Current Knowledge on the Importance of Selenium in Food for Living Organisms: A Review
Abstract
:1. Introduction
2. Selenium Contents of Foods
3. Selenium Deficiency in the Diet
4. Consequences of Excess Content of Selenium Intake with the Diet
5. Physiological Importance of Selenium in Human and Animal Organisms
6. Recommended Doses of Selenium
7. Methods of Dietary Supplementation with Selenium
8. Influence of Selenium in Cancer Prevention
9. Conclusions
Author Contributions
Conflicts of Interest
References
- Kieliszek, M.; Błażejak, S. Selenium: Significance, and outlook for supplementation. Nutrition 2013, 29, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.L. The selenium dilemma. J. Nutr. 1973, 103, 803–810. [Google Scholar] [PubMed]
- Holben, D.H.; Smith, A.M. The diverse role of selenium within selenoproteins: A review. J. Am. Diet. Assoc. 1999, 99, 836–843. [Google Scholar] [CrossRef]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Stolińska, H.; Wolańska, D. Składniki pokarmowe istotne w niedoczynności tarczycy. Żyw. Człow. Metab. 2012, 3, 221–231. [Google Scholar]
- Navarro-Alarcon, M.; Cabrera-Vique, C. Selenium in food and the human body: A review. Sci. Total Environ. 2008, 400, 115–141. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Collings, R.; Hurst, R. Selenium bioavailability: Current knowledge and future research requirements. Am. J. Clin. Nutr. 2010, 91, 1484S–1491S. [Google Scholar] [CrossRef] [PubMed]
- Tinggi, U. Essentiality and toxicity of selenium and its status in Australia: A review. Toxicol. Lett. 2003, 137, 103–110. [Google Scholar] [CrossRef]
- Zagrodzki, P.; Laszczyk, P. Selenium and cardiovascular disease: Selected issues. Postepy Hig. Med. Dosw. 2006, 60, 624–631. [Google Scholar]
- Fordyce, F. Selenium geochemistry and health. Ambio 2007, 36, 94–97. [Google Scholar] [CrossRef]
- Dumont, E.; Vanhaecke, F.; Cornelis, R. Selenium speciation from food source to metabolites: A critical review. Anal. Bioanal. Chem. 2006, 385, 1304–1323. [Google Scholar] [CrossRef] [PubMed]
- Pedrero, Z.; Madrid, Y. Novel approaches for selenium speciation in foodstuffs and biological specimens: A review. Anal. Chim. Acta. 2009, 634, 135–152. [Google Scholar] [CrossRef] [PubMed]
- Ip, C.; Thompson, H.J.; Zhu, Z.; Ganther, H.E. In vitro and in vivo studies of methylseleninic acid: Evidence that a monomethylated selenium metabolite is critical for cancer chemoprevention. Cancer Res. 2000, 60, 2882–2886. [Google Scholar] [PubMed]
- Waegeneers, N.; Thiry, C.; De Temmerman, L.; Ruttens, A. Dietary intake of selenium by a general adult population in Belgium. Food Addit. Contam. Part A 2013, 30, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Pappa, E.C.; Pappas, A.C.; Surai, P.F. Selenium content in selected foods from the Greek market and estimation of the daily intake. Sci. Total Environ. 2006, 372, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Smrkolj, P.; Pograjc, L.; Hlastan-Ribic, C.; Stibilj, V. Selenium content in selected Slovenian foodstuffs and estimated daily intakes of selenium. Food Chem. 2005, 90, 691–697. [Google Scholar] [CrossRef]
- Thiry, C.; Ruttens, A.; De Temmerman, L.; Schneider, Y.; Pussemier, L. Current knowledge in species-related bioavailability of selenium in food. Food Chem. 2012, 130, 767–784. [Google Scholar] [CrossRef]
- World Health Organization. Vitamin and Mineral Requirements in Human Nutrition. Report of a Joint FAO/WHO Expert Consultation; World Health Organization: Bangkok, Tailand; Geneva, Switzerland, 2004. [Google Scholar]
- Maseko, T.; Callahan, D.L.; Dunshea, F.R.; Doronila, A.; Kolev, S.; Ng, K. Chemical characterization and speciation of organic selenium in cultivated selenium-enriched Agaricus bisporus. Food Chem. 2013, 141, 3681–3687. [Google Scholar] [CrossRef] [PubMed]
- Wesołowski, M. Selen-pierwiastek życia. Panacea 2006, 3, 12–16. [Google Scholar]
- Finley, J.W. Selenium accumulation in plant foods. Nutr. Rev. 2005, 63, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Corona, M.T.; Sánchez-Martínez, M.; Valderrama, M.; Rodríguez, M.E.; Cámara, C.; Madrid, Y. Selenium biotransformation by Saccharomyces cerevisiae and Saccharomyces bayanus during white wine manufacture: Laboratory-scale experiments. Food Chem. 2011, 124, 1050–1055. [Google Scholar] [CrossRef]
- Kieliszek, M.; Błażejak, S.; Jędrzejczak, R. Wiązanie selenu przez drożdże paszowe Candida utilis ATCC 9950. Bromat. Chem. Toksykol. 2012, 45, 628–633. [Google Scholar]
- Alves, M.R.; Starling, A.L.; Kanufre, V.C.; Soares, R.D.; Norton, R.C.; Aguiar, M.J.; Januario, J.N. Selenium intake and nutritional status of children with phenylketonuria in Minas Gerais, Brazil. J. Pediatr. (Rio J.) 2012, 88, 396–400. [Google Scholar] [CrossRef]
- Eroglu, C.; Unal, D.; Cetin, A.; Orhan, O.; Sivgin, S.; Oztürk, A. Effect of serum selenium levels on radiotherapy-related toxicity in patients undergoing radiotherapy for head and neck cancer. Anticancer Res. 2012, 32, 3587–3590. [Google Scholar] [PubMed]
- Mistry, H.D.; Pipkin, F.B.; Redman, C.W.; Poston, L. Selenium in reproductive health. Am. J. Obstet. Gynecol. 2012, 206, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Kryczyk, J.; Zagrodzki, P. Selen w chorobie Gravesa-Basedowa. Postepy Hig. Med. Dosw. 2013, 67, 491–498. [Google Scholar] [CrossRef]
- Yao, Y.; Pei, F.; Kang, P. Selenium, iodine, and the relation with Kashin-Beck disease. Nutrition 2011, 27, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Lyons, G.; Stangoulis, J.; Graham, R. High-selenium wheat: Biofortification for better health. Nutr. Res. Rev. 2003, 16, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. The use of high-selenium yeast to raise selenium status: How does it measure up? Br. J. Nutr. 2004, 92, 557–573. [Google Scholar] [CrossRef] [PubMed]
- Włodarek, D. Znaczenie selenu dla zdrowia człowieka. Żyw. Człow. Metab. 2006, 33, 236–241. [Google Scholar]
- Patelski, P.; Dziekońska, U. Wpływ selenianu (IV) sodu na wzrost i aktywność fermentacyjną drożdży piekarskich. Zywn. Nauk. Technol. Jakosc. 2013, 3, 132–142. [Google Scholar]
- Musiał, J.; Juszczyk, P.; Rymowicz, W.; Kinal, S. Produkcja drożdży paszowych Yarrowia lipolytica wzbogaconych w selen i chrom. Acta Sci. Pol. Biotechnol. 2005, 4, 55–64. [Google Scholar]
- Oldfield, J.E. A brief history of selenium research: From alkali disease to prostate cancer (from poison to prevention). J. Anim. Sci. Online supplement. 2002, 11:1, 1–4. [Google Scholar]
- Khanal, D.R.; Knight, A.P. Selenium: Its role in livestock health and productivity. J. Agric. Environ. 2010, 11, 101–106. [Google Scholar] [CrossRef]
- Fordyce, F.M. Selenium deficiency and toxicity in the environment. In Essentials of Medical Geology; Selinus, O., Alloway, B., Centeno, J.A., Finkelman, R.B., Fuge, R., Lindh, U., Smedley, P., Eds.; Elsevier Academic Press: Amsterdam, The Netherlands, 2005; pp. 373–416. [Google Scholar]
- Fernández-Martinez, A.; Charlet, L. Selenium bioavailability and cycling in the environment: A structural chemist’s point of view. Rev. Environ. Sci. Biotechnol. 2009, 8, 81–110. [Google Scholar] [CrossRef]
- Li, S.; Xiao, T.; Zheng, B. Medical geology of arsenic, selenium and thallium in China. Sci. Total Environ. 2012, 421–422, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Nazemi, L.; Nazmara, S.; Eshraghyan, M.R.; Nasseri, S.; Djafarian, K.; Yunesian, M.; Sereshti, H.; Moameni, A.; Shahtaheri, S.J. Selenium status in soil, water and essential crops of Iran. Iran. J. Environ. Health Sci. Eng. 2012, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Letavayová, L.; Vlasáková, D.; Spallholz, J.E.; Brozmanová, J.; Chovanec, M. Toxicity and mutagenicity of selenium compounds in Saccharomyces cerevisiae. Mutat. Res. 2008, 638, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zwolak, I.; Zaporowska, H. Selenium interactions and toxicity: A review. Selenium interactions and toxicity. Cell Biol. Toxicol. 2012, 28, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Drutel, A.; Archambeaud, F.; Caron, P. Selenium and the thyroid gland: More good news for clinicians. Clin. Endocrinol. 2013, 78, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Błażejak, S.; Gientka, I.; Bzducha-Wróbel, A. Accumulation and metabolism of selenium by yeast cell. Appl. Microbiol. Biotechnol. 2015, 99, 5373–5382. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Błażejak, S.; Bzducha-Wróbel, A. Influence of selenium content in the culture medium on protein profile of yeast cells Candida utilis ATCC 9950. Oxid. Med. Cell. Longev. 2015, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jovanović, I.B.; Veličković, M.; Vuković, D.; Milanović, S.; Valčić, O.; Gvozdić, D. Effects of different amounts of supplemental selenium and vitamin E on the incidence of retained placenta, selenium, malondialdehyde, and thyronines status in cows treated with prostaglandin F2α for the induction of parturition. J. Vet. Med. 2013, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ruseva, B.; Himcheva, I.; Nankova, D. Importance of selenoproteins for the function of the thyroid gland. Medicine 2013, 3, 60–64. [Google Scholar]
- Roman, M.; Jitarub, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Ogawa-Wong, A.N.; Berry, M.J.; Seale, L.A. Selenium and metabolic disorders: An emphasis on type 2 diabetes risk. Nutrients 2016, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Żarczyńska, K.; Sobiech, P.; Radwińska, J.; Rękawek, W. Effects of selenium on animal health. J. Elem. 2013, 18, 329–340. [Google Scholar] [CrossRef]
- Dokoupilová, A.; Marounek, M.; Skřivanová, V.; Březina, P. Selenium content in tissues and meat quality in rabbits fed selenium yeast. Czech J. Anim. Sci. 2007, 52, 165–169. [Google Scholar]
- Saxena, R.; Jaiswal, G. Selenium and its role in health and disease. Kuwait Med. J. 2007, 1, 10–18. [Google Scholar]
- Yaroshenko, F.O.; Dvorska, J.E.; Surai, P.F.; Sparks, N.H.C. Selenium-enriched eggs as a source of selenium for human consumption. Appl. Biotechnol. Food Sci. Policy 2003, 1, 13–23. [Google Scholar]
- Rosen, B.R.; Liu, Z.J. Transport pathways for arsenic and selenium: A minireview. Environ. Int. 2009, 35, 512–515. [Google Scholar] [CrossRef] [PubMed]
- Shini, S.; Sultan, A.; Bryden, W.L. Selenium biochemistry and bioavailability: Implications for animal agriculture. Agriculture 2015, 5, 1277–1288. [Google Scholar] [CrossRef]
- Tamura, T.; Stadtman, T.C. A new selenoprotein from human lung adenocarcinoma cells: Purification, properties and thioredoxin reductase activity. Proc. Natl. Acad. Sci. USA 1996, 93, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Kamwesiga, J.; Mutabazi, V.; Kayumba, J.; Tayari, J.C.K.; Uwimbabazi, C.J.; Batanage, G.; Uwera, G.; Baziruwiha, M.; Ntizimira, C.; Murebwayire, A.; et al. Effect of selenium supplementation on CD4+ T-cell recovery, viral suppression and morbidity of HIV-infected patients in Rwanda: A randomized controlled trial. AIDS 2015, 29, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Szucik, K.; Gondek, M.; Belkot, Z.; Kursa, K. Zawartość selenu w mięśniach i narządach wewnętrznych koni rzeźnych w zależności od ich wieku i płci. Zywn. Nauk. Technol. Jakosc 2014, 5, 63–71. [Google Scholar]
- Lipiński, B. Can selenite be an ultimate inhibitor of Ebola and other viral infections. Br. J. Med. Med. Res. 2015, 6, 319–324. [Google Scholar] [CrossRef]
- Brozmanová, J.; Mániková, D.; Vlčková, V.; Chovanec, M. Selenium: A double-edged sword for defense and offence in cancer. Arch. Toxicol. 2010, 84, 919–938. [Google Scholar] [CrossRef] [PubMed]
- Papp, L.V.; Lu, J.; Holmgren, A.; Khanna, K.K. From selenium to selenoproteins: Synthesis, identity, and their role in human health. Antioxid. Redox Sign. 2007, 9, 775–806. [Google Scholar] [CrossRef] [PubMed]
- Ferreiro, A.; Quijano-Roy, S.; Pichereau, C.; Moghadaszadeh, B.; Goemans, N.; Bönnemann, C.; Jungbluth, H.; Straub, V.; Villanova, M.; Leroy, J.P.; et al. Mutations of the selenoprotein N gene, which is implicated in rigid spine muscular dystrophy, cause the classical phenotype of multiminicore disease: Reassessing the nosology of early-onset myopathies. Am. J. Hum. Genet. 2002, 71, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F., Jr. Biomarkers of selenium status. Nutrients 2015, 7, 2209–2236. [Google Scholar] [CrossRef] [PubMed]
- Gromer, S.; Eubel, J.K.; Lee, B.L.; Jacob, J. Human selenoproteins at a glance. Cell. Mol. Life Sci. 2005, 62, 2414–2437. [Google Scholar] [CrossRef] [PubMed]
- Clark, L.C.; Dalkin, B.; Krongrad, A.; Combs, G.F., Jr.; Turnbull, B.W.; Slate, E.H.; Witherington, R.; Herlong, J.H.; Janosko, E.; Carpenter, D.; et al. Decreased incidence of prostate cancer with selenium supplementation: Results of a double-blind cancer prevention trial. Br. J. Urol. 1998, 81, 730–734. [Google Scholar] [CrossRef] [PubMed]
- Hatfield, D.L.; Tsuji, P.A.; Carlson, B.A.; Gladyshev, V.N. Selenium and selenocysteine: Roles in cancer, health, and development. Trends Biochem. Sci. 2014, 39, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Duffield-Lillico, A.J.; Reid, M.E.; Turnbull, B.W.; Combs, G.F.; Slate, E.H.; Fischbach, L.A.; Marshall, J.R.; Clark, L.C. Baseline characteristics and the effect of selenium supplementation on cancer incidence in a randomized clinical trial A summary report of the nutritional prevention of cancer trial. Cancer Epidem. Biomar. 2002, 11, 630–639. [Google Scholar]
- Jönsson-Videsäter, K.; Björkhem-Bergman, L.; Hossain, A.; Söderberg, A.; Eriksson, L.C.; Paul, C.; Rosén, A.; Björnstedt, M. Selenite-induced apoptosis in doxorubicin-resistant cells and effects on the thioredoxin system. Biochem. Pharmacol. 2004, 67, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Hendrickx, W.; Decock, J.; Mulholland, F.; Bao, Y.; Fairweather-Tait, S. Selenium biomarkers in prostate cancer cell lines and influence of selenium on invasive potential of PC3 cells. Front. Oncol. 2013, 3, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Wąsowicz, W.; Gromadzińska, J.; Rydzyński, K.; Tomczak, J. Selenium status of low-selenium area residents: Polish experience. Toxicol. Lett. 2003, 137, 95–101. [Google Scholar] [CrossRef]
- Combs, G.F. Selenium in global food systems. Br. J. Nutr. 2001, 85, 517–547. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Scientific opinion of the panel on food additives, flavourings, processing aids and materials in contact with food [AFC] on a request from the commission on selenium-enriched yeast as source for selenium. EFSA J. 2008, 766, 1–42. [Google Scholar] [CrossRef]
- Alfthan, G.; Eurola, M.; Ekholm, P.; Venäläinen, E.R.; Root, T.; Korkalainen, K.; Hartikainen, H.; Salminen, P.; Hietaniemi, V.; Aspila, P.; Aro, A. Effects of nationwide addition of selenium to fertilizers on foods, and animal and human health in Finland: From deficiency to optimal selenium status of the population. J. Trace Elem. Med. Biol. 2015, 31, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Achibat, H.; AlOmari, N.A.; Messina, F.; Sancineto, L.; Khouili, M.; Santi, C. Organoselenium compounds as phytochemicals from the Natural Kingdom. Nat. Prod. Commun. 2015, 10, 1885–1892. [Google Scholar] [PubMed]
- Ros, G.H.; van Rotterdam, A.M.D.; Bussink, D.W.; Bindraban, P.S. Selenium fertilization strategies for bio-fortification of food: An agro-ecosystem approach. Plant Soil 2016. [Google Scholar] [CrossRef]
- Ramos, S.J.; Faquin, V.; Guilherme, L.R.G.; Castro, E.M.; Ávila, F.W.; Carvalho, G.S.; Bastos, C.E.A.; Oliveira, C. Selenium biofortification and antioxidant activity in lettuce plants fed with selenate and selenite. Plant Soil Environ. 2010, 56, 584–588. [Google Scholar]
- Şlencu, B.G.; Ciobanu, C.; Cuciureanu, R. Selenium content in foodstuffs and its nutritional requirement for humans. Clujul Med. 2012, 85, 139–145. [Google Scholar]
- Bitterli, C.; Bañuelos, G.S.; Schulin, R. Use of transfer factors to characterize uptake of selenium by plants. J. Geochem. Explor. 2010, 107, 206–216. [Google Scholar] [CrossRef]
- Niwińska, B.; Andrzejewski, M. Selen w żywieniu bydła. Rocz. Nauk. Zoot. 2013, 40, 97–107. [Google Scholar]
- Diowksz, A.; Pęczkowska, B.; Wŀodarczyk, M.; Ambroziak, W. Bacteria/yeast and plant biomass enriched in Se via bioconversion process as a source of Se supplementation in food. Progr. Biotechnol. 2000, 17, 295–300. [Google Scholar]
- Steinbrenner, H.; Al-Quraishy, S.; Dkhil, M.A.; Wunderlich, F.; Sies, H. Dietary selenium in adjuvant therapy of viral and bacterial infections. Adv. Nutr. 2015, 6, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Jach, M.E.; Serefko, A.; Sajnaga, E.; Kozak, E.; Poleszak, E.; Malm, A. Dietary supplements based on the yeast biomass. Curr. Top. Nutraceutical Res. 2015, 13, 83–88. [Google Scholar]
- Van Bakel, M.M.; Printzen, G.; Wermuth, B.; Wiesmann, U.N. Antioxidant and thyroid hormone status in selenium-deficient phenylketonuric and hyperphenylalaninemic patients. Am. J. Clin. Nutr. 2000, 72, 976–981. [Google Scholar] [PubMed]
- Kieliszek, M.; Błażejak, S.; Płaczek, M. Spectrophotometric evaluation of selenium binding by Saccharomyces cerevisiae ATCC MYA-2200 and Candida utilis ATCC 9950 yeast. J. Trace Elem. Med. Biol. 2016, 35, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Mehdi, Y.; Hornick, J.L.; Istasse, L.; Dufrasne, I. Selenium in the environment, metabolism and involvement in body functions. Molecules 2013, 18, 3292–3311. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Błażejak, S.; Bzducha-Wróbel, A.; Kurcz, A. Effects of selenium on morphological changes in Candida utilis ATCC 9950 yeast cells. Biol. Trace Elem. Res. 2016, 169, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Stabnikova, O.; Ivanov, V.; Larionova, I.; Stabnikov, V.; Bryszewska, M.A.; Lewis, J. Ukrainian dietary bakery product with selenium-enriched yeast. LWT-Food Sci. Technol. 2008, 41, 890–895. [Google Scholar] [CrossRef]
- McSheehy, S.; Yang, L.; Mester, Z. Selenomethionine extraction from selenized yeast: An LC-MS. Study of the acid hydrolysis of a synthetic selenopeptide. Microchim. Acta (MCA) 2006, 155, 373–377. [Google Scholar] [CrossRef]
- Finley, J.W. Bioavailability of selenium from foods. Nutr. Rev. 2006, 64, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Fisinin, V.I.; Papazyan, T.T.; Surai, P.F. Producing selenium-enriched eggs and meat to improve the selenium status of thegeneral population. Crit. Rev. Biotechnol. 2009, 29, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Bourre, J.M.; Galea, F. An important source of omega-3 fatty acids, vitamins D and E, carotenoids, iodine and selenium: A new natural multi-enriched egg. J. Nutr. Health. Aging. 2006, 10, 371–376. [Google Scholar] [PubMed]
- FDA. Agency Response Letter GRAS Notice No GRN 000260; FDA: Silver Spring, MD, USA, 2008. [Google Scholar]
- Reilly, C. Selenium: A new entrant into the functional food arena. Trends Food Sci. Technol. 1998, 9, 114–118. [Google Scholar] [CrossRef]
- Wu, M.; Kang, M.M.; Schoene, N.W.; Cheng, W.H. Selenium compounds activate early barriers of tumorigenesis. J. Biol. Chem. 2010, 285, 12055–12062. [Google Scholar] [CrossRef] [PubMed]
- Lei, C.; Niu, X.; Wei, J.; Zhu, J.; Zhu, Y. Interaction of glutathione peroxidase-1 and selenium in endemic dilated cardiomyopathy. Clin. Chim. Acta 2009, 399, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Prabhu, K.S.; Mastro, A.M. Is selenium a potential treatment for cancer metastasis? Nutrients 2013, 5, 1149–1168. [Google Scholar] [CrossRef] [PubMed]
- Selol—Cancer Prevention. Available online: http://biosel.pl/index.html (accessed on 25 March 2016).
- Flis, A.; Suchocki, P.; Królikowska, M.A.; Suchocka, Z.; Remiszewska, M.; Śliwka, L.; Książek, I.; Sitarz, K.; Sochacka, M.; Hoser, G.; et al. Selenitetriglycerides—Redox-active agents. Pharmacol. Rep. 2015, 67, 1–8. [Google Scholar] [CrossRef] [PubMed]
- De Souza, L.R.; Muehlmann, L.A.; Matos, L.C.; Simón-Vázquez, R.; Lacava, Z.G.; De-Paula, A.M.; Mosiniewicz-Szablewska, E.; Suchocki, P.; Morais, P.C.; González-Fernández, Á.; et al. Antitumor activity and systemic effects of PVM/MA-shelled selol nanocapsules in lung adenocarcinoma-bearing mice. Nanotechnology 2015, 26, 505101. [Google Scholar] [CrossRef] [PubMed]
- Ślusarczyk, J.; Wierzbicka, M.; Suchocki, P.; Kuraś, M. Ultrastructural changes in onion (Allium cepa L.) root tip meristem cells treated with Selol and sodium selenate (IV). Caryologia 2016, 68, 1–11. [Google Scholar] [CrossRef]
- Zabłocka, K.; Biernat, J. The influence of selected nutritional components in lung cancer risk—unsaturated fatty acids, isothiocyanates, selenium. Współczesna Onkol. 2010, 14, 54–58. [Google Scholar] [CrossRef]
- Venza, M.; Visalli, M.; Beninati, C.; De Gaetano, G.V.; Teti, D.; Venza, I. Cellular mechanisms of oxidative stress and action in melanoma. Oxid. Med. Cell. Longev. 2015, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zachara, B.A. Selenium and selenium-dependent antioxidants in chronic kidney disease. Adv. Clin. Chem. 2015, 68, 131–151. [Google Scholar] [PubMed]
- Gromadzinska, J.; Reszka, E.; Bruzelius, K.; Wasowicz, W.; Akesson, B. Selenium and cancer: Biomarkers of selenium status and molecular action of selenium supplements. Eur. J. Clin. Nutr. 2008, 47 (Suppl. S2), 29–50. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Selenium in cancer prevention: A review of the evidence and mechanism of action. Proc. Nutr. Soc. 2005, 64, 527–542. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Wu, M.; Botnen, J.H. Methylselenol, a selenium metabolite, induces cell cycle arrest in G1 phase and apoptosis via the extracellular-regulated kinase 1/2 pathway and other cancer signaling genes. J. Nutr. 2009, 139, 1613–1618. [Google Scholar] [CrossRef] [PubMed]
- Lavu, R.V.S.; Van De Wiele, T.; Pratti, V.L.; Tack, F.; Du Laing, G. Selenium bioaccessibility in stomach, small intestine and colon: Comparison between pure Se compounds, Se-enriched food crops and food supplements. Food Chem. 2016, 197, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Reszka, E.; Jablonska, E.; Gromadzinska, J.; Wasowicz, W. Relevance of selenoprotein transcripts for selenium status in humans. Genes Nutr. 2012, 7, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, P.R.; Berry, M.J. The influence of selenium on immune responses. Mol. Nutr. Food Res. 2008, 52, 1273–1280. [Google Scholar] [CrossRef] [PubMed]
- Kenfield, S.A.; Van Blarigan, E.L.; DuPre, N.; Stampfer, M.J.; Giovannucci, E.L.; Chan, J.M. Selenium supplementation and prostate cancer mortality. J. Natl. Cancer Inst. 2015, 107, 360. [Google Scholar] [CrossRef] [PubMed]
- Gromadzinska, J.; Sklodowska, M.; Wolkanin, P.; Wasowicz, W.; Quispe, O.Z.; Pluzanska, A. Lipid peroxide levels and antioxidant enzyme activities in blood of breast cancer patients. J. Clin. Biochem. Nutr. 1992, 13, 127–135. [Google Scholar] [CrossRef]
- Méplan, C.; Crosley, L.K.; Nicol, F.; Beckett, G.J.; Howie, A.F.; Hill, K.E.; Hesketh, J.E. Genetic polymorphisms in the human selenoprotein P gene determine the response of selenoprotein markers to selenium supplementation in a gender-specific manner (the SELGEN study). FASEB J. 2007, 21, 3063–3074. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, A.; Kim, D.H.; Relton, C.; Ahn, Y.O.; Hesketh, J. Polymorphisms in the selenoprotein S and 15-kDa selenoprotein genes are associated with altered susceptibility to colorectal cancer. Genes Nutr. 2010, 5, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.; Méplan, C.; Gautrey, H.; Hall, J.; Hesketh, J.E. Differential effects of selenium and knock-down of glutathione peroxidases on TNFα and flagellin inflammatory responses in gut epithelial cells. Genes Nutr. 2012, 7, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Heras, I.L.; Palomo, M.; Madrid, Y. Selenoproteins: The key factor in selenium essentiality. State of the art analytical techniques for selenoprotein studies. Anal. Bioanal. Chem. 2011, 400, 1717–1727. [Google Scholar] [CrossRef] [PubMed]
| Food Products | Selenium Content (µg/g) | Reference |
|---|---|---|
| Selenium yeast | 3000 | [12] |
| Brazil nuts | 0.85–6.86 | [7] |
| Garlic | 0.5 | [13] |
| Onion | 0.5 | [7] |
| Salmon | 0.21–0.27 | [14] |
| Eggs | 0.17 | [15] |
| Beef | 0.35–0.47 | [16] |
| Chicken | 0.57 | [17] |
| Milk products | 0.01–0.55 | [7] |
| Name | Molecular Formula | Chemical Structures | Abbreviation |
|---|---|---|---|
| Sodium Selenite (IV) | Na2SeO3 | 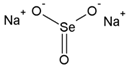 | Se(IV) |
| Sodium Selenate (VI) | Na2SeO4 | 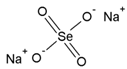 | Se(VI) |
| Selenomethionine | C5H11NO2Se | 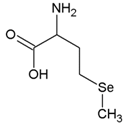 | SeMet |
| Selenocysteine | C3H7NO2Se |  | SeCys |
| γ-glutamyl- Se-methylselenocysteine | C9H16N2O5Se | 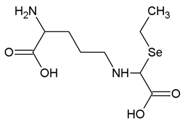 | γ-glutamyl- SeMeSeCys |
| Se-methyl selenocysteine | C4H9NO2Se | 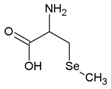 | SeMeSeCys |
| Methylselenol | CH4Se | 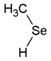 | MeSeH |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kieliszek, M.; Błażejak, S. Current Knowledge on the Importance of Selenium in Food for Living Organisms: A Review. Molecules 2016, 21, 609. https://doi.org/10.3390/molecules21050609
Kieliszek M, Błażejak S. Current Knowledge on the Importance of Selenium in Food for Living Organisms: A Review. Molecules. 2016; 21(5):609. https://doi.org/10.3390/molecules21050609
Chicago/Turabian StyleKieliszek, Marek, and Stanisław Błażejak. 2016. "Current Knowledge on the Importance of Selenium in Food for Living Organisms: A Review" Molecules 21, no. 5: 609. https://doi.org/10.3390/molecules21050609
APA StyleKieliszek, M., & Błażejak, S. (2016). Current Knowledge on the Importance of Selenium in Food for Living Organisms: A Review. Molecules, 21(5), 609. https://doi.org/10.3390/molecules21050609







