β-Amyloid and the Pathomechanisms of Alzheimer’s Disease: A Comprehensive View
Abstract
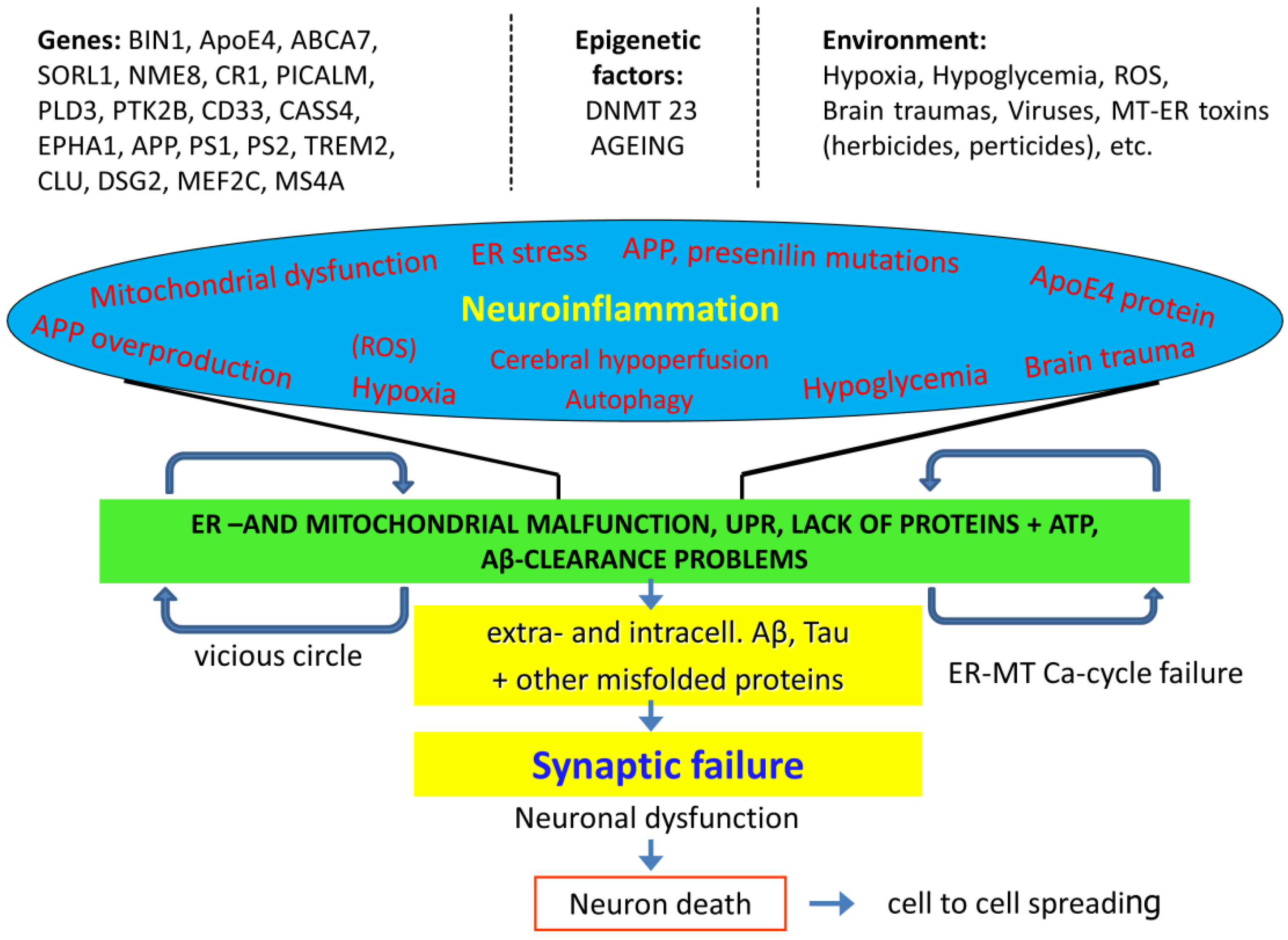
1. An Overview of the Possible Pathomechanisms of Alzheimer’s Disease (AD)
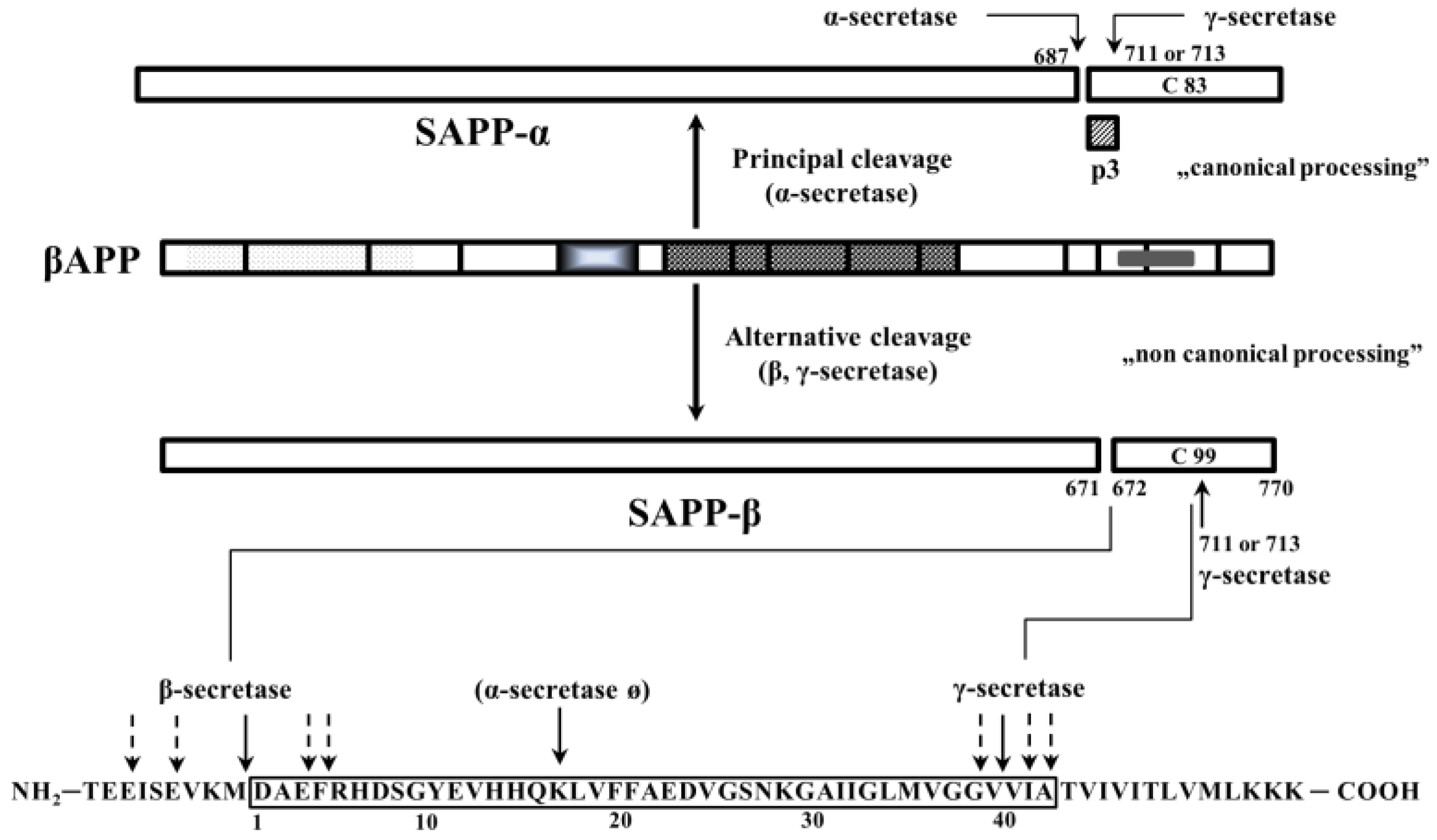
2. Physiological Functions of the Amyloid Precursor Protein (APP) and Its Metabolites (Amyloid Fragments)
- Late Golgi compartments could be mainly involved in the generation of intracellular Aβ (iAβ) [30].
- Recent studies have demonstrated that APP is rapidly transported from the Golgi apparatus to the lysosome, where it is processed into Aβ [31]. The adaptor protein 3 (AP-3) mediates rapid delivery of APP to lysosomes.
- Several studies demonstrated, however, that α- and β-secretases are present in ER allowing APP processing [32,33]. It was shown that presenilin 1 and 2 (PSN1, PSN2) and γ-secretase activity are located in the special subcompartment of the ER, called mitochondrial associated membrane (MAM). It was hypothesized that MAM is a subcellular site of the amyloidogenic processing of APP and Aβ generation [34,35]. Most recent results demonstrate that vascular dysfunctions, exposures to hypoxia/ischemia increase the amyloidogenic processing of APP and provoke AD pathology by activating β- and γ-secretases [19].
- APP plays a key role in neuronal homeostasis [38] including intracellular transport and signaling. APP acts as a signaling connection that transduces information about extracellular conditions (e.g., neuronal damage) and induces intracellular signaling events. Minute disruptions in APP signaling functions may be major contributors to neuronal dysfunction leading to AD.
- Most recent studies proved the results on the key role of APP as a neuroprotective factor [39]. Acute (stroke, cardiac arrest) or chronic (cerebrovascular disease) hypoxic-ischemic conditions cause up-regulation of APP. The protein itself and its soluble extracellular fragment SAPPα can promote neuronal survival. The underlying mechanism is very probably APP-mediated regulation of calcium homeostasis.
- Among the three major splice isoforms of APP (APP695, APP751, APP770), the predominant neuronal form is APP695. Beyond these, closely related APP-like proteins (APLP1, APLP2) exist as members of the APP family. All the APP family members are truly multifunctional proteins and can form large signaling complexes with various transmembrane proteins and intracellular binding partners [40].
- Canonical and non-canonical processing of APP results in a series of APP fragments, the most important fragments are SAPPα, SAPPβ, Aβ (40 and 42 AA length) and AICD. Their in vivo functions in the CNS are widely reviewed by Müller et al. [40], here we give a very short summary of their most important role:
- SAPPα:
- rescues memory and spine density in aged rats
- reduces plaque deposition and tau phosphorylation
- protects against traumatic brain injury (TBI), neuronal death in transient ischemia and hypoxia
- stimulates adult neurogenesis
- AICD:
- modulator of gene expression apoptosis and cytoskeletal dynamics [41]
- indirect regulation of DNA damage response
- Aβ 40/42:
3. Formation and Propagation of Hypertoxic Aβ Structures and the Prionoid Hypothesis
- Fibrillar Aβ
- Soluble, oligomeric Aβ: Aβ dimers; Aβ trimer, tetramer, hexamer; Aβ dodecamer (56kD, Aβ*56); large Aβ oligomers (“high-n” oligomers)
- Protofibrils
4. Short View of the Genetic Background of AD
- Lipid metabolism:
APOE SORL1 ABCA7 DSG2 CLU - Inflammatory response. Neuroinflammation is a pathological hallmark of AD and several genes were found in GWASs:
CD33 MEF2c HLA-DRB5/HLA-DRB1 CR1 MS4A ABCA7 TREM2 JNPP5D CLU EPHA1 - Endocytosis:
PICALM CDZAP BIN1 SORL1
5. Effect of Lipids in APP Processing and the Role of APP Metabolites in Lipid Homeostasis
- Cholesterol: A mutual regulatory feedback cycle exists, in which cholesterol influences APP processing to Aβ, while de novo, cholesterol synthesis is inhibited by high level of Aβ. Modification of cholesterol levels has effects on multiple proteins, not only APP and Aβ [115].
- Docosahexaenoic acid (DHA) is a polyunsaturated fatty acid. There are several epidemiological and experimental indications for a beneficial effects of DHA in preventing AD, at least at very early disease stages.
- Plasmalogens (PLs) represent 22% of the total phospholipid mass in human brain tissue. In AD pathogenesis a vicious circle between PLs and Aβ-generation might be postulated. Accumulated Aβ reduces cerebral PL contact and that stimulates γ-secretase activity, which leads to further Aβ-production [114].
- Sphingolipid (SL) impact on AD. Sphingolipids modulate APP-processing and Aβ-aggregation and, inversely, AICD downregulates total sphingolipid biosynthesis [116]. Sphingolipids are major components of lipid raft and the different SL classes have impact in AD-pathogenesis:
- Ceramide is accumulated in AD-patients. A feed-forward cycle between ceramide and Aβ exists in AD brain: increased ceramide level leads to enhanced Aβ production and that elevates ceramide synthesis, which stimulates Aβ-production. Sphingosine content elevates during the progress of AD [117]. In contrast, cerebral sphingosine-1-phosphate (S1P) content declines in AD patients, and negatively correlates with the level of Aβ.
- Gangliosides, sialic acid containing glycosphingolipids represent a heterogeneous group of different subtypes (GM1, GM1a, GD1b, GT1b GM3, etc.). There is strong link between ganglioside homeostasis and AD, however, further studies are needed to identify the most promising molecular target for AD therapeutic approaches.
6. Dysregulation of Protein Homeostasis in AD
6.1. The Double Edged Sword: Neurotrophic APP as A Source of Toxic Aβ
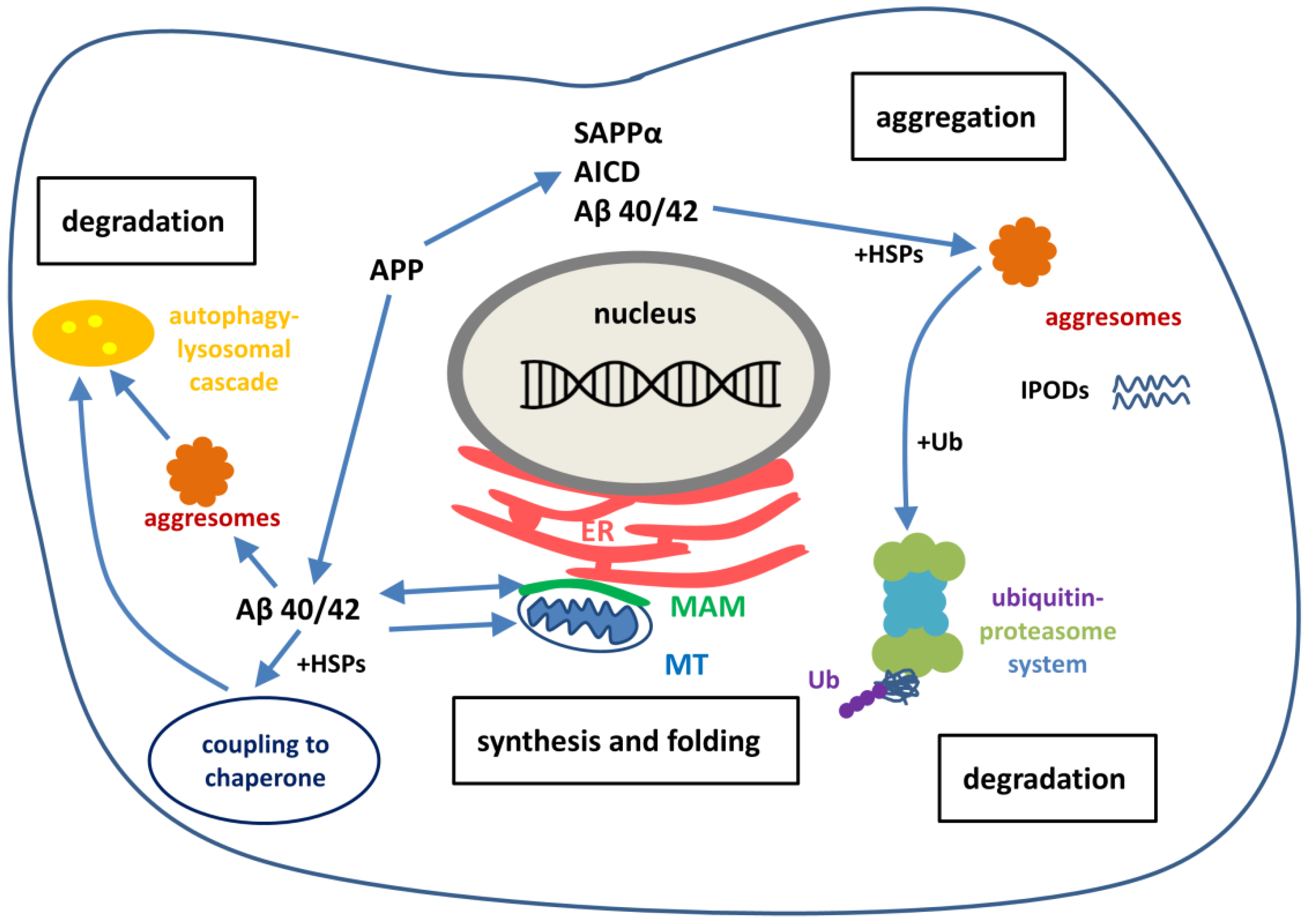
6.2. Formation of Intracellular Aβ in Neurons
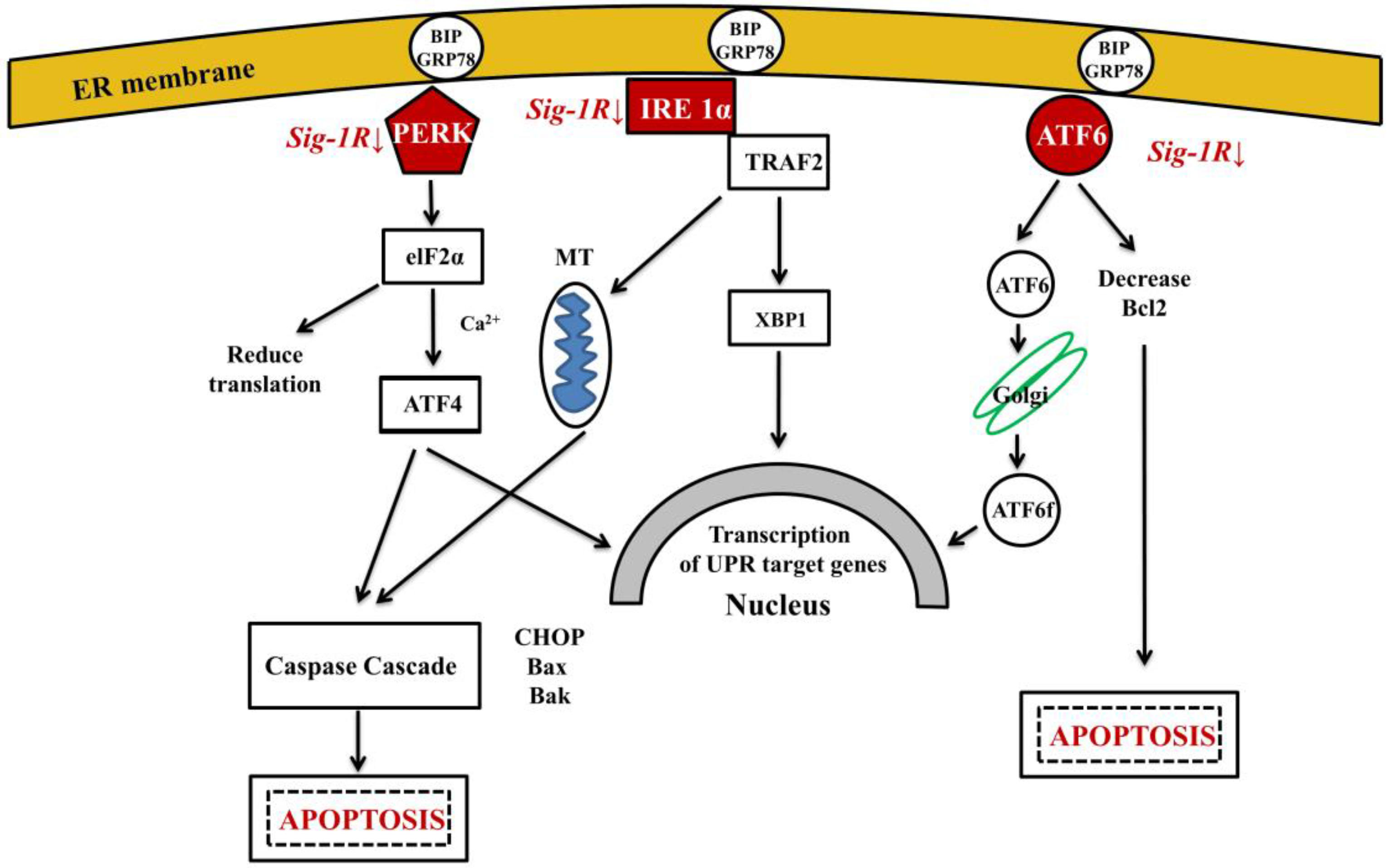
6.3. Intracellular Aβ Affects the Function of Subcellular Organelles: Ca2+ Dysregulation, the ER-Mithochondria Axis and Cross-Talk
- -
- phosphatidylserine synthase-1 (PSS1)
- -
- fatty acid EoA ligase 4 (FACL4)
- -
- inositol -1,4,5-triphosphate receptors (IP3Rs)
- -
- voltage-dependent anion channel (VDAC1)
- -
- sigma-1 receptor (Sig-1R)
6.4. Molecular Chaperones in AD
6.5. Aggresome Activity for Maintaining Proteostasis
- Insoluble protein deposits (IPODs): the proteins within are not ubiquitinated and mostly form amyloid fibrils [209].
- Juxtanuclear qualitity control (JUNQ) [209] inclusions contain ubiquitinated proteins, proteasome subunits and chaperones (e.g., HSP70). JUNQ was proposed to be a cellular quality control center in which soluble misfolded proteins or aggregates from the cytosol accumulate for proteosomal degradation and refolding [209].
6.6. Balancing Proteostasis by Protein Degradation
7. Vascular Risk Factors: The Role of the Neurovascular Unit and the BBB in AD
- Decreased bulk flow occurs because of reduced production of CSF. With aging (and even more so in AD) the reabsorption of CSF back into the circulation is reduced [235]. CSF levels of neurotoxic substances, such as Aβ increase, this process is further aggravated by a slowing of the glymphatic circulation [234,236] and BBB disruption. Neuroinflammation might be the background of these changes.
- In AD, the BBB undergoes functional and structural changes that disrupt its gate function, impair energy supply to the brain, reduce the clearance of Aβ and produce neurotoxic molecules. Decreased BBB clearance of Aβ leads to an increased amyloid burden in the brain and the initiation of AD [237]. The brain-to-blood efflux of Aβ is mediated by the lipoprotein receptor related protein (LRP-1), and the ABC subfamily B member 1, also termed P-glycoprotein (P-gp). Data from mouse models suggest that LRP-1 and P-gp expressions decline with age [238] suggesting a possible path for increased Aβ deposition and decreased clearance. Patients with AD show decreased levels of LRP-1 and P-gp [239] in BBB cells, leading to increased oxidation of LRP-1 and decreased level of P-gp activity. Knockdown of LRP-1 in mice recapitulates the key features of AD predicted by the neurovascular hypothesis (decreased Aβ-clearance, increased brain levels of Aβ and impaired cognition). Once initiated, decreased clearance of Aβ can further impair LRP-1 function by oxidation. Decreasing Aβ levels in animal models of AD restores Aβ clearance and cognition [240]. Inflammation also impairs Aβ-clearance from the brain and increases pericyte uptake of Aβ. This mechanism may account for the loss of pericytes seen in AD [241]. Taken together, BBB impairment can promote or even initiate AD: BBB dysfunction causes oxidative stress. Inflammation and mitochondrial dysfunction can mediate a vicious circle causing neuronal damage and increase of Aβ level in the brain [233]. BBB-mediated Aβ accumulation is predominantly based on reduced efflux across the barrier.
- The brain is dependent on BBB transport of insulin; this hormone has neurotrophic and neuroprotective functions in the CNS. In AD, the transport of insulin across the BBB is decreased. Such a transport defect reinforces that insulin resistance occurs in the brain of AD patients [242]. In clinical studies, delivery of insulin to CNS could improve some aspects of cognition of AD patients within 15 min [243].
8. AD as An Inflammatory Disease: The Role of Microglia
9. Conclusions: Major Trends in AD Prevention and Drug Discovery
- Management of modifiable risk factors for dementia [255]. Beyond regular physical activity and decreasing cardiovascular risk, the nutritional factors are in the center of the research. Nutritional approaches have shown limited beneficial effects in clinical studies. The identification of apoE4 as the most prevalent genetic risk factor for AD emphasized the crucial role of lipids in the pathomechanism of AD (see Section 5). Based on the results of apoE and lipid experiments, novel methods are offered: different supplements can be combined in multinutrional approaches [113].
- Development of compounds acting on the main stages of the pathogenesis of disease (the so called “disease modifying agents”). These drugs could potentially slow down the development of functional (and structural) abnormalities in the CNS, providing improvements of cognitive functions. Potential proteostatis-based therapeutics improving the imbalance between protein production and degradation belong to this group.
- Focused design of multitargeted drugs acting on multiple molecular targets involved in the pathogenesis of Aβ. Some enzyme inhibitors, (e.g., Sig-1R agonists), anti-inflammatory agents and antioxidants are in these groups.
- Repositioning of old drugs for new, anti-AD application offers a very attractive approach facilitating clinical trials. The oncolytic drugs bexarotene, tamibarotene, isotretinoin and long-acting insulin analogs belong to this group.
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| Aβ | β-amyloid peptide |
| AD | Alzheimer’s disease |
| AICD | APP intracellular domain |
| ALS | Amyotrophic lateral sclerosis |
| APP | Amyloid precursor protein |
| ATF 6 | activating transcription factor 6 |
| ATP | Adenosine triphosphate |
| BACE | Beta-site APP cleaving enzyme |
| Bak | Bcl-2 homologous antagonist killer |
| Bax | Bcl-2 like protein 4 |
| BBB | Blood brain barrier |
| Bcl-2 | B-cell lymphoma 2 |
| BiP (GRP78) | Binding immunoglobulin protein |
| CCH | Chronic cerebral hypoperfusion |
| CNS | Central nervous system |
| CSF | Cerebrospinal fluid |
| DNMT | DNA methyl transferum |
| EOAD | Early onset AD |
| ER | Endoplasmic reticulum |
| ERAD | ER-associated degradation |
| GWAS | Genome wide association studies |
| HD | Huntington disease |
| HSP | heat shock protein |
| IRE1α | inositol requiring enzyme 1α |
| iAβ | Intracellular Aβ |
| IP3R | Inositol-triphosphate receptor |
| IL1β | Interleukin 1β |
| ID | Intrinsically disordered |
| IPOD | Insoluble protein deposits |
| JNK | c-Jun amino terminal kinase |
| LBD | Lewy body disease |
| LOAD | Late onset AD |
| LRP1 | Lipoprotein receptor related protein-1 |
| LTP | Long term potentiation |
| MAM | Mitochondrion associated membrane |
| MVB | Multivesicular body |
| NDD | Neurodegenerative disease |
| NVU | Neurovascular unit |
| PD | Parkinson disease |
| P-gp | P-glycoprotein |
| PERK | Protein kinase RNA like ER-kinase |
| PM | Plasma membrane |
| PSN1, PSN2 | Presenilin 1 and 2 |
| PrP | prion protein |
| ROS | Reactive oxygene species |
| SAPPα | Soluble APP-α |
| Sig-1R | Sigma1 receptor |
| SERCA | Sarcoendoplasmic reticulum ATP-ase |
| SOS | Superoxide dismutase |
| TLR4 | Toll-like receptor 4 |
| TBI | Traumatic brain injury |
| TGN | Trans Golgi network |
| TNFa | Tumor necrosis factor a |
| Ub | Ubiquitin |
| UPR | Unfolded protein response |
| UPS | Ubiqitin-proteasome system |
References
- Goloubinoff, P. Mechanisms of protein homeostatis in health, aging and disease. Swiss Med. Wkly. 2016, 146, w14306. [Google Scholar] [PubMed]
- Khanam, H.; Ali, A.; Asif, M.; Shamsuzzaman. Neurodegenerative diseases linked to misfolded proteins and their therapeutic approaches: A review. Eur. J. Med. Chem. 2016, 124, 1121–1141. [Google Scholar] [CrossRef] [PubMed]
- Penke, B.; Bogar, F.; Fulop, L. Protein folding and misfolding, endoplasmic reticulum stress in neurodegenerative diseases: In trace of novel drug targets. Curr. Protein Pept. Sci. 2016, 17, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, G.G. Molecular pathological classification of neurodegenerative diseases: Turning towards precision medicine. Int. J. Mol. Sci. 2016, 17, 189. [Google Scholar] [CrossRef] [PubMed]
- Arneric, S.P.; Batrla-Utermann, R.; Beckett, L.; Bittner, T.; Blennow, K.; Carter, L.; Dean, R.; Engelborghs, S.; Genius, J.; Gordon, M.F.; et al. Cerebrospinal fluid biomarkers for Alzheimer’s disease: A view of the regulatory science qualification landscape from the coalition against major diseases csf biomarker team. J. Alzheimers Dis. 2017, 55, 19–35. [Google Scholar] [CrossRef] [PubMed]
- Rathore, S.; Habes, M.; Iftikhar, M.A.; Shacklett, A.; Davatzikos, C. A review on neuroimaging-based classification studies and associated feature extraction methods for Alzheimer’s disease and its prodromal stages. NeuroImage 2017, 155, 530–548. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Shu, H.; Ward, B.D.; Antuono, P.G.; Zhang, Z.; Li, S.J. Staging Alzheimer’s disease risk by sequencing brain function and structure, cerebrospinal fluid, and cognition biomarkers. J. Alzheimers Dis. 2016, 54, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Lauwers, E.; Verstreken, P. Presynaptic protein homeostasis and neuronal function. Curr. Opin. Genet. Dev. 2017, 44, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Ciryam, P.; Kundra, R.; Freer, R.; Morimoto, R.I.; Dobson, C.M.; Vendruscolo, M. A transcriptional signature of Alzheimer’s disease is associated with a metastable subproteome at risk for aggregation. Proc. Natl. Acad. Sci. USA 2016, 113, 4753–4758. [Google Scholar] [CrossRef] [PubMed]
- Freer, R.; Sormanni, P.; Vecchi, G.; Ciryam, P.; Dobson, C.M.; Vendruscolo, M. A protein homeostasis signature in healthy brains recapitulates tissue vulnerability to Alzheimer’s disease. Sci. Adv. 2016, 2, e1600947. [Google Scholar] [CrossRef] [PubMed]
- Boehm, J.; Fernandes, K.; Leclerc, N.; Robitaille, R. The neurobiology of aging and Alzheimer’s disease: Walking down the same road? Eur. J. Neurosci. 2013, 37, 1885–1886. [Google Scholar] [CrossRef] [PubMed]
- Neill, D. Should Alzheimer’s disease be equated with human brain ageing?: A maladaptive interaction between brain evolution and senescence. Ageing Res. Rev. 2012, 11, 104–122. [Google Scholar] [CrossRef] [PubMed]
- Doehner, J.; Genoud, C.; Imhof, C.; Krstic, D.; Knuesel, I. Extrusion of misfolded and aggregated proteins—A protective strategy of aging neurons? Eur. J. Neurosci. 2012, 35, 1938–1950. [Google Scholar] [CrossRef] [PubMed]
- Sturm, A.; Perczel, A.; Ivics, Z.; Vellai, T. The piwi-pirna pathway: Road to immortality. Aging Cell 2017, 16, 906–911. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.; Allsop, D. Amyloid deposition as the central event in the etiology of alzheimers-disease. Trends Pharmacol. Sci. 1991, 12, 383–388. [Google Scholar] [CrossRef]
- Karran, E.; Mercken, M.; De Strooper, B. The amyloid cascade hypothesis for Alzheimer’s disease: An appraisal for the development of therapeutics. Nat. Rev. Drug Discov. 2011, 10, 698–712. [Google Scholar] [CrossRef] [PubMed]
- Aisen, P.S.; Cummings, J.; Jack, C.R.; Morris, J.C.; Sperling, R.; Frolich, L.; Jones, R.W.; Dowsett, S.A.; Matthews, B.R.; Raskin, J.; et al. On the path to 2025: Understanding the Alzheimer’s disease continuum. Alzheimers Res. Ther. 2017, 9, 60. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, T.; Atwal, J.K.; Steinberg, S.; Snaedal, J.; Jonsson, P.V.; Bjornsson, S.; Stefansson, H.; Sulem, P.; Gudbjartsson, D.; Maloney, J.; et al. A mutation in app protects against Alzheimer’s disease and age-related cognitive decline. Nature 2012, 488, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Salminen, A.; Kauppinen, M.; Kaamiranta, K. Hypoxia/ischemia activate processing of amyloid precursor protein: Impact of vascular dysfunction in the pathogenesis of Alzheimer’s disease. J. Neurochem. 2017, 140, 536–549. [Google Scholar] [CrossRef] [PubMed]
- Reinhard, C.; Hebert, S.S.; De Strooper, B. The amyloid-beta precursor protein: Integrating structure with biological function. EMBO J. 2005, 24, 3996–4006. [Google Scholar] [CrossRef] [PubMed]
- Thinakaran, G.; Koo, E.H. Amyloid precursor protein trafficking, processing, and function. J. Biol. Chem. 2008, 283, 29615–29619. [Google Scholar] [CrossRef] [PubMed]
- Leyssen, M.; Ayaz, D.; Hebert, S.S.; Reeve, S.; De Strooper, B.; Hassan, B.A. Amyloid precursor protein promotes post-developmental neurite arborization in the drosophila brain. EMBO J. 2005, 24, 2944–2955. [Google Scholar] [CrossRef] [PubMed]
- Roch, J.M.; Masliah, E.; Rochlevecq, A.C.; Sundsmo, M.P.; Otero, D.A.C.; Veinbergs, I.; Saitoh, T. Increase of synaptic density and memory retention by a peptide representing the trophic domain of the amyloid beta/a4 protein-precursor. Proc. Natl. Acad. Sci. USA 1994, 91, 7450–7454. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.R.; O’Connor, K.; Tate, W.P.; Abraham, W.C. Roles of amyloid precursor protein and its fragments in regulating neural activity, plasticity and memory. Prog. Neurobiol. 2003, 70, 1–32. [Google Scholar] [CrossRef]
- Lee, K.J.; Moussa, C.E.H.; Lee, Y.; Sung, Y.; Howell, B.W.; Turner, R.S.; Pak, D.T.S.; Hoe, H.S. Beta amyloid-independent role of amyloid precursor protein in generation and maintenance of dendritic spines. Neuroscience 2010, 169, 344–356. [Google Scholar] [CrossRef] [PubMed]
- Randall, A.D.; Witton, J.; Booth, C.; Hynes-Allen, A.; Brown, J.T. The functional neurophysiology of the amyloid precursor protein (app) processing pathway. Neuropharmacology 2010, 59, 243–267. [Google Scholar] [CrossRef] [PubMed]
- Lammich, S.; Kojro, E.; Postina, R.; Gilbert, S.; Pfeiffer, R.; Jasionowski, M.; Haass, C.; Fahrenholz, F. Constitutive and regulated alpha-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc. Natl. Acad. Sci. USA 1999, 96, 3922–3927. [Google Scholar] [CrossRef] [PubMed]
- Lai, A.; Sisodia, S.S.; Trowbridge, I.S. Characterization of sorting signals in the beta-amyloid precursor protein cytoplasmic domain. J. Biol. Chem. 1995, 270, 3565–3573. [Google Scholar] [CrossRef] [PubMed]
- Perez, R.G.; Soriano, S.; Hayes, J.D.; Ostaszewski, B.; Xia, W.M.; Selkoe, D.J.; Chen, X.H.; Stokin, G.B.; Koo, E.H. Mutagenesis identifies new signals for beta-amyloid precursor protein endocytosis, turnover, and the generation of secreted fragments, including a beta 42. J. Biol. Chem. 1999, 274, 18851–18856. [Google Scholar] [CrossRef] [PubMed]
- Grimm, H.S.; Beher, D.; Lichtenthaler, S.F.; Shearman, M.S.; Beyreuther, K.; Hartmann, T. Gamma-secretase cleavage site specificity differs for intracellular and secretory amyloid beta. J. Biol. Chem. 2003, 278, 13077–13085. [Google Scholar] [CrossRef] [PubMed]
- Tam, J.H.K.; Seah, C.; Pasternak, S.H. The amyloid precursor protein is rapidly transported from the golgi apparatus to the lysosome and where it is processed into beta-amyloid. Mol. Brain 2014, 7, 54. [Google Scholar] [CrossRef] [PubMed]
- Chyung, A.S.C.; Greenberg, B.D.; Cook, D.G.; Doms, R.W.; Lee, V.M.Y. Novel beta-secretase cleavage of beta-amyloid precursor protein in the endoplasmic reticulum intermediate compartment of nt2n cells. J. Cell. Biol. 1997, 138, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Shin, R.W.; Saido, T.C.; Maeda, M.; Kitamoto, T. Novel alpha-secretase cleavage of Alzheimer’s amyloid beta precursor protein in the endoplasmic reticulum of cos7 cells. Neurosci. Lett. 2005, 376, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Placido, A.I.; Pereira, C.M.F.; Duarte, A.I.; Candeias, E.; Correia, S.C.; Santos, R.X.; Carvalho, C.; Cardoso, S.; Oliveira, C.R.; Moreira, P.I. The role of endoplasmic reticulum in amyloid precursor protein processing and trafficking: Implications for Alzheimer’s disease. BBA-Mol. Basis Dis. 2014, 1842, 1444–1453. [Google Scholar] [CrossRef] [PubMed]
- Vetrivel, K.S.; Thinakaran, G. Membrane rafts in Alzheimer’s disease beta-amyloid production. BBA-Mol. Cell. Biol. Lipid 2010, 1801, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Nalivaeva, N.N.; Turner, A.J. The amyloid precursor protein: A biochemical enigma in brain development, function and disease. FEBS Lett. 2013, 587, 2046–2054. [Google Scholar] [CrossRef] [PubMed]
- Dawkins, E.; Small, D.H. Insights into the physiological function of the beta-amyloid precursor protein: Beyond Alzheimer’s disease. J. Neurochem. 2014, 129, 756–769. [Google Scholar] [CrossRef] [PubMed]
- van der Kant, R.; Goldstein, L.S. Cellular functions of the amyloid precursor protein from development to dementia. Dev. Cell 2015, 32, 502–515. [Google Scholar] [CrossRef] [PubMed]
- Hefter, D.; Draguhn, A. App as a protective factor in acute neuronal insults. Front. Mol. Neurosci. 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Müller, U.C.; Deller, T.; Korte, M. Not just amyloid: Physiological functions of the amyloid precursor protein family. Nat. Rev. Neurosci. 2017, 18, 281–298. [Google Scholar] [CrossRef] [PubMed]
- Bukhari, H.; Glotzbach, A.; Kolbe, K.; Leonhardt, G.; Loosse, C.; Muller, T. Small things matter: Implications of app intracellular domain aicd nuclear signaling in the progression and pathogenesis of Alzheimer’s disease. Prog. Neurobiol. 2017, 156, 189–213. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.; Grimm, H.S.; Hartmann, T. Amyloid beta as a regulator of lipid homeostasis. Trends Mol. Med. 2007, 13, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Pearson, H.A.; Peers, C. Physiological roles for amyloid beta peptides. J. Physiol.-Lond. 2006, 575, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Hiltunen, M.; van Groen, T.; Jolkkonen, J. Functional roles of amyloid-beta protein precursor and amyloid-beta peptides: Evidence from experimental studies. J. Alzheimers Dis. 2009, 18, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, M.L.; Caraci, F.; De Bona, P.; Pappalardo, G.; Nicoletti, F.; Rizzarelli, E.; Copani, A. The monomer state of beta-amyloid: Where the Alzheimer’s disease protein meets physiology. Rev. Neurosci. 2010, 21, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Wogulis, M.; Wright, S.; Cunningham, D.; Chilcote, T.; Powell, K.; Rydel, R.E. Nucleation-dependent polymerization is an essential component of amyloid-mediated neuronal cell death. J. Neurosci. 2005, 25, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Sakono, M.; Zako, T. Amyloid oligomers: Formation and toxicity of a beta oligomers. FEBS J. 2010, 277, 1348–1358. [Google Scholar] [CrossRef] [PubMed]
- Upadhaya, A.R.; Capetillo-Zarate, E.; Kosterin, I.; Abramowski, D.; Kumar, S.; Yamaguchi, H.; Walter, J.; Fandrich, M.; Staufenbiel, M.; Thal, D.R. Dispersible amyloid beta-protein oligomers, protofibrils, and fibrils represent diffusible but not soluble aggregates: Their role in neurodegeneration in amyloid precursor protein (app) transgenic mice. Neurobiol. Aging 2012, 33, 2641–2660. [Google Scholar] [CrossRef] [PubMed]
- Jan, A.; Hartley, D.M.; Lashuel, H.A. Preparation and characterization of toxic a beta aggregates for structural and functional studies in Alzheimer’s disease research. Nat. Protoc. 2010, 5, 1186–1209. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J. The amyloid hypothesis for Alzheimer’s disease: A critical reappraisal. J. Neurochem. 2009, 110, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Benilova, I.; Karran, E.; De Strooper, B. The toxic a beta oligomer and Alzheimer’s disease: An emperor in need of clothes. Nat. Neurosci. 2012, 15, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Brouillette, J.; Caillierez, R.; Zommer, N.; Alves-Pires, C.; Benilova, I.; Blum, D.; De Strooper, B.; Buee, L. Neurotoxicity and memory deficits induced by soluble low-molecular-weight amyloid-beta(1-42) oligomers are revealed in vivo by using a novel animal model. J. Neurosci. 2012, 32, 7852–7861. [Google Scholar] [CrossRef] [PubMed]
- Wildburger, N.C.; Esparza, T.J.; LeDuc, R.D.; Fellers, R.T.; Thomas, P.M.; Cairns, N.J.; Kelleher, N.L.; Bateman, R.J.; Brody, D.L. Diversity of amyloid-beta proteoforms in the Alzheimer’s disease brain. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, F.S.; Habchi, J.; Cerreta, A.; Dietler, G. Afm-based single molecule techniques: Unraveling the amyloid pathogenic species. Curr. Pharm. Des. 2016, 22, 3950–3970. [Google Scholar] [CrossRef] [PubMed]
- Dobson, C.M. The structural basis of protein folding and its links with human disease. Philos. T. Roy. Soc. B 2001, 356, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Dobson, C.M. Protein folding and misfolding. Nature 2003, 426, 884–890. [Google Scholar] [CrossRef] [PubMed]
- Goloubinoff, P. Recent and future grand challenges in protein folding, misfolding and degradation. Front. Mol. Biosci. 2014, 1. [Google Scholar] [CrossRef] [PubMed]
- Stroo, E.; Koopman, M.; Nollen, E.A.A.; Mata-Cabana, A. Cellular regulation of amyloid formation in aging and disease. Front. Neurosci. 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Nagai, Y.; Inui, T.; Popiel, H.A.; Fujikake, N.; Hasegawa, K.; Urade, Y.; Goto, Y.; Naiki, H.; Toda, T. A toxic monomeric conformer of the polyglutamine protein. Nat. Struct. Mol. Biol. 2007, 14, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Kovari, E.; Horvath, J.; Bouras, C. Neuropathology of lewy body disorders. Brain Res. Bull. 2009, 80, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Breydo, L.; Wu, J.W.; Uversky, V.N. Alpha-synuclein misfolding and parkinson’s disease. BBA-Mol. Basis Dis. 2012, 1822, 261–285. [Google Scholar] [CrossRef] [PubMed]
- Lundblad, M.; Decressac, M.; Mattsson, B.; Bjorklund, A. Impaired neurotransmission caused by overexpression of alpha-synuclein in nigral dopamine neurons. Proc. Natl. Acad. Sci. USA 2012, 109, 3213–3219. [Google Scholar] [CrossRef] [PubMed]
- Sherzai, A.; Edland, S.D.; Masliah, E.; Hansen, L.; Pizzo, D.P.; Corey-Bloom, J. Spongiform change in dementia with lewy bodies and Alzheimer disease. Alzheimer Dis. Assoc. Disord. 2013, 27, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Uversky, V.N. Mysterious oligomerization of the amyloidogenic proteins. FEBS J. 2010, 277, 2940–2953. [Google Scholar] [CrossRef] [PubMed]
- Uversky, V.N. Targeting intrinsically disordered proteins in neurodegenerative and protein dysfunction diseases: Another illustration of the d-2 concept. Expert Rev. Proteom. 2010, 7, 543–564. [Google Scholar] [CrossRef] [PubMed]
- Turoverov, K.K.; Kuznetsova, I.M.; Uversky, V.N. The protein kingdom extended: Ordered and intrinsically disordered proteins, their folding, supramolecular complex formation, and aggregation. Prog. Biophys. Mol. Biol. 2010, 102, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Uversky, V.N. Intrinsically disordered proteins and novel strategies for drug discovery. Expert Opin. Drug Dis. 2012, 7, 475–488. [Google Scholar] [CrossRef] [PubMed]
- Griffith, J.S. Self-replication and scrapie. Nature 1967, 215, 1043–1044. [Google Scholar] [CrossRef] [PubMed]
- Prusiner, S.B. Some speculations about prions, amyloid, and Alzheimer’s-disease. N. Engl. J. Med. 1984, 310, 661–663. [Google Scholar] [CrossRef] [PubMed]
- Nussbaum, J.M.; Schilling, S.; Cynis, H.; Silva, A.; Swanson, E.; Wangsanut, T.; Tayler, K.; Wiltgen, B.; Hatami, A.; Ronicke, R.; et al. Prion-like behaviour and tau-dependent cytotoxicity of pyroglutamylated amyloid-beta. Nature 2012, 485, 651–655. [Google Scholar] [CrossRef] [PubMed]
- Eisele, Y.S.; Bolmont, T.; Heikenwalder, M.; Langer, F.; Jacobson, L.H.; Yan, Z.X.; Roth, K.; Aguzzi, A.; Staufenbiel, M.; Walker, L.C.; et al. Induction of cerebral beta-amyloidosis: Intracerebral versus systemic a beta inoculation. Proc. Natl. Acad. Sci. USA 2009, 106, 12926–12931. [Google Scholar] [CrossRef] [PubMed]
- Eisele, Y.S.; Obermuller, U.; Heilbronner, G.; Baumann, F.; Kaeser, S.A.; Wolburg, H.; Walker, L.C.; Staufenbiel, M.; Heikenwalder, M.; Jucker, M. Peripherally applied a beta-containing inoculates induce cerebral beta-amyloidosis. Science 2010, 330, 980–982. [Google Scholar] [CrossRef] [PubMed]
- Jucker, M.; Walker, L.C. Pathogenic protein seeding in Alzheimer disease and other neurodegenerative disorders. Ann. Neurol. 2011, 70, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Stohr, J.; Watts, J.C.; Mensinger, Z.L.; Oehler, A.; Grillo, S.K.; DeArmond, S.J.; Prusiner, S.B.; Giles, K. Purified and synthetic Alzheimer’s amyloid beta (a beta) prions. Proc. Natl. Acad. Sci. USA 2012, 109, 11025–11030. [Google Scholar] [CrossRef] [PubMed]
- Auer, S.; Dobson, C.M.; Vendruscolo, M.; Maritan, A. Self-templated nucleation in peptide and protein aggregation. Phys. Rev. Lett. 2008, 101. [Google Scholar] [CrossRef] [PubMed]
- Polymenidou, M.; Cleveland, D.W. Prion-like spread of protein aggregates in neurodegeneration. J. Exp. Med. 2012, 209, 889–893. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.H.; Warmlander, S.K.T.S.; Graslund, A.; Abrahams, J.P. Cross-interactions between the Alzheimer disease amyloid-beta peptide and other amyloid proteins: A further aspect of the amyloid cascade hypothesis. J. Biol. Chem. 2016, 291, 16485–16493. [Google Scholar] [CrossRef] [PubMed]
- Ugalde, C.L.; Finkelstein, D.I.; Lawson, V.A.; Hill, A.F. Pathogenic mechanisms of prion protein, amyloid-beta and alpha-synuclein misfolding: The prion concept and neurotoxicity of protein oligomers. J. Neurochem. 2016, 139, 162–180. [Google Scholar] [CrossRef] [PubMed]
- Aguzzi, A.; Rajendran, L. The transcellular spread of cytosolic amyloids, prions, and prionoids. Neuron 2009, 64, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Aguzzi, A.; Falsig, J. Prion propagation, toxicity and degradation. Nat. Neurosci. 2012, 15, 936–939. [Google Scholar] [CrossRef] [PubMed]
- Soto, C.; Saborio, G.P.; Anderes, L. Cyclic amplification of protein misfolding: Application to prion-related disorders and beyond. Trends Neurosci. 2002, 25, 390–394. [Google Scholar] [CrossRef]
- Saa, P.; Castilla, J.; Soto, C. Ultra-efficient replication of infectious prions by automated protein misfolding cyclic amplification. J. Biol. Chem. 2006, 281, 35245–35252. [Google Scholar] [CrossRef] [PubMed]
- Murayama, Y.; Yoshioka, M.; Yokoyama, T.; Iwamaru, Y.; Imamura, M.; Masujin, K.; Yoshiba, S.; Mohri, S. Efficient in vitro amplification of a mouse-adapted scrapie prion protein. Neurosci. Lett. 2007, 413, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Montalban, N.; Makarava, N.; Ostapchenko, V.G.; Savtchenk, R.; Alexeeva, I.; Rohwer, R.G.; Baskakov, I.V. Highly efficient protein misfolding cyclic amplification. PLoS Pathog. 2011, 7, e1001277. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Gonzalez, I.; Soto, C. Misfolded protein aggregates: Mechanisms, structures and potential for disease transmission. Semin. Cell Dev. Biol. 2011, 22, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Soto, C.; Estrada, L.; Castilla, J. Amyloids, prions and the inherent infectious nature of misfolded protein aggregates. Trends Biochem. Sci. 2006, 31, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Nath, S.; Agholme, L.; Kurudenkandy, F.R.; Granseth, B.; Marcusson, J.; Hallbeck, M. Spreading of neurodegenerative pathology via neuron-to-neuron transmission of beta-amyloid. J. Neurosci. 2012, 32, 8767–8777. [Google Scholar] [CrossRef] [PubMed]
- Condello, C.; Stohr, J. Abeta propagation and strains: Implications for the phenotypic diversity in Alzheimer’s disease. Neurobiol. Dis. 2017. [Google Scholar] [CrossRef] [PubMed]
- Qiang, W.; Yau, W.M.; Lu, J.X.; Collinge, J.; Tycko, R. Structural variation in amyloid-beta fibrils from Alzheimer’s disease clinical subtypes. Nature 2017, 541, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Brundin, P.; Melki, R.; Kopito, R. Prion-like transmission of protein aggregates in neurodegenerative diseases. Nat. Rev. Mol. Cell. Biol. 2010, 11, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Clavaguera, F.; Bolmont, T.; Crowther, R.A.; Abramowski, D.; Frank, S.; Probst, A.; Fraser, G.; Stalder, A.K.; Beibel, M.; Staufenbiel, M.; et al. Transmission and spreading of tauopathy in transgenic mouse brain. Nat. Cell Biol. 2009, 11, 909–913. [Google Scholar] [CrossRef] [PubMed]
- Frost, B.; Jacks, R.L.; Diamond, M.I. Propagation of tau misfolding from the outside to the inside of a cell. J. Biol. Chem. 2009, 284, 12845–12852. [Google Scholar] [CrossRef] [PubMed]
- Novak, P.; Prcina, M.; Kontsekova, E. Tauons and prions: Infamous cousins? J. Alzheimers Dis. 2011, 26, 413–430. [Google Scholar] [PubMed]
- Liu, L.; Drouet, V.; Wu, J.W.; Witter, M.P.; Small, S.A.; Clelland, C.; Duff, K. Trans-synaptic spread of tau pathology in vivo. PLoS ONE 2012, 7, e31302. [Google Scholar] [CrossRef] [PubMed]
- De Calignon, A.; Polydoro, M.; Suarez-Calvet, M.; William, C.; Adamowicz, D.H.; Kopeikina, K.J.; Pitstick, R.; Sahara, N.; Ashe, K.H.; Carlson, G.A.; et al. Propagation of tau pathology in a model of early Alzheimer’s disease. Neuron 2012, 73, 685–697. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, T.; Kügler, S.; Van Leuven, F. Modeling of tau-mediated synaptic and neuronal degeneration in Alzheimer’s disease. Int. J. Alzheimers Dis. 2010, 2010, 573138. [Google Scholar] [CrossRef] [PubMed]
- Hall, G.F.; Patuto, B.A. Is tau ready for admission to the prion club? Prion 2012, 6, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Blennow, K.; de Leon, M.J.; Zetterberg, H. Alzheimer’s disease. Lancet 2006, 368, 387–403. [Google Scholar] [CrossRef]
- Tanzi, R.E. The genetics of Alzheimer disease. CSH Perspect. Med. 2012, 2, a006296. [Google Scholar] [CrossRef] [PubMed]
- Piaceri, I.; Nacmias, B.; Sorbi, S. Genetics of familial and sporadic Alzheimer’s disease. Front. Biosci. (Elite Ed.) 2013, 5, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Bettens, K.; Sleegers, K.; Van Broeckhoven, C. Genetic insights in Alzheimer’s disease. Lancet Neurol. 2013, 12, 92–104. [Google Scholar] [CrossRef]
- Giri, M.; Zhang, M.; Lu, Y. Genes associated with Alzheimer’s disease: An overview and current status. Clin. Interv. Aging 2016, 11, 665–681. [Google Scholar] [CrossRef] [PubMed]
- Karch, C.M.; Cruchaga, C.; Goate, A.M. Alzheimer’s disease genetics: From the bench to the clinic. Neuron 2014, 83, 11–26. [Google Scholar] [CrossRef] [PubMed]
- De Jonghe, C.; Esselens, C.; Kumar-Singh, S.; Craessaerts, K.; Serneels, S.; Checler, F.; Annaert, W.; Van Broeckhoven, C.; De Strooper, B. Pathogenic app mutations near the gamma-secretase cleavage site differentially affect abeta secretion and app c-terminal fragment stability. Hum. Mol. Genet. 2001, 10, 1665–1671. [Google Scholar] [CrossRef] [PubMed]
- Esler, W.P.; Wolfe, M.S. Biomedicine—A portrait of Alzheimer secretases—New features and familiar faces. Science 2001, 293, 1449–1454. [Google Scholar] [CrossRef] [PubMed]
- Cacace, R.; Sleegers, K.; Van Broeckhoven, C. Molecular genetics of early-onset Alzheimer’s disease revisited. Alzheimers Dement. 2016, 12, 733–748. [Google Scholar] [CrossRef] [PubMed]
- Solomon, A.; Kivipelto, M.; Wolozin, B.; Zhou, J.F.; Whitmer, R.A. Midlife serum cholesterol and increased risk of Alzheimer’s and vascular dementia three decades later. Dement. Geriatr. Cogn. Disord. 2009, 28, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Tran, L.; Emilsson, V.; Zhu, J. Characterization of genetic networks associated with Alzheimer’s disease. Methods Mol. Biol. 2016, 1303, 459–477. [Google Scholar] [PubMed]
- Holtzman, D.M.; Herz, J.; Bu, G.J. Apolipoprotein e and apolipoprotein e receptors: Normal biology and roles in Alzheimer disease. CSH Perspect. Med. 2012, 2, a006312. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, N.; Hatsuta, H.; Murayama, S.; Suzuki, A.; Yanagisawa, K. Influence of apoe genotype and the presence of Alzheimer’s pathology on synaptic membrane lipids of human brains. J. Neurosci. Res. 2014, 92, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Kojro, E.; Gimpl, G.; Lammich, S.; Marz, W.; Fahrenholz, F. Low cholesterol stimulates the nonamyloidogenic pathway by its effect on the alpha-secretase adam 10. Proc. Natl. Acad. Sci. USA 2001, 98, 5815–5820. [Google Scholar] [CrossRef] [PubMed]
- Harris, B.; Pereira, I.; Parkin, E. Targeting adam10 to lipid rafts in neuroblastoma sh-sy5y cells impairs amyloidogenic processing of the amyloid precursor protein. Brain Res. 2009, 1296, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.; Michaelson, D.; Hartmann, T. Omega-3 fatty acids, lipids and apoe lipidation in Alzheimer’s disease: A rationale for multi-nutrient dementia prevention. J. Lipid Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.W.; Mett, J.; Grimm, H.S.; Hartmann, T. App function and lipids: A bidirectional link. Front. Mol. Neurosci. 2017, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- Wood, W.G.; Li, L.; Muller, W.E.; Eckert, G.P. Cholesterol as a causative factor in Alzheimer’s disease: A debatable hypothesis. J. Neurochem. 2014, 129, 559–572. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.; Grosgen, S.; Rothhaar, T.L.; Burg, V.K.; Hundsdorfer, B.; Haupenthal, V.J.; Friess, P.; Muller, U.; Fassbender, K.; Riemenschneider, M.; et al. Intracellular app domain regulates serine-palmitoyl-coa transferase expression and is affected in Alzheimer’s disease. Int. J. Alzheimers Dis. 2011, 2011, 695413. [Google Scholar] [CrossRef] [PubMed]
- He, X.X.; Huang, Y.; Li, B.; Gong, C.X.; Schuchman, E.H. Deregulation of sphingolipid metabolism in Alzheimer’s disease. Neurobiol. Aging 2010, 31, 398–408. [Google Scholar] [CrossRef] [PubMed]
- Balch, W.E.; Morimoto, R.I.; Dillin, A.; Kelly, J.W. Adapting proteostasis for disease intervention. Science 2008, 319, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Yerbury, J.J.; Ooi, L.; Dillin, A.; Saunders, D.N.; Hatters, D.M.; Beart, P.M.; Cashman, N.R.; Wilson, M.R.; Ecroyd, H. Walking the tightrope: Proteostasis and neurodegenerative disease. J. Neurochem. 2016, 137, 489–505. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.; Selkoe, D.J. The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 2002, 297, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Krafft, G.A.; Klein, W.L. Addls and the signaling web that leads to Alzheimer’s disease. Neuropharmacology 2010, 59, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Schliebs, R.; Arendt, T. The cholinergic system in aging and neuronal degeneration. Behav. Brain Res. 2011, 221, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.N.; Jhamandas, J.H. Neuronal receptors as targets for the action of amyloid-beta protein (a beta) in the brain. Expert Rev. Mol. Med. 2012, 14. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.Q.; Yan, J.Q.; Zhou, P.; Li, J.J.; Gao, H.M.; Xia, Y.; Wang, Q. Neurotransmitter receptors and cognitive dysfunction in Alzheimer’s disease and parkinson’s disease. Prog. Neurobiol. 2012, 97, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, T. Intracellular biology of Alzheimer’s disease amyloid beta peptide. Eur. Arch. Psychiatry Clin. Neurosci. 1999, 249, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, R.P.; Tepper, K.; Roznicke, R.; Soom, M.; Westermann, M.; Reymann, K.; Kaether, C.; Fandrich, M. Mechanism of amyloid plaque formation suggests an intracellular basis of a beta pathogenicity. Proc. Natl. Acad. Sci. USA 2010, 107, 1942–1947. [Google Scholar] [CrossRef] [PubMed]
- Gimenez-Llort, L.; Blazquez, G.; Canete, T.; Johansson, B.; Oddo, S.; Tobena, A.; LaFerla, F.M.; Fernandez-Teruel, A. Modeling behavioral and neuronal symptoms of Alzheimer’s disease in mice: A role for intraneuronal amyloid. Neurosci. Biobehav. Rev. 2007, 31, 125–147. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Chen, L.; Lee, D.H.S.; Yu, L.C.; Zhang, Y. The role of intracellular amyloid beta in Alzheimer’s disease. Prog. Neurobiol. 2007, 83, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Gouras, G.K.; Tampellini, D.; Takahashi, R.H.; Capetillo-Zarate, E. Intraneuronal beta-amyloid accumulation and synapse pathology in Alzheimer’s disease. Acta Neuropathol. 2010, 119, 523–541. [Google Scholar] [CrossRef] [PubMed]
- Bayer, T.A.; Wirths, O. Intracellular accumulation of amyloid-beta—A predictor for synaptic dysfunction and neuron loss in Alzheimer’s disease. Front. Aging Neurosci. 2010, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Intracellular amyloid ß: A modification to the amyloid hypothesis in Alzheimer’s disease. In Lipids and Cellular Membranes in Amyloid Diseases; Jelinek, R., Ed.; Wiley: New York, NY, USA, 2011; pp. 143–159. [Google Scholar]
- Zheng, L.; Cedazo-Minguez, A.; Hallbeck, M.; Jerhammar, F.; Marcusson, J.; Terman, A. Intracellular distribution of amyloid beta peptide and its relationship to the lysosomal system. Transl. Neurodegener. 2012, 1, 19. [Google Scholar] [CrossRef] [PubMed]
- Foldi, I.; Datki, Z.L.; Szabo, Z.; Bozso, Z.; Penke, B.; Janaky, T. Proteomic study of the toxic effect of oligomeric abeta1-42 in situ prepared from iso-abeta1-42. J. Neurochem. 2011, 117, 691–702. [Google Scholar] [PubMed]
- Juhasz, G.; Foldi, I.; Penke, B. Systems biology of Alzheimer’s disease: How diverse molecular changes result in memory impairment in ad. Neurochem. Int. 2011, 58, 739–750. [Google Scholar] [CrossRef] [PubMed]
- Verdier, Y.; Penke, B. Polypeptide and protein aggregates with neurotoxic properties in neurodegenerative disorders: Emphasis on beta-amyloid-induced dysfunction of receptor-mediated signal transduction pathways. Curr. Signal Transduct. Ther. 2006, 1, 97–112. [Google Scholar] [CrossRef]
- Verdier, Y.; Foldi, I.; Sergeant, N.; Fulop, L.; Penke, Z.; Janaky, T.; Szucs, M.; Penke, B. Characterization of the interaction between a beta 1-42 and glyceraldehyde phosphodehydrogenase. J. Pept. Sci. 2008, 14, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Penke, B.; Toth, A.M.; Foldi, I.; Szucs, M.; Janaky, T. Intraneuronal ss-amyloid and its interactions with proteins and subcellular organelles. Electrophoresis 2012, 33, 3608–3616. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, R.H.; Nagao, T.; Gouras, G.K. Plaque formation and the intraneuronal accumulation of beta-amyloid in Alzheimer’s disease. Pathol. Int. 2017, 67, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Casas, C.; Sergeant, N.; Itier, J.M.; Blanchard, V.; Wirths, O.; van der Kolk, N.; Vingtdeux, V.; van de Steeg, E.; Ret, G.; Canton, T.; et al. Massive ca1/2 neuronal loss with intraneuronal and n-interminal truncated a beta(42) accumulation in a novel Alzheimer transgenic model. Am. J. Pathol. 2004, 165, 1289–1300. [Google Scholar] [CrossRef]
- Billings, L.M.; Oddo, S.; Green, K.N.; McGaugh, J.L.; LaFerla, F.M. Intraneuronal abeta causes the onset of early Alzheimer’s disease-related cognitive deficits in transgenic mice. Neuron 2005, 45, 675–688. [Google Scholar] [CrossRef] [PubMed]
- Tomiyama, T.; Matsuyama, S.; Iso, H.; Umeda, T.; Takuma, H.; Ohnishi, K.; Ishibashi, K.; Teraoka, R.; Sakama, N.; Yamashita, T.; et al. A mouse model of amyloid beta oligomers: Their contribution to synaptic alteration, abnormal tau phosphorylation, glial activation, and neuronal loss in vivo. J. Neurosci. 2010, 30, 4845–4856. [Google Scholar] [CrossRef] [PubMed]
- Abramowski, D.; Rabe, S.; Upadhaya, A.R.; Reichwald, J.; Danner, S.; Staab, D.; Capetillo-Zarate, E.; Yamaguchi, H.; Saido, T.C.; Wiederhold, K.H.; et al. Transgenic expression of intraneuronal a beta(42) but not a beta(40) leads to cellular a beta lesions, degeneration, and functional impairment without typical Alzheimer’s disease pathology. J. Neurosci. 2012, 32, 1273–1283. [Google Scholar] [CrossRef] [PubMed]
- Claeysen, S.; Cochet, M.; Donneger, R.; Dumuis, A.; Bockaert, J.; Giannoni, P. Alzheimer culprits: Cellular crossroads and interplay. Cell Signal. 2012, 24, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Burgos, P.V.; Mardones, G.A.; Rojas, A.L.; daSilva, L.L.P.; Prabhu, Y.; Hurley, J.H.; Bonifacino, J.S. Sorting of the Alzheimer’s disease amyloid precursor protein mediated by the ap-4 complex. Dev. Cell 2010, 18, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A. Autophagy in neurodegenerative disease: Friend, foe or turncoat? Trends Neurosci. 2006, 29, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Lopez, E.M.; Bell, K.F.S.; Ribeiro-da-Silva, A.; Cuello, A.C. Early changes in neurons of the hippocampus and neocortex in transgenic rats expressing intracellular human a-beta. J. Alzheimers Dis. 2004, 6, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Gajdusek, D.C. Transmissible and non-transmissible amyloidoses: Autocatalytic post-translational conversion of host precursor proteins to beta-pleated sheet configurations. J. Neuroimmunol. 1988, 20, 95–110. [Google Scholar] [CrossRef]
- Prusiner, S.B.; Dearmond, S.J. Prions causing nervous-system degeneration. Lab. Investig. 1987, 56, 349–363. [Google Scholar] [PubMed]
- Glabe, C. Intracellular mechanisms of amyloid accumulation and pathogenesis in Alzheimer’s disease. J. Mol. Neurosci. 2001, 17, 137–145. [Google Scholar] [CrossRef]
- Nagele, R.G.; D’Andrea, M.R.; Anderson, W.J.; Wang, H.Y. Intracellular accumulation of beta-amyloid(1-42) in neurons is facilitated by the alpha 7 nicotinic acetylcholine receptor in Alzheimer’s disease. Neuroscience 2002, 110, 199–211. [Google Scholar] [CrossRef]
- Burdick, D.; Kosmoski, J.; Knauer, M.F.; Glabe, C.G. Preferential adsorption, internalization and resistance to degradation of the major isoform of the Alzheimer’s amyloid peptide, a beta 1–42, in differentiated pc12 cells. Brain Res. 1997, 746, 275–284. [Google Scholar] [CrossRef]
- Ditaranto, K.; Tekirian, T.L.; Yang, A.J. Lysosomal membrane damage in soluble a beta-mediated cell death in Alzheimer’s disease. Neurobiol. Dis. 2001, 8, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Lustbader, J.W.; Cirilli, M.; Lin, C.; Xu, H.W.; Takuma, K.; Wang, N.; Caspersen, C.; Chen, X.; Pollak, S.; Chaney, M.; et al. Abad directly links a beta to mitochondrial toxicity in Alzheimer’s disease. Science 2004, 304, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yan, S.D. Mitochondrial abeta: A potential cause of metabolic dysfunction in Alzheimer’s disease. IUBMB Life 2006, 58, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.T.; Beal, M.F. Alzheimer’s app mangles mitochondria. Nat. Med. 2006, 12, 1241–1243. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, R.J.; Wong, P.C. Amyloid precursor protein processing and Alzheimer’s disease. Annu. Rev. Neurosci. 2011, 34, 185–204. [Google Scholar] [CrossRef] [PubMed]
- Blair, J.A.; Siedlak, S.L.; Wolfram, J.A.; Nunomura, A.; Castellani, R.J.; Ferreira, S.T.; Klein, W.L.; Wang, Y.; Casadesus, G.; Smith, M.A.; et al. Accumulation of intraneuronal amyloid-beta is common in normal brain. Curr. Alzheimer Res. 2014, 11, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Alberdi, E.; Wyssenbach, A.; Alberdi, M.; Sanchez-Gomez, M.V.; Cavaliere, F.; Rodriguez, J.J.; Verkhratsky, A.; Matute, C. Ca2+-dependent endoplasmic reticulum stress correlates with astrogliosis in oligomeric amyloid-treated astrocytes and in a model of Alzheimer’s disease. Aging Cell 2013, 12, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Endres, K.; Reinhardt, S. Er-stress in Alzheimer’s disease: Turning the scale? Am. J. Neurodegener Dis. 2013, 2, 247–265. [Google Scholar] [PubMed]
- Oh, H.; Madison, C.; Baker, S.; Rabinovici, G.; Jagust, W. Dynamic relationships between age, amyloid-beta deposition, and glucose metabolism link to the regional vulnerability to Alzheimer’s disease. Brain 2016, 139, 2275–2289. [Google Scholar] [CrossRef] [PubMed]
- Roussel, B.D.; Kruppa, A.J.; Miranda, E.; Crowther, D.C.; Lomas, D.A.; Marciniak, S.J. Endoplasmic reticulum dysfunction in neurological disease. Lancet Neurol. 2013, 12, 105–118. [Google Scholar] [CrossRef]
- Volgyi, K.; Juhasz, G.; Kovacs, Z.; Penke, B. Dysfunction of endoplasmic reticulum (er) and mitochondria (mt) in Alzheimer’s disease: The role of the er-mt cross-talk. Curr. Alzheimer Res. 2015, 12, 655–672. [Google Scholar] [CrossRef] [PubMed]
- Mossmann, D.; Vogtle, F.N.; Taskin, A.A.; Teixeira, P.F.; Ring, J.; Burkhart, J.M.; Burger, N.; Pinho, C.M.; Tadic, J.; Loreth, D.; et al. Amyloid-beta peptide induces mitochondrial dysfunction by inhibition of preprotein maturation. Cell Metab. 2014, 20, 662–669. [Google Scholar] [CrossRef] [PubMed]
- Cenini, G.; Rub, C.; Bruderek, M.; Voos, W. Amyloid beta-peptides interfere with mitochondrial preprotein import competence by a coaggregation process. Mol. Biol. Cell. 2016, 27, 3257–3272. [Google Scholar] [CrossRef] [PubMed]
- Hosoi, T.; Matsuzaki, S.; Miyahara, T.; Shimizu, K.; Hasegawa, Y.; Ozawa, K. Possible involvement of 15-deoxy-delta(12,14)-prostaglandin j(2) in the development of leptin resistance. J. Neurochem. 2015, 133, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Ohta, K.; Mizuno, A.; Li, S.M.; Itoh, M.; Ueda, M.; Ohta, E.; Hida, Y.; Wang, M.X.; Furoi, M.; Tsuzuki, Y.; et al. Endoplasmic reticulum stress enhances gamma-secretase activity. Biochem. Biophys. Res. Commun. 2011, 416, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C. The unfolded protein response: Controlling cell fate decisions under er stress and beyond. Nat. Rev. Mol. Cell Biol. 2012, 13, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.M. Crosstalk between endoplasmic reticulum stress and protein misfolding in neurodegenerative diseases. ISRN Cell Biol. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Scheper, W.; Hoozemans, J.J.M. The unfolded protein response in neurodegenerative diseases: A neuropathological perspective. Acta Neuropathol. 2015, 130, 315–331. [Google Scholar] [CrossRef] [PubMed]
- Bahar, E.; Kim, H.; Yoon, H. Er stress-mediated signaling: Action potential and ca(2+) as key players. Int. J. Mol. Sci. 2016, 17, 1558. [Google Scholar] [CrossRef] [PubMed]
- Safra, M.; Ben-Hamo, S.; Kenyon, C.; Henis-Korenblit, S. The ire-1 er stress-response pathway is required for normal secretory-protein metabolism in c. Elegans. J. Cell Sci. 2013, 126, 4136–4146. [Google Scholar] [CrossRef] [PubMed]
- Magi, S.; Castaldo, P.; Macri, M.L.; Maiolino, M.; Matteucci, A.; Bastioli, G.; Gratteri, S.; Amoroso, S.; Lariccia, V. Intracellular calcium dysregulation: Implications for Alzheimer’s disease. Biomed. Res. Int. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Calcium regulation of neural rhythms, memory and Alzheimer’s disease. J. Physiol.-Lond. 2014, 592, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Popugaeva, E.; Pchitskaya, E.; Bezprozvanny, I. Dysregulation of neuronal calcium homeostasis in Alzheimer’s disease—A therapeutic opportunity? Biochem. Biophys. Res. Commun. 2017, 483, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Briggs, C.A.; Chakroborty, S.; Stutzmann, G.E. Emerging pathways driving early synaptic pathology in Alzheimer’s disease. Biochem. Biophys. Res. Commun. 2017, 483, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Dong, Y.; Cheng, J. The function of the mitochondrial calcium uniporter in neurodegenerative disorders. Int. J. Mol. Sci. 2017, 18, 248. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, D.; Rizzuto, R.; Pozzan, T. Enjoy the trip: Calcium in mitochondria back and forth. Annu. Rev. Biochem. 2016, 85, 161–192. [Google Scholar] [CrossRef] [PubMed]
- Filadi, R.; Theurey, P.; Pizzo, P. The endoplasmic reticulum-mitochondria coupling in health and disease: Molecules, functions and significance. Cell Calcium 2017, 62, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Paillusson, S.; Stoica, R.; Gomez-Suaga, P.; Lau, D.H.W.; Mueller, S.; Miller, T.; Miller, C.C.J. There’s something wrong with my mam; the er-mitochondria axis and neurodegenerative diseases. Trends Neurosci. 2016, 39, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.U.; Kornfeld, O.S.; Mochly-Rosen, D. The entangled er-mitochondrial axis as a potential therapeutic strategy in neurodegeneration: A tangled duo unchained. Cell Calcium 2016, 60, 218–234. [Google Scholar] [CrossRef] [PubMed]
- Schon, E.A.; Area-Gomez, E. Mitochondria-associated er membranes in Alzheimer disease. Mol. Cell. Neurosci. 2013, 55, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Schon, E.A.; Area-Gomez, E. Is Alzheimer’s disease a disorder of mitochondria-associated membranes? J. Alzheimers Dis. 2010, 20, S281–S292. [Google Scholar] [CrossRef] [PubMed]
- Csordas, G.; Renken, C.; Varnai, P.; Walter, L.; Weaver, D.; Buttle, K.F.; Balla, T.; Mannella, C.A.; Hajnoczky, G. Structural and functional features and significance of the physical linkage between er and mitochondria. J. Cell Biol. 2006, 174, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Rizzuto, R.; Hajnoczky, G.; Su, T.P. Mam: More than just a housekeeper. Trends Cell Biol. 2009, 19, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Cardenas, C.; Miller, R.A.; Smith, I.; Bui, T.; Molgo, J.; Muller, M.; Vais, H.; Cheung, K.H.; Yang, J.; Parker, I.; et al. Essential regulation of cell bioenergetics by constitutive insp(3) receptor ca2+ transfer to mitochondria. Cell 2010, 142, 270–283. [Google Scholar] [CrossRef] [PubMed]
- Szabadkai, G.; Bianchi, K.; Varnai, P.; De Stefani, D.; Wieckowski, M.R.; Cavagna, D.; Nagy, A.I.; Balla, T.; Rizzuto, R. Chaperone-mediated coupling of endoplasmic reticulum and mitochondrial ca2+ channels. J. Cell Biol. 2006, 175, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Filadi, R.; Pozzan, T. Generation and functions of second messengers microdomains. Cell Calcium 2015, 58, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Domingues, S.C.; Henriques, A.G.; Wu, W.; Da Cruz e Silva, E.F.; Da Cruz e Silva, O.A. Altered subcellular distribution of the Alzheimer’s amyloid precursor protein under stress conditions. Ann. N. Y. Acad. Sci. 2007, 1096, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Katayama, T.; Imaizumi, K.; Sato, N.; Miyoshi, K.; Kudo, T.; Hitomi, J.; Morihara, T.; Yoneda, T.; Gomi, F.; Mori, Y.; et al. Presenilin-1 mutations downregulate the signalling pathway of the unfolded-protein response. Nat. Cell Biol. 1999, 1, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Area-Gomez, E.; Del Carmen Lara Castillo, M.; Tambini, M.D.; Guardia-Laguarta, C.; de Groof, A.J.; Madra, M.; Ikenouchi, J.; Umeda, M.; Bird, T.D.; Sturley, S.L.; et al. Upregulated function of mitochondria-associated er membranes in Alzheimer disease. EMBO J. 2012, 31, 4106–4123. [Google Scholar] [CrossRef] [PubMed]
- Hedskog, L.; Pinho, C.M.; Filadi, R.; Ronnback, A.; Hertwig, L.; Wiehager, B.; Larssen, P.; Gellhaar, S.; Sandebring, A.; Westerlund, M.; et al. Modulation of the endoplasmic reticulum-mitochondria interface in Alzheimer’s disease and related models. Proc. Natl. Acad. Sci. USA 2013, 110, 7916–7921. [Google Scholar] [CrossRef] [PubMed]
- Hartl, F.U.; Bracher, A.; Hayer-Hartl, M. Molecular chaperones in protein folding and proteostasis. Nature 2011, 475, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Brehme, M.; Voisine, C.; Rolland, T.; Wachi, S.; Soper, J.H.; Zhu, Y.; Orton, K.; Villella, A.; Garza, D.; Vidal, M.; et al. A chaperome subnetwork safeguards proteostasis in aging and neurodegenerative disease. Cell Rep. 2014, 9, 1135–1150. [Google Scholar] [CrossRef] [PubMed]
- Baird, N.A.; Douglas, P.M.; Simic, M.S.; Grant, A.R.; Moresco, J.J.; Wolff, S.C.; Yates, J.R.; Manning, G.; Dillin, A. Hsf-1-mediated cytoskeletal integrity determines thermotolerance and life span. Science 2014, 346, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Brandvold, K.R.; Morimoto, R.I. The chemical biology of molecular chaperones-implications for modulation of proteostasis. J. Mol. Biol. 2015, 427, 2931–2947. [Google Scholar] [CrossRef] [PubMed]
- Duncan, E.J.; Cheetham, M.E.; Chapple, J.P.; van der Spuy, J. The role of hsp70 and its co-chaperones in protein misfolding, aggregation and disease. Subcell. Biochem. 2015, 78, 243–273. [Google Scholar] [PubMed]
- Pratt, W.B.; Gestwicki, J.E.; Osawa, Y.; Lieberman, A.P. Targeting hsp90/hsp70-based protein quality control for treatment of adult onset neurodegenerative diseases. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 353–371. [Google Scholar] [CrossRef] [PubMed]
- Agarraberes, F.A.; Dice, J.F. A molecular chaperone complex at the lysosomal membrane is required for protein translocation. J. Cell Sci. 2001, 114, 2491–2499. [Google Scholar] [PubMed]
- Hoshino, T.; Murao, N.; Namba, T.; Takehara, M.; Adachi, H.; Katsuno, M.; Sobue, G.; Matsushima, T.; Suzuki, T.; Mizushima, T. Suppression of Alzheimer’s disease-related phenotypes by expression of heat shock protein 70 in mice. J. Neurosci. 2011, 31, 5225–5234. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmus, M.M.M.; Boelens, W.C.; Otte-Holler, I.; Kamps, B.; Kusters, B.; Maat-Schieman, M.L.C.; Waal, R.M.W.; Verbeek, M.M. Small heat shock protein hspb8: Its distribution in Alzheimer’s disease brains and its inhibition of amyloid-beta protein aggregation and cerebrovascular amyloid-beta toxicity. Acta Neuropathol. 2006, 111, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Walther, D.M.; Kasturi, P.; Zheng, M.; Pinkert, S.; Vecchi, G.; Ciryam, P.; Morimoto, R.I.; Dobson, C.M.; Vendruscolo, M.; Mann, M.; et al. Widespread proteome remodeling and aggregation in aging c-elegans. Cell 2015, 161, 919–932. [Google Scholar] [CrossRef] [PubMed]
- Knowles, T.P.J.; Vendruscolo, M.; Dobson, C.M. The amyloid state and its association with protein misfolding diseases. Nat. Rev. Mol. Cell Biol. 2014, 15, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Gidalevitz, T.; Stevens, F.; Argon, Y. Orchestration of secretory protein folding by er chaperones. BBA-Mol. Cell Res. 2013, 1833, 2410–2424. [Google Scholar] [CrossRef] [PubMed]
- Jansen, G.; Maattanen, P.; Denisov, A.Y.; Scarffe, L.; Schade, B.; Balghi, H.; Dejgaard, K.; Chen, L.Y.; Muller, W.J.; Gehring, K.; et al. An interaction map of endoplasmic reticulum chaperones and foldases. Mol. Cell. Proteom. 2012, 11, 710–723. [Google Scholar] [CrossRef] [PubMed]
- Penke, B.; Fülöp, L.; Szűcs, M.; Frecska, E. The role of sigma-1 receptor, an intracellular chaperone in neurodegenerative diseases. Curr. Neuropharmacol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Leigh, P.N.; Whitwell, H.; Garofalo, O.; Buller, J.; Swash, M.; Martin, J.E.; Gallo, J.M.; Weller, R.O.; Anderton, B.H. Ubiquitin-immunoreactive intraneuronal inclusions in amyotrophic-lateral-sclerosis-morphology, distribution, and specificity. Brain 1991, 114, 775–788. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.A.; Ward, C.L.; Kopito, R.R. Aggresomes: A cellular response to misfolded proteins. J. Cell Biol. 1998, 143, 1883–1898. [Google Scholar] [CrossRef] [PubMed]
- Kopito, R.R. Aggresomes, inclusion bodies and protein aggregation. Trends Cell Biol. 2000, 10, 524–530. [Google Scholar] [CrossRef]
- Kaganovich, D.; Kopito, R.; Frydman, J. Misfolded proteins partition between two distinct quality control compartments. Nature 2008, 454, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Matsuda, N. Proteostasis and neurodegeneration: The roles of proteasomal degradation and autophagy. BBA-Mol. Cell Res. 2014, 1843, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.S.; Maldonado, M.A. The ubiquitin-proteasome system and its role in inflammatory and autoimmune diseases. Cell. Mol. Immunol. 2006, 3, 255–261. [Google Scholar] [PubMed]
- Mabb, A.M.; Ehlers, M.D. Ubiquitination in postsynaptic function and plasticity. Annu Rev. Cell Dev. Biol 2010, 26, 179–210. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, E.; Stoj, J.; Karpowicz, P.; Osmulski, P.A.; Gaczynska, M. The proteasome in health and disease. Curr. Pharm. Des. 2013, 19, 1010–1028. [Google Scholar] [PubMed]
- Gadhave, K.; Bolshette, N.; Ahire, A.; Pardeshi, R.; Thakur, K.; Trandafir, C.; Istrate, A.; Ahmed, S.; Lahkar, M.; Muresanu, D.F.; et al. The ubiquitin proteasomal system: A potential target for the management of Alzheimer’s disease. J. Cell. Mol. Med. 2016, 20, 1392–1407. [Google Scholar] [CrossRef] [PubMed]
- Ciechanover, A.; Kwon, Y.T. Degradation of misfolded proteins in neurodegenerative diseases: Therapeutic targets and strategies. Exp. Mol. Med. 2015, 47, e147. [Google Scholar] [CrossRef] [PubMed]
- Ryu, K.Y.; Sinnar, S.A.; Reinholdt, L.G.; Vaccari, S.; Hall, S.; Garcia, M.A.; Zaitseva, T.S.; Bouley, D.M.; Boekelheide, K.; Handel, M.A.; et al. The mouse polyubiquitin gene ubb is essential for meiotic progression. Mol. Cell. Biol. 2008, 28, 1136–1146. [Google Scholar] [CrossRef] [PubMed]
- Vilchez, D.; Saez, I.; Dillin, A. The role of protein clearance mechanisms in organismal ageing and age-related diseases. Nat. Commun. 2014, 5, 5659. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Baehrecke, E.H.; Ballabio, A.; Boya, P.; Bravo-San Pedro, J.M.; Cecconi, F.; Choi, A.M.; Chu, C.T.; Codogno, P.; Colombo, M.I.; et al. Molecular definitions of autophagy and related processes. EMBO J. 2017, 36, 1811–1836. [Google Scholar] [CrossRef] [PubMed]
- Ravikumar, B.; Sarkar, S.; Davies, J.E.; Futter, M.; Garcia-Arencibia, M.; Green-Thompson, Z.W.; Jimenez-Sanchez, M.; Korolchuk, V.I.; Lichtenberg, M.; Luo, S.Q.; et al. Regulation of mammalian autophagy in physiology and pathophysiology. Physiol. Rev. 2010, 90, 1383–1435. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A. The role of autophagy in neurodegenerative disease. Nat. Med. 2013, 19, 983–997. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, D.M.; Lee, J.H.; Kumar, A.; Lee, S.; Orenstein, S.J.; Nixon, R.A. Autophagy failure in Alzheimer’s disease and the role of defective lysosomal acidification. Eur. J. Neurosci. 2013, 37, 1949–1961. [Google Scholar] [CrossRef] [PubMed]
- Whyte, L.S.; Lau, A.A.; Hemsley, K.M.; Hopwood, J.J.; Sargeant, T.J. Endo-lysosomal and autophagic dysfunction: A driving factor in Alzheimer’s disease? J. Neurochem. 2017, 140, 703–717. [Google Scholar] [CrossRef] [PubMed]
- Moreau, K.; Fleming, A.; Imarisio, S.; Ramirez, A.L.; Mercer, J.L.; Jimenez-Sanchez, M.; Bento, C.F.; Puri, C.; Zavodszky, E.; Siddiqi, F.; et al. Picalm modulates autophagy activity and tau accumulation. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A.; Wegiel, J.; Kumar, A.; Yu, W.H.; Peterhoff, C.; Cataldo, A.; Cuervo, A.M. Extensive involvement of autophagy in Alzheimer disease: An immuno-electron microscopy study. J. Neuropathol. Exp. Neurol. 2005, 64, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Boland, B.; Kumar, A.; Lee, S.; Platt, F.M.; Wegiel, J.; Yu, W.H.; Nixon, R.A. Autophagy induction and autophagosome clearance in neurons: Relationship to autophagic pathology in Alzheimer’s disease. J. Neurosci. 2008, 28, 6926–6937. [Google Scholar] [CrossRef] [PubMed]
- Khalil, H.; Tazi, M.; Caution, K.; Ahmed, A.; Kanneganti, A.; Assani, K.; Kopp, B.; Marsh, C.; Dakhlallah, D.; Amer, A.O. Aging is associated with hypermethylation of autophagy genes in macrophages. Epigenetics 2016, 11, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Jing, Y.Y.; Kang, X.; Yang, L.; Wang, D.L.; Zhang, W.; Zhang, L.; Chen, P.; Chang, J.F.; Yang, X.M.; et al. Histone h2b monoubiquitination is a critical epigenetic switch for the regulation of autophagy. Nucleic Acids Res. 2017, 45, 1144–1158. [Google Scholar] [CrossRef] [PubMed]
- Fullgrabe, J.; Klionsky, D.J.; Joseph, B. The return of the nucleus: Transcriptional and epigenetic control of autophagy. Nat. Rev. Mol. Cell Biol. 2014, 15, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Friedman, L.G.; Qureshi, Y.H.; Yu, W.H. Promoting autophagic clearance: Viable therapeutic targets in Alzheimer’s disease. Neurotherapeutics 2015, 12, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Zlokovic, B.V. Clearing amyloid through the blood-brain barrier. J. Neurochem. 2004, 89, 807–811. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, J.C. Vascular risk factor detection and control may prevent Alzheimer’s disease. Ageing Res. Rev. 2010, 9, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Zenaro, E.; Piacentino, G.; Constantin, G. The blood-brain barrier in Alzheimer’s disease. Neurobiol. Dis. 2016, 41–56. [Google Scholar] [CrossRef] [PubMed]
- Di Marco, L.Y.; Venneri, A.; Farkas, E.; Evans, P.C.; Marzo, A.; Frangi, A.F. Vascular dysfunction in the pathogenesis of Alzheimer’s disease—A review of endothelium-mediated mechanisms and ensuing vicious circles. Neurobiol. Dis. 2015, 82, 593–606. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A. From blood-brain barrier to blood-brain interface: New opportunities for cns drug delivery. Nat. Rev. Drug Discov. 2016, 15, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Alafuzoff, I.; Adolfsson, R.; Grundke-Iqbal, I.; Winblad, B. Blood-brain barrier in Alzheimer dementia and in non-demented elderly. An immunocytochemical study. Acta Neuropathol. 1987, 73, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Kress, B.T.; Iliff, J.J.; Xia, M.S.; Wang, M.H.; Wei, H.L.S.; Zeppenfeld, D.; Xie, L.L.; Kang, H.Y.; Xu, Q.W.; Liew, J.A.; et al. Impairment of paravascular clearance pathways in the aging brain. Ann. Neurol. 2014, 76, 845–861. [Google Scholar] [CrossRef] [PubMed]
- Zlokovic, B.V. Neurovascular mechanisms of Alzheimer’s neurodegeneration. Trends Neurosci. 2005, 28, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, G.D.; Messier, A.A.; Miller, M.C.; Machan, J.T.; Majmudar, S.S.; Stopa, E.G.; Donahue, J.E.; Johanson, C.E. Amyloid efflux transporter expression at the blood-brain barrier declines in normal aging. J. Neuropathol. Exp. Neurol. 2010, 69, 1034–1043. [Google Scholar] [CrossRef] [PubMed]
- Wijesuriya, H.C.; Bullock, J.Y.; Faull, R.L.M.; Hladky, S.B.; Barrand, M.A. Abc efflux transporters in brain vasculature of Alzheimer’s subjects. Brain Res. 2010, 1358, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A.; Kumar, V.B.; Farr, S.A.; Nakaoke, R.; Robinson, S.M.; Morley, J.E. Impairments in brain-to-blood transport of amyloid-beta and reabsorption of cerebrospinal fluid in an animal model of Alzheimer’s disease are reversed by antisense directed against amyloid-beta protein precursor. J. Alzheimers Dis. 2011, 23, 599–605. [Google Scholar] [PubMed]
- Sengillo, J.D.; Winkler, E.A.; Walker, C.T.; Sullivan, J.S.; Johnson, M.; Zlokovic, B.V. Deficiency in mural vascular cells coincides with blood-brain barrier disruption in Alzheimer’s disease. Brain Pathol. 2013, 23, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Talbot, K.; Wang, H.Y.; Kazi, H.; Han, L.Y.; Bakshi, K.P.; Stucky, A.; Fuino, R.L.; Kawaguchi, K.R.; Samoyedny, A.J.; Wilson, R.S.; et al. Demonstrated brain insulin resistance in Alzheimer’s disease patients is associated with igf-1 resistance, irs-1 dysregulation, and cognitive decline. J. Clin. Investig. 2012, 122, 1316–1338. [Google Scholar] [CrossRef] [PubMed]
- Reger, M.A.; Watson, G.S.; Green, P.S.; Baker, L.D.; Cholerton, B.; Fishel, M.A.; Plymate, S.R.; Cherrier, M.M.; Schellenberg, G.D.; Frey, W.H.; et al. Intranasal insulin administration dose-dependently modulates verbal memory and plasma amyloid-beta in memory-impaired older adults. J. Alzheimers Dis. 2008, 13, 323–331. [Google Scholar] [CrossRef] [PubMed]
- McGeer, P.L.; McGeer, E.G. The amyloid cascade-inflammatory hypothesis of Alzheimer disease: Implications for therapy. Acta Neuropathol. 2013, 126, 479–497. [Google Scholar] [CrossRef] [PubMed]
- Takata, K.; Kitamura, Y.; Saeki, M.; Terada, M.; Kagitani, S.; Kitamura, R.; Fujikawa, Y.; Maelicke, A.; Tomimoto, H.; Taniguchi, T.; et al. Galantamine-induced amyloid-beta clearance mediated via stimulation of microglial nicotinic acetylcholine receptors. J. Biol. Chem. 2010, 285, 40180–40191. [Google Scholar] [CrossRef] [PubMed]
- Go, M.; Kou, J.H.; Lim, J.E.; Yang, J.L.; Fukuchi, K.I. Microglial response to lps increases in wild-type mice during aging but diminishes in an Alzheimer’s mouse model: Implication of tlr4 signaling in disease progression. Biochem. Biophys. Res. Commun. 2016, 479, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Sheedy, F.J.; Grebe, A.; Rayner, K.J.; Kalantari, P.; Ramkhelawon, B.; Carpenter, S.B.; Becker, C.E.; Ediriweera, H.N.; Mullick, A.E.; Golenbock, D.T.; et al. Cd36 coordinates nlrp3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nat. Immunol. 2013, 14, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Lenart, N.; Brough, D.; Denes, A. Inflammasomes link vascular disease with neuroinflammation and brain disorders. J. Cereb. Blood Flow Metab. 2016, 36, 1668–1685. [Google Scholar] [CrossRef] [PubMed]
- Matcovitch-Natan, O.; Winter, D.R.; Giladi, A.; Aguilar, S.V.; Spinrad, A.; Sarrazin, S.; Ben-Yehuda, H.; David, E.; Gonzalez, F.Z.; Perrin, P.; et al. Microglia development follows a stepwise program to regulate brain homeostasis. Science 2016, 353. [Google Scholar] [CrossRef] [PubMed]
- Ransohoff, R.M.; El Khoury, J. Microglia in health and disease. CSH Perspect Biol. 2016, 8, a020560. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Beja-Glasser, V.F.; Nfonoyim, B.M.; Frouin, A.; Li, S.M.; Ramakrishnan, S.; Merry, K.M.; Shi, Q.Q.; Rosenthal, A.; Barres, B.A.; et al. Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science 2016, 352, 712–716. [Google Scholar] [CrossRef] [PubMed]
- Elali, A.; Rivest, S. Microglia in Alzheimer’s disease: A multifaceted relationship. Brain Behav. Immun. 2016, 55, 138–150. [Google Scholar] [CrossRef] [PubMed]
- McDade, E.; Bateman, R.J. Stop Alzheimer’s before it starts. Nature 2017, 547, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Bachurin, S.O.; Bovina, E.V.; Ustyugov, A.A. Drugs in clinical trials for Alzheimer’s disease: The major trends. Med. Res. Rev. 2017, 37, 1186–1225. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, M.; Snyder, H.M.; Carrillo, M.C.; Fazio, S.; Kim, H.; Johns, H. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimers Dement. 2015, 11, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.; Aisen, P.S.; DuBois, B.; Frolich, L.; Jack, C.R., Jr.; Jones, R.W.; Morris, J.C.; Raskin, J.; Dowsett, S.A.; Scheltens, P. Drug development in Alzheimer’s disease: The path to 2025. Alzheimers Res. Ther. 2016, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Cazarim, M.D.; Moriguti, J.C.; Ogunjimi, A.T.; Pereira, L.R.L. Perspectives for treating Alzheimer’s disease: A review on promising pharmacological substances. Sao Paulo Med. J. 2016, 134, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Parsons, C.G.; Rammes, G. Preclinical to phase 2 amyloid beta (a) peptide modulators under investigation for Alzheimer’s disease. Expert Opin. Inv. Drug 2017, 26, 579–591. [Google Scholar] [CrossRef] [PubMed]
| List of Diseases | Misfolded Proteins |
|---|---|
| Alzheimer’s disease | β-amyloid (Aβ) |
| hyperphosphorylated Tau (pTau) | |
| α-synuclein | |
| TDP-43 | |
| Parkinson’s disease | α-synuclein |
| Huntington disease | huntingtin |
| Lewy-body dementia | α-synuclein |
| Amyotrophic lateral scelerosis | TDP-43 |
| Prion diseases | superoxide dismutase |
| prion protein (PrPsc) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Penke, B.; Bogár, F.; Fülöp, L. β-Amyloid and the Pathomechanisms of Alzheimer’s Disease: A Comprehensive View. Molecules 2017, 22, 1692. https://doi.org/10.3390/molecules22101692
Penke B, Bogár F, Fülöp L. β-Amyloid and the Pathomechanisms of Alzheimer’s Disease: A Comprehensive View. Molecules. 2017; 22(10):1692. https://doi.org/10.3390/molecules22101692
Chicago/Turabian StylePenke, Botond, Ferenc Bogár, and Lívia Fülöp. 2017. "β-Amyloid and the Pathomechanisms of Alzheimer’s Disease: A Comprehensive View" Molecules 22, no. 10: 1692. https://doi.org/10.3390/molecules22101692
APA StylePenke, B., Bogár, F., & Fülöp, L. (2017). β-Amyloid and the Pathomechanisms of Alzheimer’s Disease: A Comprehensive View. Molecules, 22(10), 1692. https://doi.org/10.3390/molecules22101692






