3.4. Analytical Characterization Data of Products
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-phenylalanine Schiff Base Complex (6a). Red solid (138 mg, yield 98%); mp 116–118 °C; [α]20D = +2502 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.08 (d, J = 2.1 Hz, 1H), 8.32 (d, J = 9.3 Hz, 1H), 7.61–7.49 (m, 3H), 7.42–7.27 (m, 6H), 7.15–7.02 (m, 3H), 6.65 (d, J = 7.7 Hz, 1H), 6.61 (d, J = 2.6 Hz, 1H), 4.22 (t, J = 5.2 Hz, 1H), 3.54 (dd, J = 59.9, 13.0 Hz, 2H), 3.36–3.24 (m, 1H), 3.14 (dt, J = 17.6, 6.8 Hz, 2H), 2.78 (dd, J = 13.8, 5.4 Hz, 1H), 2.42–2.23 (m, 1H), 2.14–2.03 (m, 1H), 1.98 (dd, J = 19.7, 9.7 Hz, 1H), 1.87–1.73 (m, 1H), 1.33 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.5, 178.1, 170.8, 141.5, 135.8, 134.0, 133.4, 133.1, 133.0, 132.7, 132.6, 131.1, 130.5, 130.3, 129.8, 129.5, 129.3, 129.1, 127.9, 127.7, 127.5, 125.8, 124.3, 74.3, 71.8, 57.3, 54.7, 41.4, 39.8, 20.7, 17.8. LRMS (ESI, m/z): 704.0 [M + H]+. HRMS (ESI, m/z): calcd for C35H31Cl3N3NiO3, 704.0605 [M + H]+; found 704.0625. The dr was determined by LC-MS using a system equipped with a binary pump, and a photodiode array detector (DAD), using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 15.966 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-2-methoxyphenylalanine Schiff Base Complex (6b). Red solid (132 mg, yield 90%); mp 125–128 °C; [α]20D = +2021 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.00 (d, J = 2.0 Hz, 1H), 8.36 (d, J = 9.3 Hz, 1H), 7.57–7.53 (m, 2H), 7.52–7.48 (m, 1H), 7.41–7.37 (m, 1H), 7.36–7.31 (m, 1H), 7.31–7.27 (m, 2H), 7.22–7.19 (m, 1H), 7.10 (dd, J = 9.3, 2.6 Hz, 1H), 6.96 (t, J = 7.4 Hz, 1H), 6.88–6.82 (m, 2H), 6.62 (d, J = 2.6 Hz, 1H), 4.15 (t, J = 4.9 Hz, 1H), 3.52 (q, J = 13.4 Hz, 2H), 3.31–3.22 (m, 4H), 3.21–3.16 (m, 1H), 3.11–3.06 (m, 1H), 2.91 (dd, J = 13.6, 4.9 Hz, 1H), 2.31–2.20 (m, 1H), 2.10–2.01 (m, 1H), 1.94 (q, J = 9.9 Hz, 1H), 1.83–1.73 (m, 1H), 1.31 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.5, 178.6, 171.1, 158.5, 141.4, 135.9, 134.1, 133.7, 133.2, 133.1, 132.9, 132.7, 132.5, 131.2, 130.1, 130.0, 129.4, 129.3, 129.3, 128.4, 128.0, 127.7, 125.7, 124.5, 124.2, 121.5, 110.7, 74.6, 72.0, 57.7, 54.9, 54.6, 41.4, 34.8, 20.9, 18.1. LRMS (ESI, m/z): 734.0 [M − H]−. HRMS (ESI, m/z): calcd for C36H31Cl3N3NiO4, 734.0739 [M − H]−; found 734.0741. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 19.059 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-3-methoxyphenylalanine Schiff Base Complex (6c). Red solid (127 mg, yield 90%); mp 118–121 °C; [α]20D = +3344 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.07–9.05 (m, 1H), 8.30 (d, J = 9.3 Hz, 1H), 7.59–7.45 (m, 3H), 7.36–7.27 (m, 3H), 7.23–7.18 (m, 1H), 7.09 (dd, J = 9.3, 2.6 Hz, 1H), 6.88–6.83 (m, 1H), 6.64 (d, J = 7.5 Hz, 1H), 6.59–6.56 (m, 2H), 6.55–6.50 (m, 1H), 4.19 (t, J = 5.3 Hz, 1H), 3.63 (s, 3H), 3.60 (d, J = 13.1 Hz, 1H), 3.45 (d, J = 13.2 Hz, 1H), 3.42–3.34 (m, 1H), 3.22–3.09 (m, 2H), 2.78 (dd, J = 13.8, 5.1 Hz, 1H), 2.49–2.36 (m, 1H), 2.17–2.08 (m, 1H), 2.04–1.94 (m, 1H), 1.91–1.79 (m, 1H), 1.33 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.8, 178.3, 171.0, 141.6, 135.9, 135.8, 134.1, 133.5, 133.2, 133.1, 132.8, 132.7, 131.2, 130.4, 129.9, 129.7, 129.6, 129.4, 127.8, 127.5, 126.5, 125.9, 124.4, 124.4, 74.5, 71.4, 57.5, 54.9, 54.6, 41.6, 34.2, 21.0, 18.0. LRMS (ESI, m/z): 734.0 [M − H]−. HRMS (ESI, m/z): calcd for C36H31Cl3N3NiO4, 734.0739 [M − H]−; found 734.0741. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 16.298 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-3-methylphenylalanine Schiff Base Complex (6d). Red solid (126 mg, yield 88%); mp 135–136 °C; [α]20D = +2588 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.06–9.03 (m, 1H), 8.33 (d, J = 9.3 Hz, 1H), 7.56 (t, J = 7.7 Hz, 2H), 7.51 (t, J = 7.5 Hz, 1H), 7.37 (t, J = 7.5 Hz, 1H), 7.33–7.27 (m, 2H), 7.24–7.18 (m, 1H), 7.16–7.09 (m, 2H), 6.89–6.83 (m, 2H), 6.64 (d, J = 7.6 Hz, 1H), 6.61 (d, J = 2.5 Hz, 1H), 4.20 (t, J = 5.1 Hz, 1H), 3.63–3.55 (m, 1H), 3.50–3.42 (m, 1H), 3.35–3.24 (m, 1H), 3.16–3.06 (m, 2H), 2.74 (dd, J = 13.7, 5.4 Hz, 1H), 2.41–2.29 (m, 1H), 2.26 (s, 3H), 2.15–2.03 (m, 1H), 2.02–1.94 (m, 1H), 1.88–1.76 (m, 1H), 1.32 (s, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.2, 178.0, 170.5, 141.3, 138.5, 135.6, 135.4, 133.8, 133.1, 132.9, 132.8, 132.4, 132.4, 131.1, 131.1, 130.9, 130.0, 129.6, 129.3, 128.9, 128.7, 128.2, 127.7, 127.5, 127.3, 127.3, 125.5, 124.0, 74.1, 71.6, 57.1, 54.5, 41.0, 39.6, 21.4, 20.4, 17.6. LRMS (ESI, m/z): 716.0 [M − H]−. HRMS (ESI, m/z): calcd for C36H31Cl3N3NiO3, 716.0790 [M − H]−; found 716.0798. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 21.599 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-4-fluorophenylalanine Schiff Base Complex (6e). Red solid (132 mg, yield 91%); mp 188–190 °C; [α]20D = +1786 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.09–9.06 (m, 1H), 8.33 (d, J = 9.3 Hz, 1H), 7.62–7.52 (m, 3H), 7.47–7.41 (m, 1H), 7.34 (d, J = 7.0 Hz, 1H), 7.31 (d, J = 8.2 Hz, 1H), 7.14 (dd, J = 9.3, 2.5 Hz, 1H), 7.08–7.03 (m, 4H), 6.74 (d, J = 7.5 Hz, 1H), 6.63 (d, J = 2.5 Hz, 1H), 4.21 (t, J = 5.0 Hz, 1H), 3.64–3.58 (m, 1H), 3.52–3.46 (m, 1H), 3.40–3.30 (m, 1H), 3.17–3.03 (m, 2H), 2.75 (dd, J = 14.0, 5.4 Hz, 1H), 2.42–2.30 (m, 1H), 2.23–2.14 (m, 1H), 2.06–1.96 (m, 1H), 1.90–1.84 (m, 1H), 1.35 (s, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.3, 177.7, 170.7, 162.3 (d, J = 246.68 Hz), 141.3, 135.5, 133.7, 133.1, 132.9, 132.8, 132.5, 132.4, 131.7, 131.7, 131.3, 131.3, 130.9, 130.2, 129.6, 129.4, 129.1, 127.5, 127.4, 127.3, 125.7, 124.1, 115.8, 115.7, 74.2, 71.4, 57.2, 54.4, 41.0, 38.6, 20.5, 17.6. LRMS (ESI, m/z): 720.0 [M − H]−. HRMS (ESI, m/z): calcd for C35H28Cl3FN3NiO3, 720.0539 [M − H]−; found 720.0547. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 17.940 min, tminor = not found, dr > 99:1.
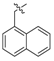
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-3,5-diiodotyrosine Schiff Base Complex (6f). Red solid (175 mg, yield 90%); mp 286–288 °C; [α]20D = +2037 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.05 (d, J = 2.0 Hz, 1H), 8.43 (d, J = 9.3 Hz, 1H), 7.66–7.53 (m, 3H), 7.47 (dd, J = 10.8, 5.7 Hz, 1H), 7.33 (t, J = 7.2 Hz, 2H), 7.27 (s, 2H), 7.16 (dd, J = 9.3, 2.6 Hz, 1H), 6.78 (d, J = 7.4 Hz, 1H), 6.66 (d, J = 2.5 Hz, 1H), 5.82 (s, 1H), 4.14–4.07 (m, 1H), 3.56 (dd, J = 29.2, 13.0 Hz, 2H), 3.46–3.34 (m, 1H), 3.17 (t, J = 8.8 Hz, 1H), 2.89 (dd, J = 14.1, 4.4 Hz, 1H), 2.67 (dd, J = 13.9, 6.2 Hz, 1H), 2.54–2.39 (m, 1H), 2.24 (dd, J = 13.8, 9.8 Hz, 1H), 2.02 (td, J = 18.7, 9.4 Hz, 2H), 1.37 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 177.6, 171.0, 153.4, 141.8, 140.6, 135.7, 134.0, 133.2, 133.1, 133.0, 132.6, 131.8, 131.2, 130.6, 129.8, 129.7, 129.4, 127.6, 127.5, 127.4, 125.8, 124.4, 82.8, 74.5, 71.2, 57.5, 54.7, 41.0, 38.0, 21.0, 17.9. LRMS (ESI, m/z): 971.8 [M + H]+. HRMS (ESI, m/z): calcd for C35H29Cl3I2N3NiO4, 971.8487 [M − H]−; found 971.8509. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 17.421 min, tminor = not found, dr > 99:1.
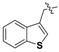
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-3-(1-naphthyl)alanine Schiff Base Complex (6g). Red solid (139 mg, yield 92%); mp 182–184 °C; [α]20D = +2432 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.12–9.09 (m, 1H), 8.31 (d, J = 9.3 Hz, 1H), 7.80 (t, J = 8.0 Hz, 2H), 7.69 (dd, J = 8.2, 2.0 Hz, 1H), 7.58 (d, J = 8.6 Hz, 1H), 7.42–7.37 (m, 1H), 7.36–7.28 (m, 4H), 7.21 (t, J = 7.7 Hz, 1H), 7.19–7.12 (m, 2H), 7.09 (dd, J = 9.3, 2.6 Hz, 1H), 6.69 (t, J = 7.6 Hz, 1H), 6.45 (d, J = 2.6 Hz, 1H), 5.65 (d, J = 7.7 Hz, 1H), 4.45–4.40 (m, 1H), 4.12–4.04 (m, 1H), 3.83–3.77 (m, 1H), 3.71 (d, J = 13.2 Hz, 1H), 3.67–3.57 (m, 1H), 3.47 (d, J = 13.2 Hz, 1H), 3.30–3.20 (m, 1H), 2.84–2.54 (m, 1H), 2.35–2.25 (m, 1H), 2.13 (q, J = 9.6 Hz, 1H), 2.07–1.95 (m, 1H), 1.41 (s, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.1, 178.1, 170.3, 141.1, 135.6, 134.0, 133.6, 133.1, 132.7, 132.6, 132.4, 132.3, 132.1, 131.5, 131.0, 129.5, 129.4, 128.6, 128.5, 128.4, 128.3, 128.3, 127.5, 127.3, 127.2, 126.3, 125.8, 125.5, 125.4, 123.9, 123.2, 74.2, 71.4, 56.7, 54.9, 41.4, 40.0, 20.7, 17.7. LRMS (ESI, m/z): 752.0 [M − H]−. HRMS (ESI, m/z): calcd for C39H31Cl3N3NiO3, 752.0790 [M − H]−; found 752.0799. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 24.156 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-3-(3-benzothienyl)alanine Schiff Base Complex (6h). Red solid (142 mg, yield 94%); mp 138–141 °C; [α]20D = +2452 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.08–9.05 (m, 1H), 8.35 (d, J = 9.3 Hz, 1H), 7.87 (d, J = 8.0 Hz, 1H), 7.76–7.56 (m, 1H), 7.50 (t, J = 7.5 Hz, 1H), 7.43 (d, J = 8.0 Hz, 1H), 7.38 (t, J = 7.5 Hz, 1H), 7.34–7.28 (m, 3H), 7.19–7.10 (m, 4H), 6.60 (d, J = 2.6 Hz, 1H), 6.49 (d, J = 7.5 Hz, 1H), 4.32 (t, J = 5.5 Hz, 1H), 3.65–3.57 (m, 2H), 3.48–3.41 (m, 1H), 3.23–3.15 (m, 1H), 3.11–2.98 (m, 2H), 2.13–1.90 (m, 3H), 1.82–1.70 (m, 1H), 1.31 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.0, 178.1, 170.6, 141.3, 140.4, 139.1, 135.5, 133.7, 133.0, 132.8, 132.7, 132.5, 130.9, 130.3, 129.9, 129.6, 129.1, 128.8, 127.3, 127.3, 125.7, 125.5, 124.6, 124.3, 124.0, 122.7, 121.9, 74.1, 70.7, 57.0, 54.6, 40.8, 33.8, 20.2, 17.6. LRMS (ESI, m/z): 758.0 [M − H]−. HRMS (ESI, m/z): calcd for C37H29Cl3N3NiO3S, 758.0354 [M − H]−; found 758.0362. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 27.103 min, tminor = 24.123, dr > 99:1.
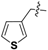
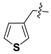
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-3-(3-thienyl)alanine Schiff Base Complex (6i). Red solid (126 mg, yield 89%); mp 178–180 °C; [α]20D = +2012 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.10 (d, J = 2.1 Hz, 1H), 8.35 (d, J = 9.3 Hz, 1H), 7.61–7.51 (m, 3H), 7.46–7.41 (m, 1H), 7.37–7.30 (m, 3H), 7.14 (dd, J = 9.3, 2.6 Hz, 1H), 7.06–7.04 (m, 1H), 6.86 (dd, J = 4.9, 1.2 Hz, 1H), 6.73–6.69 (m, 1H), 6.63 (d, J = 2.6 Hz, 1H), 4.18 (t, J = 5.1 Hz, 1H), 3.67–3.61 (m, 1H), 3.56–3.46 (m, 2H), 3.22–3.15 (m, 2H), 2.82–2.75 (m, 1H), 2.58–2.44 (m, 1H), 2.25–2.15 (m, 1H), 2.03 (q, J = 9.6 Hz, 1H), 1.97–1.87 (m, 1H), 1.37 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 178.1, 170.7, 141.3, 135.6, 135.5, 133.8, 133.2, 132.9, 132.8, 132.5, 132.4, 130.9, 130.1, 129.6, 129.4, 129.3, 129.1, 127.5, 127.2, 126.2, 125.6, 124.1, 124.1, 74.2, 71.1, 57.2, 54.6, 41.3, 33.9, 20.7, 17.7. LRMS (ESI, m/z): 708.0 [M − H]−. HRMS (ESI, m/z): calcd for C33H27Cl3N3NiO3S, 708.0198 [M − H]−; found 708.0200. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 15.723 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-2-(3-methoxyphenyl)glycine Schiff Base Complex (6j). Red solid (124 mg, yield 87%); mp 153–155 °C; [α]20D = +2183 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.21–9.17 (m, 1H), 8.24 (d, J = 9.3 Hz, 1H), 7.82–7.78 (m, 1H), 7.58–7.51 (m, 1H), 7.44–7.36 (m, 3H), 7.30 (d, J = 7.5 Hz, 1H), 7.23 (t, J = 7.9 Hz, 1H), 7.16 (dd, J = 9.3, 2.5 Hz, 1H), 7.11–7.05 (m, 2H), 6.83–6.78 (m, 1H), 6.67 (d, J = 2.5 Hz, 1H), 6.15 (d, J = 7.9 Hz, 1H), 4.72 (s, 1H), 4.08–3.99 (m, 1H), 3.84–3.78 (m, 1H), 3.72 (s, 3H), 3.65–3.58 (m, 1H), 3.39–3.31 (m, 1H), 3.26–3.08 (m, 1H), 2.51–2.41 (m, 1H), 2.27–2.08 (m, 2H), 1.50 (s, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.6, 177.2, 171.5, 159.6, 141.1, 139.0, 135.6, 133.8, 133.1, 133.1, 132.9, 132.4, 132.2, 131.0, 129.8, 129.7, 129.5, 128.8, 128.7, 127.5, 126.8, 126.5, 125.8, 124.6, 118.2, 74.5, 74.2, 57.0, 55.2, 55.0, 41.5, 21.0, 18.1. LRMS (ESI, m/z): 718.0 [M − H]−. HRMS (ESI, m/z): calcd for C35H29Cl3N3NiO4, 718.0583 [M − H]−; found 718.0602. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 16.728 min, tminor = 18.308, dr = 97:3.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-2-(3-bromophenyl)glycine Schiff Base Complex (6k). Red solid (131 mg, yield 85%); mp 119–121 °C; [α]20D = +2543 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.20 (d, J = 2.1 Hz, 1H), 8.24 (d, J = 9.3 Hz, 1H), 8.17 (t, J = 1.9 Hz, 1H), 7.81 (dd, J = 8.2, 2.2 Hz, 1H), 7.62–7.55 (m, 1H), 7.49–7.40 (m, 3H), 7.36 (d, J = 7.9 Hz, 1H), 7.32 (d, J = 7.6 Hz, 1H), 7.22–7.10 (m, 3H), 6.69 (d, J = 2.5 Hz, 1H), 6.17–6.12 (m, 1H), 4.72 (s, 1H), 4.11–4.01 (m, 1H), 3.86–3.81 (m, 1H), 3.67–3.61 (m, 1H), 3.41–3.35 (m, 1H), 3.22–3.10 (m, 1H), 2.57–2.46 (m, 1H), 2.28–2.16 (m, 2H), 1.52 (s, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.6, 176.6, 171.9, 141.2, 139.7, 135.6, 133.8, 133.2, 132.7, 132.6, 132.2, 131.4, 131.1, 130.1, 130.0, 129.8, 129.1, 129.1, 128.9, 127.4, 126.7, 126.5, 125.9, 125.1, 124.7, 122.7, 74.3, 73.8, 57.1, 55.1, 41.6, 21.0, 18.1. LRMS (ESI, m/z): 765.9 [M − H]−. HRMS (ESI, m/z): calcd for C34H26BrCl3N3NiO3, 765.9582 [M − H]−; found 765.9579. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/ H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 25.652 min, tminor = 28.939, dr = 98:2.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-2-cyclobutylglycine Schiff Base Complex (6l). Red solid (118 mg, yield 89%); mp 307–308 °C; [α]20D = +3252 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.12 (s, 1H), 8.23 (d, J = 9.3 Hz, 1H), 7.70 (d, J = 8.2 Hz, 1H), 7.60–7.38 (m, 3H), 7.29 (t, J = 6.9 Hz, 2H), 7.09 (dd, J = 9.3, 2.5 Hz, 1H), 6.87 (d, J = 7.4 Hz, 1H), 6.58 (d, J = 2.5 Hz, 1H), 4.21–3.95 (m, 1H), 3.82–3.69 (m, 2H), 3.51 (d, J = 13.0 Hz, 1H), 3.40 (t, J = 8.7 Hz, 1H), 3.17–2.88 (m, 1H), 2.67 (q, J = 17.3 Hz, 1H), 2.58–2.33 (m, 3H), 2.29–2.12 (m, 2H), 2.09–1.96 (m, 1H), 1.81 (dd, J = 18.1, 8.2 Hz, 1H), 1.70 (dd, J = 18.3, 9.0 Hz, 2H), 1.44 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.3, 177.2, 169.6, 140.9, 135.8, 133.9, 133.3, 133.1, 133.0, 132.5, 132.3, 131.1, 130.3, 129.8, 129.4, 129.1, 128.1, 128.0, 127.8, 125.8, 124.2, 74.5, 74.0, 57.2, 55.0, 41.9, 40.7, 26.0, 25.6, 20.7, 18.1, 17.5. LRMS (ESI, m/z): 668.0 [M + H]+. HRMS (ESI, m/z): calcd for C32H31Cl3N3NiO3, 668.0604 [M + H]+; found 668.0624. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 16.283 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-2-amino-4,4,4-trifluorobutyric acid Schiff Base Complex (6m). Red solid (121 mg, yield 87%); mp 244–246 °C; [α]20D = +2830 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.17 (d, J = 1.8 Hz, 1H), 8.35 (d, J = 9.3 Hz, 1H), 7.66–7.55 (m, 3H), 7.55–7.50 (m, 1H), 7.36 (d, J = 8.1 Hz, 1H), 7.30 (d, J = 6.8 Hz, 1H), 7.17 (dd, J = 9.3, 2.5 Hz, 1H), 6.90 (d, J = 7.4 Hz, 1H), 6.61 (d, J = 2.5 Hz, 1H), 4.23–4.11 (m, 2H), 3.74–3.57 (m, 2H), 3.39 (t, J = 8.7 Hz, 1H), 3.06–2.92 (m, 1H), 2.63–2.37 (m, 2H), 2.21–2.10 (m, 2H), 2.05–1.93 (m, 1H), 1.44 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 177.2, 172.4, 141.9, 135.8, 134.0, 133.2, 133.2, 133.2, 133.1, 132.5, 131.3, 130.8, 129.9, 129.8, 129.7, 127.6, 127.6, 126.8, 125.8, 124.6, 74.9, 64.5, 57.8, 54.8, 41.2, 35.6 (q, J = 28.9 Hz), 20.3, 17.4. LRMS (ESI, m/z): 694.0 [M − H]−. HRMS (ESI, m/z): calcd for C30H24Cl3F3N3NiO3, 694.0194 [M − H]−; found 694.0203. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 13.938 min, tminor = 15.440, dr = 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-norvaline Schiff Base Complex (6n). Red solid (121 mg, yield 92%); mp 244–247 °C; [α]20D = +2842 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.14–9.12 (m, 1H), 8.22 (d, J = 9.3 Hz, 1H), 7.75 (dd, J = 8.2, 2.0 Hz, 1H), 7.60–7.51 (m, 2H), 7.50–7.45 (m, 1H), 7.35 (d, J = 8.2 Hz, 1H), 7.33–7.29 (m, 1H), 7.13 (dd, J = 9.3, 2.6 Hz, 1H), 6.89–6.85 (m, 1H), 6.62 (d, J = 2.6 Hz, 1H), 4.09–3.99 (m, 1H), 3.89 (dd, J = 7.6, 3.6 Hz, 1H), 3.77 (d, J = 13.0 Hz, 1H), 3.57 (d, J = 13.1 Hz, 1H), 3.40 (t, J = 8.8 Hz, 1H), 3.22–3.10 (m, 1H), 2.50–2.38 (m, 1H), 2.29–2.16 (m, 2H), 2.15–1.99 (m, 1H), 1.95–1.82 (m, 1H), 1.71 (dd, J = 13.1, 6.3 Hz, 1H), 1.62–1.56 (m, 1H), 1.47 (s, 3H), 0.81 (t, J = 7.2 Hz, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.2, 178.8, 169.6, 140.7, 135.6, 133.7, 133.0, 132.9, 132.8, 132.1, 131.0, 130.1, 129.7, 129.3, 129.1, 127.9, 127.3, 127.3, 125.7, 124.3, 74.3, 70.3, 57.0, 54.8, 41.4, 37.2, 20.8, 18.2, 17.7, 13.7. LRMS (ESI, m/z): 654.1 [M − H]−. HRMS (ESI, m/z): calcd for C31H29Cl3N3NiO3, 654.0633 [M − H]−; found 654.0643. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 14.871 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-valine Schiff Base Complex (6o). Red solid (122 mg, yield 93%); mp 267–269 °C; [α]20D = +3016 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.17 (d, J = 2.1 Hz, 1H), 8.28 (d, J = 9.3 Hz, 1H), 7.65 (dd, J = 8.2, 2.1 Hz, 1H), 7.54 (dt, J = 16.1, 7.9 Hz, 2H), 7.45 (t, J = 6.9 Hz, 1H), 7.31 (dd, J = 7.7, 4.2 Hz, 2H), 7.10 (dd, J = 9.3, 2.6 Hz, 1H), 6.85 (d, J = 7.5 Hz, 1H), 6.61 (d, J = 2.6 Hz, 1H), 4.14–3.97 (m, 1H), 3.83–3.72 (m, 2H), 3.53 (d, J = 13.1 Hz, 1H), 3.40 (t, J = 9.0 Hz, 1H), 3.00 (td, J = 12.5, 4.0 Hz, 1H), 2.53–2.34 (m, 1H), 2.27–2.08 (m, 2H), 1.84 (d, J = 6.6 Hz, 3H), 1.80–1.67 (m, 1H), 1.45 (s, 3H), 0.73 (d, J = 6.8 Hz, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 177.4, 170.3, 141.0, 135.9, 134.0, 133.2, 133.2, 133.0, 132.5, 132.4, 131.2, 130.2, 129.8, 129.5, 129.1, 127.9, 127.9, 127.5, 125.8, 124.2, 75.6, 74.4, 57.3, 54.9, 41.7, 34.4, 20.5, 19.9, 17.8, 17.7. LRMS (ESI, m/z): 656.0 [M + H]+. HRMS (ESI, m/z): calcd for C31H31Cl3N3NiO3, 656.0604 [M + H]+; found 656.0606. The dr was determined by LC-MS with binary pump, photodiode array detector (DAD), using Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 14.594 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-leucine Schiff Base Complex (6p). Red solid (127 mg, yield 95%); mp 156–159 °C; [α]20D = +2731 (c = 1, CHCl3); 1H-NMR (500 MHz, CDCl3) δ 9.14–9.11 (m, 1H), 8.16 (d, J = 9.3 Hz, 1H), 7.80 (dd, J = 8.2, 2.1 Hz, 1H), 7.59–7.55 (m, 1H), 7.53 (t, J = 7.4 Hz, 1H), 7.50–7.45 (m, 1H), 7.36–7.32 (m, 2H), 7.12 (dd, J = 9.3, 2.6 Hz, 1H), 6.88 (d, J = 7.6 Hz, 1H), 6.62 (d, J = 2.6 Hz, 1H), 4.06–3.97 (m, 1H), 3.85 (dd, J = 10.5, 4.1 Hz, 1H), 3.82 (d, J = 13.2 Hz, 1H), 3.54 (d, J = 13.1 Hz, 1H), 3.40 (t, J = 8.8 Hz, 1H), 3.34–3.17 (m, 1H), 2.48–2.34 (m, 2H), 2.28–2.18 (m, 2H), 1.97–1.86 (m, 1H), 1.47 (s, 3H), 1.39–1.32 (m, 1H), 0.83 (d, J = 6.7 Hz, 3H), 0.40 (d, J = 6.5 Hz, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.2, 178.6, 169.0, 140.6, 135.6, 133.7, 133.1, 132.9, 132.7, 132.0, 130.9, 130.1, 129.7, 129.2, 129.1, 127.9, 127.6, 127.4, 125.7, 124.3, 74.1, 69.1, 56.7, 55.1, 46.0, 41.6, 24.3, 23.7, 21.2, 20.8, 17.9. LRMS (ESI, m/z): 668.0 [M − H]−. HRMS (ESI, m/z): calcd for C32H31Cl3N3NiO3, 668.0955 [M − H]−; found 668.0947. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 20.065 min, tminor = not found, dr > 99:1.
Nickel(II)-(S)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(S)-methionine Schiff Base Complex (6q). Red solid (127 mg, yield 93%); mp 143–145 °C; [α]20D = +1883 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.15–9.11 (m, 1H), 8.23 (d, J = 9.3 Hz, 1H), 7.76–7.71 (m, 1H), 7.62–7.52 (m, 2H), 7.52–7.46 (m, 1H), 7.38–7.34 (m, 1H), 7.34–7.30 (m, 1H), 7.14 (dd, J = 9.3, 2.5 Hz, 1H), 6.90 (d, J = 7.5 Hz, 1H), 6.62 (d, J = 2.5 Hz, 1H), 4.07–3.98 (m, 1H), 3.97–3.92 (m, 1H), 3.81–3.72 (m, 1H), 3.61–3.54 (m, 1H), 3.39 (t, J = 8.9 Hz, 1H), 3.23–3.12 (m, 1H), 3.11–3.03 (m, 1H), 2.65–2.54 (m, 1H), 2.50–2.38 (m, 1H), 2.30–2.14 (m, 3H), 1.99 (s, 3H), 1.93–1.83 (m, 1H), 1.47 (s, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.4, 178.3, 170.3, 141.1, 135.7, 133.9, 133.3, 133.1, 132.9, 132.5, 132.4, 131.2, 130.4, 129.9, 129.6, 129.6, 129.4, 127.9, 127.5, 127.4, 125.9, 124.6, 74.5, 69.8, 57.3, 55.0, 41.6, 35.1, 29.8, 21.2, 18.0, 15.8. LRMS (ESI, m/z): 686.0 [M − H]−. HRMS (ESI, m/z): calcd for C31H29Cl3N3NiO3S, 686.0354 [M − H]−; found 686.0371. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 13.877 min, tminor = 15.012, dr = 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-phenylalanine Schiff Base Complex (6a). Red solid (128 mg, yield 91%); mp 121–124 °C; [α]20D = –1684 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.09 (d, J = 2.1 Hz, 1H), 8.34 (d, J = 9.3 Hz, 1H), 7.62–7.56 (m, 2H), 7.56–7.51 (m, 1H), 7.43–7.32 (m, 5H), 7.31–7.29 (m, 1H), 7.16–7.09 (m, 3H), 6.68–6.64 (m, 1H), 6.62 (d, J = 2.6 Hz, 1H), 4.24 (t, J = 5.2 Hz, 1H), 3.65–3.60 (m, 1H), 3.51–3.45 (m, 1H), 3.36–3.26 (m, 1H), 3.20–3.09 (m, 2H), 2.83–2.76 (m, 1H), 2.41–2.27 (m, 1H), 2.16–2.05 (m, 1H), 2.05–1.95 (m, 1H), 1.88–1.77 (m, 1H), 1.35 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.3, 177.9, 170.6, 141.3, 135.6, 133.8, 133.2, 132.9, 132.8, 132.4, 132.4, 130.9, 130.3, 130.1, 129.6, 129.3, 129.1, 128.9, 127.7, 127.5, 127.3, 125.6, 124.1, 74.1, 71.6, 57.1, 54.5, 41.2, 39.6, 20.5, 17.6. LRMS (ESI, m/z): 702.0 [M − H]−. HRMS (ESI, m/z): calcd for C35H29Cl3N3NiO3, 702.0633 [M − H]−; found 702.0650. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 15.665 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-2-methoxyphenylalanine Schiff Base Complex (6b). Red solid (129 mg, yield 88%); mp 112–114 °C; [α]20D = –1818 (c = 1, CHCl3); 1H-NMR (500 MHz, CDCl3) δ 9.01 (d, J = 2.0 Hz, 1H), 8.37 (d, J = 9.3 Hz, 1H), 7.60–7.53 (m, 2H), 7.51 (t, J = 7.5 Hz, 1H), 7.39 (t, J = 7.4 Hz, 1H), 7.36–7.32 (m, 1H), 7.30 (d, J = 8.1 Hz, 2H), 7.21 (dd, J = 7.4, 1.3 Hz, 1H), 7.11 (dd, J = 9.3, 2.6 Hz, 1H), 6.97 (t, J = 7.4 Hz, 1H), 6.86 (dd, J = 11.1, 8.1 Hz, 2H), 6.63 (d, J = 2.6 Hz, 1H), 4.15 (t, J = 4.9 Hz, 1H), 3.52 (dd, J = 30.2, 13.0 Hz, 2H), 3.31–3.23 (m, 4H), 3.19 (dd, J = 13.6, 5.0 Hz, 1H), 3.09 (t, J = 8.9 Hz, 1H), 2.91 (dd, J = 13.6, 4.9 Hz, 1H), 2.33–2.20 (m, 1H), 2.10–2.02 (m, 1H), 1.95 (dd, J = 19.5, 9.9 Hz, 1H), 1.84–1.72 (m, 1H), 1.32 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 178.4, 170.9, 158.3, 141.2, 135.8, 134.0, 133.5, 133.0, 132.9, 132.7, 132.6, 132.3, 131.0, 130.0, 129.8, 129.3, 129.2, 129.2, 128.3, 127.9, 127.6, 125.6, 124.3, 124.1, 121.3, 110.5, 74.5, 71.9, 57.5, 54.7, 54.5, 41.3, 34.6, 20.8, 17.9. LRMS (ESI, m/z): 734.0 [M + H]+. HRMS (ESI, m/z): calcd for C36H33Cl3N3NiO4, 734.0885, [M + H]+; found 734.0887. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 19.227 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-3-methoxyphenylalanine Schiff Base Complex (6c). Red solid (131 mg, yield 93%); mp 104–106 °C; [α]20D = –2923 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.08 (d, J = 2.0 Hz, 1H), 8.33 (d, J = 9.3 Hz, 1H), 7.62–7.47 (m, 3H), 7.40–7.28 (m, 3H), 7.26–7.18 (m, 1H), 7.12 (dd, J = 9.3, 2.6 Hz, 1H), 6.88 (dd, J = 8.3, 2.3 Hz, 1H), 6.67 (d, J = 7.5 Hz, 1H), 6.60 (d, J = 2.6 Hz, 2H), 6.55 (d, J = 7.9 Hz, 1H), 4.21 (t, J = 5.3 Hz, 1H), 3.65 (s, 3H), 3.55 (dd, J = 59.3, 13.1 Hz, 2H), 3.45–3.31 (m, 1H), 3.26–3.03 (m, 2H), 2.81 (dd, J = 13.8, 5.1 Hz, 1H), 2.46 (dq, J = 14.1, 7.2 Hz, 1H), 2.25–2.11 (m, 1H), 2.07–1.96 (m, 1H), 1.95–1.78 (m, 1H), 1.35 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.6, 178.3, 170.9, 141.5, 135.8, 135.7, 134.0, 133.4, 133.1, 133.0, 132.7, 132.6, 131.1, 130.3, 129.8, 129.6, 129.5, 129.3, 127.7, 127.4, 126.4, 125.8, 124.3, 124.3, 74.4, 71.3, 57.4, 54.8, 41.5, 34.1, 20.9, 17.9. LRMS (ESI, m/z): 734.0 [M + H]+. HRMS (ESI, m/z): calcd for C36H33Cl3N3NiO4, 734.0711 [M + H]+; found 734.0718. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 16.492 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-3-methylphenylalanine Schiff Base Complex (6d). Red solid (129 mg, yield 90%); mp 105–107 °C; [α]20D = –1898 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.06 (d, J = 1.5 Hz, 1H), 8.34 (d, J = 9.3 Hz, 1H), 7.55 (dt, J = 17.5, 7.4 Hz, 3H), 7.38 (t, J = 7.5 Hz, 1H), 7.31 (t, J = 6.7 Hz, 2H), 7.23 (t, J = 7.5 Hz, 1H), 7.17–7.09 (m, 2H), 6.87 (d, J = 7.8 Hz, 2H), 6.63 (dd, J = 12.2, 5.1 Hz, 2H), 4.21 (t, J = 5.1 Hz, 1H), 3.53 (dd, J = 47.7, 13.1 Hz, 2H), 3.31 (td, J = 12.6, 5.8 Hz, 1H), 3.11 (dd, J = 13.7, 4.7 Hz, 2H), 2.75 (dd, J = 13.7, 5.4 Hz, 1H), 2.47–2.31 (m, 1H), 2.27 (s, 3H), 2.13–2.06 (m, 1H), 1.99 (dd, J = 19.4, 9.7 Hz, 1H), 1.84 (dt, J = 14.9, 6.7 Hz, 1H), 1.33 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 178.2, 170.7, 141.5, 138.7, 135.8, 135.6, 134.0, 133.3, 133.1, 133.0, 132.6, 132.6, 131.3, 131.1, 130.2, 129.8, 129.5, 129.2, 128.9, 128.4, 127.9, 127.7, 127.5, 127.5, 125.7, 124.2, 74.3, 71.8, 57.3, 54.7, 41.2, 39.8, 21.6, 20.6, 17.8. LRMS (ESI, m/z): 718.0 [M + H]+. HRMS (ESI, m/z): calcd for C36H33Cl3N3NiO3, 718.0762 [M + H]+; found 718.0773. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 21.866 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-4-fluorophenylalanine Schiff Base Complex (6e). Red solid (129 mg, yield 89%); mp 235–237 °C; [α]20D = −2300 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.06 (d, J = 1.5 Hz, 1H), 8.32 (d, J = 9.3 Hz, 1H), 7.56 (p, J = 7.5 Hz, 3H), 7.43 (t, J = 7.3 Hz, 1H), 7.37–7.28 (m, 2H), 7.13 (dd, J = 9.3, 2.5 Hz, 1H), 7.05 (d, J = 7.0 Hz, 4H), 6.73 (d, J = 7.5 Hz, 1H), 6.62 (d, J = 2.5 Hz, 1H), 4.21 (t, J = 5.0 Hz, 1H), 3.54 (dd, J = 47.7, 13.0 Hz, 2H), 3.34 (td, J = 12.6, 6.0 Hz, 1H), 3.20–3.01 (m, 2H), 2.74 (dd, J = 14.0, 5.4 Hz, 1H), 2.45–2.26 (m, 1H), 2.26–2.12 (m, 1H), 2.00 (dd, J = 19.3, 9.6 Hz, 1H), 1.90–1.83 (m, 1H), 1.35 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.5, 177.9, 170.9, 163.4, 161.8, 141.5, 135.7, 133.9, 133.3, 133.1, 133.0, 132.7, 132.6, 131.9, 131.9, 131.5, 131.5, 131.1, 130.4, 129.8, 129.6, 129.3, 127.7, 127.6, 127.4, 125.9, 124.3, 116.0, 115.8, 74.3, 71.6, 57.4, 54.6, 41.2, 38.8, 20.7, 17.8. LRMS (ESI, m/z): 722.0 [M + H]+. HRMS (ESI, m/z): calcd for C35H30Cl3FN3NiO3, 722.0511 [M + H]+; found 722.0517. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 18.447 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-3,5-diiodotyrosine Schiff Base Complex (6f): red solid (169 mg, yield 87%); mp 233–235 °C; [α]20D = –1774 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.08–9.05 (m, 1H), 8.47–8.42 (m, 1H), 7.63–7.59 (m, 2H), 7.59–7.55 (m, 1H), 7.52–7.46 (m, 1H), 7.37–7.32 (m, 2H), 7.29–7.27 (m, 2H), 7.20–7.15 (m, 1H), 6.79 (d, J = 7.4 Hz, 1H), 6.67 (d, J = 2.5 Hz, 1H), 5.83 (s, 1H), 4.14–4.10 (m, 1H), 3.57 (q, J = 13.8 Hz,2H), 3.48–3.38 (m, 1H), 3.23–3.16 (m, 1H), 2.94–2.87 (m, 1H), 2.73–2.65 (m, 1H), 2.56–2.46 (m, 1H), 2.31–2.19 (m, 1H), 2.10–1.96 (m, 2H), 1.38 (s, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.4, 177.6, 171.0, 153.4, 141.7, 140.6, 135.6, 134.0, 133.2, 133.1, 133.0, 132.6, 131.8, 131.1, 130.6, 129.8, 129.7, 129.4, 127.6, 127.5, 127.4, 125.8, 124.4, 82.8, 74.5, 71.2, 57.5, 54.6, 41.0, 38.0, 21.0, 17.9. LRMS (ESI, m/z): 969.8 [M − H]−. HRMS (ESI, m/z): calcd for C35H28Cl3I2N3NiO4, 969.8515 [M − H]−; found 969.8511. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 17.454 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-3-(1-naphthyl)alanine Schiff Base Complex (6g). Red solid (142 mg, yield 94%); mp 114–116 °C; [α]20D = −1812 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.09 (d, J = 2.0 Hz, 1H), 8.30 (d, J = 9.3 Hz, 1H), 7.78 (t, J = 8.0 Hz, 2H), 7.68 (dd, J = 8.2, 2.0 Hz, 1H), 7.57 (d, J = 8.6 Hz, 1H), 7.43–7.35 (m, 1H), 7.30 (dd, J = 15.6, 7.6 Hz, 4H), 7.23–7.11 (m, 3H), 7.08 (dd, J = 9.3, 2.6 Hz, 1H), 6.68 (t, J = 7.6 Hz, 1H), 6.44 (d, J = 2.6 Hz, 1H), 5.64 (d, J = 7.7 Hz, 1H), 4.41 (dd, J = 8.3, 4.3 Hz, 1H), 4.07 (dd, J = 14.2, 8.5 Hz, 1H), 3.79 (dd, J = 14.1, 4.3 Hz, 1H), 3.70 (d, J = 13.2 Hz, 1H), 3.61 (td, J = 13.1, 5.9 Hz, 1H), 3.46 (d, J = 13.2 Hz, 1H), 3.24 (t, J = 9.1 Hz, 1H), 2.83–2.53 (m, 1H), 2.39–2.22 (m, 1H), 2.12 (dd, J = 19.8, 9.6 Hz, 1H), 2.00 (dt, J = 15.0, 8.9 Hz, 1H), 1.40 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.3, 178.3, 170.5, 141.3, 135.8, 134.2, 133.8, 133.3, 133.0, 132.8, 132.6, 132.5, 132.3, 131.7, 131.2, 129.8, 129.6, 128.8, 128.7, 128.6, 128.6, 128.5, 127.7, 127.5, 127.4, 126.5, 126.1, 125.7, 125.6, 124.1, 123.4, 74.4, 71.6, 56.9, 55.1, 41.6, 40.2, 20.9, 17.9. LRMS (ESI, m/z): 754.0 [M + H]+. HRMS (ESI, m/z): calcd for C39H33Cl3N3NiO3, 754.0763 [M + H]+; found 754.0779. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 23.723 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-3-(3-benzothienyl)alanine Schiff Base Complex (6h). Red solid (148 mg, yield 98%); mp 127–129 °C; [α]20D = –1836 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.06 (d, J = 2.0 Hz, 1H), 8.34 (d, J = 9.3 Hz, 1H), 7.86 (d, J = 8.0 Hz, 1H), 7.57 (dd, J = 8.2, 2.0 Hz, 1H), 7.49 (t, J = 7.5 Hz, 1H), 7.45–7.34 (m, 2H), 7.30 (dd, J = 11.9, 6.2 Hz, 3H), 7.19–7.08 (m, 4H), 6.59 (d, J = 2.6 Hz, 1H), 6.48 (d, J = 7.5 Hz, 1H), 4.31 (t, J = 5.5 Hz, 1H), 3.71–3.59 (m, 1H), 3.51 (dd, J = 57.6, 9.8 Hz, 2H), 3.18 (dd, J = 14.5, 4.7 Hz, 1H), 3.11–2.97 (m, 2H), 2.12–1.90 (m, 3H), 1.81–1.69 (m, 1H), 1.30 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.3, 178.3, 170.8, 141.5, 140.6, 139.3, 135.7, 133.9, 133.2, 133.0, 132.9, 132.7, 131.1, 130.5, 130.1, 129.8, 129.3, 129.0, 127.5, 127.5, 125.9, 125.8, 124.8, 124.5, 124.3, 122.9, 122.1, 74.3, 70.9, 57.2, 54.8, 41.0, 34.0, 20.4, 17.8. LRMS (ESI, m/z): 760.0 [M + H]+. HRMS (ESI, m/z): calcd for C37H31Cl3N3NiO3S, 760.0326 [M + H]+; found 760.0333. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 27.103 min, tminor = not dound, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-3-(3-thienyl)alanine Schiff Base Complex (6i). Red solid (128 mg, yield 92%); mp 208–210 °C; [α]20D = −2419 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.09 (d, J = 2.1 Hz, 1H), 8.34 (d, J = 9.3 Hz, 1H), 7.63–7.49 (m, 3H), 7.46–7.39 (m, 1H), 7.38–7.28 (m, 3H), 7.13 (dd, J = 9.3, 2.6 Hz, 1H), 7.04 (d, J = 1.8 Hz, 1H), 6.86 (dd, J = 4.9, 1.2 Hz, 1H), 6.70 (d, J = 7.7 Hz, 1H), 6.62 (d, J = 2.6 Hz, 1H), 4.18 (t, J = 5.1 Hz, 1H), 3.63 (d, J = 13.1 Hz, 1H), 3.50 (dq, J = 11.9, 5.9 Hz, 2H), 3.25–3.05 (m, 2H), 2.77 (dd, J = 14.3, 5.4 Hz, 1H), 2.62–2.41 (m, 1H), 2.29–2.12 (m, 1H), 2.09–1.98 (m, 1H), 1.96–1.84 (m, 1H), 1.36 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.6, 178.3, 170.9, 141.5, 135.8, 135.7, 134.0, 133.4, 133.1, 133.0, 132.7, 132.6, 131.1, 130.3, 129.8, 129.6, 129.5, 129.3, 127.7, 127.4, 126.4, 125.8, 124.3, 124.3, 74.4, 71.3, 57.4, 54.8, 41.5, 34.1, 20.9, 17.9. LRMS (ESI, m/z): 710.0 [M + H]+. HRMS (ESI, m/z): calcd for C33H29Cl3N3NiO3S, 710.0168 [M + H]+; found 710.0175. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 16.027 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-2-(3-methoxyphenyl)glycine Schiff Base Complex (6j). Red solid (99 mg, yield 70%); mp 135–137 °C; [α]20D = –2183 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.18 (d, J = 1.6 Hz, 1H), 8.23 (d, J = 9.3 Hz, 1H), 7.79 (d, J = 8.2 Hz, 1H), 7.54 (t, J = 7.6 Hz, 1H), 7.39 (dd, J = 15.2, 7.2 Hz, 3H), 7.29 (d, J = 7.5 Hz, 1H), 7.22 (t, J = 7.9 Hz, 1H), 7.16 (dd, J = 9.3, 2.5 Hz, 1H), 7.11–7.02 (m, 2H), 6.80 (dd, J = 8.2, 2.3 Hz, 1H), 6.66 (d, J = 2.5 Hz, 1H), 6.14 (d, J = 7.9 Hz, 1H), 4.71 (s, 1H), 4.08–3.95 (m, 1H), 3.80 (d, J = 13.0 Hz, 1H), 3.71 (s, 3H), 3.61 (d, J = 13.0 Hz, 1H), 3.34 (t, J = 8.9 Hz, 1H), 3.14 (dd, J = 18.8, 9.9 Hz, 1H), 2.46 (dd, J = 14.1, 9.9 Hz, 1H), 2.19 (dd, J = 17.6, 7.9 Hz, 2H), 1.49 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.80, 177.4, 171.7, 159.8, 141.3, 139.2, 135.8, 134.0, 133.3, 133.3, 133.1, 132.6, 132.4, 131.2, 130.0, 129.9, 129.7, 129.0, 128.9, 127.7, 127.1, 126.7, 126.0, 124.9, 118.4, 113.8, 112.6, 74.7, 74.4, 57.3, 55.4, 55.3, 41.7, 21.2, 18.3. LRMS (ESI, m/z): 720.0 [M + H]+. HRMS (ESI, m/z): calcd for C35H31Cl3N3NiO4, 720.0554 [M + H]+; found 720.0574. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 16.851 min, tminor = 18.454, dr = 98:2.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-2-(3-bromophenyl)glycine Schiff Base Complex (6k). Red solid (134 mg, yield 87%); mp 102–104 °C; [α]20D = –2069 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.18 (d, J = 2.1 Hz, 1H), 8.22 (d, J = 9.3 Hz, 1H), 8.15 (s, 1H), 7.80 (dd, J = 8.2, 2.2 Hz, 1H), 7.57 (t, J = 7.6 Hz, 1H), 7.50–7.38 (m, 3H), 7.32 (dd, J = 18.2, 7.8 Hz, 2H), 7.20–7.08 (m, 3H), 6.67 (d, J = 2.5 Hz, 1H), 6.13 (d, J = 7.8 Hz, 1H), 4.71 (s, 1H), 4.04 (t, J = 10.9 Hz, 1H), 3.72 (dd, J = 78.9, 13.1 Hz, 2H), 3.41–3.29 (m, 1H), 3.14 (dd, J = 21.0, 13.1 Hz, 1H), 2.50 (dd, J = 13.7, 10.0 Hz, 1H), 2.30–2.04 (m, 2H), 1.50 (s, 3H), 13C-NMR (125 MHz, CDCl3) δ 180.8, 176.8, 172.1, 141.4, 139.9, 135.8, 134.0, 133.4, 132.9, 132.9, 132.4, 131.6, 131.3, 130.3, 130.2, 130.0, 129.3, 129.3, 129.1, 127.6, 127.0, 126.7, 126.1, 125.3, 125.0, 122.9, 74.5, 74.1, 57.3, 55.3, 41.8, 21.2, 18.3. LRMS (ESI, m/z): 767.9 [M + H]+. HRMS (ESI, m/z): calcd for C34H28BrCl3N3NiO3, 767.9557 [M + H]+; found 767.9561. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 25.867 min, tminor = 29.203, dr = 97:3.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-2-cyclobutylglycine Schiff Base Complex (6l). Red solid (121 mg, yield 91%); mp 266–269 °C; [α]20D = –2560 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.14 (s, 1H), 8.24 (d, J = 9.3 Hz, 1H), 7.74–7.69 (m, 1H), 7.58–7.49 (m, 2H), 7.49–7.43 (m, 1H), 7.34–7.27 (m, 2H), 7.10 (dd, J = 9.3, 2.5 Hz, 1H), 6.88 (d, J = 7.4 Hz, 1H), 6.60 (d, J = 2.5 Hz, 1H), 4.21–4.05 (m, 1H), 3.84–3.75 (m, 2H), 3.52 (d, J = 13.0 Hz, 1H), 3.45–3.38 (m, 1H), 3.17–3.00 (m, 1H), 2.78–2.60 (m, 1H), 2.59–2.34 (m, 3H), 2.31–2.14 (m, 2H), 2.11–2.09 (m, 1H), 1.91–1.77 (m, 1H), 1.75–1.62 (m, 2H), 1.46 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.1, 177.0, 169.4, 140.7, 135.6, 133.7, 133.1, 132.9, 132.8, 132.3, 132.1, 130.9, 130.0, 129.6, 129.2, 128.9, 127.9, 127.8, 127.5, 125.6, 124.0, 74.3, 73.8, 57.0, 54.8, 41.7, 40.5, 25.8, 25.4, 20.5, 17.9, 17.3. LRMS (ESI, m/z): 666.0 [M − H]−. HRMS (ESI, m/z): calcd for C32H29Cl3N3NiO3, 666.0633 [M − H]−; found 666.0632. The dr was determined by LC-MS using Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 16.198 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-2-amino-4,4,4-trifluorobutyric acid Schiff Base Complex (6m). Red solid (129 mg, yield 93%); mp 274–276 °C; [α]20D = –3150 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.16 (d, J = 1.8 Hz, 1H), 8.35 (d, J = 9.3 Hz, 1H), 7.68–7.47 (m, 4H), 7.36 (d, J = 8.1 Hz, 1H), 7.30 (d, J = 6.8 Hz, 1H), 7.16 (dd, J = 9.3, 2.5 Hz, 1H), 6.90 (d, J = 7.4 Hz, 1H), 6.60 (d, J = 2.5 Hz, 1H), 4.22–4.10 (m, 2H), 3.74–3.49 (m, 2H), 3.38 (t, J = 8.7 Hz, 1H), 3.09–2.80 (m, 1H), 2.63–2.46 (m, 1H), 2.42 (dd, J = 13.7, 9.8 Hz, 1H), 2.23–2.09 (m, 2H), 2.06–1.90 (m, 1H), 1.44 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.5, 177.2, 172.4, 141.9, 135.8, 134.0, 133.2, 133.2, 133.2, 133.1, 132.5, 131.3, 130.8, 129.9, 129.8, 129.7, 127.6, 127.6, 126.8, 125.8, 124.6, 74.9, 64.5, 57.8, 54.8, 41.2, 35.6 (d, J = 29.2 Hz), 20.3, 17.4. LRMS (ESI, m/z): 696.0 [M + H]+. HRMS (ESI, m/z): calcd for C30H26Cl3F3N3NiO3, 696.0164 [M + H]+; found 696.0171. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 13.464 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-norvaline Schiff Base Complex (6n). Red solid (118 mg, yield 90%); mp 227–228 °C; [α]20D = –2430 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.11 (d, J = 2.0 Hz, 1H), 8.21 (d, J = 9.3 Hz, 1H), 7.73 (dd, J = 8.2, 2.0 Hz, 1H), 7.61–7.49 (m, 2H), 7.46 (t, J = 7.4 Hz, 1H), 7.31 (dd, J = 15.9, 7.6 Hz, 2H), 7.12 (dd, J = 9.3, 2.6 Hz, 1H), 6.86 (d, J = 7.5 Hz, 1H), 6.60 (d, J = 2.6 Hz, 1H), 4.11–3.98 (m, 1H), 3.87 (dd, J = 7.6, 3.6 Hz, 1H), 3.65 (dd, J = 82.3, 13.0 Hz, 2H), 3.38 (t, J = 8.8 Hz, 1H), 3.25–3.02 (m, 1H), 2.48–2.37 (m, 1H), 2.27–2.14 (m, 2H), 2.13–1.97 (m, 1H), 1.84 (dt, J = 13.1, 8.4 Hz, 1H), 1.69 (dd, J = 13.1, 6.3 Hz, 1H), 1.59–1.49 (m, 1H), 1.45 (s, 3H), 0.79 (t, J = 7.2 Hz, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 179.0, 169.8, 140.9, 135.8, 133.9, 133.3, 133.1, 133.1, 132.4, 131.2, 130.3, 129.9, 129.5, 129.3, 128.1, 127.5, 127.5, 125.9, 124.5, 74.5, 70.5, 57.2, 55.0, 41.6, 37.4, 21.0, 18.4, 17.9, 13.9. LRMS (ESI, m/z): 656.1 [M + H]+. HRMS (ESI, m/z): calcd for C31H31Cl3N3NiO3, 656.0604 [M + H]+; found 656.0601. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 14.850 min, tminor = 17.260, dr = 98:2.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-valine Schiff Base Complex (6o). Red solid (126 mg, yield 96%); mp 224–226 °C; [α]20D = −2566 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.18 (d, J = 2.1 Hz, 1H), 8.29 (d, J = 9.3 Hz, 1H), 7.68–7.64 (m, 1H), 7.60–7.50 (m, 2H), 7.49–7.43 (m, 1H), 7.35–7.30 (m, 2H), 7.12 (dd, J = 9.3, 2.6 Hz, 1H), 6.88–6.84 (m, 1H), 6.62 (d, J = 2.6 Hz, 1H), 4.13–4.01 (m, 1H), 3.82–3.76 (m, 2H), 3.58–3.52 (m, 1H), 3.45–3.38 (m, 1H), 3.07–2.87 (m, 1H), 2.49–2.39 (m, 1H), 2.25–2.10 (m, 2H), 1.86 (d, J = 6.6 Hz, 3H), 1.81–1.70 (m, 1H), 1.46 (s, 3H), 0.75 (d, J = 6.8 Hz, 3H). 13C-NMR (150 MHz, CDCl3) δ 180.2, 177.1, 170.1, 140.8, 135.7, 133.8, 133.0, 133.0, 132.8, 132.3, 132.2, 130.9, 130.0, 129.6, 129.3, 128.9, 127.7, 127.6, 127.3, 125.6, 124.0, 75.4, 74.2, 57.1, 54.7, 41.5, 34.2, 20.3, 19.7, 17.6, 17.5. LRMS (ESI, m/z): 656.0 [M − H]−. HRMS (ESI, m/z): calcd for C31H29Cl3N3NiO3, 656.0633 [M − H]−; found 656.0629. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 14.706 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-leucine Schiff Base Complex (6p). Red solid (127 mg, yield 95%); mp 126–127 °C; [α]20D = −1898 (c = 1, CHCl3); 1H-NMR (500 MHz, CDCl3) δ 9.11 (d, J = 2.0 Hz, 1H), 8.15 (d, J = 9.3 Hz, 1H), 7.78 (dd, J = 8.2, 2.1 Hz, 1H), 7.60–7.49 (m, 2H), 7.46 (td, J = 7.5, 1.0 Hz, 1H), 7.32 (t, J = 6.8 Hz, 2H), 7.10 (dd, J = 9.3, 2.6 Hz, 1H), 6.87 (d, J = 7.6 Hz, 1H), 6.60 (d, J = 2.6 Hz, 1H), 4.06–3.98 (m, 1H), 3.83 (dd, J = 10.5, 4.1 Hz, 1H), 3.80 (d, J = 13.2 Hz, 1H), 3.53 (d, J = 13.1 Hz, 1H), 3.39 (t, J = 8.8 Hz, 1H), 3.32–3.15 (m, 1H), 2.49–2.31 (m, 2H), 2.29–2.16 (m, 2H), 1.99–1.81 (m, 1H), 1.45 (s, 3H), 1.38–1.32 (m, 1H), 0.81 (d, J = 6.7 Hz, 3H), 0.38 (d, J = 6.5 Hz, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 178.8, 169.2, 140.8, 135.8, 133.9, 133.3, 133.1, 132.9, 132.2, 131.1, 130.3, 129.9, 129.4, 129.3, 128.1, 127.8, 127.6, 125.9, 124.5, 74.4, 69.3, 56.9, 55.3, 46.2, 41.8, 24.5, 23.9, 21.4, 21.0, 18.1. LRMS (ESI, m/z): 669.9 [M + H]+. HRMS (ESI, m/z): calcd for C32H33Cl3N3NiO3, 670.0935 [M + H]+; found 670.0935. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 19.979 min, tminor = not found, dr > 99:1.
Nickel(II)-(R)-N-(2-benzoyl-4-chlorophenyl)-1-(3,4-dichlorobenzyl)-2-methylpyrrolidine-2-carboxamide/(R)-methionine Schiff Base Complex (6q). Red solid (120 mg, yield 88%); mp 123–125 °C; [α]20D = −2202 (c = 1, CHCl3); 1H-NMR (400 MHz, CDCl3) δ 9.11 (d, J = 1.5 Hz, 1H), 8.21 (d, J = 9.3 Hz, 1H), 7.72 (d, J = 6.7 Hz, 1H), 7.60–7.51 (m, 2H), 7.47 (t, J = 7.4 Hz, 1H), 7.32 (dd, J = 14.6, 7.6 Hz, 2H), 7.13 (dd, J = 9.3, 2.5 Hz, 1H), 6.88 (d, J = 7.5 Hz, 1H), 6.60 (d, J = 2.5 Hz, 1H), 4.09–3.96 (m, 1H), 3.93 (dd, J = 8.3, 3.6 Hz, 1H), 3.66 (dd, J = 74.8, 13.0 Hz, 2H), 3.37 (t, J = 8.9 Hz, 1H), 3.17 (dd, J = 16.5, 10.3 Hz, 1H), 3.09–3.01 (m, 1H), 2.67–2.53 (m, 1H), 2.49–2.37 (m, 1H), 2.19 (dd, J = 15.7, 6.7 Hz, 3H), 1.97 (s, 3H), 1.91–1.77 (m, 1H), 1.45 (s, 3H). 13C-NMR (125 MHz, CDCl3) δ 180.4, 178.3, 170.3, 141.1, 135.7, 133.9, 133.3, 133.2, 132.9, 132.6, 132.4, 131.2, 130.4, 129.9, 129.6, 129.4, 127.9, 127.5, 127.4, 125.9, 124.6, 74.5, 69.8, 57.3, 55.0, 41.6, 35.1, 29.8, 21.3, 18.0, 15.8. LRMS (ESI, m/z): 688.0 [M − H]−. HRMS (ESI, m/z): calcd for C31H31Cl3N3NiO3S, 688.0324 [M − H]−; found 688.0339. The dr was determined by LC-MS using an Eclipse XDB-C18 column (250 × 4.6 mm, 5 µm) (CH3CN/H2O = 65:35, flow rate 1.0 mL/min, λ = 254 nm), tmajor = 13.870 min, tminor = 14.997, dr = 98:2.
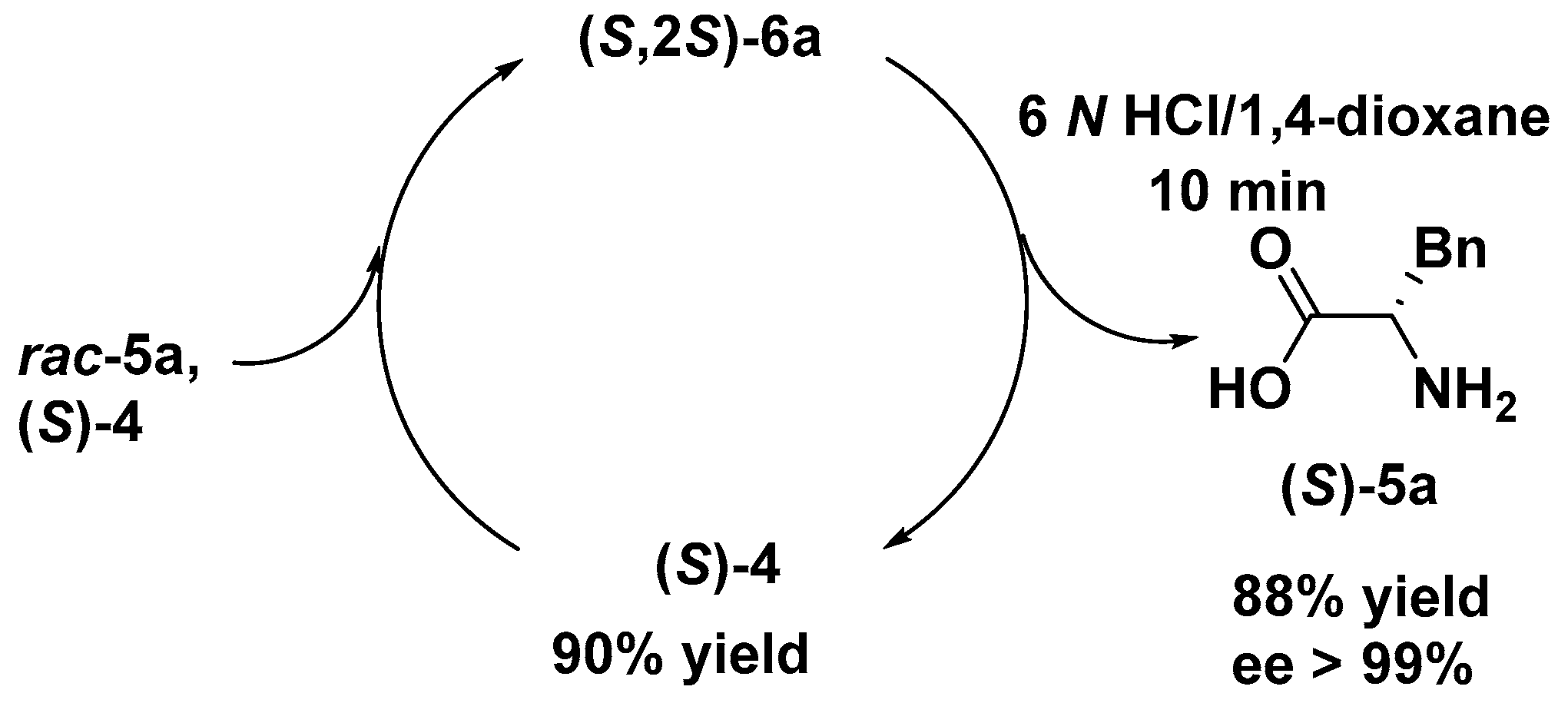
(S)-Phenylalanine (5a). White solid (16.5 mg, yield 88%); mp 273–275 °C; [α]20D = – 38 (c = 1, MeOH); 1H-NMR (500 MHz, D2O) δ 7.41 (m, J = 14.6, 7.2 Hz, 3H), 7.33 (d, J = 7.1 Hz, 2H), 4.00 (dd, J = 7.9, 5.2 Hz, 1H), 3.30 (dd, J = 14.5, 5.2 Hz, 1H), 3.13 (dd, J = 14.5, 8.0 Hz, 1H), 2.00 (s, 1H), 1.96 (s, 2H). 13C-NMR (125 MHz, D2O) δ 173.4, 134.6, 128.8, 128.6, 127.2, 55.5, 35.8, 22.1, 20.7. LRMS (ESI, m/z): 166.0 [M + H]+. HRMS (ESI, m/z): calcd for C9H11NO2, 166.0863 [M + H]+; found 166.0865. The ee was determined by HPLC using an Astec CHIROBIOTIC™ T chiral HPLC column (4.6 mm × 25 cm, 5 μm) (MeOH/H2O = 90/10, λ = 210 nm, 1 mL/min). tS = 7.472 min, tR = not found, ee > 99 %.
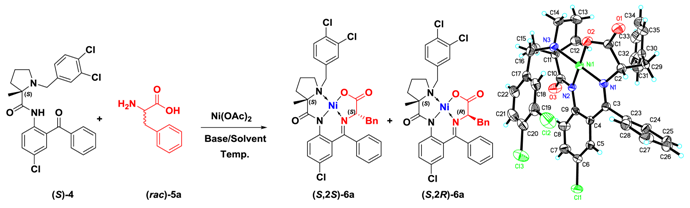
X-ray Single Crystal Structure Analysis of (
S,2
S)-
6a: (
S,2
S)-
6a (100 mg) was dissolved in DCM (10 mL) and crystals suitable for X-ray single crystal structure determination were obtained after all the solvent had evaporated at room temperature. The X-ray structure of (
S,2
S)
-6a was resolved at T = 293(2) K: C
35H
30Cl
3N
3NiO
3,
Mr = 705.68, monoclinic. Space group
P2 (1), a = 10.1494 (12) Å, b = 15.7644 (19) Å, c = 22.968 (3) Å, α = 90°, β = 90°, γ = 90°,
V = 3674.9 (8) Å
3,
Z = 4. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via
www.ccdc.cam.ac.uk/data_request/cif, the CCDC number is 1472366.