Panax Notoginseng Saponins: A Review of Its Mechanisms of Antidepressant or Anxiolytic Effects and Network Analysis on Phytochemistry and Pharmacology
Abstract
1. Introduction
2. Mechanisms of Antidepression
2.1. Panax Notoginseng Saponins
2.2. Single Component: Rg1
2.3. Single Component: Rb1
2.4. Single Component: Rb3
2.5. Single Component: Rg3
3. Mechanisms of Antianxiety
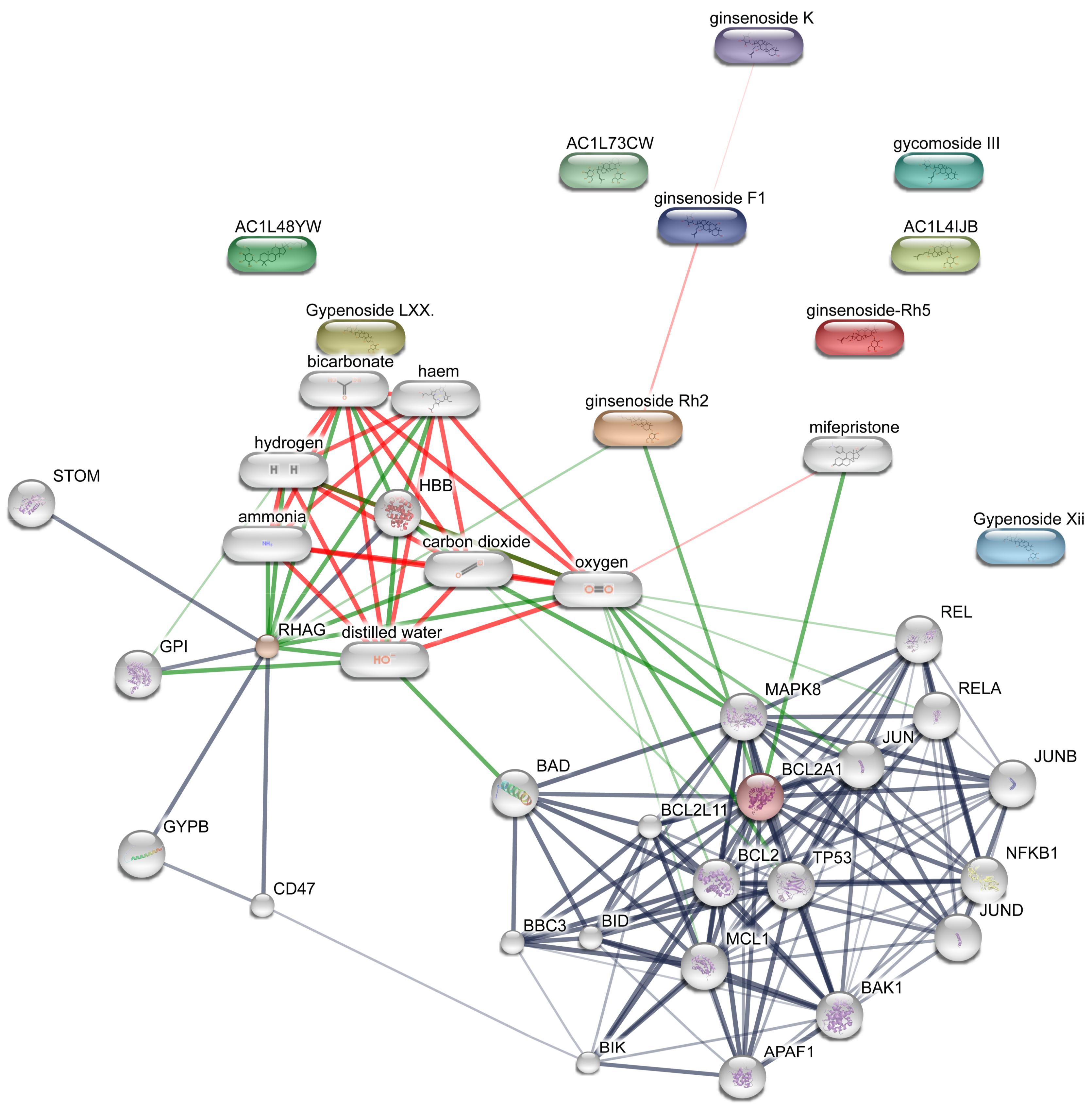
4. Network Target Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Dean, J.; Keshavan, M. The neurobiology of depression: An integrated view. Asian J. Psychiatry 2017, 27, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Dell’Osso, L.; Carmassi, C.; Mucci, F.; Marazziti, D. Depression, Serotonin and Tryptophan. Curr. Pharm. Des. 2016, 22, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Dunn, E.C.; Brown, R.C.; Dai, Y.; Rosand, J.; Nugent, N.R.; Amstadter, A.B.; Smoller, J.W. Genetic determinants of depression: Recent findings and future directions. Harv. Rev. Psychiatry 2015, 23, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Hashmi, A.M.; Butt, Z.; Umair, M. Is depression an inflammatory condition? A review of available evidence. J. Pak. Med. Assoc. 2013, 63, 899–906. [Google Scholar] [PubMed]
- Catena-Dell’Osso, M.; Caserta, A.; Baroni, S.; Nisita, C.; Marazziti, D. The relationship between epilepsy and depression: An update. Curr. Med. Chem. 2013, 20, 2861–2867. [Google Scholar] [PubMed]
- Limon, A.; Mamdani, F.; Hjelm, B.E.; Vawter, M.P.; Sequeira, A. Targets of polyamine dysregulation in major depression and suicide: Activity-dependent feedback, excitability, and neurotransmission. Neurosci. Biobehav. Rev. 2016, 66, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Novais, F.; Starkstein, S. Phenomenology of Depression in Alzheimer’s Disease. J. Alzheimers Dis. 2015, 47, 845–855. [Google Scholar] [CrossRef] [PubMed]
- Stepanichev, M.; Dygalo, N.N.; Grigoryan, G.; Shishkina, G.T.; Gulyaeva, N. Rodent models of depression: Neurotrophic and neuroinflammatory biomarkers. BioMed Res. Int. 2014, 2014, 932757. [Google Scholar] [CrossRef] [PubMed]
- Uchida, S.; Yamagata, H.; Seki, T.; Watanabe, Y. Epigenetic mechanisms of major depression: Targeting neuronal plasticity. Psychiatry Clin. Neurosci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Ma, S.; Kang, A.; Wu, M.; Wang, L.; Wang, Q.; Wang, G.; Hao, H. Chemical dampening of Ly6C(hi) monocytes in the periphery produces anti-depressant effects in mice. Sci. Rep. 2016, 6, 19406. [Google Scholar] [CrossRef] [PubMed]
- Zong, S.; Hoffmann, C.; Mane-Damas, M.; Molenaar, P.; Losen, M.; Martinez, P. Neuronal Surface Autoantibodies in Neuropsychiatric Disorders: Are There Implications for Depression? Front. Immunol. 2017, 8, 752. [Google Scholar] [CrossRef] [PubMed]
- Chi, S.; Yu, J.T.; Tan, M.S.; Tan, L. Depression in Alzheimer’s disease: Epidemiology, mechanisms, and management. J. Alzheimers Dis. 2014, 42, 739–755. [Google Scholar] [PubMed]
- Flaster, M.; Sharma, A.; Rao, M. Poststroke depression: A review emphasizing the role of prophylactic treatment and synergy with treatment for motor recovery. Top. Stroke Rehabil. 2013, 20, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ling, S.; Yang, Y.; Hu, Z.; Davies, H.; Fang, M. Systematic hypothesis for post-stroke depression caused inflammation and neurotransmission and resultant on possible treatments. Neuro Endocrinol. Lett. 2014, 35, 104–109. [Google Scholar] [PubMed]
- Benson, C.; Mifflin, K.; Kerr, B.; Jesudasan, S.J.; Dursun, S.; Baker, G. Biogenic Amines and the Amino Acids GABA and Glutamate: Relationships with Pain and Depression. Mod. Trends Pharmacopsychiatry 2015, 30, 67–79. [Google Scholar] [PubMed]
- Jeon, S.W.; Kim, Y.K. Neuroinflammation and cytokine abnormality in major depression: Cause or consequence in that illness? World J. Psychiatry 2016, 6, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Oakes, P.; Loukas, M.; Oskouian, R.J.; Tubbs, R.S. The neuroanatomy of depression: A review. Clin. Anat. 2017, 30, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.B.; Hou, D.R.; Tang, Q.P. Research progress of depression and the application of esketamine. J. South. Med. Univ. 2016, 37, 567. [Google Scholar]
- Zhang, H.; Zhou, Z.; Chen, Z.; Zhong, Z.; Li, Z. Ginsenoside Rg3 exerts anti-depressive effect on an NMDA-treated cell model and a chronic mild stress animal model. J. Pharmacol. Sci. 2017, 134, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Allison, D.J.; Ditor, D.S. The common inflammatory etiology of depression and cognitive impairment: A therapeutic target. J. Neuroinflamm. 2014, 11, 151. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M. Neurodegeneration in Parkinson’s disease: Interactions of oxidative stress, tryptophan catabolites and depression with mitochondria and sirtuins. Mol. Neurobiol. 2014, 49, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M. TRYCAT pathways link peripheral inflammation, nicotine, somatization and depression in the etiology and course of Parkinson’s disease. CNS Neurol. Disord. Drug Targets 2014, 13, 137–149. [Google Scholar] [CrossRef]
- Anderson, G.; Seo, M.; Berk, M.; Carvalho, A.F.; Maes, M. Gut Permeability and Microbiota in Parkinson’s Disease: Role of Depression, Tryptophan Catabolites, Oxidative and Nitrosative Stress and Melatonergic Pathways. Curr. Pharm. Des. 2016, 22, 6142–6151. [Google Scholar] [CrossRef] [PubMed]
- Braga, J.; Campar, A. Biological causes of depression in Systemic Lupus Erythematosus. Acta Reumatol. Port. 2014, 39, 218–226. [Google Scholar] [PubMed]
- Cui, J.; Jiang, L.; Xiang, H. Ginsenoside Rb3 exerts antidepressant-like effects in several animal models. J. Psychopharmacol. 2012, 26, 697–713. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Liu, Y.; Zhang, B.; Huang, J.; Li, Y.; Yang, B.; Huang, Z.; Xiang, F.; Zhang, H. The antidepressant effects and mechanism of action of total saponins from the caudexes and leaves of Panax notoginseng in animal models of depression. Phytomedicine 2011, 18, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, Z.; Zhou, Z.; Yang, H.; Zhong, Z.; Lou, C. Antidepressant-like effects of ginsenosides: A comparison of ginsenoside Rb3 and its four deglycosylated derivatives, Rg3, Rh2, K, and 20(S)-protopanaxadiol in mice models of despair. Pharmacol. Biochem. Behav. 2016, 140, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Su, P.; Wang, L.; Du, S.J.; Xin, W.F.; Zhang, W.S. Advance in studies of Panax notoginseng saponins on pharmacological mechanism of nervous system disease. Zhongguo Zhong Yao Za Zhi 2014, 39, 4516–4521. [Google Scholar] [PubMed]
- Bahramsoltani, R.; Farzaei, M.H.; Farahani, M.S.; Rahimi, R. Phytochemical constituents as future antidepressants: A comprehensive review. Rev. Neurosci. 2015, 26, 699–719. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Guo, R.; Zhou, G.; Zhou, X.; Kou, Z.; Sui, F.; Li, C.; Tang, L.; Wang, Z. Traditional uses, botany, phytochemistry, pharmacology and toxicology of Panax notoginseng (Burk.) F.H. Chen: A review. J. Ethnopharmacol. 2016, 188, 234–258. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ding, L.; Wang, B.; Ren, G.; Sun, A.; Deng, C.; Wei, X.; Mani, S.; Wang, Z.; Dou, W. Notoginsenoside R1 attenuates experimental inflammatory bowel disease via pregnane X receptor activation. J. Pharmacol. Exp. Ther. 2015, 352, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Liang, Y.; Kang, A.; Ma, S.J.; Xing, L.; Zhou, Y.Y.; Dai, C.; Xie, H.; Xie, L.; Wang, G.J.; et al. Peripheral immunomodulation with ginsenoside Rg1 ameliorates neuroinflammation-induced behavioral deficits in rats. Neuroscience 2014, 256, 210–222. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Kim, K.Y.; Jeong, H.J.; Kim, H.M. Antidepressant-like effect of altered Korean red ginseng in mice. Behav. Med. 2011, 37, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Zhou, X.P.; Lin, L.L.; Xu, J.Y.; Shen, C.S.; Feng, Z.; Zhou, L.L.; Shan, J.J. Metabolomics analysis of Tripterygium wilfordii formulation based on theory of detoxicity compatibility. Zhongguo Zhong Yao Za Zhi 2016, 41, 1124–1129. [Google Scholar] [PubMed]
- Pang, H.H.; Li, M.Y.; Wang, Y.; Tang, M.K.; Ma, C.H.; Huang, J.M. Effect of compatible herbs on the pharmacokinetics of effective components of Panax notoginseng in Fufang Xueshuantong Capsule. J. Zhejiang Univ. Sci. B 2017, 18, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.; Pang, H.; Du, S.; Lu, Y.; Zhang, L.; Wu, H.; Guo, S.; Wang, M.; Zhang, Q. Effect of Panax notoginseng saponins on the pharmacokinetics of aspirin in rats. J. Chromatogr. B 2017, 1040, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zheng, M.X.; Chen, H.E.; Wu, C.Y.; Wang, W.T. Effect of panax notoginseng saponins injection on the p38MAPK pathway in lung tissue in a rat model of hypoxic pulmonary hypertension. Chin. J. Integr. Med. 2015, 21, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.L.; Wang, Y.; Li, Y.B.; Hu, X.L.; Wang, S.L. Protective effect of notoginsenoside R1 on neuron injury induced by OGD/R through ATF6/Akt signaling pathway. Zhongguo Zhong Yao Za Zhi 2017, 42, 1167–1174. [Google Scholar] [PubMed]
- Yang, X.; Yang, S.; Hong, C.; Yu, W.; Guonian, W. Panax Notoginseng Saponins attenuates sevofluraneinduced nerve cell injury by modulating AKT signaling pathway. Mol. Med. Rep. 2017, 16, 7829–7834. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.A.; Kim, D.-E.; Choi, H.J.; Kim, N.J.; Kim, D.-H. Anti-stress Effects of 20(S)-Protopanaxadiol and 20(S)-Protopanaxatriol in Immobilized Mice. Biol. Pharm. Bull. 2015, 38, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Albert, P.R.; Fiori, L.M. Transcriptional dys-regulation in anxiety and major depression: 5-HT1A gene promoter architecture as a therapeutic opportunity. Curr. Pharm Des. 2014, 20, 3738–3750. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.Y.; Zhang, L.J.; Liu, M.C. Effect of ginsenoside pre-treatment on 5-hydroxytryptamine system in SD rats with myocardial infarction and depression. Zhonghua Nei Ke Za Zhi 2016, 55, 700–704. [Google Scholar] [PubMed]
- Jiang, B.; Xiong, Z.; Yang, J.; Wang, W.; Wang, Y.; Hu, Z.L.; Wang, F.; Chen, J.G. Antidepressant-like effects of ginsenoside Rg1 are due to activation of the BDNF signalling pathway and neurogenesis in the hippocampus. Br. J. Pharmacol. 2012, 166, 1872–1887. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Shim, I.; Lee, H.; Hahm, D.H. Effect of ginsenoside Re on depression- and anxiety-like behaviors and cognition memory deficit induced by repeated immobilization in rats. J. Microbiol. Biotechnol. 2012, 22, 708–720. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Qi, Y.; Cheng, Z.; Zhu, X.; Fan, C.; Yu, S.Y. The effects of ginsenoside Rg1 on chronic stress induced depression-like behaviors, BDNF expression and the phosphorylation of PKA and CREB in rats. Neuroscience 2016, 322, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Si, Y.; Zhu, J.; Huang, X.; Zhu, P.; Xie, C. Effects of Panax notoginseng saponins on proliferation and differentiation of rat embryonic cortical neural stem cells. J. Chin. Med. Assoc. 2016, 79, 256–263. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhao, M.; Chen, L.; Yang, J.; Han, D.; Fang, D.; Qiu, X.; Yang, X.; Qiao, Z.; Ma, J.; Wang, L.; et al. BDNF Val66Met polymorphism, life stress and depression: A meta-analysis of gene-environment interaction. J. Affect. Disord. 2017, 227, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Berk, M.; Dean, O.; Moylan, S.; Maes, M. Role of immune-inflammatory and oxidative and nitrosative stress pathways in the etiology of depression: therapeutic implications. CNS Drugs 2014, 28, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M.; Berk, M. Schizophrenia is primed for an increased expression of depression through activation of immuno-inflammatory, oxidative and nitrosative stress, and tryptophan catabolite pathways. Prog. Neuropsychopharm. Biol. Psychiatry 2013, 42, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.G.; Jin, Y.H.; Lee, H.; Oh, T.W.; Yim, N.H.; Cho, W.K.; Ma, J.Y. Protective Effect of Panax notoginseng Root Water Extract against Influenza A Virus Infection by Enhancing Antiviral Interferon-Mediated Immune Responses and Natural Killer Cell Activity. Front. Immunol. 2017, 8, 1542. [Google Scholar] [CrossRef] [PubMed]
- Han, J.Y.; Li, Q.; Ma, Z.Z.; Fan, J.Y. Effects and mechanisms of compound Chinese medicine and major ingredients on microcirculatory dysfunction and organ injury induced by ischemia/reperfusion. Pharmacol. Ther. 2017, 177, 146–173. [Google Scholar] [CrossRef] [PubMed]
- Kang, A.; Hao, H.; Zheng, X.; Liang, Y.; Xie, Y.; Xie, T.; Dai, C.; Zhao, Q.; Wu, X.; Xie, L.; et al. Peripheral anti-inflammatory effects explain the ginsenosides paradox between poor brain distribution and anti-depression efficacy. J. Neuroinflamm. 2011, 8, 100. [Google Scholar] [CrossRef] [PubMed]
- Kang, A.; Xie, T.; Zhu, D.; Shan, J.; Di, L.; Zheng, X. Suppressive Effect of Ginsenoside Rg3 against Lipopolysaccharide-Induced Depression-Like Behavior and Neuroinflammation in Mice. J. Agric. Food Chem. 2017, 65, 6861–6869. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Zhang, Y.; Lu, J.; Geng, Z.; Jia, L.; Rong, X.; Wang, Z.; Zhao, Q.; Wu, R.; Chu, M.; et al. Panax Notoginseng Saponins Ameliorates Coxsackievirus B3-Induced Myocarditis by Activating the Cystathionine-gamma-Lyase/Hydrogen Sulfide Pathway. J. Cardiovasc. Transl. Res. 2015, 8, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lu, L.; Wang, Y.; Wu, Y.; Han, J.; Wang, W.; Li, C.; Tu, P. Qishenyiqi Dropping Pill attenuates myocardial fibrosis in rats by inhibiting RAAS-mediated arachidonic acid inflammation. J. Ethnopharmacol. 2015, 176, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Wenxi, D.; Shufang, D.; Xiaoling, Y.; Liming, Y. Panax notoginseng saponins suppress radiation-induced osteoporosis by regulating bone formation and resorption. Phytomedicine 2015, 22, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.X.; Chen, J.X. Negative chronotropic and inotropic effects of Panax notoginseng saponins. Zhongguo Yao Li Xue Bao 1988, 9, 409–412. [Google Scholar] [PubMed]
- Ge, Z.R.; Xu, M.C.; Huang, Y.U.; Zhang, C.J.; Lin, J.E.; Ruan, C.W. Cardioprotective effect of notoginsenoside R1 in a rabbit lung remote ischemic postconditioning model via activation of the TGF-beta1/TAK1 signaling pathway. Exp. Ther. Med. 2016, 11, 2341–2348. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Wan, D.; Shao, L.; Dai, J.; Jiang, C. Notoginsenoside R1 stimulates osteogenic function in primary osteoblasts via estrogen receptor signaling. Biochem. Biophys. Res. Commun. 2015, 466, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ren, Y.; Xing, L.; Dai, X.; Liu, S.; Yu, B.; Wang, Y. Endothelium-dependent vasodilation effects of Panax notoginseng and its main components are mediated by nitric oxide and cyclooxygenase pathways. Exp. Ther. Med. 2016, 12, 3998–4006. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Lin, L.; Tang, L.; Zheng, M.; Ma, Y.; Huang, L.; Meng, W.; Wang, W. Notoginsenoside R1 attenuates hypoxia and hypercapnia-induced vasoconstriction in isolated rat pulmonary arterial rings by reducing the expression of ERK. Am. J. Chin. Med. 2014, 42, 799–816. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Min, S.; Wei, K.; Zhang, J.; Liu, Y. Propofol interacts with stimulus intensities of electroconvulsive shock to regulate behavior and hippocampal BDNF in a rat model of depression. Psychiatry Res. 2012, 198, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Gazal, M.; Ortmann, C.F.; Martins, F.A.; Streck, E.L.; Quevedo, J.; de Campos, A.M.; Stefanello, F.M.; Kaster, M.P.; Ghisleni, G.; Reginatto, F.H.; et al. Antidepressant-like effects of aqueous extract from Cecropia pachystachya leaves in a mouse model of chronic unpredictable stress. Brain Res. Bull. 2014, 108, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Pechlivanova, D.; Tchekalarova, J.; Nikolov, R.; Yakimova, K. Dose-dependent effects of caffeine on behavior and thermoregulation in a chronic unpredictable stress model of depression in rats. Behav. Brain Res. 2010, 209, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Taksande, B.G.; Faldu, D.S.; Dixit, M.P.; Sakaria, J.N.; Aglawe, M.M.; Umekar, M.J.; Kotagale, N.R. Agmatine attenuates chronic unpredictable mild stress induced behavioral alteration in mice. Eur. J. Pharmacol. 2013, 720, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Wang, J.Y.; Luo, F. Depression shows divergent effects on evoked and spontaneous pain behaviors in rats. J. Pain 2010, 11, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.F.; Zhu, C.H.; Guo, J.Y. Effect of ginsenoside Rg1 on behaviors and hippocampal amino acids in depressive-like rats. Zhongguo Zhong Yao Za Zhi 2012, 37, 3117–3121. [Google Scholar] [PubMed]
- Carr, M.N.; Bekku, N.; Yoshimura, H. Identification of anxiolytic ingredients in ginseng root using the elevated plus-maze test in mice. Eur. J. Pharmacol. 2006, 531, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Cha, H.Y.; Park, J.H.; Hong, J.T.; Yoo, H.S.; Song, S.; Hwang, B.Y.; Eun, J.S.; Oh, K.W. Anxiolytic-like effects of ginsenosides on the elevated plus-maze model in mice. Biol. Pharm. Bull. 2005, 28, 1621–1625. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.P.; Ding, H.; Lu, J.D.; Tang, Y.H.; Deng, B.X.; Deng, C.Q. Effects of the Combination of the Main Active Components of Astragalus and Panax notoginseng on Inflammation and Apoptosis of Nerve Cell after Cerebral Ischemia-Reperfusion. Am. J. Chin. Med. 2015, 43, 1419–1438. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.P.; Ding, H.; Wang, B.; Qiu, Y.Y.; Tang, Y.H.; Zeng, R.; Deng, C.Q. Effects of the main active components combinations of Astragalus and Panax notoginseng on energy metabolism in brain tissues after cerebral ischemia-reperfusion in mice. Pharmacogn. Mag. 2015, 11, 732–739. [Google Scholar] [PubMed]
- Qi, H.; Huang, Y.; Yang, Y.; Dou, G.; Wan, F.; Zhang, W.; Yang, H.; Wang, L.; Wu, C.; Li, L. Anti-platelet activity of panaxatriol saponins is mediated by suppression of intracellular calcium mobilization and ERK2/p38 activation. BMC Complement. Altern. Med. 2016, 16, 174. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Guan, Y.; Ma, H.J.; Guo, Z.; He, R.R.; Zhang, Y. Influences of Panax notoginsenosid on spontaneous contraction of small intestine smooth muscle of rabbits in vitro. Zhongguo Ying Yong Sheng Li Xue Za Zhi 2010, 26, 117–120. [Google Scholar] [PubMed]
- You, Z.; Yao, Q.; Shen, J.; Gu, Z.; Xu, H.; Wu, Z.; Chen, C.; Li, L. Antidepressant-like effects of ginsenoside Rg3 in mice via activation of the hippocampal BDNF signaling cascade. J. Nat. Med. 2017, 71, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Leff-Gelman, P.; Mancilla-Herrera, I.; Flores-Ramos, M.; Cruz-Fuentes, C.; Reyes-Grajeda, J.P.; Garcia-Cuetara Mdel, P.; Bugnot-Perez, M.D.; Pulido-Ascencio, D.E. The Immune System and the Role of Inflammation in Perinatal Depression. Neurosci. Bull. 2016, 32, 398–420. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, D.; Hou, J.; Lei, H. Protective effects of geniposide and ginsenoside Rg1 combination treatment on rats following cerebral ischemia are mediated via microglial microRNA1555p inhibition. Mol. Med. Rep. 2018, 17, 3186–3193. [Google Scholar] [PubMed]
- Xu, C.; Teng, J.; Chen, W.; Ge, Q.; Yang, Z.; Yu, C.; Yang, Z.; Jia, W. 20(S)-protopanaxadiol, an active ginseng metabolite, exhibits strong antidepressant-like effects in animal tests. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 1402–1411. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Cheng, Y.; Li, T.; Dong, M.; Zhao, H.; Liu, G. Effects of notoginsenoside R1 on CYP1A2, CYP2C11, CYP2D1, and CYP3A1/2 activities in rats by cocktail probe drugs. Pharm. Biol. 2016, 54, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Dang, Y.; Zhu, C. Simultaneous determination of three major bioactive saponins of Panax notoginseng using liquid chromatography-tandem mass spectrometry and a pharmacokinetic study. Chin. Med. 2010, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Ducottet, C.; Aubert, A.; Belzung, C. Susceptibility to subchronic unpredictable stress is related to individual reactivity to threat stimuli in mice. Behav. Brain Res. 2004, 155, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, C.; Chen, S.; Li, Z.; Ma, L.; Jia, X.; Wang, K.; Bao, J.; Liang, Y.; Chen, M.; et al. Hormetic effect of panaxatriol saponins confers neuroprotection in PC12 cells and zebrafish through PI3K/AKT/mTOR and AMPK/SIRT1/FOXO3 pathways. Sci. Rep. 2017, 7, 41082. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Yu, W.; Yang, T.; Liu, W.; Zhao, Y.; Sun, Y.; Chai, L.; Gao, Y.; Dong, B.; Zhu, L. Panax notoginseng saponins provide neuroprotection by regulating NgR1/RhoA/ROCK2 pathway expression, in vitro and in vivo. J. Ethnopharmacol. 2016, 190, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Yamada, N.; Araki, H.; Yoshimura, H. Identification of antidepressant-like ingredients in ginseng root (Panax ginseng C.A. Meyer) using a menopausal depressive-like state in female mice: participation of 5-HT2A receptors. Psychopharmacology 2011, 216, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Choi, H.J.; Kim, N.J.; Kim, D.H. Anxiolytic-like effects of ginsenosides Rg3 and Rh2 from red ginseng in the elevated plus-maze model. Planta Med. 2009, 75, 836–839. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Wang, C.; Zhao, W.; Gao, S.; Cui, Z. Antidepressant-like effects of ginsenoside Rg5 in mice: Involving of hippocampus BDNF signaling pathway. Neurosci. Lett. 2017, 645, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Miao, B.; Song, X.; Jiang, Z. Inactivation of GABA(A) receptor reduces ginsenoside Rb3 neuroprotection in mouse hippocampal slices after oxygen-glucose deprivation. J. Ethnopharmacol. 2011, 133, 914–916. [Google Scholar] [CrossRef] [PubMed]
- Cheong, J.H.; Song, M.-R.; Lee, Y.S.; Her, Y.; Yim, D.S.; Ko, H.S.; Ryu, Y.S.; Choi, E.J.; Yoon, S.Y.; Choi, J.H. Anxiolytic and Antidepressant Activities of Ginsenoside Rb1. Biomol. Ther. 2007, 15, 27–33. [Google Scholar]
- Churchill, J.D.; Gerson, J.L.; Hinton, K.A.; Mifek, J.L.; Walter, M.J.; Winslow, C.L.; Deyo, R.A. The nootropic properties of ginseng saponin Rb1 are linked to effects on anxiety. Integr. Physiol. Behav. Sci. 2002, 37, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.J.; Song, L.; Guo, L.C.; Yin, M.; Sun, Y.N. Induction of differentiation by panaxydol in human hepatocarcinoma SMMC-7721 cells via cAMP and MAP kinase dependent mechanism. Yakugaku Zasshi 2011, 131, 993–1000. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wu, C.F.; Liu, Y.L.; Song, M.; Liu, W.; Wang, J.H.; Li, X.; Yang, J.Y. Protective effects of pseudoginsenoside-F11 on methamphetamine-induced neurotoxicity in mice. Pharmacol. Biochem. Behav. 2003, 76, 103–109. [Google Scholar] [CrossRef]
- Lee, B.; Sur, B.; Cho, S.G.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.H. Ginsenoside Rb1 rescues anxiety-like responses in a rat model of post-traumatic stress disorder. J. Nat. Med. 2016, 70, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.-H.; Kim, H.-J.; Chung, L.; Nah, S.-Y. Ginsenoside Rg(3) regulates GABA(A) receptor channel activity: Involvement of interaction with the gamma(2) subunit. Eur. J. Pharmacol. 2013, 705, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Yue, S.-J.; Xin, L.-T.; Fan, Y.-C.; Li, S.-J.; Tang, Y.-P.; Duan, J.-A.; Guan, H.-S.; Wang, C.-Y. Herb pair Danggui-Honghua: Mechanisms underlying blood stasis syndrome by system pharmacology approach. Sci. Rep. 2017, 7, 40318. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhao, P.; Li, Y.; Tian, Y.; Wang, Y. Systems pharmacology-based dissection of mechanisms of Chinese medicinal formula Bufei Yishen as an effective treatment for chronic obstructive pulmonary disease. Sci. Rep. 2015, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Rhee, D.K. Effects of ginseng on stress-related depression, anxiety, and the hypothalamic-pituitary-adrenal axis. J. Ginseng Res. 2017, 41, 589–594. [Google Scholar] [CrossRef] [PubMed]
| Name | Methods | Models | Neurobehavioral Effects and Mechanisms |
|---|---|---|---|
| PNS (58.6%) | OFT; TST; FST; SPT [26,53] | CMS-induced mice; CUMS-induced rats [26,53] | ↓ Immobility time in FST and TST; ↑ sucrose intake in sucrose preference test; ↑ level of animal activity; ↑ Expression of BDNF [40,60,68,69,70]. |
| Purified PNS | SPT; OFT [45,71] | CUMS-induced SD rats [45,71] | Regulate upstream neural signal transduction pathways: AC/cAMP/PKA, TrKB/MAPK/PSK and Ca2+/CaM/CaMK signaling pathway; ↓ Concentration of Ca2+ in nerve cells; ↑ release of neurotransmitter 5-HT, DA, and NE [45,71]. |
| PNS and GTS | FST; SPT [53,75,76,77,78]. | LPS-induced KM mice; CD-1 mice [53,75,76,77,78] | ↓ mRNA of IL-1β, IL-6, TNF-α, and IDO; ↓ Inflammation in the brain [53,75,76,77,78]. |
| Ginsenoside Rg1 | OFT; FST; SPT [19,45] | CMS and CUMS-induced depression Animal Model [19,45] | ↓ CMS-induced increasement of corticosterone levels in serum; ↑ CUMS-induced CREB phosphorylation in the amygdala of the brain [45]; ↑ expression of BDNF [33,43]; regulate hyperactivity of the HPA axis in depressed states; ↑neuronal status, neurons quantity and density of dendritic spines [19,45]. |
| Ginsenoside Rg1 | FST; SPT [79] | CMS and CUMS-induced depression animal model [80] | ↑ Expression of connexin and gap junction of astrocytes in the prefrontal cortex of the brain; ↑ dense ultrastructure; ↓ permeability and depression-like behaviors induced by CUS in rats [79]. |
| Ginsenoside Rg1 | FST; SPT [10] | Cerebral inflammatory model mice [10] | ↓ Recruitment of Ly6C (hi) monocytes in inflammatory brain model mice [10,16]; activate THP-1 monocytes; blocks the feedback-regulated release of CCL2 from astroglial cells; ↓ CCL2 signaling pathways: p38/MAPK and PI3K/Akt activities [10,81]. |
| Ginsenoside Rg1 | TST; FST; SPT [15,67] | CUMS-induced depression rats [67] | ↓ Contents of Glu and Asp in hippocampus, ↑ contents of GABA and Tau; ↓ depression-like Behavior in CUMS model rats [15,67] |
| Ginsenoside Rb1 and ginsenoside K | TST; FST [42] | CUMS-induced depression rats [42] | ↑ Expression of 5-HT2AR mRNA and activate of 5-HT2AR [42]; the antidepressant effects of Rb1 and the metabolite ginsenoside K may be antagonized by 5-HT2AR antagonists (Ritanerin), indicating that Rb1 has a similar 5-HT transmitter activation effect [68,82,83]. |
| Ginsenoside Rg3 and ginsenoside Rh2 | OFT; SPT [19,53,74] | LPS-induced mice [19,53,74] and CSDS-induced mice [74] | ↓ IL-6 and TNF-α in plasma and the expression of indoleamine 2,3-dioxygenase (IDO) in brain; ↓ turnover of tryptophan and 5-HT in hippocampal tissue; regulate secretory activities of microglia and transcription of NF-kappa-B in nuclear; ↑ expression of BNDF; ↓ depressive behavior or symptoms [53,74,84]. |
| Ginsenoside Rg5 | OFT; FST; SPT [85] | CSDS-induced mice [85] | ↓ Trk and AchE; ↑expression of BNDF [69,85]. |
| Ginsenoside Re | OFT; TST [44] | CMS-induced rat model [44] | Regulate the central adrenergic system; ↓ expression of TH in the locus coeruleus area; ↓ decrease of BDNF in the hippocampus, and regulates the secretion of corticosterone from the HPA axis [44]. |
| Name | Methods | Models | Neurobehavioral Effects and Mechanisms |
|---|---|---|---|
| PNS | OFT; SPT [26,28] | l-DOPA-induced mice [26,28]; 5-HTP-induced rats [89] | ↓ Basal [Ca2+]i levels; ↓ immobility time; ↑ levels of 5-HT, DA and NE [26,28,89]. |
| Ginsenoside Rb1 | EPM test [44,91] | SPS model and rat model of post-traumatic stress disorder [44,91] | ↓ Anxiety index;↑ Risk assessment; ↓ grooming behaviors in EPMT; ↑ total number of line crossings of an open field after SPS; ↓ SPS-induced decreasement in hypothalamic neuropeptide Y expression; ↑ in locus cerulean tyrosine hydroxylase expression; ↓ expression of BDNF [44,91]. |
| Ginsenoside Rb1, Re; ginsenoside Rh1, PPD | EPM test [27,40] | Immobilization stress-induced ICR mice [27,40] | ↑ Time spent in open arms and open arm entries in EPM tests; ↓ Immobilization stress-induced serum levels of corticosterone and IL-6; ↓ anxiolytic effect via γ-aminobutyrate A (GABA A) receptor(s) [27,40]. |
| Ginsenoside Rg3 | Two electrode voltage-clamp technique [86,92] | Xenopus laevis frogs [86,92] | ↓ Anxiolytic effect via γ-aminobutyrate A (GABA A) receptor(s) [86,92]. |
| Ginsenoside Re | FST; EPM; AAT [44] | Repeated immobilization stress-induced rats [44] | ↓ Stress-induced behavioral deficits in these behavioral tests; ↓ TH expression in LC; ↓ mRNA expression of BDNF in the hippocampus; modulate central noradrenergic system in rats [44]. |
| Ginsenoside Rg3 and ginsenoside Rh2 | EPM test [27,84] | Male ICR mice [27,84] | ↑ Time spent on the open arms and the number of open-arm entries; antagonize GABA/benzodiazepines [27,84]. |
| Ginsenoside Rb1, Rg1, and Ro | EPM test [68,84] | Male ICR albino mice [68,84] | ↑ Both the frequency and duration of open arm entries [68,84]. |
| Pseudoginsenoside-F11 | Light-dark box test; FST [90] | MA-induced behavioral and neurochemical toxicities in mice [90] | ↓ Anxiety-like behavior induced by methamphetamine (MA); ↓ MA-induced prolonged latency; ↓ the error counts; ↓ contents of DOPAC, HVA, and 5-HIAA in the brain of MA-treated mice; antagonize decreases of DA [90]. |
| No. | Name | Structure |
|---|---|---|
| PNS-1 | Ginsenoside Rg1 | 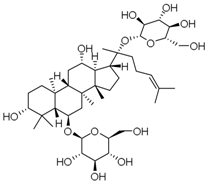 |
| PNS-2 | Ginsenoside Rg3 |  |
| PNS-3 | Ginsenoside Rg5 | 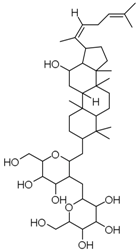 |
| PNS-4 | Ginsenoside Rb1 | 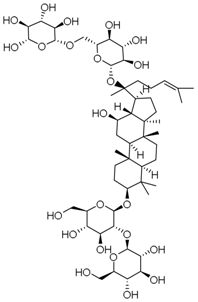 |
| PNS-5 | Ginsenoside Rb3 | 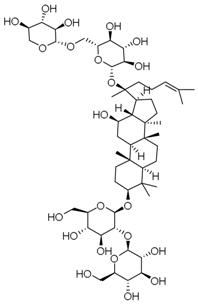 |
| PNS-6 | Ginsenoside Re |  |
| PNS-7 | Ginsenoside Rh1 | 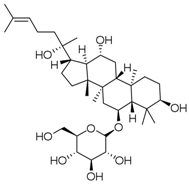 |
| PNS-8 | Ginsenoside Rh2 | 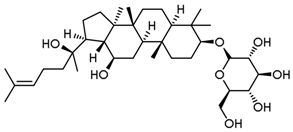 |
| PNS-9 | Pseudoginsenoside-F11 | 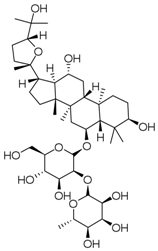 |
| PNS-10 | Ginsenoside Ro | 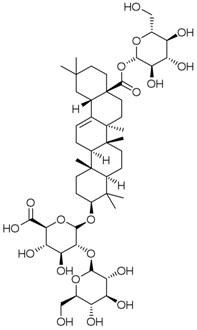 |
| PNS-11 | Ginsenoside K |  |
| PNS-12 | Notoginsenoside R1 | 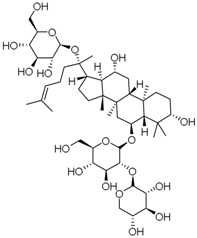 |
| Node1 | Node2 | Node1 Accession | Node2 Accession | Score |
|---|---|---|---|---|
| APAF1 | BAK1 | ENSP00000448165 | ENSP00000353878 | 0.997 |
| APAF1 | BCL2 | ENSP00000448165 | ENSP00000329623 | 0.999 |
| APAF1 | BCL2A1 | ENSP00000448165 | ENSP00000267953 | 0.912 |
| APAF1 | BIK | ENSP00000448165 | ENSP00000216115 | 0.932 |
| APAF1 | JUN | ENSP00000448165 | ENSP00000360266 | 0.582 |
| APAF1 | MAPK8 | ENSP00000448165 | ENSP00000353483 | 0.611 |
| APAF1 | MCL1 | ENSP00000448165 | ENSP00000358022 | 0.95 |
| APAF1 | NFKB1 | ENSP00000448165 | ENSP00000226574 | 0.608 |
| APAF1 | TP53 | ENSP00000448165 | ENSP00000269305 | 0.996 |
| BAK1 | APAF1 | ENSP00000353878 | ENSP00000448165 | 0.997 |
| BAK1 | BBC3 | ENSP00000353878 | ENSP00000404503 | 0.414 |
| BAK1 | BCL2 | ENSP00000353878 | ENSP00000329623 | 0.999 |
| BAK1 | BCL2A1 | ENSP00000353878 | ENSP00000267953 | 0.987 |
| BAK1 | BIK | ENSP00000353878 | ENSP00000216115 | 0.583 |
| BAK1 | JUN | ENSP00000353878 | ENSP00000360266 | 0.852 |
| BAK1 | JUNB | ENSP00000353878 | ENSP00000303315 | 0.658 |
| BAK1 | JUND | ENSP00000353878 | ENSP00000252818 | 0.671 |
| BAK1 | MAPK8 | ENSP00000353878 | ENSP00000353483 | 0.83 |
| BAK1 | MCL1 | ENSP00000353878 | ENSP00000358022 | 0.99 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, W.; Meng, X.; Zhai, Y.; Zhou, P.; Ye, T.; Wang, Z.; Sun, G.; Sun, X. Panax Notoginseng Saponins: A Review of Its Mechanisms of Antidepressant or Anxiolytic Effects and Network Analysis on Phytochemistry and Pharmacology. Molecules 2018, 23, 940. https://doi.org/10.3390/molecules23040940
Xie W, Meng X, Zhai Y, Zhou P, Ye T, Wang Z, Sun G, Sun X. Panax Notoginseng Saponins: A Review of Its Mechanisms of Antidepressant or Anxiolytic Effects and Network Analysis on Phytochemistry and Pharmacology. Molecules. 2018; 23(4):940. https://doi.org/10.3390/molecules23040940
Chicago/Turabian StyleXie, Weijie, Xiangbao Meng, Yadong Zhai, Ping Zhou, Tianyuan Ye, Zhen Wang, Guibo Sun, and Xiaobo Sun. 2018. "Panax Notoginseng Saponins: A Review of Its Mechanisms of Antidepressant or Anxiolytic Effects and Network Analysis on Phytochemistry and Pharmacology" Molecules 23, no. 4: 940. https://doi.org/10.3390/molecules23040940
APA StyleXie, W., Meng, X., Zhai, Y., Zhou, P., Ye, T., Wang, Z., Sun, G., & Sun, X. (2018). Panax Notoginseng Saponins: A Review of Its Mechanisms of Antidepressant or Anxiolytic Effects and Network Analysis on Phytochemistry and Pharmacology. Molecules, 23(4), 940. https://doi.org/10.3390/molecules23040940





