Effects of Rhodomyrtus tomentosa Extract on Killing Activity of Human Neutrophils and Membrane Integrity of Enterohaemorrhagic Escherichia coli O157:H7
Abstract
:1. Introduction
2. Results and Discussion
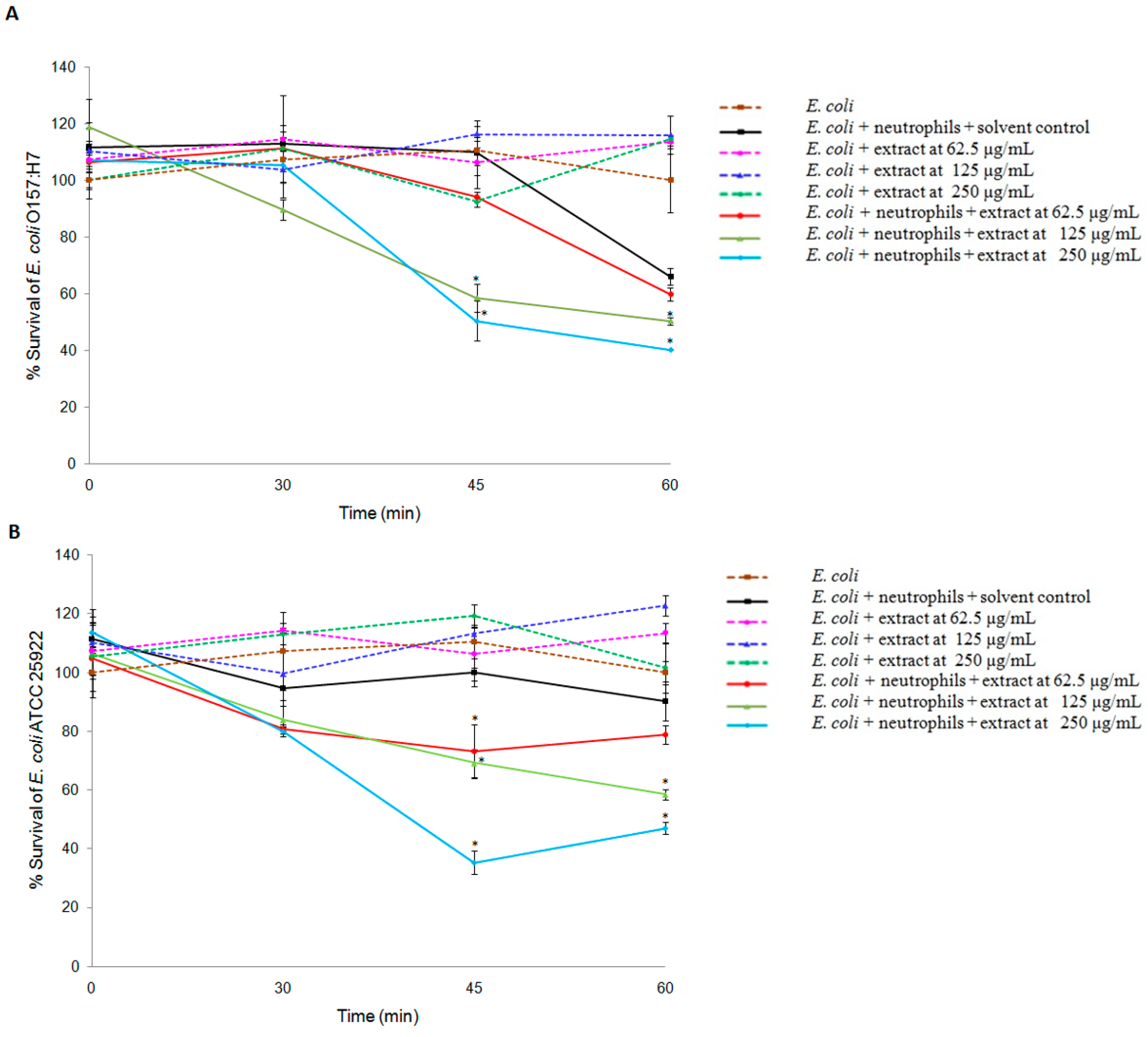
2.1. Effects of R. tomentosa Extract on Killing Activity of Neutrophils
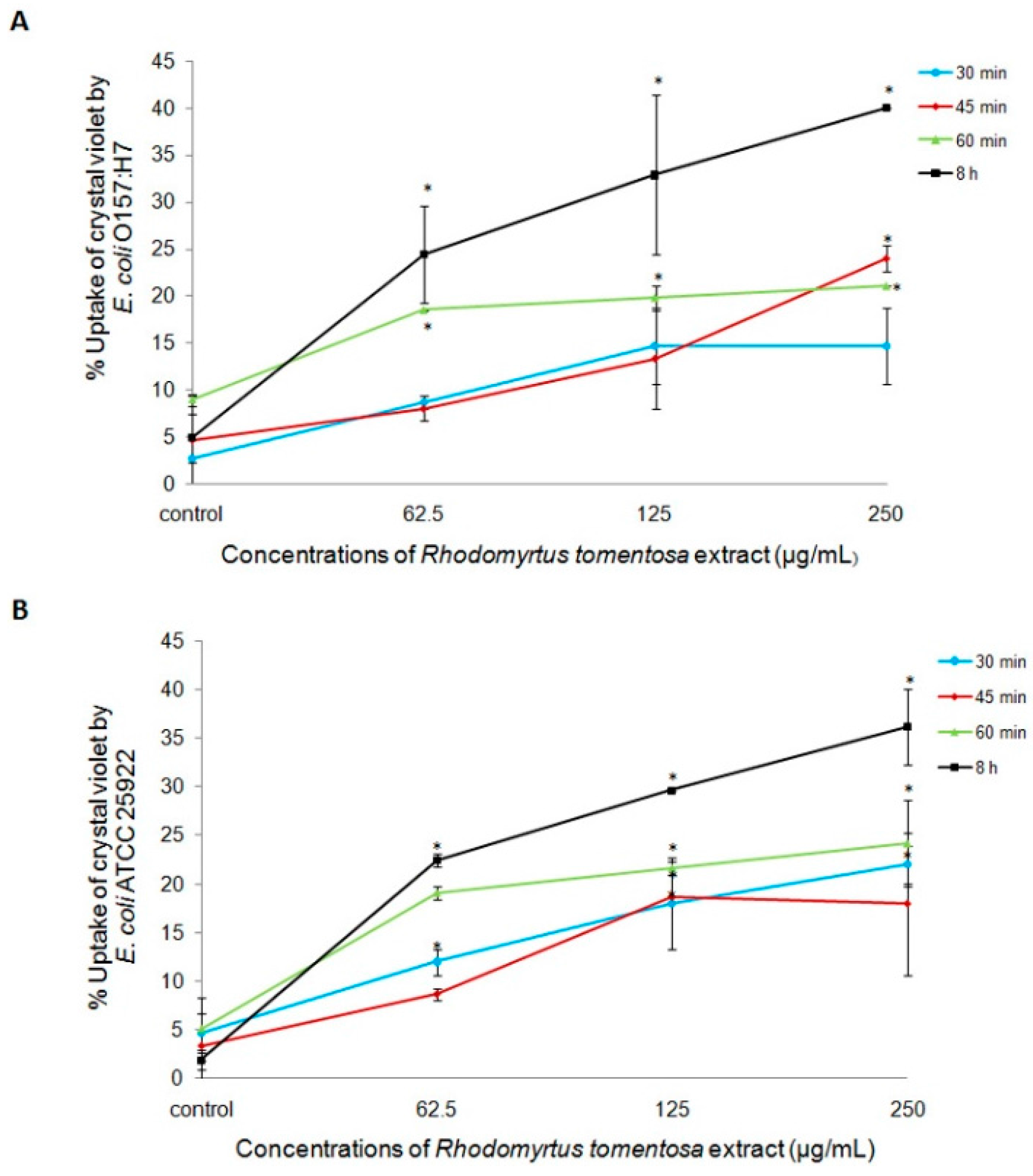
2.2. Effects of R. tomentosa Extract on Membrane Integrity of E. coli
3. Experimental Section
3.1. Pathogens Used and Culture Conditions
3.2. Preparation of R. tomentosa Leaf Extract
3.3. Serum Preparation and Opsonization
3.4. Isolation of Human Neutrophils
3.5. Cell Survival Assay
3.6. Membrane Permeability Assay
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Griffin, P.M.; Tauxe, R.V. The epidemiology of infections caused by Escherichia coli O157:H7, other enterohemorrhagic E. coli, and the associated hemolytic uremic syndrome. Epidemiol. Rev. 1991, 13, 60–98. [Google Scholar] [PubMed]
- Nguyen, Y.; Sperandio, V. Enterohemorrhagic E. coli (EHEC) pathogenesis. Front. Cell Infect. Microbiol. 2012, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.E.; Wilker, P.R.; Reiter, P.L.; Hedican, E.B.; Bender, J.B.; Hedberg, C.W. Antibiotic treatment of Escherichia coli O157 infection and the risk of hemolytic uremic syndrome, minnesota. Pediatr. Infect. Dis. J. 2012, 31, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Yoon, J.; Hovde, C.J.; Hovde, C.J. A brief overview of Escherichia coli O157:H7 and its plasmid O157. J. Microbiol. Biotechnol. 2010, 20, 5–14. [Google Scholar] [PubMed]
- Voravuthikunchai, S.; Lortheeranuwat, A.; Jeeju, W.; Sririrak, T.; Phongpaichit, S.; Supawita, T. Effective medicinal plants against enterohaemorrhagic Escherichia coli O157:H7. J. Ethnopharmacol. 2004, 94, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Baskaran, S.A.; Venkitanarayanan, K. Plant-derived antimicrobials reduce E. coli O157:H7 virulence factors critical for colonization in cattle gastrointestinal tract in vitro. Biomed. Res. Int. 2014, 2014, 212395. [Google Scholar] [CrossRef]
- Razali, F.N.; Ismail, A.; Abidin, N.Z.; Shuib, A.S. Stimulatory effects of polysaccharide fraction from Solanum nigrum on RAW 264.7 murine macrophage cells. PLoS ONE 2014, 9, e108988. [Google Scholar] [CrossRef] [PubMed]
- Rafiul Haque, M.; Ansari, S.H.; Rashikh, A. Coffea arabica seed extract stimulate the cellular immune function and cyclophosphamide-induced immunosuppression in mice. Iran. J. Pharm. Res. 2013, 12, 101–108. [Google Scholar] [PubMed]
- Chitemerere, T.A.; Mukanganyama, S. Evaluation of cell membrane integrity as a potential antimicrobial target for plant products. BMC Complement. Altern. Med. 2014, 14, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ghule, B.V.; Yeole, P.G. In vitro and in vivo immunomodulatory activities of iridoids fraction from Barleria prionitis Linn. J. Ethnopharmacol. 2012, 141, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Wei, F. Manufacture of Traditional Chinese Medicine Composition for Treating Urinary Tract Infection (Gungxi Huahong Pharmaceutical Co., Ltd., People’s Republic of China; Shanghai Fosun Pharmaceutical (Group) Co., Ltd.), Faming Zhuanli Shenqing Gongkai Shuomingshu. China Patent CN1853687, 1 November 2006. [Google Scholar]
- Ong, H.C.; Nordiana, M. Malay ethno-medico botany in Machang, Kelantan, Malaysia. Fitoterapia 1999, 70, 502–513. [Google Scholar] [CrossRef]
- Wei, F. Manufacture of Oral Liquid Containing Traditional Chinese Medicine Extract for Treating Gynecopathy (Guangxi Huahong Pharmaceutical Co., Ltd., People’s Republic of China; Shanghai Fosun Pharmaceutical (Group) Co., Ltd.), Faming Zhuanli Shenqing Gongkai Shuomingshu. China Patent CN1846715, 18 October 2006. [Google Scholar]
- Lavanya, G.; Voravuthikunchai, S.P.; Towatana, N.H. Acetone extract from Rhodomyrtus tomentosa: A potent natural antioxidant. Evid. Based Complement. Altern. Med. 2012, 2012, 535479. [Google Scholar] [CrossRef] [PubMed]
- Geetha, K.M.; Sridhar, C.; Murugan, V. Antioxidant and gastroprotective activities of R. tomentosa (Ait.) Hassk. Int. J. PharmTech. Res. 2010, 2, 283–291. [Google Scholar]
- Limsuwan, S.; Trip, E.N.; Kouwen, T.R.; Piersma, S.; Hiranrat, A.; Mahabusarakam, W.; Voravuthikunchai, S.P.; van Dijl, J.M.; Kayser, O. Rhodomyrtone: A new candidate as natural antibacterial drug from Rhodomyrtus tomentosa. Phytomedicine 2009, 16, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Voravuthikunchai, S.P.; Dolah, S.; Charernjiratrakul, W. Control of Bacillus cereus in foods by R. tomentosa (Ait.) Hassk. leaf extract and its purified compound. J. Food Prot. 2010, 73, 1907–1912. [Google Scholar] [PubMed]
- Saising, J.; Ongsakul, M.; Voravuthikunchai, S.P. R. tomentosa (Aiton) Hassk. ethanol extract and rhodomyrtone: A potential strategy for the treatment of biofilm-forming staphylococci. J. Med. Microbiol. 2011, 60, 1793–1800. [Google Scholar] [CrossRef] [PubMed]
- Czuprynski, C.J.; Henson, P.M.; Campbell, P.A. Effect of dimethyl sulfoxide on the in vitro and in vivo bactericidal activity of human and mouse neutrophils and mononuclear phagocytes. Inflammation 1984, 8, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Serafino, A.; Vallebona, P.S.; Andreola, F.; Zonfrillo, M.; Mercuri, L.; Federici, M.; Rasi, G.; Garaci, E.; Pierimarchi, P. Stimulatory effect of Eucalyptus essential oil on innate cell-mediated immune response. BMC Immunol. 2008, 9, 17. [Google Scholar] [CrossRef] [PubMed]
- Yadav, M.K.; Chae, S.W.; Im, G.J.; Chung, J.W.; Song, J.J. Eugenol: A phyto-compound effective against methicillin-resistant and methicillin-sensitive Staphylococcus aureus clinical strain biofilms. PLoS ONE 2015, 10, e0119564. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Luo, M.; Fu, Y.J.; Zu, Y.G.; Wang, W.; Zhang, L.; Yao, L.P.; Zhao, C.J.; Sun, Y. Effect of corilagin on membrane permeability of Escherichia coli, Staphylococcus aureus and Candida albicans. Phytother. Res. 2013, 27, 1517–1523. [Google Scholar] [PubMed]
- Srisuwan, S.; Tongtawe, P.; Srimanote, P.; Voravuthikunchai, S.P. Rhodomyrtone modulates innate immune responses of THP-1 monocytes to assist in clearing methicillin-resistant Staphylococcus aureus. PLoS ONE 2014, 9, e110321. [Google Scholar] [CrossRef] [PubMed]
- Strober, W. Trypan blue exclusion test of cell viability. In Current Protocols in Immunology; John Wiley & Sons, Inc.: New York, NY, USA, 2001; pp. A.3B.1–A.3B.2. [Google Scholar]
- Sample Availability: Samples of R. tomentosa extract are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hmoteh, J.; Syed Musthafa, K.; Pomwised, R.; Voravuthikunchai, S.P. Effects of Rhodomyrtus tomentosa Extract on Killing Activity of Human Neutrophils and Membrane Integrity of Enterohaemorrhagic Escherichia coli O157:H7. Molecules 2016, 21, 692. https://doi.org/10.3390/molecules21060692
Hmoteh J, Syed Musthafa K, Pomwised R, Voravuthikunchai SP. Effects of Rhodomyrtus tomentosa Extract on Killing Activity of Human Neutrophils and Membrane Integrity of Enterohaemorrhagic Escherichia coli O157:H7. Molecules. 2016; 21(6):692. https://doi.org/10.3390/molecules21060692
Chicago/Turabian StyleHmoteh, Jutharat, Khadar Syed Musthafa, Rattanaruji Pomwised, and Supayang Piyawan Voravuthikunchai. 2016. "Effects of Rhodomyrtus tomentosa Extract on Killing Activity of Human Neutrophils and Membrane Integrity of Enterohaemorrhagic Escherichia coli O157:H7" Molecules 21, no. 6: 692. https://doi.org/10.3390/molecules21060692
APA StyleHmoteh, J., Syed Musthafa, K., Pomwised, R., & Voravuthikunchai, S. P. (2016). Effects of Rhodomyrtus tomentosa Extract on Killing Activity of Human Neutrophils and Membrane Integrity of Enterohaemorrhagic Escherichia coli O157:H7. Molecules, 21(6), 692. https://doi.org/10.3390/molecules21060692