Metabolomics Strategy Using High Resolution Mass Spectrometry Reveals Novel Biomarkers and Pain-Relief Effect of Traditional Chinese Medicine Prescription Wu-Zhu-Yu Decoction Acting on Headache Modelling Rats
Abstract
1. Introduction
2. Results
2.1. Behavior Study Results
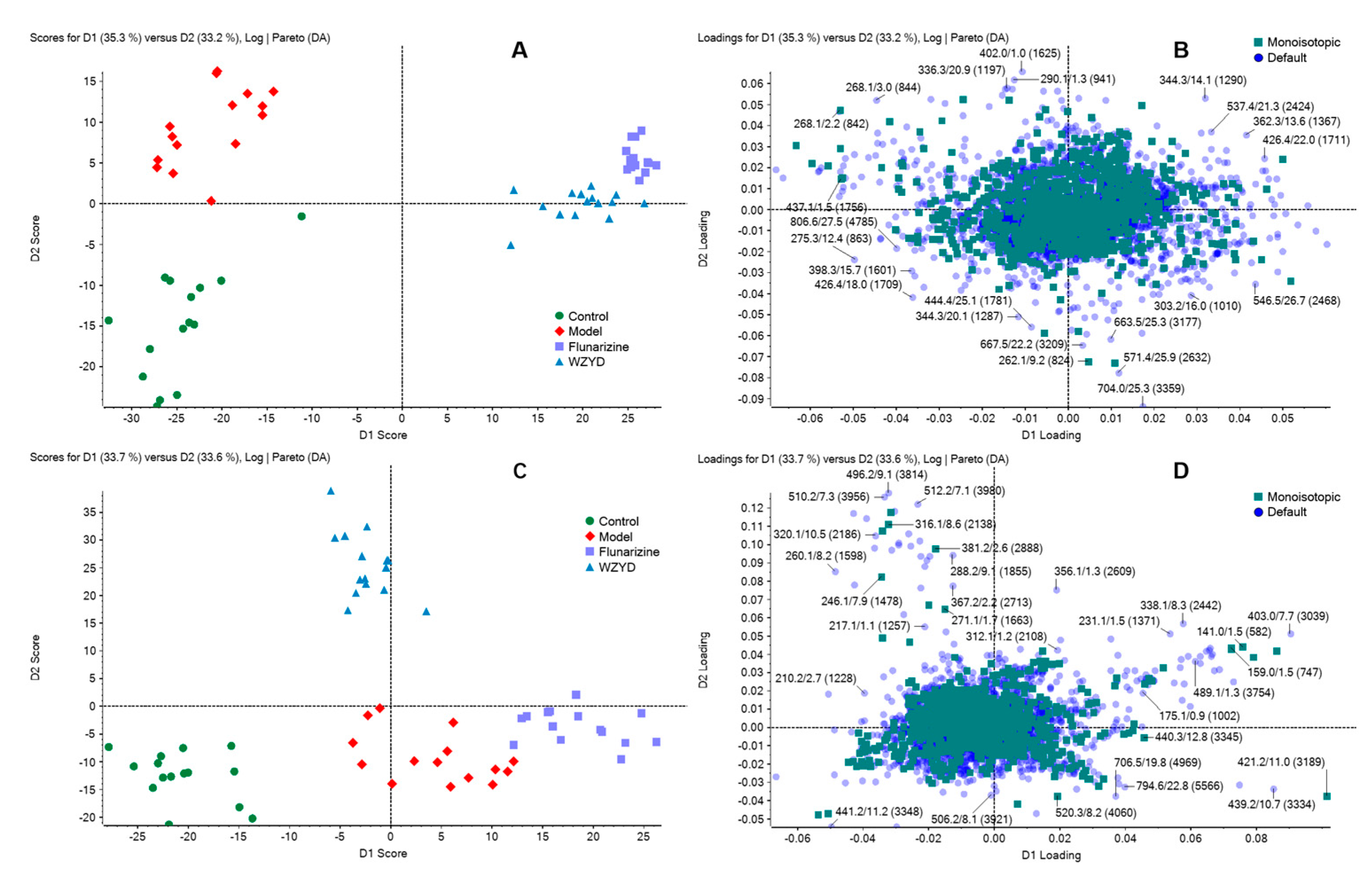
2.2. Multivariate Statistical Analysis of Metabolite Profiling
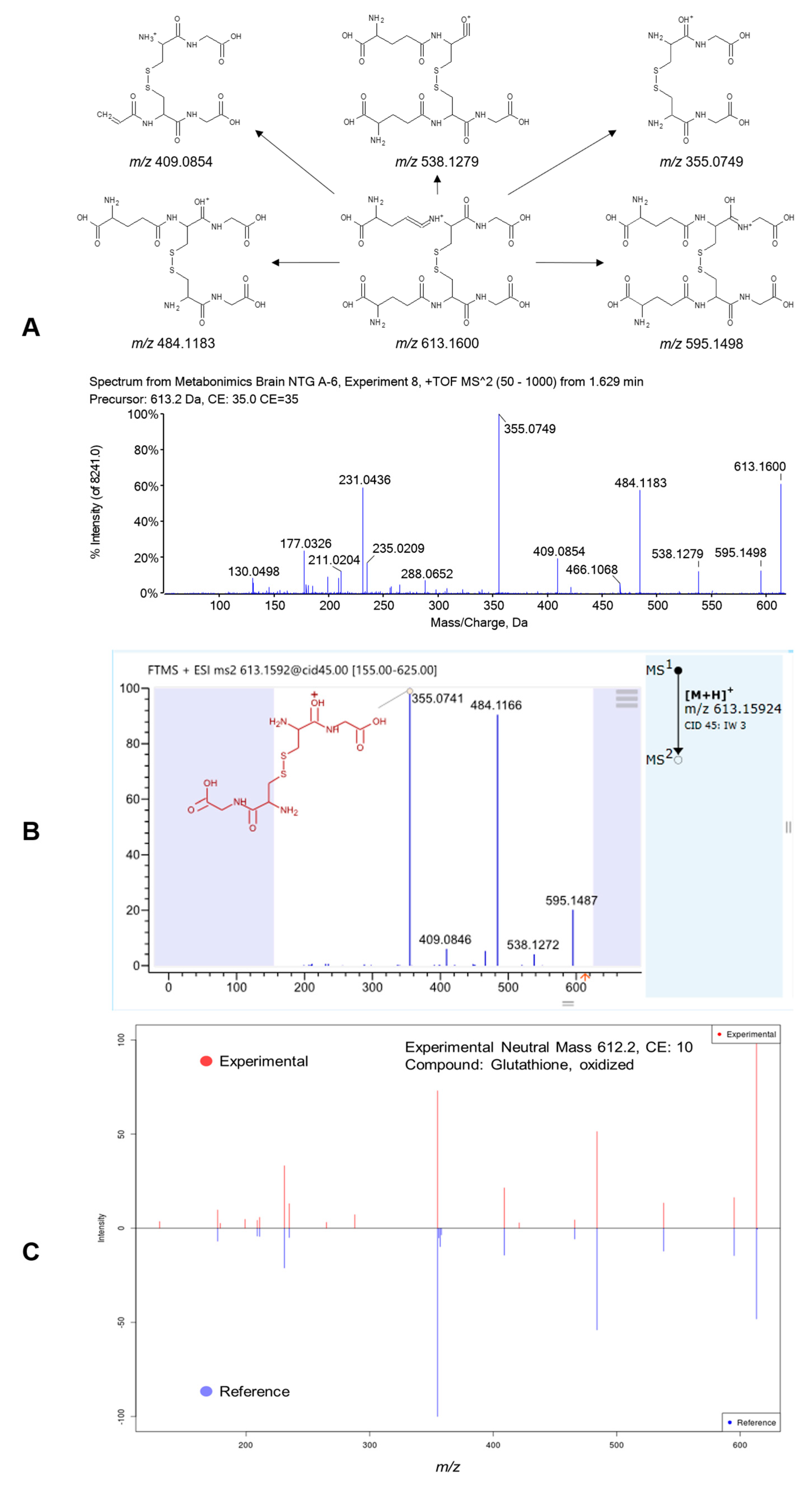
2.3. Metabolite Identification
2.4. Differential Metabolites Related to Headache
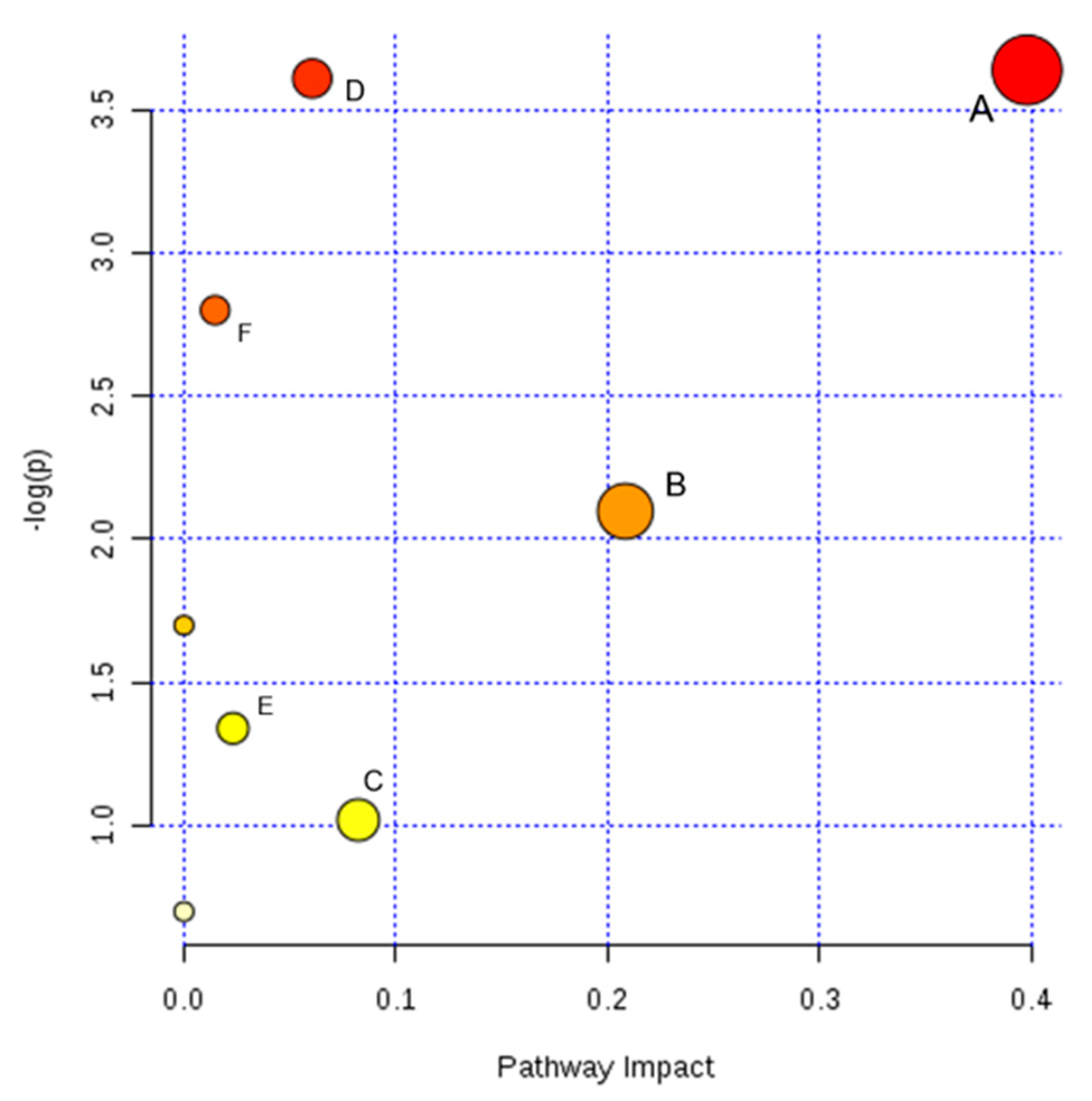
2.5. Pathway Analysis and Metabolic Network
3. Discussion
3.1. Characterized Potential Biomarkers
3.2. Possible Mechanism of Pain-Relif Action by WZYD
4. Materials and Methods
4.1. Materials and Preparation of Wu-Zhu-Yu Decoction
4.2. Animal Modelling and Treatments
4.3. Collection and Preparation of Biosamples
4.4. Liquid Chromatography-High Resolution Mass Mpectrometry Analysis
4.5. Data Processing and Multivariate Data Analysis
4.6. Identification of Biomarkers and Metabolic Pathway
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
Abbreviations
| 5-HIAA | 5-hydroxyindoleacetic acid |
| 5-HT | 5-hydroxytryptamine |
| AMP | adenosine monophosphate |
| CID | collision induced dissociation |
| CNS | central nervous system |
| DBS | dynamic background subtraction |
| GPC | glycerophosphocholine |
| GSH | glutathione |
| GSSG | oxidized glutathione |
| HCA | hierarchical cluster analysis |
| HDAC | histone deacetylases |
| IAA | indoleacetic acid |
| IDA | information dependent acquisition |
| LC-HRMS | liquid chromatography-high resolution mass spectrometry |
| NAD | nicotinamide adenine dinucleotide |
| NO | nitric oxide |
| NTG | nitroglycerin |
| PCA-DA | principal component analysis–discriminant analysis |
| PLS-DA | partial least squares-discriminant analysis |
| TCM | Traditional Chinese medicine |
| QTOF | quadrupole time-of-flight |
| WZYD | Wu-Zhu-Yu decoction |
References
- Odaguchi, H.; Wakasugi, A.; Ito, H.; Shoda, H.; Gono, Y.; Sakai, F.; Hanawa, T. The efficacy of goshuyuto, a typical Kampo (Japanese herbal medicine) formula, in preventing episodes of headache. Curr. Med. Res. Opin. 2006, 22, 11. [Google Scholar] [CrossRef] [PubMed]
- Schwedt, T.J. Chronic migraine. BMJ 2014, 348, g1416. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, M.A.; Buzzi, M.G. Migraine general aspects. Handb. Clin. Neurol. 2010, 97, 253–266. [Google Scholar] [PubMed]
- World Health Organization (WHO). Headache disorders. In Fact Sheet No. 277; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Goadsby, P.J. The vascular theory of migraine—A great story wrecked by the facts. Brain J. Neurol. 2009, 132, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Brennan, K.C.; Charles, A. An update on the blood vessel in migraine. Curr. Opin. Neurol. 2010, 23, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Lipton, R.B.; Ferrari, M.D. Migraine—Current understanding and treatment. N. Engl. J. Med. 2002, 346, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Olesen, J. The Headaches; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; p. 276. [Google Scholar]
- Gasparini, C.F.; Smith, R.A.; Griffiths, L.R. Genetic and biochemical changes of the serotonergic system in migraine pathobiology. J. Headache Pain 2017, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Liu, Q.; Zhao, H.; Zhou, X.; Sun, H.; Nan, Y.; Zou, S.; Ma, C.W.; Wang, X. Phenotypic characterization of nanshi oral liquid alters metabolic signatures during disease prevention. Sci. Rep. 2016, 6, 19333. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Zhang, A.; Han, Y.; Lu, S.; Kong, L.; Han, J.; Liu, Z.; Sun, H.; Wang, X. Metabolomics approach to explore the effects of Kai-Xin-San on Alzheimer’s disease using UPLC/ESI-Q-TOF mass spectrometry. J. Chromatogr. B 2016, 1015–1016, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Lionetto, L.; Gentile, G.; Bellei, E.; Capi, M.; Sabato, D.; Marsibilio, F.; Simmaco, M.; Pini, L.A.; Martelletti, P. The omics in migraine. J. Headache Pain 2013, 14, 55. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Zhang, A.; Sun, H.; Zhou, X.; Guan, Y.; Liu, Q.; Kong, L.; Wang, X. Metabolomics-proteomics profiles delineate metabolic changes in kidney fibrosis disease. Proteomics 2015, 15, 3699–3710. [Google Scholar] [CrossRef] [PubMed]
- Nan, Y.; Zhou, X.; Liu, Q.; Zhang, A.; Guan, Y.; Lin, S.; Kong, L.; Han, Y.; Wang, X. Serum metabolomics strategy for understanding pharmacological effects of ShenQi pill acting on kidney yang deficiency syndrome. J. Chromatogr. B 2016, 1026, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. Using MetaboAnalyst 3.0 for Comprehensive Metabolomics Data Analysis. Curr. Protoc. Bioinform. 2016, 55, 14.10.1–14.10.91. [Google Scholar]
- MetaboAnalyst—A Comprehensive Tool for Metabolomics Analysis and Interpretation. Available online: http://www.metaboanalyst.ca (accessed on 16 June 2017).
- Gokce Cokal, B.; Aytac, B.; Durak, Z.E.; Gunes, H.N.; Ozturk, B.; Keskin Guler, S.; Durak, I.; Yoldas, T.K. Serum oxidant and antioxidant status of patients with chronic tension-type headache: Possible effects of medical treatment. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2015, 36, 1771–1775. [Google Scholar]
- Srikiatkhachorn, A. Pathophysiology of chronic daily headache. Curr. Pain Headache Rep. 2001, 5, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Rogers, K.L.; Grice, I.D.; Griffiths, L.R. Inhibition of platelet aggregation and 5-HT release by extracts of Australian plants used traditionally as headache treatments. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2000, 9, 355–363. [Google Scholar] [CrossRef]
- Zwilling, D.; Huang, S.Y.; Sathyasaikumar, K.V.; Notarangelo, F.M.; Guidetti, P.; Wu, H.Q.; Lee, J.; Truong, J.; Andrews-Zwilling, Y.; Hsieh, E.W.; et al. Kynurenine 3-monooxygenase inhibition in blood ameliorates neurodegeneration. Cell 2011, 145, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Zadori, D.; Klivenyi, P.; Vamos, E.; Fulop, F.; Toldi, J.; Vecsei, L. Kynurenines in chronic neurodegenerative disorders: Future therapeutic strategies. J. Neural Transm. 2009, 116, 1403–1409. [Google Scholar] [CrossRef] [PubMed]
- Bynoe, M.S.; Viret, C.; Yan, A.; Kim, D.G. Adenosine receptor signaling: A key to opening the blood-brain door. Fluids Barriers CNS 2015, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Berger, F.; Ramirez-Hernandez, M.H.; Ziegler, M. The new life of a centenarian: Signalling functions of NAD(P). Trends Biochem. Sci. 2004, 29, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Corda, D.; Di Girolamo, M. Functional aspects of protein mono-ADP-ribosylation. EMBO J. 2003, 22, 1953–1958. [Google Scholar] [CrossRef] [PubMed]
- Tarighat Esfanjani, A.; Mahdavi, R.; Ebrahimi Mameghani, M.; Talebi, M.; Nikniaz, Z.; Safaiyan, A. The effects of magnesium, L-carnitine, and concurrent magnesium-L-carnitine supplementation in migraine prophylaxis. Biol. Trace Elem. Res. 2012, 150, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S.; Mehta, N.C.; Dabhi, A.S.; Lakhani, O.; Khilari, M.; Shah, N.D. The prevalence of headache may be related with the latitude: A possible role of Vitamin D insufficiency? J. Headache Pain 2010, 11, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Kjaergaard, M.; Eggen, A.E.; Mathiesen, E.B.; Jorde, R. Association between headache and serum 25-hydroxyvitamin D: The Tromso Study: Tromso 6. Headache 2012, 52, 1499–1505. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.K.; Hooten, W.M.; Schmidt, J.E.; Kerkvliet, J.L.; Townsend, C.O.; Bruce, B.K. Prevalence and clinical correlates of vitamin D inadequacy among patients with chronic pain. Pain Med. 2008, 9, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Li, Q.; Yin, Y.; Lv, C.; Sun, W.; He, B.; Liu, R.; Chen, X.; Bi, K. Simultaneous determination of three alkaloids, four ginsenosides and limonin in the plasma of normal and headache rats after oral administration of Wu-Zhu-Yu decoction by a novel ultra fast liquid chromatography-tandem mass spectrometry method: Application to a comparative pharmacokinetics and ethological study. J. Mass Spectrom. 2013, 48, 519–532. [Google Scholar] [PubMed]
Sample Availability: Samples of the reference standards mentioned in the paper are available from the authors. |

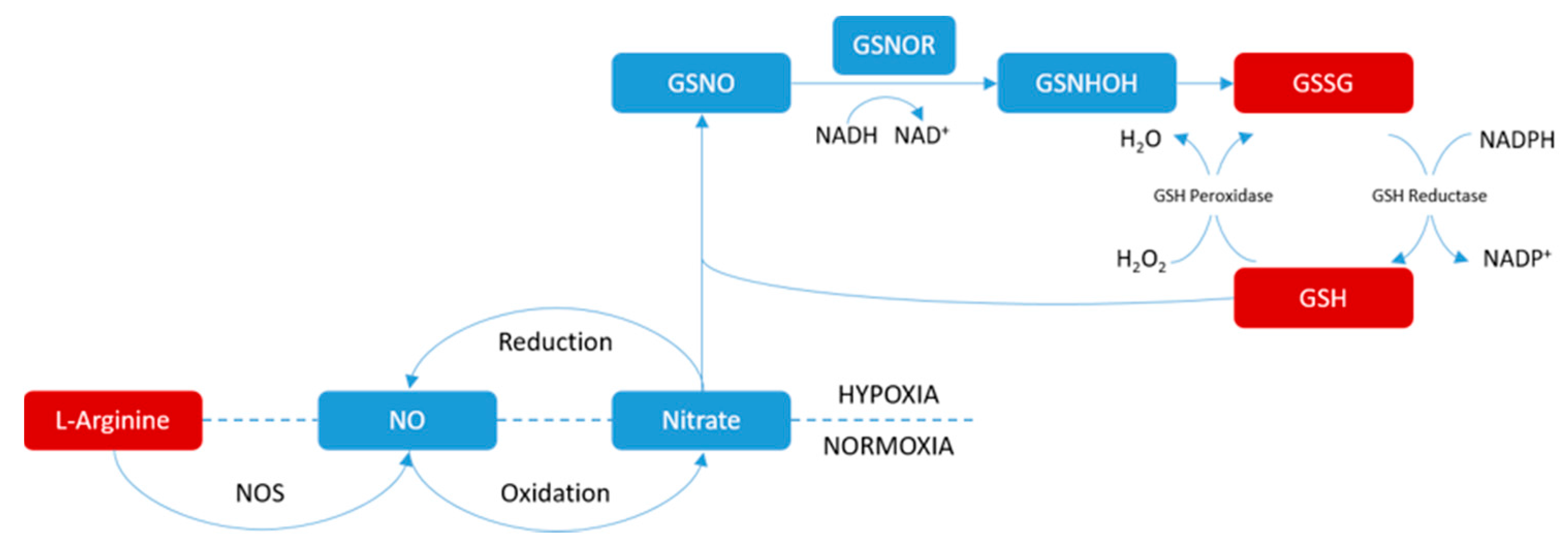
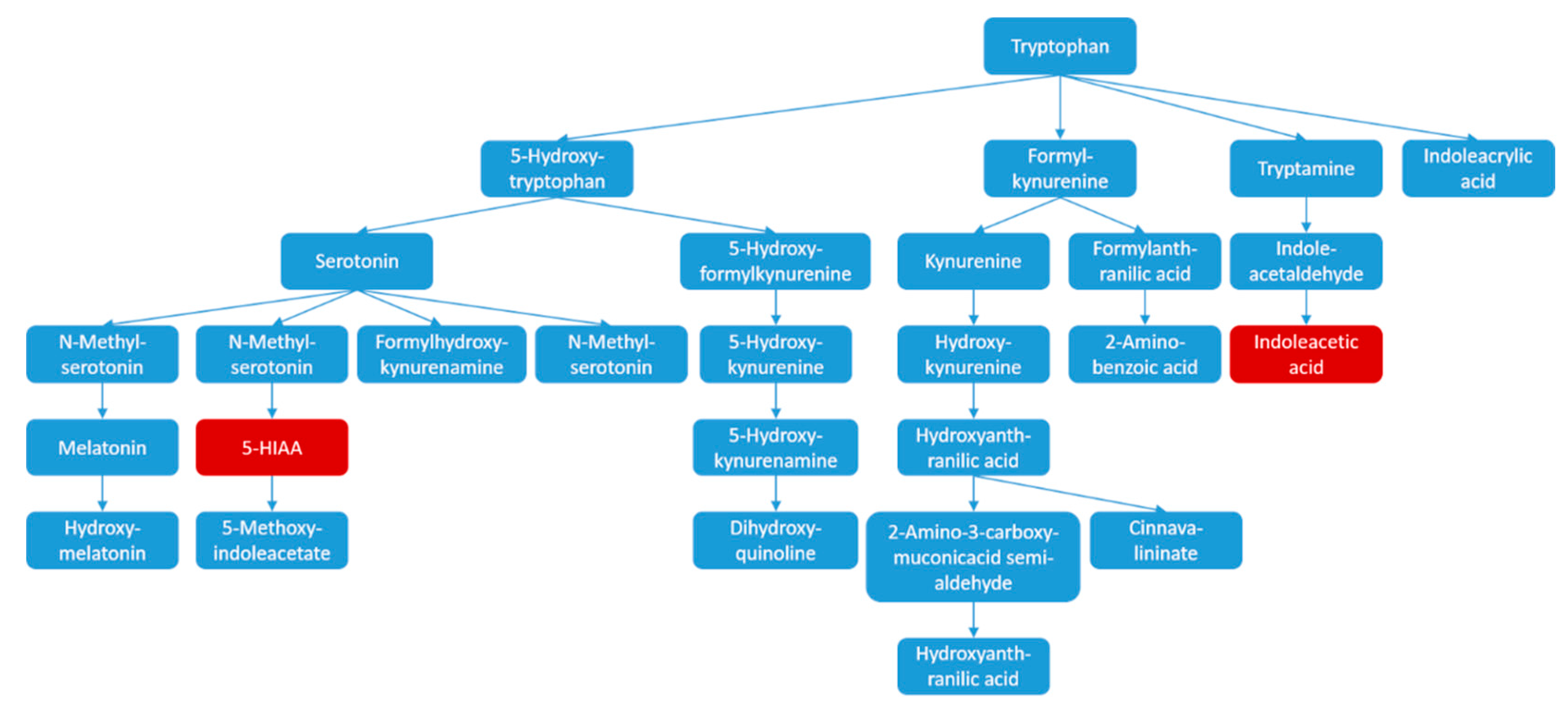
| RT | m/z | Compound | Peak Intensity (Mean ± SD) | |||
|---|---|---|---|---|---|---|
| Control | Model | WZYD | Flunarizine | |||
| 0.81 | 258.1108 | GPC | 6.61 × 104 ± 3.41 × 104 | 1.13 × 105 ± 5.40 × 104 ## | 2.17 × 104 ± 4.03 × 103 ** | 2.42 × 104 ± 7.77 × 103 ** |
| 0.82 | 175.1183 | Arginine | 1.68 × 105 ± 4.88 × 104 | 1.31 × 105 ± 2.10 × 104 ## | 2.77 × 105 ± 4.33 × 104 ** | 1.91 × 105 ± 2.29 × 104 ** |
| 1.11 | 348.0707 | AMP | 3.14 × 105 ± 2.05 × 105 | 5.60 × 105 ± 2.04 × 105 ## | 1.62 × 105 ± 3.37 × 104 ** | 1.82 × 105 ± 6.37 × 104 ** |
| 1.14 | 664.1144 | NAD | 5.89 × 104 ± 2.29 × 104 | 7.67 × 104 ± 2.99 × 104 # | 2.29 × 104 ± 4.99 × 103 ** | 3.70 × 104 ± 1.70 × 104 ** |
| 1.18 | 308.0895 | GSH | 2.33 × 106 ± 1.14 × 106 | 3.06 × 106 ± 1.09 × 106 # | 9.56 × 104 ± 4.70 × 104 ** | 6.50 × 104 ± 2.42 × 104 ** |
| 1.34 | 613.1597 | GSSG | 1.39 × 106 ± 7.23 × 105 | 1.72 × 106 ± 4.48 × 105 # | 3.19 × 105 ± 2.32 × 105 ** | 2.32 × 105 ± 1.81 × 105 ** |
| 2.20 | 136.0616 | Adenine | 4.61 × 105 ± 2.49 × 105 | 6.69 × 105 ± 1.75 × 105 ## | 2.74 × 105 ± 4.40× 104 ** | 2.08 × 105 ± 2.54 × 104 ** |
| 2.24 | 268.1025 | Adenosine | 2.70 × 106 ± 2.37 × 106 | 5.20 × 106 ± 2.05 × 106 ## | 1.97 × 105 ± 5.66 × 104 ** | 2.70 × 105 ± 1.41 × 105 ** |
| 3.51 | 100.0764 | Valerolactam | 9.97 × 103 ± 3.32 × 103 | 2.19 × 104 ± 2.18 × 104 ## | 1.02 × 104 ± 3.93 × 103 ** | 1.03 × 104 ± 5.05 × 103 ** |
| 6.56 | 232.1540 | Butyrylcarnitine | 5.87 × 104 ± 3.35 × 104 | 8.25 × 104 ± 4.18 × 104 ## | 1.58 × 104 ± 3.90 × 103 ** | 2.58 × 104 ± 8.02 × 103 ** |
| 6.74 | 114.0914 | Caprolactam | 8.00 × 105 ± 1.45 × 105 | 8.98 × 105 ± 1.31 × 105 ## | 2.17 × 105 ± 1.26 × 104 ** | 2.27 × 105 ± 2.44 × 104 ** |
| 20.38 | 385.3465 | Cholecalciferol | 1.82 × 105 ± 8.48 × 104 | 1.24 × 105 ± 7.58 × 104 # | 1.75 × 105 ± 5.27 × 104 * | 1.23 × 105 ± 3.25 × 104 |
| RT | m/z | Compound | Peak Intensity (Mean ± SD) | |||
|---|---|---|---|---|---|---|
| Control | Model | WZYD | Flunarizine | |||
| 0.93 | 175.1188 | Arginine | 2.91 × 105 ± 2.47 × 104 | 2.28 × 105 ± 4.79 × 104 ## | 2.60 × 105 ± 3.45 × 104 * | 2.05 × 105 ± 3.01 × 104 |
| 0.94 | 162.1122 | Carnitine | 7.47 × 105 ± 1.17 × 105 | 5.87 × 105 ± 8.45 × 104 ## | 6.87 × 105 ± 1.65 × 105 * | 5.70 × 105 ± 9.79 × 104 |
| 3.12 | 192.0660 | 5-HIAA | 4.63 × 104 ± 1.07 × 104 | 3.92 × 104 ± 8.96 × 103 # | 4.24 × 104 ± 9.62 × 103 | 4.01 × 104 ± 8.89 × 103 |
| 6.64 | 132.0808 | 3-Methyl-indole | 9.68 × 104 ± 1.69 × 104 | 8.37 × 104 ± 1.04 × 104 # | 1.06 × 105 ± 1.43 × 104 ** | 8.39 × 104 ± 1.22 × 104 |
| 7.17 | 134.0592 | Hydroxy-indole | 7.31 × 104 ± 3.61 × 104 | 4.49 × 104 ± 2.38 × 104 ## | 6.31 × 104 ± 3.39 × 104 | 3.65 × 104 ± 1.43 × 104 |
| 9.2 | 176.0700 | IAA | 2.03 × 104 ± 1.06 × 104 | 1.19 × 104 ± 4.27 × 103 ## | 1.37 × 104 ± 3.87 × 103 | 8.95 × 103 ± 2.36 × 103 |
| Pathway Name | Total | Expected | Hits | Raw p | Impact |
|---|---|---|---|---|---|
| Glutathione metabolism | 26 | 0.25963 | 2 | 0.026253 | 0.39790 |
| Purine metabolism | 68 | 0.67903 | 3 | 0.027048 | 0.06056 |
| Tryptophan metabolism | 41 | 0.40942 | 2 | 0.060817 | 0.01473 |
| Nicotinate and nicotinamide metabolism | 13 | 0.12981 | 1 | 0.122810 | 0.20833 |
| Lysine degradation | 20 | 0.19971 | 1 | 0.182990 | 0.00000 |
| Glycerophospholipid metabolism | 30 | 0.29957 | 1 | 0.262330 | 0.02315 |
| Arginine and proline metabolism | 44 | 0.43937 | 1 | 0.361430 | 0.08228 |
| Aminoacyl-tRNA biosynthesis | 67 | 0.66904 | 1 | 0.497840 | 0.00000 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, R.; Xu, H.; Zhang, X.; Wang, X.; Yuan, Z.; Sui, Z.; Wang, D.; Bi, K.; Li, Q. Metabolomics Strategy Using High Resolution Mass Spectrometry Reveals Novel Biomarkers and Pain-Relief Effect of Traditional Chinese Medicine Prescription Wu-Zhu-Yu Decoction Acting on Headache Modelling Rats. Molecules 2017, 22, 2110. https://doi.org/10.3390/molecules22122110
Liu R, Xu H, Zhang X, Wang X, Yuan Z, Sui Z, Wang D, Bi K, Li Q. Metabolomics Strategy Using High Resolution Mass Spectrometry Reveals Novel Biomarkers and Pain-Relief Effect of Traditional Chinese Medicine Prescription Wu-Zhu-Yu Decoction Acting on Headache Modelling Rats. Molecules. 2017; 22(12):2110. https://doi.org/10.3390/molecules22122110
Chicago/Turabian StyleLiu, Ran, Huarong Xu, Xiaowen Zhang, Xiaotong Wang, Ziyue Yuan, Zhenyu Sui, Dong Wang, Kaishun Bi, and Qing Li. 2017. "Metabolomics Strategy Using High Resolution Mass Spectrometry Reveals Novel Biomarkers and Pain-Relief Effect of Traditional Chinese Medicine Prescription Wu-Zhu-Yu Decoction Acting on Headache Modelling Rats" Molecules 22, no. 12: 2110. https://doi.org/10.3390/molecules22122110
APA StyleLiu, R., Xu, H., Zhang, X., Wang, X., Yuan, Z., Sui, Z., Wang, D., Bi, K., & Li, Q. (2017). Metabolomics Strategy Using High Resolution Mass Spectrometry Reveals Novel Biomarkers and Pain-Relief Effect of Traditional Chinese Medicine Prescription Wu-Zhu-Yu Decoction Acting on Headache Modelling Rats. Molecules, 22(12), 2110. https://doi.org/10.3390/molecules22122110