Aromatic Constituents from the Stems of Astragalus membranaceus (Fisch.) Bge. var. Mongholicus (Bge.) Hsiao
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Compound Characterization
3.5. Acid Hydrolysis of 2 and 3
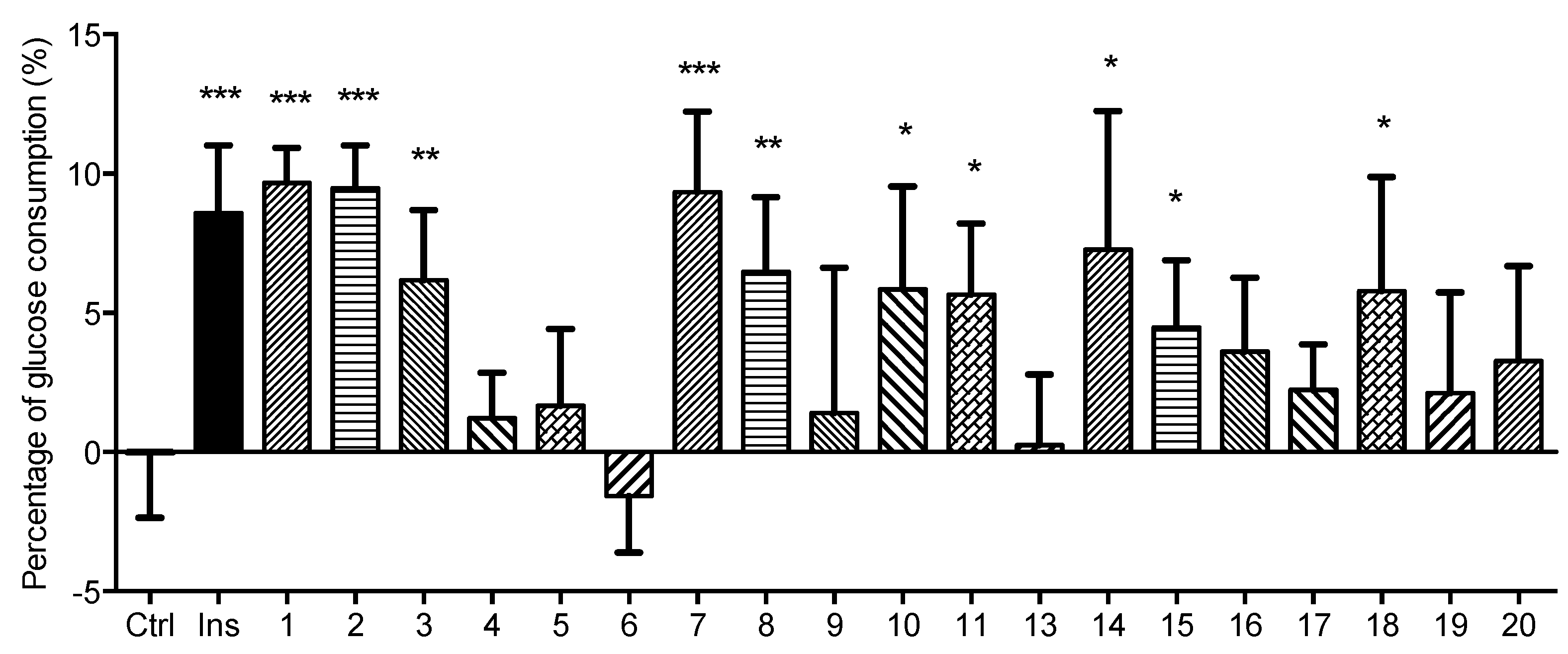
3.6. Glucose Consumption Assay of Compounds Obtained from AM
3.6.1. Materials
3.6.2. Cell Culture and Differentiation
3.6.3. Determination of Glucose Consumption in L6 Myotubes
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, Y.; Li, X.; Ruan, J.; Wang, T.; Dong, Y.; Hao, J.; Liu, E.; Han, L.; Gao, X.; Wang, T. Oleanane type saponins from the stems of Astragalus membranaceus (Fisch.) Bge. var. mongholicus (Bge.) Hsiao. Fitoterapia 2015, 109, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Lu, G.; Tan, X.; Chen, J.; Huang, Z. Chemical constituents of Desmodium caudatum. J. Chin. Med. Mater. 2013, 36, 1953–1956. [Google Scholar]
- Wei, J.; Chang, X.; Wang, W.; Kang, W. Chemical constituents from Lysimachia circaeoides. J. Chin. Med. Mater. 2013, 36, 1441–1443. [Google Scholar]
- Zhao, D.; Li, D.; Wei, J.; Hua, H.; Li, Z. Isolation and structural identification of the chemical constituents from the flowers of Xanthoceras sorbifolia Bunge. J. Shenyang Pharm. Univ. 2012, 29, 514–518. [Google Scholar]
- Xia, X.; Li, W. Chemical constituents of Lespedeza virgate. Chin. J. Exp. Tradit. Med. Formulae 2012, 18, 127–129. [Google Scholar]
- Lu, C.; Zhou, G.; Wang, H.; Li, Y.; Yao, X. Studies on the chemical constituents of Nerviliae fordii. J. Chin. Med. Mater. 2009, 32, 373–375. [Google Scholar]
- Gohar, A.A. Flavonol glycosides from Cadaba glandulosa. Z. Naturforsch C 2002, 57, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Zhou, G.; Wang, H.; Y, W.; Yao, X. Water-soluble constituents from Nerviliae fordii. Li Shizhen Med. Mater. Med. Res. 2010, 21, 3087–3088. [Google Scholar]
- Cai, Q.; Wang, L.; Liu, Y.; Wang, C.; Gu, Y. Study on chemical constituents from leaves of Exochorda serratifolia. Chin. Tradit. Herb. Drugs 2012, 43, 673–675. [Google Scholar]
- Zhang, L.; Zhang, Y.; Chen, Y.; Jia, S.; Dai, J.; Meng, W.; Li, L.; Deng, Y. Isoflavones in leaves of Belamcanda chinensis. Nat. Prod. Res. Dev. 2011, 23, 69–71. [Google Scholar]
- Wang, C.; Tang, Y.; Zhou, L.; Gao, H. A study on flavonoid in barks of Sophora japonica. Jiangsu Zhongyiyao 2008, 40, 65–67. [Google Scholar]
- Zhang, S.; Fan, C.; Wang, L.; Liu, X.; Sun, X.; Ye, W. Chemical constituents of Shenqi Fuzheng Injection. Chin. Tradit. Pat. Med. 2011, 33, 1743–1747. [Google Scholar]
- Song, C.; Zhen, Z.; Liu, D.; Hu, Z. Isoflavones from Astragalus membranaceus. Acta Bot. Sin. 1997, 39, 764–768. [Google Scholar]
- Zheng, W.; Tan, X.Q.; Guo, L.J.; Kong, F.F.; Lu, P.; Ni, D.J.; Wang, P. Chemical constituents from Monochasma savatieri. Chin. J. Nat. Med. 2012, 10, 102–104. [Google Scholar] [CrossRef]
- Gaffield, W. Circular dichroism, optical rotatory dispersion, and absolute configuration of flavanones, 3-hydroxyflavanones, and their glycosides. Determination of aglycone chirality in flavanone glycosides. Tetrahedron 1970, 26, 4093–4108. [Google Scholar] [CrossRef]
- Chu, H.; He, W.; Zhang, Y.; Ji, C.; Tan, N. Flavonoids and nor-sesquiterpenes of Pedicularis densispica. China J. Chin. Mater. Med. 2011, 36, 2672–2675. [Google Scholar]
- Otsuka, H.; Takeuchi, M.; Inoshiri, S.; Sato, T.; Yamasaki, K. Phenolic compounds from Coix lachryma-jobi var. Ma-yuen. Phytochemistry 1989, 28, 883–886. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kim, K.H.; Lee, I.K.; Lee, K.H.; Choi, S.U.; Lee, K.R. A new flavonol glycoside from Hylomecon vernalis. Arch. Pharm. Res. 2012, 35, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Fujii, I.; Ebizuka, Y.; Li, X.; Shimomak, I.S.; Sakano, Y. Flavonoids from the seeds of Sphaerophysa Salsula. J. Asian Nat. Prod. Res. 2004, 6, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.H.; Ren, X.L.; Xu, F.; Ma, X.Q.; Liu, G.X.; Li, C.H.; Li, C.; Cai, S.Q. Absorptive constituents and their metabolites in drug-containing urine samples from Wuzhishan miniature pigs orally administered with Buyang Huanwu decoction. J. Nat. Med. 2014, 68, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, Y.; Wang, S.; Dong, Y.; Wang, T.; Qu, L.; Li, N.; Wang, T. Bioactive constituents from the aerial parts of Lippia triphylla. Molecules 2015, 20, 21946–21959. [Google Scholar] [CrossRef] [PubMed]
- Cheong, S.H.; Furuhashi, K.; Ito, K.; Nagaoka, M.; Yonezawa, T.; Miura, Y.; Yagasaki, K. Daidzein promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in L6 myocytes and improves glucose homeostasis in Type 2 diabetic model mice. J. Nutr. Biochem. 2014, 25, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Minakawa, M.; Miura, Y.; Yagasaki, K. Piceatannol, a resveratrol derivative, promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in L6 myocytes and suppresses blood glucose levels in type 2 diabetic model db/db mice. Biochem. Biophys. Res. Commun. 2012, 422, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Gao, Z.; Liu, D.; Liu, Z.; Ye, J. Berberine improves glucose metabolism through induction of glycolysis. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E148–E156. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.
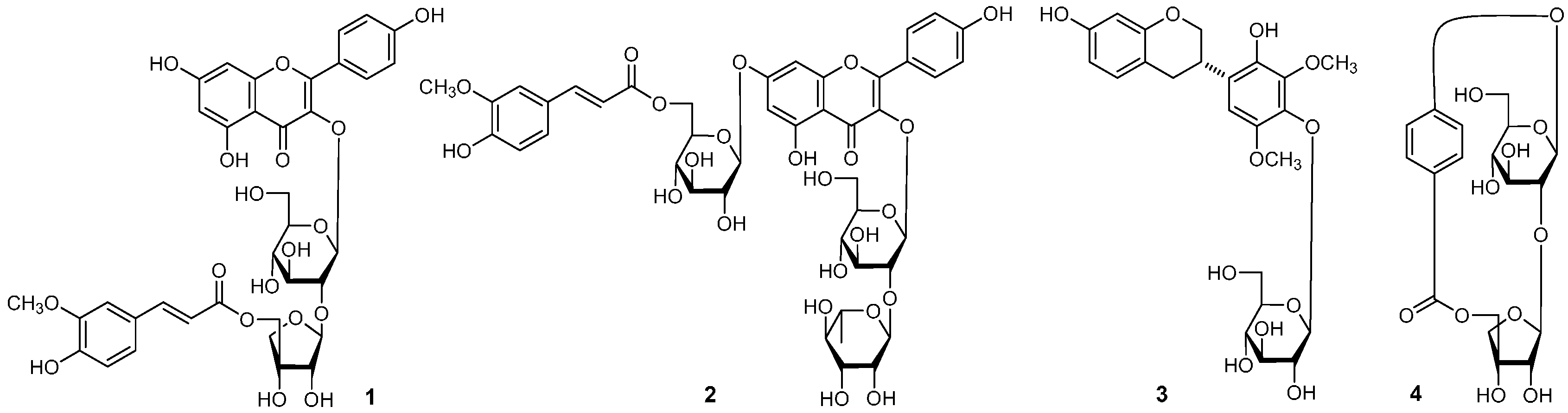
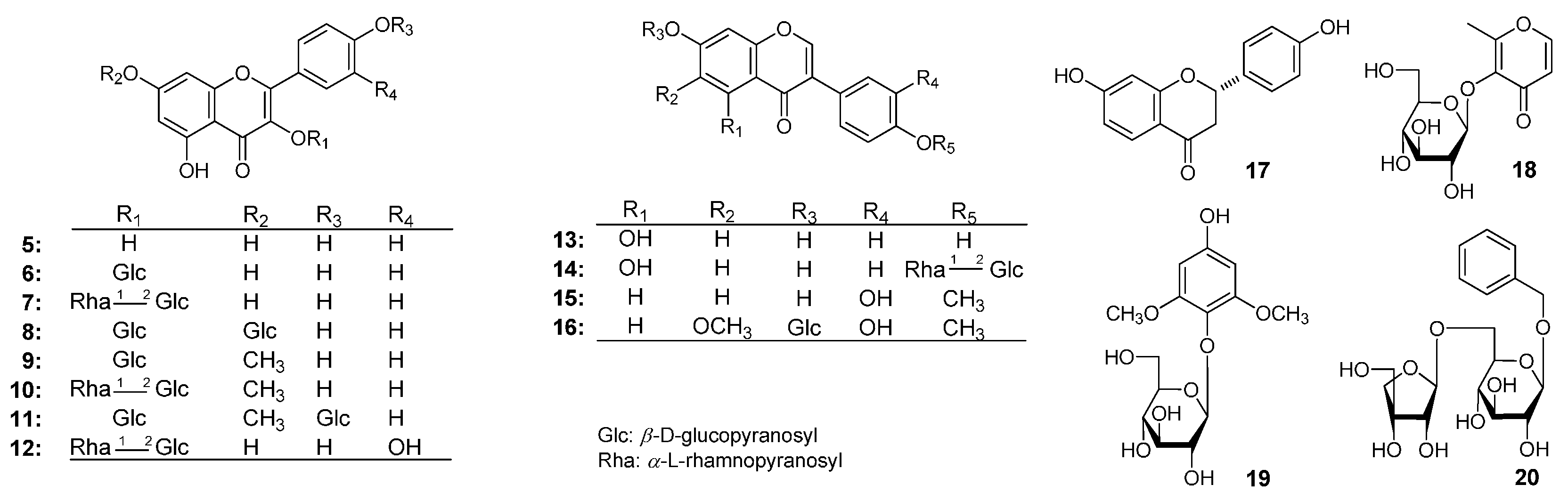
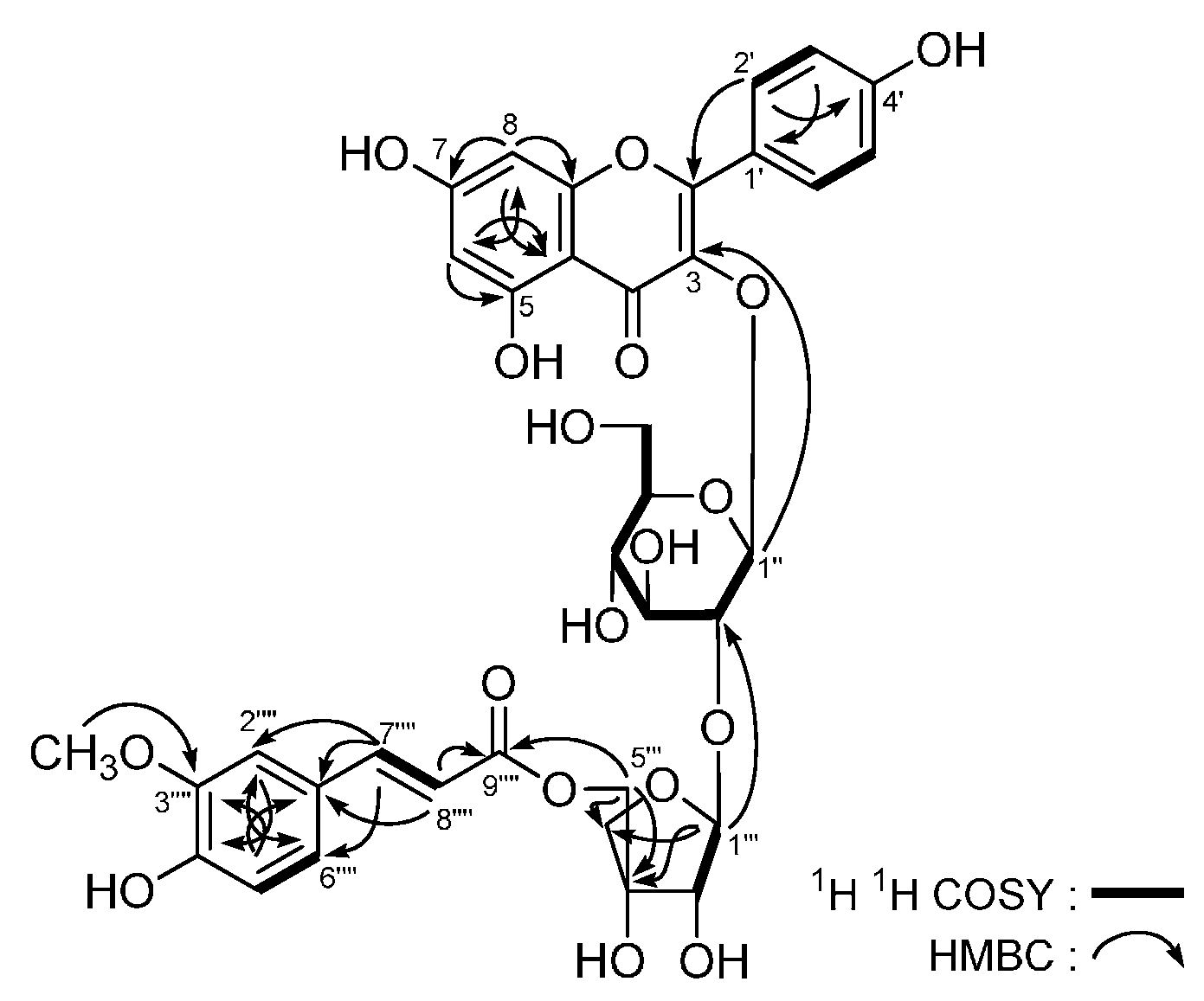
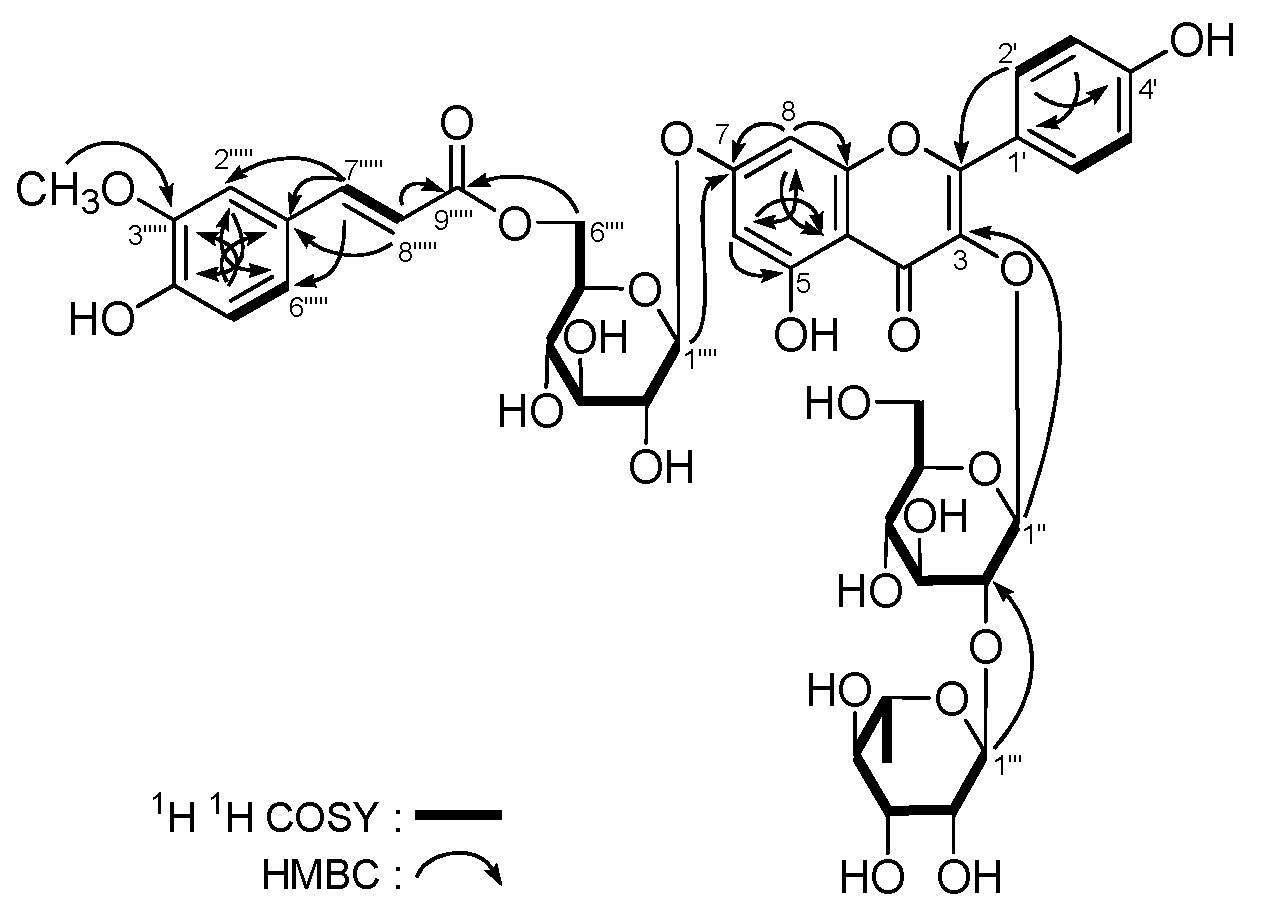
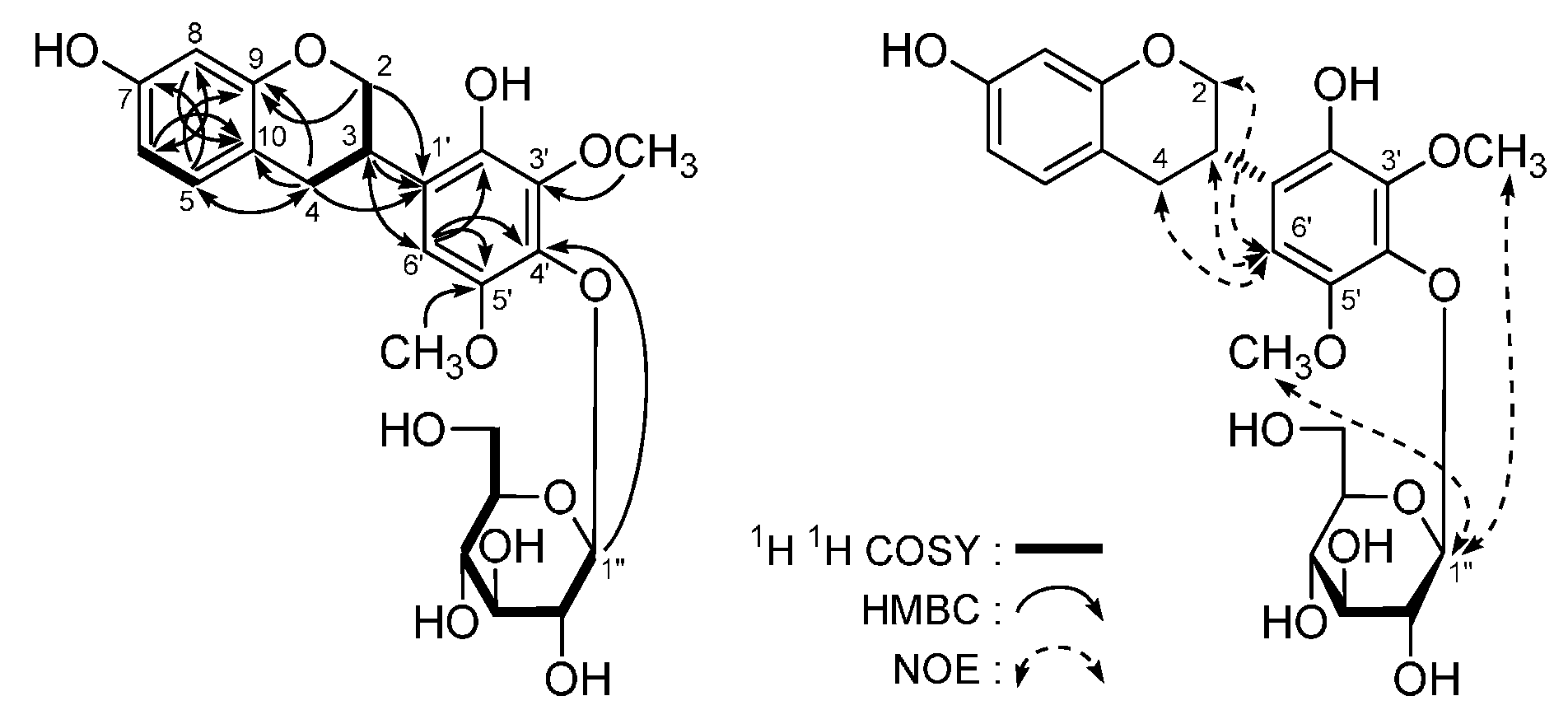
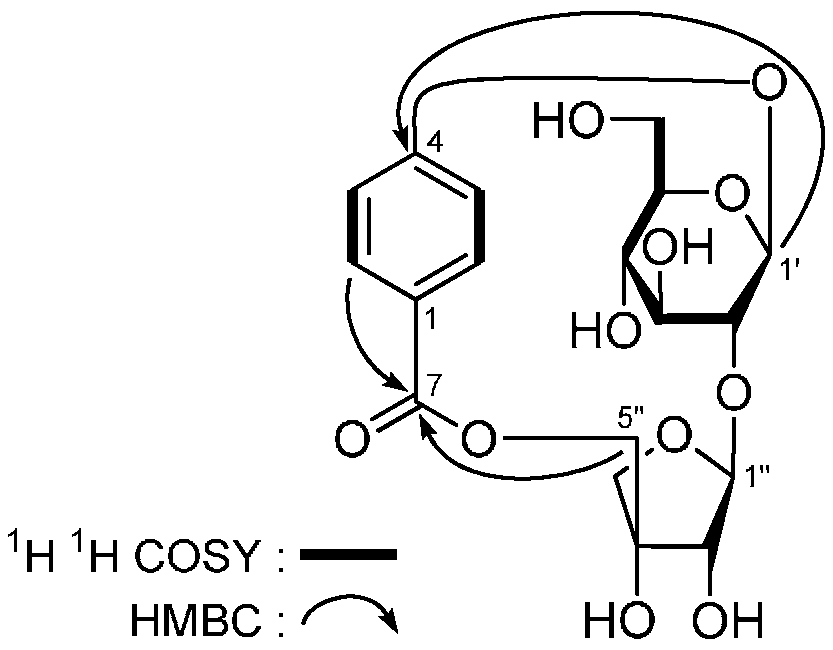
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 2 | 155.4 | - | 6′′ | 60.6 | 3.29 (m, overlapped) |
| 3 | 132.7 | - | 3.55 (br. d, ca. 12) | ||
| 4 | 177.1 | - | 1′′′ | 107.7 | 5.40 (br. s) |
| 5 | 161.2 | - | 2′′′ | 76.3 | 3.75 (br. s) |
| 6 | 98.5 | 6.16 (d, 1.5) | 3′′′ | 77.5 | - |
| 7 | 163.9 | - | 4′′′ | 73.6 | 3.57 (d, 9.5) |
| 8 | 93.3 | 6.24 (d, 1.5) | 3.95 (d, 9.5) | ||
| 9 | 156.0 | - | 5′′′ | 67.6 | 4.15 (d, 11.0) |
| 10 | 103.9 | - | 4.27 (d, 11.0) | ||
| 1′ | 120.9 | - | 1′′′′ | 125.4 | - |
| 2′,6′ | 130.6 | 7.99 (d, 9.0) | 2′′′′ | 110.7 | 7.14 (d, 1.5) |
| 3′,5′ | 114.9 | 6.86 (d, 9.0) | 3′′′′ | 147.7 | - |
| 4′ | 159.8 | - | 4′′′′ | 149.1 | - |
| 5-OH | - | 12.58 (br. s) | 5′′′′ | 115.3 | 6.77 (d, 8.0) |
| 1′′ | 98.3 | 5.66 (d, 7.5) | 6′′′′ | 122.9 | 6.93 (dd, 1.5, 8.0) |
| 2′′ | 75.9 | 3.49 (dd, 7.5, 9.0) | 7′′′′ | 144.5 | 7.25 (d, 16.0) |
| 3′′ | 76.9 | 3.42 (dd, 9.0, 9.0) | 8′′′′ | 114.0 | 6.21 (d, 16.0) |
| 4′′ | 70.2 | 3.10 (dd, 9.0, 9.0) | 9′′′′ | 166.2 | - |
| 5′′ | 77.4 | 3.09 (m) | 3′′′′-OCH3 | 55.5 | 3.81 (s) |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 2 | 155.8 | - | 2′′′ | 70.5 | 3.75 (br. s) |
| 3 | 132.9 | - | 3′′′ | 70.4 | 3.46 (dd, 3.0, 9.0) |
| 4 | 177.4 | - | 4′′′ | 71.8 | 3.14 (dd, 9.0, 9.0) |
| 5 | 160.9 | - | 5′′′ | 68.2 | 3.72 (m) |
| 6 | 99.6 | 6.51 (d, 1.5) | ′′′ | 17.2 | 0.79 (d, 6.0) |
| 7 | 162.6 | - | 1′′′′ | 99.1 | 5.18 (d, 7.0) |
| 8 | 94.4 | 6.81 (d, 1.5) | 2′′′′ | 73.0 | 3.32 (dd, 7.0, 9.0) |
| 9 | 156.6 | - | 3′′′′ | 76.1 | 3.36 (dd, 9.0, 9.0) |
| 10 | 105.6 | - | 4′′′′ | 69.3 | 3.31 (m, overlapped) |
| 1′ | 120.6 | - | 5′′′′ | 73.8 | 3.80 (m) |
| 2′,6′ | 130.7 | 8.06 (d, 9.0) | 6′′′′ | 63.0 | 4.20 (dd, 5.5, 12.0) |
| 3′,5′ | 115.0 | 6.87 (d, 9.0) | 4.44 (br. d, ca. 12) | ||
| 4′ | 160.0 | - | 1′′′′′ | 125.4 | - |
| 5-OH | - | 12.63 (br. s) | 2′′′′′ | 110.9 | 7.28 (d, 1.5) |
| 1′′ | 98.2 | 5.67 (d, 7.5) | 3′′′′′ | 147.8 | - |
| 2′′ | 77.5 | 3.46 (dd, 7.5, 9.0) | 4′′′′′ | 149.3 | - |
| 3′′ | 77.2 | 3.40 (dd, 9.0, 9.0) | 5′′′′′ | 115.4 | 6.77 (d, 8.5) |
| 4′′ | 70.1 | 3.09 (dd, 9.0, 9.0) | 6′′′′′ | 123.1 | 7.04 (dd, 1.5, 8.5) |
| 5′′ | 77.4 | 3.09 (m) | 7′′′′′ | 145.2 | 7.54 (d, 16.0) |
| 6′′ | 60.7 | 3.28 (dd, 4.5, 11.5) | 8′′′′′ | 114.0 | 6.46 (d, 16.0) |
| 3.56 (br. d, ca. 12) | 9′′′′′ | 166.5 | - | ||
| 1′′′ | 100.5 | 5.10 (br. s) | 3′′′′′-OCH3 | 55.6 | 3.80 (s) |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 2 | 71.6 | 3.84 (dd, 8.5, 9.0) | 4′ | 141.3 | - |
| 4.33 (dd, 2.0, 9.0) | 5′ | 141.6 | - | ||
| 3 | 32.4 | 3.80 (m) | 6′ | 109.8 | 6.42 (s) |
| 4 | 32.5 | 2.75 (m) | 1′′ | 105.6 | 4.85 (d, 9.0) |
| 5 | 131.3 | 6.85 (d, 8.0) | 2′′ | 75.6 | 3.43 (m, overlapped) |
| 6 | 109.2 | 6.32 (dd, 2.5, 8.0) | 3′′ | 77.9 | 3.43 (m, overlapped) |
| 7 | 157.6 | - | 4′′ | 71.5 | 3.36 (dd, 6.5, 9.5) |
| 8 | 103.9 | 6.25 (d, 2.5) | 5′′ | 78.2 | 3.24 (m) |
| 9 | 156.3 | - | 6′′ | 62.8 | 3.70 (dd, 5.0, 11.5) |
| 10 | 114.9 | - | 3.85 (dd, 2.0, 11.5) | ||
| 1′ | 133.1 | - | 3′-OCH3 | 61.9 | 3.90 (s) |
| 2′ | 148.8 | - | 5′-OCH3 | 61.3 | 3.83 (s) |
| 3′ | 147.6 | - |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 122.3 | - | 6′ | 60.2 | 3.45 (dd, 5.5, 12.0) |
| 2,6 | 130.9 | 7.53 (d, 9.0) | 3.65 (br. d, ca. 12) | ||
| 3,5 | 114.8 | 6.97 (d, 9.0) | 1′′ | 107.7 | 5.34 (br. s) |
| 4 | 160.2 | - | 2′′ | 76.3 | 3.65 (br. s) |
| 7 | 164.6 | - | 3′′ | 77.5 | - |
| 1′ | 96.8 | 5.18 (d, 7.0) | 4′′ | 73.4 | 3.80 (d, 9.5) |
| 2′ | 75.3 | 3.53 (dd, 7.0, 9.0) | 4.31 (d, 9.5) | ||
| 3′ | 76.7 | 3.52 (dd, 9.0, 9.0) | 5′′ | 68.0 | 3.87 (d, 11.0) |
| 4′ | 69.5 | 3.20 (dd, 9.0, 9.0) | 4.32 (d, 11.0) | ||
| 5′ | 76.7 | 3.52 (m) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, J.; Li, J.; Li, X.; Liu, Y.; Ruan, J.; Dong, Y.; Zhang, Y.; Wang, T. Aromatic Constituents from the Stems of Astragalus membranaceus (Fisch.) Bge. var. Mongholicus (Bge.) Hsiao. Molecules 2016, 21, 354. https://doi.org/10.3390/molecules21030354
Hao J, Li J, Li X, Liu Y, Ruan J, Dong Y, Zhang Y, Wang T. Aromatic Constituents from the Stems of Astragalus membranaceus (Fisch.) Bge. var. Mongholicus (Bge.) Hsiao. Molecules. 2016; 21(3):354. https://doi.org/10.3390/molecules21030354
Chicago/Turabian StyleHao, Jia, Jian Li, Xiaoxia Li, Yanxia Liu, Jingya Ruan, Yongzhe Dong, Yi Zhang, and Tao Wang. 2016. "Aromatic Constituents from the Stems of Astragalus membranaceus (Fisch.) Bge. var. Mongholicus (Bge.) Hsiao" Molecules 21, no. 3: 354. https://doi.org/10.3390/molecules21030354
APA StyleHao, J., Li, J., Li, X., Liu, Y., Ruan, J., Dong, Y., Zhang, Y., & Wang, T. (2016). Aromatic Constituents from the Stems of Astragalus membranaceus (Fisch.) Bge. var. Mongholicus (Bge.) Hsiao. Molecules, 21(3), 354. https://doi.org/10.3390/molecules21030354





