A New Approach for the Prevention and Treatment of Cardiovascular Disorders. Molecular Hydrogen Significantly Reduces the Effects of Oxidative Stress
Abstract
1. Introduction
2. ROS and Cardiovascular Diseases
2.1. Radiation and Radiation-Induced Heart Disease
Methods of Radiation Protection
2.2. Ischemia and Reperfusion Injury
2.3. Heart Transplantation
Antioxidants for Graft Preservation
3. New Approach for Prevention and Treatment of Cardiovascular Disorders
3.1. Medical Gases and Hydrogen on the Cardiovascular System
3.2. Molecular Hydrogen
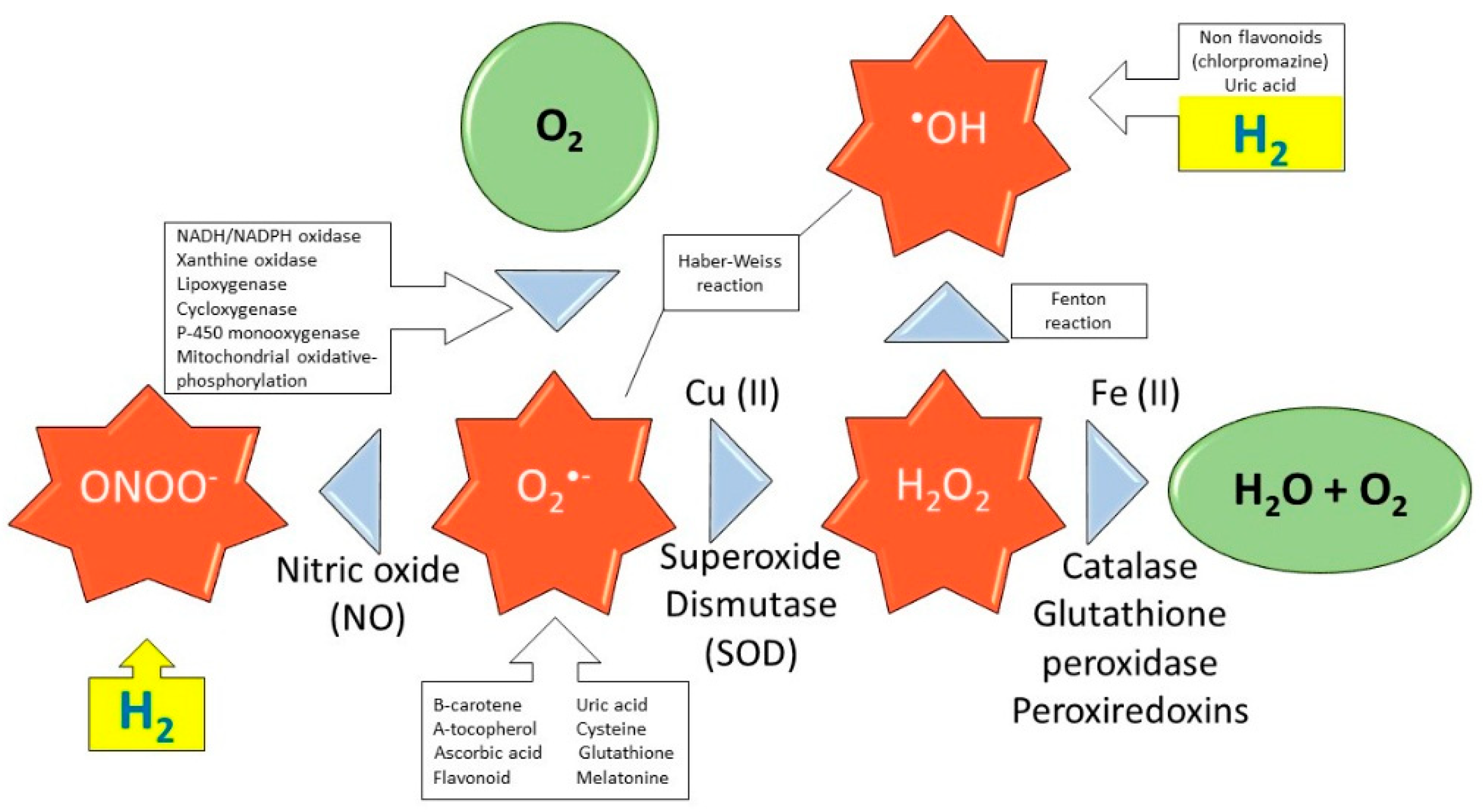
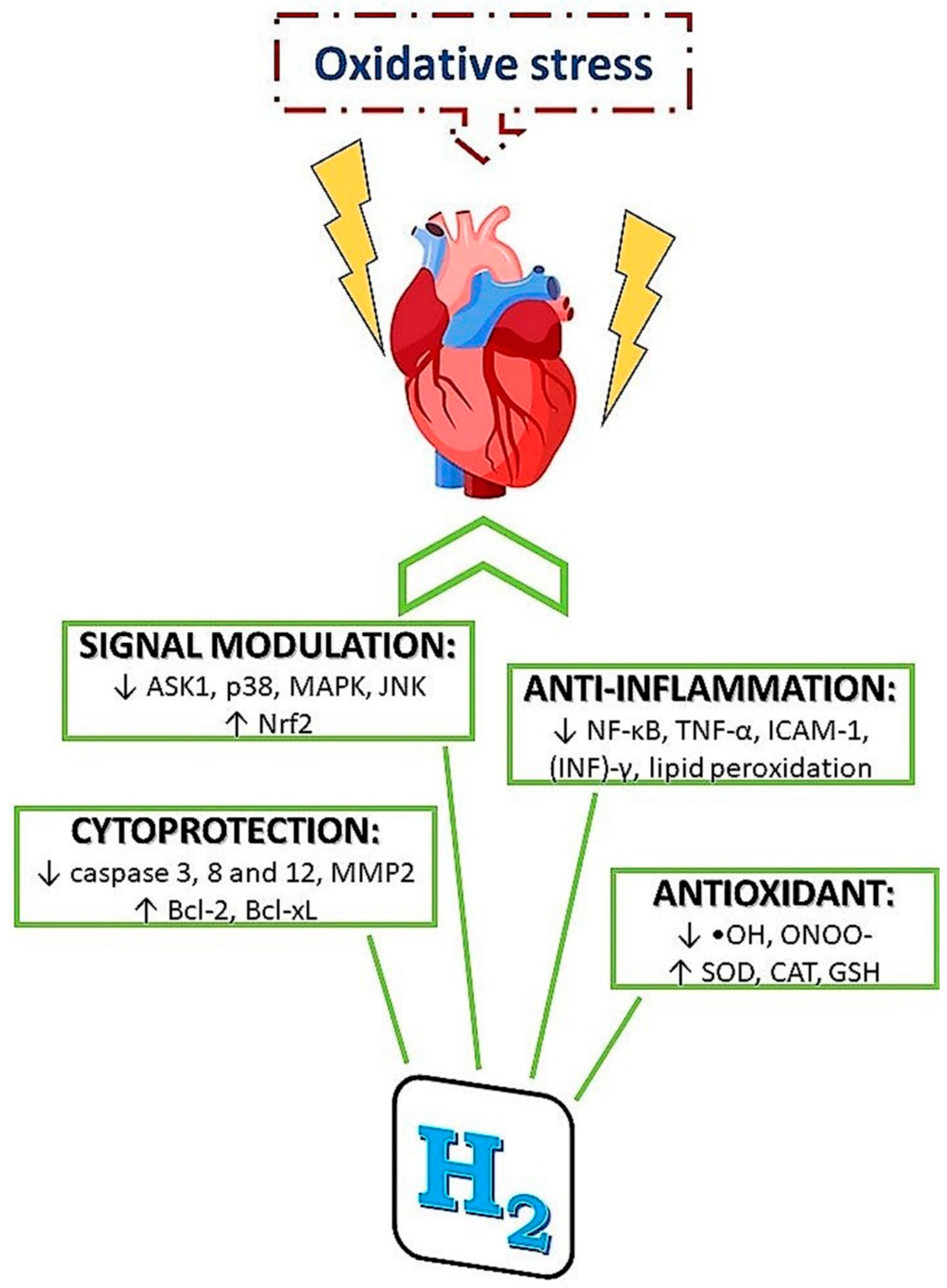
3.2.1. Mechanisms of H2 Action
- (1)
- specific scavenging activity of hydroxyl radicals and peroxynitrite
- (2)
- reduction of inflammatory reactions
- (3)
- modulation of signal transduction
- (4)
- alterations of gene expressions
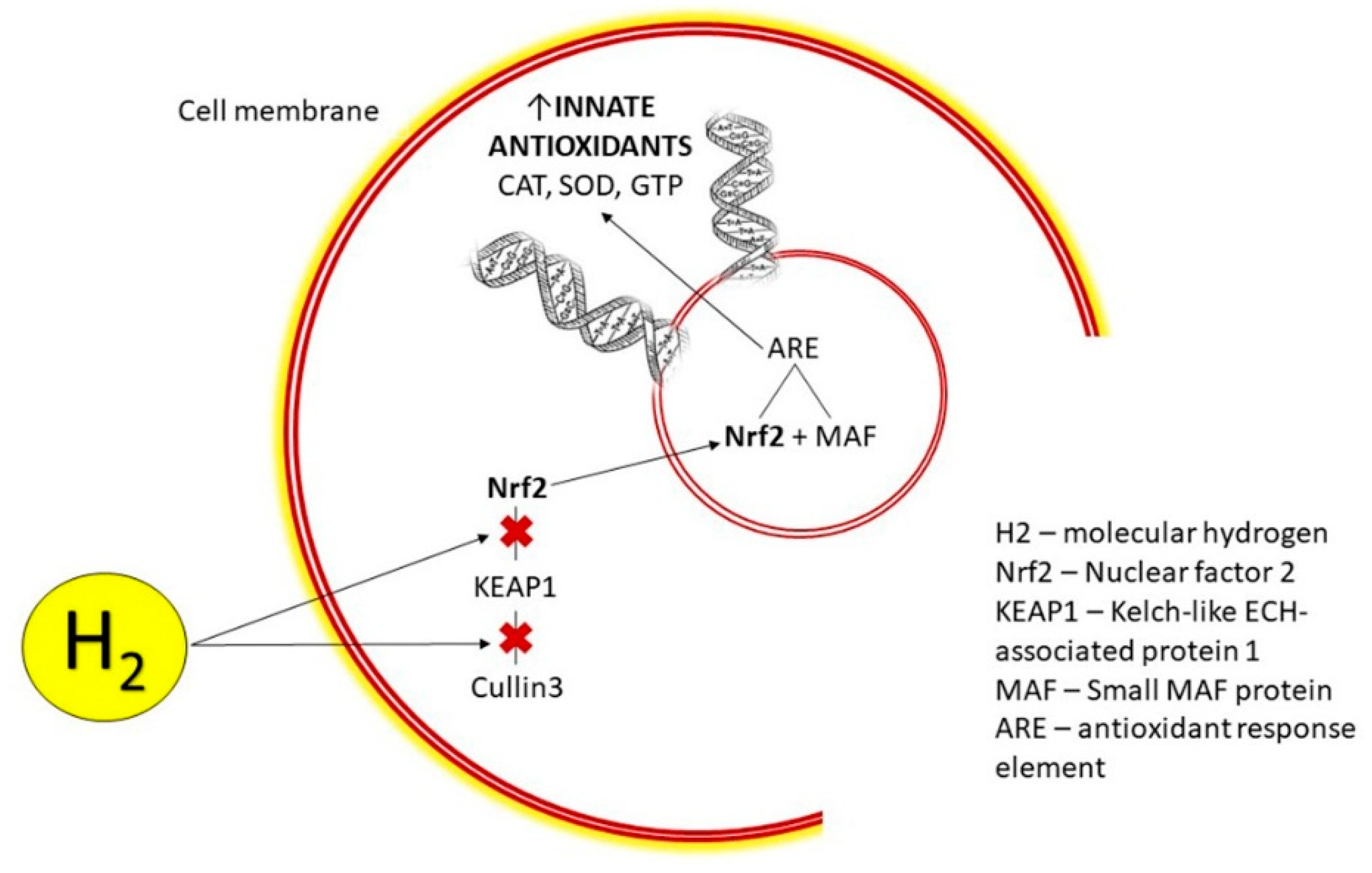
3.2.2. H2 Modulates Nrf2 Pathway
3.2.3. H2 Induces Hormesis
3.2.4. H2 and miRNAs
4. Potential Usage of H2 Against Diseases
4.1. Therapeutic and Protective Function of H2 in Chemotherapy and Radiotherapy
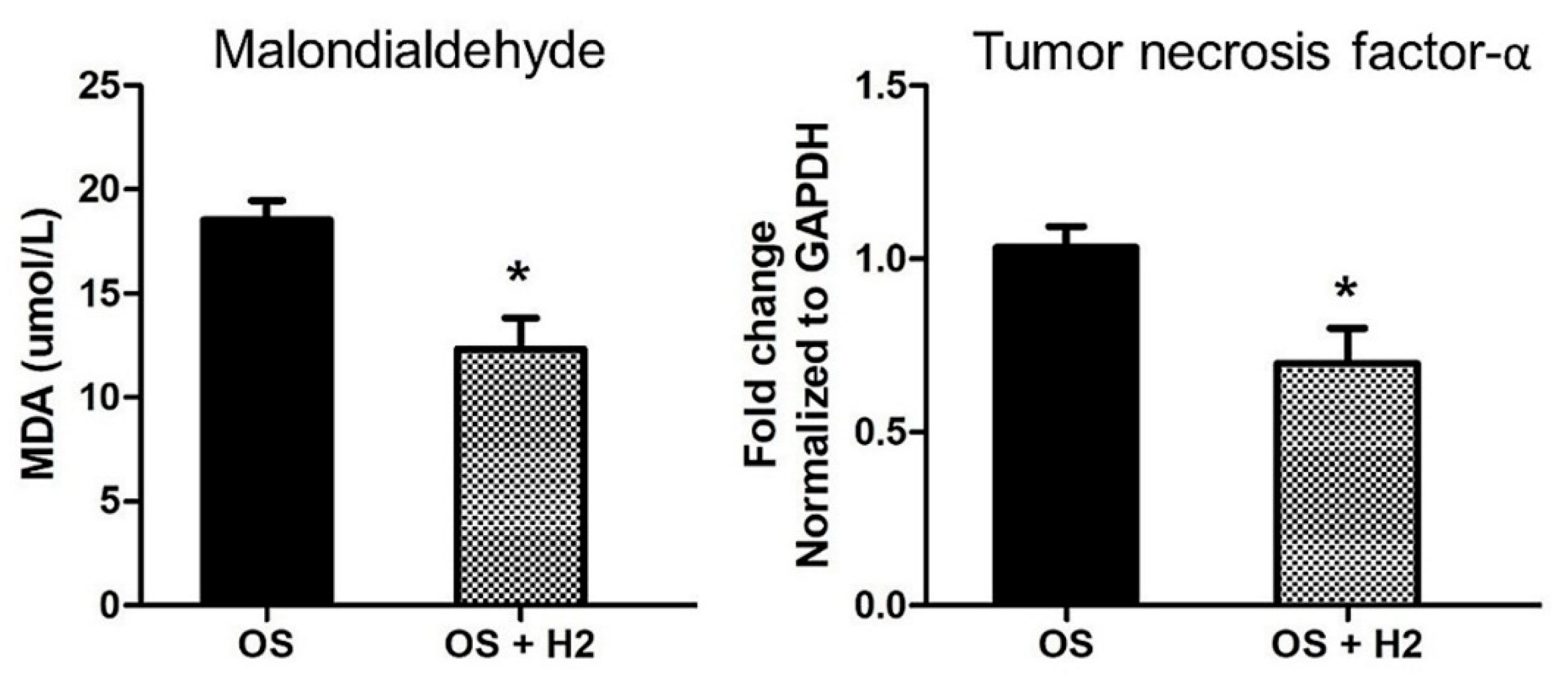
4.2. H2 Benefits in I/R Injury
4.3. H2 for Graft Preservation
5. Conclusions
Funding
Conflicts of Interest
References
- Slezak, J.; Kura, B.; Babal, P.; Barancik, M.; Ferko, M.; Frimmel, K.; Kalocayova, B.; Kukreja, R.C.; Lazou, A.; Mezesova, L.; et al. Potential markers and metabolic processes involved in the mechanism of radiation-induced heart injury. Can. J. Physiol. Pharmacol. 2017, 95, 1190–1203. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. Available online: https://ec.europa.eu/ (accessed on 14 August 2016).
- Kura, B.; Babal, P.; Slezak, J. Implication of microRNAs in the development and potential treatment of radiation-induced heart disease. Can. J. Physiol. Pharmacol. 2017, 95, 1236–1244. [Google Scholar] [CrossRef] [PubMed]
- Slezak, J.; Kura, B.; Ravingerová, T.; Tribulova, N.; Okruhlicova, L.; Barancik, M. Mechanisms of cardiac radiation injury and potential preventive approaches. Can. J. Physiol. Pharmacol. 2015, 93, 737–753. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.R.; Fajardo, L.F. Radiation-induced heart disease: An update. Prog. Cardiovasc. Dis. 1984, 27, 173–194. [Google Scholar] [CrossRef]
- Gillani, S.; Cao, J.; Suzuki, T.; Hak, D.J. The effect of ischemia reperfusion injury on skeletal muscle. Injury 2012, 43, 670–675. [Google Scholar] [CrossRef]
- Ohta, S. Molecular hydrogen as a preventive and therapeutic medical gas: Initiation, development and potential of hydrogen medicine. Pharmacol. Ther. 2014, 144, 1–11. [Google Scholar] [CrossRef]
- Kura, B.; Bagchi, A.K.; Singal, P.K.; Barancik, M.; LeBaron, T.W.; Valachova, K.; Šoltés, L.; Slezák, J. Molecular hydrogen: Potential in mitigating oxidative-stress-induced radiation injury. Can. J. Physiol. Pharmacol. 2019, 97, 287–292. [Google Scholar] [CrossRef]
- Slezák, J.; Kura, B.; Frimmel, K.; Zálešák, M.; Ravingerová, T.; Viczenczová, C.; Okruhlicová, Ľ.; Tribulová, N. Preventive and therapeutic application of molecular hydrogen in situations with excessive production of free radicals. Physiol. Res. 2016, 65 (Suppl. 1), S11–S28. [Google Scholar]
- Ichihara, M.; Sobue, S.; Ito, M.; Ito, M.; Hirayama, M.; Ohno, K. Beneficial biological effects and the underlying mechanisms of molecular hydrogen-Comprehensive review of 321 original articles. Med. Gas Res. 2015, 5, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Ohta, S. Recent Progress Toward Hydrogen Medicine: Potential of Molecular Hydrogen for Preventive and Therapeutic Applications. Curr. Pharm. Des. 2011, 17, 2241–2252. [Google Scholar] [CrossRef]
- Ďuračková, Z. Some current insights into oxidative stress. Physiol. Res. 2010, 8408, 459–469. [Google Scholar]
- Moris, D.; Spartalis, M.; Spartalis, E.; Karachaliou, G.-S.; Karaolanis, G.I.; Tsourouflis, G.; Tsilimigras, D.I.; Tzatzaki, E.; Theocharis, S. The role of reactive oxygen species in the pathophysiology of cardiovascular diseases and the clinical significance of myocardial redox. Ann. Transl. Med. 2017, 5, 326. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Kajiyama, S.; Amano, A.; Kondo, Y.; Sasaki, T.; Handa, S.; Takahashi, R.; Fukui, M.; Hasegawa, G.; Nakamura, N.; et al. Hydrogen-rich pure water prevents superoxide formation in brain slices of vitamin C-depleted SMP30/GNL knockout mice. Biochem. Biophys. Res. Commun. 2008, 375, 346–350. [Google Scholar] [CrossRef]
- Jabs, T. Reactive oxygen intermediates as mediators of programmed cell death in plants and animals. Biochem. Pharmacol. 1999, 57, 231–245. [Google Scholar] [CrossRef]
- Roos, W.P.; Thomas, A.D.; Kaina, B. DNA damage and the balance between survival and death in cancer biology. Nat. Rev. Cancer 2016, 16, 20–33. [Google Scholar] [CrossRef]
- Zhang, G.; Miura, Y.; Yagasaki, K. Suppression of adhesion and invasion of hepatoma cells in culture by tea compounds through antioxidative activity. Cancer Lett. 2000, 159, 169–173. [Google Scholar] [CrossRef]
- Fridovich, I. The biology of oxygen radicals. Science 1978, 201, 875–880. [Google Scholar] [CrossRef]
- Endo, J.; Sano, M.; Katayama, T.; Hishiki, T.; Shinmura, K.; Morizane, S.; Matsuhashi, T.; Katsumata, Y.; Zhang, Y.; Ito, H.; et al. Metabolic remodeling induced by mitochondrial aldehyde stress stimulates tolerance to oxidative stress in the heart. Circ. Res. 2009, 105, 1118–1127. [Google Scholar] [CrossRef]
- Jazwa, A.; Cuadrado, A. Targeting Heme Oxygenase-1 for Neuroprotection and Neuroinflammation in Neurodegenerative Diseases. Curr. Drug Targets 2010, 11, 1517–1531. [Google Scholar] [CrossRef]
- Hakim, J. Reactive Oxygen Species and Inflammation. Comptes Rendus des séances la Soc. Biol. ses Fil. 1993, 187, 286–295. [Google Scholar]
- Poderoso, J.J.; Carreras, M.C.; Lisdero, C.; Riobó, N.; Schöpfer, F.; Boveris, A. Nitric oxide inhibits electron transfer and increases superoxide radical production in rat heart mitochondria and submitochondrial particles. Arch. Biochem. Biophys. 1996, 328, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.D.; Ridnour, L.A.; Isenberg, J.S.; Flores-Santana, W.; Switzer, C.H.; Donzelli, S.; Hussain, P.; Vecoli, C.; Paolocci, N.; Ambs, S.; et al. The chemical biology of nitric oxide: Implications in cellular signaling. Free Radic. Biol. Med. 2008, 45, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T. Signal transduction by reactive oxygen species. J. Cell Biol. 2011, 194, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Cao, F.; Cui, J.; Huang, Y.; Zhou, X.; Liu, S.; Cai, J. Radioprotective effect of hydrogen in cultured cells and mice. Free Radic. Res. 2010, 44, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Kirsch, D.G.; Santiago, P.M.; Di Tomaso, E.; Sullivan, J.M.; Hou, W.S.; Dayton, T.; Jeffords, L.B.; Sodha, P.; Mercer, K.L.; Cohen, R.; et al. P53 controls radiation-induced gastrointestinal syndrome in mice independent of apoptosis. Science 2010, 327, 593–596. [Google Scholar] [CrossRef]
- Ward, J.F. DNA Damage Produced by Ionizing Radiation in Mammalian Cells: Identities, Mechanisms of Formation, and Reparability. Prog. Nucleic Acid Res. Mol. Biol. 1988, 35, 95–125. [Google Scholar]
- Chuai, Y.; Qian, L.; Sun, X.; Cai, J. Molecular hydrogen and radiation protection. Free Radic. Res. 2012, 46, 1061–1067. [Google Scholar] [CrossRef]
- Kura, B.; Yin, C.; Frimmel, K.; Krizak, J.; Okruhlicova, L.; Kukreja, R.C.; Slezak, J. Changes of microRNA-1, -15b and -21 levels in irradiated rat hearts after treatment with potentially radioprotective drugs. Physiol. Res. 2016, 65 (Suppl. 1), S129–S137. [Google Scholar]
- Kasai, H. Analysis of a form of oxidative DNA damage, 8-hydroxy-2X -deoxyguanosine, as a marker of cellular oxidative stress during carcinogenesis. Mutat. Res. 1997, 387, 147–163. [Google Scholar] [CrossRef]
- Floyd, R.A. Tine role off 8-hydroxyguanine to carcinogenesis. Carcinogenesis 1990, 11, 1447–1450. [Google Scholar] [CrossRef]
- Bansal, D.; Bhalla, A.; Bhasin, D.; Pandhi, P.; Sharma, N.; Rana, S.; Malhotra, S. Safety and efficacy of vitamin-based antioxidant therapy in patients with severe acute pancreatitis: A randomized controlled trial. Saudi J. Gastroenterol. 2011, 17, 174–179. [Google Scholar]
- Fan, X. Ionizing radiation induces formation of malondialdehyde, formaldehyde, and acetaldehyde from carbohydrates and organic acid. J. Agric. Food Chem. 2003, 51, 5946–5949. [Google Scholar] [CrossRef]
- Dubner, D.; Gisone, P.; Jaitovich, I.; Perez, M. Free radicals production and estimation of oxidative stress related to γ irradiation. Biol. Trace Elem. Res. 1995, 47, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.P.; Sonwalkar, N. Structural Changes in Plasma Membranes Prepared from Irradiated Chinese Hamster V79 Cells as Revealed by Raman Spectroscopy. Radiat. Res. 1991, 126, 27–35. [Google Scholar] [CrossRef]
- Giusti, A.M.; Raimondi, M.; Ravagnan, G.; Sapora, O.; Parasassi, T. Human cell membrane oxidative damage induced by single and fractionated doses of ionizing radiation: A fluorescence spectroscopy study. Int. J. Radiat. Biol. 1998, 74, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Quarmby, S.; Hunter, R.D.; Kumar, S. Irradiation induced expression of CD31, ICAM-1 and VCAM-1 in human microvascular endothelial cells. Anticancer Res. 2000, 20, 3375–3381. [Google Scholar] [PubMed]
- Stewart, F.A.; Hoving, S.; Russell, N.S. Vascular damage as an underlying mechanism of cardiac and cerebral toxicity in irradiated cancer patients. Radiat. Res. 2010, 174, 865–869. [Google Scholar] [CrossRef]
- Te Poele, J.A.; Van Kleef, E.M.; Van Der Wal, A.F.; Dewit, L.G.H.; Stewart, F.A. Radiation-induced glomerular thrombus formation and nephropathy are not prevented by the ADP receptor antagonist clopidogrel. Int. J. Radiat. Oncol. Biol. Phys. 2001, 50, 1332–1338. [Google Scholar] [CrossRef]
- Wang, J.; Zheng, H.; Ou, X.; Fink, L.M.; Hauer-Jensen, M. Deficiency of microvascular thrombomodulin and up-regulation of protease-activated receptor-1 in irradiated rat intestine: Possible link between endothelial dysfunction and chronic radiation fibrosis. Am. J. Pathol. 2002, 160, 2063–2072. [Google Scholar] [CrossRef]
- Adams, M.J.; Hardenbergh, P.H.; Constine, L.S.; Lipshultz, S.E. Radiation-associated cardiovascular disease. Crit. Rev. Oncol. Hematol. 2003, 45, 55–75. [Google Scholar] [CrossRef]
- Stewart, J.R.; Fajardo, L.F.; Gillette, S.M.; Constine, L.S. Radiation injury to the heart. Int. J. Radiat. Oncol. Biol. Phys. 1995, 31, 1205–1211. [Google Scholar] [CrossRef]
- Lee, C.K.K.; Aeppli, D.; Nierengarten, M.E. The need for long-term surveillance for patients treated with curative radiotherapy for Hodgkin’s disease: University of Minnesota experience. Int. J. Radiat. Oncol. Biol. Phys. 2000, 48, 169–179. [Google Scholar] [CrossRef]
- Cuomo, J.R.; Sharma, G.K.; Conger, P.D.; Weintraub, N.L. Novel concepts in radiation-induced cardiovascular disease. World J. Cardiol. 2016, 8, 504–519. [Google Scholar] [CrossRef]
- Beyzadeoglu, M.; Oysul, K.; Dirican, B.; Arpaci, F.; Balkan, A.; Surenkok, S.; Pak, Y. Effect of Dose-Rate and Lung Dose in Total Body Irradiation on Interstitial Pneumonitis after Bone Marrow Transplantation. Tohoku J. Exp. Med. 2004, 202, 255–263. [Google Scholar] [CrossRef]
- Sampath, S.; Schultheiss, T.E.; Wong, J. Dose response and factors related to interstitial pneumonitis after bone marrow transplant. Int. J. Radiat. Oncol. Biol. Phys. 2005, 63, 876–884. [Google Scholar] [CrossRef]
- Qian, L.; Shen, J.; Chuai, Y.; Cai, J. Hydrogen as a new class of radioprotective agent. Int. J. Biol. Sci. 2013, 9, 887–894. [Google Scholar] [CrossRef]
- Vijayalaxmi; Reiter, R.J.; Tan, D.X.; Herman, T.S.; Thomas, C.R. Melatonin as a radioprotective agent: A review. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 639–653. [Google Scholar] [CrossRef]
- Aisin, J.R.M. Bacq and A lexander Aw ard Lecture Chem ical radioprotection: Past, present and future prospects. Int. J. Radiat. Biol. 1998, 73, 443–450. [Google Scholar]
- Citrin, D.; Cotrim, A.P.; Hyodo, F.; Baum, B.J.; Krishna, M.C.; Mitchell, J.B. Radioprotectors and Mitigators of Radiation-Induced Normal Tissue Injury. Oncologist 2010, 15, 360–371. [Google Scholar] [CrossRef]
- Noda, M.; Fujita, K.; Lee, C.-H.; Yoshioka, T. The Principle and the Potential Approach to ROS-dependent Cytotoxicity by Non-pharmaceutical Therapies: Optimal Use of Medical Gases with Antioxidant Properties. Curr. Pharm. Des. 2011, 17, 2253–2263. [Google Scholar] [CrossRef]
- Su, F.; Zhao, L.; Zhang, S.; Wang, J.; Chen, N.; Gong, Q.; Tang, J.; Wang, H.; Yao, J.; Wang, Q.; et al. Cardioprotection by PI3K-mediated signaling is required for anti-arrhythmia and myocardial repair in response to ischemic preconditioning in infarcted pig hearts. Lab. Investig. 2015, 95, 860–871. [Google Scholar] [CrossRef]
- Smyth, J.W.; Hong, T.-T.; Gao, D.; Vogan, J.M.; Jensen, B.C.; Fong, T.S.; Simpson, P.C.; Stainier, D.Y.R.; Chi, N.C.; Shaw, R.M. Limited forward trafficking of connexin 43 reduces cell-cell coupling in stressed human and mouse myocardium. J. Clin. Invest. 2010, 120, 266–279. [Google Scholar] [CrossRef]
- Egan Benova, T.; Szeiffova Bacova, B.; Viczenczova, C.; Diez, E.; Barancik, M.; Tribulova, N. Protection of cardiac cell-to-cell coupling attenuate myocardial remodeling and proarrhythmia induced by hypertension. Physiol. Res. 2016, 65 (Suppl. 1), S29–S42. [Google Scholar]
- Tribulova, N.; Szeiffova Bacova, B.; Benova, T.; Viczenczova, C. Can we protect from malignant arrhythmias by modulation of cardiac cell-to-cell coupling? J. Electrocardiol. 2015, 48, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Viczenczova, C.; Kura, B.; Benova, T.E.; Yin, C.; Kukreja, R.C.; Slezak, J.; Tribulova, N.; Bacova, B.S. Irradiation-induced cardiac connexin-43 and miR-21 responses are hampered by treatment with atorvastatin and aspirin. Int. J. Mol. Sci. 2018, 19, 1–10. [Google Scholar] [CrossRef]
- Qiang, B.; Toma, J.; Fujii, H.; Osherov, A.B.; Nili, N.; Sparkes, J.D.; Fefer, P.; Samuel, M.; Butany, J.; Leong-Poi, H.; et al. Statin therapy prevents expansive remodeling in venous bypass grafts. Atherosclerosis 2012, 223, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Werner-Wasik, M. Future development of amifostine as a radioprotectant. Semin. Oncol. 1999, 26, 129–134. [Google Scholar]
- Genvresse, I.; Lange, C.; Schanz, J.; Schwelgert, M.; Harder, H.; Possinger, K.; Späth-Schwalbe, E. Tolerability of the cytoprotective agent amifostine in elderly patients receiving chemotherapy: A comparative study. Anticancer. Drugs 2001, 12, 345–349. [Google Scholar] [CrossRef]
- Turer, A.T.; Hill, J.A. Pathogenesis of Myocardial Ischemia-Reperfusion Injury and Rationale for Therapy. Am. J. Cardiol. 2010, 37, 761–771. [Google Scholar] [CrossRef]
- Kucharská, J.; Gvozdjáková, A.; Kura, B.; Rausová, Z.; Slezák, J. Effect of molecular hydrogen on coenzyme Q in plasma, myocardial tissue and mitochondria of rats. J. Nutr. Heal. Food Eng. 2018, 8, 362–364. [Google Scholar]
- Xia, Y.; Zweier, J.L. Substrate control of free radical generation from xanthine oxidase in the postischemic heart. J. Biol. Chem. 1995, 270, 18797–18803. [Google Scholar] [CrossRef]
- Zorov, D.B.; Filburn, C.R.; Klotz, L.-O.; Zweier, J.L.; Sollott, S.J. Reactive Oxygen Species (ROS)-induced ROS Release: A New Phenomenon Accompanying Induction of the Mitochondrial Permeability Transition in Cardiac Myocytes. J. Exp. Med. Bull. 2000, 192, 1001–1014. [Google Scholar] [CrossRef]
- Zhang, Y.; Tan, S.; Xu, J.; Wang, T. Hydrogen Therapy in Cardiovascular and Metabolic Diseases: From Bench to Bedside. Cell. Physiol. Biochem. 2018, 47, 1–10. [Google Scholar] [CrossRef]
- Burchfield, J.S.; Xie, M.; Hill, J.A. Pathological ventricular remodeling: Mechanisms: Part 1 of 2. Circulation 2013, 128, 388–400. [Google Scholar] [CrossRef]
- Pedram, A.; Razandi, M.; O’Mahony, F.; Lubahn, D.; Levin, E.R. Estrogen Receptor-β Prevents Cardiac Fibrosis. Mol. Endocrinol. 2010, 24, 2152–2165. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H. Reprogramming Interferon Regulatory Factor Signaling in Cardiometabolic Diseases. Physiology 2017, 32, 210–223. [Google Scholar] [CrossRef]
- Keeley, E.C.; Boura, J.A.; Grines, C.L. Primary angioplasty versus intravenous thrombolytic therapy for acute myocardial infarction: A quantitative review of 23 randomised trials. Lancet 2003, 361, 13–20. [Google Scholar] [CrossRef]
- Braunwald, E.; Kloner, R.A. Myocardial reperfusion: A double-edged sword? J. Clin. Invest. 1985, 76, 1713–1719. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Yellon, D.M. Preconditioning and postconditioning: United at reperfusion. Pharmacol. Ther. 2007, 116, 173–191. [Google Scholar] [CrossRef]
- Loke, K.E.; McConnell, P.I.; Tuzman, J.M.; Shesely, E.G.; Smith, C.J.; Stackpole, C.J.; Thompson, C.I.; Kaley, G.; Wolin, M.S.; Hintze, T.H. Endogenous Endothelial Nitric Oxide Synthase–Derived Nitric Oxide Is a Physiological Regulator of Myocardial Oxygen Consumption. Circ. Res. 1999, 84, 840–845. [Google Scholar] [CrossRef]
- Radomski, M.W.; Palmer, R.M.J.; Moncada, S. Endogenous Nitric Oxide Inhibits Human Platelet Adhesion To Vascular Endothelium. Lancet 1987, 330, 1057–1058. [Google Scholar] [CrossRef]
- Ma, X.L.; Weyrich, A.S.; Lefer, D.J.; Lefer, A.M. Diminished basal nitric oxide release after myocardial ischemia and reperfusion promotes neutrophil adherence to coronary endothelium. Circ. Res. 1993, 72, 403–412. [Google Scholar] [CrossRef]
- Beckman, J.S.; Beckman, T.W.; Chen, J.; Marshall, P.A.; Freeman, B.A. Apparent hydroxyl radical production by peroxynitrite: Implications for endothelial injury from nitric oxide and superoxide. Proc. Natl. Acad. Sci. United States Am. 1990, 87, 1620–1624. [Google Scholar] [CrossRef]
- Wang, P.; Zweier, J.L. Measurement of Nitric Oxide and Peroxynitrite Generation in the Postischemic Heart. J. Biol. Chem. 1996, 271, 29223–29230. [Google Scholar] [CrossRef]
- Shi, S.; Xue, F. Current Antioxidant Treatments in Organ Transplantation. Oxid. Med. Cell. Longev. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
- Panieri, E.; Santoro, M.M. ROS signaling and redox biology in endothelial cells. Cell. Mol. Life Sci. 2015, 72, 3281–3303. [Google Scholar] [CrossRef]
- León, J.; Acuña-Castroviejo, D.; Escames, G.; Tan, D.X.; Reiter, R.J. Melatonin mitigates mitochondrial malfunction. J. Pineal. Res. 2005, 38, 1–9. [Google Scholar]
- Koh, P.O. Melatonin regulates nitric oxide synthase expression in ischemic brain injury. J. Vet. Med. Sci. 2008, 70, 747–750. [Google Scholar] [CrossRef]
- Ahmad, A.; Sattar, M.Z.A.; Rathore, H.A.; Hussain, A.I.; Khan, S.A.; Fatima, T.; Afzal, S.; Abdullah, N.A.; Johns, E.J. Antioxidant activity and free radical scavenging capacity of L-arginine and NaHS: A comparative in vitro study. Acta Pol. Pharm. Drug Res. 2015, 72, 245–252. [Google Scholar]
- Lemasters, J.J.; Qian, T.; He, L.; Kim, J.-S.; Elmore, S.P.; Cascio, W.E.; Brenner, D.A. Role of Mitochondrial Inner Membrane Permeabilization in Necrotic Cell Death, Apoptosis, and Autophagy. Antioxid. Redox Signal. 2002, 4, 769–781. [Google Scholar] [CrossRef]
- Zaoualí, M.A.; Reiter, R.J.; Padrissa-Altés, S.; Boncompagni, E.; García, J.J.; Ben Abnennebi, H.; Freitas, I.; García-Gil, F.A.; Rosello-Catafau, J. Melatonin protects steatotic and nonsteatotic liver grafts against cold ischemia and reperfusion injury. J. Pineal Res. 2011, 50, 213–221. [Google Scholar] [CrossRef]
- Abu-Amara, M.; Gurusamy, K.S.; Glantzounis, G.; Fuller, B.; Davidson, B.R.R. Pharmacological interventions versus no pharmacological intervention for ischaemia reperfusion injury in liver resection surgery performed under vascular control. Cochrane Database Syst. Rev. 2009, CD008154. [Google Scholar] [CrossRef]
- Álvarez-Ayuso, L.; Gómez-Heras, S.G.; Jorge, E.; Guardiola, J.M.; Torralba, A.; Granado, F.; Millán, I.; Roda, J.R.; Calero, P.; Fernández-García, H.; et al. Vitamin E action on oxidative state, endothelial function and morphology in long-term myocardial preservation. Histol. Histopathol. 2010, 25, 577–587. [Google Scholar]
- Miller, E.R., 3rd; Pastor-Barriuso, R.; Dalal, D.; Riemersma, R.A.; Appel, L.J.; Guallar, E. Meta-Analysis: High-Dosage Vitamin E Supplementation May Increase All-Cause Mortality Background: Experimental models and observational studies. Ann. Intern. Med. 2005, 142, 37–46. [Google Scholar] [CrossRef]
- Schulz, R. Intracellular Targets of Matrix Metalloproteinase-2 in Cardiac Disease: Rationale and Therapeutic Approaches. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 211–242. [Google Scholar] [CrossRef]
- Burwell, L.S.; Nadtochiy, S.M.; Tompkins, A.J.; Young, S.; Brookes, P.S. Direct evidence for S-nitrosation of mitochondrial complex I. Biochem. J. 2006, 394, 627–634. [Google Scholar] [CrossRef]
- Gladwin, M.T.; Schechter, A.N.; Kim-Shapiro, D.B.; Patel, R.P.; Hogg, N.; Shiva, S.; Cannon, R.O.; Kelm, M.; Wink, D.A.; Espey, M.G.; et al. The Emerging Biology of the Nitrite Anion. Nat. Chem. Biol. 2005, 1, 308–314. [Google Scholar] [CrossRef]
- Downey, J.M.; Cohen, M.V. A really radical observation. A comment on Penna et al. in Basic Res Cardiol (2006) 101:180-189. Basic Res. Cardiol. 2006, 101, 190–191. [Google Scholar] [CrossRef]
- Wang, R. Gasotransmitters: Growing pains and joys. Trends Biochem. Sci. 2014, 39, 227–232. [Google Scholar] [CrossRef]
- Dong, B.M.; Abano, J.B.; Egan, T.M. Nitric oxide ventilation of rat lungs from non-heart-beating donors improves posttransplant function. Am. J. Transplant. 2009, 9, 2707–2715. [Google Scholar] [CrossRef]
- Ozaki, K.S.; Yoshida, J.; Ueki, S.; Pettigrew, G.L.; Ghonem, N.; Sico, R.M.; Lee, L.Y.; Shapiro, R.; Lakkis, F.G.; Pacheco-Silva, A.; et al. Carbon monoxide inhibits apoptosis during cold storage and protects kidney grafts donated after cardiac death. Transpl. Int. 2012, 25, 107–117. [Google Scholar] [CrossRef]
- Nath, K.A. Preservation of the kidney by carbon monoxide: A black swan phenomenon. Kidney Int. 2008, 74, 989–991. [Google Scholar] [CrossRef]
- Wu, D.; Wang, J.; Li, H.; Xue, M.; Ji, A.; Li, Y. Role of hydrogen sulfide in ischemia-reperfusion injury. Oxid. Med. Cell. Longev. 2015, 2015, 186908. [Google Scholar] [CrossRef]
- Kajimura, M.; Nakanishi, T.; Takenouchi, T.; Morikawa, T.; Hishiki, T.; Yukutake, Y.; Suematsu, M. Gas biology: Tiny molecules controlling metabolic systems. Respir. Physiol. Neurobiol. 2012, 184, 139–148. [Google Scholar] [CrossRef]
- Iida, A.; Nosaka, N.; Yumoto, T.; Knaup, E.; Naito, H.; Nishiyama, C.; Yamakawa, Y.; Tsukahara, K.; Terado, M.; Sato, K.; et al. The clinical application of hydrogen as a medical treatment. Acta Med. Okayama 2016, 70, 331–338. [Google Scholar]
- Abraini, J.H.; Gardette-Chauffour, M.C.; Martinez, E.; Rostain, J.C.; Lemaire, C. Psychophysiological reactions in humans during an open sea dive to 500 m with a hydrogen-helium-oxygen mixture. J. Appl. Physiol. 1994, 76, 1113–1118. [Google Scholar] [CrossRef]
- Dole, M.; Wilson, F.R.; Fife, W.P. Hyperbaric Hydrogen Therapy: A possible Treatment for Cancer. Science 1975, 190, 152–154. [Google Scholar] [CrossRef]
- Ohsawa, I.; Ishikawa, M.; Takahashi, K.; Watanabe, M.; Nishimaki, K.; Yamagata, K.; Katsura, K.I.; Katayama, Y.; Asoh, S.; Ohta, S. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat. Med. 2007, 13, 688–694. [Google Scholar] [CrossRef]
- Li, Q.; Yu, P.; Zeng, Q.; Luo, B.; Cai, S.; Hui, K.; Yu, G.; Zhu, C.; Chen, X.; Duan, M.; et al. Neuroprotective Effect of Hydrogen-Rich Saline in Global Cerebral Ischemia/Reperfusion Rats: Up-Regulated Tregs and Down-Regulated miR-21, miR-210 and NF-κB Expression. Neurochem. Res. 2016, 41, 2655–2665. [Google Scholar] [CrossRef]
- Suzuki, Y.; Sano, M.; Hayashida, K.; Ohsawa, I.; Ohta, S.; Fukuda, K. Are the effects of α-glucosidase inhibitors on cardiovascular events related to elevated levels of hydrogen gas in the gastrointestinal tract? FEBS Lett. 2009, 583, 2157–2159. [Google Scholar] [CrossRef]
- Tamasawa, A.; Mochizuki, K.; Hariya, N.; Saito, M.; Ishida, H.; Doguchi, S.; Yanagiya, S.; Osonoi, T. Hydrogen gas production is associated with reduced interleukin-1β mRNA in peripheral blood after a single dose of acarbose in Japanese type 2 diabetic patients. Eur. J. Pharmacol. 2015, 762, 96–101. [Google Scholar] [CrossRef]
- Oharazawa, H.; Igarashi, T.; Yokota, T.; Fujii, H.; Suzuki, H.; Machide, M.; Takahashi, H.; Ohta, S.; Ohsawa, I. Protection of the retina by rapid diffusion of hydrogen: Administration of hydrogen-loaded eye drops in retinal ischemia-reperfusion injury. Investig. Ophthalmol. Vis. Sci. 2010, 51, 487–492. [Google Scholar] [CrossRef]
- Hayashida, K.; Sano, M.; Ohsawa, I.; Shinmura, K.; Tamaki, K.; Kimura, K.; Endo, J.; Katayama, T.; Kawamura, A.; Kohsaka, S.; et al. Inhalation of hydrogen gas reduces infarct size in the rat model of myocardial ischemia-reperfusion injury. Biochem. Biophys. Res. Commun. 2008, 373, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, J.; Long, Z.; Wang, C.; Wang, L.; Sun, P.; Li, P.; Wang, T. Hydrogen (H2) inhibits isoproterenol-induced cardiac hypertrophy via antioxidative pathways. Front. Pharmacol. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Cardinal, J.S.; Zhan, J.; Wang, Y.; Sugimoto, R.; Tsung, A.; McCurry, K.R.; Billiar, T.R.; Nakao, A. Oral hydrogen water prevents chronic allograft nephropathy in rats. Kidney Int. 2010, 77, 101–109. [Google Scholar] [CrossRef]
- LeBaron, T.W.; Larson, A.J.; Ohta, S.; Mikami, T.; Barlow, J.; Bulloch, J.; DeBeliso, M. Acute Supplementation with Molecular Hydrogen Benefits Submaximal Exercise Indices. Randomized, Double-Blinded, Placebo-Controlled Crossover Pilot Study. J. Lifestyle Med. 2019, 9, 36–43. [Google Scholar] [CrossRef]
- LeBaron, T.W.; Laher, I.; Kura, B.; Slezak, J. Hydrogen gas: From clinical medicine to an emerging ergogenic molecule for sports athletes. Can. J. Physiol. Pharmacol. 2019. [Epub ahead of print]. [Google Scholar] [CrossRef]
- Ito, M.; Hirayama, M.; Yamai, K.; Goto, S.; Ito, M.; Ichihara, M.; Ohno, K. Drinking hydrogen water and intermittent hydrogen gas exposure, but not lactulose or continuous hydrogen gas exposure, prevent 6-hydorxydopamine-induced Parkinson’s disease in rats. Med. Gas Res. 2012, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, M.; Ito, M.; Minato, T.; Yoritaka, A.; LeBaron, T.; Ohno, K. Inhalation of hydrogen gas elevates urinary 8-hydroxy-2′-deoxyguanine in Parkinson’s disease. Med. Gas Res. 2018, 8, 144–149. [Google Scholar]
- Ohta, S. Molecular hydrogen is a novel antioxidant to efficiently reduce oxidative stress with potential for the improvement of mitochondrial diseases. Biochim. Biophys. Acta Gen. Subj. 2012, 1820, 586–594. [Google Scholar] [CrossRef]
- Ono, H.; Nishijima, Y.; Adachi, N.; Sakamoto, M.; Kudo, Y.; Kaneko, K.; Nakao, A.; Imaoka, T. A basic study on molecular hydrogen (H2) inhalation in acute cerebral ischemia patients for safety check with physiological parameters and measurement of blood H2 level. Med. Gas Res. 2012, 2, 21. [Google Scholar] [CrossRef]
- Huang, C.S.; Kawamura, T.; Toyoda, Y.; Nakao, A. Recent advances in hydrogen research as a therapeutic medical gas. Free Radic. Res. 2010, 44, 971–982. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yu, Q.; Liu, Y.; Zhang, R.; Xue, L. Hydrogen gas alleviates oxygen toxicity by reducing hydroxyl radical levels in PC12 cells. PLoS ONE 2017, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Song, H.; Wang, X.T.; Liang, Y.; Xi, Y.J.; Gao, Y.; Guo, Q.J.; Lebaron, T.; Luo, Y.X.; Li, S.C.; et al. Molecular hydrogen increases resilience to stress in mice. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric Oxide and Peroxynitrite in Health and Disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef] [PubMed]
- Hanaoka, T.; Kamimura, N.; Yokota, T.; Takai, S.; Ohta, S. Molecular hydrogen protects chondrocytes from oxidative stress and indirectly alters gene expressions through reducing peroxynitrite derived from nitric oxide. Med. Gas Res. 2011, 1, 18. [Google Scholar] [CrossRef] [PubMed]
- Huang, L. Molecular hydrogen: A therapeutic antioxidant and beyond. Med. Gas Res. 2016, 6, 219–222. [Google Scholar] [CrossRef]
- Hood, E.; Simone, E.; Wattamwar, P.; Dziubla, T.; Muzykantov, V. Nanocarriers for vascular delivery of antioxidants. Nanomedicine 2011, 6, 1257–1272. [Google Scholar] [CrossRef]
- Penders, J.; Kissner, R.; Koppenol, W.H. ONOOH does not react with H2: Potential beneficial effects of H2 as an antioxidant by selective reaction with hydroxyl radicals and peroxynitrite. Free Radic. Biol. Med. 2014, 75, 191–194. [Google Scholar] [CrossRef]
- Wood, K.C.; Gladwin, M.T. The hydrogen highway to reperfusion therapy. Nat. Med. 2007, 13, 673–674. [Google Scholar] [CrossRef]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B.; Tsang, W. Critical Review of rate constants for reactions of hydrated electronsChemical Kinetic Data Base for Combustion Chemistry. Part 3: Propane. J. Phys. Chem. Ref. Data 1988, 17, 513. [Google Scholar]
- Dixon, B.J.; Tang, J.; Zhang, J.H. The evolution of molecular hydrogen: A noteworthy potential therapy with clinical significance. Med. Gas Res. 2013, 3, 10. [Google Scholar] [CrossRef]
- Kamimura, N.; Nishimaki, K.; Ohsawa, I.; Ohta, S. Molecular hydrogen improves obesity and diabetes by inducing hepatic FGF21 and stimulating energy metabolism in db/db mice. Obesity 2011, 19, 1396–1403. [Google Scholar] [CrossRef]
- Kamimura, N.; Ichimiya, H.; Iuchi, K.; Ohta, S. Molecular hydrogen stimulates the gene expression of transcriptional coactivator pgc-1α to enhance fatty acid metabolism. npj Aging Mech. Dis. 2016, 2, 16008. [Google Scholar] [CrossRef]
- Ge, L.; Yang, M.; Yang, N.-N.; Yin, X.-X.; Song, W.-G. Molecular hydrogen: A preventive and therapeutic medical gas for various diseases. Oncotarget 2017, 8, 102653–102673. [Google Scholar] [CrossRef]
- Ohta, S. Molecular hydrogen as a novel antioxidant: Overview of the advantages of hydrogen for medical applications. Methods Enzymol. 2015, 555, 289–317. [Google Scholar]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef]
- Itoh, K.; Wakabayashi, N.; Katoh, Y.; Ishii, T.; Igarashi, K.; Engel, J.D.; Yamamoto, M. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999, 13, 76–86. [Google Scholar] [CrossRef]
- Baird, L.; Dinkova-Kostova, A.T. The cytoprotective role of the Keap1-Nrf2 pathway. Arch. Toxicol. 2011, 85, 241–272. [Google Scholar] [CrossRef] [PubMed]
- Raghunath, A.; Sundarraj, K.; Nagarajan, R.; Arfuso, F.; Bian, J.; Kumar, A.P.; Sethi, G.; Perumal, E. Antioxidant response elements: Discovery, classes, regulation and potential applications. Redox Biol. 2018, 17, 297–314. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q. Role of NRF2 in oxidative stress ans toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef]
- Yuan, J.; Wang, D.; Liu, Y.; Chen, X.; Zhang, H.; Shen, F.; Liu, X.; Fu, J. Hydrogen-rich water attenuates oxidative stress in rats with traumatic brain injury via Nrf2 pathway. J. Surg. Res. 2018, 228, 238–246. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, G.; Zhang, L.; Cheng, S.; Luo, C.; Liao, Y.; Guo, S. Therapeutic efficacy of hydrogen-rich saline alone and in combination with PI3K inhibitor in non-small cell lung cancer. Mol. Med. Rep. 2018, 18, 2182–2190. [Google Scholar] [CrossRef] [PubMed]
- López-Bernardo, E.; Anedda, A.; Sánchez-Pérez, P.; Acosta-Iborra, B.; Cadenas, S. 4-Hydroxynonenal induces Nrf2-mediated UCP3 upregulation in mouse cardiomyocytes. Free Radic. Biol. Med. 2015, 88, 427–438. [Google Scholar] [CrossRef]
- Bell, K.F.; Al-Mubarak, B.; Fowler, J.H.; Baxter, P.S.; Gupta, K.; Tsujita, T.; Chowdhry, S.; Patani, R.; Chandran, S.; Horsburgh, K.; et al. Mild oxidative stress activates Nrf2 in astrocytes, which contributes to neuroprotective ischemic preconditioning. Proc. Natl. Acad. Sci. 2011, 108, E1–E2. [Google Scholar] [CrossRef]
- Jo, S.-K.; Ko, G.J.; Boo, C.S.; Cho, W.Y.; Kim, H.K. Heat Preconditioning Attenuates Renal Injury in Ischemic ARF in Rats: Role of Heat-Shock Protein 70 on NF-κB–Mediated Inflammation and on Tubular Cell Injury. J. Am. Soc. Nephrol. 2006, 17, 3082–3092. [Google Scholar] [CrossRef] [PubMed]
- Zálešák, M.; Kura, B.; Graban, J.; Farkašová, V.; Slezák, J.; Ravingerová, T. Molecular hydrogen potentiates beneficial anti-infarct effect of hypoxic postconditioning in isolated rat hearts: A novel cardioprotective intervention. Can. J. Physiol. Pharmacol. 2017, 95, 888–893. [Google Scholar] [CrossRef] [PubMed]
- Ristow, M.; Zarse, K. How increased oxidative stress promotes longevity and metabolic health: The concept of mitochondrial hormesis (mitohormesis). Exp. Gerontol. 2010, 45, 410–418. [Google Scholar] [CrossRef]
- Murakami, Y.; Ito, M.; Ohsawa, I. Molecular hydrogen protects against oxidative stress-induced SH-SY5Y neuroblastoma cell death through the process of mitohormesis. PLoS ONE 2017, 12, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kura, B.; Kalocayova, B.; LeBaron, T.W.; Frimmel, K.; Buday, J.; Surovy, J.; Slezak, J. Regulation of microRNAs by molecular hydrogen contributes to the prevention of radiation-induced damage in the rat myocardium. Mol. Cell. Biochem. 2019, 0, 1–12. [Google Scholar] [CrossRef]
- Zhang, C.B.; Tang, Y.C.; Xu, X.J.; Guo, S.X.; Wang, H.Z. Hydrogen gas inhalation protects against liver ischemia/reperfusion injury by activating the NF-κB signaling pathway. Exp. Ther. Med. 2015, 9, 2114–2120. [Google Scholar] [CrossRef]
- Huang, C.S.; Kawamura, T.; Peng, X.; Tochigi, N.; Shigemura, N.; Billiar, T.R.; Nakao, A.; Toyoda, Y. Hydrogen inhalation reduced epithelial apoptosis in ventilator-induced lung injury via a mechanism involving nuclear factor-kappa B activation. Biochem. Biophys. Res. Commun. 2011, 408, 253–258. [Google Scholar] [CrossRef]
- Nishiwaki, H.; Ito, M.; Negishi, S.; Sobue, S.; Ichihara, M.; Ohno, K. Molecular hydrogen upregulates heat shock response and collagen biosynthesis, and downregulates cell cycles: Meta-analyses of gene expression profiles. Free Radic. Res. 2018, 52, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Sobue, S.; Inoue, C.; Hori, F.; Qiao, S.; Murate, T.; Ichihara, M. Molecular hydrogen modulates gene expression via histone modification and induces the mitochondrial unfolded protein response. Biochem. Biophys. Res. Commun. 2017, 493, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Magenta, A.; Greco, S.; Gaetano, C.; Martelli, F. Oxidative stress and microRNAs in vascular diseases. Int. J. Mol. Sci. 2013, 14, 17319–17346. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Kang, Z.; Cai, J.; Liu, W.; Liu, Y.; Zhang, J.H.; Denoble, P.J.; Tao, H.; Sun, X. Hydrogen-Rich Saline Protects Myocardium Against Ischemia/Reperfusion Injury in Rats. Exp. Biol. Med. 2009, 234, 1212–1219. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, S.; Kong, S.W.; Lu, J.; Bisping, E.; Zhang, H.; Allen, P.D.; Golub, T.R.; Pieske, B.; Pu, W.T. Altered microRNA expression in human heart disease. Physiol. Genomics 2007, 31, 367–373. [Google Scholar] [CrossRef]
- Qin, Z.X.; Yu, P.; Qian, D.H.; Song, M.B.; Tan, H.; Yu, Y.; Li, W.; Wang, H.; Liu, J.; Wang, Q.; et al. Hydrogen-rich saline prevents neointima formation after carotid balloon injury by suppressing ROS and the TNF-α/NF-κB pathway. Atherosclerosis 2012, 220, 343–350. [Google Scholar] [CrossRef]
- Lan, J.; Huang, Z.; Han, J.; Shao, J.; Huang, C. Redox regulation of microRNAs in cancer. Cancer Lett. 2018, 418, 250–259. [Google Scholar] [CrossRef]
- Gong, Y.-Y.; Luo, J.-Y.; Wang, L.; Huang, Y. MicroRNAs Regulating Reactive Oxygen Species in Cardiovascular Diseases. Antioxid. Redox Signal. 2018, 29, 1092–1107. [Google Scholar] [CrossRef]
- Simone, N.L.; Soule, B.P.; Ly, D.; Saleh, A.D.; Savage, J.E.; DeGraff, W.; Cook, J.; Harris, C.C.; Gius, D.; Mitchell, J.B. Ionizing radiation-induced oxidative stress alters miRNA expression. PLoS ONE 2009, 4, 1–7. [Google Scholar] [CrossRef]
- Zhu, H.; Fan, G.C. Role of microRNAs in the reperfused myocardium towards post-infarct remodelling. Cardiovasc. Res. 2012, 94, 284–292. [Google Scholar] [CrossRef]
- Viczenczova, C.; Szeiffova Bacova, B.; Egan Benova, T.; Kura, B.; Yin, C.; Weismann, P.; Kukreja, R.; Slezak, J.; Tribulova, N. Myocardial connexin-43 and PKC signalling are involved in adaptation of the heart to irradiation-induced injury: Implication of miR-1 and miR-21. Gen. Physiol. Biophys. 2016, 35, 215–222. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, H.; Yang, X.; Ge, Y.; Zhang, C.; Qin, Q.; Lu, J.; Zhan, L.; Cheng, H.; Sun, X. MicroRNA-21 is a novel promising target in cancer radiation therapy. Tumor Biol. 2014, 35, 3975–3979. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, J.; Miao, H.; Chen, X.; Sun, X.; Li, Y. Hydrogen-rich saline attenuates vascular smooth muscle cell proliferation and neointimal hyperplasia by inhibiting reactive oxygen species production and inactivating the Ras-ERK1/2-MEK1/2 and Akt pathways. Int. J. Mol. Med. 2013, 31, 597–606. [Google Scholar]
- Riley, P.A. Free Radicals in Biology: Oxidative Stress and the Effects of Ionizing Radiation Free radicals in biology: Oxidative stress and the effects of ionizing radiation. Int. J. Radiat. Biol. 1994, 65, 27–33. [Google Scholar] [CrossRef]
- Zhao, W.; Robbins, M. Inflammation and Chronic Oxidative Stress in Radiation-Induced Late Normal Tissue Injury: Therapeutic Implications. Curr. Med. Chem. 2009, 16, 130–143. [Google Scholar] [CrossRef]
- Yang, Y.; Zhu, Y.; Xi, X. Anti-inflammatory and antitumor action of hydrogen via reactive oxygen species. Oncol. Lett. 2018, 16, 2771–2776. [Google Scholar]
- Xie, K.; Yu, Y.; Pei, Y.; Hou, L.; Chen, S.; Xiong, L.; Wang, G. Protective effects of hydrogen gas on murine polymicrobial sepsis via reducing oxidative stress and HMGB1 release. Shock 2010, 34, 90–97. [Google Scholar] [CrossRef]
- Sun, H.; Chen, L.; Zhou, W.; Hu, L.; Li, L.; Tu, Q.; Chang, Y.; Liu, Q.; Sun, X.; Wu, M.; et al. The protective role of hydrogen-rich saline in experimental liver injury in mice. J. Hepatol. 2011, 54, 471–480. [Google Scholar] [CrossRef]
- Hattori, Y.; Kotani, T.; Tsuda, H.; Mano, Y.; Tu, L.; Li, H.; Hirako, S.; Ushida, T.; Imai, K.; Nakano, T.; et al. Maternal molecular hydrogen treatment attenuates lipopolysaccharide-induced rat fetal lung injury. Free Radic. Res. 2015, 49, 1026–1037. [Google Scholar] [CrossRef]
- Varga, V.; Németh, J.; Oláh, O.; Tóth-Szűki, V.; Kovács, V.; Remzső, G.; Domoki, F. Molecular hydrogen alleviates asphyxia-induced neuronal cyclooxygenase-2 expression in newborn pigs. Acta Pharmacol. Sin. 2018, 39, 1273–1283. [Google Scholar] [CrossRef]
- Qian, L.; Li, B.; Cao, F.; Huang, Y.; Liu, S.; Cai, J.; Gao, F. Hydrogen-rich PBS protects cultured human cells from ionizing radiation-induced cellular damage. Nucl. Technol. Radiat. Prot. 2010, 25, 23–29. [Google Scholar] [CrossRef]
- Xiao, H.W.; Li, Y.; Luo, D.; Dong, J.L.; Zhou, L.X.; Zhao, S.Y.; Zheng, Q.S.; Wang, H.C.; Cui, M.; Fan, S.J. Hydrogen-water ameliorates radiation-induced gastrointestinal toxicity via MyD88’s effects on the gut microbiota. Exp. Mol. Med. 2018, 50, e433. [Google Scholar] [CrossRef]
- Qian, L.; Cao, F.; Cui, J.; Wang, Y.; Huang, Y.; Chuai, Y.; Zaho, L.; Jiang, H.; Cai, J. The Potential Cardioprotective Effects of Hydrogen in Irradiated Mice. J. Radiat. Res. 2010, 51, 741–747. [Google Scholar] [CrossRef]
- Qian, L.; Li, B.; Cai, J.; Gao, F. The Hypothesis of an Effective Safe and Novel Radioprotective Agent Hydrogen-rich Solution La Hipótesis de una Solución rica en Hidrógeno como Agente Radioprotector Novedoso, Seguro y Efectivo. Am. J. Transplant. 2010, 59, 122–124. [Google Scholar]
- Mei, K.; Zhao, S.; Qian, L.; Li, B.; Ni, J.; Cai, J. Hydrogen protects rats from dermatitis caused by local radiation. J. Dermatolog. Treat. 2014, 25, 182–188. [Google Scholar] [CrossRef]
- Watanabe, S.; Fujita, M.; Ishihara, M.; Tachibana, S.; Yamamoto, Y.; Kaji, T.; Kawauchi, T.; Kanatani, Y. Protective effect of inhalation of hydrogen gas on radiation-induced dermatitis and skin injury in rats. J. Radiat. Res. 2014, 55, 1107–1113. [Google Scholar] [CrossRef]
- Ramachandran, A.; Madesh, M.; Balasubramanian, K.A. Apoptosis in the intestinal epithelium: Its relevance in normal and pathophysiological conditions. J. Gastroenterol. Hepatol. 2000, 15, 109–120. [Google Scholar] [CrossRef]
- Kang, K.M.; Kang, Y.N.; Choi, I.B.; Gu, Y.; Kawamura, T.; Toyoda, Y.; Nakao, A. Effects of drinking hydrogen-rich water on the quality of life of patients treated with radiotherapy for liver tumors. Med. Gas Res. 2011, 1, 11. [Google Scholar] [CrossRef]
- Conklin, K.A. Dietary Antioxidants During Cancer Chemotherapy: Impact on Chemotherapeutic Effectiveness and Development of Side Effects. Nutr. Cancer 2000, 37, 1–18. [Google Scholar] [CrossRef]
- Kitamura, A.; Kobayashi, S.; Matsushita, T.; Fujinawa, H.; Murase, K. Experimental verification of protective effect of hydrogen-rich water against cisplatin-induced nephrotoxicity in rats using dynamic contrast-enhanced CT. Br. J. Radiol. 2010, 83, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Shinbo, T.; Kokubo, K.; Sato, Y.; Hagiri, S.; Hataishi, R.; Hirose, M.; Kobayashi, H. Breathing nitric oxide plus hydrogen gas reduces ischemia-reperfusion injury and nitrotyrosine production in murine heart. Am. J. Physiol. Circ. Physiol. 2013, 305, H542–H550. [Google Scholar] [CrossRef][Green Version]
- Fukuda, K.I.; Asoh, S.; Ishikawa, M.; Yamamoto, Y.; Ohsawa, I.; Ohta, S. Inhalation of hydrogen gas suppresses hepatic injury caused by ischemia/reperfusion through reducing oxidative stress. Biochem. Biophys. Res. Commun. 2007, 361, 670–674. [Google Scholar] [CrossRef]
- Sakai, K.; Cho, S.; Shibata, I.; Yoshitomi, O.; Maekawa, T.; Sumikawa, K. Inhalation of hydrogen gas protects against myocardial stunning and infarction in swine. Scand. Cardiovasc. J. 2012, 46, 183–189. [Google Scholar] [CrossRef]
- Cole, A.R.; Perry, D.A.; Raza, A.; Nedder, A.P.; Pollack, E.; Regan, W.L.; van den Bosch, S.J.; Polizzotti, B.D.; Yang, E.; Davila, D.; et al. Perioperatively Inhaled Hydrogen Gas Diminishes Neurologic Injury Following Experimental Circulatory Arrest in Swine. JACC Basic to Transl. Sci. 2019, 4, 176–187. [Google Scholar] [CrossRef]
- Lefer, D.J.; Nakanishi, K.; Johnston, W.E.; Vinten-Johansen, J. Antineutrophil and myocardial protecting actions of a novel nitric oxide donor after acute myocardial ischemia and reperfusion in dogs. Circulation 1993, 88, 2337–2350. [Google Scholar] [CrossRef]
- Wong, P.S.Y.; Eiserich, J.P.; Reddy, S.; Lopez, C.L.; Cross, C.E.; van der VlieT, A. Inactivation of glutathione S-Transferases by nitric oxide-derived oxidants: Exploring a role for tyrosine nitration. Arch. Biochem. Biophys. 2001, 394, 216–228. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, Q.; He, B.; Xiao, J.; Wang, Z.; Sun, X. Anti-inflammatory effect of hydrogen-rich saline in a rat model of regional myocardial ischemia and reperfusion. Int. J. Cardiol. 2011, 148, 91–95. [Google Scholar] [CrossRef]
- Javadov, S.; Karmazyn, M. Mitochondrial permeability transition pore opening as an endpoint to initiate cell death and as a putative target for cardioprotection. Cell. Physiol. Biochem. 2007, 20, 1–22. [Google Scholar] [CrossRef]
- Jing, L.; Wang, Y.; Zhao, X.M.; Zhao, B.; Han, J.J.; Qin, S.C.; Sun, X.J. Cardioprotective Effect of Hydrogen-rich Saline on Isoproterenol-induced Myocardial Infarction in Rats. Hear. Lung Circ. 2015, 24, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Long, Z.; Xu, J.; Tan, S.; Zhang, N.; Li, A.; Wang, L.; Wang, T. Hydrogen inhibits isoproterenol-induced autophagy in cardiomyocytes in vitro and in vivo. Mol. Med. Rep. 2017, 16, 8253–8258. [Google Scholar] [CrossRef]
- Gao, Y.; Yang, H.; Chi, J.; Xu, Q.; Zhao, L.; Yang, W.; Liu, W.; Yang, W. Hydrogen Gas Attenuates Myocardial Ischemia Reperfusion Injury Independent of Postconditioning in Rats by Attenuating Endoplasmic Reticulum Stress-Induced Autophagy. Cell. Physiol. Biochem. 2017, 43, 1503–1514. [Google Scholar] [CrossRef]
- Gao, Y.; Yang, H.; Fan, Y.; Li, L.; Fang, J.; Yang, W. Hydrogen-Rich Saline Attenuates Cardiac and Hepatic Injury in Doxorubicin Rat Model by Inhibiting Inflammation and Apoptosis. Mediators Inflamm. 2016, 2016, 3675910. [Google Scholar] [CrossRef] [PubMed]
- Huo, T.T.; Zeng, Y.; Liu, X.N.; Sun, L.; Han, H.Z.; Chen, H.G.; Lu, Z.H.; Huang, Y.; Nie, H.; Dong, H.L.; et al. Hydrogen-rich saline improves survival and neurological outcome after cardiac arrest and cardiopulmonary resuscitation in Rats. Anesth. Analg. 2014, 119, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Hayashida, K.; Sano, M.; Kamimura, N.; Yokota, T.; Suzuki, M.; Maekawa, Y.; Kawamura, A.; Abe, T.; Ohta, S.; Fukuda, K.; et al. H(2) gas improves functional outcome after cardiac arrest to an extent comparable to therapeutic hypothermia in a rat model. J. Am. Heart Assoc. 2012, 1, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.S.; Zheng, H. Chronic hydrogen-rich saline treatment reduces oxidative stress and attenuates left ventricular hypertrophy in spontaneous hypertensive rats. Mol. Cell. Biochem. 2012, 365, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Kato, R.; Nomura, A.; Sakamoto, A.; Yasuda, Y.; Amatani, K.; Nagai, S.; Sen, Y.; Ijiri, Y.; Okada, Y.; Yamaguchi, T.; et al. Hydrogen gas attenuates embryonic gene expression and prevents left ventricular remodeling induced by intermittent hypoxia in cardiomyopathic hamsters. Am. J. Physiol. Circ. Physiol. 2014, 307, H1626–H1633. [Google Scholar] [CrossRef] [PubMed]
- Noda, K.; Shigemura, N.; Tanaka, Y.; Kawamura, T.; Hyun Lim, S.; Kokubo, K.; Billiar, T.R.; Bermudez, C.A.; Kobayashi, H.; Nakao, A. A novel method of preserving cardiac grafts using a hydrogen-rich water bath. J. Hear. Lung Transplant. 2013, 32, 241–250. [Google Scholar] [CrossRef]
- Ohno, K.; Ito, M.; Ichihara, M.; Ito, M. Molecular Hydrogen as an Emerging Therapeutic Medical Gas for Neurodegenerative and Other Diseases. Oxid. Med. Cell. Longev. 2012, 2012, 1–11. [Google Scholar] [CrossRef]
- Tan, M.; Sun, X.; Guo, L.; Su, C.; Sun, X.; Xu, Z. Hydrogen as additive of HTK solution fortifies myocardial preservation in grafts with prolonged cold ischemia. Int. J. Cardiol. 2013, 167, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Noda, K.; Tanaka, Y.; Shigemura, N.; Kawamura, T.; Wang, Y.; Masutani, K.; Sun, X.; Toyoda, Y.; Bermudez, C.A.; Nakao, A. Hydrogen-supplemented drinking water protects cardiac allografts from inflammation-associated deterioration. Transpl. Int. 2012, 25, 1213–1222. [Google Scholar] [CrossRef]
- Nakao, A.; Kaczorowski, D.J.; Wang, Y.; Cardinal, J.S.; Buchholz, B.M.; Sugimoto, R.; Tobita, K.; Lee, S.; Toyoda, Y.; Billiar, T.R.; et al. Amelioration of rat cardiac cold ischemia/reperfusion injury with inhaled hydrogen or carbon monoxide, or both. J. Hear. Lung Transplant. 2010, 29, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, L.; Tao, K.; Vizcaychipi, M.P.; Lloyd, D.M.; Sun, X.; Irwin, M.G.; Ma, D.; Yu, W. Protective effects of hydrogen enriched saline on liver ischemia reperfusion injury by reducing oxidative stress and HMGB1 release. BMC Gastroenterol. 2014, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bolli, R.; Becker, L.; Gross, G.; Mentzer, R.; Balshaw, D.; Lathrop, D.A. Myocardial Protection at a Crossroads: The Need for Translation Into Clinical Therapy Roberto. Circ. Res. 2004, 95, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Katsumata, Y.; Sano, F.; Abe, T.; Tamura, T.; Fujisawa, T.; Shiraishi, Y.; Kohsaka, S.; Ueda, I.; Homma, K.; Suzuki, M.; et al. The Effects of Hydrogen Gas Inhalation on Adverse Left Ventricular Remodeling After Percutaneous Coronary Intervention for ST-Elevated Myocardial Infarction ― First Pilot Study in Humans. Circ. J. 2017, 81, 940–947. [Google Scholar] [CrossRef] [PubMed]
| Alteration Caused by I/R Injury | Mechanism |
|---|---|
| Changes in ion flux | Accumulation of intracellular calcium Ca2+-induced “stone-heart” |
| Increased sodium influx | |
| Abnormal potassium flux | |
| Drop in pH followed by normalization upon reperfusion | |
| Loss of mitochondrial membrane potential | Opening of mitochondrial permeability transition pore (mPTP) |
| Cytochrome c release | |
| Reduction of ATP synthesis | |
| Reactive oxygen species (ROS) | Substrate-level induction of xanthine oxidase resulting in more ROS |
| Impaired mitochondrial function | |
| Neutrophil infiltration | |
| ROS-induced ROS | |
| Dysregulated nitric oxide (NO) metabolism | Loss of NO-vasodilation |
| Production of peroxynitrite | |
| Abnormal S-nitrosation | |
| Apoptosis | JNK pathway |
| Ceramide generation | |
| Cytoplasm acidification | |
| Caspase activation | |
| Autophagic cell death | Excessive AMPK activation |
| Excessive induction of HIF-1α | |
| Endothelial dysfunction | Cytokine, myokine, chemokine signaling |
| Expression of cellular adhesion markers | |
| Impaired vasodilation | |
| Platelet aggregation | |
| Immune activation | Innate immunity (e.g., complement activation, induction of Toll-like receptors) |
| Neutrophil accumulation |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
LeBaron, T.W.; Kura, B.; Kalocayova, B.; Tribulova, N.; Slezak, J. A New Approach for the Prevention and Treatment of Cardiovascular Disorders. Molecular Hydrogen Significantly Reduces the Effects of Oxidative Stress. Molecules 2019, 24, 2076. https://doi.org/10.3390/molecules24112076
LeBaron TW, Kura B, Kalocayova B, Tribulova N, Slezak J. A New Approach for the Prevention and Treatment of Cardiovascular Disorders. Molecular Hydrogen Significantly Reduces the Effects of Oxidative Stress. Molecules. 2019; 24(11):2076. https://doi.org/10.3390/molecules24112076
Chicago/Turabian StyleLeBaron, Tyler W., Branislav Kura, Barbora Kalocayova, Narcis Tribulova, and Jan Slezak. 2019. "A New Approach for the Prevention and Treatment of Cardiovascular Disorders. Molecular Hydrogen Significantly Reduces the Effects of Oxidative Stress" Molecules 24, no. 11: 2076. https://doi.org/10.3390/molecules24112076
APA StyleLeBaron, T. W., Kura, B., Kalocayova, B., Tribulova, N., & Slezak, J. (2019). A New Approach for the Prevention and Treatment of Cardiovascular Disorders. Molecular Hydrogen Significantly Reduces the Effects of Oxidative Stress. Molecules, 24(11), 2076. https://doi.org/10.3390/molecules24112076








