A Novel Aqueous Two Phase System Composed of Surfactant and Xylitol for the Purification of Lipase from Pumpkin (Cucurbita moschata) Seeds and Recycling of Phase Components
Abstract
:1. Introduction
2. Result and Discussion
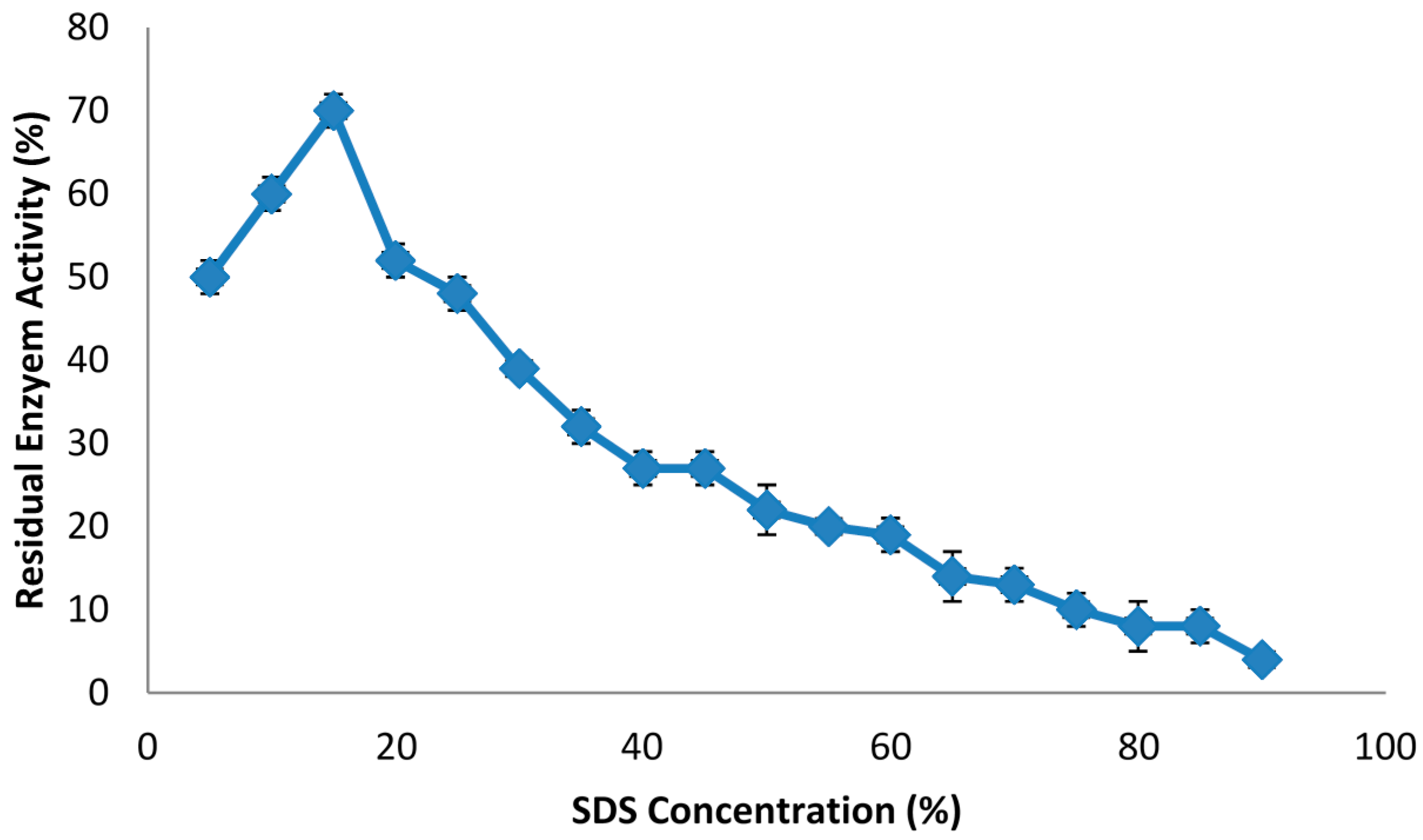
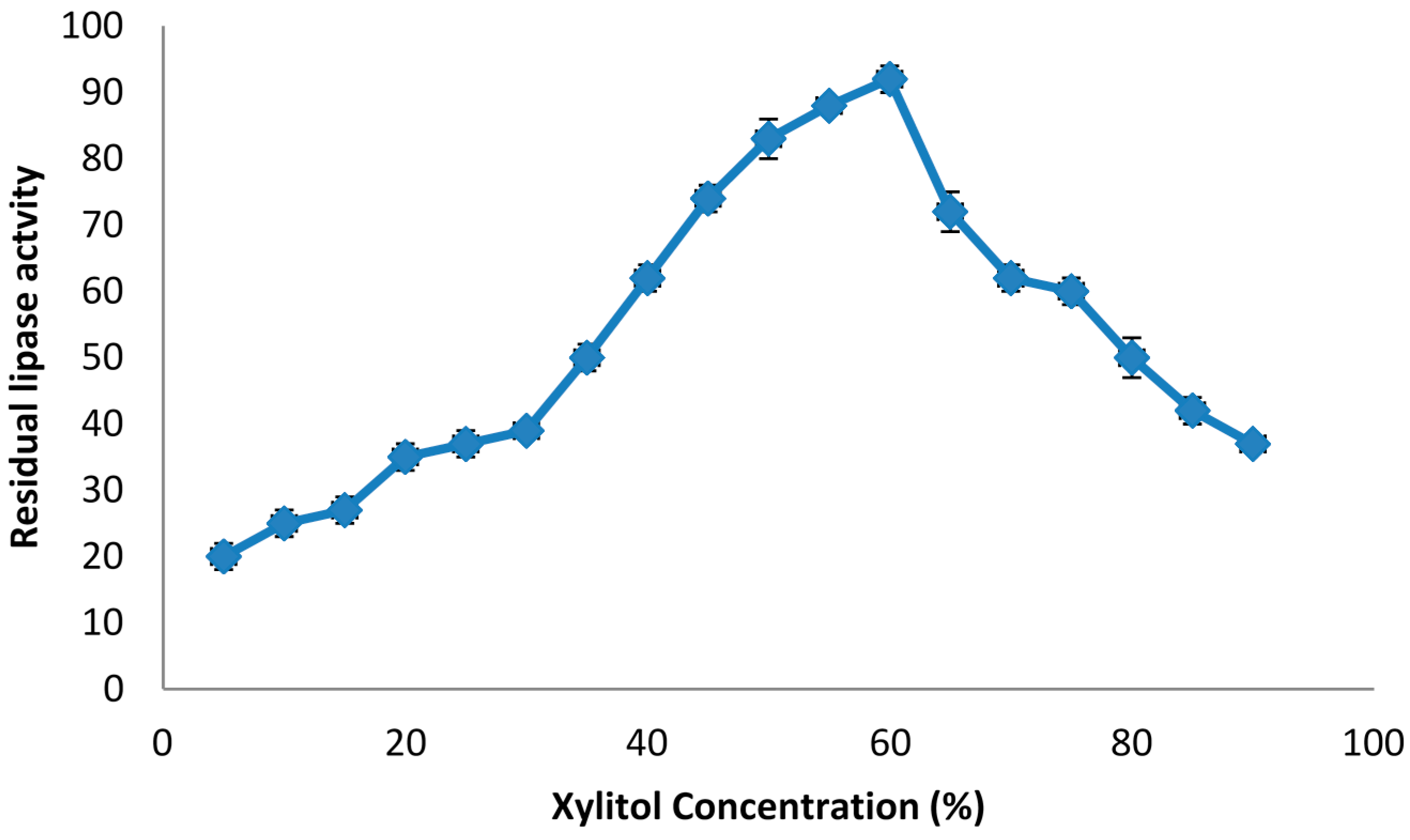
2.1. The Effect of Phase Components on Lipase Stability
| Phase Compositions | Concentration (% w/w) | Lipase Activity (U/mL) |
|---|---|---|
| Triton X-100 | 15 | 102.1 ± 0.23 a |
| 30 | 110.1 ± 0.22 a | |
| 45 | 118.4 ± 0.11 b | |
| 60 | 142.2 ± 0.01 c | |
| 75 | 103.2 ± 0.01 d | |
| Tween 80 | 15 | 82.3 ± 0.11 a |
| 30 | 81.1 ± 0.21 b | |
| 45 | 88.4 ± 0.22 c | |
| 60 | 95.6 ± 0.57 ab | |
| 75 | 92.3 ± 0.17 d | |
| SDS | 15 | 52.3 ± 0.32 a |
| 30 | 41.2 ± 1.21 b | |
| 45 | 38.2 ± 0.03 a | |
| 60 | 32.1 ± 0.32 a | |
| 75 | 27.2 ± 0.01 a | |
| Xylitol | 15 | 132.1 ± 0.07 a |
| 30 | 112.6 ± 1.10 b | |
| 45 | 98.1 ± 0.22 ab | |
| 60 | 92.3 ± 0.52 c | |
| 75 | 84.2 ± 0.12 d |
2.2. Selection of the Optimal Surfactants/Xylitol ATPS for the Lipase Partitioning
| System | Concentration of Surfactant/Xylitol (% w/w) | TLL (% w/w) | Selectivity | Purification Factor |
|---|---|---|---|---|
| TritonX-100/xylitol | 15/17 | 54.3 | 51.01 ± 0.2 a | 7.31 ± 1.1 a |
| 22/25 | 56.2 | 88.11 ± 0.1 b | 10.09 ± 1.1 b | |
| 26/28 | 59.1 | 66.15 ± 0.3 c | 8.12 ± 0.3 c | |
| 32/30 | 63.4 | 31.01 ± 1.1 d | 7.84 ± 0.2 d | |
| Tween-80/xylitol | 17/18 | 22.4 | 22.20 ± 1.1 e | 3.33 ± 1.1 ab |
| 22/20 | 33.3 | 32.12 ± 0.2 e | 3.12 ± 1.1 e | |
| 24/22 | 42.4 | 22.11 ± 0.1 ed | 2.81 ± 0.1 d | |
| 29/25 | 47.2 | 18.11 ± 0.3 e | 1.62 ± 0.3 e | |
| SDS/xylitol | 13/12 | 33.2 | 12.23 ± 1.1 j | 2.12 ± 0.4 g |
| 20/18 | 35.4 | 9.15 ± 2.3 k | 1.07 ± 0.2 h | |
| 22/19 | 46.1 | 7.11 ± 1.1 k | 0.72 ± 0.3 i | |
| 27/25 | 52.4 | 5.42 ± 0.2 jk | 0.34 ± 1.1 j |
| Triton X-100 (% w/w) | Xylitol (% w/w) | Purification Factor | Yield (%) |
|---|---|---|---|
| 22 | 18 | 3.11 ± 0.2 a | 75.3 ± 0.2 a |
| 22 | 20 | 4.12 ± 1.1 b | 66.6 ± 0.3 b |
| 22 | 22 | 5.21 ± 0.1 c | 59.4 ± 10 c |
| 22 | 24 | 5.13 ± 0.2 d | 48.3 ± 1.3 ab |
| 22 | 26 | 5.10 ± 1.1 e | 43.3 ± 1.2 d |
| 23 | 18 | 6.01 ± 0.1 f | 32.6 ± 0.2 e |
| 23 | 20 | 6.13 ± 1.1 g | 23.4 ± 0.1 f |
| 23 | 22 | 6.42 ± 1.1 g | 21.2 ± 1.3 g |
| 23 | 24 | 7.72 ± 0.2 h | 18.3 ± 0.1 g |
| 23 | 26 | 7.81 ± 0.1 b | 10.6 ± 0.2 e |
| 24 | 18 | 8.83 ± 1.1 ab | 68.1 ± 0.1 f |
| 24 | 20 | 12.28 ± 0.1 i | 93.1 ± 0.1 g |
| 24 | 22 | 10.32 ± 0.2 i | 82.2 ± 0.1 h |
| 24 | 24 | 10.11 ± 0.1 i | 64.3 ± 0.2 i |
| 24 | 26 | 9.09 ± 0.2 i | 53.2 ± 1.1 j |
| 25 | 18 | 8.23 ± 0.1 j | 31.3 ± 1.1 k |
| 25 | 20 | 4.21 ± 1.1 i | 28.3 ± 0.3 k |
| 25 | 22 | 3.32 ± 0.2 k | 22.6 ± 1.2 j |
| 25 | 24 | 2.12 ± 0.3 l | 18.4 ± 1.0 l |
| 25 | 26 | 1.01 ± 2.1 m | 14.3 ± 0.2 m |
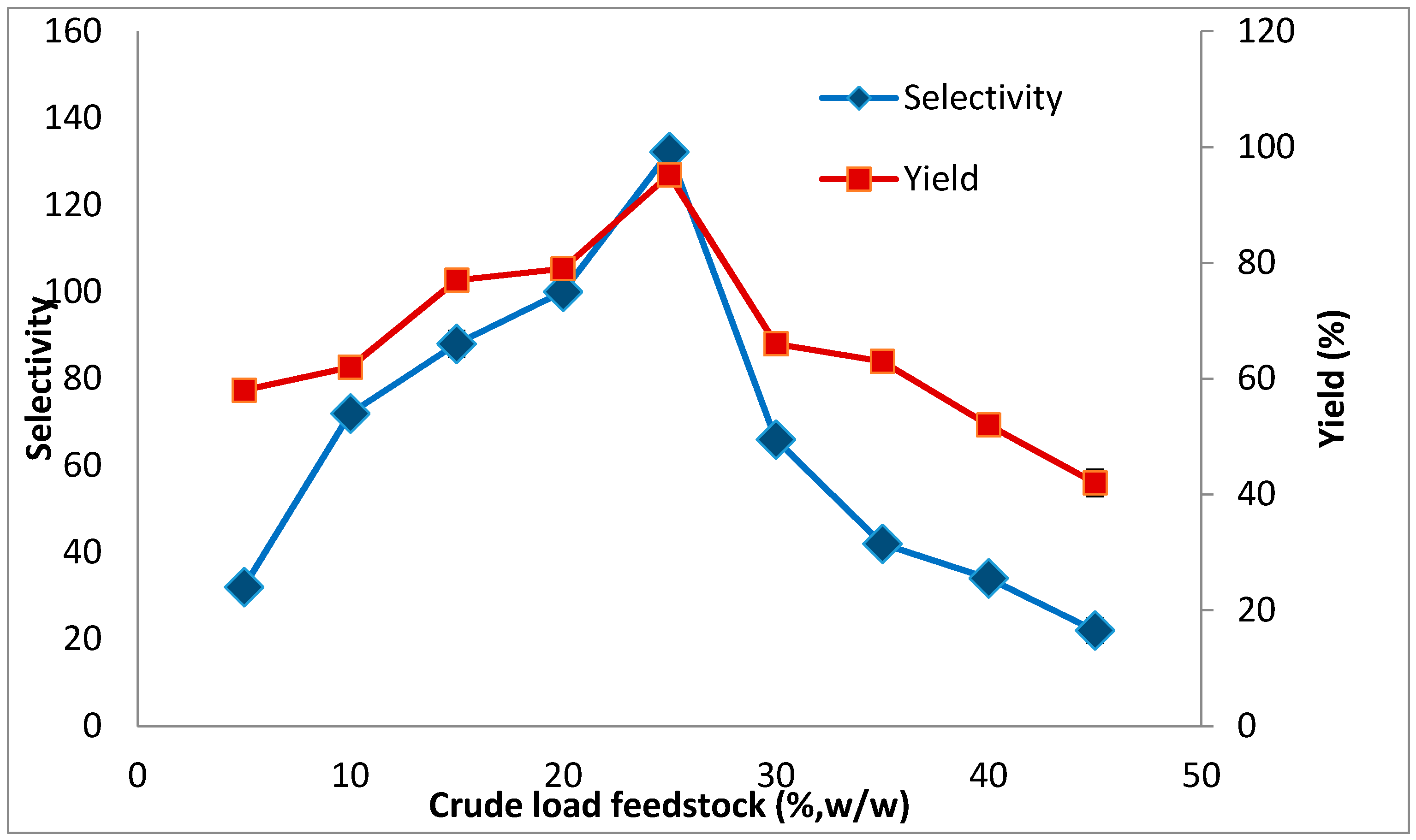
2.3. The Effect of Crude Feedstock Concentration on Lipase Partitioning
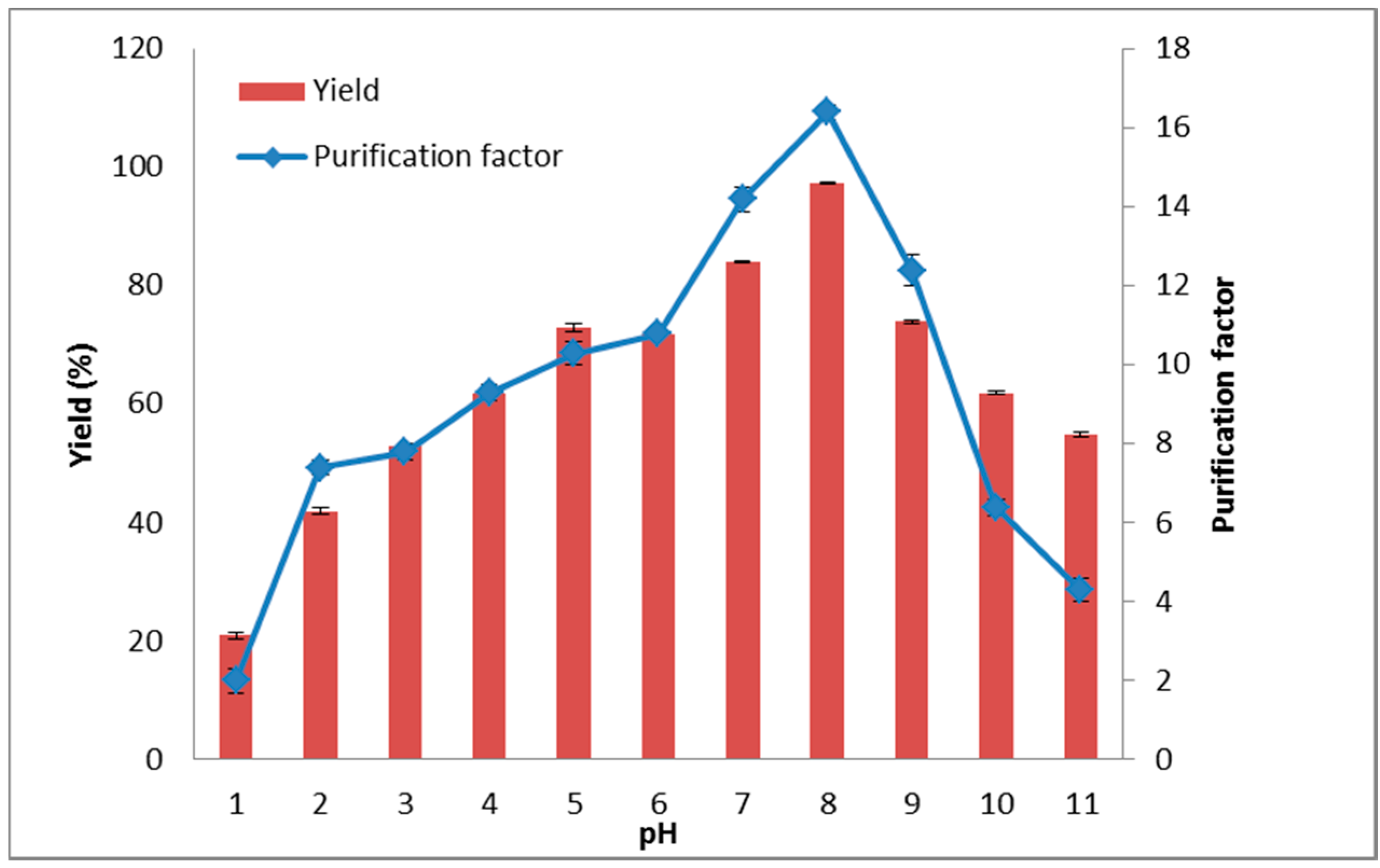
2.4. The Effect of System pH on Lipase Partitioning
2.5. Recycling of Phase Components
| System | Initial | Recycle Systems | ||||||
|---|---|---|---|---|---|---|---|---|
| First | Second | Third | Forth | Fifth | Sixth | Seventh | ||
| Recovery of surfactant (%) | 99.4 | 98.8 ± 0.02 | 98.5 ± 1.1 | 98.1 ± 0.21 | 97.8 ± 0.06 | 97.3 ± 0.05 | 83.2 ± 1.15 | 62.2 ± 1.11 |
| Recovery of xylitol (%) | 99.3 | 99.1 ± 0.03 | 98.6 ± 0.2 | 97.8 ± 0.01 | 97.2 ± 0.13 | 97.0 ± 0.11 | 88.2 ± 0.12 | 78.4 ± 0.11 |
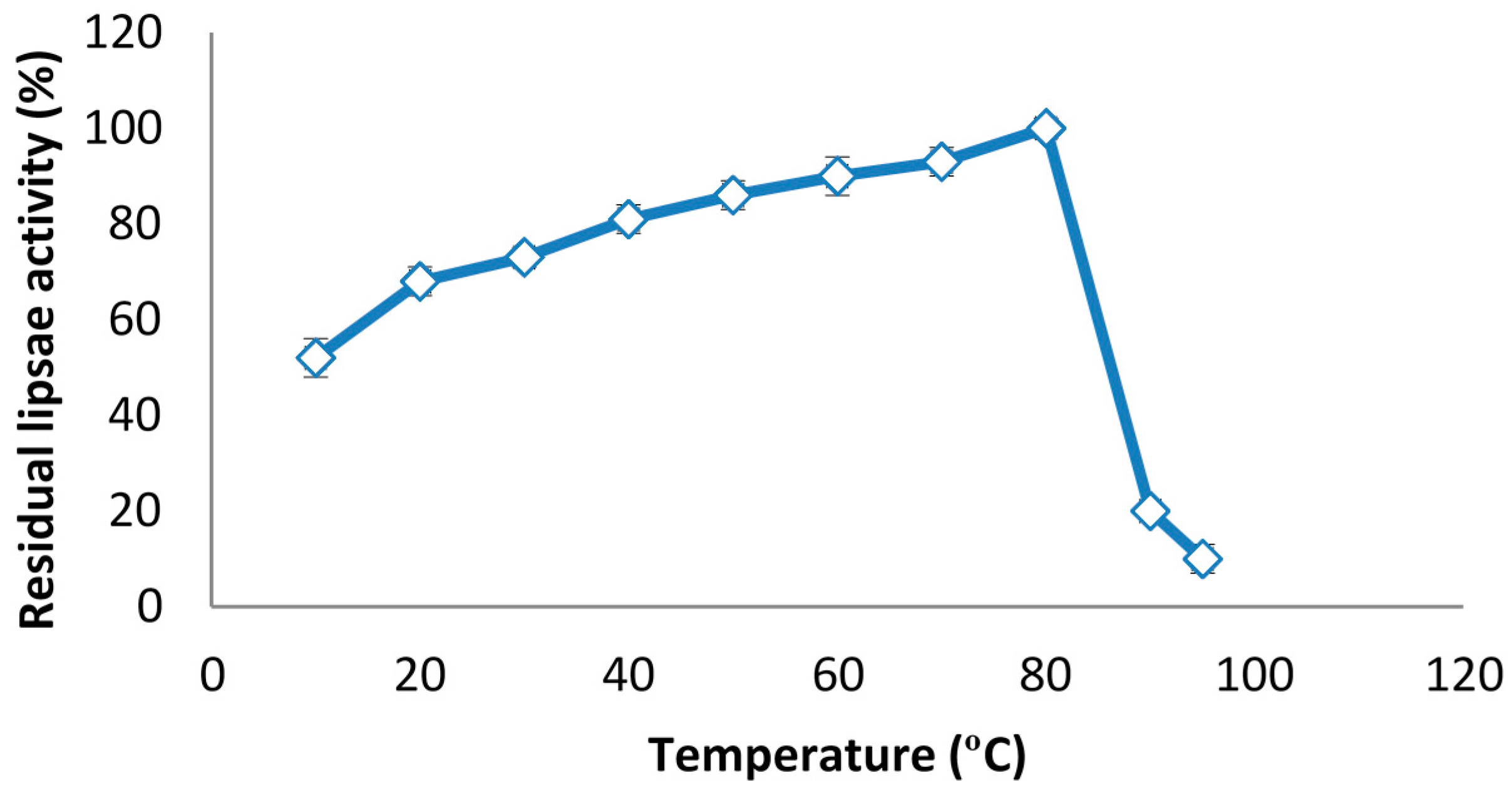
2.6. The Effect of Temperature on Lipase Activity
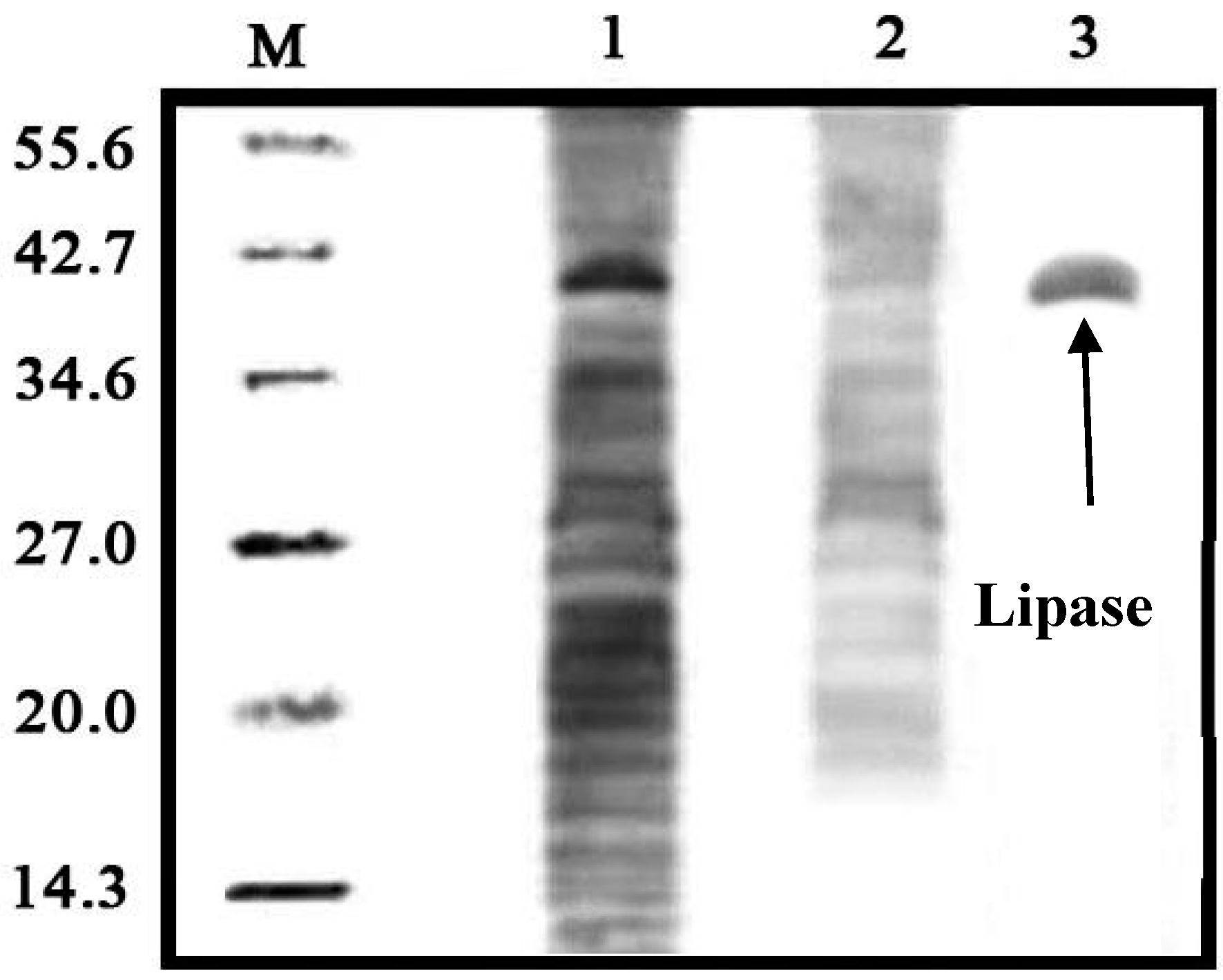
2.7. Lipase Recovery
3. Experimental Section
3.1. Materials
3.2. Lipase Extraction from Pumpkin Seed
3.3. ATPS Composed of Non-Ionic Surfactant and Xylitol
3.4. Lipase Assay and Protein Determination
3.5. Determination of Optimum Temperature of Lipase
3.6. Determination of the Enzyme Partitioning
3.7. Recycling of Phase Components
3.8. Electrophoresis
3.9. Statistical Design and Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
References
- Galante, Y.M.; Formantici, C. Enzyme applications in detergency and in manufacturing industries. Curr. Org. Chem. 2003, 7, 1399–1422. [Google Scholar] [CrossRef]
- Prinza, A.; Zeiner, T.; Vössinga, T.; Schüttmannb, I.; Zornb, H.; Góraka, A. Experimental investigation of laccase purification using aqueous two-phase extraction. Chem. Eng. 2012, 27, 349–354. [Google Scholar]
- Silvério, S.C.; Wegrzyn, A.; Lladosa, E.; Rodríguez, O.; Macedo, E.A. Effect of aqueous two-phase system constituents in different poly (ethylene glycol)–salt phase diagrams. J. Chem. Eng. Data 2012, 57, 1203–1208. [Google Scholar] [CrossRef]
- Benavides, J.; Rito-Palomares, M. Practical experiences from the development of aqueous two-phase processes for the recovery of high value biological products. J. Chem. Technol. Biotechnol. 2008, 83, 133–142. [Google Scholar] [CrossRef]
- Asenjo, J.A.; Andrews, B.A. Aqueous two-phase systems for protein separation: A perspective. J. Chromatogr. A 2011, 1218, 8826–8835. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, J.M.P.; Souza, R.L.; Fricks, A.T.; Zanin, G.M.; Soares, C.M.F.; Lima, Á.S. Purification of lipase produced by a new source of Bacillus in submerged fermentation using an aqueous two-phase system. J. Chromatogr. B 2011, 879, 3853–3858. [Google Scholar] [CrossRef] [PubMed]
- Schnitzhofera, W.; Weber, H.J.; Vrsanska, M.; Biely, P.; Cavaco-Paulo, A.; Guebitz, G.M. Purification and mechanistic characterisation of two pectinases from Sclerotium rolfsii. J. Enzym. Microb. Technol. 2007, 40, 1739–1747. [Google Scholar] [CrossRef] [Green Version]
- Platis, D.; Labrou, N.E. Development of an aqueous two-phase partitioning system for fractionating therapeutic proteins from tobacco extract. J. Chromatogr. A 2006, 1128, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Goja, A.M.; Yang, H.; Cui, M.; Li, C. Aqueous Two-Phase Extraction Advances for Bioseparation. J. Bioprocess. Biotechnol. 2013, 4, 1–8. [Google Scholar]
- Patel, V.; Nambiar, S.; Madamwar, D. An extracellular solvent stable alkaline lipase from Pseudomonas sp. DMVR46: Partial purification, characterization and application in non-aqueous environment. Process Biochem. 2014, 49, 1673–1681. [Google Scholar] [CrossRef]
- Aravindan, R.; Anbumathi, P.; Viruthagiri, T. Lipase applications in food industry. Indian J. Biotechnol. 2007, 6, 141–158. [Google Scholar]
- Kawakami, K.; Oda, Y.; Takahashi, R. Application of a Burkholderia cepacia lipase-immobilized silica monolith to batch and continuous biodiesel production with a stoichiometric mixture of methanol and crude Jatropha oil. Biotechnol. Biofuels 2011, 4, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Velez, A.M.; Horta, A.C.L.; da Silva, A.J.; Iemma, M.R.D.C.; Giordano, R.D.L.C.; Zangirolami, T.C. Enhanced production of recombinant thermo-stable lipase in Escherichia coli at high induction temperature. Protein Expr. Purif. 2013, 90, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Calli, F.; Huan, S.; Quanhong, L. A review on pharmacological activities and utilization technologies of pumpkin. Plant Foods. Hum. Nutr. 2006, 61, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Fissore, E.N.; Matkovic, L.; Wider, E.; Rojas, A.M.; Gerschenson, L.N. Rheological properties of pectin-enriched products isolated from butternut (Cucurbita moschata Duch ex Poiret). LWT-Food Sci. Technol. 2009, 42, 1413–1421. [Google Scholar] [CrossRef]
- Maran, P.J.; Mekala, V.; Manikandan, S. Modeling and optimization of ultrasound-assisted extraction of polysaccharide from Cucurbita moschata. Carbohydr. Polym. 2013, 92, 2018–2026. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.C.A.; Araújo, K.B.; Zubiolo, C.; Soares, C.M.F.; Lima, A.S.; de Santana Aquino, L.C.L. Microbial lipase obtained from the fermentation of pumpkin seeds: Immobilization potential of hydrophobic matrices. Acta Sci. Technol. 2014, 36, 193–201. [Google Scholar] [CrossRef]
- Mitra, P.; Ramaswamy, H.S.; Chang, K.S. Pumpkin (Cucurbita maxima) seed oil extraction using supercritical carbon dioxide and physicochemical properties of the oil. J. Food. Eng. 2009, 95, 208–213. [Google Scholar] [CrossRef]
- Jiao, J.; Li, Z.G.; Gai, Q.Y.; Li, X.J.; Wei, F.Y.; Fu, Y.J.; Ma, W. Microwave-assisted aqueous enzymatic extraction of oil from pumpkin seeds and evaluation of its physicochemical properties, fatty acid compositions and antioxidant activities. Food. Chem. 2014, 147, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Amid, M.; Murshid, F.S.; Manap, M.Y.; Hussin, M. A Novel Aqueous Micellar Two-Phase System Composed of Surfactant and Sorbitol for Purification of Pectinase Enzyme from Psidium guajava and Recycling Phase Components. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Salabat, A.; Far, M.R.; Moghadam, S.T. Partitioning of amino acids in surfactant based aqueous two-phase systems containing the nonionic surfactant (triton X-100) and salts. J. Solut. Chem. 2011, 40, 61–66. [Google Scholar] [CrossRef]
- Amid, M.; Manap, M.Y.A.; Shuhaimi, M. Purification of a novel protease enzyme from kesinai plant (Streblus asper) leaves using a surfactant-salt aqueous micellar two-phase system: A potential low cost source of enzyme and purification method. Eur. Food. Res. Technol. 2013, 237, 601–608. [Google Scholar] [CrossRef]
- Ooi, C.W.; Tey, B.T.; Hii, S.L.; Kamal, S.M.M.; Lan, J.C.W.; Ariff, A.; Ling, T.C. Purification of lipase derived from Burkholderia pseudomallei with alcohol/salt-based aqueous two-phase systems. Process Biochem. 2009, 44, 1083–1087. [Google Scholar] [CrossRef]
- Escobar, S.; Bernal, C.; Mesa, M. Kinetic study of the colloidal and enzymatic stability of β-galactosidase, for designing its encapsulation route through sol-gel route assisted by Triton X-100 surfactant. Biochem. Eng. J. 2013, 75, 32–38. [Google Scholar] [CrossRef]
- Mirjana, G.A. Partitioning of pectinase produced by Polyporus squamosus in aqueous two phase system polyethylene glycol 4000 crude dextran at different initial pH values. J. Carbohydr. Polym. 2004, 56, 295–300. [Google Scholar]
- Padma, V.I.; Laxmi, A. Enzyme stability and stabilization-Aqueous and non-aqueous environment. Process Biochem. 2008, 43, 1019–1032. [Google Scholar]
- Das, D.; Das, P.K. Superior activity of structurally deprived enzyme-carbon nanotube hybrids in cationic reverse micelles. Langmuir 2009, 25, 4421–4428. [Google Scholar] [CrossRef] [PubMed]
- Nasirpour, N.; Mousavi, S.M.; Shojaosadati, S.A. A novel surfactant-assisted ionic liquid pretreatment of sugarcane bagasse for enhanced enzymatic hydrolysis. Bioresour. Technol. 2014, 169, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Granström, T.B.; Izumori, K.; Leisola, M. A rare sugar xylitol. Part II: Biotechnological production and future applications of xylitol. Appl. Microbiol. Biotechnol. 2007, 74, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Ermakova, E.A.; Zakhartchenko, N.L.; Zuev, Y.F. Effect of surface potential of reverse micelle on enzyme–substrate complex formation. Colloids Surf. A Physicochem. Eng. Asp. 2007, 317, 297–302. [Google Scholar] [CrossRef]
- Wang, W.W.; Yuan, X.Z.; Zeng, G.M.; Liang, Y.S.; Chao, Y. Enzymatic hydrolysis of cellulose in reverse micelles. Environ. Sci. 2010, 31, 2202–2207. [Google Scholar]
- Bonomo, R.C.F.; Minim, L.A.; Coimbra, J.S.R.; Fontan, R.C.I.; Mendes da Silva, L.H.; Minim, V.P.R. Hydrophobic interaction adsorption of whey proteins: Effect of temperature and salt concentration and thermodynamic analysis. J. Chromatogr. B 2006, 844, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Ketnawa, S.; Benjakul, S.; Ling, T.C.; Martínez-Alvarez, O.; Rawdkuen, S. Enhanced recovery of alkaline protease from fish viscera by phase partitioning and its application. Chem. Cent. J. 2013, 7, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaushik, V.; Roos, Y.H. Limonene encapsulation in freeze-drying of Gum Arabic sucrose-gelatin systems. LWT-Food Sci. Technol. 2007, 40, 1381–1391. [Google Scholar] [CrossRef]
- Liu, J.G.; Xing, J.M.; Shen, R.; Yang, C.L.; Liu, H.Z. Reverse micelles extraction of nattokinase from fermentation broth. Biochem. Eng. J. 2004, 21, 273–278. [Google Scholar] [CrossRef]
- Streitner, N.; Flaschel, V.C. Reverse micellar extraction systems for the purification of pharmaceutical grade plasmid DNA. J. Biotechnol. 2007, 131, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Shahbaz, H.; Omidinia, M.E. Process integration for the recovery and purification of recombinant Pseudomonas fluorescens proline dehydrogenase using aqueous two-phase systems. J. Chromatogr. B 2013, 15, 11–17. [Google Scholar]
- Amid, M.; Manap, M.Y.A.; Shuhaimi, M. Purification of pectinase from mango (Mangifera indica L. cv. Chokanan) waste using an aqueous organic phase system: A potential low cost source of the enzyme. Chromatogr. B 2013, 931, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Andrews, B.A.; Schmidt, A.S.; Asenjo, J.A. Correlation for the partition behavior of proteins in aqueous two-phase systems: Effect of surface hydrophobicity and charge. Biotechnol. Bioeng. 2005, 90, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Amid, M.; Manap, Y.; Zohdi, N.K. A Novel Aqueous Two Phase System Composed of a Thermo-Separating Polymer and an Organic Solvent for Purification of Thermo-Acidic Amylase Enzyme from Red Pitaya (Hylocereus polyrhizus) Peel. Molecules 2014, 19, 6635–6650. [Google Scholar] [CrossRef] [PubMed]
- Show, P.L.; Ooi, C.W.; Anuar, M.S.; Ariff, A.; Yusof, Y.A.; Chen, S.K.; Mohamad Anuar, M.S.; Ling, T.C. Recovery of lipase derived from Burkholderia cenocepacia ST8 using sustainable aqueous two-phase flotation composed of recycling hydrophilic organic solvent and inorganic salt. Sep. Purif. Technol. 2013, 110, 112–118. [Google Scholar] [CrossRef] [Green Version]
- Zhong, H.; Zeng, G.M.; Yuan, X.Z.; Fu, H.Y.; Huang, G.H.; Ren, F.Y. Adsorption of dirhamnolipid on four microorganisms and the effect on cell surface hydrophobicity. Appl. Microb. Biotechnol. 2007, 77, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Zohdi, N.K.; Amid, M. Optimization of Extraction of Novel Pectinase Enzyme Discovered in Red Pitaya (Hylocereus polyrhizus) Peel. Molecules 2013, 18, 14366–14380. [Google Scholar] [CrossRef] [PubMed]
- Silva, G.M.D.M.; Marques, D.D.A.V.; Porto, T.S.; Lima Filho, J.L.; Teixeira, J.A.C.; Porto, A.L.F.; Pessoa-Júnior, A. Extraction of fibrinolytic proteases from Streptomyces sp. DPUA1576 using PEG-phosphate aqueous two-phase systems. Fluid Phase Equilibria 2013, 339, 52–57. [Google Scholar] [CrossRef]
- Silva, W.O.B.; Mitidieri, S.; Schrank, A.; Vainstein, M.H. Production and extraction of an extracellular lipase from the entomopathogenic fungus Metarhizium anisopliae. Process Biochem. 2005, 40, 321–326. [Google Scholar] [CrossRef]
- Barbosa, H.; Slater, N.K.; Marcos, J.C. Protein quantification in the presence of poly (ethylene glycol) and dextran using the Bradford method. J. Anal. Biochem. 2009, 395, 108–110. [Google Scholar] [CrossRef] [PubMed]
- Mohamed Ali, S.; Ling, T.C.; Muniandy, S.; Tan, Y.S.; Raman, J.; Sabaratnam, V. Recovery and partial purification of fibrinolytic enzymes of Auricularia polytricha (Mont.) Sacc by an aqueous two-phase system. J. Sep. Purif. Technol. 2014, 122, 359–366. [Google Scholar] [CrossRef]
- Amid, M.; Manap, M.Y.A.; Shuhaimi, M. Optimization of Freeze Drying Conditions for Purified Pectinase from Mango (Mangifera indica cv. Chokanan) Peel. Molecules 2012, 13, 2939–2950. [Google Scholar]
- Sample Availability: Samples of the pumpkin (Curcubita moschata) seed are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amid, M.; Manap, M.Y.; Hussin, M.; Mustafa, S. A Novel Aqueous Two Phase System Composed of Surfactant and Xylitol for the Purification of Lipase from Pumpkin (Cucurbita moschata) Seeds and Recycling of Phase Components. Molecules 2015, 20, 11184-11201. https://doi.org/10.3390/molecules200611184
Amid M, Manap MY, Hussin M, Mustafa S. A Novel Aqueous Two Phase System Composed of Surfactant and Xylitol for the Purification of Lipase from Pumpkin (Cucurbita moschata) Seeds and Recycling of Phase Components. Molecules. 2015; 20(6):11184-11201. https://doi.org/10.3390/molecules200611184
Chicago/Turabian StyleAmid, Mehrnoush, Mohd Yazid Manap, Muhaini Hussin, and Shuhaimi Mustafa. 2015. "A Novel Aqueous Two Phase System Composed of Surfactant and Xylitol for the Purification of Lipase from Pumpkin (Cucurbita moschata) Seeds and Recycling of Phase Components" Molecules 20, no. 6: 11184-11201. https://doi.org/10.3390/molecules200611184
APA StyleAmid, M., Manap, M. Y., Hussin, M., & Mustafa, S. (2015). A Novel Aqueous Two Phase System Composed of Surfactant and Xylitol for the Purification of Lipase from Pumpkin (Cucurbita moschata) Seeds and Recycling of Phase Components. Molecules, 20(6), 11184-11201. https://doi.org/10.3390/molecules200611184





