5-Hydroxycyclopenicillone Inhibits β-Amyloid Oligomerization and Produces Anti-β-Amyloid Neuroprotective Effects In Vitro
Abstract
:1. Introduction
2. Results
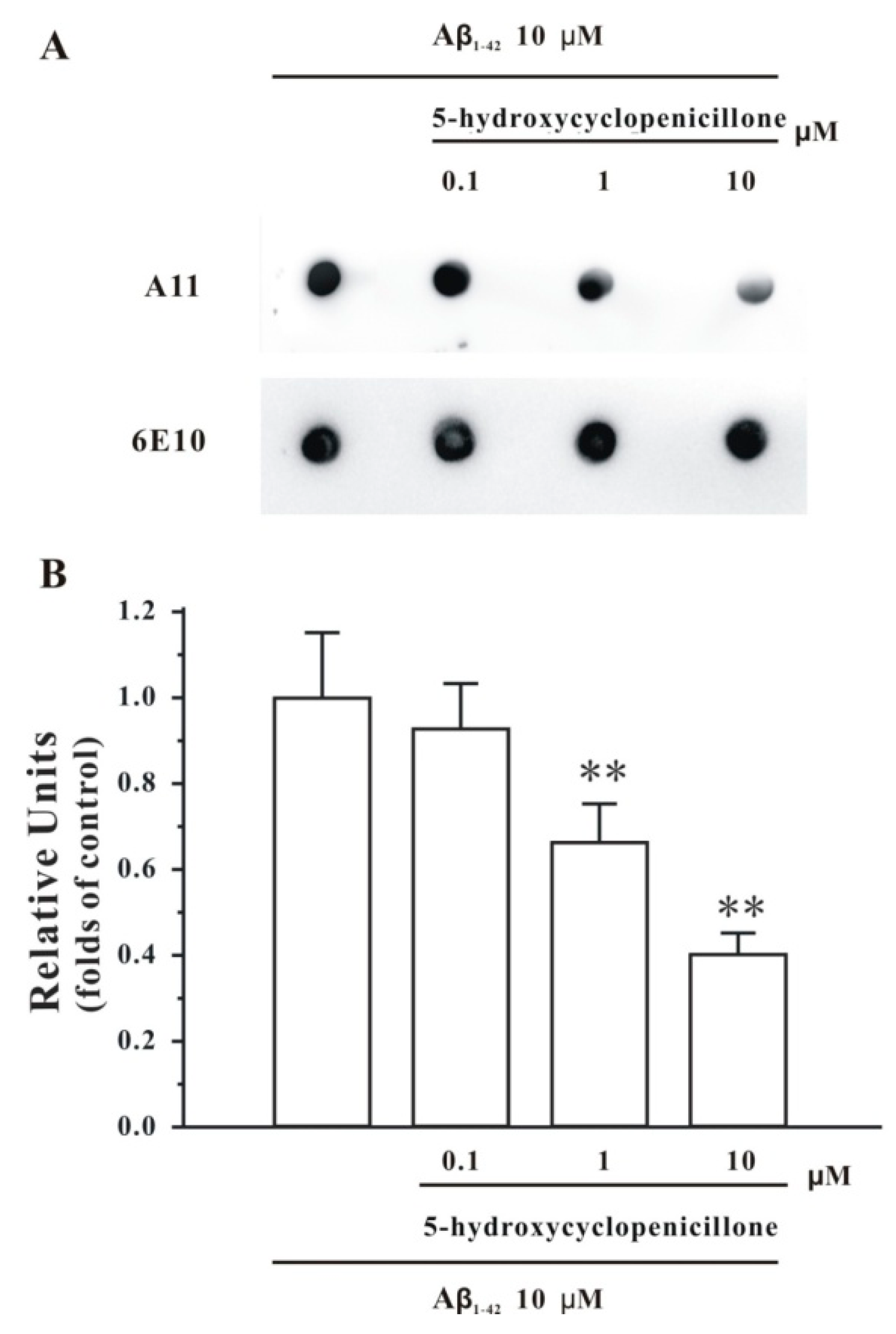
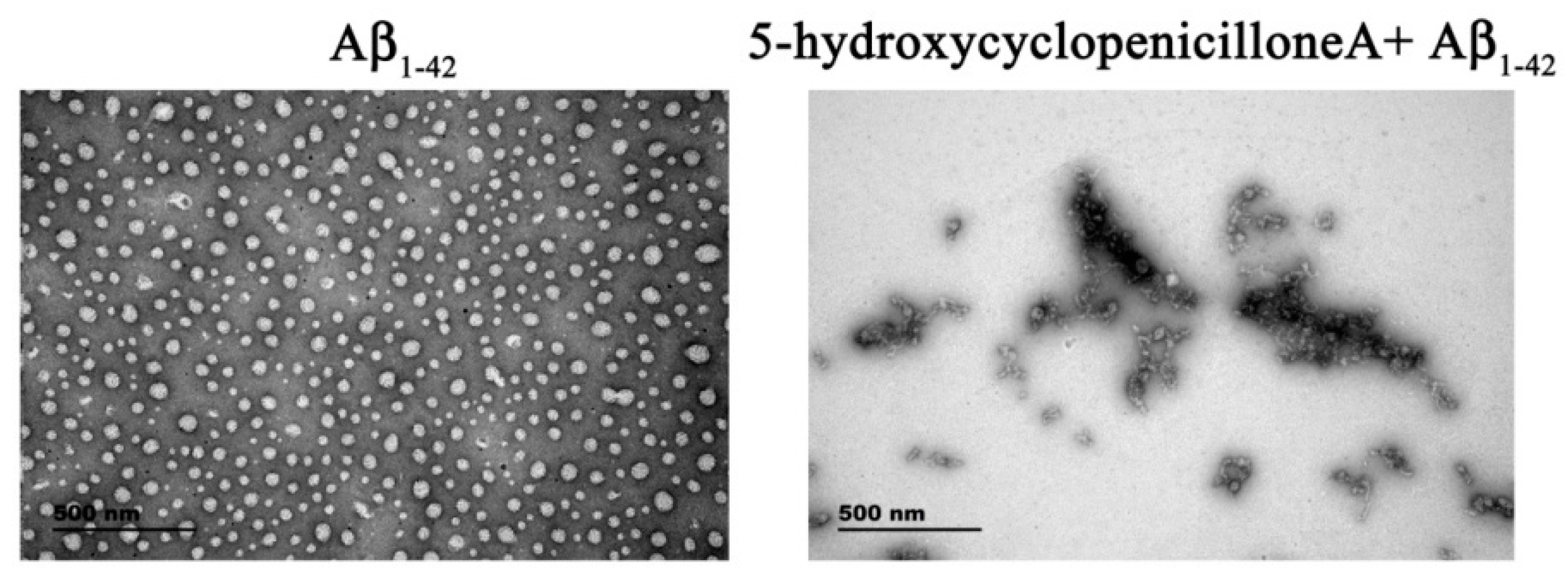
2.1. 5-Hydroxycyclopenicillone Inhibits Aβ1-42 Oligomer Formation
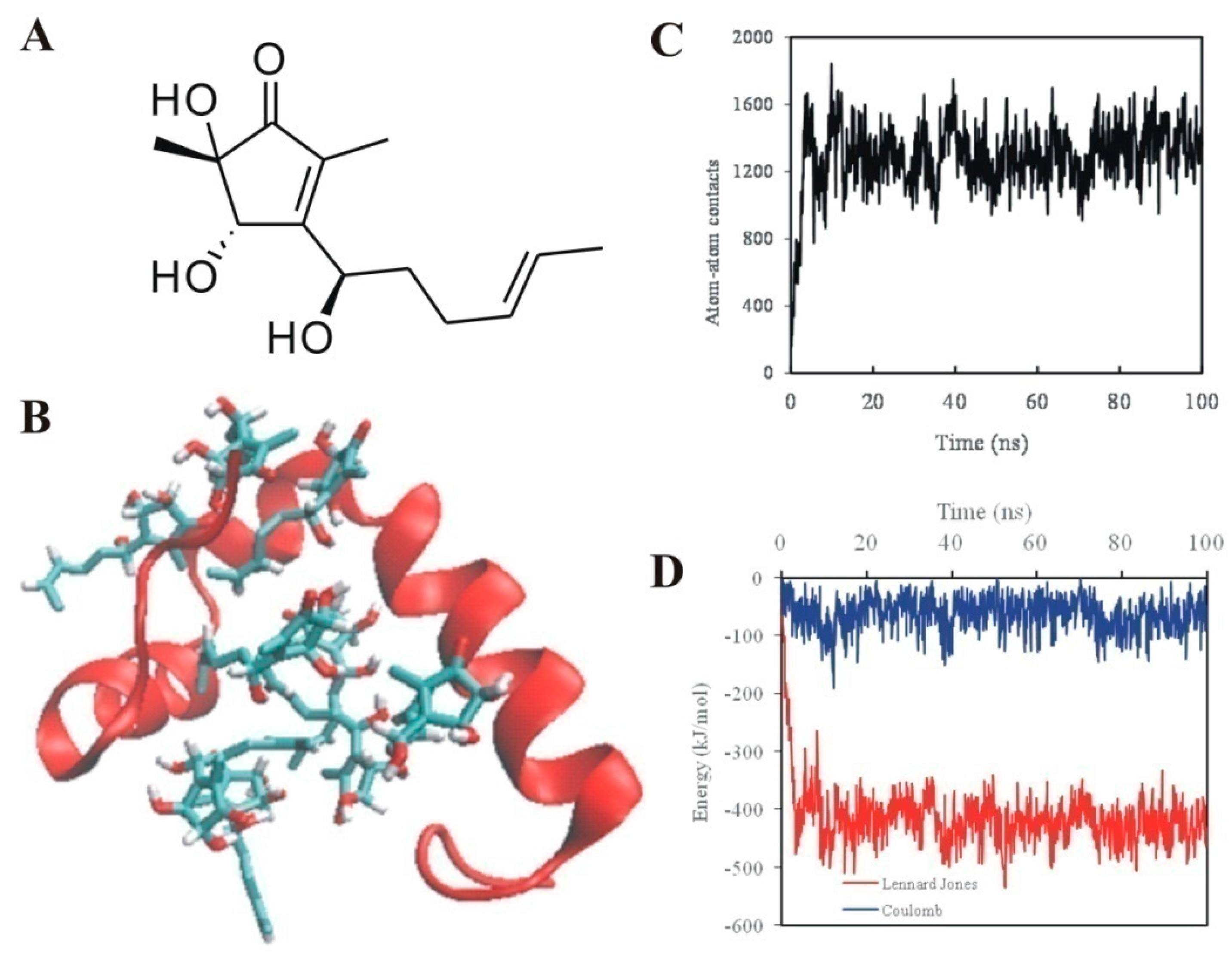
2.2. 5-Hydroxycyclopenicillone Likely Binds to Aβ1-42 Peptides via Hydrophobic Interactions
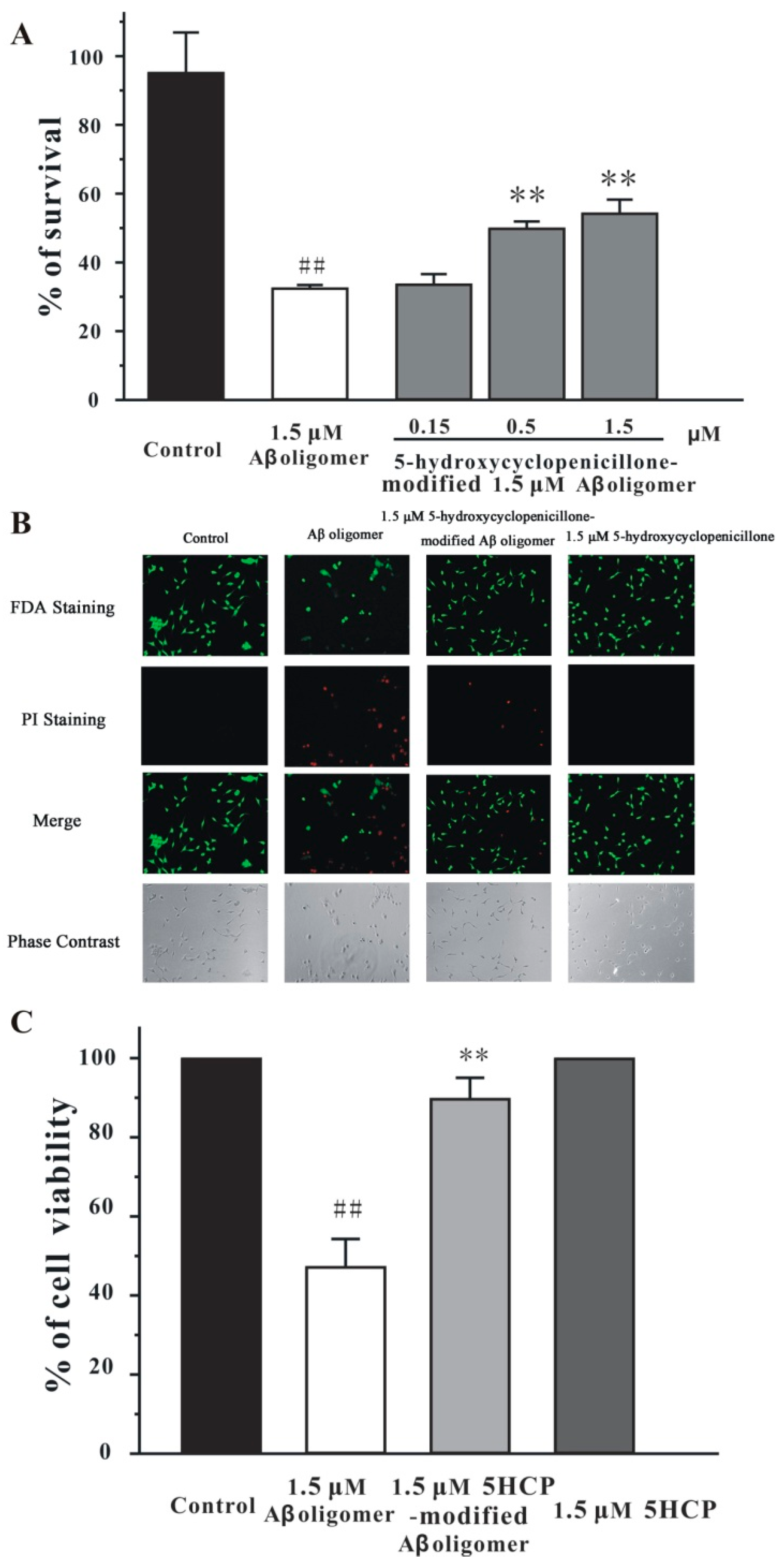
2.3. 5-Hydroxycyclopenicillone Decreased the Neurotoxicity of the Aβ1-42 Oligomer in SH-SY5Y Cells
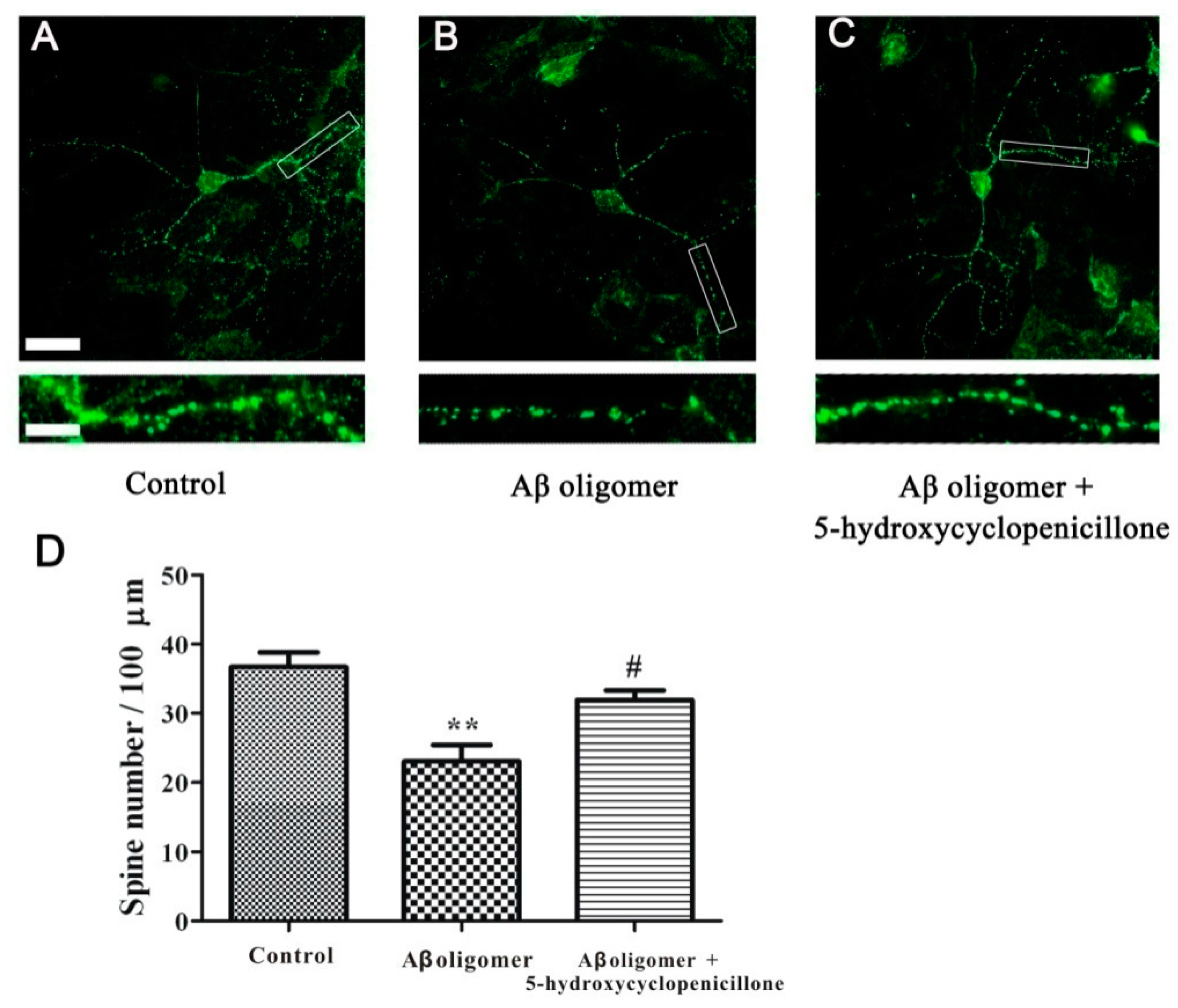
2.4. 5-Hydroxycyclopenicillone Prevents Synaptic Toxicity of Aβ1-42 Oligomer in Primary Hippocampal Neurons
3. Discussion
4. Materials and Methods
4.1. Preparation of 5-Hydroxycyclopenicillone
4.2. Preparation of the Aβ1-42 Oligomer
4.3. Dot Blotting Analysis
4.4. TEM Analysis
4.5. Simulation System
4.6. Molecular Dynamics Simulation
4.7. Molecular Dynamics Simulation Analysis
4.8. Culture of SH-SY5Y Cells
4.9. Cell Viability Measurement
4.10. FDA/PI Double Staining
4.11. Primary Hippocampal Neuronal Cultures
4.12. Immunocyto Chemisty
4.13. Confocal Imaging and Analysis
4.14. Data Analysis and Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Scheltens, P.; Blennow, K.; Breteler, M.M.B.; de Strooper, B.; Frisoni, G.B.; Salloway, S.; van der Flier, W.M. Alzheimer’s disease. Lancet 2016, 388, 505–517. [Google Scholar] [CrossRef]
- Viola, K.L.; Klein, W.L. Amyloid beta oligomers in Alzheimer’s disease pathogenesis, treatment, and diagnosis. Acta Neuropathol. 2015, 129, 183–206. [Google Scholar] [CrossRef] [PubMed]
- Selkoe, D.J.; Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol. Med. 2016, 8, 595–608. [Google Scholar] [CrossRef] [PubMed]
- Salahuddin, P.; Fatima, M.T.; Abdelhameed, A.S.; Nusrat, S.; Khan, R.H. Structure of amyloid oligomers and their mechanisms of toxicities: Targeting amyloid oligomers using novel therapeutic approaches. Eur. J. Med. Chem. 2016, 114, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Thapa, A.; Jett, S.D.; Chi, E.Y. Curcumin Attenuates Arnyloid-beta Aggregate Toxicity and Modulates Amyloid-beta Aggregation Pathway. ACS Chem. Neurosci. 2016, 7, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Du, W.J.; Guo, J.J.; Gao, M.T.; Hu, S.Q.; Dong, X.Y.; Han, Y.F.; Liu, F.F.; Jiang, S.Y.; Sun, Y. Brazilin inhibits amyloid beta-protein fibrillogenesis, remodels amyloid fibrils and reduces amyloid cytotoxicity functional. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
- Bieschke, J.; Herbst, M.; Wiglenda, T.; Friedrich, R.P.; Boeddrich, A.; Schiele, F.; Kleckers, D.; del Amo, J.M.L.; Gruning, B.A.; Wang, Q.W.; et al. Small-molecule conversion of toxic oligomers to nontoxic beta-sheet-rich amyloid fibrils. Nat. Chem. Biol. 2012, 8, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Torres, V.; Encinar, J.A.; Herranz-Lopez, M.; Perez-Sanchez, A.; Galiano, V.; Barrajon-Catalan, E.; Micol, V. An Updated Review on Marine Anticancer Compounds: The Use of Virtual Screening for the Discovery of Small-Molecule Cancer Drugs. Molecules 2017, 22, 1037. [Google Scholar] [CrossRef] [PubMed]
- Shanmuganathan, B.; Malar, D.S.; Sathya, S.; Devi, K.P. Antiaggregation Potential of Padina gymnospora against the Toxic Alzheimer’s Beta-Amyloid Peptide(25–35) and Cholinesterase Inhibitory Property of Its Bioactive Compounds. PLoS ONE 2015, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.Y.; Liu, F.F.; Lin, J.J.; Chen, H.X.; Huang, C.H.; Chen, L.P.; Zhou, Y.Y.; Ye, L.Y.; Zhang, K.; Jin, J.K.; et al. Fucoxanthin Inhibits beta-Amyloid Assembly and Attenuates beta-Amyloid Oligomer-Induced Cognitive Impairments. J. Agric. Food Chem. 2017, 65, 4092–4102. [Google Scholar] [CrossRef] [PubMed]
- Bryukhovetskiy, I.; Lyakhova, I.; Mischenko, P.; Milkina, E.; Zaitsev, S.; Khotimchenko, Y.; Bryukhovetskiy, A.; Polevshchikov, A.; Kudryavtsev, I.; Khotimchenko, M.; et al. Alkaloids of fascaplysin are effective conventional chemotherapeutic drugs, inhibiting the proliferation of C6 glioma cells and causing their death in vitro. Oncol. Lett. 2017, 13, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Zhao, J.Y.; Ding, L.J.; Huang, C.H.; Naman, C.B.; He, S.; Wu, B.; Zhu, P.; Luo, Q.J.; Gerwick, W.H.; et al. 5-Hydroxycyclopenicillone, a New beta-Amyloid Fibrillization Inhibitor from a Sponge-Derived Fungus Trichoderma sp HPQJ-34. Mar. Drugs 2017, 15, 260. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhao, C.Q.; Yang, X.J.; Ren, J.S.; Xu, C.; Qu, X.G. In Situ Monitoring Alzheimer’s Disease ss-Amyloid Aggregation and Screening of A ss Inhibitors Using a Perylene Probe. Small 2013, 9, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Culyba, E.K.; Powers, E.T.; Kelly, J.W. Amyloid-beta forms fibrils by nucleated conformational conversion of oligomers. Nat. Chem. Biol. 2011, 7, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Laganowsky, A.; Liu, C.; Sawaya, M.R.; Whitelegge, J.P.; Park, J.; Zhao, M.L.; Pensalfini, A.; Soriaga, A.B.; Landau, M.; Teng, P.K.; et al. Atomic View of a Toxic Amyloid Small Oligomer. Science 2012, 335, 1228–1231. [Google Scholar] [CrossRef] [PubMed]
- Morgado, I.; Wieligmann, K.; Bereza, M.; Ronicke, R.; Meinhardt, K.; Annamalai, K.; Baumann, M.; Wacker, J.; Hortschansky, P.; Malesevic, M.; et al. Molecular basis of beta-amyloid oligomer recognition with a conformational antibody fragment. Proc. Natl. Acad. Sci. USA 2012, 109, 12503–12508. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.Q.; Wang, R.; Cui, W.; Mak, S.H.; Li, G.; Hu, Y.J.; Lee, M.Y.; Pang, Y.P.; Han, Y.F. Dimeric bis (heptyl)-Cognitin Blocks Alzheimer’s beta-Amyloid Neurotoxicity Via the Inhibition of A beta Fibrils Formation and Disaggregation of Preformed Fibrils. CNS Neurosci. Ther. 2015, 21, 953–961. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.T.; Huang, M.; Xu, S.J.; Wang, Y.; An, P.Y.; Feng, C.X.; Chen, X.W.; Wei, X.F.; Han, Y.F.; Wang, Q.W. Bis(propyl)-cognitin Prevents beta-amyloid-induced Memory Deficits as Well as Synaptic Formation and Plasticity Impairments via the Activation of PI3-K Pathway. Mol. Neurobiol. 2016, 53, 3832–3841. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Cui, W.; Yang, Y.; Xu, S.J.; Zhou, W.H.; Fu, H.J.; Hu, S.Q.; Mak, S.H.; Hu, J.W.; Wang, Q.; et al. Protection against beta-amyloid-induced synaptic and memory impairments via altering beta-amyloid assembly by bis(heptyl)-cognitin. Sci. Rep-Uk 2015, 5, 10256. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.J.; Liu, G.L.; Bao, X.M.; Wu, J.; Li, S.M.; Zheng, B.X.; Anwyl, R.; Wang, Q.W. Rosiglitazone Prevents Amyloid-beta Oligomer-Induced Impairment of Synapse Formation and Plasticity via Increasing Dendrite and Spine Mitochondrial Number. J. Alzheimers Dis. 2014, 39, 239–251. [Google Scholar] [PubMed]
- Laport, M.S.; Santos, O.C.S.; Muricy, G. Marine Sponges: Potential Sources of New Antimicrobial Drugs. Curr. Pharm. Biotechnol. 2009, 10, 86–105. [Google Scholar] [CrossRef] [PubMed]
- Santos, O.C.S.; Soares, A.R.; Machado, F.L.S.; Romanos, M.T.V.; Muricy, G.; Giambiagi-deMarval, M.; Laport, M.S. Investigation of biotechnological potential of sponge-associated bacteria collected in Brazilian coast. Lett. Appl. Microbiol. 2015, 60, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Indraningrat, A.A.G.; Smidt, H.; Sipkema, D. Bioprospecting Sponge-Associated Microbes for Antimicrobial Compounds. Marinedrugs 2016, 14, 87. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.W.; Radax, R.; Steger, D.; Wagner, M. Sponge-associated microorganisms: Evolution, ecology, and biotechnological potential. Microbiol. Mol. Biol. Rev. 2007, 71, 295. [Google Scholar] [CrossRef] [PubMed]
- Fuerst, J.A. Diversity and biotechnological potential of microorganisms associated with marine sponges. Appl. Microbiol. Biotechnol. 2014, 98, 7331–7347. [Google Scholar] [CrossRef] [PubMed]
- Jarosz-Griffiths, H.H.; Noble, E.; Rushworth, J.V.; Hooper, N.M. Amyloid- Receptors: The Good, the Bad, and the Prion Protein. J. Biol. Chem. 2016, 291, 3174–3183. [Google Scholar] [CrossRef] [PubMed]
- Hielscher-Michael, S.; Griehl, C.; Buchholz, M.; Demuth, H.U.; Arnold, N.; Wessjohann, L.A. Natural Products from Microalgae with Potential against Alzheimer’s Disease: Sulfolipids Are Potent Glutaminyl Cyclase Inhibitors. Marinedrugs 2016, 14, 203. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Song, L.; Jope, R.S. Cholinergic stimulation of AP-1 and NF kappa B transcription factors is differentially sensitive to oxidative stress in SH-SY5Y neuroblastoma: relationship to phosphoinositide hydrolysis. J. Neurosci. 1996, 16, 5914–5922. [Google Scholar] [PubMed]
- Nirmaladevi, D.; Venkataramana, M.; Chandranayaka, S.; Ramesha, A.; Jameel, N.M.; Srinivas, C. Neuroprotective effects of bikaverin on H2O2-induced oxidative stress mediated neuronal damage in SH-SY5Y cell line. Cell. Mol. Neurobiol. 2014, 34, 973–985. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Guo, L.P.; Hu, X.L.; Huang, J.; Fan, Y.H.; Ren, T.S.; Zhao, Q.C. Protective effects of Arctium lappa L. roots against hydrogen peroxide-induced cell injury and potential mechanisms in SH-SY5Y cells. Cell. Mol. Neurobiol. 2015, 35, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Tomaselli, S.; Esposito, V.; Vangone, P.; van Nuland, N.A.; Bonvin, A.M.; Guerrini, R.; Tancredi, T.; Temussi, P.A.; Picone, D. The alpha-to-beta conformational transition of Alzheimer’s Abeta-(1–42) peptide in aqueous media is reversible: A step by step conformational analysis suggests the location of beta conformation seeding. ChemBioChem 2006, 7, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Zhang, Z.; Li, W.; Hu, S.; Mak, S.; Zhang, H.; Han, R.; Yuan, S.; Li, S.; Sa, F.; et al. The anti-cancer agent SU4312 unexpectedly protects against MPP(+) -induced neurotoxicity via selective and direct inhibition of neuronal NOS. Br. J. Pharmacol. 2013, 168, 1201–1214. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Zhang, Z.J.; Hu, S.Q.; Mak, S.H.; Xu, D.P.; Choi, C.L.; Wang, Y.Q.; Tsim, W.K.; Lee, M.Y.; Rong, J.H.; et al. Sunitinib produces neuroprotective effect via inhibiting nitric oxide overproduction. CNS Neurosci. Ther. 2014, 20, 244–252. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, J.; Liu, F.; Huang, C.; Shentu, J.; Wang, M.; Sun, C.; Chen, L.; Yan, S.; Fang, F.; Wang, Y.; et al. 5-Hydroxycyclopenicillone Inhibits β-Amyloid Oligomerization and Produces Anti-β-Amyloid Neuroprotective Effects In Vitro. Molecules 2017, 22, 1651. https://doi.org/10.3390/molecules22101651
Zhao J, Liu F, Huang C, Shentu J, Wang M, Sun C, Chen L, Yan S, Fang F, Wang Y, et al. 5-Hydroxycyclopenicillone Inhibits β-Amyloid Oligomerization and Produces Anti-β-Amyloid Neuroprotective Effects In Vitro. Molecules. 2017; 22(10):1651. https://doi.org/10.3390/molecules22101651
Chicago/Turabian StyleZhao, Jiaying, Fufeng Liu, Chunhui Huang, Jieyi Shentu, Minjun Wang, Chenkai Sun, Liping Chen, Sicheng Yan, Fang Fang, Yuanyuan Wang, and et al. 2017. "5-Hydroxycyclopenicillone Inhibits β-Amyloid Oligomerization and Produces Anti-β-Amyloid Neuroprotective Effects In Vitro" Molecules 22, no. 10: 1651. https://doi.org/10.3390/molecules22101651
APA StyleZhao, J., Liu, F., Huang, C., Shentu, J., Wang, M., Sun, C., Chen, L., Yan, S., Fang, F., Wang, Y., Xu, S., Naman, C. B., Wang, Q., He, S., & Cui, W. (2017). 5-Hydroxycyclopenicillone Inhibits β-Amyloid Oligomerization and Produces Anti-β-Amyloid Neuroprotective Effects In Vitro. Molecules, 22(10), 1651. https://doi.org/10.3390/molecules22101651







