Effect of the Aged Garlic Extract on Cardiovascular Function in Metabolic Syndrome Rats
Abstract
:1. Introduction
2. Results
2.1. Effect of AGE on the Cluster of Risk Factors That Constitute MS
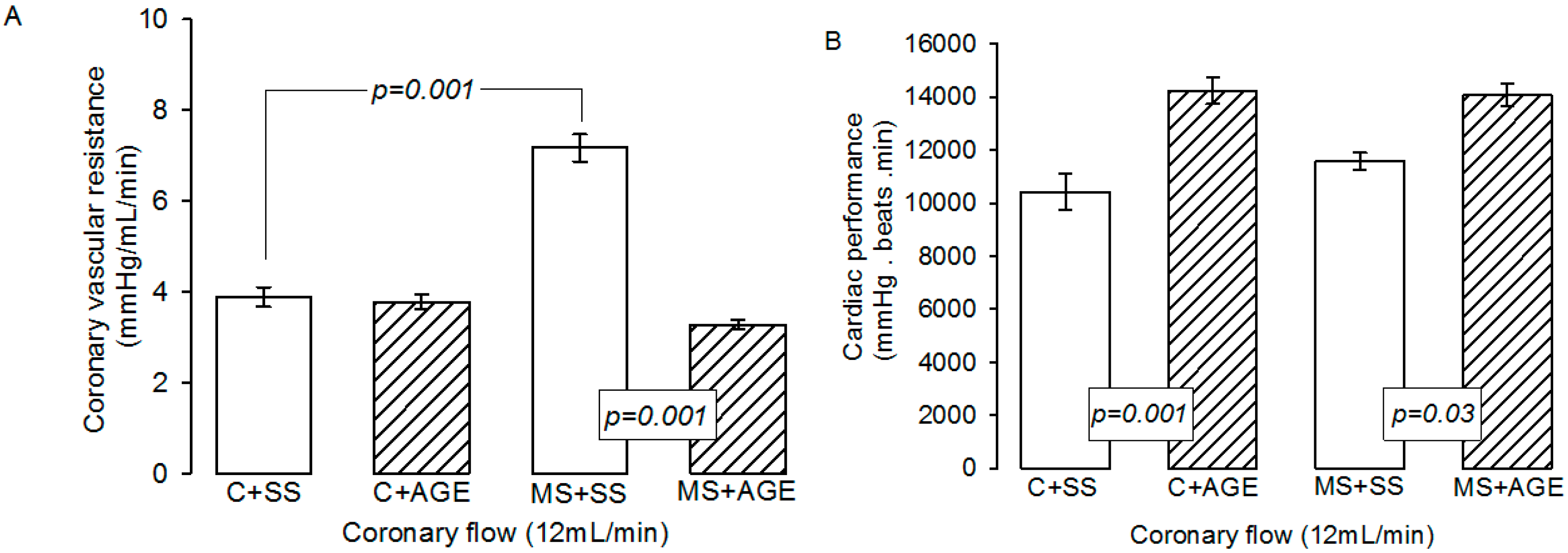
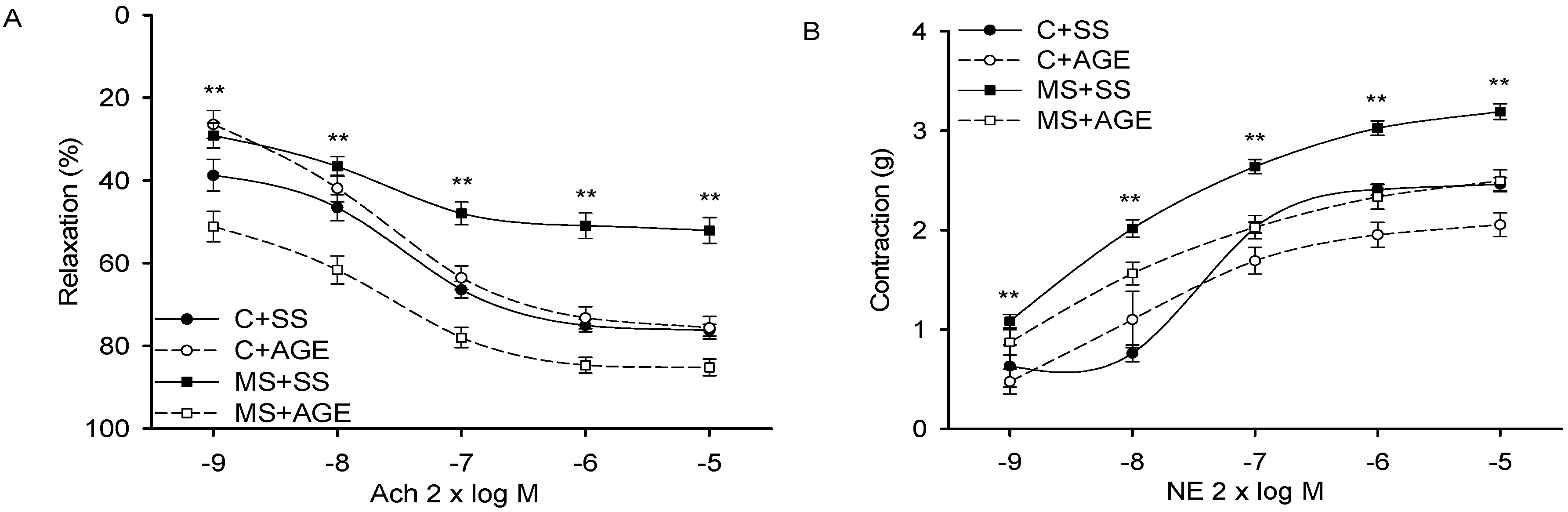
2.2. Effect of AGE on Heart Functioning and Vascular Reactivity
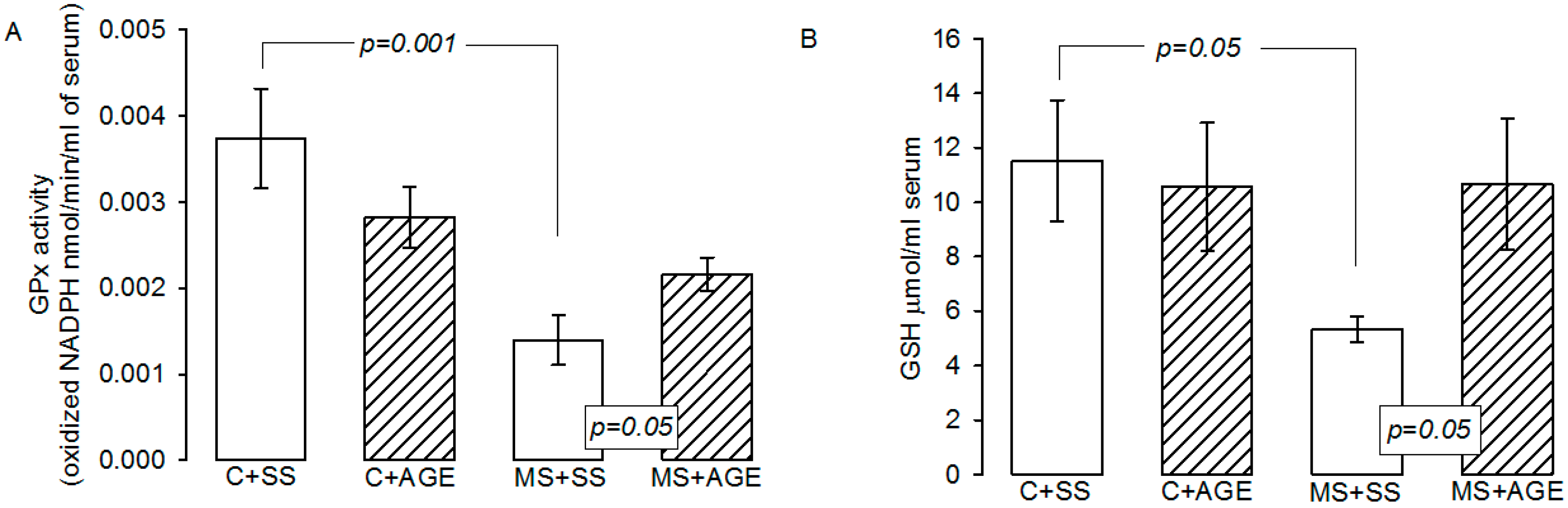
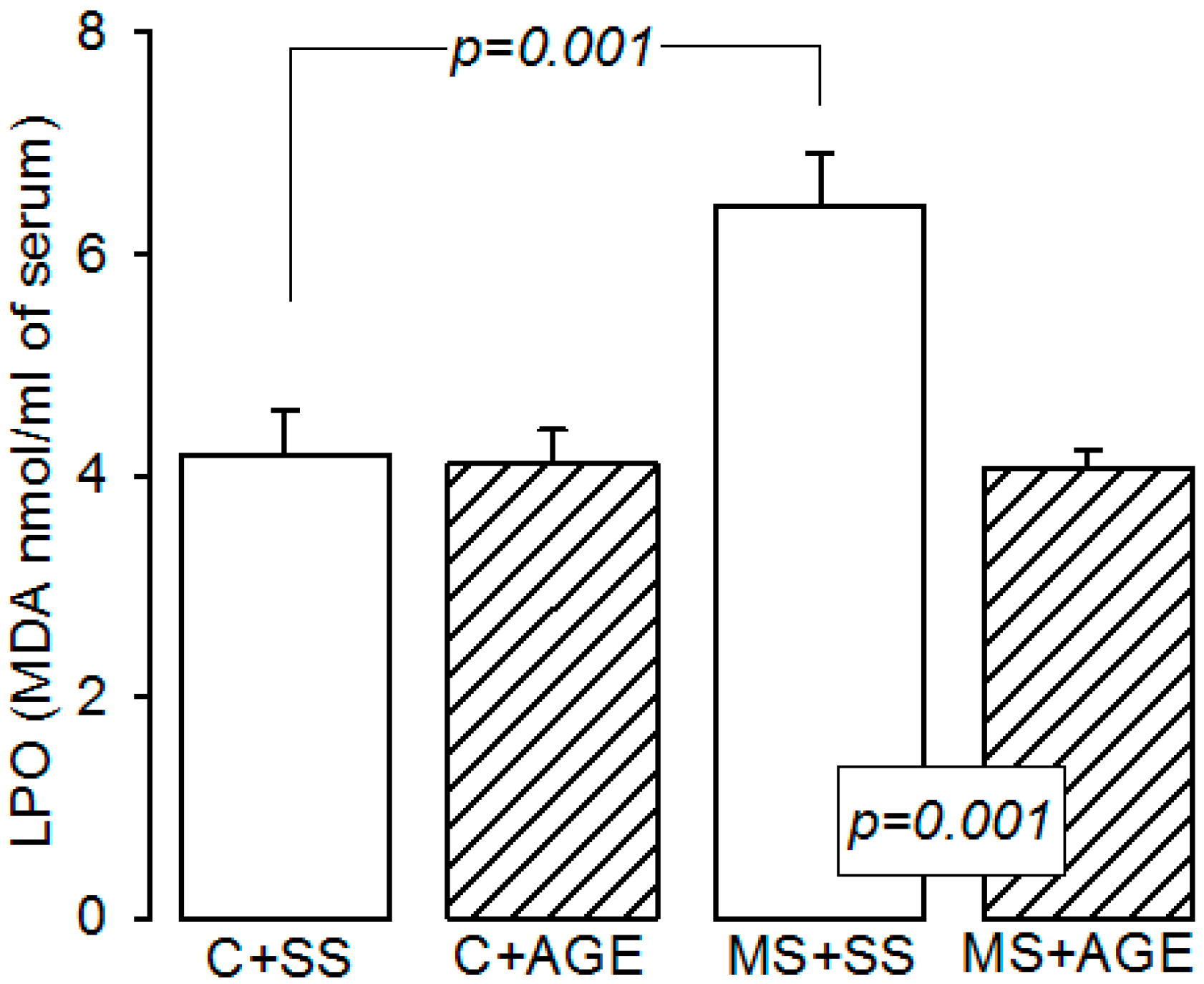
2.3. Glutathione Peroxidase Activity and Glutathione Concentration
3. Materials and Methods
3.1. Animals
3.2. Biochemical Variables
3.3. Nitric Oxide Quantification
3.4. Citrulline Determination
3.5. Nitrate and Nitrite Quantification
3.6. Isolated Heart Perfused by the Langendorff Method
3.7. Vascular Reactivity
3.8. Glutathione Peroxidase Activity
3.9. Determination of GSH Concentration
3.10. Lipid Peroxidation Levels
3.11. Statistical Analysis
4. Discussion
4.1. Biochemical Variables
4.2. Vascular Reactivity and Cardiovascular Function
4.3. Hypertension
4.4. GPx Activity and GSH Concentration
4.5. Lipid Peroxidation
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ginsberg, H.N.; MacCallum, P.R. The obesity, metabolic syndrome, and type 2 diabetes mellitus pandemic: Part I. Increased cardiovascular disease risk and the importance of atherogenic dyslipidemia in persons with the metabolic syndrome and type 2 diabetes mellitus. J. Cardiometab. Syndr. 2009, 4, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Rice, M.W.; Cai, D. Neuroinflammatory and autonomic mechanisms in diabetes and hypertension. Am. J. Physiol. Endocrinol. Metab. 2016, 311, E32–E41. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Torres, I.; Zúñiga, M.A.; Beltrán-Rodríguez, U.; Díaz-Díaz, E.; Martínez-Memije, R.; Guarner, L.V. Modification of the liver fatty acids by Hibiscus sabdariffa Linnaeus (Malvaceae) infusion, its possible effect on vascular reactivity in a metabolic syndrome model. Clin. Exp. Hypertens. 2014, 36, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Nava, P.; Collados, M.T.; Massó, F.; Guarner, V. Endothelin mediation of insulin and glucose induced changes in vascular contractility. Hypertension 1997, 30, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Borek, C. Antioxidant health effects of aged garlic extract. J. Nutr. 2001, 131, 1010S–1015S. [Google Scholar] [PubMed]
- Qu, Z.; Mossine, V.V.; Cui, J.; Sun, G.Y.; Gu, Z. Protective effects of AGE and its components on neuroinflammation and neurodegeneration. NeuroMol. Med. 2016, 18, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Harauma, A.; Moriguchi, T. Aged garlic extract improves blood pressure in spontaneously hypertensive rats more safely than raw garlic. J. Nutr. 2006, 136, 769S–773S. [Google Scholar] [PubMed]
- Jeong, Y.Y.; Ryu, J.H.; Shin, J.H.; Kang, M.J.; Kang, J.R.; Han, J.; Kang, D. Comparison of anti-oxidant and anti-inflammatory effects between fresh and aged black garlic extracts. Molecules 2016, 21, 430. [Google Scholar] [CrossRef] [PubMed]
- Varshney, R.; Budoff, M.J. Garlic and heart disease. J. Nutr. 2016, 146, 416S–421S. [Google Scholar] [CrossRef] [PubMed]
- Atkin, M.; Laight, D.; Cummings, M.H. The effects of garlic extract upon endothelial function, vascular inflammation, oxidative stress and insulin resistance in adults with type 2 diabetes at high cardiovascular risk. A pilot double blind randomized placebo controlled trial. J. Diabetes Complicat. 2016, 30, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity: Implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes. Res. Clin. Pract. 2013, 7, e330–e341. [Google Scholar] [CrossRef] [PubMed]
- Majewski, M. Allium sativum: Facts and myths regarding human health. Rocz. Panstw. Zakl. Hig. 2014, 65, 1–8. [Google Scholar] [PubMed]
- Kyolic Aged Garlic Extract from Wakunaga Reserve. Available online: http://www.kyolic.com (access on 24 October 2016).
- Arnault, I.; Christides, J.P.; Mandon, N.; Haffner, T.; Kahane, R.; Auger, J. High-performance ion-pair chromatography method for simultaneous analysis of alliin, deoxyalliin, allicin and dipeptide precursors in garlic products using multiple mass spectrometry and UV detection. J. Chromatogr. A 2003, 991, 69–75. [Google Scholar] [CrossRef]
- Tenorio, L.F.A.; del Valle, M.L.; Pastelín, H.G. Validación de un método espectrofotométrico para la cuantificación de metabolitos estables de óxido nítrico en fluidos biológicos. Rev. Mex. Cienc. Farm. 2005, 36, 31–41. [Google Scholar]
- Li, D.; Li, N.S.; Chen, Q.Q.; Guo, R.; Xu, P.S.; Deng, H.W.; Li, Y.J. Calcitonin gene-related peptide-mediated cardioprotection of postconditioning in isolated rat hearts. Regul. Pept. 2008, 147, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Döring, H.J.; Dehvert, H. The isolated perfused warm-blooded heart according to Langendorff. In Biological Measurement Techniques, 1st ed.; Dehnert, H., Ed.; Biomes Technik-Verlag: Baden-Wurtemberg, Germany, 1988; pp. 1–131. [Google Scholar]
- Soto, M.E.; Zuñiga-Muñoz, A.; Guarner, L.V.; Duran-Hernández, E.J.; Pérez-Torres, I. Infusion of Hibiscus sabdariffa L. modulates oxidative stress in patients with Marfan syndrome. Mediat. Inflamm. 2016, 2016, 8625203. [Google Scholar] [CrossRef] [PubMed]
- Ilkun, O.; Boudina, S. Cardiac dysfunction and oxidative stress in the metabolic syndrome: An update on antioxidant therapies. Curr. Pharm. Des. 2013, 19, 4806–4817. [Google Scholar] [CrossRef] [PubMed]
- Kones, R. Primary prevention of coronary heart disease: Integration of new data, evolving views, revised goals, and role of rosuvastatin in management. A comprehensive survey. Drug. Des. Dev. Ther. 2011, 5, 325–380. [Google Scholar] [CrossRef] [PubMed]
- López, Y.R.; Pérez-Torres, I.; Zúñiga-Muñoz, A.; Lans, V.G.; Díaz-Díaz, E.; Castro, E.S.; Espejel, R.V. Effect of glycine on adipocyte hypertrophy in a metabolic syndrome rat model. Curr. Drug. Deliv. 2016, 13, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Ried, K.; Travica, N.; Sali, A. The effect of aged garlic extract on blood pressure and other cardiovascular risk factors in uncontrolled hypertensives: The AGE at Heart trial. Integr. Blood Press. Control 2016, 9, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Al-Qattan, K.K.; Al-Enezi, F.; Khanafer, R.M.; Mustafa, T. Effect of allicin from garlic powder on serum lipids and blood pressure in rats fed with a high cholesterol diet. Prostaglandins Leukot. Essent. Fatty Acids 2000, 62, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Padiya, R.; Khatua, T.N.; Bagul, P.K.; Kuncha, M.; Banerjee, S.K. Garlic improves insulin sensitivity and associated metabolic syndromes in fructose fed rats. Nutr. Metab. Lond. 2011, 8, 53. [Google Scholar] [CrossRef] [PubMed]
- Brace, L.D. Cardiovascular benefits of garlic (Allium sativum L.). J. Cardiovasc. Nurs. 2002, 16, 33–49. [Google Scholar] [CrossRef] [PubMed]
- Zúñiga-Muñoz, A.M.; Guarner, L.V.; Soria-Castro, E.; Diaz-Diaz, E.; Torrico-Lavayen, R.; Tena-Betancourt, E.; Pérez-Torres, I. 17βEstradiol modulates perfusion pressure and expression of 5-LOX and CYP450 4A in the isolated kidney of metabolic syndrome female rats. Int. J. Endocrinol. 2015, 2015, 149408. [Google Scholar] [CrossRef] [PubMed]
- Reaven, G.M.; Ho, H. Sugar-induced hypertension in Sprague-Dawley rats. Am. J. Hypertens. 1991, 4, 610–614. [Google Scholar] [PubMed]
- Peredo-Escárcega, A.E.; Guarner-Lans, V.; Pérez-Torres, I.; Ortega-Ocampo, S.; Carreón-Torres, E.; Castrejón-Tellez, V.; Díaz-Díaz, E.; Rubio-Ruiz, M.E. The combination of resveratrol and quercetin attenuates metabolic syndrome in rats by modifying the serum fatty acid composition and by upregulating SIRT 1 and SIRT 2 expression in white adipose tissue. Evid. Based Complement. Altern. Med. 2015, 2015, 474032. [Google Scholar] [CrossRef] [PubMed]
- Ambati, S. Garlic derivatives: Role in obesity and related disorders. OA Biotechnol. 2013, 2, 1–5. [Google Scholar] [CrossRef]
- Keophiphath, M.; Priem, F.; Jacquemond-Collet, I.; Clément, K.; Lacasa, D. 1,2-Vinyldithiin from garlic inhibits differentiation and inflammation of human preadipocytes. J. Nutr. 2009, 139, 2055–2060. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.S.; Pischetsrieder, M.; Ahmed, N. Aged garlic extract and S-allyl cysteine prevent formation of advanced glycation end products. Eur. J. Pharmacol. 2007, 561, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Zhang, Y.; Chen, L.; Liu, L.; Zhang, X.; Wu, Z.; Bai, B.; Ji, S. Decreased glycation and structural protection properties of γ-glutamyl-S-allyl-cysteine peptide isolated from fresh garlic scales (Allium sativum L.). Nat. Prod. Res. 2015, 29, 2219–2222. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Prieto, M.A.; Rodriguez, L.C.; Lembo, C.; Galmarini, C.R.; Miatello, R.M. Garlic and onion attenuates vascular inflammation and oxidative stress in fructose-fed rats. J. Nutr. Metab. 2011, 2011, 475216. [Google Scholar] [CrossRef] [PubMed]
- Al-Qattan, K.K.; Alnaqeeb, M.A.; Ali, M. The antihypertensive effect of garlic (Allium sativum) in the rat two-kidney—One-clip Gold blatt model. J. Ethnopharmacol. 1999, 66, 217–222. [Google Scholar] [CrossRef]
- Thomson, M.; Mustafa, T.; Ali, M. Thromboxane-B(2) levels in serum of rabbits receiving a single intravenous dose of aqueous extract of garlic and onion. Prostaglandins Leukot. Essent. Fatty Acids 2000, 63, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Kaye, A.D.; De Witt, B.J.; Anwar, M.; Smith, D.E.; Feng, C.J.; Kadowitz, P.J.; Nossaman, B.D. Analysis of responses of garlic derivatives in the pulmonary vascular bed of the rat. J. Appl. Physiol. 1985, 89, 353–358. [Google Scholar]
- Panza, J.A.; García, C.E.; Kilcoyne, C.M.; Quyyumi, A.A.; Cannon, R.O. Impaired endothelium-dependent vasodilation in patients with essential hypertension. Evidence that nitric oxide abnormality is not localized to a single signal transduction pathway. Circulation 1995, 91, 1732–1738. [Google Scholar] [CrossRef] [PubMed]
- Weiss, N.; Papatheodorou, L.; Morihara, N.; Hilge, R.; Ide, N. Aged garlic extract restores nitric oxide bioavailability in cultured human endothelial cell seven under conditions of homocysteine elevation. J. Ethnopharmacol. 2013, 145, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Das, I.; Hirani, J.; Sooranna, S. Arginine is not responsible for the activation of nitric oxide synthase by garlic. J. Ethnopharmacol. 1996, 53, 5–9. [Google Scholar] [CrossRef]
- Dirsch, V.M.; Kiemer, A.K.; Wagner, H.; Vollmar, A.M. Effect of allicin and ajoene, two compounds of garlic, on inducible nitric oxide synthase. Atherosclerosis 1998, 139, 333–339. [Google Scholar] [CrossRef]
- Pedraza-Chaverrí, J.; Tapia, E.; Medina-Campos, O.N.; Granados, F.M. Garlic prevents hypertension induced by chronic inhibition of nitric oxide synthesis. Life Sci. 1998, 62, 71–77. [Google Scholar] [CrossRef]
- Breithaupt-Grögler, K.; Ling, M.; Boudoulas, H.; Belz, G.G. Protective effect of chronic garlic in take on elastic properties of aorta in the elderly. Circulation 1997, 96, 2649–2655. [Google Scholar] [CrossRef] [PubMed]
- Kojima, R.; Toyama, Y.; Ohnishi, S.T. Protective effects of an aged garlic extract on doxorubicin-induced cardiotoxicity in the mouse. Nutr. Cancer 1994, 22, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.K.; Dinda, A.K.; Manchanda, S.C.; Maulik, S.K. Chronic garlic administration protects rat heart against oxidative stress induced by ischemic reperfusion injury. BMC Pharmacol. 2002, 2, 16. [Google Scholar] [CrossRef]
- Rubio-Ruiz, M.E.; Pérez-Torres, I.; Soto, M.E.; Pastelín, G.; Guarner-Lans, V. Aging in blood vessels. Medicinal agents for systemic arterial hypertension in the elderly. Ageing Res. Rev. 2014, 18, 132–147. [Google Scholar] [CrossRef] [PubMed]
- Manna, P.; Jain, S.K. Hydrogen sulfide and l-cysteine increase phosphatidylinositol 3,4,5-trisphosphate (PIP3) and glucose utilization by inhibiting phosphatase and tensin homolog (PTEN) protein and activating phosphoinositide 3-kinase (PI3K)/serine/threonine protein kinase (AKT)/protein kinase Cζ/λ (PKCζ/λ) in 3T3l1 adipocytes. J. Biol. Chem. 2011, 286, 39848–39859. [Google Scholar] [PubMed]
- King, A.L.; Polhemus, D.J.; Bhushan, S.; Otsuka, H.; Kondo, K.; Nicholson, C.K.; Bradley, J.M.; Islam, K.N.; Calvert, J.W.; Tao, Y.X.; et al. Hydrogen sulfide cytoprotective signaling is endothelial nitric oxide synthase-nitric oxide dependent. Proc. Natl. Acad. Sci. USA 2014, 111, 3182–3187. [Google Scholar] [CrossRef] [PubMed]
- Bradley, J.M.; Organ, C.L.; Lefer, D.J. Garlic-derived organic polysulfides and myocardial protection. J. Nutr. 2016, 146, 403S–409S. [Google Scholar] [CrossRef] [PubMed]
- Morihara, N.; Sumioka, I.; Moriguchi, T.; Uda, N.; Kyo, E. Aged garlic extract enhances production of nitric oxide. Life Sci. 2002, 71, 509–517. [Google Scholar] [CrossRef]
- Elkayam, A.; Mirelman, D.; Peleg, E.; Wilchek, M.; Miron, T.; Rabinkov, A.; Sadetzki, S.; Rosenthal, T. The effects of allicin and enalapril in fructose-induced hyperinsulinemic hyperlipidemic hypertensive rats. Am. J. Hypertens. 2001, 14, 377–381. [Google Scholar] [CrossRef]
- Hara, Y.; Noda, A.; Miyata, S.; Minoshima, M.; Sugiura, M.; Kojima, J.; Otake, M.; Furukawa, M.; Cheng, X.W.; Nagata, K.; et al. Effects of aged garlic extract on left ventricular diastolic function and fibrosis in a rat hypertension model. Exp. Anim. 2013, 62, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Blumand, J.; Fridovich, I. Inactivation of glutathione peroxidase by superoxide radical. Arch. Biochem. Biophys. 1985, 240, 500–508. [Google Scholar]
- Guerra, R.C.; Zuñiga-Muñoz, A.; Guarner, L.V.; Díaz-Díaz, E.; Tena Betancourt, C.A.; Pérez-Torres, I. Modulation of the activities of catalase, Cu-Zn, Mn superoxide dismutase, and glutathione peroxidase in adipocyte from ovariectomized female rats with metabolic syndrome. Int. J. Endocrinol. 2014, 2014, 175080. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D. Fish, mercury, selenium and cardiovascular risk: Current evidence and unanswered questions. Int. J. Environ. Res. Public Health 2009, 6, 1894–1916. [Google Scholar] [CrossRef] [PubMed]
- Benstoem, C.; Goetzenich, A.; Kraemer, S.; Borosch, S.; Manzanares, W.; Hardy, G.; Stoppe, C. Selenium and its supplementation in cardiovascular disease-what do we know? Nutrients 2015, 7, 3094–3118. [Google Scholar] [CrossRef] [PubMed]
- Colín-González, A.L.; Santana, R.A.; Silva-Islas, C.A.; Chánez-Cárdenas, M.E.; Santamaría, A.; Maldonado, P.D. The antioxidant mechanisms underlying the aged garlic extract and S-allylcysteine-induced protection. Oxid. Med. Cell. Longev. 2012, 2012, 907162. [Google Scholar] [CrossRef] [PubMed]
- Rybka, J.; Kupczyk, D.; Kędziora-Kornatowska, K.; Motyl, J.; Czuczejko, J.; Szewczyk-Golec, K.; Kozakiewicz, M.; Pawluk, H.; Carvalho, L.A.; Kędziora, J. Glutathione-related antioxidant defense system in elderly patients treated for hypertension. Cardiovasc. Toxicol. 2011, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ribas, V.; García-Ruiz, C.; Fernández-Checa, J.C. Glutathione and mitochondria. Front. Pharmacol. 2014, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Matsuda, M.; Fukuhara, A.; Komuro, R.; Shimomura, I. Dysregulated glutathione metabolism links to impaired insulin action in adipocytes. Am. J. Phys. Endocrinol. Metab. 2009, 296, E1326–E1334. [Google Scholar] [CrossRef] [PubMed]
- Ide, N.; Lau, B.H. Garlic compounds minimize intracellular oxidative stress and inhibit nuclear factor-κB activation. J. Nutr. 2001, 131, 1020S–1026S. [Google Scholar] [PubMed]
- Dillon, S.A.; Lowe, G.M.; Billington, D.; Rahman, K. Dietary supplementation with aged garlic extract reduces plasma and urine concentrations of 8-iso-prostaglandin F(2α) in smoking and nonsmoking men and women. J. Nutr. 2002, 132, 168–171. [Google Scholar] [PubMed]
- Sample Availability: Samples of the compounds Kyolic are not available from the authors.
| Variables | C + SS | C + AGE | MS + SS | MS + AGE |
|---|---|---|---|---|
| SBP (mmHg) | 122.3 ± 4.2 | 122.6 ± 1.5 | 142.0 ± 2.8 * | 127.9 ± 3.0 * |
| Glucose (mmol/L) | 7.1 ± 0.5 | 6.1 ± 0.4 | 6.5 ± 0.1 | 6.4 ± 0.4 |
| TG (mg/dL) | 82.0 ± 6.9 | 67.1 ± 7.4 | 121.2 ± 4.7 ** | 93.7 ± 7.9 * |
| Cholesterol (mg/dL) | 40.4 ± 3.0 | 33.7 ± 1.8 * | 44.1 ± 1.2 | 35.2 ± 1.7 * |
| Insulin (µU/mL) | 5.2 ± 0.6 | 6.2 ± 1.2 | 7.2 ± 0.3 † | 5.7 ± 0.5 † |
| HOMA index | 1.5 ± 0.2 | 1.6 ± 0.3 | 2.2 ± 0.1 † | 1.5 ± 0.1 † |
| Leptin (ng/mL) | 10.3 ± 0.7 | 11.1 ± 1.1 | 17.8 ± 1.3 ** | 12.4 ± 1.4 ** |
| Advanced glycation end products (µU/mL) | 420.8 ± 42.7 | 458.0 ± 67.8 | 1100.4 ± 103.9 ** | 643.0 ± 110.7 † |
| Produts of the nitric oxide pathway | ||||
| Citrulline (µmol/L) | 67.5 ± 6.1 | 77.9 ± 6.2 | 40.5 ± 5.2 ** | 74.6 ± 5.7 ** |
| NO3−/NO2− (µg/mL serum) | 25.6 ± 1.5 | 26.6 ± 2.4 | 21.2 ± 0.8 * | 24.9 ± 1.1 * |
| NO (pmol/mL perfusion liquid) | 20.5 ± 1.3 | 30.2 ± 1.9 ** | 17.1 ± 0.5 * | 27.7 ± 8.1 ** |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Torres, I.; Torres-Narváez, J.C.; Pedraza-Chaverri, J.; Rubio-Ruiz, M.E.; Díaz-Díaz, E.; Del Valle-Mondragón, L.; Martínez-Memije, R.; Varela López, E.; Guarner-Lans, V. Effect of the Aged Garlic Extract on Cardiovascular Function in Metabolic Syndrome Rats. Molecules 2016, 21, 1425. https://doi.org/10.3390/molecules21111425
Pérez-Torres I, Torres-Narváez JC, Pedraza-Chaverri J, Rubio-Ruiz ME, Díaz-Díaz E, Del Valle-Mondragón L, Martínez-Memije R, Varela López E, Guarner-Lans V. Effect of the Aged Garlic Extract on Cardiovascular Function in Metabolic Syndrome Rats. Molecules. 2016; 21(11):1425. https://doi.org/10.3390/molecules21111425
Chicago/Turabian StylePérez-Torres, Israel, Juan Carlos Torres-Narváez, José Pedraza-Chaverri, María Esther Rubio-Ruiz, Eulises Díaz-Díaz, Leonardo Del Valle-Mondragón, Raúl Martínez-Memije, Elvira Varela López, and Verónica Guarner-Lans. 2016. "Effect of the Aged Garlic Extract on Cardiovascular Function in Metabolic Syndrome Rats" Molecules 21, no. 11: 1425. https://doi.org/10.3390/molecules21111425
APA StylePérez-Torres, I., Torres-Narváez, J. C., Pedraza-Chaverri, J., Rubio-Ruiz, M. E., Díaz-Díaz, E., Del Valle-Mondragón, L., Martínez-Memije, R., Varela López, E., & Guarner-Lans, V. (2016). Effect of the Aged Garlic Extract on Cardiovascular Function in Metabolic Syndrome Rats. Molecules, 21(11), 1425. https://doi.org/10.3390/molecules21111425









