Beta Glucan: Supplement or Drug? From Laboratory to Clinical Trials
Abstract
1. Why Dietary Supplements
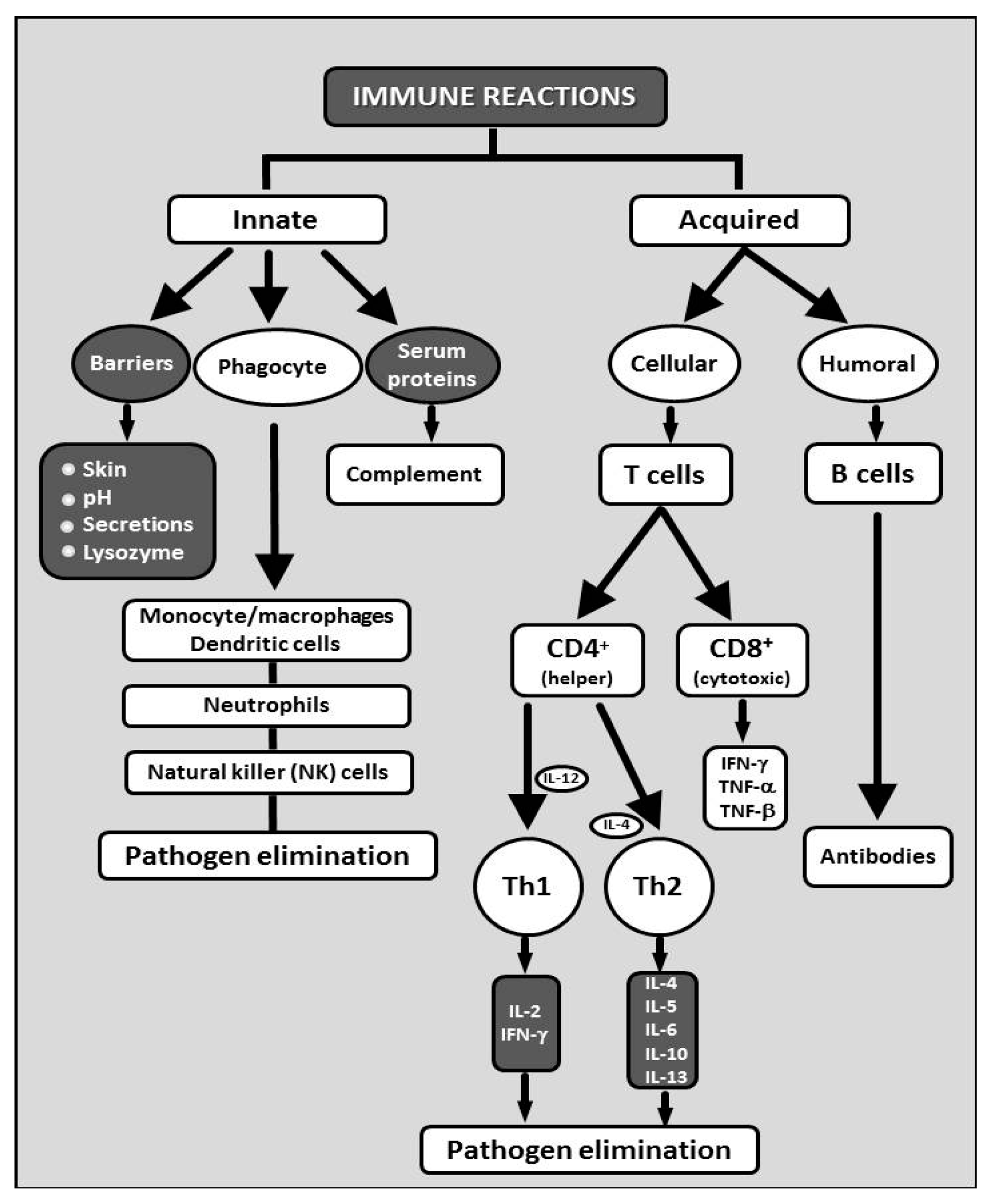
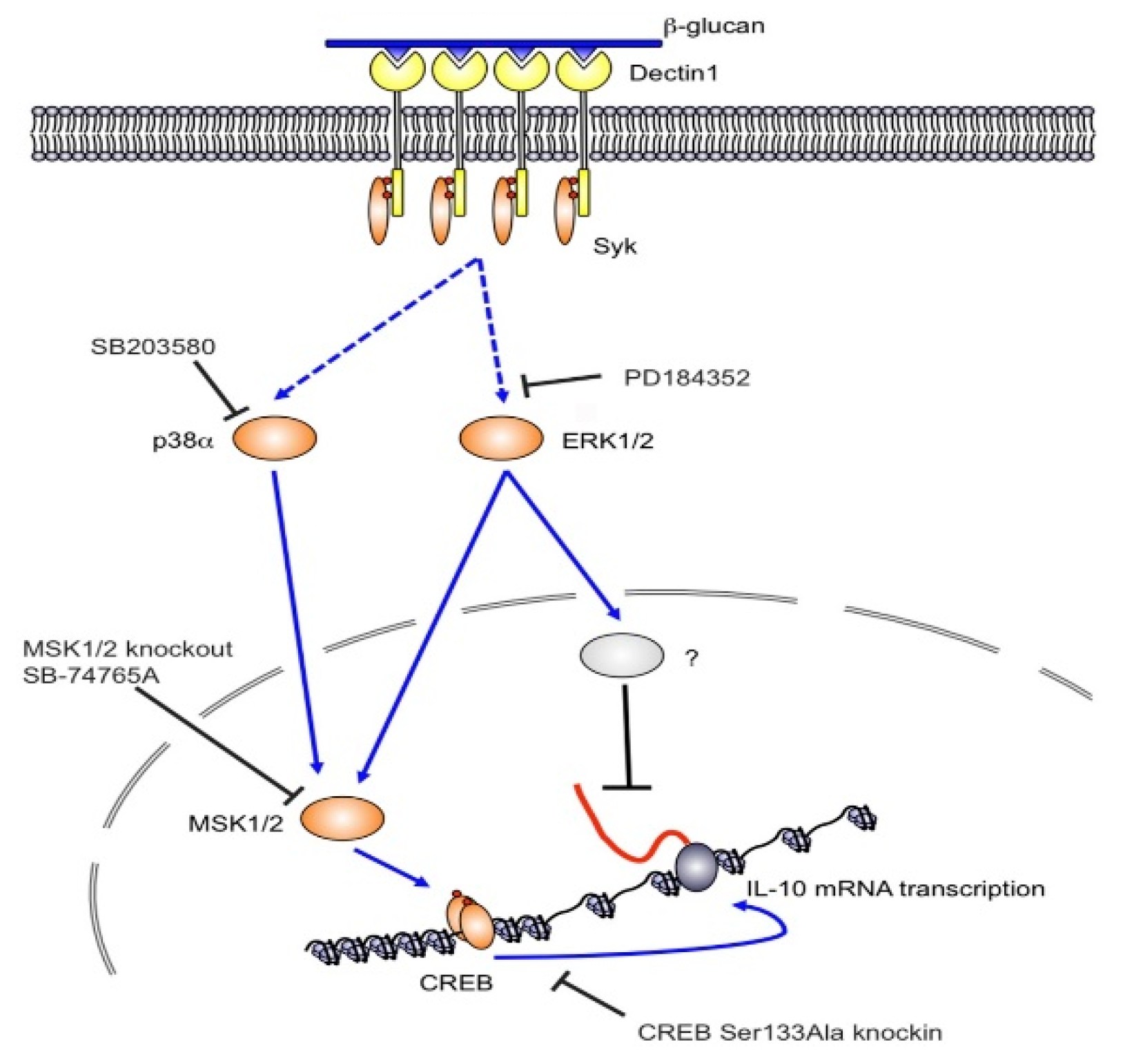
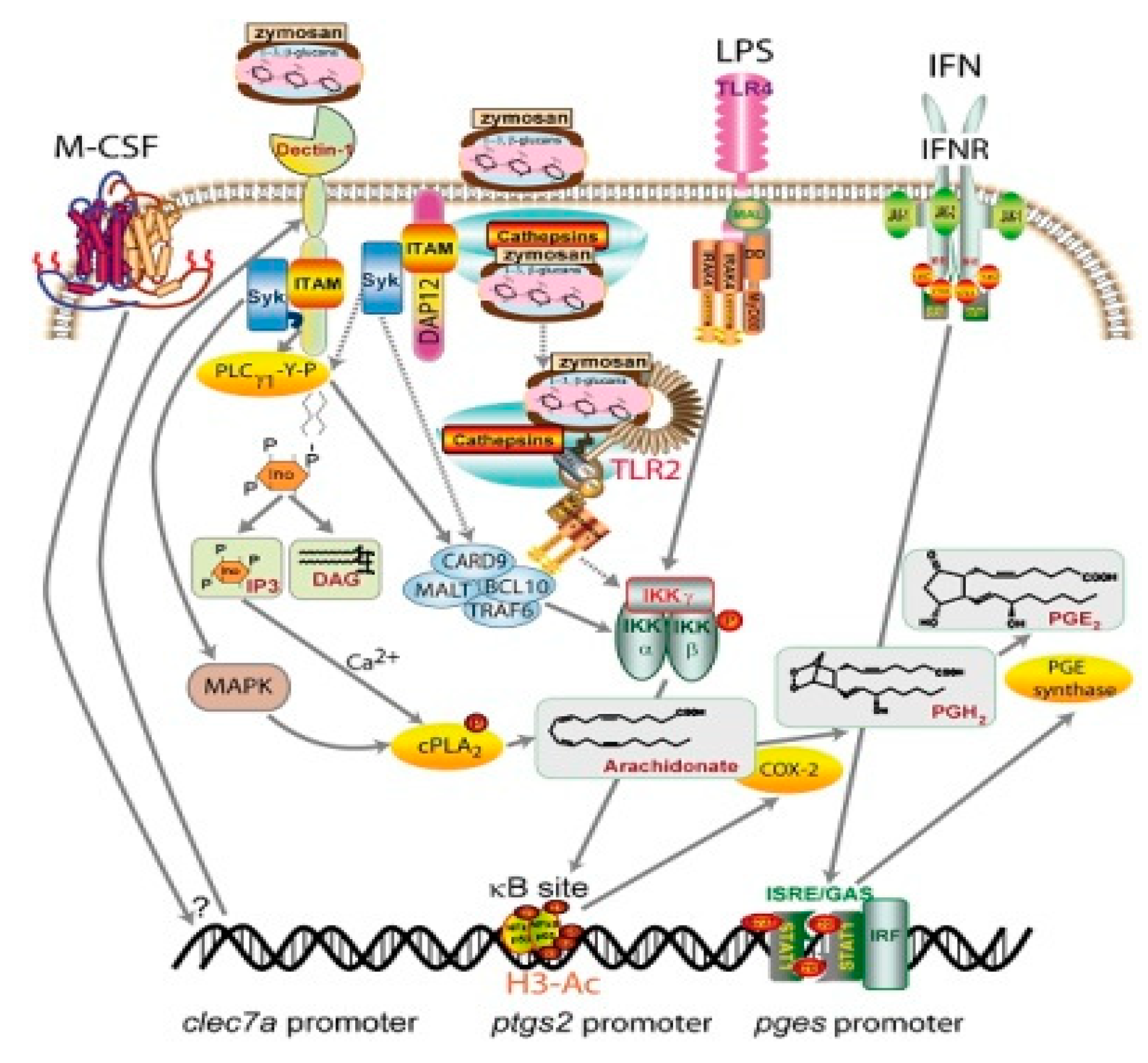
2. Glucans in the Immunity
3. Glucan in Dermatology
4. Glucan in Clinical Trials
5. Beta Glucan as Food Supplement
6. Transport through the Gut
7. Glucan and Medicine
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Scrimshaw, N.S. Historical concepts of interactions, synergism and antagonism between nutrition and infection. J. Nutr. 2003, 133, 316S–321S. [Google Scholar] [CrossRef]
- Chandra, R.K. Nutrition and immunology: From the clinic to cellular biology and back again. Proc. Nutr. Soc. 1999, 58, 681–683. [Google Scholar] [CrossRef] [PubMed]
- Mainous, M.R.; Deitch, E.A. Nutrition and infection. Surg. Clin. N. Am. 1994, 74, 659–676. [Google Scholar] [CrossRef]
- Chandra, R.K. Nutrition and Immunology; ARTS Biomedical: St John’s, NL, Canada, 1992. [Google Scholar]
- Plata-Salaman, C.R. Cytokines and feeding. Int. J. Obes. Relat. Metab. Disord. 2001, 25 (Suppl. 5), S48–S52. [Google Scholar] [CrossRef] [PubMed]
- Donabedian, H. Nutritional therapy and infectious diseases: A two-edged sword. Nutr. J. 2006, 5, 21. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. Natural immunomodulators and their stimulation of immune reaction: True or false? Anticancer Res. 2014, 34, 2275–2282. [Google Scholar] [PubMed]
- Zipfel, C.; Robatzek, S. Pathogen-associated molecular pattern-triggered immunity: Veni, vidi...? Plant Physiol. 2010, 154, 551–554. [Google Scholar] [CrossRef]
- Shear, M.J.; Turner, F.C.; Perrault, A.; Shovelton, T. Chemical treatment of tumors. V. Isolation of the hemorrhage-producing fraction from Serratia marcescens (Bacillus prodigiosus) culture filtrate. J. Natl. Cancer Inst. 1943, 4, 81–97. [Google Scholar] [CrossRef]
- Rathgeb, P.; Sylven, B. Fractionation studies on the tumor-necrotizing agent from Serratia marcescens (Shear’s polysaccharide). J. Natl. Cancer Inst. 1954, 14, 1099–1108. [Google Scholar]
- Yang, J.; Tu, J.; Liu, H.; Wen, L.; Jiang, Y.; Yang, B. Identification of an immunostimulatory polysaccharide in banana. Food Chem. 2019, 277, 46–53. [Google Scholar] [CrossRef]
- Iwanaga, S.; Lee, B.L. Recent advances in the innate immunity of invertebrate animals. J. Biochem. Mol. Biol. 2005, 38, 128–150. [Google Scholar] [CrossRef]
- Soltanian, S.; Stuyven, E.; Cox, E.; Sorgeloos, P.; Bossier, P. Beta-glucans as immunostimulant in vertebrates and invertebrates. Crit. Rev. Microbiol. 2009, 35, 109–138. [Google Scholar] [CrossRef] [PubMed]
- Riggi, S.J.; Di Luzio, N.R. Identification of a reticuloendothelial stimulating agent in zymosan. Am. J. Physiol. 1961, 200, 297–300. [Google Scholar] [CrossRef]
- Bohn, J.A.; BeMiller, J.N. (1→3)-β-d-Glucans as biological response modifiers: A review of structure-functional activity relationships. Carbohydr. Polym. 1995, 28, 3–14. [Google Scholar] [CrossRef]
- Novak, M.; Vetvicka, V. Development of views on β-glucan composition and structure. In Biology and Chemistry of Beta Glucan; Vetvicka, V., Novak, M., Eds.; Bentham Science: Sharjah, UAE, 2011; Volume 1, pp. 1–9. [Google Scholar]
- Vetvicka, V.; Dvorak, B.; Vetvickova, J.; Richter, J.; Krizan, J.; Sima, P.; Yvin, J.C. Orally administered marine (1-->3)-beta-d-glucan Phycarine stimulates both humoral and cellular immunity. Int. J. Biol. Macromol. 2007, 40, 291–298. [Google Scholar] [CrossRef]
- Hong, F.; Yan, J.; Baran, J.T.; Allendorf, D.J.; Hansen, R.D.; Ostroff, G.R.; Xing, P.X.; Cheung, N.K.; Ross, G.D. Mechanism by which orally administered beta-1,3-glucans enhance the tumoricidal activity of antitumor monoclonal antibodies in murine tumor models. J. Immunol. 2004, 173, 797–806. [Google Scholar] [CrossRef]
- Chan, G.C.; Chan, W.K.; Sze, D.M. The effects of beta-glucan on human immune and cancer cells. J. Hematol. Oncol. 2009, 2, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Moriyuki, H.; Ichimura, M. Acute toxicity of lentinan in mice and rats (author’s transl). J. Toxicol. Sci. 1980, 5, 1–9. [Google Scholar] [CrossRef]
- Vetvicka, V.; Richter, J.; Svozil, V.; Rajnohova Dobiasova, L.; Kral, V. Placebo-driven clinical trials of Transfer Point Glucan #300 in children with chronic respiratory problems: Antibody production. Am. J. Immunol. 2013, 9, 43–47. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Eweedah, N.M.; Moustafa, E.M.; Shahin, M.G. Synbiotic effects of Aspergillus oryzae and beta-glucan on growth and oxidative and immune responses of Nile tilapia, Oreochromis niloticus. Probiot. Antimicrob. Proteins 2019. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhya, A.; O’Doherty, J.V.; Sweeney, T. A combination of yeast beta-glucan and milk hydrolysate is a suitable alternative to zinc oxide in the race to alleviate post-weaning diarrhoea in piglets. Sci. Rep. 2019, 9, 616. [Google Scholar] [CrossRef] [PubMed]
- Anusuya, S.; Sathiyabama, M. Foliar application of beta-D-glucan nanoparticles to control rhizome rot disease of turmeric. Int. J. Biol. Macromol. 2015, 72, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Menard, R.; de Ruffray, P.; Fritig, B.; Yvin, J.C.; Kauffmann, S. Defense and resistance-inducing activities in tobacco of the sulfated beta-1,3 glucan PS3 and its synergistic activities with the unsulfated molecule. Plant Cell Physiol. 2005, 46, 1964–1972. [Google Scholar] [CrossRef] [PubMed]
- Al Tuwaijri, A.S.; Mahmoud, A.A.; Al Mofleh, I.A.; Al Khuwaitir, S.A. Effect of glucan on Leishmania major infection in BALB/c mice. J. Med. Microbiol. 1987, 23, 363–365. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.A.; Holbrook, T.W. Immunogenicity of soluble and particulate antigens from Leishmania donovani: Effect of glucan as an adjuvant. Infect. Immun. 1983, 40, 1038–1043. [Google Scholar]
- Bacon, J.S.; Farmer, V.C. The presence of a predominantly beta(1-6)component in preparations of yeast glucan. Biochem. J. 1968, 110, 34P–35P. [Google Scholar] [CrossRef]
- Bousquet, M.; Escoula, L.; Pipy, B.; Bessieres, M.H.; Chavant, L.; Seguela, J.P. Enhancement of resistance of mice Toxoplasma gondii by 2 polysaccharides beta 1-3, beta 1-6 (PSAT and Scleroglucan). Ann. Parasitol. Hum. Comp. 1988, 63, 398–409. [Google Scholar] [CrossRef] [PubMed]
- Dritz, S.S.; Shi, J.; Kielian, T.L.; Goodband, R.D.; Nelssen, J.L.; Tokach, M.D.; Chengappa, M.M.; Smith, J.E.; Blecha, F. Influence of dietary beta-glucan on growth performance, nonspecific immunity, and resistance to Streptococcus suis infection in weanling pigs. J. Anim. Sci. 1995, 73, 3341–3350. [Google Scholar] [CrossRef]
- Kumar, P.; Ahmad, S. Glucan-induced immunity in mice against Plasmodium berghei. Ann. Trop. Med. Parasitol. 1985, 79, 211–213. [Google Scholar] [CrossRef]
- Liang, J.; Melican, D.; Cafro, L.; Palace, G.; Fisette, L.; Armstrong, R.; Patchen, M.L. Enhanced clearance of a multiple antibiotic resistant Staphylococcus aureus in rats treated with PGG-glucan is associated with increased leukocyte counts and increased neutrophil oxidative burst activity. Int. J. Immunopharmacol. 1998, 20, 595–614. [Google Scholar] [CrossRef]
- Rasmussen, L.T.; Seljelid, R. Dynamics of blood components and peritoneal fluid during treatment of murine E. coli sepsis with beta-1,3-D-polyglucose derivatives. I. Cells. Scand. J. Immunol. 1990, 32, 321–331. [Google Scholar] [CrossRef] [PubMed]
- White, T.R.; Thompson, R.C.; Penhale, W.J.; Chihara, G. The effect of lentinan on the resistance of mice to Mesocestoides corti. Parasitol. Res. 1988, 74, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.L.; Yaeger, R.G.; Pretus, H.A.; Browder, I.W.; McNamee, R.B.; Jones, E.L. Immunization against Trypanosoma cruzi: Adjuvant effect of glucan. Int. J. Immunopharmacol. 1989, 11, 403–410. [Google Scholar] [CrossRef]
- Yun, C.H.; Estrada, A.; Van Kessel, A.; Gajadhar, A.; Redmond, M.; Laarveld, B. Immunomodulatory effects of oat beta-glucan administered intragastrically or parenterally on mice infected with Eimeria vermiformis. Microbiol. Immunol. 1998, 42, 457–465. [Google Scholar]
- Vetvicka, V.; Terayam, K.; Mandeville, R.; Brousseau, P.; Kournikakis, B.; Ostroff, G. Pilot study: Orally-administered yeast β1,3-glucan prophylactically protects against anthrax infectin and cancer in mice. JANA 2002, 5, 1–5. [Google Scholar]
- Nakao, I.; Uchino, H.; Orita, K.; Kaido, I.; Kimura, T.; Goto, Y.; Kondo, T.; Takino, T.; Taguchi, T.; Nakajima, T.; et al. Clinical evaluation of schizophyllan (SPG) in advanced gastric cancer—A randomized comparative study by an envelope method. Gan To Kagaku Ryoho 1983, 10, 1146–1159. [Google Scholar] [PubMed]
- Malyarenko, O.S.; Usoltseva, R.V.; Zvyagintseva, T.N.; Ermakova, S.P. Laminaran from brown alga Dictyota dichotoma and its sulfated derivative as radioprotectors and radiosensitizers in melanoma therapy. Carbohydr. Polym. 2019, 206, 539–547. [Google Scholar] [CrossRef]
- Bouike, G.; Nishitani, Y.; Shiomi, H.; Yoshida, M.; Azuma, T.; Hashimoto, T.; Kanazawa, K.; Mizuno, M. Oral treatment with extract of Agaricus blazei murill enhanced Th1 Response through intestinal epithelial cells and suppressed OVA-sensitized allergy in mice. Evid. Based Complement. Altern. Med. 2011, 2011, 532180. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Hansen, R.D.; Yan, J.; Allendorf, D.J.; Baran, J.T.; Ostroff, G.R.; Ross, G.D. Beta-glucan functions as an adjuvant for monoclonal antibody immunotherapy by recruiting tumoricidal granulocytes as killer cells. Cancer Res. 2003, 63, 9023–9031. [Google Scholar]
- Ross, G.D.; Vetvicka, V.; Yan, J.; Xia, Y.; Vetvickova, J. Therapeutic intervention with complement and beta-glucan in cancer. Immunopharmacology 1999, 42, 61–74. [Google Scholar] [CrossRef]
- Xu, H.; Zou, S.; Xu, X. The beta-glucan from Lentinus edodes suppresses cell proliferation and promotes apoptosis in estrogen receptor positive breast cancers. Oncotarget 2017, 8, 86693–86709. [Google Scholar] [CrossRef]
- Fortin, O.; Aguilar-Uscanga, B.R.; Vu, K.D.; Salmieri, S.; Lacroix, M. Effect of Saccharomyces boulardii cell wall extracts on colon cancer prevention in male F344 rats treated with 1,2-dimethylhydrazine. Nutr. Cancer 2018, 70, 632–642. [Google Scholar] [CrossRef] [PubMed]
- Větvička, V.C. [Beta]-Glucans as Natural Biological Response Modifiers; Nova Science Publishers, Inc.: New York, NY, USA, 2013. [Google Scholar]
- Schepetkin, I.A.; Quinn, M.T. Botanical polysaccharides: Macrophage immunomodulation and therapeutic potential. Int. Immunopharmacol. 2006, 6, 317–333. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vetvickova, J. Physiological effects of different types of beta-glucan. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2007, 151, 225–231. [Google Scholar] [CrossRef]
- Ali, M.F.; Driscoll, C.B.; Walters, P.R.; Limper, A.H.; Carmona, E.M. Beta-glucan-activated human B lymphocytes participate in innate immune responses by releasing proinflammatory cytokines and stimulating neutrophil chemotaxis. J. Immunol. 2015, 195, 5318–5326. [Google Scholar] [CrossRef]
- Elcombe, S.E.; Naqvi, S.; Van Den Bosch, M.W.; MacKenzie, K.F.; Cianfanelli, F.; Brown, G.D.; Arthur, J.S. Dectin-1 regulates IL-10 production via a MSK1/2 and CREB dependent pathway and promotes the induction of regulatory macrophage markers. PLoS ONE 2013, 8, e60086. [Google Scholar] [CrossRef]
- Municio, C.; Alvarez, Y.; Montero, O.; Hugo, E.; Rodriguez, M.; Domingo, E.; Alonso, S.; Fernandez, N.; Crespo, M.S. The response of human macrophages to beta-glucans depends on the inflammatory milieu. PLoS ONE 2013, 8, e62016. [Google Scholar] [CrossRef] [PubMed]
- Bashir, K.M.I.; Choi, J.S. Clinical and physiological perspectives of beta-glucans: The past, present, and future. Int. J. Mol. Sci. 2017, 18, 1906. [Google Scholar] [CrossRef]
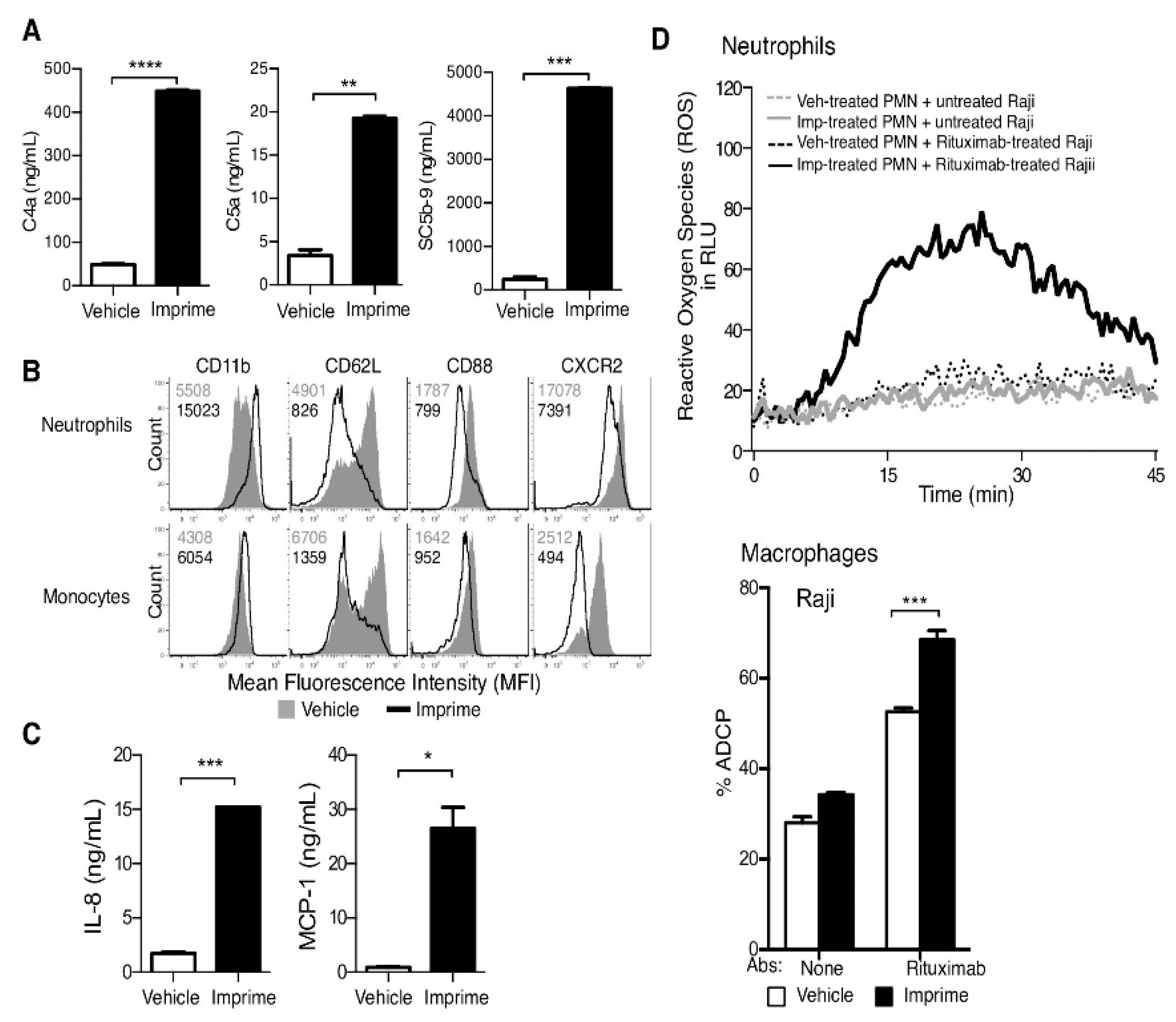
- Chan, A.S.; Jonas, A.B.; Qiu, X.; Ottoson, N.R.; Walsh, R.M.; Gorden, K.B.; Harrison, B.; Maimonis, P.J.; Leonardo, S.M.; Ertelt, K.E.; et al. Imprime PGG-mediated anti-cancer immune activation requires immune complex formation. PLoS ONE 2016, 11, e0165909. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Novak, M. Biology and Chemistry of Beta Glucan; Bentham Science: Sharjah, UAE, 2011; Volume 1. [Google Scholar]
- Li, X.; Chen, P.; Zhang, P.; Chang, Y.; Cui, M.; Duan, J. Protein-bound beta-glucan from Coriolus versicolor has potential for use against obesity. Mol. Nutr. Food Res. 2019, e1801231. [Google Scholar] [CrossRef]
- Vetvicka, V.; Gover, O.; Karpovsky, M.; Hayby, H.; Danay, O.; Ezov, N.; Hadar, Y.; Schwartz, B. Immune-modulating activities of glucans extracted from Pleurotus ostreatus and Pleurotus eryngii. J. Funct. Foods 2019, 54, 81–91. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vetvickova, J. Glucan supplementation ameliorates some health problems related to the development of Lyme disease. World J. Pathol. 2019, 8, 7–13. [Google Scholar]
- Berdal, M.; Appelbom, H.I.; Eikrem, J.H.; Lund, A.; Zykova, S.; Busund, L.T.; Seljelid, R.; Jenssen, T. Aminated beta-1,3-D-glucan improves wound healing in diabetic db/db mice. Wound Repair Regen. 2007, 15, 825–832. [Google Scholar] [CrossRef] [PubMed]
- Gulcelik, M.A.; Dincer, H.; Sahin, D.; Faruk Demir, O.; Yenidogan, E.; Alagol, H. Glucan improves impaired wound healing in diabetic rats. Wounds 2010, 22, 12–16. [Google Scholar] [PubMed]
- Takatsuki, F.; Namiki, R.; Kikuchi, T.; Suzuki, M.; Hamuro, J. Lentinan augments skin reaction induced by bradykinin: Its correlation with vascular dilatation and hemorrhage responses and antitumor activities. Int. J. Immunopharmacol. 1995, 17, 465–474. [Google Scholar] [CrossRef]
- Gaspar, L.R.; Camargo, F.B., Jr.; Gianeti, M.D.; Maia Campos, P.M. Evaluation of dermatological effects of cosmetic formulations containing Saccharomyces cerevisiae extract and vitamins. Food Chem. Toxicol. 2008, 46, 3493–3500. [Google Scholar] [CrossRef]
- Pillai, R.; Redmond, M.; Röding, J. Anti-wrinkle therapy: Significant new findings in the non-invasive cosmetic treatment of skin wrinkles with beta-glucan. Int. J. Cosmet. Sci. 2005, 27, 292. [Google Scholar] [CrossRef]
- O’Day, S.; Stopeck, A.; Huhn, R.; Gargano, M.; Prathikanti, R.; Ma, B.; Mattson, P.; Lowe, J.; Bose, N.; Ertelt, K.; et al. Abstract OT1-01-04: A phase 2, open-label study of imprime PGG (Imprime), a novel beta glucan, with pembrolizumab (Pembro) in chemotherapy-resistant metastatic triple negative breast cancer (TNBC). Cancer Res. 2018, 78. [Google Scholar] [CrossRef]
- Tamayo, M.E.; Cornelio, G.H.; Bautista, J.B.; Flores, M.L.; Kurman, M.R.; Paul, M.M.; Gargano, M.A.; Patchen, M.L. A phase Ib/2, dose-escalating, safety, and efficacy study of imprime PGG, cetuximab and irinotecan in patients with advanced colorectal cancer (CRC). J. Clin. Oncol. 2009, 27, e15062. [Google Scholar] [CrossRef]
- Wesa, K.M.; Cunningham-Rundles, S.; Klimek, V.M.; Vertosick, E.; Coleton, M.I.; Yeung, K.S.; Lin, H.; Nimer, S.; Cassileth, B.R. Maitake mushroom extract in myelodysplastic syndromes (MDS): A phase II study. Cancer Immunol. Immunother. 2015, 64, 237–247. [Google Scholar] [CrossRef]
- Aleem, E. β-Glucans and their applications in cancer therapy: Focus on human studies. Anticancer Agents Med. Chem. 2013, 13, 709–719. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vetvickova, J. Glucans and cancer: Comparison of commercially available β-glucans—Part IV. Anticancer Res. 2018, 38, 1327–1333. [Google Scholar] [CrossRef]
- Richter, J.; Kral, V.; Stiborova, I.; Rajnohova, D.; Vetvicka, V. Anti-inflammatory effects of β-glucan in cancer related fatigue. J. Nutr. Health Sci. 2015, 2, 304–310. [Google Scholar] [CrossRef]
- Richter, J.; Kral, V.; Svozil, V.; Rajnohova Dobiasova, L.; Pohorska, J.; Stiborova, I.; Vetvicka, V. Effect of Transfer Point Glucan #300 supplementation on children exposed to passive smoking: Placebo-driven double-blind trials. J. Nutr. Health 2014, 1, 105–111. [Google Scholar]
- Richter, J.; Svozil, V.; Kral, V.; Rajnohova Dobiasova, L.; Stiborova, I.; Vetvicka, V. Clinical trials of yeast-derived beta-(1,3) glucan in children: Effects on innate immunity. Ann. Transl. Med. 2014, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Fuller, R.; Moore, M.V.; Lewith, G.; Stuart, B.L.; Ormiston, R.V.; Fisk, H.L.; Noakes, P.S.; Calder, P.C. Yeast-derived beta-1,3/1,6 glucan, upper respiratory tract infection and innate immunity in older adults. Nutrition 2017, 39–40, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Richter, J.; Kral, V.; Vetvicka, V.; Lucie, R.D.; Fernandez-Botran, R. Effect of beta-glucan supplementation on levels of IgM, IgA, IgG and its subclasses IgG1, IgG2, IgG3, and IgG4 in cancer patients. J. Tumor 2016, 4, 469–473. [Google Scholar]
- Vetvicka, V.; Richter, J.; Kral, V.; Rajnohova Dobiasova, L.; Stiborova, I.; Pohorska, J. Regulation of hematopoiesis in cancer patients: Placebor driven, double-blind clinical trials of β-glucan. J. Tumor 2015, 3, 305–308. [Google Scholar]
- Pohorska, J.; Richter, J.; Kral, V.; Rajonohova Dobiasova, L.; Stiborova, I.; Vetvicka, V. Reconstruction of NK cells during complex cancer treatment. J. Tumor 2016, 4, 398–402. [Google Scholar] [CrossRef]
- Stier, H.; Ebbeskotte, V.; Gruenwald, J. Immune-modulatory effects of dietary yeast beta-1,3/1,6-D-glucan. Nutr. J. 2014, 13, 38. [Google Scholar] [CrossRef]
- Wang, Y.; Ames, N.P.; Tun, H.M.; Tosh, S.M.; Jones, P.J.; Khafipour, E. High molecular weight barley beta-glucan alters gut microbiota toward reduced cardiovascular disease risk. Front. Microbiol. 2016, 7, 129. [Google Scholar] [CrossRef] [PubMed]
- Richter, J.; Zavorkova, M.; Vetvicka, V.; Liehneova, I.; Kral, V.; Dobiasova, L.R. Effects of B-glucan and vitamin D supplementation on inflammatory parameters in patients with diabetic retinopathy. J. Diet. Suppl. 2018, 19, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Richter, J.; Zavorkova, M.; Vetvicka, V.; Vlastimil, K.; Stiborova, I.; Liehneova, I.; Pohorska, J.; Rajnohova, D.L. IgG4-related orbital disease in patients with diabetic retinopathy: Effects of glucan and vitamin D supplementation. Pathol. Discov. 2018, 6, 1–7. [Google Scholar]
- Markovina, N.; Banjari, I.; Bucevic Popovic, V.; Jelicic Kadic, A.; Puljak, L. Efficacy and safety of oral and inhalation commercial beta-glucan products: Systematic review of randomized controlled trials. Clin. Nutr. 2019. [Google Scholar] [CrossRef]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef]
- de Jesus Raposo, M.F.; de Morais, A.M.; de Morais, R.M. Emergent sources of prebiotics: Seaweeds and microalgae. Mar. Drugs 2016, 14, 27. [Google Scholar] [CrossRef]
- Malkki, Y.; Virtanen, E. Gastrointestinal effects of oat bran and oat gum—A review. Lebensm.-Wiss. Technol. 2001, 34, 337–347. [Google Scholar] [CrossRef]
- Ngamkala, S.; Futami, K.; Endo, M.; Maita, M.; Katagiri, T. Immunological effects of glucan and Lactobacillus rhamnosus GG, a probiotic bacterium, on Nile tilapia Oreochromis niloticus intestine with oral Aeromonas challenges. Fish. Sci. 2010, 76, 833–840. [Google Scholar] [CrossRef]
- Rosburg, V.; Boylston, T.; White, P. Viability of bifidobacteria strains in yogurt with added oat beta-glucan and corn starch during cold storage. J. Food Sci. 2010, 75, C439–C444. [Google Scholar] [CrossRef]
- Szymańska-Czerwińska, M.; Bednarek, D. Effect of tylosin and prebiotics on the selected humoral immunological parameters in calves. Medycyna Wet. 2011, 67, 275–278. [Google Scholar]
- Nakashima, A.; Yamada, K.; Iwata, O.; Sugimoto, R.; Atsuji, K.; Ogawa, T.; Ishibashi-Ohgo, N.; Suzuki, K. β-Glucan in foods and its physiological functions. J. Nutr. Sci. Vitaminol. (Tokyo) 2018, 64, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Kendall, C.W.; Axelsen, M.; Augustin, L.S.; Vuksan, V. Viscous and nonviscous fibres, nonabsorbable and low glycaemic index carbohydrates, blood lipids and coronary heart disease. Curr. Opin. Lipidol. 2000, 11, 49–56. [Google Scholar] [CrossRef]
- Fadel, J.G.; Newman, R.K.; Newman, C.W.; Barnes, A.E. Hypocholesterolemic effects of beta-glucans in different barley diets fed to broiler chicks. Nutr. Rep. Int. 1987, 35, 1049–1058. [Google Scholar]
- Lim, M.K.; Ku, S.K.; Choi, J.S.; Kim, J.W. Effect of polycan, a beta-glucan originating from Aureobasidium, on a high-fat diet-induced hyperlipemic hamster model. Exp. Ther. Med. 2015, 9, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Kusmiati; Dhewantara, F.X. Cholesterol-lowering effect of beta glucan extracted from Saccharomyces cerevisiae in rats. Sci. Pharm. 2016, 84, 153–165. [Google Scholar] [CrossRef]
- Anderson, J.W.; Gustafson, N.J. Hypocholesterolemic effects of oat and bean products. Am. J. Clin. Nutr. 1988, 48, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Story, L.; Sieling, B.; Chen, W.J.; Petro, M.S.; Story, J. Hypocholesterolemic effects of oat-bran or bean intake for hypercholesterolemic men. Am. J. Clin. Nutr. 1984, 40, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Huang, X.F. The effects of diets enriched in beta-glucans on blood lipoprotein concentrations. J. Clin. Lipidol. 2009, 3, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Opizzi, A.; Monteferrario, F.; Klersy, C.; Cazzola, R.; Cestaro, B. Beta-glucan- or rice bran-enriched foods: A comparative crossover clinical trial on lipidic pattern in mildly hypercholesterolemic men. Eur. J. Clin. Nutr. 2011, 65, 864–871. [Google Scholar] [CrossRef]
- Sima, P.; Vannucci, L.; Vetvicka, V. β-glucans and cholesterol (Review). Int. J. Mol. Med. 2018, 41, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Vetvickova, J. A comparison of injected and orally administered beta glucans. JANA 2008, 11, 42–49. [Google Scholar]
- Hashimoto, K.; Suzuki, I.; Yadomae, T. Oral administration of SSG, a beta-glucan obtained from Sclerotinia sclerotiorum, affects the function of Peyer’s patch cells. Int. J. Immunopharmacol. 1991, 13, 437–442. [Google Scholar] [CrossRef]
- Rice, P.J.; Adams, E.L.; Ozment-Skelton, T.; Gonzalez, A.J.; Goldman, M.P.; Lockhart, B.E.; Barker, L.A.; Breuel, K.F.; Deponti, W.K.; Kalbfleisch, J.H.; et al. Oral delivery and gastrointestinal absorption of soluble glucans stimulate increased resistance to infectious challenge. J. Pharmacol. Exp. Ther. 2005, 314, 1079–1086. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Kim, J.A.; Huang, A.Y. Optimizing Tumor microenvironment for cancer immunotherapy: Beta-glucan-based nanoparticles. Front. Immunol. 2018, 9, 341. [Google Scholar] [CrossRef] [PubMed]
- Ostroff, G.R.; Easson, D.D.; Jamas, S. A new β-glucan-based macrophage-targeted adjuvant. In Polymeric Drugs and Drug Delivery Systems; American Chemical Society: Washington, DC, USA, 1991; Volume 469, pp. 52–59. [Google Scholar]
- Fujiwara, N.; Izumi, H.; Morimoto, Y.; Sakurai, K.; Mochizuki, S. Complex consisting of antisense DNA and beta-glucan promotes internalization into cell through Dectin-1 and hybridizes with target mRNA in cytosol. Cancer Gene Ther. 2019, 26, 32–40. [Google Scholar] [CrossRef]
- Jones, D.H.; Corris, S.; McDonald, S.; Clegg, J.C.; Farrar, G.H. Poly(DL-lactide-co-glycolide)-encapsulated plasmid DNA elicits systemic and mucosal antibody responses to encoded protein after oral administration. Vaccine 1997, 15, 814–817. [Google Scholar] [CrossRef]
- Aouadi, M.; Tesz, G.J.; Nicoloro, S.M.; Wang, M.; Chouinard, M.; Soto, E.; Ostroff, G.R.; Czech, M.P. Orally delivered siRNA targeting macrophage Map4k4 suppresses systemic inflammation. Nature 2009, 458, 1180–1184. [Google Scholar] [CrossRef]
- De Smet, R.; Demoor, T.; Verschuere, S.; Dullaers, M.; Ostroff, G.R.; Leclercq, G.; Allais, L.; Pilette, C.; Dierendonck, M.; De Geest, B.G.; et al. β-Glucan microparticles are good candidates for mucosal antigen delivery in oral vaccination. J. Control. Release 2013, 172, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Specht, C.A.; Lee, C.K.; Huang, H.; Tipper, D.J.; Shen, Z.T.; Lodge, J.K.; Leszyk, J.; Ostroff, G.R.; Levitz, S.M. Protection against experimental cryptococcosis following vaccination with glucan particles containing cryptococcus alkaline extracts. MBio 2015, 6, e01905. [Google Scholar] [CrossRef]
- Soares, E.; Jesus, S.; Borges, O. Oral hepatitis B vaccine: Chitosan or glucan based delivery systems for efficient HBsAg immunization following subcutaneous priming. Int. J. Pharm. 2018, 535, 261–271. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, L.; Yang, R.; Fei, C.; Wang, X.; Zhang, K.; Wang, C.; Zheng, W.; Xue, F. Improvement of immune responses to influenza vaccine (H5N1) by sulfated yeast beta-glucan. Int. J. Biol. Macromol. 2016, 93, 203–207. [Google Scholar] [CrossRef]
- Sima, P.; Vetvicka, V. β-Glucan in Allergies. Am. J. Immunol. 2017, 13, 73–80. [Google Scholar]
- Kofuji, K.; Aoki, A.; Tsubaki, K.; Konishi, M.; Isobe, T.; Murata, Y. Antioxidant activity of beta-glucan. ISRN Pharm. 2012, 2012, 125864. [Google Scholar] [CrossRef]
- Yamada, J.; Hamuro, J.; Hatanaka, H.; Hamabata, K.; Kinoshita, S. Alleviation of seasonal allergic symptoms with superfine beta-1,3-glucan: A randomized study. J. Allergy Clin. Immunol. 2007, 119, 1119–1126. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, S.; Hirose, K.; Iwata, A.; Takahashi, K.; Ohkubo, A.; Tamachi, T.; Ikeda, K.; Kagami, S.; Nakajima, H. beta-glucan curdlan induces IL-10-producing CD4+ T cells and inhibits allergic airway inflammation. J. Immunol. 2012, 189, 5713–5721. [Google Scholar] [CrossRef]
- Jesenak, M.; Hrubisko, M.; Majtan, J.; Rennerova, Z.; Banovcin, P. Anti-allergic effect of Pleuran (beta-glucan from Pleurotus ostreatus) in children with recurrent respiratory tract infections. Phytother. Res. 2014, 28, 471–474. [Google Scholar] [CrossRef]
- Miyamoto, K.; Watanabe, Y.; Iizuka, N.; Sakaguchi, E.; Okita, K. Effect of a hot water extract of Agaricus blazei fruiting bodies (CJ-01) on the intracellular cytokines level in a patient with bronchitis. J. Trad Med. 2002, 19, 142–147. [Google Scholar]
- Talbott, S.M.; Talbott, J.A.; Talbott, T.L.; Dingler, E. beta-Glucan supplementation, allergy symptoms, and quality of life in self-described ragweed allergy sufferers. Food Sci. Nutr. 2013, 1, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, E.; Shoji, S.; Nishihara, M.; Shimoda, T.; Nishima, S. Contribution of lung fibroblast migration in the fibrotic process of airway remodeling in asthma. Allergol. Int. 2008, 57, 73–78. [Google Scholar] [CrossRef]
- Fuller, R.; Butt, H.; Noakes, P.S.; Kenyon, J.; Yam, T.S.; Calder, P.C. Influence of yeast-derived 1,3/1,6 glucopolysaccharide on circulating cytokines and chemokines with respect to upper respiratory tract infections. Nutrition 2012, 28, 665–669. [Google Scholar] [CrossRef] [PubMed]
- Jesenak, M.; Banovcin, P.; Rennerova, Z.; Majtan, J. β-Glucans in the treatment and prevention of allergic diseases. Allergol. Immunopathol. (Madr.) 2014, 42, 149–156. [Google Scholar] [CrossRef]
- Wichers, H. Immunomodulation by food: Promising concept for mitigating allergic disease? Anal. Bioanal. Chem. 2009, 395, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Sarinho, E.; Medeiros, D.; Schor, D.; Rego Silva, A.; Sales, V.; Motta, M.E.; Costa, A.; Azoubel, A.; Rizzo, J.A. Production of interleukin-10 in asthmatic children after beta-1-3-glucan. Allergol. Immunopathol. (Madr.) 2009, 37, 188–192. [Google Scholar] [CrossRef]
- Miraglia Del Giudice, M.; Maiello, N.; Capristo, C.; Alterio, E.; Capasso, M.; Perrone, L.; Ciprandi, G. Resveratrol plus carboxymethyl-beta-glucan reduces nasal symptoms in children with pollen-induced allergic rhinitis. Curr. Med. Res. Opin. 2014, 30, 1931–1935. [Google Scholar] [CrossRef] [PubMed]
- Miraglia Del Giudice, M.; Maiello, N.; Decimo, F.; Capasso, M.; Campana, G.; Leonardi, S.; Ciprandi, G. Resveratrol plus carboxymethyl-beta-glucan may affect respiratory infections in children with allergic rhinitis. Pediatr. Allergy Immunol. 2014, 25, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Jin, X.; Liu, B.; Zhuang, W.; Scalabrin, D. Follow-up formula consumption in 3- to 4-year-olds and respiratory infections: An RCT. Pediatrics 2014, 133, e1533–e1540. [Google Scholar] [CrossRef]
- Bobovcak, M.; Kuniakova, R.; Gabriz, J.; Majtan, J. Effect of Pleuran (beta-glucan from Pleurotus ostreatus) supplementation on cellular immune response after intensive exercise in elite athletes. Appl. Physiol. Nutr. Metab. 2010, 35, 755–762. [Google Scholar] [CrossRef]
- Majtan, J. Pleuran (beta-glucan from Pleurotus ostreatus): An effective nutritional supplement against upper respiratory tract infections? Med. Sport Sci. 2012, 59, 57–61. [Google Scholar] [CrossRef]
- Murphy, E.A.; Davis, J.M.; Brown, A.S.; Carmichael, M.D.; Carson, J.A.; Van Rooijen, N.; Ghaffar, A.; Mayer, E.P. Benefits of oat beta-glucan on respiratory infection following exercise stress: Role of lung macrophages. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1593–R1599. [Google Scholar] [CrossRef]
- Davis, J.M.; Murphy, E.A.; Brown, A.S.; Carmichael, M.D.; Ghaffar, A.; Mayer, E.P. Effects of oat beta-glucan on innate immunity and infection after exercise stress. Med. Sci. Sports Exerc. 2004, 36, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
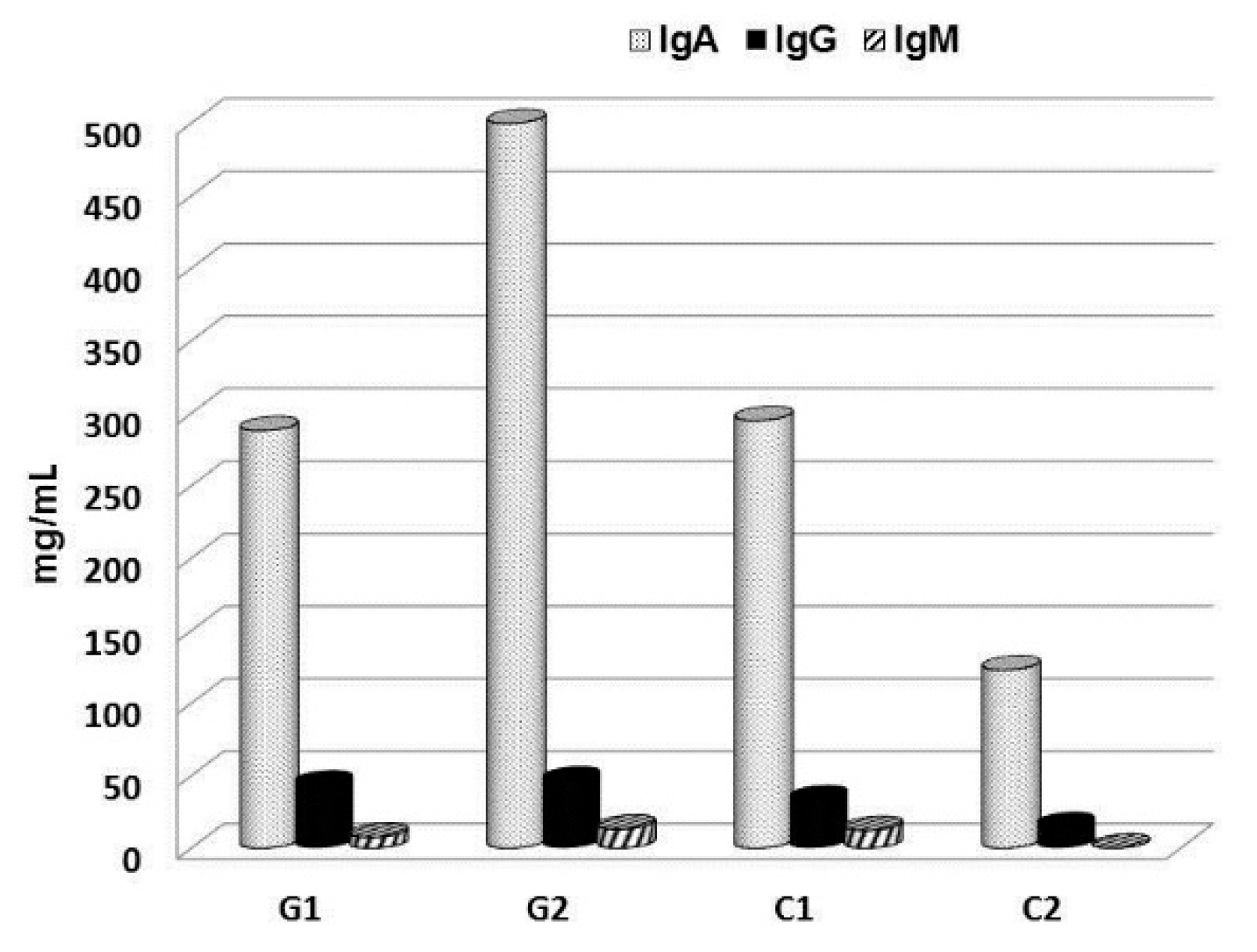
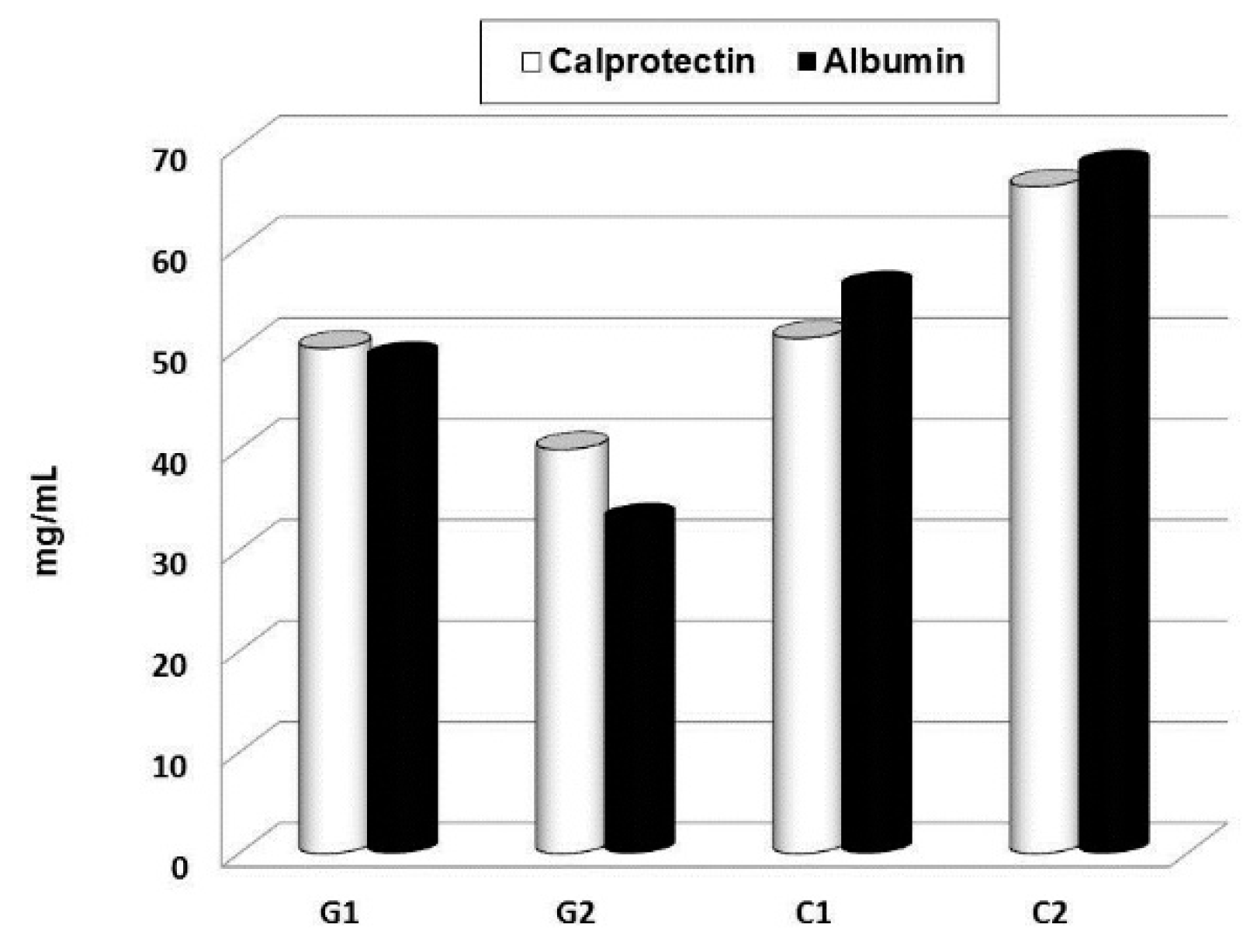
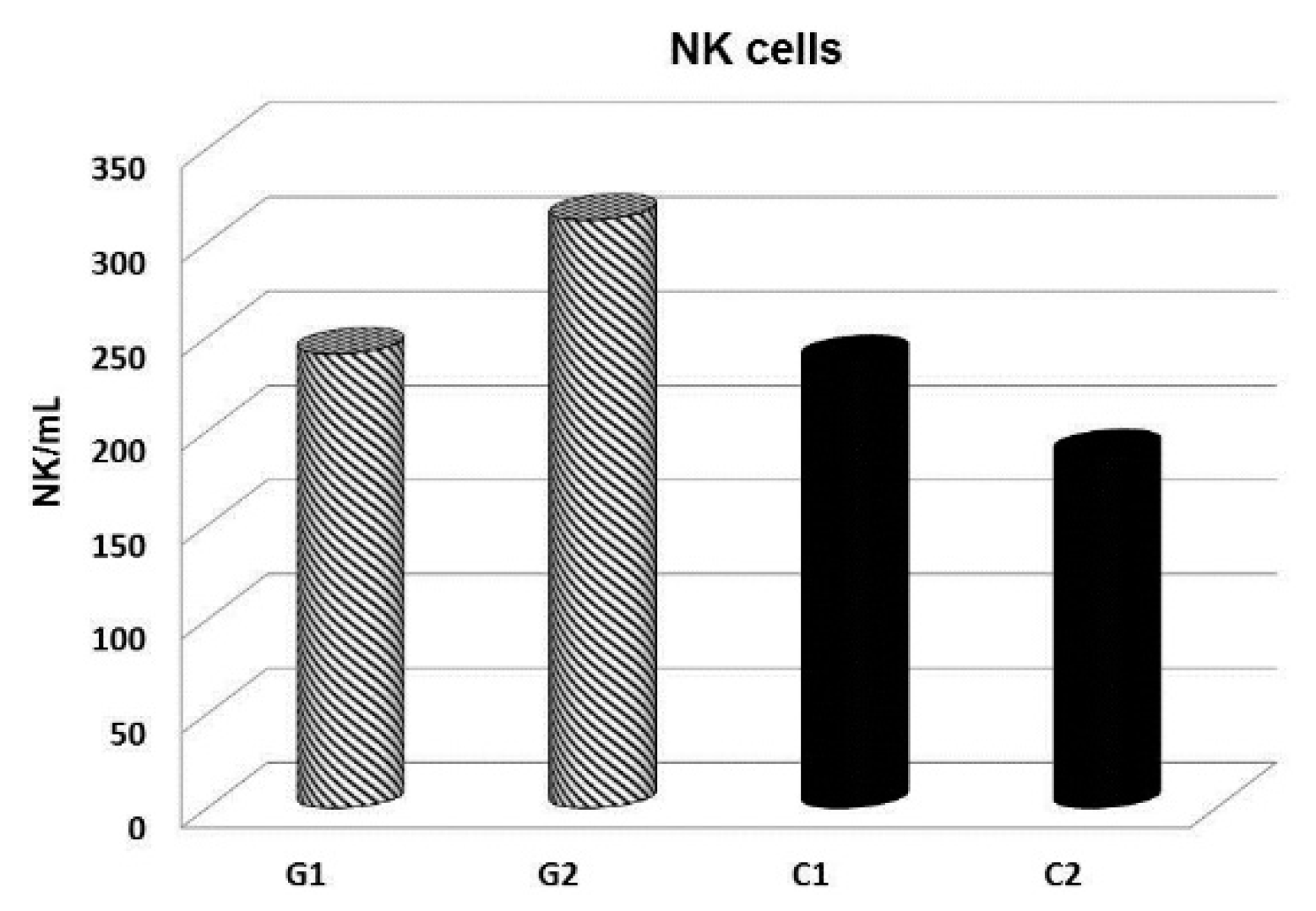
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vetvicka, V.; Vannucci, L.; Sima, P.; Richter, J. Beta Glucan: Supplement or Drug? From Laboratory to Clinical Trials. Molecules 2019, 24, 1251. https://doi.org/10.3390/molecules24071251
Vetvicka V, Vannucci L, Sima P, Richter J. Beta Glucan: Supplement or Drug? From Laboratory to Clinical Trials. Molecules. 2019; 24(7):1251. https://doi.org/10.3390/molecules24071251
Chicago/Turabian StyleVetvicka, Vaclav, Luca Vannucci, Petr Sima, and Josef Richter. 2019. "Beta Glucan: Supplement or Drug? From Laboratory to Clinical Trials" Molecules 24, no. 7: 1251. https://doi.org/10.3390/molecules24071251
APA StyleVetvicka, V., Vannucci, L., Sima, P., & Richter, J. (2019). Beta Glucan: Supplement or Drug? From Laboratory to Clinical Trials. Molecules, 24(7), 1251. https://doi.org/10.3390/molecules24071251






