Phenolic Acids in Jerusalem Artichoke (Helianthus tuberosus L.): Plant Organ Dependent Antioxidant Activity and Optimized Extraction from Leaves
Abstract
:1. Introduction
2. Results
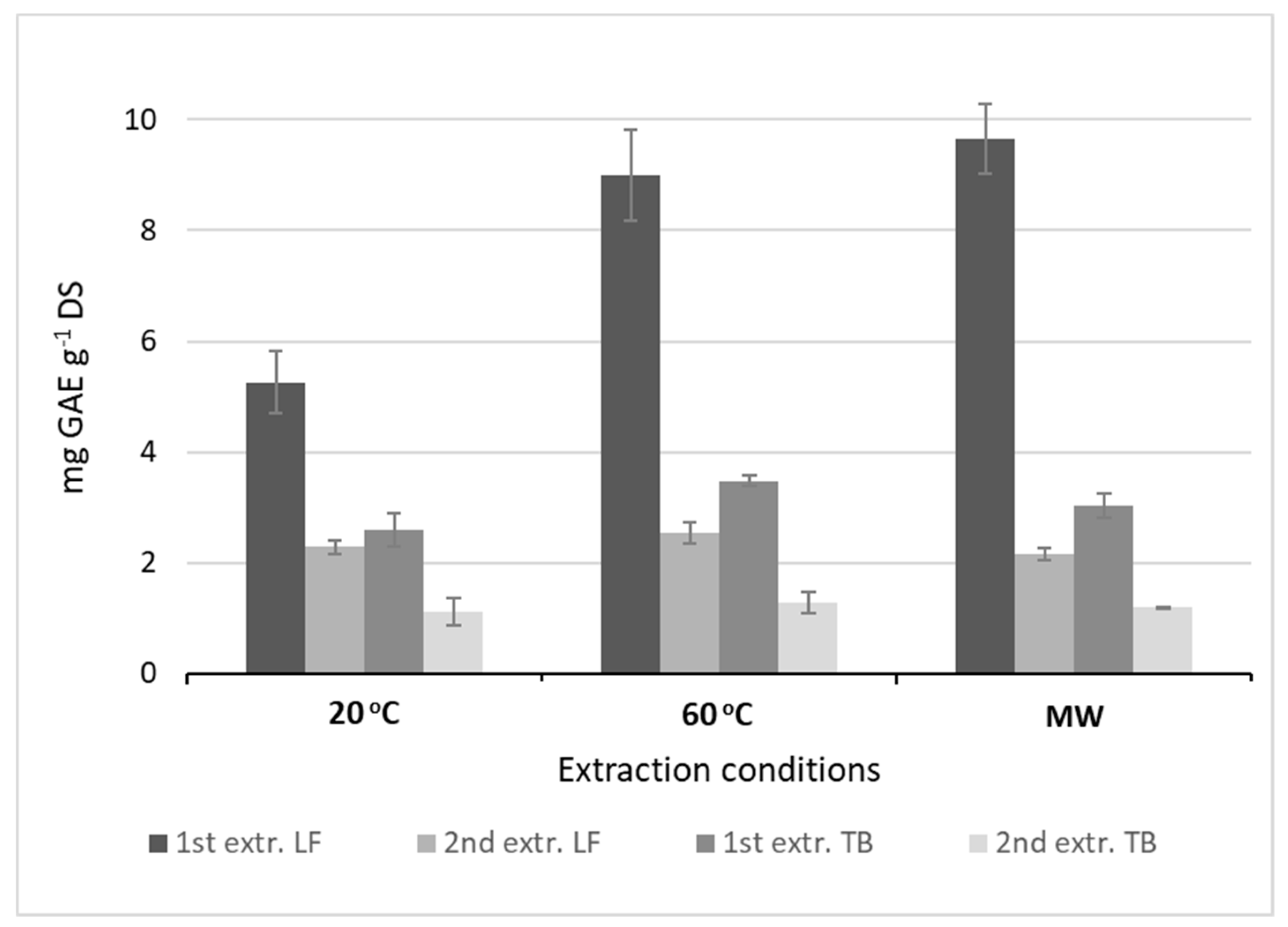
2.1. Phenolic Acids Extracted under Different Conditions
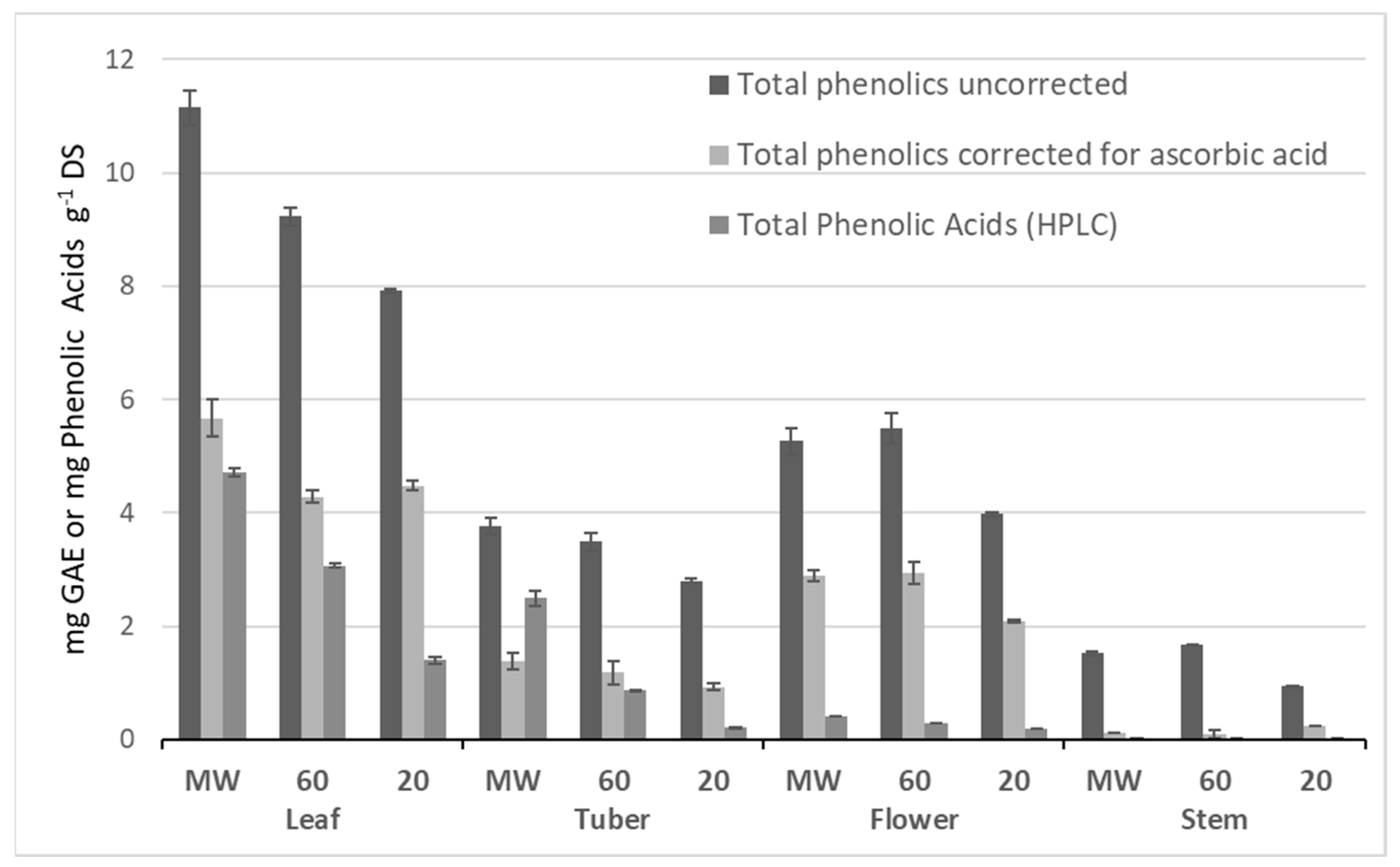
2.2. Distribution of Total Phenolic Content in the Whole Plant
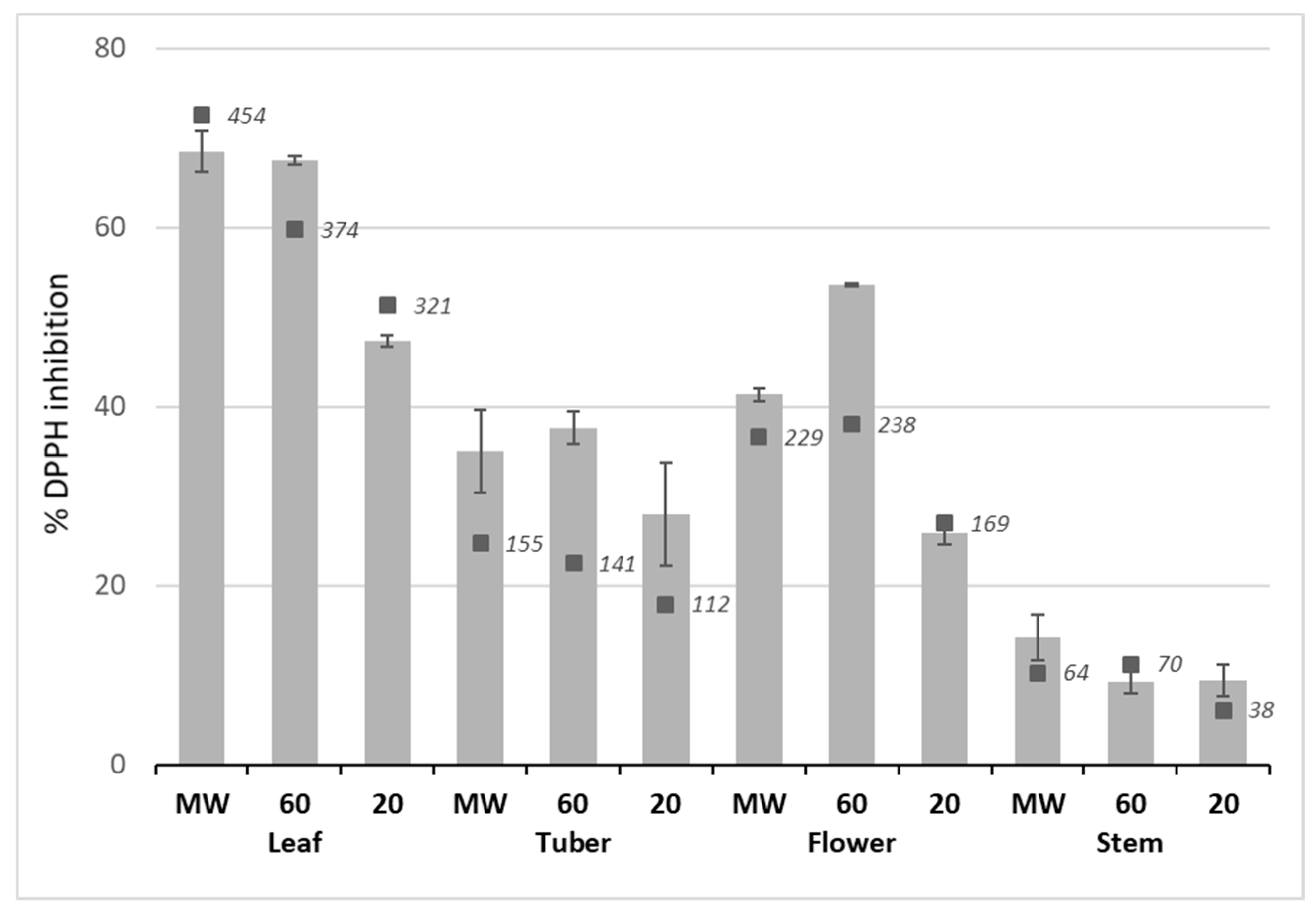
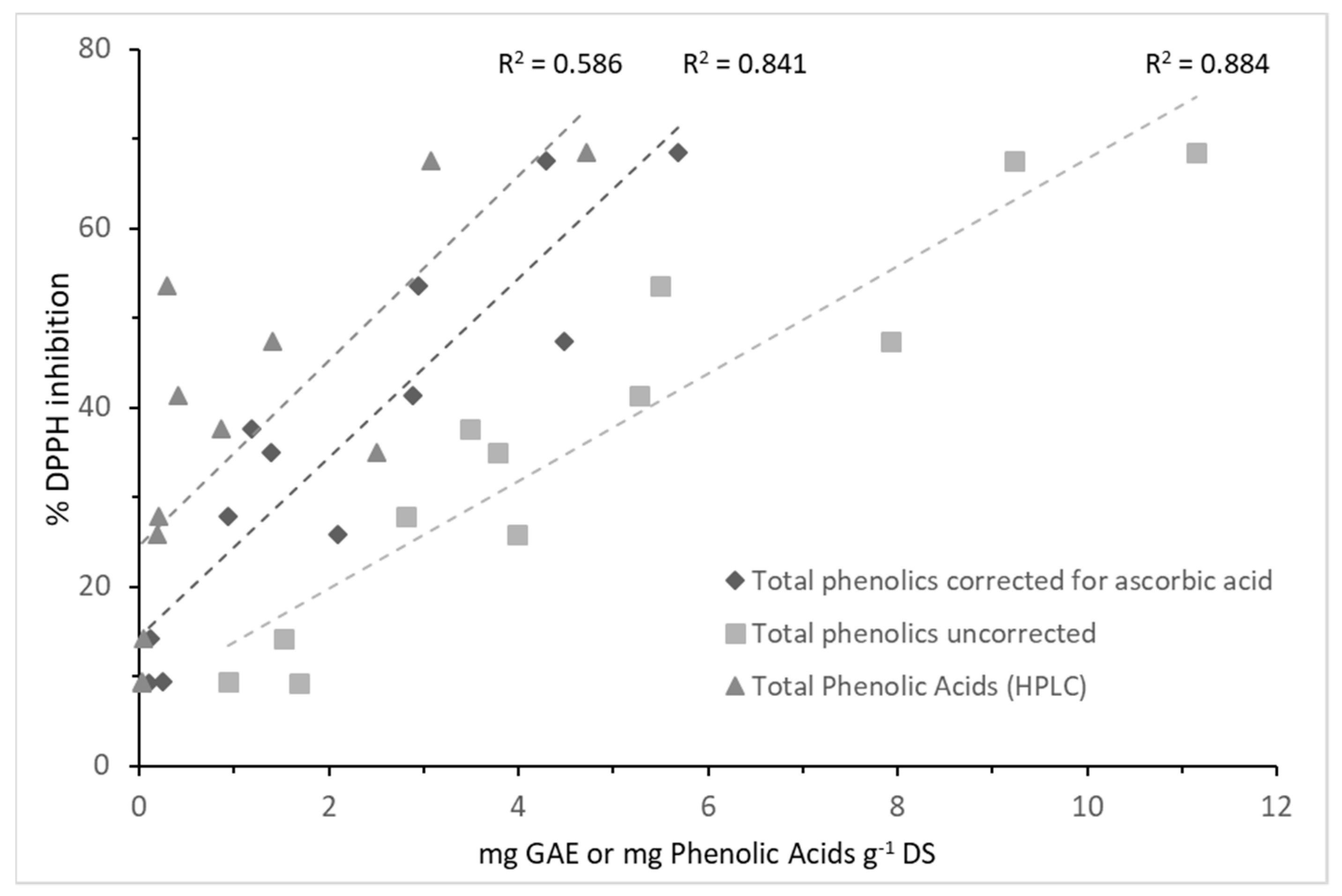
2.3. Antioxidant Activity
2.4. Identification and Quantitation of Major Phenolic Acids
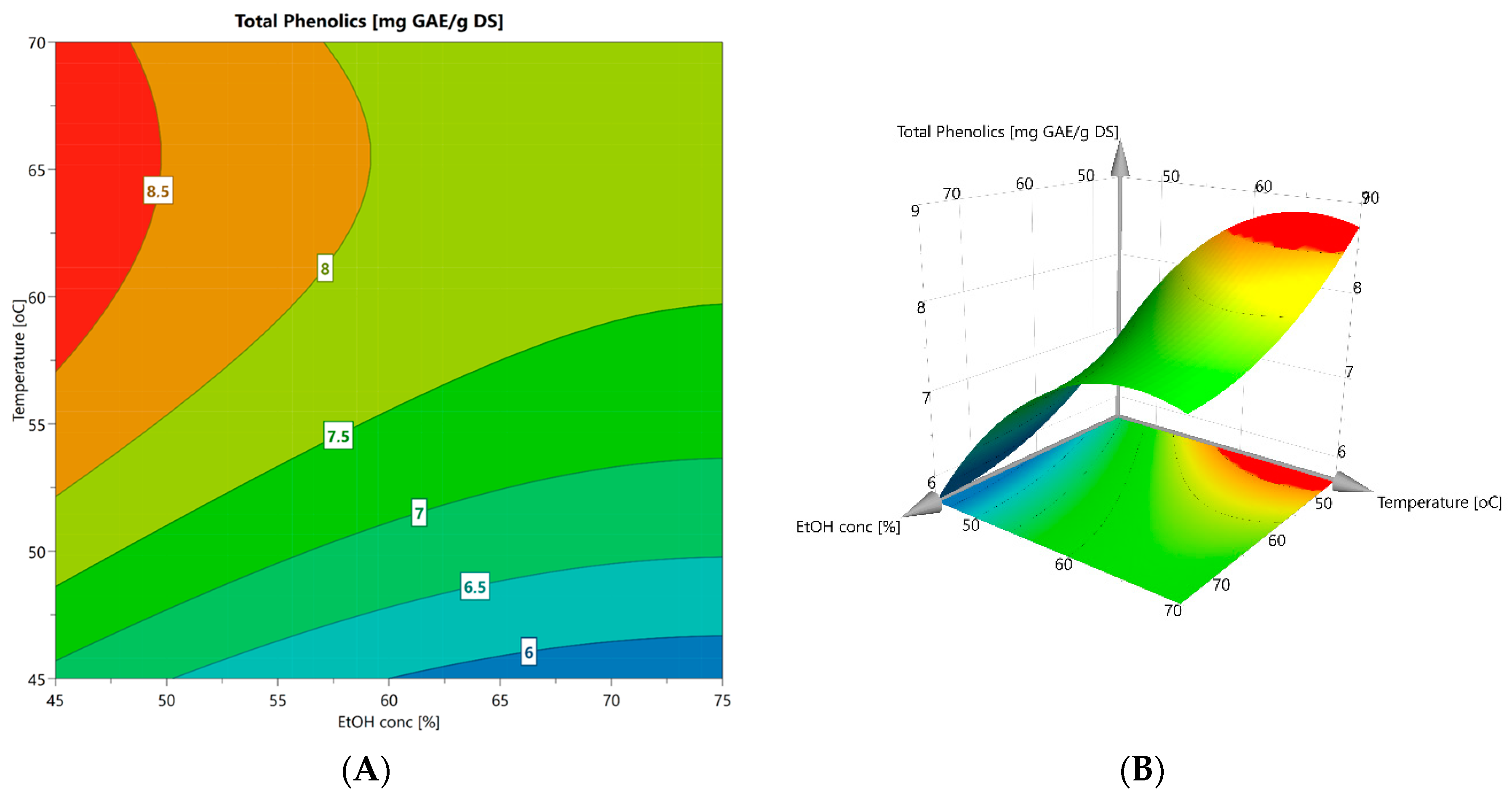
2.5. Factorial Design of Extraction Conditions
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Material
4.3. Pre-Treatment and Extraction
4.3.1. Microwave-Assisted Extraction
4.3.2. Heated Stirring Extraction
4.3.3. Room Temperature Stirring Extraction
4.3.4. Post-Extraction Treatments
4.4. Corrected Total Phenolic Content
4.5. Antioxidant Activity
4.6. Identification and Quantification of Phenolic Acids
4.7. Statistical Analysis
4.8. Experimental Factorial Design in Extraction Conditions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tchoné, M.; Bärwald, G.; Annemüller, G.; Fleischer, L.-G. Separation and identification of phenolic compounds in Jerusalem artichoke (Helianthus tuberosus L.). Sci. Aliment. 2006, 26, 394–408. [Google Scholar] [CrossRef]
- Jaiswal, R.; Deshpande, S.; Kuhnert, N. Profiling the chlorogenic acids of Rudbeckia hirta, Helianthus tuberosus, Carlina acaulis and Symphyotrichum novae-angliae leaves by LC-MSn. Phytochem. Anal. 2011, 22, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Long, X.; Liu, Z.; Shao, H.; Liu, L. Analysis of phenolic acids of Jerusalem artichoke (Helianthus tuberosus L.) responding to salt-stress by liquid chromatography/tandem mass spectrometry. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Kapusta, I.; Krok, E.; Jamro, D.; Cebulak, T.; Kaszuba, J.; Salach, R. Identification and quantification of phenolic compounds from Jerusalem artichoke (Helianthus tuberosus L.) Tubers. J. Food Agric. Environ. 2013, 11, 601–606. [Google Scholar]
- Yuan, X.; Gao, M.; Xiao, H.; Tan, C.; Du, Y. Free radical scavenging activities and bioactive substances of Jerusalem artichoke (Helianthus tuberosus L.) leaves. Food Chem. 2012, 133, 10–14. [Google Scholar] [CrossRef]
- Chen, F.; Long, X.; Yu, M.; Liu, Z.; Liu, L.; Shao, H. Phenolics and antifungal activities analysis in industrial crop Jerusalem artichoke (Helianthus tuberosus L.) leaves. Ind. Crop. Prod. 2013, 47, 339–345. [Google Scholar] [CrossRef]
- Petrova, I.; Petkova, N.; Ivanov, I. Five edible flowers—Valuable source of antioxidants in human nutrition. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 604–610. [Google Scholar]
- Petkova, N.; Ivanov, I.; Denev, P.; Pavlov, A. Bioactive substance and free radical scavenging activities of flour from Jerusalem artichoke (Helianthus tuberosus L.) Tubers—A comparative study. Türk Tarım Doğa Bilimleri 2014, 7, 1773–1778. [Google Scholar]
- Sun, P.-C.; Liu, Y.; Yi, Y.-T.; Li, H.-J.; Fan, P.; Xia, C.-H. Preliminary enrichment and separation of chlorogenic acid from Helianthus tuberosus L. leaves extract by macroporous resins. Food Chem. 2015, 168, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Sinden, M.R.; Kennedy, A.H.; Chai, H.; Watson, L.E.; Graham, T.L.; Kinghorn, A.D. Bioactive constituents of Helianthus tuberosus (Jerusalem artichoke). Phytochem. Lett. 2009, 2, 15–18. [Google Scholar] [CrossRef]
- Lim, T. Helianthus tuberosus. In Edible Medicinal and Non Medicinal Plants; Springer: Dordrecht, The Netherlands, 2015; pp. 687–716. [Google Scholar]
- Yang, L.; He, Q.S.; Corscadden, K.; Udenigwe, C.C. The prospects of Jerusalem artichoke in functional food ingredients and bioenergy production. Biotechnol. Rep. 2015, 5, 77–88. [Google Scholar] [CrossRef]
- Zhang, Q.; Kim, H.-Y. Antioxidant, anti-inflammatory and cytotoxicityon human lung epithelial A549 cells of Jerusalem artichoke (Helianthus tuberosus L.) Tuber. Korean J. Plant Res 2015, 28, 305–311. [Google Scholar] [CrossRef]
- Johansson, E.; Prade, T.; Angelidaki, I.; Svensson, S.-E.; Newson, W.R.; Gunnarsson, I.B.; Hovmalm, H.P. Economically viable components from Jerusalem artichoke (Helianthus tuberosus L.) in a biorefinery concept. Int. J. Mol. Sci. 2015, 16, 8997–9016. [Google Scholar] [CrossRef] [PubMed]
- Slimestad, R.; Seljaasen, R.; Meijer, K.; Skar, S.L. Norwegian-grown Jerusalem artichoke (Helianthus tuberosus L.): Morphology and content of sugars and fructo-oligosaccharides in stems and tubers. J. Sci. Food Agric. 2010, 90, 956–964. [Google Scholar] [CrossRef] [PubMed]
- Bach, V.; Clausen, M.R.; Edelenbos, M. Production of Jerusalem artichoke (Helianthus tuberosus L.) and impact on inulin and phenolic compounds. In Processing and Impact on Active Components in Food; Elsevier: Amsterdam, The Netherlands, 2015; pp. 97–102. [Google Scholar]
- Isabelle, M.; Lee, B.L.; Lim, M.T.; Koh, W.-P.; Huang, D.; Ong, C.N. Antioxidant activity and profiles of common vegetables in Singapore. Food Chem. 2010, 120, 993–1003. [Google Scholar] [CrossRef]
- Sánchez-Rangel, J.C.; Benavides, J.; Heredia, J.B.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. The Folin–Ciocalteu assay revisited: Improvement of its specificity for total phenolic content determination. Anal. Methods 2013, 5, 5990–5999. [Google Scholar] [CrossRef]
- Gunnarsson, I.B.; Svensson, S.E.; Johansson, E.; Karakashev, D.; Angelidaki, I. Potential of Jerusalem artichoke (Helianthus tuberosus L.) as a biorefinery crop. Ind. Crops Prod. 2014, 56, 231–240. [Google Scholar] [CrossRef]
- Long, X.-H.; Shao, H.-B.; Liu, L.; Liu, L.-P.; Liu, Z.-P. Jerusalem artichoke: A sustainable biomass feedstock for biorefinery. Renew. Sustain. Energy Rev. 2016, 54, 1382–1388. [Google Scholar] [CrossRef]
- Siatka, T.; Kašparová, M. Seasonal variation in total phenolic and flavonoid contents and DPPH scavenging activity of Bellis perennis L. flowers. Molecules 2010, 15, 9450–9461. [Google Scholar] [CrossRef]
- Prat, D.; Wells, A.; Hayler, J.; Sneddon, H.; McElroy, C.R.; Abou-Shehada, S.; Dunn, P.J. CHEM21 selection guide of classical-and less classical-solvents. Green Chem. 2015, 18, 288–296. [Google Scholar] [CrossRef]
- Sepahpour, S.; Selamat, J.; Abdul Manap, M.Y.; Khatib, A.; Abdull Razis, A.F. Comparative analysis of chemical composition, antioxidant activity and quantitative characterization of some phenolic compounds in selected herbs and spices in different solvent extraction systems. Molecules 2018, 23, 402. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-W.; Kim, J.-K.; Song, I.-S.; Kwon, E.-S.; Youn, K.-S. Comparison of antioxidant and physiological properties of Jerusalem artichoke leaves with different extraction processes. J. Korean Soc. Food Sci. Nutr. 2013, 42, 68–75. [Google Scholar] [CrossRef]
- Yuan, X.; Cheng, M.; Gao, M.; Zhuo, R.; Zhang, L.; Xiao, H. Cytotoxic constituents from the leaves of Jerusalem artichoke (Helianthus tuberosus L.) and their structure–activity relationships. Phytochem. Lett. 2013, 6, 21–25. [Google Scholar] [CrossRef]
- Sharma, O.P.; Bhat, T.K. DPPH antioxidant assay revisited. Food Chem. 2009, 113, 1202–1205. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds dried leaf, flower, tuber and stem of Jerusalem artichoke are available from the authors. |
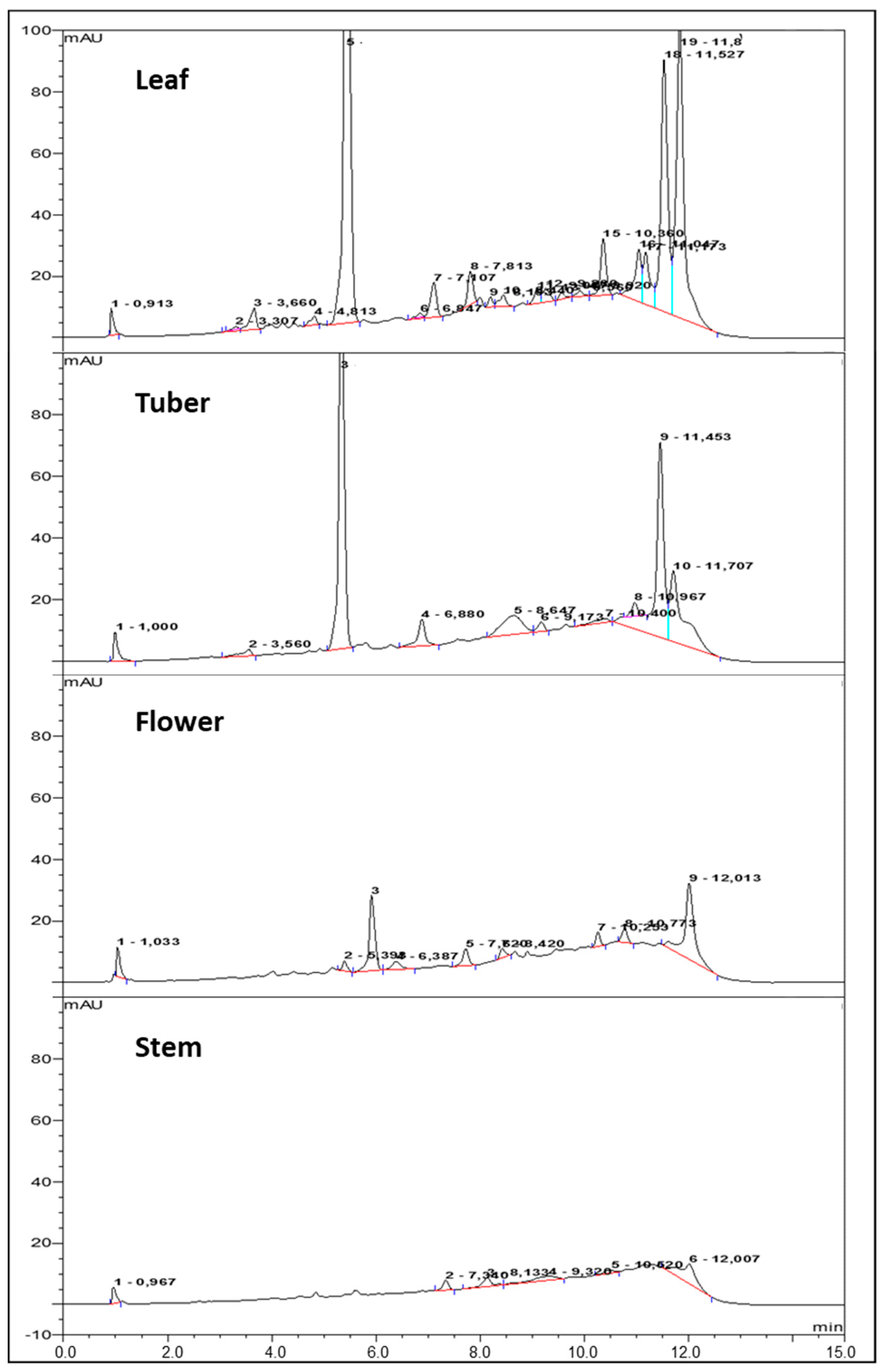
| Sample | Condition | CQA | PQA | CA | 1,5 diCQA | 3,5 diCQA | Sum | % of TPC corr. |
|---|---|---|---|---|---|---|---|---|
| Leaves | MW 60 20 | 2.23 2.52 0.91 | 0.02 0.02 0.10 | 0.00 0.00 0.00 | 1.07 0.19 0.38 | 1.39 0.34 0.01 | 4.71 3.07 1.40 | 82.7 71.5 31.2 |
| Tuber | MW 60 20 | 1.48 0.65 0.13 | 0.00 0.00 0.00 | 0.07 0.08 0.08 | 0.13 0.14 0.00 | 0.82 0.00 0.00 | 2.50 0.87 0.21 | 178.6 72.3 23.1 |
| Flower | MW 60 20 | 0.14 0.20 0.04 | 0.03 0.03 0.03 | 0.06 0.06 0.07 | 0.17 0.00 0.01 | 0.01 0.00 0.04 | 0.41 0.29 0.19 | 14.1 10.0 8.9 |
| Stem | MW 60 20 | 0.01 0.01 0.00 | 0.00 0.01 0.02 | 0.00 0.00 0.00 | 0.01 0.00 0.00 | 0.02 0.00 0.00 | 0.04 0.02 0.02 | 36.1 19.0 10.5 |
| Model Term | Coefficient (β) | p-Value |
|---|---|---|
| Intercept (βo) | 6.798 | <0.001 |
| Ethanol conc. (X1) | −0.589 | <0.001 |
| Temperature (X2) | 0.937 | <0.001 |
| Holding time (X3) | −0.567 | 0.001 |
| X1X1 | 0.281 | 0.250 |
| X2X2 | −0.730 | 0.004 |
| X3X3 | 0.301 | 0.219 |
| Source of Variation | Sum Quadratic | DF | Mean Quadratic (Variance) | F-Value | p | SD |
|---|---|---|---|---|---|---|
| Total corrected | 43.21 | 31 | 1.39 | 1.18 | ||
| Regression | 35.20 | 6 | 5.87 | 18.29 | <0.001 | 2.42 |
| Residual | 8.02 | 25 | 0.321 | 0.566 | ||
| Lack of fit (model error) | 0.894 | 6 | 0.149 | 0.397 | 0.872 | 0.386 |
| Pure error (replicate error) | 7.12 | 19 | 0.375 | 0.612 | ||
| R2 | 0.814 | |||||
| R2 adjusted | 0.770 |
| Response | Predicted from Model (Equation (1)) | Model-set Conditions (n = 3) | Standard Conditions (n = 2) |
|---|---|---|---|
| TPC uncorr. (±SD) | 8.28 ± 0.6 | 9.42 ± 0.06 | 9.36 ± 0.06 |
| TPC corrected (±SD) | n.d. | 6.08 ± 0.06 | 6.00 ± 0.04 |
| Variable | Factor | Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| Ethanol strength (%) | X1 | 45 | 60 | 75 |
| Temperature (°C) | X2 | 45 | 58 | 70 * |
| Holding time (min) | X3 | 60 | 120 | 180 |
| Run | Factor | Total Phenolics, Uncorrected | ||||
|---|---|---|---|---|---|---|
| X1 | X2 | X3 | mg GAE g−1 DS ± SD (n = 2) | |||
| 1 | − | − | − | 6.54 | ± | 0.72 |
| 2 | + | − | − | 5.76 | ± | 0.07 |
| 3 | − | + | − | 9.04 | ± | 0.52 |
| 4 | + | + | − | 5.26 | ± | 0.59 |
| 5 | − | − | + | 7.82 | ± | 0.38 |
| 6 | + | − | + | 4.84 | ± | 0.16 |
| 7 | − | + | + | 7.64 | ± | 0.61 |
| 8 | + | + | + | 6.27 | ± | 0.03 |
| 9 | − | 0 | 0 | 7.66 | ± | 0.16 |
| 10 | + | 0 | 0 | 6.30 | ± | 0.25 |
| 11 | 0 | − | 0 | 5.12 | ± | 0.62 |
| 12 | 0 | + | 0 | 7.68 | ± | 0.98 |
| 13 | 0 | 0 | − | 7.63 | ± | 0.03 |
| 14 | 0 | 0 | + | 6.39 | ± | 1,10 |
| 15 | 0 | 0 | 0 | 7.03 | ± | 0.72 |
| 16 | 0 | 0 | 0 | 5.93 | ± | 0.38 |
| 17 | 0 | 0 | 0 | 7.82 | ± | 0.17 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Showkat, M.M.; Falck-Ytter, A.B.; Strætkvern, K.O. Phenolic Acids in Jerusalem Artichoke (Helianthus tuberosus L.): Plant Organ Dependent Antioxidant Activity and Optimized Extraction from Leaves. Molecules 2019, 24, 3296. https://doi.org/10.3390/molecules24183296
Showkat MM, Falck-Ytter AB, Strætkvern KO. Phenolic Acids in Jerusalem Artichoke (Helianthus tuberosus L.): Plant Organ Dependent Antioxidant Activity and Optimized Extraction from Leaves. Molecules. 2019; 24(18):3296. https://doi.org/10.3390/molecules24183296
Chicago/Turabian StyleShowkat, Muhammad Mir, Anne Bergljot Falck-Ytter, and Knut Olav Strætkvern. 2019. "Phenolic Acids in Jerusalem Artichoke (Helianthus tuberosus L.): Plant Organ Dependent Antioxidant Activity and Optimized Extraction from Leaves" Molecules 24, no. 18: 3296. https://doi.org/10.3390/molecules24183296
APA StyleShowkat, M. M., Falck-Ytter, A. B., & Strætkvern, K. O. (2019). Phenolic Acids in Jerusalem Artichoke (Helianthus tuberosus L.): Plant Organ Dependent Antioxidant Activity and Optimized Extraction from Leaves. Molecules, 24(18), 3296. https://doi.org/10.3390/molecules24183296




