Polysaccharides from the Marine Environment with Pharmacological, Cosmeceutical and Nutraceutical Potential
Abstract
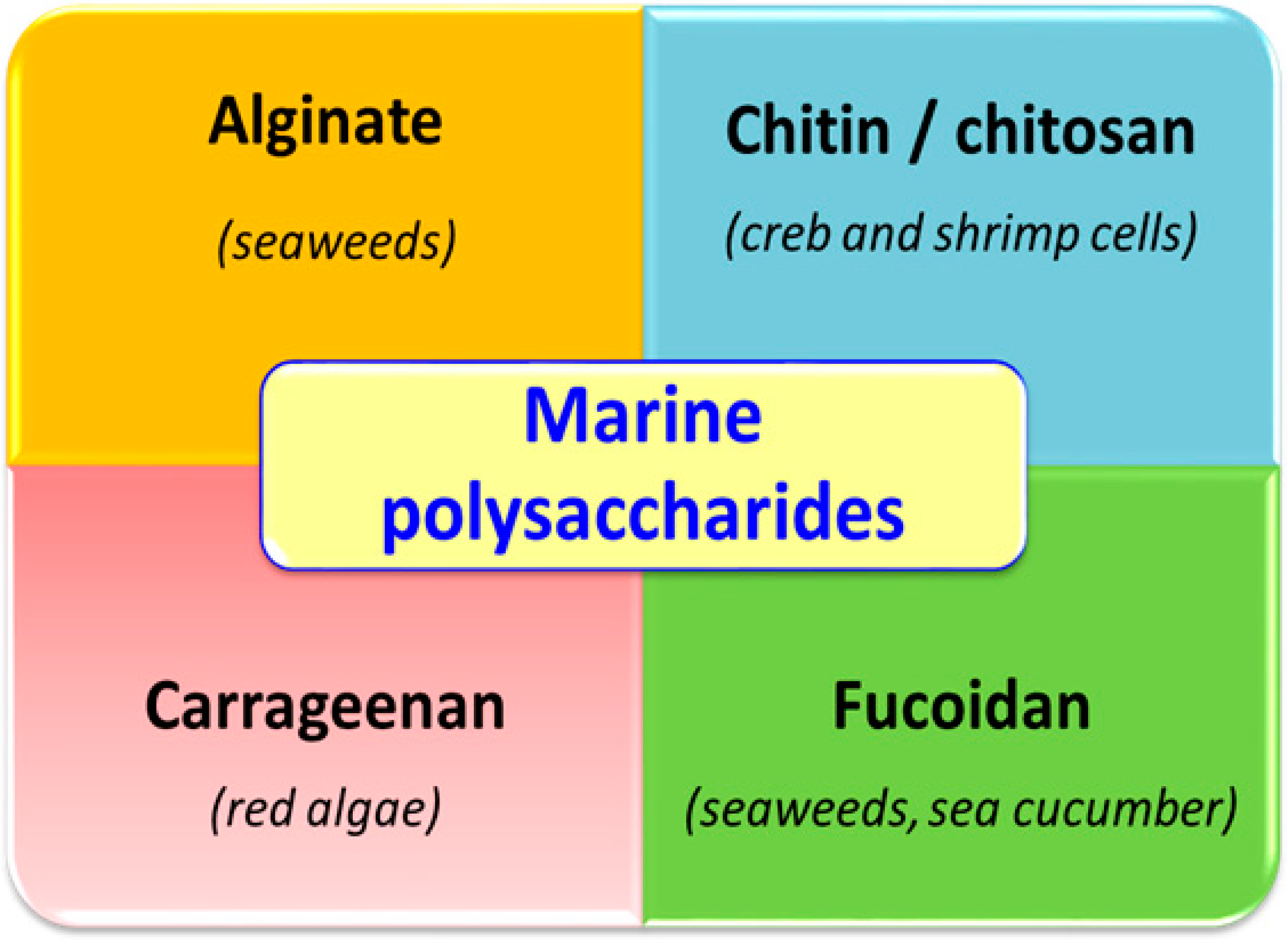
:1. Introduction
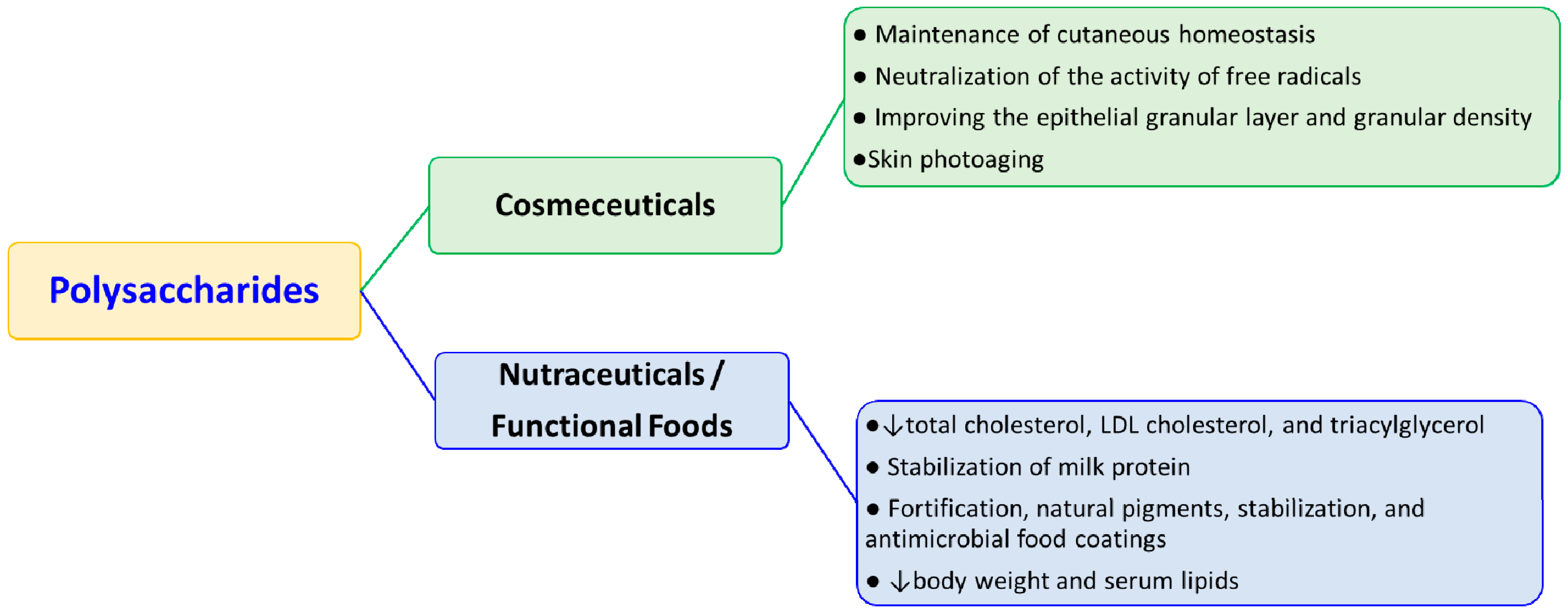
2. Cosmeceutical Applications
3. Nutraceutical Applications
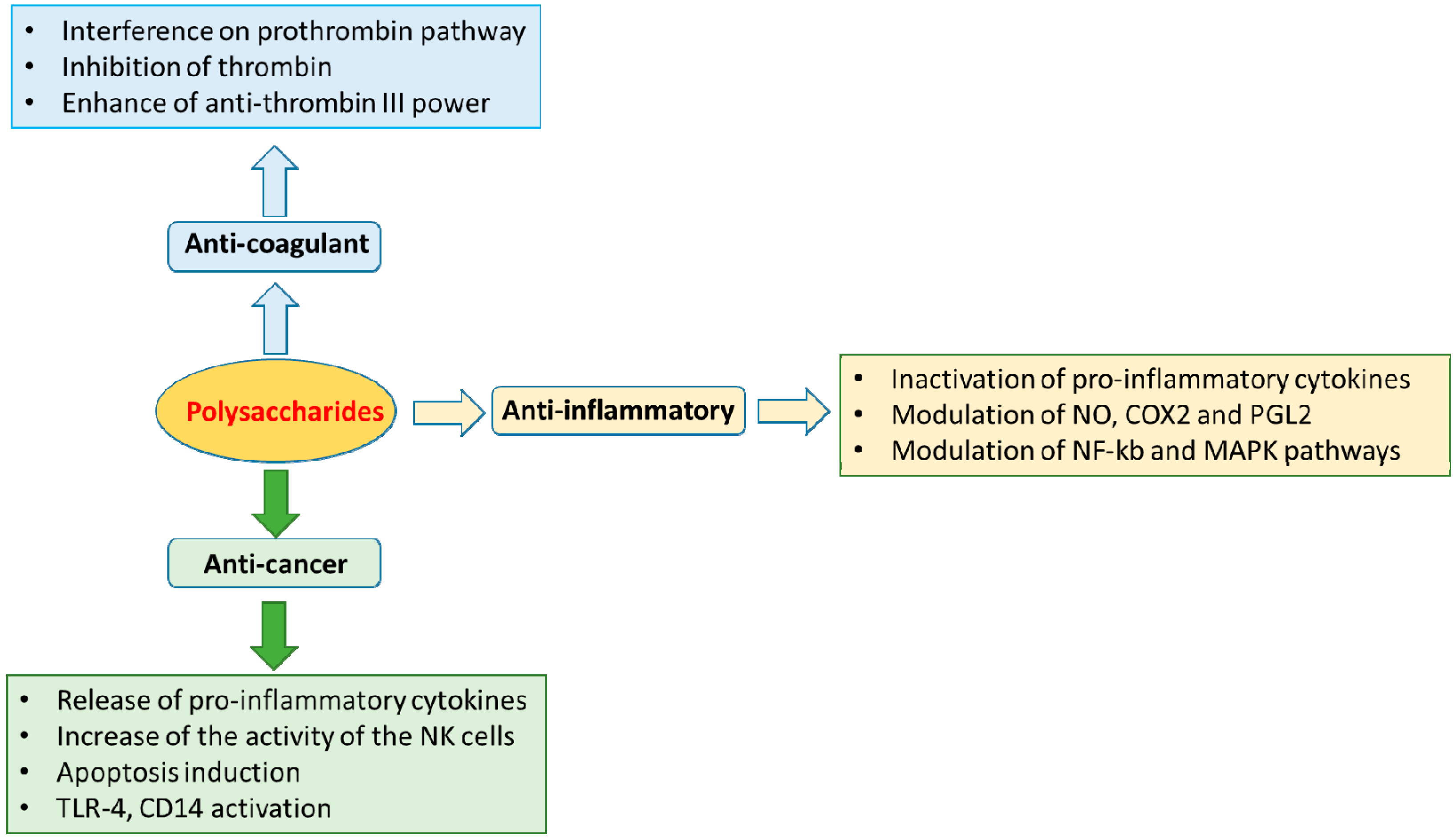
4. Pharmacological Applications
4.1. Anti-Inflammatory and Immunomodulatory Activities
4.2. Anti-Coagulant Activities
4.3. Anti-Cancer Effects
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Aneiros, A.; Garateix, A. Bioactive peptides from marine sources: Pharmacological properties and isolation procedures. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2004, 15, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, R.S.; Morrissey, M.T. Marine biotechnology for production of food ingredients. Adv. Food Nutr. Res. 2007, 52, 237–292. [Google Scholar] [PubMed]
- Vasta, G.R.; Ahmed, H. Animal lectins as cell surface receptors: Current status for invertebrate species signaling mechanisms in protozoa and invertebrates. In Progress in Molecular and Subcellular Biology; Springer: Berlin, Germany, 1996; Volume 17, pp. 158–182. [Google Scholar]
- Kilpatrick, D.C. Animal lectins: A historical introduction and overview. Biochim. Biophys. Acta 2002, 1572, 187–197. [Google Scholar] [CrossRef]
- Sharon, N.; Lis, H. History of lectins: From hemagglutinins to biological recognition molecules. Glycobiology 2004, 14, 53R–62R. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.J.; Desbois, A.P.; Dyrynda, E.A. Conventional and unconventional antimicrobials from fish, marine invertebrates and micro-algae. Mar. Drugs 2010, 8, 1213–1262. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.E. Innate host defense mechanisms of fish against viruses and bacteria. Dev. Comp. Immunol. 2001, 25, 827–839. [Google Scholar] [CrossRef]
- Montaser, R.; Luesch, H. Marine natural products: A new wave of drugs? Future Med. Chem. 2011, 3, 1475–1489. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2012, 29, 144–222. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2014, 31, 160–258. [Google Scholar] [CrossRef] [PubMed]
- Khodse, V.B.; Fernandes, L.; Bhosle, N.B.; Sardessai, S. Carbohydrates, uronic acids and alkali extractable carbohydrates in contrasting marine and estuarine sediments: Distribution, size fractionation and partial chemical characterization. Org. Geochem. 2008, 39, 265–283. [Google Scholar] [CrossRef]
- Pakulski, J.D.; Benner, R. Abundance and distribution of carbohydrates in the ocean. Limnol. Oceanogr. 1994, 39, 930–940. [Google Scholar] [CrossRef]
- Decho, A.W. Microbial exopolymer secretions in the ocean environments: Their role(s) in food webs and marine processes. Oceanogr. Mar. Biol. Ann. Rev. 1990, 28, 73–153. [Google Scholar]
- Sinninghe Damstè, J.S.; Kok, M.D.; Köster, J.; Schouten, S. Sulfurized carbohydrates: An important sedimentary sink for organic carbon. Earth Planet. Sci. Lett. 1998, 164, 7–13. [Google Scholar] [CrossRef]
- Skoog, A.; Benner, R. Aldoses in various size fractions of marine organic matter: Implications for carbon cycling. Limnol. Oceanogr. 1997, 42, 1803–1813. [Google Scholar] [CrossRef]
- Borch, N.H.; Kirchman, D.L. Concentration and composition of dissolved combined neutral sugars (polysaccharides) in seawater determined by HPLC-PAD. Mar. Chem. 1997, 57, 85–95. [Google Scholar] [CrossRef]
- Bhosle, N.B.; Bhaskar, P.V.; Ramachandran, S. Abundance of dissolved polysaccharides in the oxygen minimum layer of northern Indian Ocean. Mar. Chem. 1998, 63, 171–182. [Google Scholar] [CrossRef]
- Mecozzi, M.; Amici, M.; Pietrantonio, E.; Romanelli, G. An ultrasound assisted extraction of the available humic substance from marine sediments. Ultrason. Sonochem. 2002, 9, 11–18. [Google Scholar] [CrossRef]
- Unger, D.; Ittekkot, V.; Schäfer, P.; Tiemann, J. Biogeochemistry of particulate organic matter from the Bay of Bengal as discernible from hydrolysable neutral carbohydrates and amino acids. Mar. Chem. 2005, 96, 155–184. [Google Scholar] [CrossRef]
- Wang, D.; Henrichs, S.M.; Guo, L. Distributions of nutrients, dissolved organic carbon and carbohydrates in the western Arctic Ocean. Cont. Shelf Res. 2006, 26, 1654–1667. [Google Scholar] [CrossRef]
- Khodse, V.B.; Fernandes, L.; Gopalakrishna, V.V.; Bhosle, N.B.; Fernandes, V.; Matondkar, S.G.P.; Bhushan, R. Distribution and seasonal variation of concentrations of particulate carbohydrates and uronic acids in the northern Indian Ocean. Mar. Chem. 2007, 103, 327–346. [Google Scholar] [CrossRef]
- Hazdija, O.; Juracic, M.; Luic, M.; Tonkovic, M.; Jericevic, B. The carbohydrates in relation to mineralogic and granulometric composition of surface sediments in the Karst estuary (River Krka estuary, Yugoslavia). Estuar. Coast. Shelf Sci. 1985, 21, 701–709. [Google Scholar]
- Handa, N.; Tominaga, H. A detailed analysis of carbohydrates in marine particulate matter. Mar. Biol. 1969, 2, 228–235. [Google Scholar] [CrossRef]
- Levine, R. Monosaccharides in health and disease. Annu. Rev. Nutr. 1986, 6, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Fu, Z.-M.; Han, C.C. The potential applications of marine bioactives against diabetes and obesity. Am. J. Mar. Sci. 2014, 2, 1–8. [Google Scholar] [CrossRef]
- Kligman, A.M. Cosmetics A dermatologists looks to the future: Promises and problems. Dermatol. Clin. 2000, 18, 699–709. [Google Scholar] [CrossRef]
- Kim, S.-K. Marien cosmeceuticals. In Trends and Prospects; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Chen, Z.; Seo, J.Y.; Kim, Y.K.; Lee, S.R.; Kim, K.H.; Cho, K.H.; Eun, H.C.; Chung, J.H. Heat modulation of tropoelastin, fibrillin-1, and matrix metalloproteinase-12 in human skin in vivo. J. Investig. Dermatol. 2005, 124, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Epstein, H. Cosmeceutical vehicles. Clin. Dermatol. 2009, 27, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.B.; Adel, M.; Karimi, P.; Peidayesh, M. Pharmaceutical, cosmeceutical, and traditional applications of marine carbohydrates. Adv. Food Nutr. Res. 2014, 73, 197–220. [Google Scholar] [PubMed]
- Kim, S.-K. Marien cosmeceuticals. J. Cosm. Dermatol. 2014, 13, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Ravichandran, Y.D.; Khan, S.B.; Kim, Y.T. Prospective of the cosmeceuticals derived from marine organisms. Biotechnol. Bioprocess Eng. 2008, 13, 511–523. [Google Scholar] [CrossRef]
- Ito, I.; Osaki, T.; Ifuku, S.; Saimoto, H.; Takamori, Y.; Kurozumi, S.; Imagawa, T.; Azuma, K.; Tsuka, T.; Okamoto, Y.; et al. Evaluation of the effects of chitin nanofibrils on skin function using skin models. Carbohydr. Polym. 2014, 101, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Draelos, Z.T. Cosmeceuticals: Procedures in Cosmetic Dermatology Series; Elsevier Sauders: Philadelphia, PA, USA, 2014. [Google Scholar]
- Calderón Muñoz, L.; Lecumberri Lima, E.; Expósito Harris, R.; López Mengíbar, M.A.; Acosta Contreras, N.; Heras Caballero, A.M. Chemical properties of chitosan as a marine cosmeceutical. Mar. Cosmeceut. 2011, 39–50. [Google Scholar] [CrossRef]
- Zhemning, C.; Yan, F. Exopolysaccharides from marine bacteria. J. Ocean Univ. China 2005, 4, 67–74. [Google Scholar]
- Delbarre-Ladrat, C.; Sinquin, C.; Lebellenger, L.; Zykwinska, A.; Colliec-Jouault, S. Exopolysaccharides produced by marine bacteria and their applications as glycosaminoglycan-like molecules. Front. Chem. 2014, 2, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, W.; Jeon, Y.J. Biological activities and potential industrial applications of fucose rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Moon, H.J.; Lee, S.H.; Ku, M.J.; Yu, B.C.; Jeon, M.J.; Jeong, S.H.; Stonil, V.A.; Zvyagintseva, T.N.; Ermakova, S.P.; Lee, Y.H. Fucoidan inhibits UVB-induced MMP-1 promoter expression and down regulation of type I procollagen synthesis in human skin fibroblasts. Eur. J. Dermatol. 2009, 19, 129–134. [Google Scholar] [PubMed]
- Thomas, N.V.; Kim, S.K. Beneficial effects of marine algal compounds in cosmeceuticals. Mar. Drugs 2013, 11, 146–164. [Google Scholar] [CrossRef] [PubMed]
- Senni, K.; Gueniche, F.; Bertaud, A.F.; Tchen, S.I.; Fioretti, F.; Jouault, S.C.; Durand, P.; Guezennec, J.; Godeau, G.; Letourneur, D. Fucoidan a sulfated polysaccharide from brown algae is a potent modulator of connective tissue proteolysis. Arch. Biochem. Biophys. 2006, 445, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Belford, D.A.; Hendry, I.A.; Parish, C.R. Investigation of the ability of several naturally occurring and synthetic polyanions to bind to and potentiate the biological activity of acidic fibroblast growth factor. J. Cell. Physiol. 1993, 157, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.B.A.; Taha, R.M. Marine phytochemical compounds and their cosmeceuticals applications. In Marine Cosmeceuticals: Trends and Prospects; Kim, S., Ed.; CRC Press: Boca Raton, FL, USA, 2011; pp. 51–61. [Google Scholar]
- Xue, C.; Yu, G.; Hirata, T.; Terao, J.; Lin, H. Antioxidative activities of several marine polysaccharides evaluated in a phosphatidylcholine-liposomal suspension and organic solvents. Biosci. Biotechol. Biochem. 1998, 62, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Carvalho, A.; Vaz, D.; Gil, M.; Mendes, A.; Bartolo, P. Development of novel alginate based hydrogel films for wound healing applications. Int. J. Biol. Macromol. 2013, 52, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Pallela, R.; Park, I.K.P.K. Nutraceutical and pharmacological implications of marine carbohydrates. Adv. Food Nutr. Res. 2014, 73, 183–195. [Google Scholar] [PubMed]
- Vo, T.S.; Ngo, D.H.; Kang, K.H.; Jung, W.K.; Kim, S.K. The beneficial properties of marine polysaccharides in alleviation of allergic responses. Mol. Nutr. Food Res. 2015, 59, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Mhurchu, C.N.; Poppitt, S.; McGill, A.; Leahy, F.; Bennett, D.; Lin, R.B.; Ormrod, D.; Ward, L.; Strik, C.; Rodgers, A. The effect of the dietary supplement, Chitosan, on body weight: A randomised controlled trial in 250 overweight and obese adults. Int. J. Obes. 2004, 28, 1149–1156. [Google Scholar] [CrossRef] [PubMed]
- Ngo, D.H. Sulphated polysaccharides as bioactive agents from marine algae. Int. J. Biol. Macromol. 2013, 62, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Pleissner, D.; Lin, C.S. Valorisation of food waste in biotechnological processes. Sustain. Chem. Processes 2013, 1. [Google Scholar] [CrossRef]
- Archer, M.; Watson, R.; Garret, A.; Large, M. Strategic Framework for Seafood Waste Management; Seafish Report Number SR574; The Sea Fish Industry Authority: Edinburgh, UK, 2005. [Google Scholar]
- Ghosh, T.; Chattopadhyay, K.; Marschall, M.; Karmakar, P.; Mandal, P.; Ray, B. Focus on antivirally active sulfated polysaccharides: From structure-activity analysis to clinical evaluation. Glycobiology 2009, 19, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Kraan, S. Algal polysaccharides, novel applications and outlook. In Carbohydrates—Comprehensive Studies on Glycobiology and Glycotechnology; Chang, C.-F., Ed.; InTech: Vienna, Austria, 2012; pp. 459–532. [Google Scholar]
- Amano, H.; Kakinuma, M.; Coury, D.A.; Ohno, H.; Hara, T. Effect of a seaweed mixture on serum lipid level and platelet aggregation in rats. Fish. Sci. 2005, 71, 1160–1166. [Google Scholar] [CrossRef]
- Pasquin, P. Functional and Specialty Beverage Technology; Woodhead Publishing: Boca Raton, FL, USA, 2009. [Google Scholar]
- Shah, V.; Ray, A.; Ray, N.; Madamwar, D. Characterization of the extracellular polysaccharide produced by a marine cyanobacterium, Cyanothece sp. ATCC51142, and its exploitation toward metal removal from solutions. Curr. Microbiol. 2000, 40, 274–278. [Google Scholar]
- Freile-Pelegrín, Y.; Murano, E. Agars from three species of Gracilaria (Rhodophyta) from Yucatán Peninsula. Bioresour. Technol. 2005, 96, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Suleria, H.A.R.; Osborne, S.; Masci, P.; Gobe, G. Marine-based nutraceuticals: An innovative trend in the food and supplement industries. Mar. Drugs 2015, 13, 6336–6351. [Google Scholar] [CrossRef] [PubMed]
- De Jesus Raposo, M.F.; de Morais, A.M.B.; Santos Costa de Morais, R.M. Marine polysaccharides from algae with potential biomedical applications. Mar. Drugs 2015, 13, 2967–3028. [Google Scholar] [CrossRef] [PubMed]
- Raposo, M.F.J.; Morais, A.M.M.B.; Morais, R.M.S.C. Influence of sulphate on the composition and antibacterial and antiviral properties of the exopolysaccharide from Porphyridium cruentum. Life Sci. 2014, 101, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Liu, W.S.; Han, B.Q.; Sun, Y.Y. Antidiabetic effects of chitooligosaccharideson pancreatic islet cells in streptozotocin-induced diabetic rats. World J. Gastroenterol. 2007, 13, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.N.; Joo, E.S.; Kim, K.I.; Kim, S.K.; Yang, H.P.; Jeon, Y.J. Effect of chitosanoligosaccharides on cholesterol level and antioxidant enzyme activities in hypercholesterolemic rat. J. Korean Soc. Food Sci. Nutr. 2005, 34, 36–41. [Google Scholar]
- Cho, E.J.; Rahman, A.; Kim, S.W.; Baek, Y.M.; Hwang, H.J.; Oh, J.Y.; Hwang, H.S.; Lee, S.H.; Yun, J.W. Chitosanoligosaccharides inhibit adipogenesis in 3 T3-L1 adipocytes. J. Microbiol. Biotechnol. 2008, 18, 80–87. [Google Scholar] [PubMed]
- Xia, W.; Liu, P.; Zhang, J.; Chen, J. Biological activities of chitosan and chitooligosaccharides. Food Hydrocoll. 2010, 25, 170–179. [Google Scholar] [CrossRef]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef] [PubMed]
- Laurienzo, P. Marine polysaccharides in pharmaceutical applications: An overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, I.R.L.; Cordeiro, S.L.; Gomes, D.L.; Dreyfuss, J.L.; Filgueira, L.G.A.; Leite, E.L.; Nader, H.B.; Rocha, H.A.O. Evaluation of anti-nociceptive and anti-inflammatory activities of a heterofucan from Dictyota menstrualis. Mar. Drugs 2013, 11, 2722–2740. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.J.; Moon, J.Y.; Kim, S.S.; Yang, K.W.; Lee, W.J.; Lee, N.H.; Hyun, C.G. Jeju seaweeds suppress lipopolysaccharide-stimulated proinflammatory response in RAW264.7 murine macrophages. Asian Pac. J. Trop. Biomed. 2014, 4, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Gao, Y.; Xing, Y.; Zhu, H.; Shen, J.; Tian, J. Fucoidan, a sulfated polysaccharide from brown algae, against myocardial ischemia-reperfusion injury in rats via regulating the inflammation response. Food Chem. Toxicol. 2011, 49, 2090–2095. [Google Scholar] [CrossRef] [PubMed]
- Leiro, J.M.; Castro, R.; Arranz, J.A.; Lamas, J. Immunomodulating activities of acidic sulphated polysaccharides obtained from the seaweed Ulva rigida C. Agardh. Int. Immunopharmacol. 2007, 7, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.Y.; Yim, J.H.; Lee, H.K.; Pyo, S. Activation of murine peritoneal macrophages by sulphated exopolysaccharide from marine microalga Gyrodinium impudicum (strain KG03): Involvement of the NF-kappa B and JNK pathway. Int. Immunopharmacol. 2006, 6, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Maruyamaa, H.; Tamauchib, H.; Hashimotoc, M.; Nakano, T. Suppression of Th2 immune responses by Mekabu fucoidan from Undaria pinnatifida sporophylls. Int. Arch. Allergy Immunol. 2005, 137, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Guzman, S.; Gato, A.; Lamela, M.; Freire-Garabal, M.; Calleja, J.M. Anti-Inflammatory and immunomodulatory activities of polysaccharide from Chlorella stigmatophora and Phaeodactylum tricornutum. Phytother. Res. 2003, 17, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Tabarsa, M.; Park, G.M.; Shin, I.S.; Lee, E.; Kim, J.K.; You, S. Structure-activity relationships of sulphated glycoproteins from Codium fragile on nitric oxide releasing capacity from RAW264.7 cells. Mar. Biotechnol. 2015, 17, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wu, X.Z.; Wen, Z.Y. Sulfated polysaccharides and immune response: Promoter or inhibitor? Panminerva Med. 2008, 50, 177–183. [Google Scholar] [PubMed]
- Tsuji, R.F.; Hoshino, K.; Noro, Y.; Tsuji, N.M.; Kurokawa, T.; Masuda, T.; Akira, S.; Nowak, B. Suppression of allergic reaction by lambda-carrageenan: Toll-like receptor 4/MyD88-dependent and -independent modulation of immunity. Clin. Exp. Allergy 2003, 33, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Shibata, Y.; Foster, L.; Bradfield, J.F.; Myrvik, Q.N. Oral administration of chitin down-regulates serum IgE levels and lung eosinophilia in the allergic mouse. J. Immunol. 2000, 164, 1314–1321. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.A. Chitins and chitosans for the repair of wounded skin, nerve, cartilage and bone. Carbohydr. Polym. 2009, 76, 167–182. [Google Scholar] [CrossRef]
- Malaguarnera, L.; Musumeci, M.; di Rosa, M.; Scuto, A.; Musumeci, S. Interferon-gamma, tumor necrosis factor-alpha, and lipopolysaccharide promote chitotriosidase gene expression in human macrophages. J. Clin. Lab. Anal. 2005, 19, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Di Rosa, M.; Musumeci, M.; Scuto, A.; Musumeci, S.; Malaguarnera, L. Effect of interferon-γ, interleukin-10, lipopolysaccharide and tumor necrosis factor-α on chitotriosidase synthesis in human macrophages. Clin. Chem. Lab. Med. 2005, 43, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.; Laha, B.; Maiti, S. Boswellia gum resin/chitosan polymer composites: Controlled delivery vehicles for aceclofenac. Int. J. Biol. Macromol. 2015, 77, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, R.M.; Pereira, A.C.; Pereira, I.O.; Oliveira, M.J.; Barbosa, M.A. Macrophage response to chitosan/poly-(γ-glutamic acid) nanoparticles carrying an anti-inflammatory drug. J. Mater. Sci. Mater. Med. 2015, 26. [Google Scholar] [CrossRef] [PubMed]
- Muto, S.; Niimura, K.; Oohara, M.; Oguchi, Y.; Matsunaga, K.; Hirose, K.; Kakuchi, J.; Sugita, N.; Furusho, T. Polysaccharides from Marine Algae and Antiviral Drugs Containing the Same as Active Ingredients. European Patent EP 295,956, 21 December 1988. [Google Scholar]
- Wang, W.; Wang, S.-X.; Guan, H.-S. The antiviral activities and mechanisms of marine polysaccharides: An overview. Mar. Drugs 2012, 10, 2795–2816. [Google Scholar] [CrossRef] [PubMed]
- De Jesus Raposo, M.F.; de Morais, A.M.M.B.; de Morais, R.M.S.C. Bioactivity and applications of polysaccharides from marine microalgae. In Polysaccharides: Bioactivity and Biotechnology; Merillon, J.-M., Ramawat, K.G., Eds.; Springer: Cham, Switzerland, 2014. [Google Scholar]
- Matsubara, K.; Matsuura, Y.; Bacic, A.; Liao, M.L.; Hori, K.; Miyazawa, K. Anticoagulant properties of a sulfated galactan preparation from a marine green alga, Codium cylindricum. Int. J. Biol. Macromol. 2001, 28, 395–399. [Google Scholar] [CrossRef]
- Mao, W.; Li, H.; Li, Y.; Zhang, H.; Qi, X.; Sun, H.; Chen, Y.; Guo, S. Chemical characteristic and anticoagulant activity of the sulfated polysaccharide isolated from Monostroma latissimum (Chlorophyta). Int. J. Biol. Macromol. 2009, 44, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Maeda, M.; Uehara, T.; Harada, N.; Sekiguchi, M.; Hiraoka, A. Heparinoid-active sulphated polysaccharide from Monostroma-nitidum and their distribution in the Chlorophyta. Phytochemistry 1991, 30, 3611–3614. [Google Scholar] [CrossRef]
- Li, H.; Mao, W.; Hou, Y.; Gao, Y.; Qi, X.; Zhao, C.; Chen, Y.; Chen, Y.; Li, N.; Wang, C. Preparation, structure and anticoagulant activity of a low molecular weight fraction produced by mild acid hydrolysis of sulfated rhamnan from Monostroma latissimum. Bioresour. Technol. 2012, 114, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Wijesekara, I.; Pangestuti, R.; Kim, S.K. Biological activities and potential health benefits of sulfated polysaccharides derived from marine algae. Carbohydr. Polym. 2011, 84, 14–21. [Google Scholar] [CrossRef]
- Nishino, T.; Yokoyama, G.; Dobahi, K. Isolation, purification and characterization of fucose-containing sulfated polysaccharides from the brown seaweed Ecklonia kurome and their blood-anticoagulant activities. Carbohydr. Res. 1989, 186, 119–129. [Google Scholar] [CrossRef]
- Silva, F.R.F.; Dore, C.M.P.G.; Marques, C.T.; Nascimento, M.S.; Benevides, N.M.B.; Rocha, H.A.O.; Chavante, S.F.; Leite, E.L. Anticoagulant activity, paw edema and pleurisy induced carrageenan: Action of major types of commercial carrageenans. Carbohydr. Polym. 2010, 79, 26–33. [Google Scholar] [CrossRef]
- Chandía, N.P.; Matsuhiro, B. Characterization of a fucoidan from Lessonia vadosa (Phaeophyta) and its anticoagulant and elicitor properties. Int. J. Biol. Macromol. 2008, 42, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Pomin, V.H.; Pereira, M.S.; Valente, A.P.; Tollefsen, D.M.; Pavao, M.S.G.; Mourao, P.A.S. Selective cleavage and anticoagulant activity of a sulfated fucan: Stereospecific removal of a 2-sulfate ester from the polysaccharide by mild acid hydrolysis, preparation of oligosaccharides, and heparin cofactor II-dependent anticoagulant activity. Glycobiology 2005, 15, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, M.; Mody, K.H. Heparinoid-active sulphated polysaccharides from marine algae as potential anticoagulant agents. Curr. Sci. 2000, 79, 1672–1683. [Google Scholar]
- Wang, L.; Wang, X.; Wu, H.; Liu, R. Overview on biological activities and molecular characteristics of sulfated polysaccharides from marine green algae in recent years. Mar. Drugs 2014, 12, 4984–5020. [Google Scholar] [CrossRef] [PubMed]
- Rocha, H.A.O.; Moraes, F.A.; Trindade, E.S.; Franco, C.R.C.; Torquato, R.J.S.; Veiga, S.S.; Valente, A.P.; Mourão, P.A.; Leite, E.L.; Nader, H.B.; et al. Structural and haemostatic activities of a sulfated galactofucan from the brown alga Spatoglossum schröederi. An ideal antithrombotic agent? J. Biol. Chem. 2005, 280, 41278–41288. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, Y.; Hayashi, T.; Hayashi, K.; Osawa, T.; Niiya, K.; Sakuragawa, N. Activation of heparin cofactor II by calcium spirulan. J. Biol. Chem. 2000, 275, 11379–11382. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Luo, K.; Li, D.; Yu, S.; Cai, J.; Chen, L.; Du, Y. Preparation, characterization and in vitro anticoagulant activity of highly sulfated chitosan. Int. J. Biol. Macromol. 2013, 52, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Ilium, L. Chitosan and its use as a pharmaceutical excipient. Pharm. Res. 1998, 15, 1326–1331. [Google Scholar] [CrossRef]
- Sayari, N.; Sila, A.; Abdelmalek, B.E.; Abdallah, R.B.; Ellouz-Chaabouni, S.; Bougatef, A.; Balti, R. Chitin and chitosan from the Norway lobster by-products: Antimicrobial and anti-proliferative activities. Int. J. Biol. Macromol. 2016, 87, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Muanprasat, C.; Wongkrasant, P.; Satitsri, S.; Moonwiriyakit, A.; Pongkorpsakol, P.; Mattaveewong, T.; Pichyangkura, R.; Chatsudthipong, V. Activation of AMPK by chitosan oligosaccharide in intestinal epithelial cells: Mechanism of action and potential applications in intestinal disorders. Biochem. Pharmacol. 2015, 96, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.P.; Birundha, K.; Kaveri, K.; Devi, K.T. Antioxidant studies of chitosan nanoparticles containing naringenin and their cytotoxicity effects in lung cancer cells. Int. J. Biol. Macromol. 2015, 78, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Choi, K.C.; Kang, M.S.; Oh, J.S.; Jeong, Y.I.; Lee, H.C. Self-organized nanoparticles of caffeic acid conjugated polysaccharide and its anticancer activity. J. Nanosci. Nanotechnol. 2015, 15, 1130–1134. [Google Scholar] [CrossRef] [PubMed]
- Mazzarino, L.; Loch-Neckel, G.; Bubniak Ldos, S.; Mazzucco, S.; Santos-Silva, M.C.; Borsali, R.; Lemos-Senna, E. Curcumin-loaded chitosan-coated nanoparticles as a new approach for the local treatment of oral cavity cancer. J. Nanosci. Nanotechnol. 2015, 15, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Teruya, T.; Konishi, T.; Uechi, S.; Tamaki, H.; Tako, M. Anti-proliferative activity of oversulfated fucoidan from commercially cultured Cladosiphon okamuranus Tokida in U937 cells. Int. J. Biol. Macromol. 2007, 41, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Heneji, K.; Matsuda, T.; Tomita, M.; Kawakami, H.; Ohshiro, K.; Masuda, M.; Takasu, N.; Tanaka, Y.; Ohta, T.; Mori, N. Fucoidan extracted from Cladosiphon okamuranus tokida induces apoptosis of human T-cell leukemia virus type 1-infected T-cell lines and primary adult T-cell leukemia cells. Nutr. Cancer 2005, 52, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Khotimchenko, Y.S. Antitumor properties of non-starch polysaccharides: Fucoidans and Chitosans. Russ. J. Mar. Biol. 2010, 36, 321–330. [Google Scholar] [CrossRef]
- Atashrazm, F.; Lowenthal, R.M.; Woods, G.M.; Holloway, A.F.; Dickinson, J.L. Fucoidan and cancer: A multifunctional molecule with anti-tumor potential. Mar. Drugs 2015, 13, 2327–2346. [Google Scholar] [CrossRef] [PubMed]
- Ale, M.T.; Maruyama, H.; Tamauchi, H.; Mikkelsen, J.D.; Meyer, A.S. Fucoidan from Sargassum sp. and Fucus vesiculosus reduces cell viability of lung carcinoma and melanoma cells in vitro and activates natural killer cells in mice in vivo. Int. J. Biol. Macromol. 2011, 49, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Park, S.Y.; Lee, J.Y.; Park, J.H. Fucoidan present in brown algae induces apoptosis of human colon cancer cells. BMC Gastroenterol. 2010, 10, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki-Miyamoto, Y.; Yamasaki, M.; Tachibana, H.; Yamada, K. Fucoidan induces apoptosis through activation of caspase-8 on human breast cancer MCF-7 cells. J. Agric. Food Chem. 2009, 57, 8677–8682. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.S.; Jung, W.K.; Kim, J.A.; Choi, I.W.; Kim, S.K. Beneficial effects of fucoidan on osteoblastic MG-63 cell differentiation. Food Chem. 2009, 116, 990–994. [Google Scholar] [CrossRef]
- Ji, H.; Shao, H.; Zhang, C.; Hong, P.; Xiong, H. Separation of the polysaccharides in Caulerpa racemosa and their chemical composition and antitumor activity. J. Appl. Polym. Sci. 2008, 110, 1435–1440. [Google Scholar] [CrossRef]
- Maruyama, H.; Tamauchi, H.; Iizuka, M.; Nakano, T. The role of NK cells in antitumor activity of dietary fucoidan from Undaria pinnatifida sporophylls (Mekabu). Planta Med. 2006, 72, 1415–1417. [Google Scholar] [CrossRef] [PubMed]
- Teruya, T.; Tatemoto, H.; Konishi, T.; Tako, M. Structural characteristics and in vitro macrophage activation of acetyl fucoidan from Cladosiphon okamuranus. Glycoconj. J. 2009, 26, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Raghavendran, H.R.; Srinivasan, P.; Rekha, S. Immunomodulatory activity of fucoidan against aspirin-induced gastric mucosal damage in rats. Int. Immunopharmacol. 2011, 11, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Shibata, H.; Iimuro, M.; Uchiya, N.; Kawamori, T.; Nagaoka, M.; Ueyama, S.; Hashimoto, S.; Yokokura, T.; Sugimura, T.; Wakabayashi, K. Preventive effects of Cladosiphon fucoidan against Helicobacter pylori infection in Mongolian gerbils. Helicobacter 2003, 8, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Foley, S.A.; Szegezdi, E.; Mulloy, B.; Samali, A.; Tuohy, M.G. An unfractionated fucoidan from Ascophyllum nodosum: Extraction, characterization, and apoptotic effects in vitro. J. Nat. Prod. 2011, 74, 1851–1861. [Google Scholar] [CrossRef] [PubMed]
- Miao, H.Q.; Elkin, M.; Aingorn, E.; Ishai-Michaeli, R.; Stein, C.A.; Vlodavsky, I. Inhibition of heparanase activity and tumor metastasis by laminarin sulfate and synthetic phosphorothioate oligodeoxynucleotides. Int. J. Cancer 1999, 83, 424–431. [Google Scholar] [CrossRef]
- Ngoan, B.T.; Hanh, T.T.; Vienle, T.; Diep, C.N.; Thao, N.P.; Thao, T.; Thanh, N.V.; Cuong, N.X.; Nam, N.H.; Thung, C.; et al. Asterosaponins and glycosylated polyhydroxysteroids from the starfish Culcita novaeguineae and their cytotoxic activities. J. Asian Nat. Prod. Res. 2015, 17, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Kim, I.H.; Kim, J.; Nam, T.J. Induction of apoptosis and the regulation of ErbB signaling by laminarin in HT-29 human colon cancer cells. Int. J. Mol. Med. 2013, 32, 291–295. [Google Scholar] [PubMed]
- Park, H.K.; Kim, I.H.; Kim, J.; Nam, T.J. Induction of apoptosis by laminarin, regulating the insulin-like growth factor-IR signaling pathways in HT-29 human colon cells. Int. J. Mol. Med. 2012, 30, 734–738. [Google Scholar] [PubMed]
- Ji, Y.B.; Ji, C.F.; Zhang, H. Laminarin induces apoptosis of human colon cancer LOVO cells through a mitochondrial pathway. Molecules 2012, 17, 9947–9960. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Li, X.; Li, T.; Jiang, P.; Zhang, L.; Wu, M.; Zhang, L. Characterization and anti-tumor activity of alkali-extracted polysaccharide from Enteromorpha intestinalis. Int. Immunopharmacol. 2009, 9, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, I.; Nagumo, T.; Takahashi, M.; Fujihara, M.; Suzuki, Y.; Iizima, N. Antitumor effect of seaweeds, 3. Antitumor effect of an extract from Sargassum kjellmanianum. Jpn. J. Exp. Med. 1981, 51, 187–189. [Google Scholar] [PubMed]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruocco, N.; Costantini, S.; Guariniello, S.; Costantini, M. Polysaccharides from the Marine Environment with Pharmacological, Cosmeceutical and Nutraceutical Potential. Molecules 2016, 21, 551. https://doi.org/10.3390/molecules21050551
Ruocco N, Costantini S, Guariniello S, Costantini M. Polysaccharides from the Marine Environment with Pharmacological, Cosmeceutical and Nutraceutical Potential. Molecules. 2016; 21(5):551. https://doi.org/10.3390/molecules21050551
Chicago/Turabian StyleRuocco, Nadia, Susan Costantini, Stefano Guariniello, and Maria Costantini. 2016. "Polysaccharides from the Marine Environment with Pharmacological, Cosmeceutical and Nutraceutical Potential" Molecules 21, no. 5: 551. https://doi.org/10.3390/molecules21050551
APA StyleRuocco, N., Costantini, S., Guariniello, S., & Costantini, M. (2016). Polysaccharides from the Marine Environment with Pharmacological, Cosmeceutical and Nutraceutical Potential. Molecules, 21(5), 551. https://doi.org/10.3390/molecules21050551







