Phlorotannins: Towards New Pharmacological Interventions for Diabetes Mellitus Type 2
Abstract
:1. Introduction
2. Diabetes Mellitus Type 2
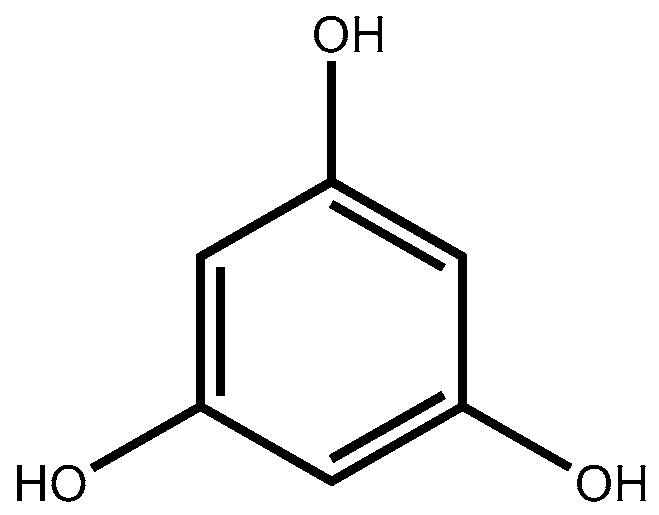
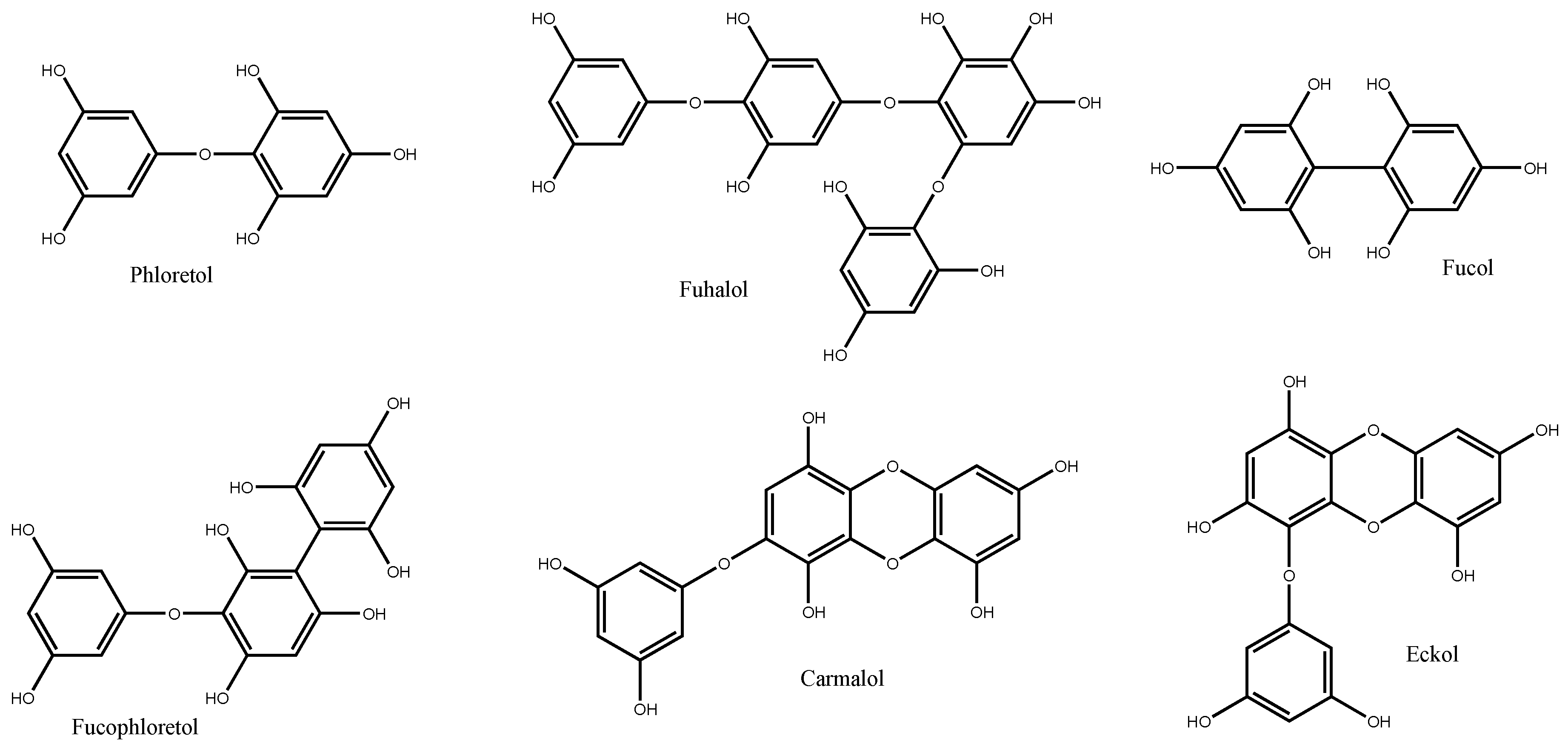
3. Phlorotannins
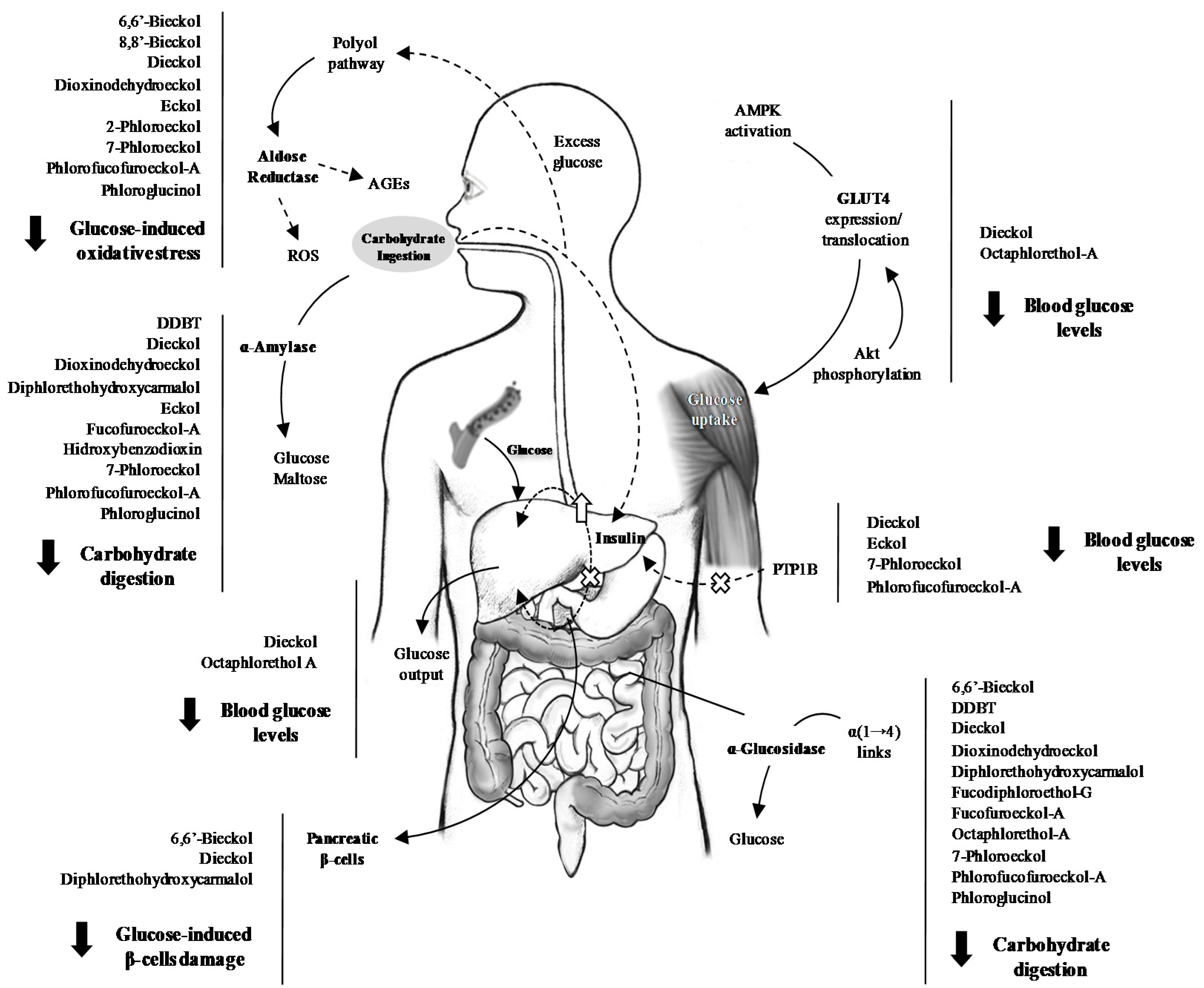
3.1. Inhibition of Glucose Absorption
3.2. Effects on Postprandial Hyperglycaemia and Insulin Levels In Vivo
3.3. Endogenous Glucose Production
3.4. Aldose Reductase and the Polyol Pathway
3.5. Glucose Uptake
3.5.1. Protein Tyrosine Phosphatase 1B (PTP1B)
3.5.2. Glucose Transporter 4 (GLUT4)
3.6. Glucose-Induced Toxicity
3.6.1. AGEs
3.6.2. Protection of Pancreatic β-Cells
4. Study Perspectives and Conclusions
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association. Classification and diagnosis of diabetes. Diabetes Care 2016, 39, 13–22. [Google Scholar]
- World Health Organization. Global Report on Diabetes; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabetes Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Emerging Risk Factors Collaboration. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: A collaborative meta-analysis of 102 prospective studies. Lancet 2010, 375, 2215–2222. [Google Scholar]
- Hickman, I.; Jonsson, J.; Prins, J.; Ash, S.; Purdie, D.; Clouston, A.; Powell, E.E. Modest weight loss and physical activity in overweight patients with chronic liver disease results in sustained improvements in alanine aminotransferase, fasting insulin, and quality of life. Gut 2004, 53, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Krentz, A.J.; Bailey, C.J. Oral Antidiabetic Agents. Drugs 2005, 65, 385–411. [Google Scholar] [CrossRef] [PubMed]
- Sapkota, S.; Brien, J.-A.; Greenfield, J.; Aslani, P. A systematic review of interventions addressing adherence to anti-diabetic medications in patients with type 2 diabetes—Impact on adherence. PLoS ONE 2015, 10, e0118296. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.; Valentão, P.; Andrade, P.B. Bioactive compounds from macroalgae in the new millennium: Implications for neurodegenerative diseases. Mar. Drugs 2014, 12, 4934–4972. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2013, 30, 237–323. [Google Scholar] [CrossRef] [PubMed]
- Leal, M.C.; Puga, J.; Serôdio, J.; Gomes, N.C.; Calado, R. Trends in the discovery of new marine natural products from invertebrates over the last two decades—Where and what are we bioprospecting? PLoS ONE 2012, 7, e30580. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef] [PubMed]
- Lannig, G.; Eilers, S.; Pörtner, H.O.; Sokolova, I.M.; Bock, C. Impact of ocean acidification on energy metabolism of oyster, Crassostrea gigas—Changes in metabolic pathways and thermal response. Mar. Drugs 2010, 8, 2318–2339. [Google Scholar] [CrossRef] [PubMed]
- Barde, S.R.; Sakhare, R.S.; Kanthale, S.B.; Chandak, P.G.; Jamkhande, P.G. Marine bioactive agents: A short review on new marine antidiabetic compounds. Asian Pac. J. Trop. Dis. 2015, 5, 209–213. [Google Scholar] [CrossRef]
- Sharifuddin, Y.; Chin, Y.-X.; Lim, P.-E.; Phang, S.-M. Potential bioactive compounds from seaweed for diabetes management. Mar. Drugs 2015, 13, 5447–5491. [Google Scholar] [CrossRef] [PubMed]
- Ceriello, A. Postprandial hyperglycemia and diabetes complications is it time to treat? Diabetes 2005, 54, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lebovitz, H.E. Insulin: Potential Negative Consequences of Early Routine Use in Patients with Type 2 Diabetes. Diabetes Care 2011, 34, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Dicker, D. DPP-4 Inhibitors. Impact on glycemic control and cardiovascular risk factors. Diabetes Care 2011, 34, 276–278. [Google Scholar] [CrossRef] [PubMed]
- Thota, S.; Morel, C.M. Recent Advances in Synthetic Chemistry of Diabetic Research. Mini Rev. Med. Chem. 2015, 15, 1159–1163. [Google Scholar] [CrossRef] [PubMed]
- Kendall, D.M. Thiazolidinediones. The case for early use. Diabetes Care 2006, 29, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.P.; Bali, A.; Singh, N.; Jaggi, A.S. Advanced glycation end products and diabetic complications. Korean J. Physiol. Pharmacol. 2014, 18, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-X.; Wijesekara, I.; Li, Y.; Kim, S.-K. Phlorotannins as bioactive agents from brown algae. Process. Biochem. 2011, 46, 2219–2224. [Google Scholar] [CrossRef]
- Lopes, G. Seaweeds from the Portuguese Coast: Chemistry, Antimicrobial and Antiinflammatory Capacity. Ph.D. Thesis, Phytochemistry and Pharmacognosy Speciality, Faculty of Pharmacy, University of Porto, Porto, Portugal, 2014. [Google Scholar]
- Martínez, J.H.I.; Castañeda, H.G.T. Preparation and chromatographic analysis of phlorotannins. J. Chromatogr. Sci. 2013, 51, 825–838. [Google Scholar] [CrossRef] [PubMed]
- He, L. Alpha-glucosidase inhibitors as agents in the treatment of diabetes. Diabetes Rev. 1998, 6, 132–145. [Google Scholar]
- Bayer. Precose Prescribing Information (08753825, R. 6). 2008. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/020482s023lbl.pdf (accessed on 20 September 2016). [Google Scholar]
- Pfizer. Glyset Prescribing Information (LAB-0167-10.0). 2012. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020682s010lbl.pdf (accessed on 20 September 2016). [Google Scholar]
- Chen, X.; Zheng, Y.; Shen, Y. Voglibose (Basen®, AO-128), one of the most important α-glucosidase inhibitors. Curr. Med. Chem. 2006, 13, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Karadeniz, F.; Kim, M.M.; Kim, S.K. α-Glucosidase and α-amylase inhibitory activities of phloroglucinol derivatives from edible marine brown alga, Ecklonia cava. J. Sci. Food Agric. 2009, 89, 1552–1558. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, S.M.; Jung, W.-S.; Song, D.-G.; Um, B.-H.; Son, J.-K.; Pan, C.-H. Phlorofucofuroeckol-A, a potent inhibitor of aldo-keto reductase family 1 member B10, from the edible brown alga Eisenia bicyclis. J. Korean Soc. Appl. Biol. Chem. 2012, 55, 721–727. [Google Scholar] [CrossRef]
- Park, M.-H.; Heo, S.-J.; Park, P.-J.; Moon, S.-H.; Sung, S.-H.; Jeon, B.-T.; Lee, S.-H. 6,6′-Bieckol Isolated from Ecklonia cava Protects Oxidative Stress Through Inhibiting Expression of ROS and Proinflammatory Enzymes in High-Glucose-Induced Human Umbilical Vein Endothelial Cells. Appl. Biochem. Biotechnol. 2014, 174, 632–643. [Google Scholar] [CrossRef] [PubMed]
- Kawamura-Konishi, Y.; Watanabe, N.; Saito, M.; Nakajima, N.; Sakaki, T.; Katayama, T.; Enomoto, T. Isolation of a new phlorotannin, a potent inhibitor of carbohydrate-hydrolyzing enzymes, from the brown alga Sargassum patens. J. Agric. Food Chem. 2012, 60, 5565–5570. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Ishimaru, A.; Suzuki, R.; Okuyama, T. A new phloroglucinol derivative from the brown alga Eisenia bicyclis: Potential for the effective treatment of diabetic complications. J. Nat. Prod. 2004, 67, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.E.; Islam, M.N.; Ahn, B.R.; Chowdhury, S.S.; Sohn, H.S.; Jung, H.A.; Cho, J.S. Protein tyrosine phosphatase 1B and α-glucosidase inhibitory phlorotannins from edible brown algae, Ecklonia stolonifera and Eisenia bicyclis. Biosci. Biotechnol. Biochem. 2011, 75, 1472–1480. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Park, M.-H.; Heo, S.-J.; Kang, S.-M.; Ko, S.-C.; Han, J.-S.; Jeon, Y.J. Dieckol isolated from Ecklonia cava inhibits α-glucosidase and α-amylase in vitro and alleviates postprandial hyperglycemia in streptozotocin-induced diabetic mice. Food Chem. Toxicol. 2010, 48, 2633–2637. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-C.; Wijesinghe, W.; Lee, S.-H.; Kang, S.-M.; Ko, S.-C.; Yang, X.; Kang, N.; Jeon, B.-T.; Kim, J.; Lee, D.-H.; et al. Dieckol isolated from brown seaweed Ecklonia cava attenuates type II diabetes in db/db mouse model. Food Chem. Toxicol. 2013, 53, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Yoon, N.Y.; Woo, M.-H.; Choi, J.S. Inhibitory activities of extracts from several kinds of seaweeds and phlorotannins from the brown alga Ecklonia stolonifera on glucose-mediated protein damage and rat lens aldose reductase. Fish. Sci. 2008, 74, 1363–1365. [Google Scholar] [CrossRef]
- Lee, S.-H.; Han, J.-S.; Heo, S.-J.; Hwang, J.-Y.; Jeon, Y.-J. Protective effects of dieckol isolated from Ecklonia cava against high glucose-induced oxidative stress in human umbilical vein endothelial cells. Toxicol. In Vitro 2010, 24, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Park, M.-H.; Kang, S.-M.; Ko, S.-C.; Kang, M.-C.; Cho, S.; Park, P.-J.; Jeon, B.-T.; Kim, S.-K.; Han, J.-S.; et al. Dieckol isolated from Ecklonia cava protects against high-glucose induced damage to rat insulinoma cells by reducing oxidative stress and apoptosis. Biosci. Biotechnol. Biochem. 2012, 76, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Eom, S.H.; Lee, S.H.; Yoon, N.Y.; Jung, W.K.; Jeon, Y.J.; Kim, S.K.; Lee, M.-S.; Kim, Y.-M. α-Glucosidase- and α-amylase-inhibitory activities of phlorotannins from Eisenia bicyclis. J. Sci. Food Agric. 2012, 92, 2084–2090. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.-J.; Hwang, J.-Y.; Choi, J.-I.; Han, J.-S.; Kim, H.-J.; Jeon, Y.-J. Diphlorethohydroxycarmalol isolated from Ishige okamurae, a brown algae, a potent α-glucosidase and α-amylase inhibitor, alleviates postprandial hyperglycemia in diabetic mice. Eur. J. Pharmacol. 2009, 615, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Choi, J.-I.; Heo, S.-J.; Park, M.-H.; Park, P.-J.; Jeon, B.-T.; Kim, S.-K.; Han, J.-S.; Jeon, Y.-J. Diphlorethohydroxycarmalol isolated from Pae (Ishige okamurae) protects high glucose-induced damage in RINm5F pancreatic β cells via its antioxidant effects. Food Sci. Biotechnol. 2012, 21, 239–246. [Google Scholar] [CrossRef]
- Lee, S.-H.; Kang, S.-M.; Ko, S.-C.; Lee, D.-H.; Jeon, Y.-J. Octaphlorethol A, a novel phenolic compound isolated from a brown alga, Ishige foliacea, increases glucose transporter 4-mediated glucose uptake in skeletal muscle cells. Biochem. Biophys. Res. Commun. 2012, 420, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Kang, S.-M.; Ko, S.-C.; Kang, M.-C.; Jeon, Y.-J. Octaphlorethol A, a novel phenolic compound isolated from Ishige foliacea, protects against streptozotocin-induced pancreatic β cell damage by reducing oxidative stress and apoptosis. Food Chem. Toxicol. 2013, 59, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Kang, S.-M.; Ko, S.-C.; Moon, S.-H.; Jeon, B.-T.; Lee, D.H.; Jeon, Y.-J. Octaphlorethol A: A potent α-glucosidase inhibitor isolated from Ishige foliacea shows an anti-hyperglycemic effect in mice with streptozotocin-induced diabetes. Food Funct. 2014, 5, 2602–2608. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Ko, S.-C.; Kang, M.-C.; Lee, D.H.; Jeon, Y.-J. Octaphlorethol A, a marine algae product, exhibits antidiabetic effects in type 2 diabetic mice by activating AMP-activated protein kinase and upregulating the expression of glucose transporter 4. Food Chem. Toxicol. 2016, 91, 58–64. [Google Scholar] [CrossRef] [PubMed]
- You, H.-N.; Lee, H.-A.; Park, M.-H.; Lee, J.-H.; Han, J.-S. Phlorofucofuroeckol A isolated from Ecklonia cava alleviates postprandial hyperglycemia in diabetic mice. Eur. J. Pharmacol. 2015, 752, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Husni, A.; Wijayanti, R. Inhibitory activity of alpha-amylase and alpha-glucosidase by Padina pavonica extracts. J. Biol. Sci. 2014, 14, 515. [Google Scholar] [CrossRef]
- Liu, H.; Gu, L. Phlorotannins from brown algae (Fucus vesiculosus) inhibited the formation of advanced glycation end products by scavenging reactive carbonyls. J. Agric. Food Chem. 2012, 60, 1326–1334. [Google Scholar] [CrossRef] [PubMed]
- Purich, D.L.; Fromm, H.J. Limitations in the use of dixon plots to evaluate enzyme inhibition. Biochim. Biophys. Acta Enzymol. 1972, 268, 1–3. [Google Scholar] [CrossRef]
- Lee, S.-H.; Jeon, Y.-J. Efficacy and safety of a dieckol-rich extract (AG-dieckol) of brown algae, Ecklonia cava, in pre-diabetic individuals: A double-blind, randomized, placebo-controlled clinical trial. Food Funct. 2015, 6, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Min, K.-H.; Han, J.-S.; Lee, D.-H.; Park, D.-B.; Jung, W.-K.; Park, P.-J.; Jeon, B.-T.; Kim, S.-K.; Jeon, Y.-J. Effects of brown alga, Ecklonia cava on glucose and lipid metabolism in C57BL/KsJ-db/db mice, a model of type 2 diabetes mellitus. Food Chem. Toxicol. 2012, 50, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Firdaus, M.; Prihanto, A.A. α-Amylase and α-glucosidase inhibition by brown seaweed (Sargassum sp.) extracts. Res. J. Life Sci. 2014, 1, 6–11. [Google Scholar]
- Kellogg, J.; Grace, M.H.; Lila, M.A. Phlorotannins from Alaskan seaweed inhibit carbolytic enzyme activity. Mar. Drugs 2014, 12, 5277–5294. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-W.; Han, J.-S. Hypoglycemic effect of Sargassum ringgoldianum extract in STZ-induced diabetic mice. Prev. Nutr. Food Sci. 2012, 17, 8. [Google Scholar] [CrossRef] [PubMed]
- Shakambari, G.; Ashokkumar, B.; Varalakshmi, P. Phlorotannins from brown algae: Inhibition of advanced glycation end products formation in high glucose induced Caenorhabditis elegans. Indian J. Exp. Biol. 2015, 53, 371–379. [Google Scholar] [PubMed]
- Pandithurai, M.; Murugesan, S.; Bhuvaneswari, S.; Thennarasan, S. In vitro α-amylase and α-glucosidase inhibition activity of methanolic extract of marine brown alga Spatoglossum asperum. Int. J. Adv. Pharm. 2015, 4, 83–87. [Google Scholar]
- Min, K.-H.; Kim, H.-J.; Jeon, Y.-J.; Han, J.-S. Ishige okamurae ameliorates hyperglycemia and insulin resistance in C57BL/KsJ-db/db mice. Diabetes Res. Clin. Pract. 2011, 93, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Pantidos, N.; Boath, A.; Lund, V.; Conner, S.; McDougall, G.J. Phenolic-rich extracts from the edible seaweed, Ascophyllum nodosum, inhibit α-amylase and α-glucosidase: Potential anti-hyperglycemic effects. J. Funct. Foods 2014, 10, 201–209. [Google Scholar] [CrossRef]
- Lordan, S.; Smyth, T.J.; Soler-Vila, A.; Stanton, C.; Ross, R.P. The α-amylase and α-glucosidase inhibitory effects of Irish seaweed extracts. Food Chem. 2013, 141, 2170–2176. [Google Scholar] [CrossRef] [PubMed]
- InnoVactive. InSea2. 2016. Available online: http://insea2.com/ (accessed on 23 September 2016).
- Roy, M.-C.; Anguenot, R.; Fillion, C.; Beaulieu, M.; Bérubé, J.; Richard, D. Effect of a commercially-available algal phlorotannins extract on digestive enzymes and carbohydrate absorption in vivo. Food Res. Int. 2011, 44, 3026–3029. [Google Scholar] [CrossRef]
- Gerich, J. Pathogenesis and management of postprandial hyperglycemia: Role of incretin-based therapies. Int. J. Gen. Med. 2013, 6, 877. [Google Scholar] [CrossRef] [PubMed]
- Hanefeld, M.; Koehler, C.; Schaper, F.; Fuecker, K.; Henkel, E.; Temelkova-Kurktschiev, T. Postprandial plasma glucose is an independent risk factor for increased carotid intima-media thickness in non-diabetic individuals. Atherosclerosis 1999, 144, 229–235. [Google Scholar] [CrossRef]
- Kang, M.-C.; Ahn, G.; Yang, X.; Kim, K.-N.; Kang, S.-M.; Lee, S.-H.; Ko, S.-C.; Ko, J.-Y.; Kim, D.; Kim, Y.-T.; et al. Hepatoprotective effects of dieckol-rich phlorotannins from Ecklonia cava, a brown seaweed, against ethanol induced liver damage in BALB/c mice. Food Chem. Toxicol. 2012, 50, 1986–1991. [Google Scholar] [CrossRef] [PubMed]
- Matschinsky, F.M. Glucokinase as glucose sensor and metabolic signal generator in pancreatic β-cells and hepatocytes. Diabetes 1990, 39, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Barzilai, N.; Rossetti, L. Role of glucokinase and glucose-6-phosphatase in the acute and chronic regulation of hepatic glucose fluxes by insulin. J. Biol. Chem. 1993, 268, 25019–25025. [Google Scholar] [PubMed]
- Barthel, A.; Schmoll, D. Novel concepts in insulin regulation of hepatic gluconeogenesis. Am. J. Physiol. Endocrinol. Metab. 2003, 285, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Domingueti, C.P.; Dusse, L.M.S.A.; das Graças Carvalho, M.; de Sousa, L.P.; Gomes, K.B.; Fernandes, A.P. Diabetes mellitus: The linkage between oxidative stress, inflammation, hypercoagulability and vascular complications. J. Diabetes Complicat. 2016, 30, 738–745. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Martin, K.A.; Hwa, J. Aldose Reductase, oxidative stress, and Diabetic mellitus. Front. Pharmacol. 2012, 3, 87. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.A.; Borja, N.L. Epalrestat: An aldose reductase inhibitor for the treatment of diabetic neuropathy. Pharmacotherapy 2008, 28, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Giannoukakis, N. Drug evaluation: Ranirestat—Analdose reductase inhibitor for the potential treatment of diabetic complications. Curr. Opin. Investig. Drugs 2006, 7, 916. [Google Scholar] [PubMed]
- Iwahori, Y.; Enomoto, S.; Okada, Y.; Tanaka, J.; Okuyama, T. Naturally Occurring Substances for Prevention of Complications of Diabetes. IV: Screening of sea vegetables for inhibitory effect on aldose reductase. Nat. Med. 1999, 53, 138–140. [Google Scholar]
- Lopes, G.; Sousa, C.; Silva, L.R.; Pinto, E.; Andrade, P.B.; Bernardo, J.; Valentão, P. Can phlorotannins purified extracts constitute a novel pharmacological alternative for microbial infections with associated inflammatory conditions? PLoS ONE 2012, 7, e31145. [Google Scholar] [CrossRef] [PubMed]
- Panzhinskiy, E.; Ren, J.; Nair, S. Protein tyrosine phosphatase 1B and insulin resistance: Role of endoplasmic reticulum stress/reactive oxygen species/nuclear factor kappa B axis. PLoS ONE 2013, 8, e77228. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, B.J. Protein-tyrosine phosphatase 1B (PTP1B): A novel therapeutic target for type 2 diabetes mellitus, obesity and related states of insulin resistance. Curr. Drug Targets Immune Endocr. Metabol. Disord. 2001, 1, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Richter, E.A.; Hargreaves, M. Exercise, GLUT4, and skeletal muscle glucose uptake. Physiol. Rev. 2013, 93, 993–1017. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.F.; Shulman, G.I. Pathogenesis of skeletal muscle insulin resistance in type 2 diabetes mellitus. Am. J. Cardiol. 2002, 90, 11–18. [Google Scholar] [CrossRef]
- Kim, E.-A.; Lee, S.-H.; Lee, J.-H.; Kang, N.; Oh, J.-Y.; Cha, S.-H.; Ahn, G.; Ko, S.C.; Fernando, S.P.; Kim, S.-Y.; et al. A marine algal polyphenol, dieckol, attenuates blood glucose levels by Akt pathway in alloxan induced hyperglycemiazebrafish model. RSC Adv. 2016, 6, 78570–78575. [Google Scholar] [CrossRef]
- Lizcano, J.M.; Alessi, D.R. The insulin signalling pathway. Curr. Biol. 2002, 12, 236–238. [Google Scholar] [CrossRef]
- Jeon, H.-J.; Yoon, K.-Y.; Koh, E.-J.; Choi, J.; Kim, K.-J.; Choi, H.-S.; Lee, B.-Y. Seapolynol and Dieckol Improve Insulin Sensitivity through the Regulation of the PI3K Pathway in C57BL/KsJ-db/db Mice. J. Food Nutr. Res. 2015, 3, 648–652. [Google Scholar] [CrossRef]
- Kang, C.; Jin, Y.B.; Lee, H.; Cha, M.; Sohn, E.-T.; Moon, J.; Park, C.; Chun, S.; Jung, E.-S.; Hong, J.-S.; et al. Brown alga Ecklonia cava attenuates type 1 diabetes by activating AMPK and Akt signaling pathways. Food Chem. Toxicol. 2010, 48, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Peppa, M.; Uribarri, J.; Vlassara, H. Glucose, advanced glycation end products, and Diabetes complications: What is new and what works. Clin. Diabetes 2003, 21, 186–187. [Google Scholar] [CrossRef]
- Wang, X.; Desai, K.; Clausen, J.T.; Wu, L. Increased methylglyoxal and advanced glycation end products in kidney from spontaneously hypertensive rats. Kidney Int. 2004, 66, 2315–2321. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Uribarri, J. Advanced glycation end products (AGE) and diabetes: Cause, effect, or both? Curr. Diabetes Rep. 2014, 14, 453. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.-J.; Hwang, J.-Y.; Choi, J.-I.; Lee, S.-H.; Park, P.-J.; Kang, D.-H.; Oh, C.; Kim, D.-W.; Han, J.-S.; Jeon, Y.-J.; et al. Protective effect of diphlorethohydroxycarmalol isolated from Ishige okamurae against high glucose-induced-oxidative stress in human umbilical vein endothelial cells. Food Chem. Toxicol. 2010, 48, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Rochette, L.; Zeller, M.; Cottin, Y.; Vergely, C. Diabetes, oxidative stress and therapeutic strategies. Biochim. Biophys. Acta Gen. Subj. 2014, 1840, 2709–2729. [Google Scholar] [CrossRef] [PubMed]
- Matés, J.M.; Pérez-Gómez, C.; de Castro, I.N. Antioxidant enzymes and human diseases. Clin. Biochem. 1999, 32, 595–603. [Google Scholar] [CrossRef]
- Robertson, R.P. Chronic oxidative stress as a central mechanism for glucose toxicity in pancreatic islet beta cells in diabetes. J. Biol. Chem. 2004, 279, 42351–42354. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.P.; Zhang, H.J.; Pyzdrowski, K.L.; Walseth, T.F. Preservation of insulin mRNA levels and insulin secretion in HIT cells by avoidance of chronic exposure to high glucose concentrations. J. Clin. Investig. 1992, 90, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Briaud, I.; Harmon, J.S.; Kelpe, C.L.; Segu, V.B.G.; Poitout, V. Lipotoxicity of the pancreatic β-cell is associated with glucose-dependent esterification of fatty acids into neutral lipids. Diabetes 2001, 50, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Kesavulu, M.M.; Rao, B.K.; Giri, R.; Vijaya, J.; Subramanyam, G.; Apparao, C. Lipid peroxidation and antioxidant enzyme status in Type 2 diabetics with coronary heart disease. Diabetes Res. Clin. Pract. 2001, 53, 33–39. [Google Scholar] [CrossRef]
- Listenberger, L.L.; Ory, D.S.; Schaffer, J.E. Palmitate-induced apoptosis can occur through a ceramide-independent pathway. J. Biol. Chem. 2001, 276, 14890–14895. [Google Scholar] [CrossRef] [PubMed]
- Federici, M.; Hribal, M.; Perego, L.; Ranalli, M.; Caradonna, Z.; Perego, C.; Usellini, L.; Nano, R.; Bonini, P.; Bertuzzi, F.; et al. High glucose causes apoptosis in cultured human pancreatic islets of langerhans. A potential role for regulation of specific Bcl family genes toward an apoptotic cell death program. Diabetes 2001, 50, 1290–1301. [Google Scholar] [CrossRef] [PubMed]
- Park, M.-H.; Heo, S.-J.; Kim, K.-N.; Ahn, G.; Park, P.-J.; Moon, S.-H.; Jeon, B.-T.; Lee, S.-H. 6,6′-Bieckol protects insulinoma cells against high glucose-induced glucotoxicity by reducing oxidative stress and apoptosis. Fitoterapia 2015, 106, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Szkudelski, T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol. Res. 2001, 50, 537–546. [Google Scholar] [PubMed]
| Compound | Source | Activity | Reference |
|---|---|---|---|
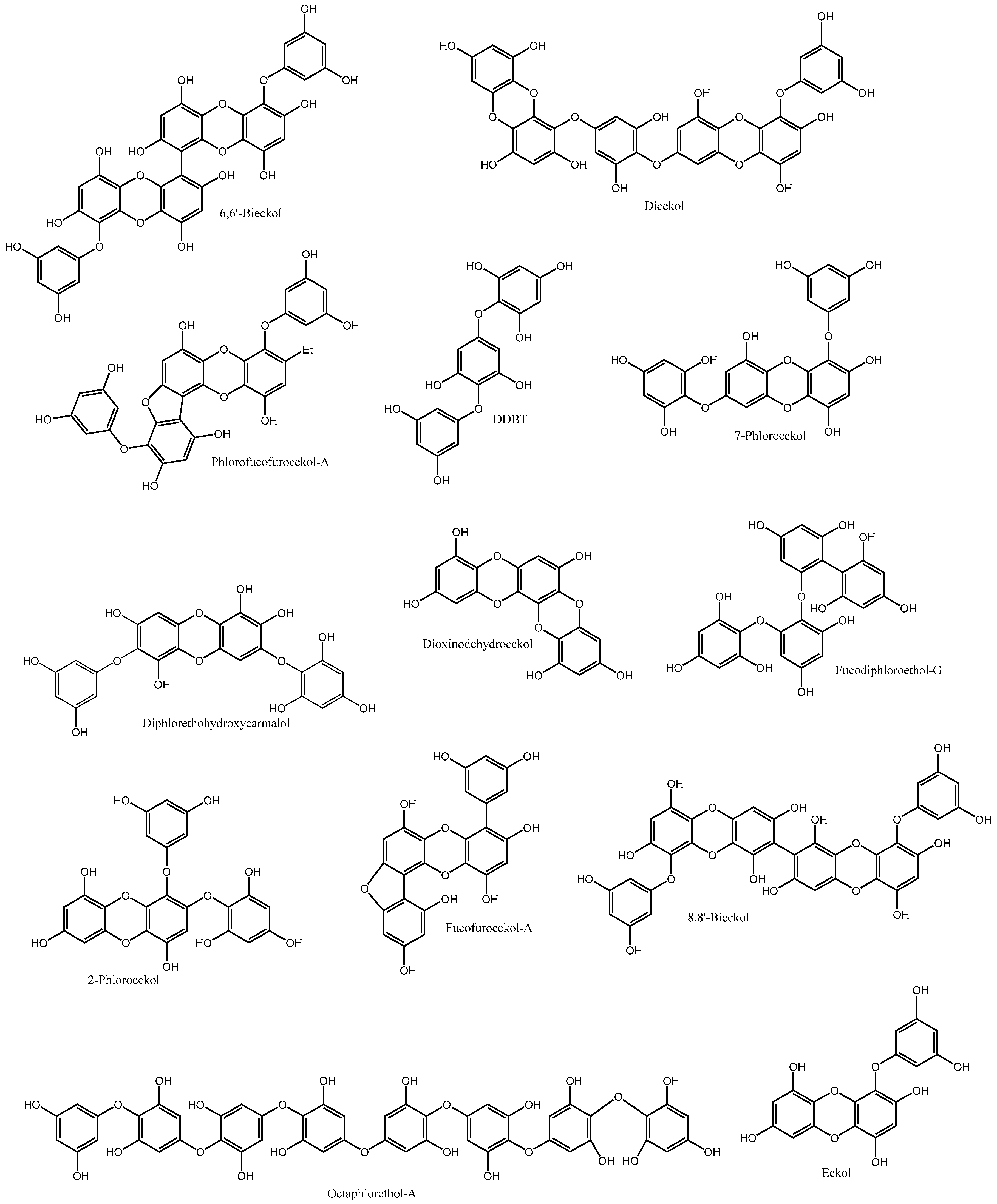
| 6,6′-Bieckol | Ecklonia cava Ecklonia bicyclis | α-Glucosidase inhibition AR inhibition Protection against GIOS ↓Glucose-induced β-cells damage | [29,30,31] |
| 8,8′-Bieckol | Ecklonia bicyclis | AR inhibition | [30] |
| DDBT | Sargassum patens | α-Glucosidase inhibition α-Amylase inhibition | [32] |
| Dieckol | Ecklonia cava Ecklonia stolonifera Eisenia bicyclis | α-Glucosidase inhibition α-Amylase inhibition ↓ Postprandial hyperglycaemia ↓ Serum insulin ↓ G6P AR inhibition PTP1B inhibition ↑ GLUT4 expression Inhibition of AGEs formation Protection against GIOS ↓Glucose-induced β-cells damage ↑ Glucose uptake Akt up-regulation ↓ C-peptide ↑ Adiponectin | [29,30,33,34,35,36,37,38,39] |
| Dioxinodehydroeckol | Ecklonia stolonifera Eisenia bicyclis | α-Glucosidase inhibition α-Amylase inhibition AR inhibition | [34,37,40] |
| Diphlorethohydroxycarmalol | Ishige okamurae | α-Glucosidase inhibition α-Amylase inhibition ↓ Postprandial hyperglycaemia Protection against GIOS ↓Glucose-induced β-cells damage | [41,42] |
| Eckol | Eisenia bicyclis Ecklonia stolonifera Ecklonia cava | α-Amylase inhibition AR inhibition PTP1B inhibition Inhibition of AGEs formation | [33,34,37] |
| Fucodiphloroethol-G | Ecklonia cava | α-Glucosidase inhibition | [29] |
| Fucofuroeckol-A | Eisenia bicyclis | α-Glucosidase inhibition α-Amylase inhibition | [40] |
| Hydroxybenzodioxin | Eisenia bicyclis | α-Amylase inhibition Inhibition of AGEs formation | [33] |
| Octaphlorethol-A | Ishige foliacea | α-Glucosidase inhibition ↓ Serum insulin ↓ G6P expression ↓PEPCK expression ↑Glut4 translocation/expression ↓Glucose-induced β-cells damage ↑ AMPK activation ↑ Glucose uptake | [43,44,45,46] |
| 2-Phloroeckol | Ecklonia stolonifera | AR inhibition | [37] |
| 7-Phloroeckol | Ecklonia cava Ecklonia stolonifera Eisenia bicyclis | α-Glucosidase inhibition α-Amylase inhibition AR inhibition PTP1B inhibition Inhibition of AGEs formation | [29,33,34,37] |
| Phlorofucofuroeckol-A | Ecklonia cava Ecklonia stolonifera Eisenia bicyclis | α-Glucosidase inhibition α-Amylase inhibition ↓ Postprandial hyperglycaemia AR inhibition PTP1B inhibition Inhibition of AGEs formation | [29,30,34,37,47] |
| Phloroglucinol | Ecklonia stolonifera Eisenia bicyclis | α-Glucosidase inhibition α-Amylase inhibition AR inhibition Inhibition of AGEs formation | [34,37,48,49] |
| Extract | Source | Activity | Reference |
|---|---|---|---|
| Acetone | Fucus vesiculosus | ↓ AGEs formation | [49] |
| Dieckol-rich extract | Ecklonia cava | ↓Postprandial hyperglycaemia ↓ Serum insulin ↑Glucokinase activity ↓ G6P activity | [51,52] |
| Ethyl acetate | Sargassum aquifolium | α-Glucosidase inhibition α-Amylase inhibition ↓Postprandial hyperglycaemia | [53] |
| Ethyl acetate | Ecklonia stolonifera | AR inhibition | [37] |
| Fucophloroethol-rich extract | Fucus distichus | α-Glucosidase inhibition α-Amylase inhibition | [54] |
| Methanol | Sargassum ringgoldianum | α-Glucosidase inhibition α-Amylase inhibition ↓Postprandial hyperglycaemia | [55] |
| Methanol | Sargassum polycystum | ↓ AGEs formation | [56] |
| Methanol | Spatoglossum asperum | α-Glucosidase inhibition α-Amylase inhibition | [57] |
| Methanol | Padina pavonica | ↓ AGEs formation | [56] |
| Methanol | Turbinaria ornata | ↓ AGEs formation | [56] |
| Methanol (80%) | Ishige okamurae | ↓ Blood glucose levels ↑ Insulin sensitivity | [58] |
| Water:acetonitrile (50:50 v/v) | Ascophyllum nodosum | α-Glucosidase inhibition α-Amylase inhibition | [59] |
| Water/Ethanol | Ascophyllum nodosum Fucus spiralis | α-Glucosidase inhibition α-Amylase inhibition | [60] |
| Water:methanol (25:75 v/v) | Padina pavonica | α-Glucosidase inhibition α-Amylase inhibition | [48] |
| Water | Ascophyllum nodosum Fucus vesiculosus | α-Glucosidase inhibition α-Amylase inhibition ↓ Postprandial hyperglycaemia ↓ Serum insulin ↓ Insulin secretion ↑ Insulin sensitivity | [61] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopes, G.; Andrade, P.B.; Valentão, P. Phlorotannins: Towards New Pharmacological Interventions for Diabetes Mellitus Type 2. Molecules 2017, 22, 56. https://doi.org/10.3390/molecules22010056
Lopes G, Andrade PB, Valentão P. Phlorotannins: Towards New Pharmacological Interventions for Diabetes Mellitus Type 2. Molecules. 2017; 22(1):56. https://doi.org/10.3390/molecules22010056
Chicago/Turabian StyleLopes, Graciliana, Paula B. Andrade, and Patrícia Valentão. 2017. "Phlorotannins: Towards New Pharmacological Interventions for Diabetes Mellitus Type 2" Molecules 22, no. 1: 56. https://doi.org/10.3390/molecules22010056
APA StyleLopes, G., Andrade, P. B., & Valentão, P. (2017). Phlorotannins: Towards New Pharmacological Interventions for Diabetes Mellitus Type 2. Molecules, 22(1), 56. https://doi.org/10.3390/molecules22010056








