelement Levels
2.3. Trace Element Levels
2.4. Trace Elements Additives
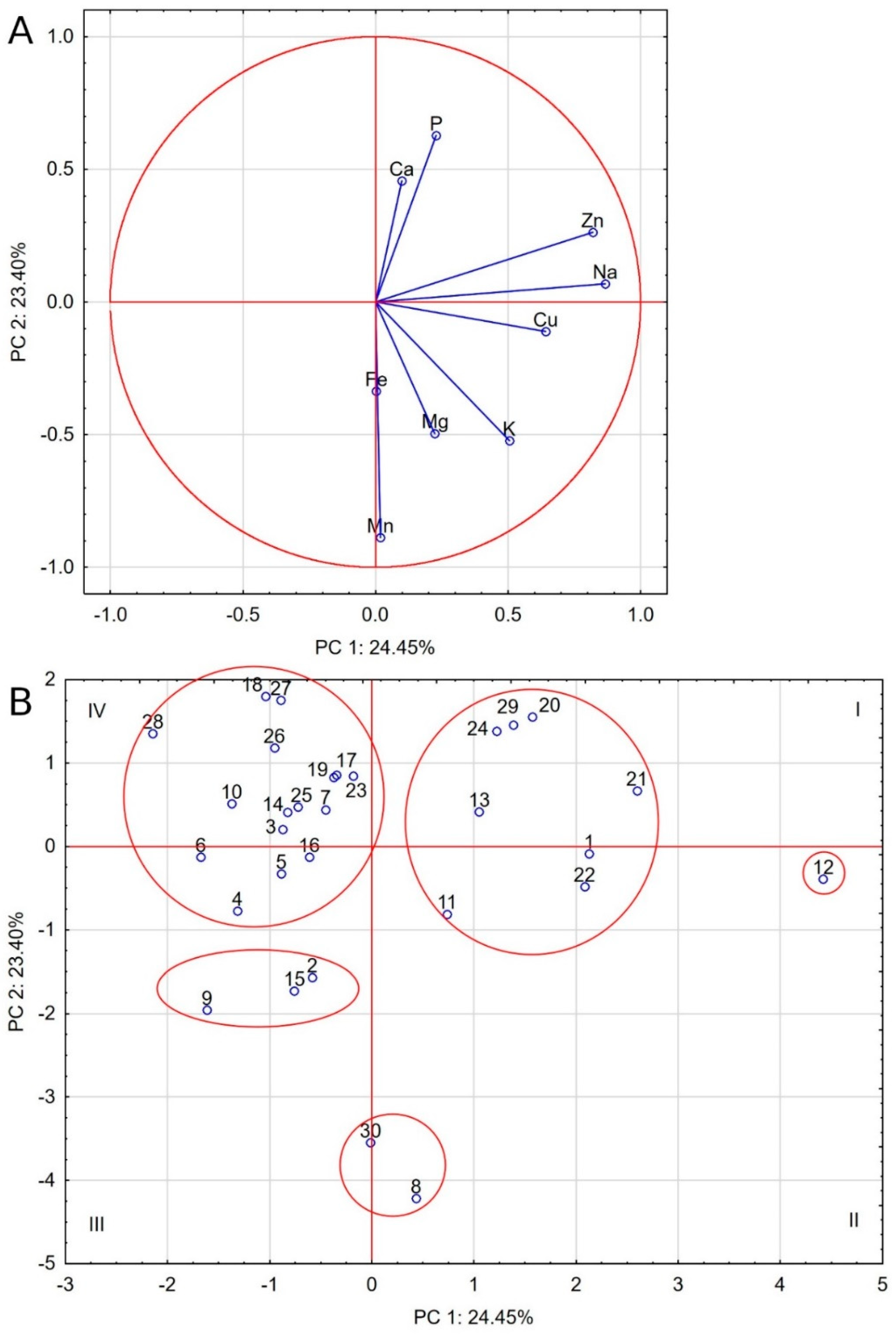
2.5. Comparative Analysis
2.6. Relations between Elements
3. Discussion
3.1. Basic Composition
3.2. Macroelements
3.3. Trace Elements
3.4. Relations between Elements
3.5. Heavy Metals Content
4. Materials and Methods
4.1. Sampling
4.2. Basic Composition
4.3. Macroelements Analysis
4.4. Microelements and Heavy Metal Analysis
4.5. Amount of Trace Elements from Feed Additives
4.6. Relations between Elements
4.7. Statistical Analysis
5. Conclusions
- All the evaluated dry dog foods met the minimum FEDIAF recommended levels for protein and fat;
- Comparative analysis of the mineral profile of foods showed differences between foods depending on the composition, which allowed to distinguish specific groups of foods—among the tested foods significant differences in the presence or absence of cereals in the composition were found, with higher P, K, Na, Fe, Zn, Mn, and Cu levels found in cereal-free foods;
- For a few individual products, mineral content often either far exceeded or did not meet the nutritional requirements, of which in two dog foods, the limit for two trace elements exceeded simultaneously and the concentration of exceeded element was supplemented with nutritional additives, which possibly led to their excess;
- Dog foods without the declared supplementation with nutritional additives met the minimum recommended levels of trace elements, and in two foods they even exceeded the legal limit;
- Diets with an insect source of animal protein had relatively high Mn content, which was supplied from raw materials, not nutritional additives; this may indicate the need for a more detailed analysis of this relatively new source of protein in dogs’ diets in order to avoid excess Mn in the finished product and thus its toxicity;
- Most of the incompatibilities concerned inappropriate mineral Ca:P ratio content (five dog foods below the minimum recommended levels and three above maximum), which may constitute a risk to the overall well-being and health of pets when they are exclusively fed with these products in the long term;
- Heavy metals (Co, Cd, Pb, Mo, Cr, Ni) levels were below detection limit in all analyzed dog foods.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Glossary
References
- American Pet Products Association. 2019–2020 APPA National Pet Owners Survey; American Pet Products Association: Stamford, CT, USA, 2020. [Google Scholar]
- FEDIAF. Annual Report 2020; The European Pet Food Industry: Bruxelles, Belgium, 2020. [Google Scholar]
- FEDIAF. Nutritional Guidelines for Complete and Complementary Pet Food for Cats and Dogs; The European Pet Food Industry: Bruxelles, Belgium, 2020. [Google Scholar]
- NRC. Nutrient Requirements of Dogs and Cats; National Academies Press; National Research Council: Washington, DC, USA, 2006. [Google Scholar]
- Rosol, T.J.; Capen, C.C. Calcium-regulating hormones and diseases of abnormal mineral (calcium, phosphorus, magnesium) metabolism. In Clinical Biochemistry of Domestic Animals; Elsevier: Amsterdam, The Netherlands, 1997; pp. 619–702. ISBN 978-0-12-396305-5. [Google Scholar]
- Nguyen, P.; Reynolds, B.; Zentek, J.; Paßlack, N.; Leray, V. Sodium in feline nutrition. J. Anim. Physiol. Anim. Nutr. 2017, 101, 403–420. [Google Scholar] [CrossRef] [PubMed]
- Atkins, P.; Ernyei, L.; Driscoll, W.; Obenauf, R.; Thomas, R. Analysis of toxic trace metals in pet foods using cryogenic grinding and quantitation by ICP-MS, Part I. Spectroscopy 2011, 26, 46–68. [Google Scholar]
- Atkins, P.; Ernyei, L.; Driscoll, W.; Obenauf, R.; Thomas, R. Analysis of toxic trace metals in pet foods using cryogenic grinding and quantitation by ICP-MS, Part II. Spectroscopy 2011, 26, 42–59. [Google Scholar]
- Kelly, D.G.; White, S.D.; Weir, R.D. Elemental composition of dog foods using nitric acid and simulated gastric digestions. Food Chem. Toxicol. 2013, 55, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Ávila, D.V.L.; Borges, A.R.; Vale, M.G.R.; Araujo, R.G.O.; Passos, E.A. Determination of Co and Cr in wet animal feeds using direct solid sample analysis by HR-CS GF AAS. Microchem. J. 2017, 133, 524–529. [Google Scholar] [CrossRef]
- Pereira, A.M.; Pinto, E.; Matos, E.; Castanheira, F.; Almeida, A.A.; Baptista, C.S.; Segundo, M.A.; Fonseca, A.J.M.; Cabrita, A.R.J. Mineral composition of dry dog foods: Impact on nutrition and potential toxicity. J. Agric. Food Chem. 2018, 66, 7822–7830. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.M.; Guedes, M.; Matos, E.; Pinto, E.; Almeida, A.A.; Segundo, M.A.; Correia, A.; Vilanova, M.; Fonseca, A.J.M.; Cabrita, A.R.J. Effect of zinc source and exogenous enzymes supplementation on zinc status in dogs fed high phytate diets. Animals 2020, 10, 400. [Google Scholar] [CrossRef]
- Tsanaktsidou, E.; Zachariadis, G. Titanium and chromium determination in feedstuffs using ICP-AES Technique. Separations 2020, 7, 1. [Google Scholar] [CrossRef]
- Schneider, J.S.; Decamp, E.; Koser, A.J.; Fritz, S.; Gonczi, H.; Syversen, T.; Guilarte, T.R. Effects of chronic manganese exposure on cognitive and motor functioning in non-human primates. Brain Res. 2006, 1118, 222–231. [Google Scholar] [CrossRef]
- Naigamwalla, D.Z.; Webb, J.A.; Giger, U. Iron deficiency anemia. Can. Vet. J. 2012, 53, 250–256. [Google Scholar]
- Tal, M.; Parr, J.M.; MacKenzie, S.; Verbrugghe, A. Dietary imbalances in a large breed puppy, leading to compression fractures, vitamin D deficiency, and suspected nutritional secondary hyperparathyroidism. Can. Vet. J. 2018, 59, 36–42. [Google Scholar]
- AAFCO. Pet Food Labeling Guide; The Association of American Feed Control Officials: Oxford, UK, 2016. [Google Scholar]
- De Nadai Fernandes, E.A.; Elias, C.; Bacchi, M.A.; Bode, P. Trace element measurement for assessment of dog food safety. Environ. Sci. Pollut. Res. 2018, 25, 2045–2050. [Google Scholar] [CrossRef]
- Kim, H.-T.; Loftus, J.P.; Mann, S.; Wakshlag, J.J. Evaluation of arsenic, cadmium, lead and mercury contamination in over-the-counter available dry dog foods with different animal ingredients (red meat, poultry, and fish). Front. Vet. Sci. 2018, 5, 264. [Google Scholar] [CrossRef] [PubMed]
- Leiva, A.; Molina, A.; Redondo-Solano, M.; Artavia, G.; Rojas-Bogantes, L.; Granados-Chinchilla, F. Pet food quality assurance and safety and quality assurance survey within the Costa Rican pet food industry. Animals 2019, 9, 980. [Google Scholar] [CrossRef] [PubMed]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdisc. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Conway, D.M.P.; Saker, K.E. Consumer attitude toward the environmental sustainability of grain-free pet foods. Front. Vet. Sci. 2018, 5. [Google Scholar] [CrossRef]
- Meineri, G.; Candellone, A.; Bello, F.D.; Gastaldi, D.; Medana, C.; Peiretti, P.G. Gluten contamination of canned and dry grain-free commercial pet foods determined by HPLC-HRMS. Ital. J. Anim. Sci. 2020, 19, 253–261. [Google Scholar] [CrossRef]
- Wrigley, C. Encyclopedia of Grain Science, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2004; ISBN 978-0-12-765490-4. [Google Scholar]
- Bedford, E. Pet Food Sales Volume U.S. by Category 2019. Statista—The Statistics Portal. 2020. Available online: https://www.statista.com/statistics/954641/pet-food-sales-volume-us-by-category/ (accessed on 5 November 2020).
- Bednar, G.E.; Murray, S.M.; Patil, A.R.; Flickinger, E.A.; Merchen, N.R.; Fahey, G.C. Selected animal and plant protein sources affect nutrient digestibility and fecal characteristics of ileally cannulated dogs. Arch. Tierernaehr. 2000, 53, 127–140. [Google Scholar] [CrossRef]
- Bosch, G.; Vervoort, J.J.M.; Hendriks, W.H. In vitro digestibility and fermentability of selected insects for dog foods. Anim. Feed Sci. Technol. 2016, 221, 174–184. [Google Scholar] [CrossRef]
- Ahlstrøm, Ø.; Krogdahl, Å.; Vhile, S.G.; Skrede, A. Fatty acid composition in commercial dog foods. J. Nutr. 2004, 134, 2145S–2147S. [Google Scholar] [CrossRef]
- Gross, K.L.; Yamka, R.M.; Khoo, C.; Friesen, K.G.; Jewell, D.E.; Schoenherr, W.D.; Debraekeleer, J.; Zicker, S.C. Macronutrients. In Small Animal Clinical Nutrition; Mark Morris Institute: Topeka, KS, USA, 2011; pp. 49–105. [Google Scholar]
- Donfrancesco, B.D.; Koppel, K. Sensory characteristics and volatile components of dry dog foods manufactured with sorghum fractions. Molecules 2017, 22, 1012. [Google Scholar] [CrossRef]
- Pezzali, J.G.; Aldrich, C.G. Effect of ancient grains and grain-free carbohydrate sources on extrusion parameters and nutrient utilization by dogs. J. Anim. Sci. 2019, 97, 3758–3767. [Google Scholar] [CrossRef]
- Koppel, K.; Adhikari, K.; Di Donfrancesco, B. Volatile Compounds in dry dog foods and their influence on sensory aromatic profile. Molecules 2013, 18, 2646–2662. [Google Scholar] [CrossRef]
- Becker, N.; Kienzle, E.; Dobenecker, B. Calcium deficiency: A problem in growing and adult dogs: Two case reports. Tierarztl. Prax. Ausg. K. Kleintiere Heimtiere 2012, 40, 135–139. [Google Scholar]
- Dobenecker, B.; Kasbeitzer, N.; Flinspach, S.; Kostlin, R.; Matis, U.; Kienzle, E. Calcium-excess causes subclinical changes of bone growth in Beagles but not in Foxhound-crossbred dogs, as measured in X-rays. J. Anim. Physiol. Anim. Nutr. 2006, 90, 394–401. [Google Scholar] [CrossRef]
- Dobenecker, B. Factors that modify the effect of excess calcium on skeletal development in puppies. Br. J. Nutr. 2011, 106, S142–S145. [Google Scholar] [CrossRef]
- Gagné, J.W.; Wakshlag, J.J.; Center, S.A.; Rutzke, M.A.; Glahn, R.P. Evaluation of calcium, phosphorus, and selected trace mineral status in commercially available dry foods formulated for dogs. J. Am. Vet. Med. Assoc. 2013, 243, 658–666. [Google Scholar] [CrossRef]
- Pedrinelli, V.; Zafalon, R.V.A.; Rodrigues, R.B.A.; Perini, M.P.; Conti, R.M.C.; Vendramini, T.H.A.; de Carvalho Balieiro, J.C.; Brunetto, M.A. Concentrations of macronutrients, minerals and heavy metals in home-prepared diets for adult dogs and cats. Sci. Rep. 2019, 9, 13058. [Google Scholar] [CrossRef]
- Kiefer-Hecker, B.; Kienzle, E.; Dobenecker, B. Effects of low phosphorus supply on the availability of calcium and phosphorus, and musculoskeletal development of growing dogs of two different breeds. J. Anim. Physiol. Anim. Nutr. 2018, 102, 789–798. [Google Scholar] [CrossRef]
- Brunetto, M.A.; Zafalon, R.V.A.; Teixeira, F.A.; Vendramini, T.H.A.; Rentas, M.F.; Pedrinelli, V.; Risolia, L.W.; Macedo, H.T. Phosphorus and sodium contents in commercial wet foods for dogs and cats. Vet. Med. Sci. 2019, 5, 494–499. [Google Scholar] [CrossRef]
- Bateman, S. Disorders of Magnesium: Magnesium Deficit and Excess. In Fluid, Electrolyte, and Acid-Base Disorders in Small Animal Practice; Elsevier: Amsterdam, The Netherlands, 2006; pp. 210–226. ISBN 978-0-7216-3949-9. [Google Scholar]
- Segev, G.; Fascetti, A.J.; Weeth, L.P.; Cowgill, L.D. Correction of hyperkalemia in dogs with chronic kidney disease consuming commercial renal therapeutic diets by a potassium-reduced home-prepared diet: Hyperkalemia in dogs with CKD. J. Vet. Intern. Med. 2010, 24, 546–550. [Google Scholar] [CrossRef]
- Chandler, M.L. Pet food safety: Sodium in pet foods. Top. Companion Anim. Med. 2008, 23, 148–153. [Google Scholar] [CrossRef]
- Brown, S.; Atkins, C.; Bagley, R.; Carr, A.; Cowgill, L.; Davidson, M.; Egner, B.; Elliott, J.; Henik, R.; Labato, M.; et al. Guidelines for the identification, evaluation, and management of systemic hypertension in dogs and cats. J. Vet. Intern. Med. 2007, 21, 542–558. [Google Scholar] [CrossRef] [PubMed]
- Sakhaee, K.; Harvey, J.A.; Padalino, P.K.; Whitson, P.; Pak, C.Y.C. The potential role of salt abuse on the risk for kidney stone formation. J. Urol. 1993, 150, 310–312. [Google Scholar] [CrossRef]
- FEDIAF. Code of Good Labelling Practice for Pet Food; The European Pet Food Industry: Bruxelles, Belgium, 2018. [Google Scholar]
- Fieten, H.; Leegwater, P.A.J.; Watson, A.L.; Rothuizen, J. Canine models of copper toxicosis for understanding mammalian copper metabolism. Mamm. Genome 2012, 23, 62–75. [Google Scholar] [CrossRef]
- Cooper, V.L.; Carlson, M.P.; Jacobson, J.; Schneider, N.R. Hepatitis and increased copper levels in a dalmatian. J. Vet. Diagn. Investig. 1997, 9, 201–203. [Google Scholar] [CrossRef]
- McDowell, L.R. Minerals in animal and human nutrition. Minerals in Animal and Human Nutrition, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review on iron and its importance for human health. J. Res. Med. Sci. 2014, 19, 164–174. [Google Scholar] [PubMed]
- Albretsen, J. The toxicity of iron, an essential element. Vet. Med. 2006, 101, 82–90. [Google Scholar]
- Andrews, N.C. Forging a field: The golden age of iron biology. Blood 2008, 112, 219–230. [Google Scholar] [CrossRef]
- McCown, J.L.; Specht, A.J. Iron Homeostasis and disorders in dogs and cats: A review. J. Am. Anim. Hosp. Assoc. 2011, 47, 151–160. [Google Scholar] [CrossRef]
- Davies, M.; Alborough, R.; Jones, L.; Davis, C.; Williams, C.; Gardner, D.S. Mineral analysis of complete dog and cat foods in the UK and compliance with European guidelines. Sci. Rep. 2017, 7, 17107. [Google Scholar] [CrossRef]
- FEDIAF. Nutritional Guidelines for Complete and Complementary Pet Food for Cats and Dogs; The European Pet Food Industry: Bruxelles, Belgium, 2013. [Google Scholar]
- FEDIAF. Nutritional Guidelines for Complete and Complementary Pet Food for Cats and Dogs; The European Pet Food Industry: Bruxelles, Belgium, 2019. [Google Scholar]
- Biel, W.; Czerniawska-Piątkowska, E.; Kowalczyk, A. Offal chemical composition from veal, beef, and lamb maintained in organic production systems. Animals 2019, 9, 489. [Google Scholar] [CrossRef] [PubMed]
- Quicke, D.L.J.; Wyeth, P.; Fawke, J.D.; Basibuyuk, H.H.; Vincent, J.F.V. Manganese and zinc in the ovipositors and mandibles of hymenopterous insects. Zool. J. Linn. Soc. 1998, 124, 387–396. [Google Scholar] [CrossRef]
- Morgan, T.D.; Baker, P.; Kramer, K.J.; Basibuyuk, H.H.; Quicke, D.L.J. Metals in mandibles of stored product insects: Do zinc and manganese enhance the ability of larvae to infest seeds? J. Stored Prod. Res. 2003, 39, 65–75. [Google Scholar] [CrossRef]
- Ben-Shahar, Y. The impact of environmental Mn exposure on insect biology. Front. Genet. 2018, 9, 70. [Google Scholar] [CrossRef] [PubMed]
- Ushakova, N.A.; Dontsov, A.E.; Sakina, N.L.; Ratnikova, I.A.; Gavrilova, N.N.; Garmash, N.Y.; Bastrakov, A.I.; Kozlova, A.A. Melanin and melanogenesis at different life stages in Hermetia illucens. Biol. Bull. Russ. Acad. Sci. 2018, 45, 47–50. [Google Scholar] [CrossRef]
- Fordahl, S.C.; Anderson, J.G.; Cooney, P.T.; Weaver, T.L.; Colyer, C.L.; Erikson, K.M. Manganese exposure inhibits the clearance of extracellular GABA and influences taurine homeostasis in the striatum of developing rats. Neurotoxicology 2010, 31, 639–646. [Google Scholar] [CrossRef]
- Peneder, T.M.; Scholze, P.; Berger, M.L.; Reither, H.; Heinze, G.; Bertl, J.; Bauer, J.; Richfield, E.K.; Hornykiewicz, O.; Pifl, C. Chronic exposure to manganese decreases striatal dopamine turnover in human alpha-synuclein transgenic mice. Neuroscience 2011, 180, 280–292. [Google Scholar] [CrossRef] [PubMed]
- Sidoryk-Wegrzynowicz, M.; Aschner, M. Manganese toxicity in the central nervous system: The glutamine/glutamate-γ-aminobutyric acid cycle. J. Intern. Med. 2013, 273, 466–477. [Google Scholar] [CrossRef]
- Al-Lozi, A.; Nielsen, S.S.; Hershey, T.; Birke, A.; Checkoway, H.; Criswell, S.R.; Racette, B.A. Cognitive control dysfunction in workers exposed to manganese-containing welding fume: Welding and cognitive impairment. Am. J. Ind. Med. 2017, 60, 181–188. [Google Scholar] [CrossRef]
- Ottoboni, M.; Spranghers, T.; Pinotti, L.; Baldi, A.; Jaeghere, W.D.; Eeckhout, M. Inclusion of Hermetia Illucens larvae or prepupae in an experimental extruded feed: Process optimisation and impact on in vitro digestibility. Ital. J. Anim. Sci. 2018, 17, 418–427. [Google Scholar] [CrossRef]
- Sanecki, R.K.; Corbin, J.E.; Forbes, R.M. Tissue changes in dogs fed a zinc-deficient ration. Am. J. Vet. Res. 1982, 43, 1642–1646. [Google Scholar]
- Soltanian, A.; Khoshnegah, J.; Heidarpour, M. Comparison of serum trace elements and antioxidant levels in terrier dogs with or without behavior problems. App. Anim. Behav. Sci. 2016, 180, 87–92. [Google Scholar] [CrossRef]
- Goi, A.; Manuelian, C.L.; Currò, S.; Marchi, M. Prediction of mineral composition in commercial extruded dry dog food by near-infrared reflectance spectroscopy. Animals 2019, 9, 640. [Google Scholar] [CrossRef] [PubMed]
- Forbes, R.M.; Erdman, J.W. Bioavailability of Trace Mineral Elements. Ann. Rev. Nutr. 1983, 3, 213–231. [Google Scholar] [CrossRef]
- Gregório, B.J.R.; Pereira, A.M.; Fernandes, S.R.; Matos, E.; Castanheira, F.; Almeida, A.A.; Fonseca, A.J.M.; Cabrita, A.R.J.; Segundo, M.A. Flow-Based dynamic approach to assess bioaccessible zinc in dry dog food samples. Molecules 2020, 25, 1333. [Google Scholar] [CrossRef]
- Kelly, O.J.; Gilman, J.C.; Ilich, J.Z. Utilizing dietary micronutrient ratios in nutritional research may be more informative than focusing on single nutrients. Nutrients 2018, 10, 107. [Google Scholar] [CrossRef]
- de Fornel-Thibaud, P.; Blanchard, G.; Escoffier-Chateau, L.; Segond, S.; Guetta, F.; Begon, D.; Delisle, F.; Rosenberg, D. Unusual case of osteopenia associated with nutritional calcium and vitamin D deficiency in an adult dog. J. Am. Anim. Hosp. Assoc. 2007, 43, 52–60. [Google Scholar] [CrossRef]
- Böswald, L.F.; Klein, C.; Dobenecker, B.; Kienzle, E. Factorial calculation of calcium and phosphorus requirements of growing dogs. PLoS ONE 2019, 14, e0220305. [Google Scholar] [CrossRef]
- Castiglioni, S.; Cazzaniga, A.; Albisetti, W.; Maier, J.A.M. Magnesium and osteoporosis: Current state of knowledge and future research directions. Nutrients 2013, 5, 3022–3033. [Google Scholar] [CrossRef]
- Howard, K.A.; Rogers, Q.R.; Morris, J.G. Magnesium requirement of kittens is increased by high dietary calcium. J. Nutr. 1998, 128, 2601S–2602S. [Google Scholar] [CrossRef]
- Abbrecht, P.H. Cardiovascular effects of chronic potassium deficiency in the dog. Am. J. Physiol. 1972, 223, 555–560. [Google Scholar] [CrossRef][Green Version]
- Kogika, M.M.; de Morais, H.A. A Quick reference on hyperkalemia. Vet. Clin. N. Am. Small Anim. Pract. 2017, 47, 223–228. [Google Scholar] [CrossRef]
- Ueda, Y.; Hopper, K.; Epstein, S.E. Incidence, Severity and prognosis associated with hyponatremia in dogs and cats. J. Vet. Intern. Med. 2015, 29, 801–807. [Google Scholar] [CrossRef]
- Pak, S.I. The clinical implication of sodium-potassium ratios in dogs. J. Vet. Sci. 2000, 1, 61–65. [Google Scholar] [CrossRef]
- Wedekind, K.J.; Titgemeyer, E.C.; Twardock, A.R.; Baker, D.H. Phosphorus, but not calcium, affects manganese absorption and turnover in chicks. J. Nutr. 1991, 121, 1776–1786. [Google Scholar] [CrossRef] [PubMed]
- Peres, T.V.; Schettinger, M.R.C.; Chen, P.; Carvalho, F.; Avila, D.S.; Bowman, A.B.; Aschner, M. Manganese-induced neurotoxicity: A review of its behavioral consequences and neuroprotective strategies. BMC Pharmacol. Toxicol. 2016, 17, 57. [Google Scholar] [CrossRef]
- Ferreira, M.; Mellanby, R.; Gow, A.; Aylor, A.; Campbell, S. Whole blood manganese concentrations in anaemic dogs with inflammatory bowel disease or confirmed iron-deficiency anaemia. In Proceedings of the BSAVA Congress Proceedings 2017; British Small Animal Veterinary Association, Birmingham, UK, 6–9 April 2017; p. 486. [Google Scholar]
- Duran, A.; Tuzen, M.; Soylak, M. Trace element concentrations of some pet foods commercially available in Turkey. Food Chem. Toxicol. 2010, 48, 2833–2837. [Google Scholar] [CrossRef]
- Luippold, A.; Gustin, M.S. Mercury concentrations in wet and dry cat and dog food. Anim. Feed Sci. Technol. 2016, 222, 190–193. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 21st ed.; Association of Official Analytical Chemists: Gaithersburg, MD, USA, 2019. [Google Scholar]
- ISO Method 6491, Animal Feeding Stuffs—Determination of Phosphorus Content—Spectrometric Method; International Organization for Standardization: Geneva, Switzerland, 1998.
- TIBCO. Available online: https://www.scribd.com/document/321061529/STATISTICA-Electronic-Manual (accessed on 23 October 2020).
- Cohen, J. rc: A profile similarity coefficient invariant over variable reflection. Psychol. Bull. 1969, 71, 281–284. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds used in this work are available from the authors. |

| No 2 | Dry Matter (g/100 g) | Crude Protein | Ether Extract | Crude Fiber | Crude Ash | NFE 3 | ME 4 |
|---|---|---|---|---|---|---|---|
| 1 | 93.77 kl | 34.14 n | 21.39 p | 13.23 op | 7.28 mn | 17.74 a | 400.0 nop |
| 2 | 92.85 fghi | 28.14 ijkl | 11.61 cdefg | 5.59 cdefg | 7.78 o | 39.74 efg | 376.0 defghi |
| 3 | 92.86 ghi | 24.40 cd | 9.82 bcdef | 5.71 defgh | 7.74 o | 45.21 hijk | 366.8 cde |
| 4 | 92.45 cde | 29.42 lm | 12.26 efgh | 7.30 ghijk | 5.92 cd | 37.56 def | 378.2 defghi |
| 5 | 92.46 cde | 25.71 def | 8.02 ab | 5.83 defghi | 9.92 r | 43.00 gh | 347.0 a |
| 6 | 92.45 cde | 28.27 ijkl | 14.74 hijklm | 5.94 defghi | 6.74 jk | 36.77 de | 392.8 jklmnop |
| 7 | 93.60 k | 26.05 efg | 14.72 hijklm | 4.12 abcde | 6.07 cde | 42.65 gh | 407.2 pq |
| 8 | 96.91 q | 21.95 a | 16.98 lmno | 11.55 mno | 6.49 hi | 39.95 efg | 400.4 nop |
| 9 | 94.51 n | 22.73 ab | 9.82 bcdef | 8.07 hijkl | 4.93 a | 48.96 jklm | 375.1 cdefghi |
| 10 | 96.82 q | 29.27 klm | 12.62 ghi | 7.30 ghijk | 5.87 c | 41.78 fgh | 397.7 lmnop |
| 11 | 95.87 o | 38.12 o | 17.98 o | 11.76 no | 7.71 o | 20.32 a | 395.5 klmnop |
| 12 | 97.39 r | 38.07 o | 16.49 klmno | 4.32 bcdef | 9.88 r | 28.64 b | 415.2 q |
| 13 | 96.34 p | 25.14 cde | 11.81 efg | 6.59 fghij | 7.19 lmn | 45.61 hijk | 389.3 ijklmno |
| 14 | 95.95 o | 24.29 cd | 11.84 efg | 6.52 efghij | 8.56 p | 44.75 hij | 382.7 fghijk |
| 15 | 93.48 jk | 28.91 jklm | 8.89 abc | 6.39 efghij | 7.08 lm | 42.23 gh | 364.5 bcd |
| 16 | 92.56 defg | 23.90 bc | 6.31 a | 6.35 efghij | 6.54 hij | 49.46 klm | 350.2 ab |
| 17 | 94.14 m | 27.92 ijk | 6.76 a | 3.22 abc | 7.17 lmn | 49.07 klm | 368.8 cdef |
| 18 | 92.82 fghi | 21.40 a | 12.33 fgh | 8.20 ijkl | 7.31 n | 43.59 ghi | 370.9 cdefg |
| 19 | 92.15 c | 26.26 efg | 15.07 ijklmn | 7.84 ghijkl | 7.30 n | 35.68 de | 383.4 ghijkl |
| 20 | 92.37 cd | 30.25 m | 11.65 defg | 8.52 jkl | 8.05 p | 33.91 cd | 361.4 abc |
| 21 | 91.11 a | 38.97 o | 16.47 klmno | 7.59 ghijkl | 7,82 o | 20.27 a | 385.2 ghijklm |
| 22 | 92.85 fghi | 34.23 n | 18.21 o | 15.14 p | 6.37 fgh | 18.92 a | 376.4 defghi |
| 23 | 93.15 ij | 34.74 n | 17.25 mno | 4.24 bcdef | 9.10 q | 27.83 b | 405.5 pq |
| 24 | 91.07 a | 30.55 m | 7.08 a | 4.86 bcdef | 7.89 o | 53.17 n | 398.58 lmnop |
| 25 | 92.53 def | 29.20 jklm | 16.81 lmno | 11.90 no | 4.80 a | 29.83 bc | 387.3 hijklmn |
| 26 | 93.57 k | 27.75 hij | 13.51 ghij | 9.13 klm | 6.68 ijk | 36.50 de | 378.6 defghij |
| 27 | 92.68 defgh | 26.39 efgh | 13.90 ghijk | 2.90 ab | 6.43 gh | 43.07 gh | 402.9 opq |
| 28 | 91.54 b | 22.58 ab | 9.55 bcde | 3.59 abcd | 5.42 b | 50.41 lmn | 377.9 defghi |
| 29 | 91.64 b | 35.41 n | 15.77 jklmno | 7.75 ghijkl | 7.10 mno | 25.62 b | 386.0 hijklmn |
| 30 | 97.55 r | 22.41 a | 17.62 no | 13.38 op | 6.66 ijk | 37.49 def | 398.2 mnop |
| MRL 5 | 18.00 | 5.50 | - | - | - | - | |
| No 2 | Ca | P | K | Na | Mg |
|---|---|---|---|---|---|
| 1 | 0.451 a | 1.075 o | 1.349 r | 0.660 s | 0.094 a |
| 2 | 0.759 cde | 0.725 efg | 0.953 kl | 0.363 d | 0.132 h |
| 3 | 1.296 m | 0.793 ij | 0.882 j | 0.407 f | 0.120 cd |
| 4 | 0.751 cde | 0.535 b | 0.831 i | 0.313 b | 0.097 a |
| 5 | 1.640 pq | 0.856 kl | 1.082 o | 0.431 hi | 0.152 k |
| 6 | 1.103 jkl | 0.535 b | 0.706 f | 0.450 l | 0.167 m |
| 7 | 0.995 ghijk | 0.717 ef | 0.794 h | 0.396 e | 0.094 a |
| 8 | 0.857 defg | 0.475 a | 1.182 q | 0.482 m | 0.184 o |
| 9 | 0.636 bc | 0.599 c | 0.939 k | 0.251 a | 0.172 n |
| 10 | 0.882 efgh | 0.897 lm | 0.588 ab | 0.395 e | 0.182 o |
| 11 | 0.964 fghij | 0.818 jk | 0.883 j | 0.687 t | 0.152 k |
| 12 | 1.522 op | 0.660 d | 1.004 m | 0.834 u | 0.181 o |
| 13 | 1.109 kl | 0.765 ghi | 0.638 c | 0.535 o | 0.138 i |
| 14 | 1.476 no | 0.777 i | 0.610 b | 0.504 n | 0.126 fg |
| 15 | 0.561 ab | 0.674 d | 0.710 f | 0.445 kl | 0.218 r |
| 16 | 0.855 def | 0.548 b | 0.849 i | 0.533 o | 0.112 b |
| 17 | 1.145 l | 0.928 mn | 0.966 l | 0.425 h | 0.128 gh |
| 18 | 1.554 op | 0.900 mn | 0.652 cd | 0.438 jk | 0.120 d |
| 19 | 1.284 m | 0.754 fghi | 0.710 f | 0.417 g | 0.139 i |
| 20 | 1.605 op | 0.896 lm | 0.827 i | 0.437 ij | 0.125 efg |
| 21 | 1.030 ijkl | 0.938 n | 1.001 m | 0.828 u | 0.189 p |
| 22 | 0.741 cd | 0.537 b | 0.997 m | 0.631 r | 0.159 m |
| 23 | 1.746 q | 0.754 fghi | 0.832 i | 0.503 n | 0.149 jk |
| 24 | 1.543 op | 0.843 k | 0.744 g | 0.575 q | 0.148 j |
| 25 | 0.539 ab | 0.735 fgh | 0.833 i | 0.417 g | 0.121 de |
| 26 | 1.315 m | 0.690 de | 0.676 e | 0.436 ij | 0.116 bc |
| 27 | 1.345 mn | 0.914 mn | 0.663 de | 0.406 f | 0.124 def |
| 28 | 0.915 fghi | 0.767 hi | 0.568 a | 0.328 c | 0.124 def |
| 29 | 0.952 fghi | 1.204 p | 1.050 n | 0.564 p | 0.163 m |
| 30 | 1.018 hijkl | 0.549 b | 1.149 p | 0.430 hi | 0.200 q |
| Contrast | |||||
| Cereal-free foods | 1.083 a | 0.771 b | 0.964 b | 0.527 b | 0.145 a |
| Cereal foods | 1.091 a | 0.750 a | 0.713 a | 0.428 a | 0.144 a |
| MRL 3 | 0.500 | 0.400 | 0.500 | 0.100 | 0.07 |
| MNL 4 | 2.500 | 1.600 | - | - | - |
| No 2 | Microelements | Heavy Metals 5 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cu | Fe | Mn | Zn | Co | Cd | Pb | Mo | Cr | Ni | |
| 1 | 2.374 kl | 55.99 s | 4.035 l | 21.22 hi | BDL | BDL | BDL | BDL | BDL | BDL |
| 2 | 2.501 mn | 99.56 x | 6.505 p | 11.11 abcd | BDL | BDL | BDL | BDL | BDL | BDL |
| 3 | 2.157 j | 49.47 p | 4.429 m | 9.52 a | BDL | BDL | BDL | BDL | BDL | BDL |
| 4 | 2.604 o | 56.25 s | 4.589 n | 10.45 ab | BDL | BDL | BDL | BDL | BDL | BDL |
| 5 | 1.277 a | 72.61 u | 5.936 o | 10.58 abc | BDL | BDL | BDL | BDL | BDL | BDL |
| 6 | 1.218 a | 29.25 f | 3.142 i | 13.02 abcde | BDL | BDL | BDL | BDL | BDL | BDL |
| 7 | 2.578 no | 33.60 i | 4.526 mn | 15.72 def | BDL | BDL | BDL | BDL | BDL | BDL |
| 8 | 2.142 j | 44.05 m | 21.210 v | 15.90 defg | BDL | BDL | BDL | BDL | BDL | BDL |
| 9 | 1.904 gh | 29.79 f | 9.522 t | 10.98 abcd | BDL | BDL | BDL | BDL | BDL | BDL |
| 10 | 1.629 ef | 31.93 gh | 3.330 j | 12.92 abcde | BDL | BDL | BDL | BDL | BDL | BDL |
| 11 | 1.693 f | 84.60 w | 7.904 s | 18.77 fghi | BDL | BDL | BDL | BDL | BDL | BDL |
| 12 | 2.825 p | 53.86 r | 7.042 q | 40.43 k | BDL | BDL | BDL | BDL | BDL | BDL |
| 13 | 2.290 k | 51.47 q | 7.127 q | 30.81 j | BDL | BDL | BDL | BDL | BDL | BDL |
| 14 | 1.583 de | 77.89 v | 7.137 q | 17.22 efgh | BDL | BDL | BDL | BDL | BDL | BDL |
| 15 | 1.497 cd | 63.65 t | 7.788 r | 17.97 fghi | BDL | BDL | BDL | BDL | BDL | BDL |
| 16 | 1.677 f | 45.90 n | 3.125 i | 17.02 efgh | BDL | BDL | BDL | BDL | BDL | BDL |
| 17 | 1.855 g | 29.33 f | 2.594 g | 15.48 cdef | BDL | BDL | BDL | BDL | BDL | BDL |
| 18 | 1.406 bc | 31.47 g | 3.600 k | 18.17 fghi | BDL | BDL | BDL | BDL | BDL | BDL |
| 19 | 2.002 i | 48.09 o | 2.350 f | 20.70 ghi | BDL | BDL | BDL | BDL | BDL | BDL |
| 20 | 2.927 q | 42.81 l | 2.071 d | 29.08 j | BDL | BDL | BDL | BDL | BDL | BDL |
| 21 | 2.288 k | 14.37 b | 0.998 a | 21.51 hi | BDL | BDL | BDL | BDL | BDL | BDL |
| 22 | 2.416 lm | 28.31 e | 2.550 g | 31.54 j | BDL | BDL | BDL | BDL | BDL | BDL |
| 23 | 1.683 f | 45.89 n | 2.916 h | 17.73 efghi | BDL | BDL | BDL | BDL | BDL | BDL |
| 24 | 2.162 j | 40.70 k | 2.508 g | 27.56 j | BDL | BDL | BDL | BDL | BDL | BDL |
| 25 | 1.634 ef | 26.91 d | 1.685 c | 20.98 hi | BDL | BDL | BDL | BDL | BDL | BDL |
| 26 | 1.716 f | 32.11 h | 2.247 e | 18.46 fghi | BDL | BDL | BDL | BDL | BDL | BDL |
| 27 | 1.685 f | 29.46 f | 2.199 e | 18.42 fghi | BDL | BDL | BDL | BDL | BDL | BDL |
| 28 | 1.395 b | 18.23 c | 1.374 b | 15.12 bcdef | BDL | BDL | BDL | BDL | BDL | BDL |
| 29 | 1.922 ghi | 12.75 a | 1.092 a | 22.53 i | BDL | BDL | BDL | BDL | BDL | BDL |
| 30 | 1.960 hi | 35.72 j | 18.300 u | 13.87 abcdef | BDL | BDL | BDL | BDL | BDL | BDL |
| Contrast | ||||||||||
| Cereal-free foods | 2.185 b | 46.91 b | 5.782 b | 19.91 b | - | - | - | - | - | - |
| Cereal foods | 1.681 a | 39.89 a | 4.272 a | 17.41 a | - | - | - | - | - | - |
| MRL 3 | 0.720 | 3.60 | 0.580 | 7.20 | - | - | - | - | - | - |
| MLL 4 | 2.800 | 68.18 | 17.000 | 22.70 | - | - | - | - | - | - |
| No | Cu | Fe | Mn | Zn |
|---|---|---|---|---|
| 1 | 17.48 | 3.11 | 34.30 | 9.33 |
| 2 | 66.36 | 5.74 | 142.36 | 102.15 |
| 3 | 76.95 | 11.55 | 209.07 | 119.23 |
| 4 | 63.74 | 10.16 | 201.78 | 108.64 |
| 5 | 129.94 | 7.87 | 155.98 | 107.35 |
| 6 | NDL | NDL | NDL | NDL |
| 7 | 16.46 | 7.61 | 66.54 | 45.43 |
| 8 | 50.74 | NDL | NDL | 68.37 |
| 9 | 20.80 | 5.88 | 14.01 | 17.69 |
| 10 | NDL | NDL | NDL | NDL |
| 11 | 12.53 | 8.68 | 68.59 | 57.06 |
| 12 | 8.01 | 13.63 | 75.76 | 26.49 |
| 13 | 9.88 | 14.26 | 76.07 | 15.77 |
| 14 | 14.29 | 9.42 | 75.96 | 28.20 |
| 15 | 14.17 | 11.53 | 69.05 | 59.60 |
| 16 | 16.87 | 8.75 | 68.84 | 70.84 |
| 17 | 15.24 | 13.70 | 82.95 | 72.09 |
| 18 | 20.12 | 12.77 | 59.75 | 66.32 |
| 19 | 15.89 | NDL | NDL | 44.10 |
| 20 | 10.87 | NDL | NDL | 50.24 |
| 21 | 54.64 | NDL | NDL | 42.45 |
| 22 | 45.00 | 38.39 | 42.63 | 48.25 |
| 23 | 64.58 | 23.69 | 37.27 | 85.82 |
| 24 | 12.80 | 8.78 | 33.56 | 20.12 |
| 25 | 16.93 | 13.27 | 49.95 | 26.42 |
| 26 | 16.30 | 11.25 | 8.89 | 27.10 |
| 27 | 16.59 | 12.26 | 9.09 | 27.16 |
| 28 | 19.83 | NDL | NDL | 66.44 |
| 29 | 15.05 | NDL | NDL | 40.53 |
| 30 | 14.11 | NDL | NDL | 62.98 |
| No | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | The Main Ingredients |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | beef, sweet potatoes, beans, beef fat |
| 2 | 0.31 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | chicken, sweet potatoes, peas, potatoes, chicken fat |
| 3 | 0.18 | 0.57 | - | - | - | - | - | - | - | - | - | - | - | - | - | pork, sweet potatoes, peas, potatoes, pork fat |
| 4 | 0.28 | 0.77 | 0.77 | - | - | - | - | - | - | - | - | - | - | - | - | beef, sweet potatoes, potatoes, peas, beef fat |
| 5 | −0.09 | 0.25 | 0.59 | 0.11 | - | - | - | - | - | - | - | - | - | - | - | lamb, sweet potatoes, peas, potatoes, lamb fat |
| 6 | −0.28 | 0.09 | 0.54 | 0.39 | 0.49 | - | - | - | - | - | - | - | - | - | - | chicken, brown rice, chicken fat |
| 7 | 0.32 | 0.49 | 0.81 | 0.90 | 0.04 | 0.37 | - | - | - | - | - | - | - | - | - | dried salmon, potatoes, salmon protein, chicken fat |
| 8 | −0.21 | 0.09 | −0.11 | 0.15 | −0.09 | 0.11 | −0.10 | - | - | - | - | - | - | - | - | potatoes, Hermetia illucens, poultry fat |
| 9 | −0.04 | 0.40 | 0.48 | 0.62 | 0.19 | 0.66 | 0.46 | 0.55 | - | - | - | - | - | - | - | Hermetia illucens, oats, potatoes, corn, peas, insects oil |
| 10 | −0.16 | 0.16 | 0.52 | 0.34 | 0.31 | 0.79 | 0.38 | −0.15 | 0.63 | - | - | - | - | - | - | salmon, brown rice, chicken fat |
| 11 | 0.17 | 0.40 | 0.16 | 0.09 | 0.40 | 0.28 | −0.11 | −0.21 | −0.11 | 0.20 | - | - | - | - | - | chicken, potatoes, dried peas, animal fat |
| 12 | −0.13 | −0.37 | −0.38 | −0.32 | −0.41 | −0.32 | −0.20 | −0.18 | −0.58 | −0.47 | −0.17 | - | - | - | - | salmon, potatoes, peas, animal fat |
| 13 | −0.19 | 0.03 | 0.18 | 0.35 | −0.35 | 0.23 | 0.51 | −0.50 | −0.08 | 0.29 | −0.03 | 0.30 | - | - | - | salmon, rice, animal fat |
| 14 | −0.31 | 0.32 | 0.59 | 0.33 | 0.61 | 0.54 | 0.32 | −0.38 | 0.03 | 0.43 | 0.55 | −0.28 | 0.41 | - | - | lamb, rice, animal fat |
| 15 | −0.31 | 0.25 | 0.03 | 0.15 | 0.14 | 0.64 | −0.09 | 0.24 | 0.62 | 0.70 | 0.40 | −0.36 | 0.05 | 0.20 | - | chicken, corn, rice, wheat, vegetable fiber, animal fat |
| 16 | 0.24 | 0.39 | 0.64 | 0.72 | 0.30 | 0.76 | 0.67 | 0.00 | 0.52 | 0.48 | 0.41 | −0.26 | 0.36 | 0.50 | 0.32 | barley, salmon, rabbit, whole grain oat flour, potato flakes, poultry fat |
| 17 | 0.36 | 0.20 | 0.81 | 0.50 | 0.51 | 0.59 | 0.68 | −0.29 | 0.47 | 0.64 | 0.01 | −0.41 | 0.12 | 0.34 | 0.05 | duck, corn flour, rice flour, salmon, liver, sugar beet molasses, poultry fat |
| 18 | −0.16 | −0.06 | 0.67 | 0.26 | 0.57 | 0.69 | 0.48 | −0.45 | 0.20 | 0.65 | 0.12 | −0.31 | 0.40 | 0.74 | 0.08 | lamb, rice, corn, poultry fat |
| 19 | −0.13 | 0.31 | 0.77 | 0.60 | 0.40 | 0.71 | 0.70 | −0.41 | 0.37 | 0.69 | 0.13 | −0.12 | 0.64 | 0.70 | 0.25 | lamb, oats, beef, pork, lamb fat, lentils, peas |
| 20 | 0.07 | 0.10 | 0.47 | 0.35 | −0.04 | −0.06 | 0.60 | −0.54 | −0.16 | 0.06 | −0.37 | 0.40 | 0.60 | 0.19 | −0.46 | lamb, peas, lentils, lamb liver, lamb fat |
| 21 | 0.29 | −0.31 | 0.08 | −0.12 | −0.10 | 0.22 | 0.09 | −0.22 | −0.02 | 0.32 | 0.05 | 0.41 | 0.08 | −0.25 | 0.06 | sardines, mackerel, hake, flounder, redfish, sole, herring, cod, blue whiting, herring oil, red lentils, green lentils, green peas, chickpeas, peas |
| 22 | 0.25 | −0.03 | 0.01 | 0.32 | −0.41 | 0.28 | 0.36 | 0.06 | 0.24 | 0.10 | −0.12 | 0.49 | 0.42 | −0.31 | 0.15 | white fish, herring, salmon, salmon oil, peas, potato flakes |
| 23 | −0.29 | 0.00 | 0.68 | 0.21 | 0.73 | 0.68 | 0.34 | −0.32 | 0.13 | 0.48 | 0.18 | 0.00 | 0.25 | 0.71 | 0.06 | turkey, rabbit, peas, pork fat, potatoes |
| 24 | −0.17 | −0.19 | 0.42 | 0.11 | 0.18 | 0.37 | 0.40 | −0.65 | −0.19 | 0.36 | −0.04 | 0.42 | 0.68 | 0.47 | −0.14 | lamb, potato flakes, peas, chicken protein, Saccharomyces cerevisiae, salmon oil |
| 25 | 0.35 | 0.24 | 0.50 | 0.61 | 0.10 | 0.68 | 0.65 | −0.12 | 0.63 | 0.66 | 0.14 | −0.42 | 0.36 | 0.22 | 0.41 | white fish, potato flakes, peas, animal fat, salmon, chicken |
| 26 | −0.08 | 0.15 | 0.77 | 0.58 | 0.42 | 0.79 | 0.72 | −0.29 | 0.40 | 0.65 | 0.12 | −0.22 | 0.56 | 0.69 | 0.17 | chicken, rice, peas, animal fat, beef |
| 27 | −0.04 | 0.06 | 0.74 | 0.41 | 0.46 | 0.68 | 0.63 | −0.48 | 0.32 | 0.74 | 0.06 | −0.32 | 0.49 | 0.65 | 0.13 | chicken, rice, peas, animal fat, beef |
| 28 | −0.02 | 0.16 | 0.66 | 0.55 | 0.33 | 0.84 | 0.65 | −0.20 | 0.62 | 0.85 | 0.11 | −0.49 | 0.45 | 0.55 | 0.44 | poultry, salmon, millet, barley, corn, rice, potatoes, dried beet pulp, animal fat |
| 29 | 0.46 | −0.25 | 0.23 | −0.08 | 0.11 | 0.20 | 0.20 | −0.44 | 0.12 | 0.49 | −0.12 | −0.10 | −0.02 | −0.19 | 0.01 | herring, sardines, flounder, cod, hake, green peas, red lentils, chickpeas, green lentils, red banded redfish, pinto beans, peas, pollock oil |
| 30 | −0.33 | 0.02 | 0.02 | 0.10 | 0.13 | 0.31 | −0.10 | 0.94 | 0.67 | 0.10 | −0.24 | −0.25 | −0.53 | −0.29 | 0.37 | potatoes, Hermetia illucens, poultry fat |
| No | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | 30 | The Main Ingredients |
| 16 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | barley, salmon, rabbit, whole grain oat flour, potato flakes, poultry fat |
| 17 | 0.61 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | duck, corn flour, rice flour, salmon, liver, sugar beet molasses, poultry fat |
| 18 | 0.54 | 0.76 | - | - | - | - | - | - | - | - | - | - | - | - | - | lamb, rice, corn, poultry fat |
| 19 | 0.67 | 0.71 | 0.82 | - | - | - | - | - | - | - | - | - | - | - | - | lamb, oats, beef, pork, lamb fat, lentils, peas |
| 20 | 0.08 | 0.40 | 0.38 | 0.60 | - | - | - | - | - | - | - | - | - | - | - | lamb, peas, lentils, lamb liver, lamb fat |
| 21 | 0.14 | 0.36 | 0.13 | 0.15 | 0.21 | - | - | - | - | - | - | - | - | - | - | sardines, mackerel, hake, flounder, redfish, sole, herring, cod, blue whiting, herring oil, red lentils, green lentils, green peas, chickpeas, peas |
| 22 | 0.48 | 0.16 | −0.09 | 0.26 | 0.26 | 0.56 | - | - | - | - | - | - | - | - | - | white fish, herring, salmon, salmon oil, peas, potato flakes |
| 23 | 0.47 | 0.63 | 0.84 | 0.79 | 0.44 | 0.23 | 0.01 | - | - | - | - | - | - | - | - | turkey, rabbit, peas, pork fat, potatoes |
| 24 | 0.29 | 0.47 | 0.69 | 0.76 | 0.77 | 0.46 | 0.31 | 0.76 | - | - | - | - | - | - | - | lamb, potato flakes, peas, chicken protein, Saccharomyces cerevisiae, salmon oil |
| 25 | 0.83 | 0.72 | 0.52 | 0.62 | 0.09 | 0.24 | 0.51 | 0.26 | 0.24 | - | - | - | - | - | - | white fish, potato flakes, peas, animal fat, salmon, chicken |
| 26 | 0.80 | 0.77 | 0.90 | 0.93 | 0.45 | 0.16 | 0.26 | 0.79 | 0.69 | 0.72 | - | - | - | - | - | chicken, rice, peas, animal fat, beef |
| 27 | 0.59 | 0.85 | 0.96 | 0.89 | 0.48 | 0.21 | 0.06 | 0.77 | 0.71 | 0.66 | 0.93 | - | - | - | - | chicken, rice, peas, animal fat, beef |
| 28 | 0.76 | 0.79 | 0.83 | 0.82 | 0.20 | 0.16 | 0.22 | 0.57 | 0.46 | 0.87 | 0.90 | 0.90 | - | - | - | poultry, salmon, millet, barley, corn, rice, potatoes, dried beet pulp, animal fat |
| 29 | 0.11 | 0.70 | 0.39 | 0.26 | 0.25 | 0.73 | 0.24 | 0.23 | 0.38 | 0.47 | 0.27 | 0.50 | 0.41 | - | - | herring, sardines, flounder, cod, hake, green peas, red lentils, chickpeas, green lentils, red banded redfish, pinto beans, peas, pollock oil |
| 30 | 0.02 | −0.08 | −0.24 | −0.23 | −0.48 | −0.09 | 0.01 | −0.07 | −0.49 | −0.05 | −0.14 | −0.26 | −0.02 | −0.24 | - | potatoes, Hermetia illucens, poultry fat |
 —cereal foods;
—cereal foods;  —cereal-free foods;
—cereal-free foods;  —x ≥ +0.75 (high similarity);
—x ≥ +0.75 (high similarity);  —+0.75 > x > +0.30 (moderate similarity);
—+0.75 > x > +0.30 (moderate similarity);  —+0.30 ≥ x ≥ -0.30 (no similarity);
—+0.30 ≥ x ≥ -0.30 (no similarity);  —−0.30 > x > −0.75 (moderate dissimilarity);
—−0.30 > x > −0.75 (moderate dissimilarity);  —x ≤ −0.75 (high dissimilarity).
—x ≤ −0.75 (high dissimilarity).| No | Ca:Mg | K:Mg | Na:K | K:Ca | Fe:Mn | Mn:Zn | P:Zn |
|---|---|---|---|---|---|---|---|
| 1 | 4.80:1 | 14.35:1 | 0.49:1 | 2.99:1 | 13.88:1 | 0.19:1 | 50.66:1 |
| 2 | 5.75:1 | 7.22:1 | 0.38:1 | 1.26:1 | 15.31:1 | 0.59:1 | 65.26:1 |
| 3 | 10.80:1 | 7.35:1 | 0.46:1 | 0.68:1 | 11.17:1 | 0.47:1 | 83.30:1 |
| 4 | 7.74:1 | 8.57:1 | 0.38:1 | 1.11:1 | 12.26:1 | 0.44:1 | 51.20:1 |
| 5 | 10.79:1 | 7.12:1 | 0.40:1 | 0.66:1 | 12.23:1 | 0.56:1 | 80.91:1 |
| 6 | 6.60:1 | 4.23:1 | 0.64:1 | 0.64:1 | 9.31:1 | 0.24:1 | 41.09:1 |
| 7 | 10.59:1 | 8.45:1 | 0.50:1 | 0.80:1 | 7.42:1 | 0.29:1 | 45.61:1 |
| 8 | 4.66:1 | 6.42:1 | 0.41:1 | 1.38:1 | 2.08:1 | 1.33:1 | 29.87:1 |
| 9 | 3.70:1 | 5.46:1 | 0.27:1 | 1.48:1 | 3.13:1 | 0.87:1 | 54.55:1 |
| 10 | 4.85:1 | 3.23:1 | 0.67:1 | 0.67:1 | 9.59:1 | 0.26:1 | 69.43:1 |
| 11 | 6.34:1 | 5.81:1 | 0.78:1 | 0.92:1 | 10.70:1 | 0.42:1 | 43.58:1 |
| 12 | 8.41:1 | 5.55:1 | 0.83:1 | 0.66:1 | 7.65:1 | 0.17:1 | 16.32:1 |
| 13 | 8.04:1 | 4.62:1 | 0.84:1 | 0.58:1 | 7.22:1 | 0.23:1 | 24.83:1 |
| 14 | 11.71:1 | 4.84:1 | 0.83:1 | 0.41:1 | 10.91:1 | 0.41:1 | 45.12:1 |
| 15 | 2.57:1 | 3.26:1 | 0.63:1 | 1.27:1 | 8.17:1 | 0.43:1 | 37.51:1 |
| 16 | 7.63:1 | 7.58:1 | 0.63:1 | 0.99:1 | 14.69:1 | 0.18:1 | 32.20:1 |
| 17 | 8.95:1 | 7.55:1 | 0.44:1 | 0.84:1 | 11.31:1 | 0.17:1 | 59.95:1 |
| 18 | 12.95:1 | 5.43:1 | 0.67:1 | 0.42:1 | 8.74:1 | 0.20:1 | 49.53:1 |
| 19 | 9.24:1 | 5.11:1 | 0.59:1 | 0.55:1 | 20.46:1 | 0.11:1 | 36.43:1 |
| 20 | 12.84:1 | 6.62:1 | 0.53:1 | 0.52:1 | 20.67:1 | 0.07:1 | 30.81:1 |
| 21 | 5.45:1 | 5.30:1 | 0.83:1 | 0.97:1 | 14.40:1 | 0.05:1 | 43.61:1 |
| 22 | 4.66:1 | 6.27:1 | 0.63:1 | 1.35:1 | 11.10:1 | 0.08:1 | 17.03:1 |
| 23 | 11.72:1 | 5.58:1 | 0.60:1 | 0.48:1 | 15.74:1 | 0.16:1 | 42.53:1 |
| 24 | 10.43:1 | 5.03:1 | 0.77:1 | 0.48:1 | 16.23:1 | 0.09:1 | 30.59:1 |
| 25 | 4.45:1 | 6.88:1 | 0.50:1 | 1.55:1 | 15.97:1 | 0.08:1 | 35.03:1 |
| 26 | 11.34:1 | 5.83:1 | 0.64:1 | 0.51:1 | 14.29:1 | 0.12:1 | 37.38:1 |
| 27 | 10.85:1 | 5.35:1 | 0.61:1 | 0.49:1 | 13.40:1 | 0.12:1 | 49.62:1 |
| 28 | 7.38:1 | 4.58:1 | 0.58:1 | 0.62:1 | 13.27:1 | 0.09:1 | 50.73:1 |
| 29 | 5.84:1 | 6.44:1 | 0.54:1 | 1.10:1 | 11.68:1 | 0.05:1 | 53.44:1 |
| 30 | 5.09:1 | 5.75:1 | 0.37:1 | 1.13:1 | 1.95:1 | 1.32:1 | 39.58:1 |
| Mean | 7.87:1 | 6.19:1 | 0.58:1 | 0.92:1 | 11.50:1 | 0.33:1 | 44.92:1 |
| According to MRL 1 | 7.14:1 | 7.14:1 | 0.20:1 | 1:1 | 6.21:1 | 0.08:1 | 55.56:1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kazimierska, K.; Biel, W.; Witkowicz, R. Mineral Composition of Cereal and Cereal-Free Dry Dog Foods versus Nutritional Guidelines. Molecules 2020, 25, 5173. https://doi.org/10.3390/molecules25215173
Kazimierska K, Biel W, Witkowicz R. Mineral Composition of Cereal and Cereal-Free Dry Dog Foods versus Nutritional Guidelines. Molecules. 2020; 25(21):5173. https://doi.org/10.3390/molecules25215173
Chicago/Turabian StyleKazimierska, Katarzyna, Wioletta Biel, and Robert Witkowicz. 2020. "Mineral Composition of Cereal and Cereal-Free Dry Dog Foods versus Nutritional Guidelines" Molecules 25, no. 21: 5173. https://doi.org/10.3390/molecules25215173
APA StyleKazimierska, K., Biel, W., & Witkowicz, R. (2020). Mineral Composition of Cereal and Cereal-Free Dry Dog Foods versus Nutritional Guidelines. Molecules, 25(21), 5173. https://doi.org/10.3390/molecules25215173







