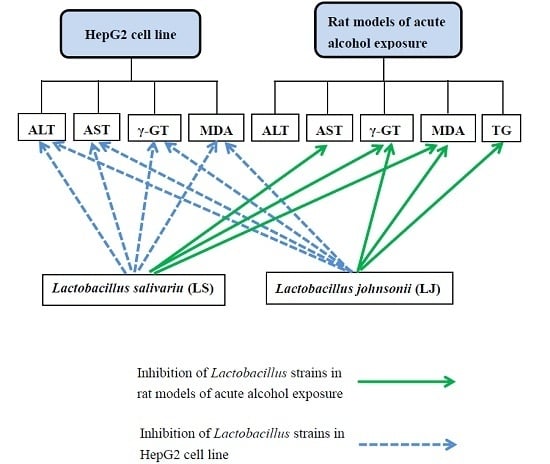
Heat-Killed Lactobacillus salivarius and Lactobacillus johnsonii Reduce Liver Injury Induced by Alcohol In Vitro and In Vivo
Abstract
:1. Introduction
2. Results
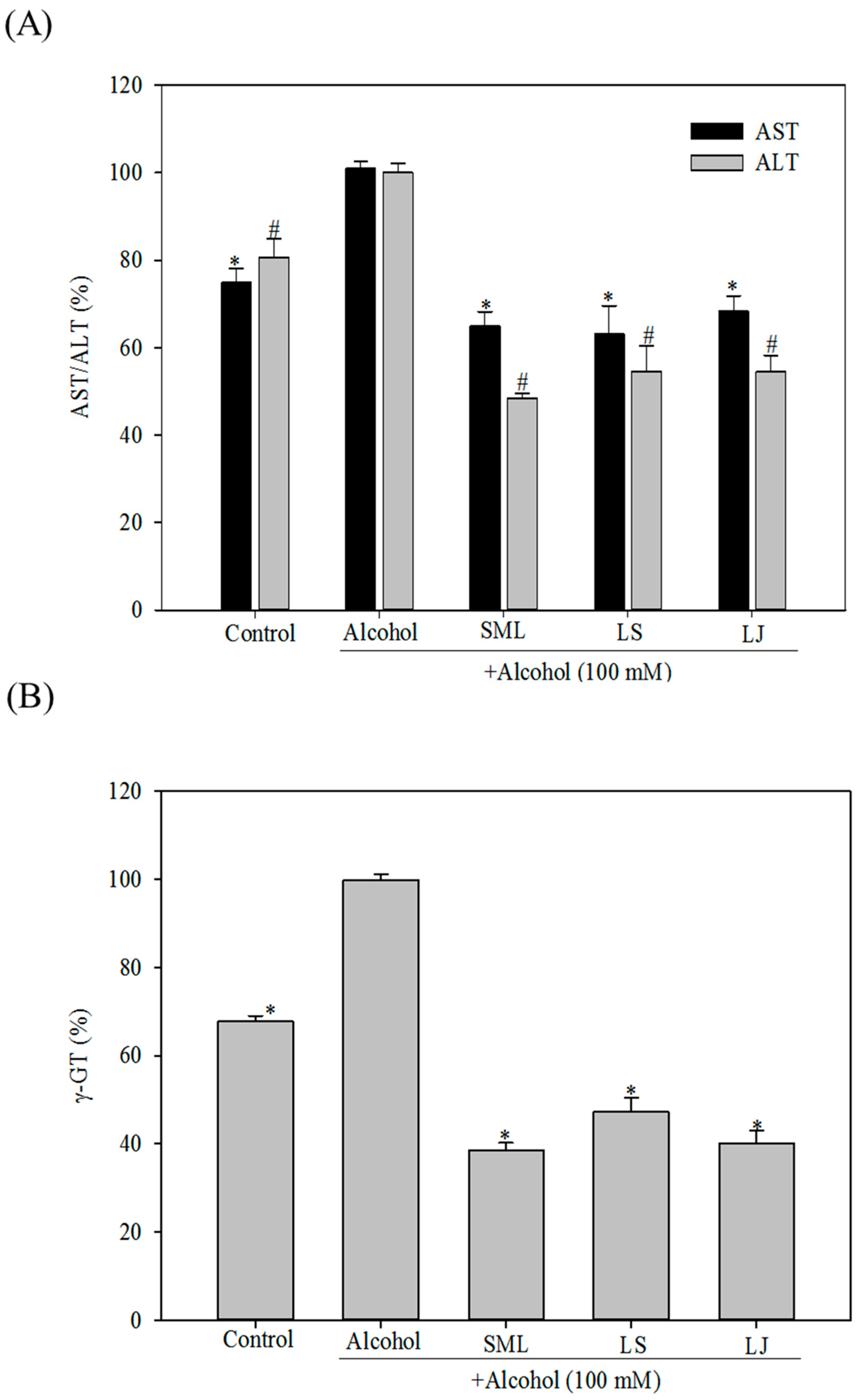
2.1. Effects of Heat-Killed LS and LJ on AST, ALT and γ-GT Levels in HepG2 Cells
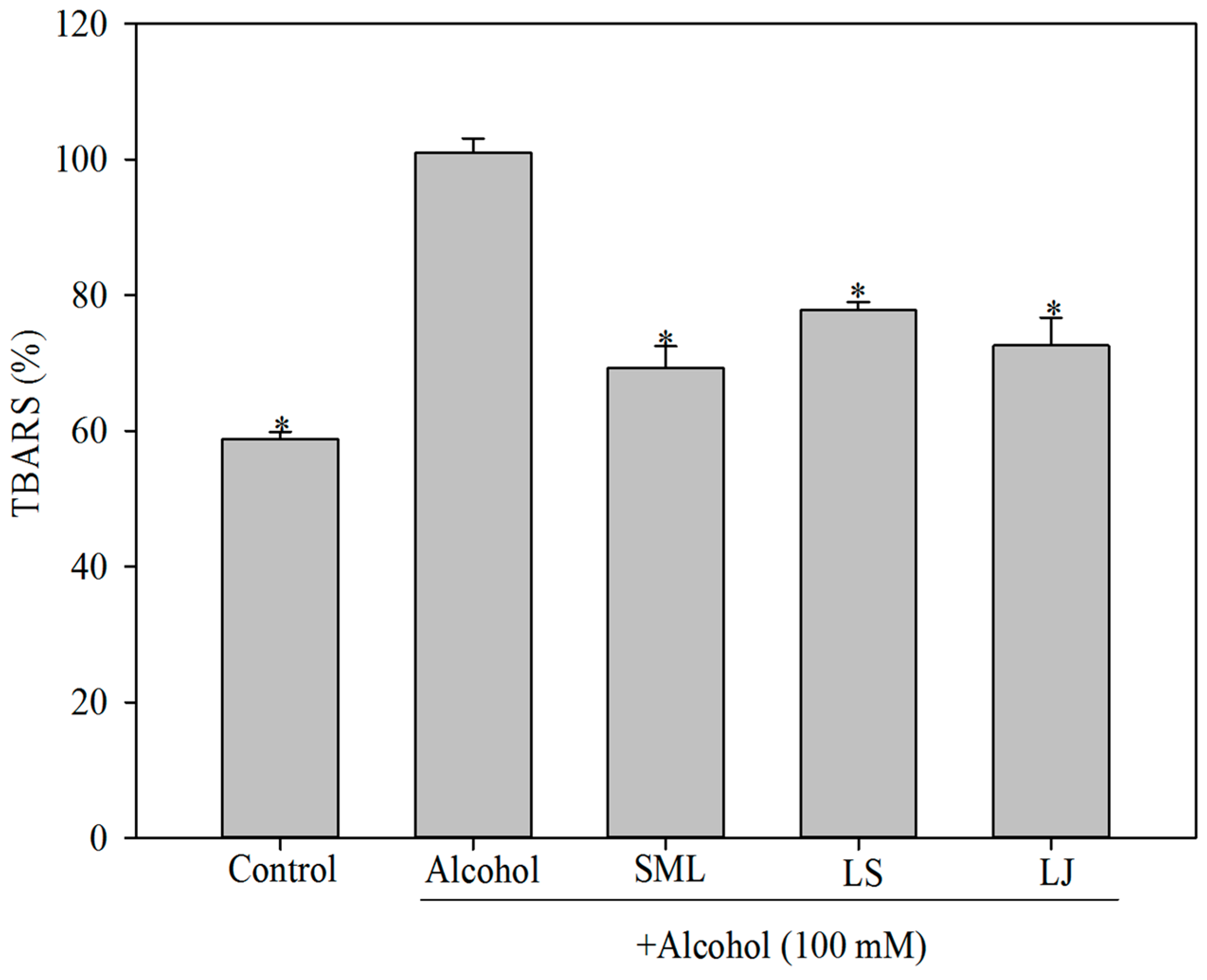
2.2. Effects of Heat-Killed LS and LJ on Lipid Peroxidation in HepG2 Cells
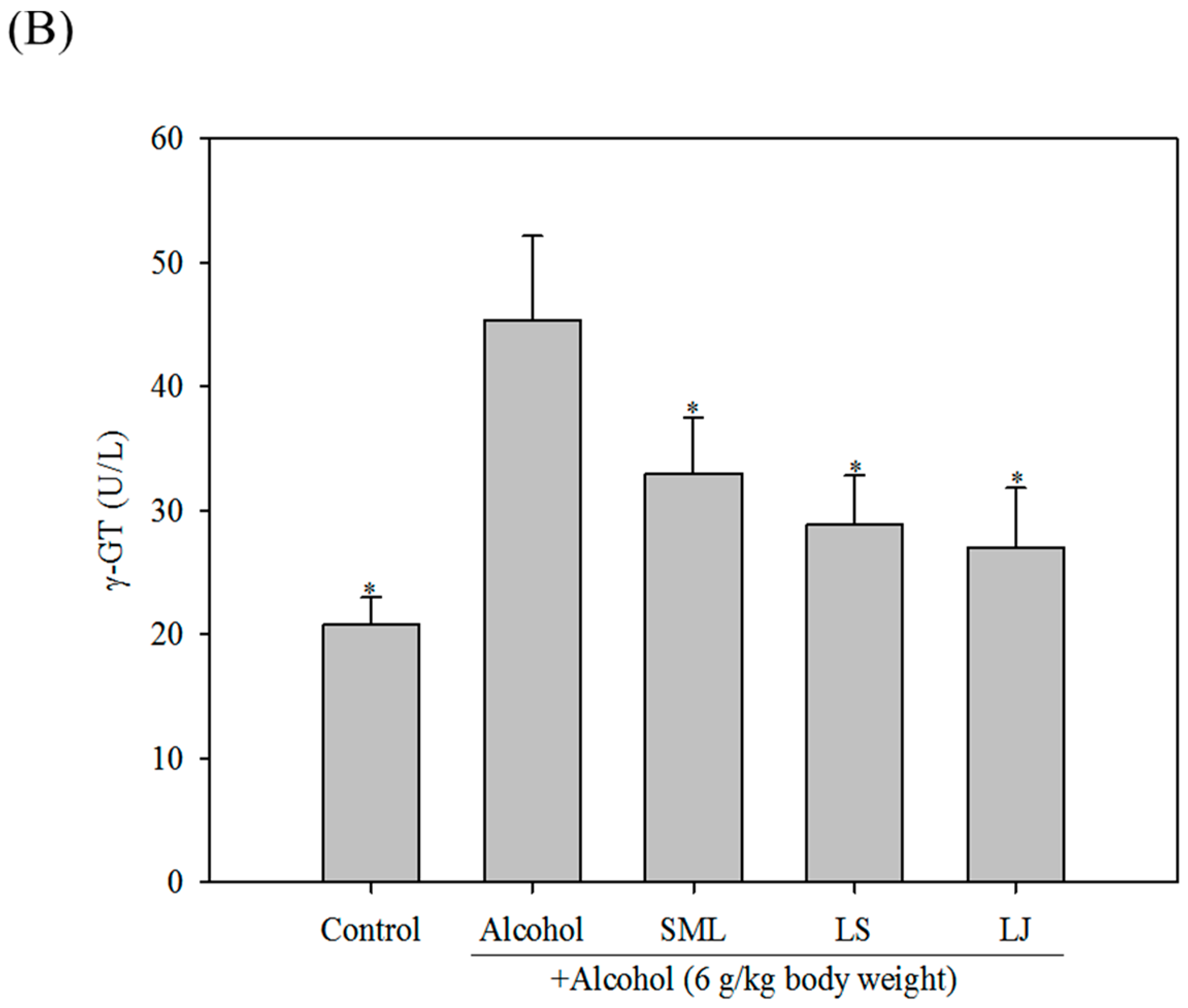
2.3. Effects of Heat-Killed LS and LJ on γ-GT Levels in Animal Models
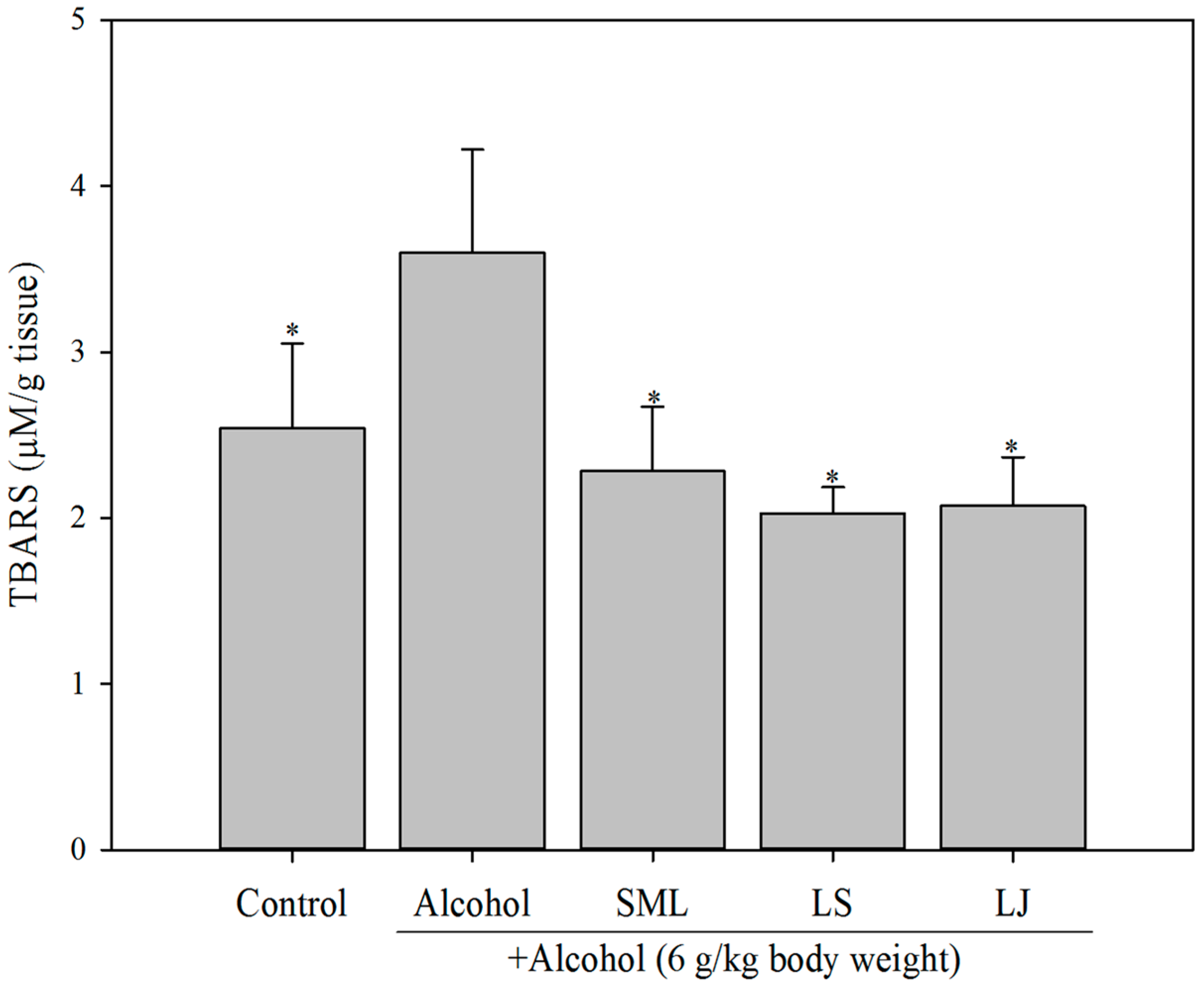
2.4. Effects of Heat-Killed LS and LJ on Lipid Peroxidation in Animal Models
2.5. Effects of Heat-Killed LS and LJ on Blood Lipid Levels in Animal Models
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Lactobacillus Cultures and Heat-Killed Products
4.3. Cell Culture and Treatments
4.4. Animals and Experimental Design
4.5. Measurement of ALT, AST and γ-GT Activity
4.6. Measurement of Lipid Peroxidation
4.7. Measurement of Serum Lipids
4.8. Determination of Liver Antioxidant Enzymes
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AST | Aspartate transaminase |
| ALT | Alanine transaminase |
| γ-GT | Gamma-glutamyl transferase |
| MDA | Malondialdehyde |
| LS | Lactobacillus salivarius |
| LJ | Lactobacillus johnsonii |
| TG | Triglycerides |
| SML | Silymarin |
| TBARS | Thiobarbituric acid-reactive substances |
| SOD | Glutathione peroxidase |
| GPx | Malondialdehyde |
| SREBP | Sterol regulatory element-binding protein |
References
- Cojocariu, C.E.; Trifan, A.V.; Gîrleanu, I.; Stanciu, C. Alcoholic liver disease-epidemiology and risk factors. Rev. Med. Chir. Soc. Med. Nat. Iasi 2014, 118, 910–917. [Google Scholar] [PubMed]
- Haber, P.S.; Seth, D.; Warner, D.; Gorrell, M.D.; McCaughan, G.W. Pathogenesis and management of alcoholic hepatitis. J. Gastroenterol. Hepatol. 2003, 18, 1332–1344. [Google Scholar] [CrossRef] [PubMed]
- Seth, D.; Hogg, P.J.; Gorrell, M.D.; McCaughan, G.W.; Haber, P.S. Direct effects of alcohol on hepatic fibrinolytic balance: Implications for alcoholic liver disease. J. Hepatol. 2008, 48, 614–627. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, S.; De la Maza, P.; Petermann, M.; Bunout, D. Lipid turnover in alcoholics before and after an ethanol load. Nutrition 1998, 14, 437–442. [Google Scholar] [CrossRef]
- Akanji, A.O.; Hockaday, T.D.R. Acetate tolerance and the kinetics of acetate utilization in diabetic and non diabetic subjects. Am. J. Clin. Nutr. 1990, 51, 112–118. [Google Scholar] [PubMed]
- Hines, I.N.; Wheeler, M.D. Recent advances in alcoholic liver disease III. Role of the innate immune response in alcoholic hepatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 287, G310–G314. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Cederbaum, A.I.; Zhang, Y.L.; Ginsberg, H.N.; Williams, K.J.; Fisher, E.A. Lipid per oxidation and oxidant stress regulate hepatic apolipoprotein B degradation and VLDL production. J. Clin. Investig. 2004, 113, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Checa, J.C.; Kaplowitz, N. Hepatic mitochondrial glutathione: Transport and role in disease and toxicity. Toxicol. Appl. Pharmacol. 2005, 204, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Chi, F.; Wang, G.; Liu, X.; Zhang, Q.; Chen, Y.; Zhang, H.; Chen, W. Lactobacillusrhamnosus CCFM1107 treatment ameliorates alcohol-induced liver injury in a mouse model of chronic alcohol feeding. J. Microbiol. 2015, 53, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A.; Yang, E.K.; Fogt, F.; Sadrzadeh, S.; Dannenberg, A.J. Medium chain triglycerides and vitamin E reduce the severity of established experimental alcoholic liver disease. J. Pharmacol. Exp. Ther. 1996, 277, 1694–1700. [Google Scholar] [PubMed]
- Ajmo, J.M.; Liang, X.; Rogers, C.Q.; Pennock, B.; You, M. Resveratrol alleviates alcoholic fatty liver in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G833–G842. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Du, R.; Wang, L.; Zhang, H. The antioxidative effects of probiotic Lactobacillus casei Zhang on the hyperlipidemic rats. Eur. Food Res. Technol. 2010, 231, 151–158. [Google Scholar] [CrossRef]
- Lin, M.Y.; Chang, F.J. Antioxidative effect of intestinal bacteria Bifidobacterium longum ATCC 15708 and Lactobacillus acidophilus ATCC 4356. Dig. Dis. Sci. 2000, 45, 1617–1622. [Google Scholar] [CrossRef] [PubMed]
- Terahara, M.; Kurama, S.; Takemoto, N. Prevention by lactic acid bacteria of the oxidation of human LDL. Biosci. Biotechnol. Biochem. 2001, 65, 1864–1868. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Kong, B.; Sun, Q.; Dong, F.; Liu, Q. Antioxidant potential of a unique LAB culture isolated from Harbin dry sausage: In vitro and in a sausage model. Meat Sci. 2015, 110, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Ewaschuk, J.; Endersby, R.; Thiel, D.; Diaz, H.; Backer, J.; Ma, M.; Churchill, T.; Madsen, K. Probiotic bacteria prevent hepatic damage and maintain colonic barrier function in a mouse model of sepsis. Hepatology 2007, 46, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Han, S.Y.; Huh, C.S.; Ahn, Y.T.; Lim, K.S.; Baek, Y.J.; Kim, D.H. Hepatoprotective effect of lactic acid bacteria, inhibitors of beta-glucuronidase production against intestinal microflora. Arch. Pharm. Res. 2005, 28, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Osman, N.; Adawi, D.; Ahrne, S.; Jeppsson, B.; Molin, G. Probiotics and blueberry attenuate the severity of dextran sulfate sodium (DSS)-induced colitis. Dig. Dis. Sci. 2008, 53, 2464–2473. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Federico, A.; Tuccillo, C.; Terracciano, F.; D’Auria, M.V.; de Simone, C.; del Vecchio Blanco, C. Beneficial effects of a probiotic VSL#3 on parameters of liver dysfunction in chronic liver diseases. J. Clin. Gastroenterol. 2005, 39, 540–543. [Google Scholar] [PubMed]
- Daubioul, C.A.; Horsmans, Y.; Lambert, P.; Danse, E.; Delzenne, N.M. Effects of oligofructose on glucose and lipid metabolism in patients with nonalcoholic steatohepatitis: Results of a pilot study. Eur. J. Clin. Nutr. 2005, 59, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Duan, Z.P.; Ha, D.K.; Bengmark, S.; Kurtovic, J.; Riordan, S.M. Synbiotic modulation of gut flora: Effect on minimal hepatic encephalopathy in patients with cirrhosis. Hepatology 2004, 39, 1441–1449. [Google Scholar] [CrossRef] [PubMed]
- Kirpich, I.A.; Solovieva, N.V.; Leikhter, S.N.; Shidakova, N.A.; Lebedeva, O.V.; Sidorov, P.I.; Bazhukova, T.A.; Soloviev, A.G.; Barve, S.S.; McClain, C.J.; et al. Probiotics restore bowel flora and improve liver enzymes in human alcohol-induced liver injury: A pilot study. Alcohol 2008, 42, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Nanji, A.A.; Khettry, U.; Sadrzadeh, S.M. Lactobacillus feeding reduces endotoxemia and severity of experimental alcoholic liver (disease). Proc. Soc. Exp. Biol. Med. 1994, 205, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Forsytha, C.B.; Farhadi, A.; Jakate, S.M.; Tang, Y.; Shaikh, M.; Keshavarzian, A. Lactobacillus GG treatment ameliorates alcohol-induced intestinal oxidative stress, gut leakiness, and liver injury in a rat model of alcoholic steatohepatitis. Alcohol 2009, 43, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Segawa, S.; Wakita, Y.; Hirata, H.; Watari, J. Oral administration of heat-killed Lactobacillusbrevis SBC8803 ameliorates alcoholic liver disease in ethanol-containing diet-fed C57BL/6N mice. Int. J. Food Microbiol. 2008, 128, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.W.; Liu, Y.Z. Molecular structure and stereochemistry of silybin A, silybin B, and isosilybin A, isosilybin B, isolated from Silybum. mariaanum. J. Nat. Prod. 2003, 66, 1171–1174. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Hong, R.; Tian, T. Silymarin’s protective effects and possible mechanisms on alcoholic fatty liver for rats. Biomol. Ther. (Seoul) 2013, 21, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Boigk, G.; Stroedter, L.; Herbst, H.; Waldschmidt, J.; Riecken, E.O.; Schuppan, D. Silymarin retards collagen accumulation in early and advanced biliary fibrosis secondary to complete bile duct obliteration in rats. Hepatology 1997, 26, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Mukamal, K.; Rimm, E.B. Alcohol consumption: Risks and benefits. Curr. Atheroscler. Rep. 2008, 10, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Pochareddy, S.; Edenberg, H.J. Chronic alcohol exposure alter gene expression in HepG2 cells. Alcohol Clin. Exp. Res. 2012, 36, 1021–1033. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.D.; Lee, S.R.; Kim, T.; Jang, S.A.; Kang, S.C.; Koo, H.J.; Sohn, E.; Bak, J.P.; Namkoong, S.; Kim, H.K.; et al. Fucoidan from Fucus vesiculosus protects against alcohol-induced liver damage by modulatin inflammatory mediators in mice and HepG2 cells. Mar. Drugs 2015, 13, 1051–1067. [Google Scholar] [CrossRef] [PubMed]
- Rishi, P.; Mavi, S.K.; Bharrhan, S.; Shukla, G.; Tewari, R. Protective efficacy of probiotic alone or in conjunction with a prebiotic in Salmonella-induced liver damage. FEMS Microbiol. Ecol. 2009, 69, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Albano, E. Alcohol, oxidative stress and free radical damage. Proc. Nutr. Soc. 2006, 65, 278–290. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Cederbaum, A.I. Alcohol, oxidative stress, and free radical damage. Alcohol Res. Health 2003, 27, 277–284. [Google Scholar] [PubMed]
- Nielsen, F.; Mikkelsen, B.B.; Nielsen, J.B.; Andersen, H.R.; Grandjean, P. Plasma malondialdehyde as biomarker for oxidative stress: Reference interval and effects of life-style factors. Clin. Chem. 1997, 43, 1209–1214. [Google Scholar] [PubMed]
- Noorani, A.A.; Kale, M.K. Pretreatment of Albin Rats with Methanolic Fruit Extract of Randia Dumetorum (L.) Protects against Alcohol Induced Liver Damage. Korean J. Physiol. Pharmacol. 2012, 16, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C.S. Ethanol metabolism, cirrhosis and alcoholism. Clin. Chim. Acta 1997, 257, 59–84. [Google Scholar] [CrossRef]
- Polavarapu, R.; Spitz, D.R.; Sim, J.E.; Follansbee, M.H.; Oberley, L.W.; Rahemtulla, A.; Nanji, A.A. Increased lipid peroxidation and impaired antioxidant enzyme function is associated with pathological liver injury in experimental alcoholic liver disease in rats fed diets high in corn oil and fish oil. Hepatology 1998, 27, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Horton, J.D.; Goldstein, J.L.; Brown, M.S. SREBPs: Activators of the complete program of cholesterol and fatty acid synthesis in the liver. J. Clin. Investig. 2002, 109, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Crabb, D.W. Molecular mechanisms of alcoholic fatty liver: Role of sterol regulatory element-binding proteins. Alcohol 2004, 34, 39–43. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Fischer, M.; Deeg, M.A.; Crabb, D.W. Ethanol induces fatty acid synthesis pathways by activation of sterol regulatory element-binding protein (SREBP). J. Biol. Chem. 2002, 277, 29342–29347. [Google Scholar] [CrossRef] [PubMed]
- Buege, A.J.; Aust, S.D. Micorsomal lipid peroxidation. Methods Enzymol. 1976, 52, 302–310. [Google Scholar]
- Imamoglu, N.; Yerer, M.B.; Donmez-Altuntas, H.; Saraymen, R. Erythrocyte antioxidant enzyme activities and lipid peroxidation in the erythrocyte membrane of stainless-steel welders exposed to welding fumes and gases. Int. J. Hyg. Environ. Health 2008, 211, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.

| Group | Serum Lipid (mg/dL) | |||
|---|---|---|---|---|
| HDL Cholesterol | LDL Cholesterol | Total Cholesterol | Triglyceride | |
| Control | 76.8 ± 6.2 | 95.6 ± 9.4 | 172.2 ± 13.5 | 43.4 ± 8.1 c |
| Alcohol | 75.2 ± 5.1 | 101.1 ± 7.1 | 175.6 ± 7.8 | 167.8 ± 27.2 a |
| SML 2 | 68.6 ± 5.2 | 98.4 ± 4.3 | 181.8 ± 5.9 | 165.5 ± 28.9 a |
| LJ | 69.6 ± 5.5 | 106.6 ± 10.7 | 171.8 ± 17.2 | 129.4 ± 39.8 ab |
| LS | 77.8 ± 7.9 | 104.5 ± 4.9 | 173.3 ± 20.0 | 102.1 ± 36.1 b |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chuang, C.-H.; Tsai, C.-C.; Lin, E.-S.; Huang, C.-S.; Lin, Y.-Y.; Lan, C.-C.; Huang, C.-C. Heat-Killed Lactobacillus salivarius and Lactobacillus johnsonii Reduce Liver Injury Induced by Alcohol In Vitro and In Vivo. Molecules 2016, 21, 1456. https://doi.org/10.3390/molecules21111456
Chuang C-H, Tsai C-C, Lin E-S, Huang C-S, Lin Y-Y, Lan C-C, Huang C-C. Heat-Killed Lactobacillus salivarius and Lactobacillus johnsonii Reduce Liver Injury Induced by Alcohol In Vitro and In Vivo. Molecules. 2016; 21(11):1456. https://doi.org/10.3390/molecules21111456
Chicago/Turabian StyleChuang, Cheng-Hung, Cheng-Chih Tsai, En-Shyh Lin, Chin-Shiu Huang, Yun-Yu Lin, Chuan-Ching Lan, and Chun-Chih Huang. 2016. "Heat-Killed Lactobacillus salivarius and Lactobacillus johnsonii Reduce Liver Injury Induced by Alcohol In Vitro and In Vivo" Molecules 21, no. 11: 1456. https://doi.org/10.3390/molecules21111456
APA StyleChuang, C.-H., Tsai, C.-C., Lin, E.-S., Huang, C.-S., Lin, Y.-Y., Lan, C.-C., & Huang, C.-C. (2016). Heat-Killed Lactobacillus salivarius and Lactobacillus johnsonii Reduce Liver Injury Induced by Alcohol In Vitro and In Vivo. Molecules, 21(11), 1456. https://doi.org/10.3390/molecules21111456