Synthesis and Characterization of Novel Cu(II), Pd(II) and Pt(II) Complexes with 8-Ethyl-2-hydroxytricyclo(7.3.1.02,7)tridecan-13-one-thiosemicarbazone: Antimicrobial and in Vitro Antiproliferative Activity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.1.1. 1H-NMR and 13C-NMR Spectra
2.1.2. Infrared Spectra and Coordination Mode
2.1.3. Electronic Spectra and Magnetic Studies
2.1.4. Mass Spectra
2.1.5. Thermal Decomposition
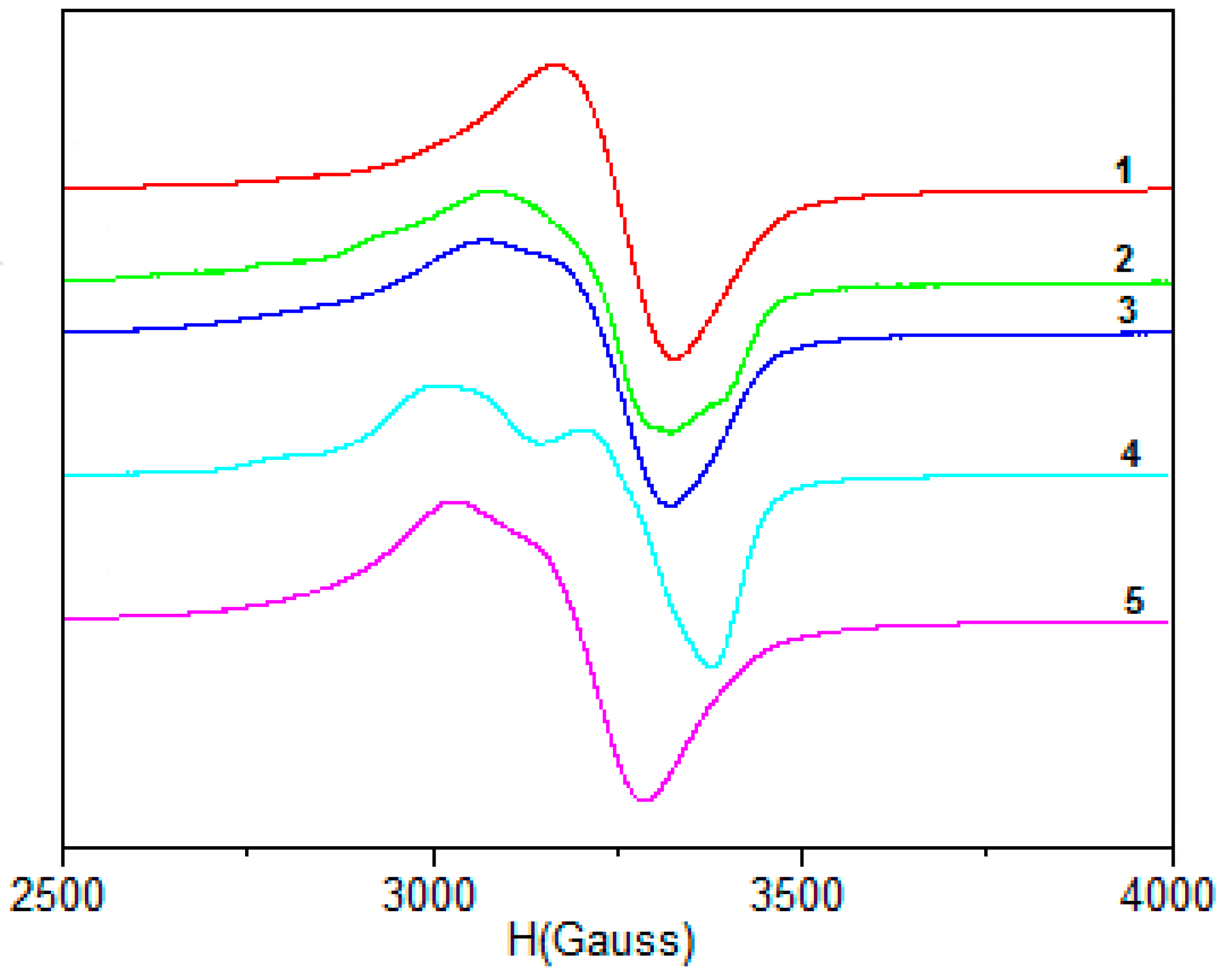
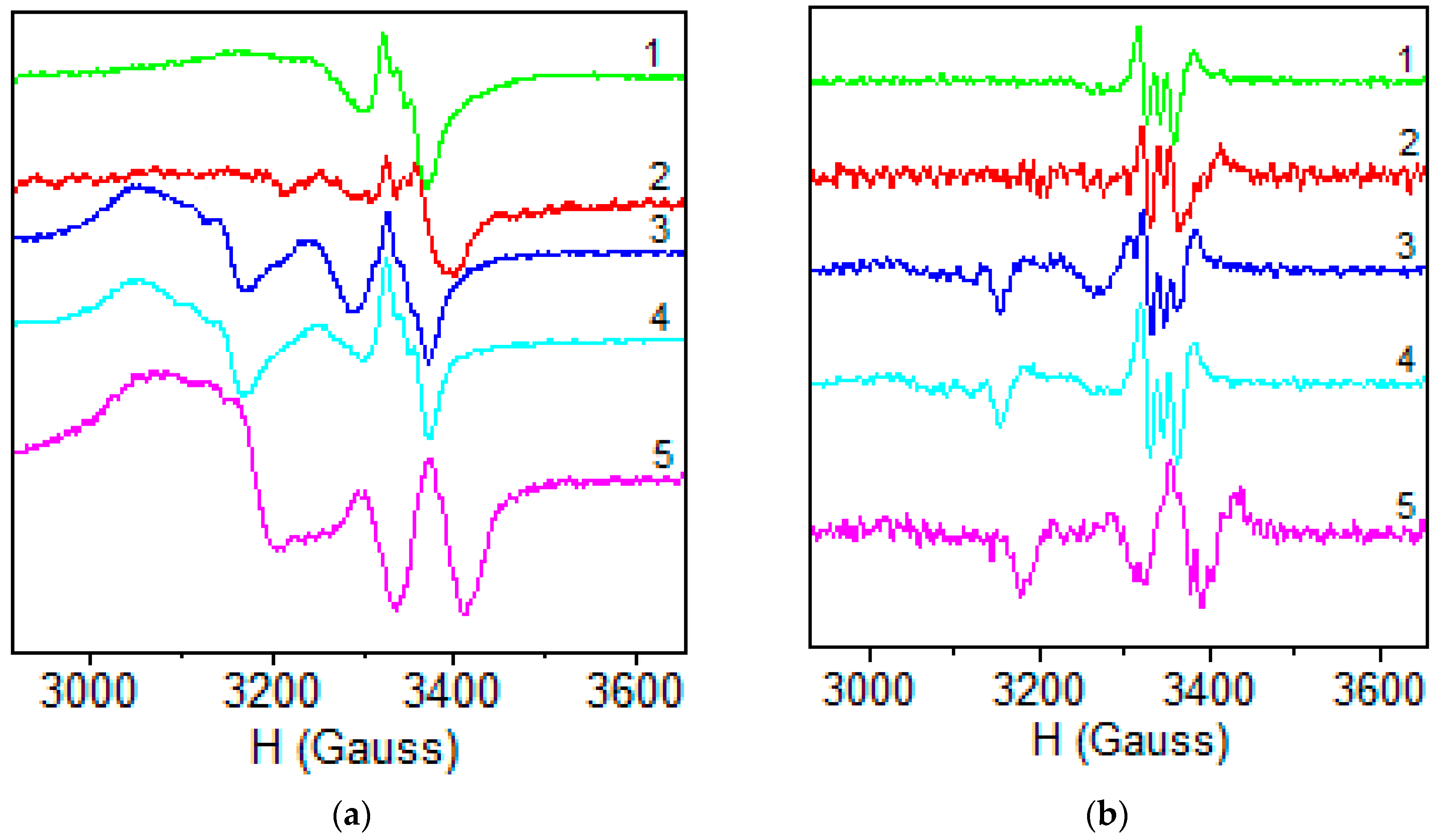
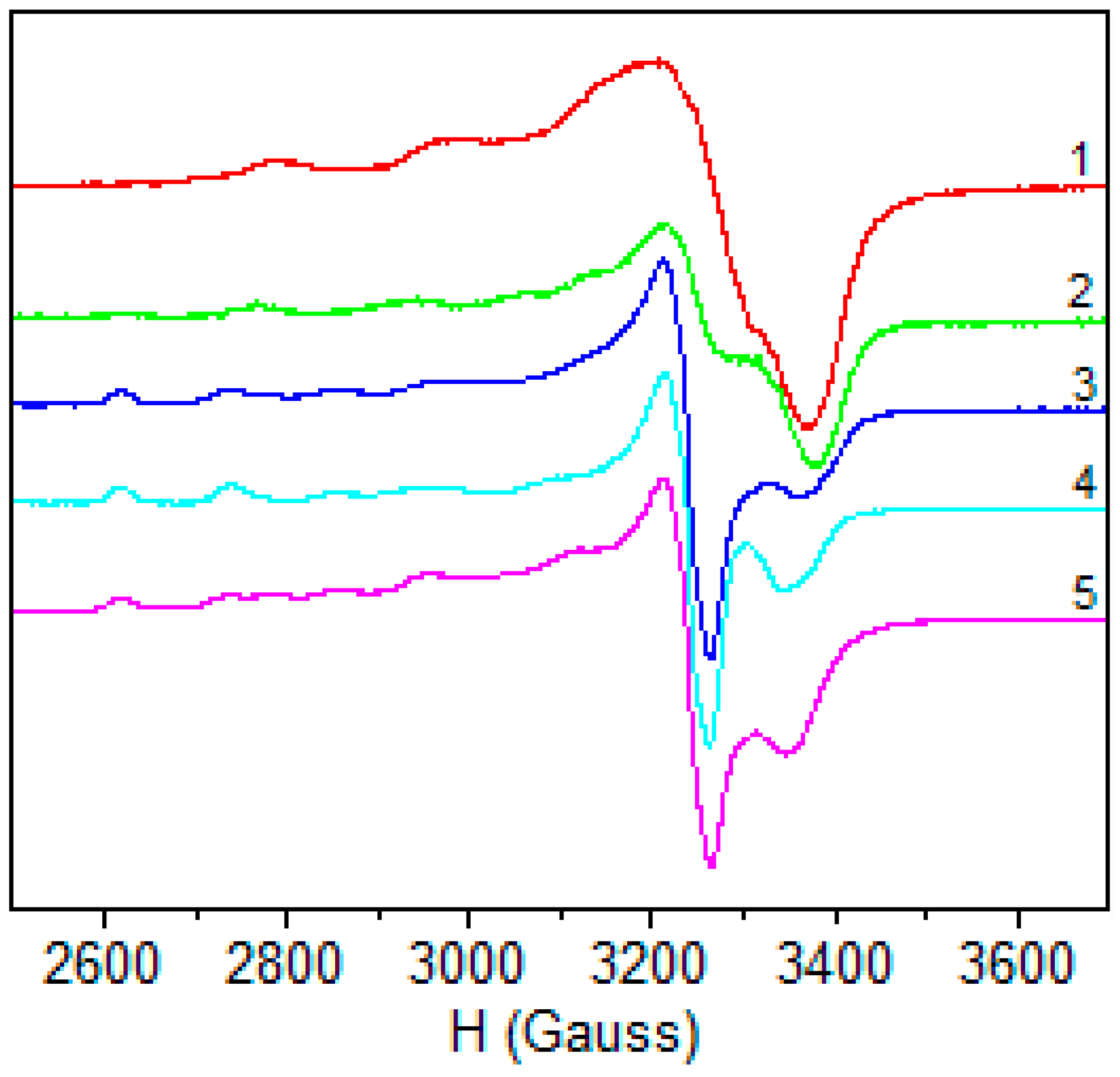
2.1.6. EPR Spectra
2.2. Antibacterial and Antifungal Activity
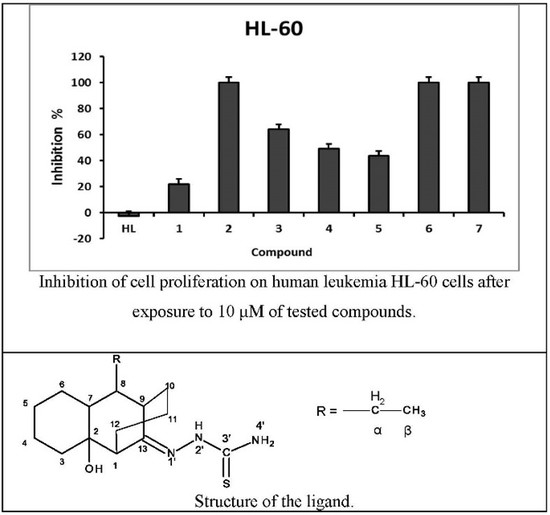
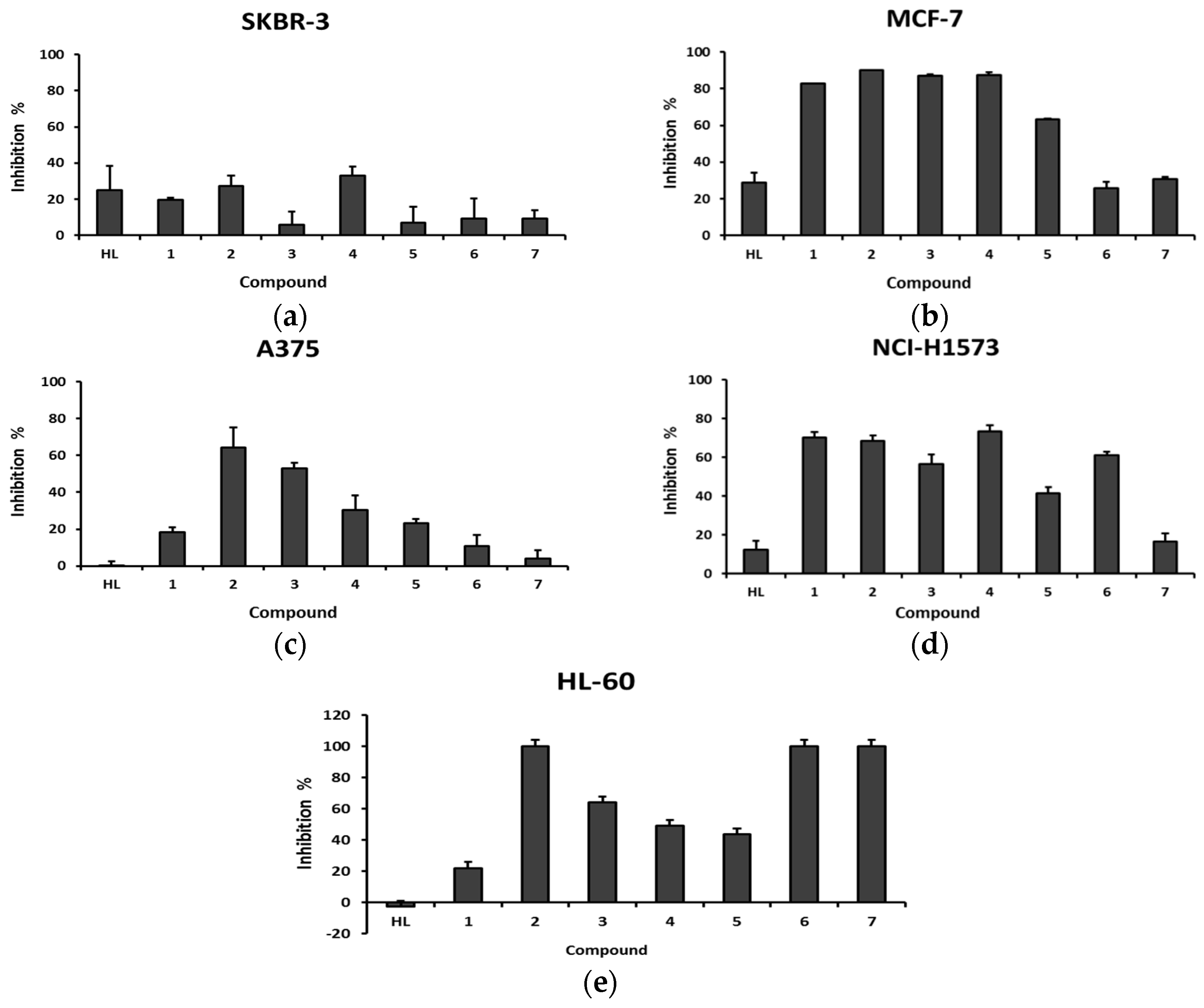
2.3. Antiproliferative Activity
3. Experimental Section
3.1. General Information
3.2. Synthesis
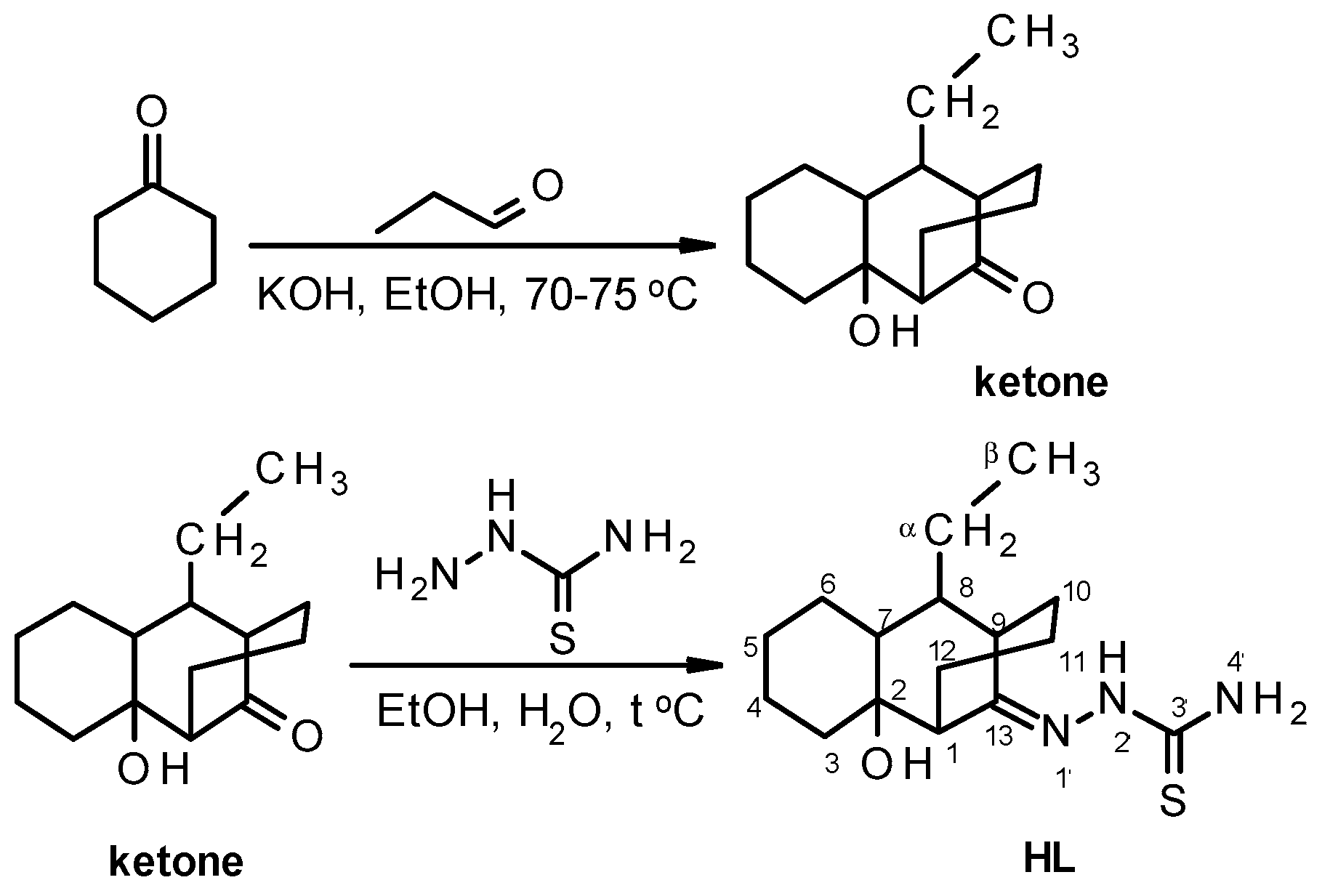
3.2.1. Synthesis of 8-Ethyl-2-hydroxytricyclo[7.3.1.02,7]tridecan-13-one
3.2.2. Synthesis of the 8-Ethyl-2-hydroxytricyclo[7.3.1.02,7]tridecane-13-one-thiosemicarbazone (HL)
3.2.3. General Procedure for the Preparation of the Metal Complexes
3.3. Antibacterial Activity
3.4. Antifungal Bioassay
3.5. Antiproliferative Activity
3.5.1. Preparation of Tested Substances Solutions
3.5.2. Cell Culture
3.5.3. Cell Proliferation Assays
Alamar Blue in Vitro Analysis
MTS Cell Proliferation Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Padhye, S.; Kauffman, G.B. Transition metal complexes of semicarbazones and thiosemicarbazones. Coord. Chem. Rev. 1985, 63, 127–160. [Google Scholar] [CrossRef]
- West, D.X.; Liberta, A.E.; Padhye, S.; Chilkate, R.C.; Sonawane, P.B.; Kumbhar, A.S.; Yerande, R.G. Thiosemicarbazone complexes of copper(II): Structural and biological studies. Coord. Chem. Rev. 1993, 123, 49–71. [Google Scholar] [CrossRef]
- Berkessel, A.; Hermann, G.; Rauch, O.-T.; Büchner, M.; Jacobi, A.; Huttner, G. Preparation and X-ray Crystal Structure of the First Trimeric Nickel Thiosemicarbazone Complex: The First Example of Oligomerization by Both Ni-O-Ni and Ni-S-Ni Bridging. Chem. Ber. 1996, 129, 1421–1423. [Google Scholar] [CrossRef]
- Casas, J.S.; Garcia-Tasende, M.S.; Sordo, J. Corrigendum to Main group metal Complexes of semicarbazones and thiosemicarbazones. A structural review. Coord. Chem. Rev. 2000, 209, 197–261. [Google Scholar] [CrossRef]
- Lobana, T.S.; Kumari, P.; Butcher, R.J.; Akitsu, T.; Aritake, Y.; Perles, J.; Fernandez, F.J.; Vega, M.C. Thiosemicarbazonates of palladium(II): The presence of methyl/phenyl substituents (R2) at C2 carbon atom induces C–H activation of R1 rings of thiosemicarbazones {R1R2C2=N3–N2H–C1(=S)–N1HR3}. J. Organometal. Chem. 2012, 701, 17–26. [Google Scholar] [CrossRef]
- Chunfang, G.; Jianguo, C.; Shaoyang, S.; Qifu, L.; Linyi, J.; Lianghua, F.; Yanmin, H. Synthesis and antiproliferative activity of some steroidal thiosemicarbazones, semicarbazones and hydrozones. Steroids 2014, 87, 99–107. [Google Scholar]
- Liberta, A.E.; West, D.X. Antifungal and antitumor activity of Heterocyclic thiosemicarbazones and their metal complexes: current status. Biometals 1992, 5, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.-C.; Lin, T.-S.; Sartorelli, A.C. Chemical and biological properties of cytotoxic alpha-(N)-heterocyclic carboxaldehyde thiosemicarbazones. Prog. Med. Chem. 1995, 32, 1–35. [Google Scholar] [PubMed]
- Rajasekaran, A.; Murugesan, S. Synthesis and antimicrobial evaluation of Thiosemicarbazones. J. Indian Chem. Soc. 2002, 79, 544–545. [Google Scholar] [CrossRef]
- Rodriguez-Arguelles, M.C.; Lopez-Silva, E.C.; Sanmartin, J.; Pelagatti, P.; Zani, F. Copper complexes of imidazole-2-, pyrrole-2- and indol-3 carbaldehyde thiosemicarbazones: Inhibitory activity against fungi and bacteria. J. Inorg. Biochem. 2005, 99, 2231–2239. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chen, Q.; Ku, X.; Meng, L.; Lin, L.; Wang, X.; Zhu, C.; Wang, Y.; Chen, Z.; Li, M.; Jiang, H.; Chen, K.; Ding, J.; Liu, H. A series of α-heterocyclic carboxaldehyde thiosemicarbazones inhibit topoisomerase IIα catalytic activity. J. Med. Chem. 2010, 53, 3048–3064. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, M.B.; Fava, G.G.; Tarasconi, P.; Albertini, R.; Pinelli, S.; Satarcich, R. Synthesis, spectroscopic and structural characterization and biological activity of aquachloro(pyridoxal thiosemicarbazone) copper(II) chloride. J. Inorg. Biochem. 1994, 53, 13–25. [Google Scholar] [CrossRef]
- Kowol, C.R.; Trondl, R.; Heffeter, P.; Arion, V.B.; Jakupec, M.A.; Roller, A.; Galanski, M.; Berger, W.; Keppler, B.K. Impact of metal coordination on cytotoxicity of 3-aminopyridine-2-carboxaldehyde thiosemicarbazone (triapine) and novel into terminal dimethylation. J. Med. Chem. 2009, 52, 5032–5043. [Google Scholar] [CrossRef] [PubMed]
- Easmon, J.; Purstinger, G.; Heinisch, G.; Roth, T.; Fiebig, H.H.; Holzer, W.; Jager, W.; Jenny, M.; Hofmann, J. Synthesis, Cytotoxicity, and Antitumor Activity of Copper(II) and Iron(II) Complexes of 4N-Azabicyclo(3.2.2)nonane Thiosemicarbazones Derived from Acyl Diazines. J. Med. Chem. 2001, 44, 2164–2171. [Google Scholar] [CrossRef] [PubMed]
- Moorthy, N.S.; Cerqueira, N.M.; Ramos, M.J.; Fernandes, P.A. Aryl- and heteroaryl-thiosemicarbazone derivatives and their metal complexes: A pharmacological template. Recent Pat Anticancer Drug Discov. 2013, 8, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Gokhale, N.; Jain, S.; Yadav, M. Design and virtual screening towards synthesis of novel substituted thiosemicarbozones as ribonuleotide reductase (RNR) inhibitors with improved cellular trafficking and anticancer activity. Curr. Top. Med. Chem. 2015, 15, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, H.P.; Hadi, J.S.; Alsalim, T.A.; Ghali, T.S.; Bolandnazar, Z. A novel series of thiosemicarbazone drugs: from synthesis to structure. Spectrochim. Acta. A Mol. Biomol. Spectrosc. 2015, 137, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Yusuf, M. Synthesis, spectral studies and in vitro antibacterial activity of steroidal thiosemicarbazone and their palladium (Pd (II)) complexes. Eur. J. Med. Chem. 2009, 44, 2270–2274. [Google Scholar] [CrossRef] [PubMed]
- Festa, R.A.; Thiele, D.J. Copper: an essential metal in biology. Curr. Biol. 2011, 21, R877–R883. [Google Scholar] [CrossRef] [PubMed]
- Paterson, B.M.; Donnelly, P.S. Copper complexes of bis(thiosemicarbazones): From chemotherapeutics to diagnostic and therapeutic radiopharmaceuticals. Chem. Soc. Rev. 2011, 40, 3005–3018. [Google Scholar] [CrossRef] [PubMed]
- Wong, E.; Giandomenico, C.M. Current status of platinum-based antitumor drugs. Chem. Rev. 1999, 99, 2451–2466. [Google Scholar] [CrossRef] [PubMed]
- Casas, J.C.; Castellano, E.E.; Ellena, J.; García-Tasende, M.S.; Pérez-Parallé, M.L.; Sánches, A.; Sánches-González, A.; Sordo, J.; Touceda, A. New Pd(II) and Pt(II) complexes with N,S-chelated pyrazolonate ligands: Molecular and supramolecular structure and preliminary study of their in vitro antitumoral activity. J. Inorg. Biochem. 2008, 102, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Jansson, P.J.; Sharpe, P.C.; Bernhardt, P.V.; Richardson, D.R. Novel Thiosemicarbazones of the ApT and DpT series and their copper complexes: Identification of pronounced redox activity and characterization of their antitumor activity. J. Med. Chem. 2010, 53, 5759–5769. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, E.; Kalaivani, P.; Prabhakaran, R.; Rath, N.P.; Brinda, S.; Poornima, P.; Vijaya Padma, V.; Natarajan, K. Synthesis, X-ray crystal structure, DNA binding, antioxidant and cytotoxicity studies of Ni(II) and Pd(II) thiosemicarbazone complexes. Metallomics. 2012, 4, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Munira Haidad Ali, S.; Yan, Y.K.; Lee, P.P.F.; Khong, K.Z.X.; Alam Sk, M.; Lim, K.H.; Klejevskaja, B.; Vilar, R. Copper(II) complexes of substituted salicylaldehyde dibenzyl semicarbazones: Synthesis, cytotoxicity and interaction with quadruplex DNA. Dalton Trans. 2014, 43, 1449–1459. [Google Scholar] [CrossRef] [PubMed]
- Santini, C.; Pellei, M.; Gandin, V.; Porchia, M.; Tisato, F.; Marzano, C. Advances in copper complexes as anticancer agents. Chem. Rev. 2014, 114, 815–862. [Google Scholar] [CrossRef] [PubMed]
- Belicchi-Ferrari, M.; Bisceglie, F.; Pelosi, G.; Tarasconi, P. Heterocyclic substituted thiosemicarbazones and their Cu(II) complexes: Synthesis, characterization and studies of substituent effects on coordination and DNA binding. Polyhedron 2008, 27, 1361–1367. [Google Scholar] [CrossRef]
- Roşu, T.; Pahonţu, E.; Păsculescu, S.; Georgescu, R.; Stănică, N.; Curaj, A.; Popescu, A.; Leabu, M. Synthesis and characterization of novel Cu(II) and Pd(II) complexes with 2-hydroxy-8-R-tricyclo(7.3.1.0.2,7)tridecane-13-one thiosemicarbazone. Study on biological activity. Eur. J. Med. Chem. 2010, 45, 1627–1634. [Google Scholar]
- Barry, A. Procedures and theoretical considerations for testing antimicrobial agents in agar media. In Antibiotics in Laboratory Medicine, 5th ed.; Williams and Wilkins: Baltimore, MD, USA, 1991; pp. 1–16. [Google Scholar]
- National Committee for Clinical Laboratory Standard. NCCLS: Methods for Anti-microbial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria, Approved Guideline; Document M45-A 26(19); NCCLS: Willanova, PA, USA, 1999. [Google Scholar]
- Geary, W.J. The use of conductivity measurements in organic solvents for the characterization of coordination compounds. Coord. Chem. Rev. 1971, 7, 81–115. [Google Scholar] [CrossRef]
- Ali, A.Q.; Teoh, S.G.; Salhin, A.; Eltayeb, N.E.; Ahamed, M.B.K.; Majid, A.M.S.A. Synthesis of platinum(II) complexes of isatin thiosemicarbazones derivatives: In vitro anti-cancer and deoxyribose nucleic acid binding activities. Inorg. Chim. Acta. 2014, 416, 235–244. [Google Scholar] [CrossRef]
- Joseph, M.; Suni, V.; Prathapachandra Kurup, M.R.; Nethaji, M.; Kishore, A.; Bhat, S.G. Structural, spectral and antimicrobial studies of copper(II) complexes of 2-benzoylpyridine N(4)-cyclohexyl thiosemicarbazone. Polyhedron 2004, 23, 3069–3080. [Google Scholar] [CrossRef]
- Youssef, N.S.; Hegab, K.H. Synthesis and Characterization of some Transition Metal Complexes of Thiosemicarbazones Derived from 2-acetylpyrrole and 2-acetylfuran. Synth. React. Inorg. Met-Org. Nano-Met. Chem. 2005, 35, 391–398. [Google Scholar] [CrossRef]
- Dilovic, I.; Rubcic, M.; Vrdoljak, V. Novel thiosemicarbazone derivatives as potential antitumor agents: Synthesis, physicochemical and structural properties, DNA interactions and antiproliferative activity. Bioorg. Med. Chem. 2008, 16, 5189–5198. [Google Scholar] [CrossRef] [PubMed]
- Latheef, L.; Prathapachandra Kurup, M.R. Spectral and structural studies of copper(II) complexes of thiosemicarbazones derived from salicylaldehyde and containing ring incorporated at N(4)-position. Spectrochim. Acta Part A 2008, 70, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, K.M.; Bekheit, M.M. Synthesis and characterization of new metal complexes of thiosemicarbazone derived from 4-phenyl-3-thiosemicarbazide and chromone-3-carboxaldehyde. Trans. Met. Chem. 1988, 13, 230–232. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, 5th ed.; Wiley-Interscience: New York, NY, USA, 1997; p. 86. [Google Scholar]
- Jayakumar, K.; Sithambaresan, M.; Ambili Aravindakshan, A.; Prathapachandra Kurup, M.R. Synthesis and spectral characterization of copper(II) complexes derived from 2-benzoylpyridine-N4,N4-dimethyl-3-thiosemicarbazone: Crystal structure of a binuclear complex. Polyhedron 2014, 75, 50–56. [Google Scholar] [CrossRef]
- Chandra, S.; Vandana. Synthesis, spectroscopic, anticancer and antibacterial studies of Ni(II) and Cu(II) complexes with 2-carboxybenzaldehyde thiosemicarbazone. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 129, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Sreekanth, A.; Sivakumar, S.; Prathapachandra Kurup, M.R. Structural studies of six and four coordinate zinc(II), nickel(II) and dioxovanadium(V) complexes with thiosemicarbazones. J. Mol. Struct. 2003, 655, 47–58. [Google Scholar] [CrossRef]
- Chandra, S.; Bargujar, S.; Nirwal, R.; Yadav, N. Synthesis, spectral characterization and biological evaluation of copper(II) and nickel(II) complexes with thiosemicarbazones derived from a bidentate Schiff base. Spectrochimica Acta Part A: Mol. Biomol. Spectrosc. 2013, 106, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Lever, A.P.B. Inorganic Electronic Spectroscopy, 2th ed.; Elsevier Science: New York, NY, USA, 1984. [Google Scholar]
- Kovala-Demertzi, D.; Alexandratos, A.; Papageorgiou, A.; Yadav, P.N.; Dalezis, P.; Demertzis, M.A. Synthesis, characterization, crystal structures, in vitro and in vivo antitumor activity of palladium(II) and zinc(II) complexes with 2-formyl and 2-acetyl pyridine N(4)-1-(2-pyridyl)-piperazinyl thiosemicarbazone. Polyhedron 2008, 27, 2731–2738. [Google Scholar] [CrossRef]
- Sreekanth, A.; Prathapachandra Kurup, M.R. Structural and spectral studies on four coordinate copper (II) complexes of 2-benzoylpyridine N(4),N(4)-(butane-1,4-diyl)thiosemicarbazone. Polyhedron 2003, 22, 3321–3332. [Google Scholar] [CrossRef]
- Hathaway, B.J.; Billing, D.E. The electronic properties and stereochemistry of mononuclear complexes of the copper(II) ion. Coord. Chem. Rev. 1970, 5, 143–207. [Google Scholar] [CrossRef]
- El-Metwally, N.M.; Al-Hazmi, G.A.A. Spectroscopic evaluation for VO(II), Ni(II), Pd(II) and Cu(II) complexes derived from thiosemicarbazide: A special emphasis on EPR study and DNA cleavage. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 107, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Joseph, M.; Kuriakose, M.; Prathapachandra Kurup, M.R.; Suresh, E.; Kishore, A.; Bhat, S.G. Structural, antimicrobial and spectral studies of copper(II) complexes of 2-benzoylpyridine N(4)-phenyl thiosemicarbazone. Polyhedron 2006, 25, 61–70. [Google Scholar] [CrossRef]
- Hathaway, B.J. Structure and Bonding; Springer Verlag: Heidelberg, Germany, 1973; p. 60. [Google Scholar]
- Pogni, R.; Bartoo, M.C.; Diaz, A.; Basosi, R. EPR characterization of mono(thiosemicarbazones) copper(II) complexes. J. Inorg. Biochem. 2000, 79, 333–337. [Google Scholar] [CrossRef]
- Fries, J.; Getrost, H. Organic Reagents for Trace Analysis; E. Merck: Darmstadt, Germany, 1977; pp. 291–298. [Google Scholar]
- Sample Availability: Samples of the compounds are not available from the authors.
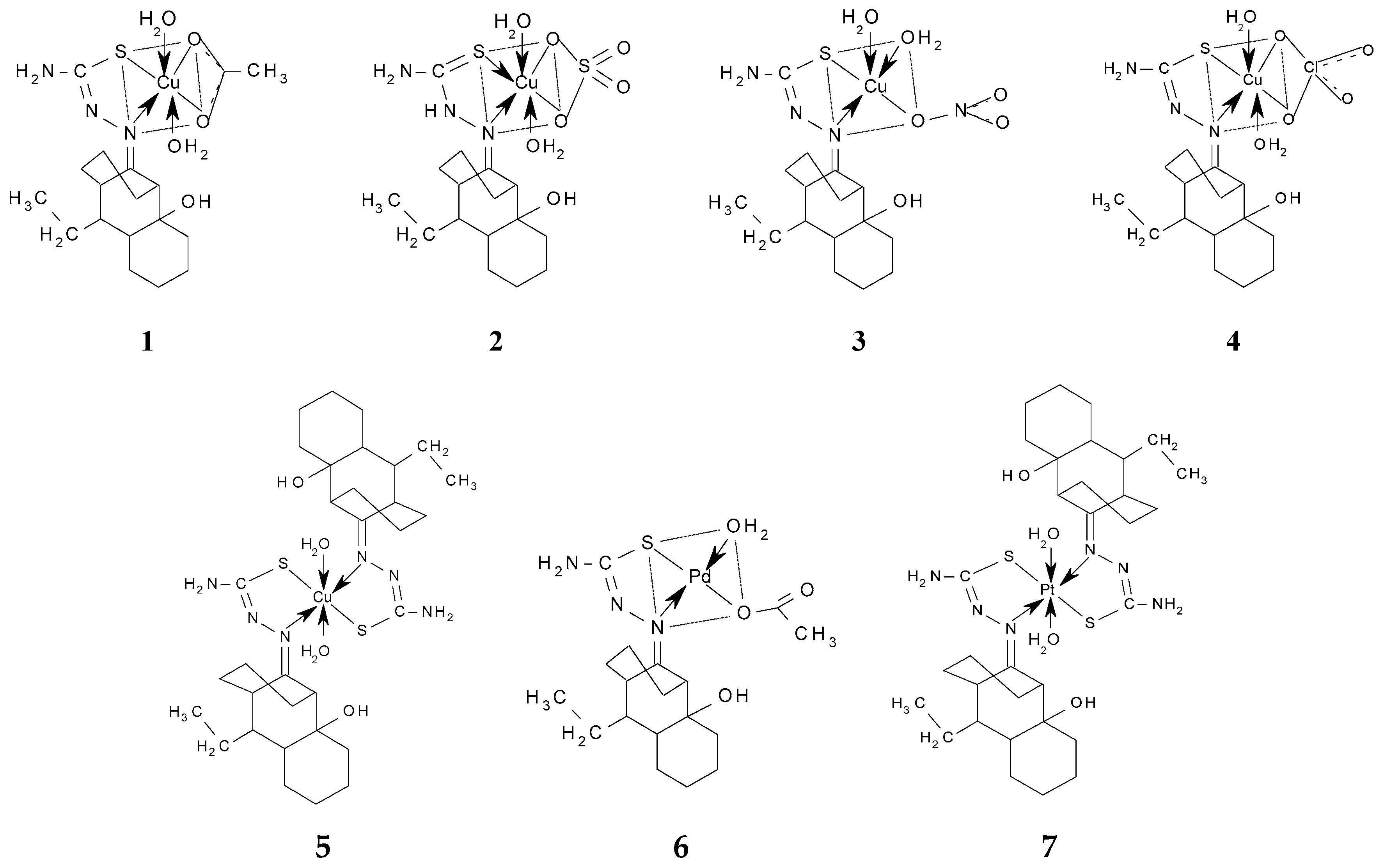

| Metal Complex Molecular Formula | Transitions d–d (cm−1) | µeff (BM) | Geometry | ||
|---|---|---|---|---|---|
| (Cu(L)(H2O)2(OAc)) (1) | 2B1g→2A1g - | 2B1g→2B2g 13,570 | 2B1g→2Eg 20,000 | 1.99 | Octahedral distorted |
| (Cu(HL)(H2O)2(SO4)) (2) | 2B1g→2A1g - | 2B1g→2B2g 13,570 | 2B1g→2Eg 20,000 | 1.83 | Octahedral distorted |
| (Cu(L)(H2O)2(NO3)) (3) | 2B1g→2A1g - | 2B1g→2B2g 12,690 | 2B1g→2Eg 20,000 | 1.69 | Square-pyramidal |
| (Cu(L)(H2O)2(ClO4)) (4) | 2B1g→2A1g - | 2B1g→2B2g 13,570 | 2B1g→2Eg 20,100 | 1.88 | Octahedral distorted |
| (Cu(L)2(H2O)2) (5) | 2B1g→2A1g - | 2B1g→2B2g 12,550 | 2B1g→2Eg 20,000 | 1.97 | Octahedral distorted |
| (Pd(L)(OAc))(H2O) (6) | 1A1g→1B1g - | 1A1g→1A2g 22,220 | 1A1g→1Eg - | * | Square-planar |
| (Pt(L)2) (7) | 1A1g→1B1g - | 1A1g→1A2g 23,250 | 1A1g→1Eg - | * | Square-planar |
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| Polycrystalline (298 K) | |||||
| g// | 2.404 | 2.371 | 2.208 | 2.409 (g3) | 2.237 |
| g⊥ | 2.087 | 2.103 | 2.041 | 2.193 (g2) | 2.065 |
| 2.011 (g1) | |||||
| DMSO (77K) | |||||
| g// | 2.394 | 2.400 | 2.399 | 2.399 | 2.400 |
| g⊥ | 2.082 | 2.088 | 2.076 | 2.078 | 2.098 |
| A// | 117.4 | 118.0 | 119.2 | 118.0 | 119.0 |
| α2 | 0.789 | 0.838 | 0.802 | 0.792 | 0.831 |
| β2 | 0.989 | 0.998 | 0.890 | 0.996 | 0.978 |
| δ2 | 0.992 | 0.879 | 0.973 | 0.969 | 0.952 |
| K// | 0.780 | 0.841 | 0.717 | 0.769 | 0.787 |
| K⊥ | 0.791 | 0.569 | 0.785 | 0.758 | 0.753 |
| Compounds | E. coli (G−) | S. enteritidis (G−) | S. aureus (G+) | E. faecalis (G+) | C. albicans | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| C16H27N3OS (HL) | 0.5 | >10.0 | >10.0 | >10.0 | 0.5 | >10.0 | >10.0 | >10.0 | 0.5 | >10.0 |
| (Cu(L)(H2O)2(OAc)) (1) | >10.0 | >10.0 | 0.5 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 |
| (Cu(HL)(H2O)2(SO4)) (2) | 0.25 | >10.0 | 0.25 | 0.12 | 0.12 | 0.12 | 0.12 | 0.12 | 0.12 | 0.12 |
| (Cu(L)(H2O)2(NO3)) (3) | >10.0 | >10.0 | >10.0 | >10.0 | 0.12 | 0.25 | 0.12 | 0.12 | 0.5 | 0.5 |
| (Cu(L)(H2O)2(ClO4)) (4) | 0.5 | >10.0 | 0.5 | >10.0 | 0.12 | >10.0 | 0.12 | >10.0 | 0.12 | 0.12 |
| (Cu(L)2(H2O)2) (5) | >10.0 | >10.0 | 0.5 | >10.0 | 0.12 | >10.0 | 0.12 | >10.0 | 0.12 | 0.12 |
| (Pd(L)(OAc))(H2O) (6) | >10.0 | >10.0 | 0.5 | >10.0 | 0.5 | >10.0 | >10.0 | >10.0 | >0.5 | >0.5 |
| (Pt(L)2) (7) | >10.0 | >10.0 | 0.5 | >10.0 | >10.0 | >10.0 | 0.12 | >10.0 | >10.0 | >10.0 |
| Cu(OAc)2·H2O | 0.5 | - | 1.024 | - | 1.024 | - | 0.5 | - | - | - |
| CuSO4·5H2O | 0.5 | - | 1.024 | - | 0.5 | - | 0.5 | - | 1.024 | - |
| Cu(NO3)2·3 H2O | 1.024 | - | 1.024 | - | 1.024 | - | 1.024 | - | - | - |
| Cu(ClO4)2·6 H2O | 1.024 | - | 1.024 | - | 1.024 | - | 0.5 | - | - | |
| CuCl2·2H2O | 1.024 | - | 0.5 | - | 0.5 | - | 0.5 | - | - | - |
| Pd(OAc)2 | 1.024 | - | 1.024 | - | 1.024 | - | 0.5 | - | - | |
| K2(PtCl4) | 1.024 | - | 1.024 | - | 1.024 | - | 0.5 | - | - | |
| Furacillinum | 0.018 | 0.037 | 0.009 | 0.009 | 0.009 | 0.009 | 0.037 | 0.037 | - | - |
| Nystatine | - | - | - | - | - | - | - | - | 0.08 | 0.08 |
| Compound | Inhibition of Cell Proliferation % | IC50 (µM) b | ||
|---|---|---|---|---|
| 10 µM | 1 µM | 0.1 µM | ||
| C16H27N3OS (HL) | −2.90 | −5.72 | 0.31 | - |
| (Cu(L)(H2O)2(OAc)) (1) | 21.97 | −2.11 | −2.58 | ≥10 |
| (Cu(HL)(H2O)2(SO4)) (2) | 100.00 | 5.14 | -5.25 | 1.6 ± 3 |
| (Cu(L)(H2O)2(NO3)) (3) | 63.91 | 8.12 | 1.35 | 6.4 ± 5 |
| (Cu(L)(H2O)2(ClO4)) (4) | 48.94 | 6.48 | 1.13 | 6.5 ± 4 |
| (Cu(L)2(H2O)2) (5) | 43.43 | 8.73 | 7.20 | 14.2 ± 3 |
| (Pd(L)(OAc))(H2O) (6) | 100.00 | −22.99 | −3.85 | 6.5 ± 4 |
| (Pt(L)2) (7) | 100.00 | −10.32 | −8.13 | 6.4 ± 2 |
| DOXO c | 91.94 | 94.55 | 77.00 | ≤0.1 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pahonțu, E.; Paraschivescu, C.; Ilieș, D.-C.; Poirier, D.; Oprean, C.; Păunescu, V.; Gulea, A.; Roșu, T.; Bratu, O. Synthesis and Characterization of Novel Cu(II), Pd(II) and Pt(II) Complexes with 8-Ethyl-2-hydroxytricyclo(7.3.1.02,7)tridecan-13-one-thiosemicarbazone: Antimicrobial and in Vitro Antiproliferative Activity. Molecules 2016, 21, 674. https://doi.org/10.3390/molecules21050674
Pahonțu E, Paraschivescu C, Ilieș D-C, Poirier D, Oprean C, Păunescu V, Gulea A, Roșu T, Bratu O. Synthesis and Characterization of Novel Cu(II), Pd(II) and Pt(II) Complexes with 8-Ethyl-2-hydroxytricyclo(7.3.1.02,7)tridecan-13-one-thiosemicarbazone: Antimicrobial and in Vitro Antiproliferative Activity. Molecules. 2016; 21(5):674. https://doi.org/10.3390/molecules21050674
Chicago/Turabian StylePahonțu, Elena, Codruța Paraschivescu, Diana-Carolina Ilieș, Donald Poirier, Camelia Oprean, Virgil Păunescu, Aurelian Gulea, Tudor Roșu, and Ovidiu Bratu. 2016. "Synthesis and Characterization of Novel Cu(II), Pd(II) and Pt(II) Complexes with 8-Ethyl-2-hydroxytricyclo(7.3.1.02,7)tridecan-13-one-thiosemicarbazone: Antimicrobial and in Vitro Antiproliferative Activity" Molecules 21, no. 5: 674. https://doi.org/10.3390/molecules21050674
APA StylePahonțu, E., Paraschivescu, C., Ilieș, D.-C., Poirier, D., Oprean, C., Păunescu, V., Gulea, A., Roșu, T., & Bratu, O. (2016). Synthesis and Characterization of Novel Cu(II), Pd(II) and Pt(II) Complexes with 8-Ethyl-2-hydroxytricyclo(7.3.1.02,7)tridecan-13-one-thiosemicarbazone: Antimicrobial and in Vitro Antiproliferative Activity. Molecules, 21(5), 674. https://doi.org/10.3390/molecules21050674