Abstract
In further study of our series of six-membered ring-containing nucleic acids, different 1',3'-di-O-methyl altropyranoside nucleoside analogs (DMANA) were synthesized comprising all four base moieties, adenine, cytosine, uracil and guanine. Following assembly into oligonucleotides (ONs), their affinity for natural oligonucleotides was evaluated by thermal denaturation of the respective duplexes. Data were compared with results obtained previously for both anhydrohexitol (HNAs) and 3'-O-methylated altrohexitol modified ONs (MANAs). We hereby demonstrate that ONs modified with DMANA monomers, unlike some of our previously described analogues with constrained 6-membered hexitol rings, did not improve thermodynamic stability of dsRNA complexes, most probably in view of an energetic penalty when forced in the required 1C4 pairing conformation. Overall, a single incorporation was more or less tolerated or even positive for the adenine congener, but incorporation of a second modification afforded a slight destabilization (except for A), while a fully modified sequence displayed a thermal stability of −0.3 °C per modification. The selectivity of pairing remained very high, and the new modification upon incorporation into a DNA strand, strongly destabilized the corresponding DNA duplexes. Unfortunately, this new modification does not bring any advantage to be further evaluated for antisense or siRNA applications.
1. Introduction
Gene silencing has become a standard technique for studying gene functions or in trying to obtain therapeutic effects and theoretically can be attained by interfering with transcription (via formation of triple stranded complexes [1] or translation processes. The latter can be obtained via steric blocking antisense oligonucleotides (ASOs) or via mRNA cleavage of double stranded complexes with RNAseH activating ASOs [2,3,4]. However, the vast majority of researchers nowadays have turned to the use of RNA interference (RNAi)-based strategies, which has recently become the technique of choice to silence gene expression in mammalian cell culture and is envisaged as a first choice for therapeutic treatment as well [5,6,7]. However, we need to point out that at the moment only one aptamer [8] and one antisense oligonucleotide have been effectively FDA approved for gene silencing [9], and also exon-skipping oligonucleotides are receiving considerable attention [10].
In cell culture in general unmodified siRNAs are highly efficient, however for in vivo application some chemical modifications are warranted to stabilise the siRNAs and to increase their selectivity and to promote delivery [11,12]. In the past, we and others have studied a wide variety of strategies for both ASO and siRNA modification as reviewed several times [13,14,15].
Our group has been very successful in increasing the affinity for RNA using modified building blocks based on 6-membered hexitol rings which resulted in the hexitol nucleic acids series [16,17,18,19]. However, the LNA monomers of the Wengel group [20] consistently showed the strongest affinity for RNA, and the series comprises many alternative structures [21]. Overall, both our hexitol nucleic acids and the LNA series of compounds take on a pre-organized conformation, fitting the A-form of dsRNA and rationalizing the strong hybridization characteristics noticed.
Herein, hexitol nucleic acids (HNA, Figure 1) are composed of 2,3-dideoxy-d-arabino-hexitol units with a nucleobase situated in the 2-(S)-position (in cis to the hydroxymethyl substituent as in natural nucleosides). Addition of a supplementary hydroxyl at the 3'-α-position resulted in d-altritol nucleic acid (ANA, Figure 1) analogs with increased affinity for RNA strands [18,22]. More recently, we finally reported on the 3'-O-methylated ANA congeners (MANA, Figure 1) resulting in a further increase of 0.5 °C/modification when evaluating melting temperatures (Tm) upon hybridisation to RNA [23]. Herein, both the heterocyclic base and the 3'-O-methyl moiety are located in an axial position with a 4C1 conformation. However, assembly of the hexitol series of nucleosides is long-routed starting with synthesis of the 1,5-anhydrohexitol ring. In view of the positive results obtained for the MANA series of congeners and the abundance of cheap α-d-methylglucoside, we now planned to prepare and evaluate “bis-methylated altritol nucleosides”, or more correctly di-O-methylated altropyranoside nucleic acids (DMANA, Figure 1).
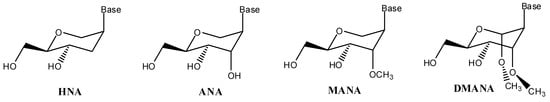
Figure 1.
Structures of the different hexitol based nucleic acids (HNA, ANA and MANA) as discussed above and the newly envisaged structure DMANA, based on methyl-altropyranoside.
2. Results and Discussion
2.1. Chemical Synthesis of the New Building Blocks
The 1'-O-methylglycosidic protected analogues 4, 6 and 9 (Scheme 1) were obtained starting from ubiquitous methyl glucopyranoside 1, which in three steps was converted to 2 in 51.4% overall yield according to literature procedures [24]. Herein, regioselective epoxide ring opening of 2 with the sodium salts of uracil or adenine in DMF at 120–130 °C afforded the corresponding altrohexitol derivatives 3 and 7 in 66%–85% yield. Chemoselective methylation of 7 and 3 was accomplished using NaH, MeI in dry THF at low temperature for 1 h to afford the methylated nucleosides 4 and 8 in 65% and 75% yield, respectively. The selective O- vs. N-methylation mainly depends upon the dielectric constant of the solvent [23,25] and the stability of sodium-enolate chelation [26]. Hence, low dielectric constant and high chelation stabilizing capacity of THF afforded a higher O-selectivity compared to DMF. The one pot conversion of compound 3 to the triazolide derivative [27] using 1,2,4-triazole, POCl3, and triethylamine, and subsequent treatment with aqueous ammonia/dioxane (1:1) at ambient temperature for 18 h yielded the cytidine derivative (5) with 58% (overall yield for 2 steps). Base protection of 5 and 8 using benzoylchloride in pyridine at rt for 3 h afforded 6 and 9 in 83 and 88% yield, respectively.
Due to the scalability, solubility and reproducibility of the reaction it proved advantageous to use the guanine derivative 10 for the epoxide ring opening reaction. The latter was obtained via Mitsunobu reaction of N2-acetylguanine [28] and 2-(trimethylsilyl)ethanol in analogy with the previously described protocol for O6-[2-(p-nitrophenyl)ethyl]guanine [29]. Selective epoxide ring opening was accomplished with the lithium salt of 10 (Scheme 2) in DMF at 130 °C and afforded 41% of 11 along with 26% of recovered 10. Remarkably, the acetyl protection was lost in 11 upon the prolonged heating in DMF. Further chemoselective methylation using NaH (60%) and MeI in DMF and DCM at low temperature for 4 h gave 12 in 94% yield. Deprotection of 12 was done by 1 M TBAF in THF at rt for 2 h to yield 75% of 13. The more base labile N2-dimethylformamidine (dmf) [30,31,32] group was introduced using N,N-dimethylformamide diethylacetal in methanol under reflux for 12 h to afford 14 with 85% yield.
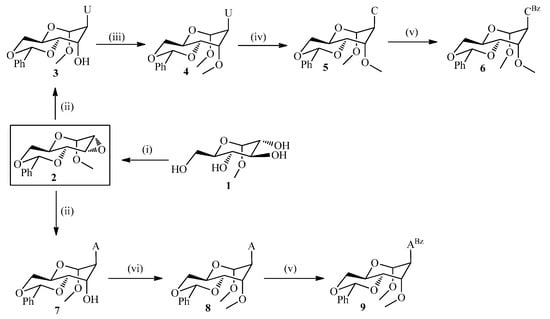
Scheme 1.
Synthetic scheme of the protected DMANA congeners for uracil, cytosine and adenine.
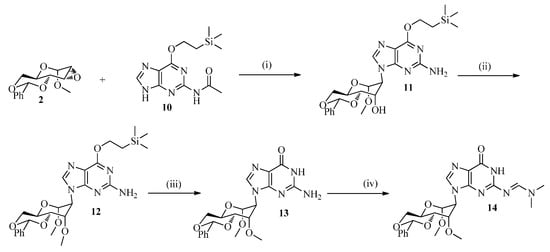
Scheme 2.
Synthetic scheme for the protected guanine containing analog.
Deprotection of the benzylidene protecting group under mild conditions using AcOH:H2O (3:1) at 45 °C for 12 h afforded 15a–d in 50%–94% yield (Scheme 3), which was followed by classical dimethoxytritylation and phosphitylation. Hereto, the nucleosides 15a–d were selectively protected at the 6'-OH by reaction with DMTrCl in pyridine at rt for 3 h to furnish the corresponding protected derivatives 16a–d in 74%–93% yield. Phosphitylation of 16a–d at the 4'-OH with 2-cyanoethyl N,N-diisopropylchlorophosphoramidite in anhydrous CH2Cl2 at 0 °C for 1.5 h afforded the corresponding phosphoramidite building blocks 17a–d in 65%–93% yield, to be used for oligomer assembly. Assembly of all oligonucleotides and purification was carried out as described before [33].
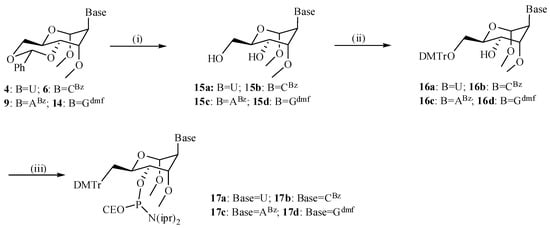
Scheme 3.
Scheme for assembly of the different phosporamidites.
2.2. Oligonucleotide Affinity Measurements
First, following incorporation of a single DMANA modification into an dsRNA nonamer sequence [5'-GCGU-X*-UGCG/5'-CGCAYACGC], the respective affinities for complementary RNA were studied in a 0.1 M NaCl buffer and were compared with the melting temperatures (Tm) of previously studied six-membered ring structures substituting for the ribose ring (Table 1). Within the context of all four natural bases, the HNA (anhydrohexitol) and ANA (altritol) substitutions proved advantageous and considerably stabilized the RNA helix. Where ANA modifications were either slightly less stabilizing (for pyrimidines) or more stabilizing (as with purines) versus HNA modifications, methylation of the 3'-hydroxyl moiety of ANA congeners further improved the affinity for RNA systematically with approximately 0.5 °C. However, converting the hexitol into a methyl hexopyranoside via attachment of a second “methoxy substituent” at the 1'-position as in our DMANA constructs, wiped out the advantage which was gained before in using constrained hexitol moieties. The obtained affinities of these DMANA containing constructs more or less matched those of the corresponding fully complementary RNA sequences. Incorporation of a single DMANA building block slightly destabilized the RNA duplex for substitution of a pyrimidine, but on contrast slightly stabilized the duplex in case of guanine and to a larger extent upon substitution of adenine within this sequence context (Table 1 and Figure 2, base matches).
As on average the DMNA pairing affinity to RNA was not advantageous nor really detrimental, we further studied their mismatch behavior within the same RNA nonamer constructs (5'-GCGU-X*-UGCG/5'-CGCAYACGC; Figure 2) in comparison to different constructs. The graphical chart displays the destabilization in relation to the respective matched sequence as obtained for DMANA building blocks in comparison to discrimination properties for HNA and MANA ([23] and within dsRNA duplexes. Analogous discrimination properties were noted, and especially for dmanaC very selective pairing to guanosine was obtained with −22 °C to −24 °C of destabilization for the different mismatches. However, no overall advantage can be seen in terms of either pairing selectivity or universal base pairing capabilities. Hence, normal WC pairing can be assumed.

Table 1.
Tm values of complementary RNA duplexes [5'-GCGU-X*-UGCG/5'-CGCAYACGC].
| X* | Structure | Tm (°C) | X* | Structure | Tm (°C) |
|---|---|---|---|---|---|
| U* | RNA | 50.4 ± 0.0 | G* | RNA | 60.4 ± 0.0 |
| HNA | 53.4 ± 0.1 | HNA | 62.4 ± 0.1 | ||
| ANA | 53.0 ± 0.2 | ANA | 62.9 ± 0.1 | ||
| MANA | 53.8 ± 0.2 | MANA | 63.4 ± 0.1 | ||
| DMANA | 50.6 ± 0.2 | DMANA | 61.6 ± 0.1 | ||
| C* | RNA | 60.8 ± 0.1 | A* | RNA | 52.5 ± 0.1 |
| HNA | 62.0 ± 0.0 | HNA | 55.0 ± 0.2 | ||
| ANA | 60.9 ± 0.1 | ANA | 56.5 ± 0.1 | ||
| MANA | 61.4 ± 0.0 | MANA | 57.0 ± 0.1 | ||
| DMANA | 60.1 ± 0.2 | DMANA | 55.5 ± 0.1 |
Conditions: as determined in 100 mM NaCl buffer containing 20 mM KH2PO4 and 0.1 mM EDTA, pH 7.5, with a duplex concentration of 4 µM. Annotations U*, C*, A* and G* denote either a RNA, HNA, ANA, MANA or DMANA monomer respectively, versus the natural complementary base Y.
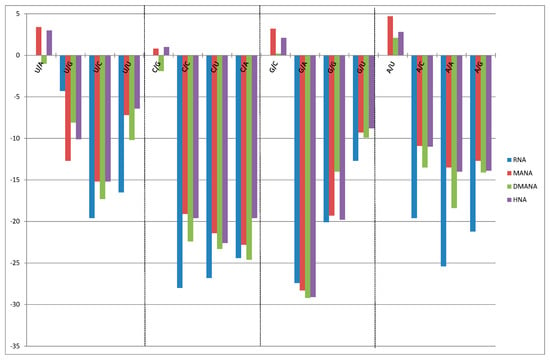
Figure 2.
Base pairing selectivity for different 6-membered ring analogues. Conditions: graphical overview of the (de)stabilization of the matched and mismatched nonamer sequences following a single incorporation of a sugar modified nucleoside (MANA in red, DMANA in green and HNA in purple) wherein the selectivity of pairing is shown by destabilization of the respective mismatches. The pairing selectivity for RNA is shown in blue.
This picture was confirmed with a second substitution within different nonamer dsRNA sequences (Table 2), with the largest destabilization noted for dmanaC building blocks (−1.8 °C/modification) while still an increase in stability of 1.1 °C/modification was seen with incorporation of 2 dmanaA blocks. The stabilizing effects of the HNA and 3'-methylated ANA constructs (MANA) are included for comparative reasons. We therefore decided to prepare a fully modified DMANA octamer and hybridized it to the complementary RNA sequence (Table 2, bottom). Where the natural RNA duplex displayed a Tm of 40.6 °C, the DMANA construct still paired albeit with slightly lower affinity (−0.3 °C/modification). The different hexitol constructs on the other hand strongly increased the affinity for the RNA complement as documented before [23,34]. It hence can be concluded that the DMANA analogues are different from the previous hexitol series of compounds with a constrained 6-membered ring conformation fit for pairing to RNA.

Table 2.
Thermal stability for RNA duplexes containing a double modification.
| Sequences | X* | Tm (°C) | ∆Tm/Modification (°C) |
|---|---|---|---|
| 5'-GCU*GUGU*CG-3' | RNA | 55.6 ± 0.4 | Reference |
| MANA | 62.6 ± 0.2 | 3.5 | |
| DMANA | 54.5 ± 0.1 | −0.5 | |
| HNA | 60.2 ± 0.1 | 2.3 | |
| 5'-GCC*AUAC*CG-3' | RNA | 57.1 ± 0.1 | Reference |
| MANA | 59.3 ± 0.1 | 1.1 | |
| DMANA | 53.4 ± 0.1 | −1.8 | |
| HNA | 58.2 ± 0.1 | 0.6 | |
| 5'-GCG*UUUG*CG-3' | RNA | 51.3 ± 0.2 | Reference |
| MANA | 54.3 ± 0.1 | 1.5 | |
| DMANA | 51.1 ± 0.1 | −0.1 | |
| HNA | 53.0 ± 0.2 | 0.8 | |
| 5'-GCA*CUCA*CG-3' | RNA | 57.1 ± 0.1 | Reference |
| MANA | 63.5 ± 0.1 | 3.2 | |
| DMANA | 59.3 ± 0.1 | 1.1 | |
| HNA | 62.1 ± 0.1 | 2.5 | |
| 5'-G*C*G*U*A*G*C*G*-3' | RNA | 40.6 ± 0.1 | Reference |
| MANA | 61.1 ± 0.3 | 2.6 | |
| DMANA | 38.1 ± 0.2 | −0.3 | |
| ANA | 59.6 [34] | 2.4 | |
| HNA | 52.0 [34] | 1.4 |
Conditions: Tm as determined in 100 mM NaCl buffer containing 20 mM KH2PO4 and 0.1 mM EDTA, pH 7.5, with a duplex concentration of 4 µM. U*, C*, A* and G* denote either a RNA, MANA, DMANA, ANA or a HNA monomer, respectively. At the bottom the results are given for the fully modified strands paired to the complementary RNA strand.
Finally, incorporation of a single DMANA building block into dsDNA 13-mer sequences [5'-CACCGX*TGCTACC-3'/3'-GTGGCYACGATGG-5'] was evaluated at 0.1 M salt concentration (Table 3) for both match and mismatch sequences. However, a single DMANA incorporation already afforded respectively 9 °C (for dmanaU and dmanaC), 5 °C (for dmanaA) or 6 °C (for dmanaG) of destabilization for the different matched pairs within this sequence context. A slightly better result could be expected if we could compare the dmanaT construct instead of dmanaU in view of the stabilizing effect of a 5-methyl substituent on the base, but this still would have resulted in a destabilization of 7 to 8 °C. The selectivity of pairing is adequate but gives a mixed picture, with selectivity being dependent probably on the base and the sequence context. In view of the fairly strong destabilization versus DNA sequences it is clear however that these DMANA blocks do not have a DNA like conformation.

Table 3.
Hybridization studies following incorporation into a DNA strand.
| Y | A | T | G | C | ||||
|---|---|---|---|---|---|---|---|---|
| X* (X) | Tm | ΔTm | Tm | ΔTm | Tm | ΔTm | Tm | ΔTm |
| U* | 47.7 | - | 40.9 | −6.8 | 41.7 | −6.0 | 38.3 | −9.4 |
| T | 57.1 | - | 46.7 | −10.4 | 50.3 | −6.8 | 44.2 | −12.9 |
| A* | 41.9 | −10.8 | 52.7 | - | 45.3 | −7.4 | 41.4 | −11.3 |
| A | 46.6 | −10.7 | 57.3 | - | 52.9 | −4.4 | 44.4 | −12.9 |
| C* | 41.8 | −10.3 | 40.8 | −11.3 | 52.1 | - | 38.5 | −13.6 |
| C | 44.4 | −16.5 | 45.6 | −15.3 | 60.9 | - | 40.6 | −20.3 |
| G* | 42.9 | −11.0 | 48.9 | −5.0 | 46.0 | −7.9 | 53.9 | - |
| G | 51.6 | −8.4 | 51.0 | −9.0 | 53.5 | −6.5 | 60.0 | - |
Conditions: Tm values are provided for a single incorporation of a DMANA (X*) modification into a mixed DNA sequence [5'-CACCGX*TGCTACC-3'/3'-GTGGCYACGATGG-5'] for the match and the different mismatch sequences (Y) at 0.1 M salt and 4 M of duplex concentration.
2.3. Discussion
The MedChem group of the Rega Institute has already been elaborating for many years on nucleoside analogues with a 6-membered ring system substituting for ribose for various applications. Especially the analogues with a 1,5-anhydrohexitol ring having the base at the C2' in syn orientation with the remaining hydroxymethyl substituent (HNA, ANA and MANA) turned out to be well pre-organized for pairing with RNA, and are thus strongly stabilizing for RNA duplexes. Several biological studies have been undertaken with both HNA and ANA [22,35,36] and interesting results were obtained more recently regarding their use as xeno-nucleic acids [37,38]. Highest affinities so far however were obtained with MANA building blocks [23]. We therefore started to study the influence of DMANA analogues carrying an additional methoxy substituent on the 6-membered ring system. However, as shown by the various Tm studies, incorporation of the new modification does not further increase the affinity for RNA, but overall rather tends to slightly destabilize dsRNA complexes, while strongly destabilizing DNA upon incorporation. Therefore the modification still resembles more closely RNA monomers with their 3'-endo conformation as in RNA duplexes.
These findings are further corroborated by inserting in silico a modified building block into a dsRNA. As can be seen in Figure 3, no steric hindrance occurs when substituting a DMANA residue having two OMe groups at the sugar 1' and 3' positions for a uridine into an RNA duplex for which the model of Mooers was used [39]. The HNA sugar conformation upon pairing with RNA is 1C4 having an axial base orientation. Likewise, for the DMANA structure incorporated into the dsRNA, both methoxy substituents likewise are oriented axially with no apparent steric clashes. However, it is well known that apart from forces like the anomeric effect, substituents in 6-membered rings prefer the equatorial orientation to avoid 1,3-diaxial interactions. Energy calculations for the monomers using Amber force field [40] indeed show a slight preference for having the base and both methoxy groups in an all-equatorial configuration (with 4'-OH and 6'-CH2OH in axial orientation), opposite to what is found for HNA and ANA building blocks with an axial oriented heterocyclic base. Increasing the number of OMe substituents therefore may destabilize the 1C4 chair conformation giving preference to 4C1, which is less compatible with the RNA duplex. Hence, the energy penalty to preserve the DMANA monomer in a 1C4 conformation to allow for pairing within a dsRNA strand, might upset the entropic gain as expected of a pre-organized monomer.
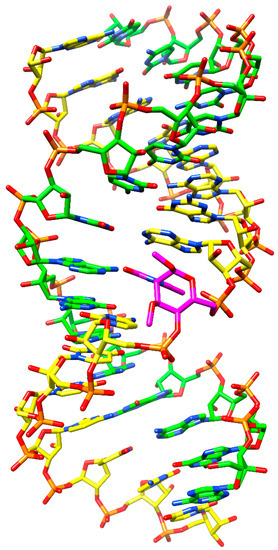
Figure 3.
RNA duplex following insertion of a DMANA modification (DMANA modification in purple; picture generated using Chimera (UCSF Chimera—a visualization system for exploratory research and analysis [41]).
Table 4 indeed shows the largest energy difference for both chair conformations for DMANA constructs. This energetic penalty for a forced change in conformation could be the basis of the reduced fitness of DMANA analogues for pairing with RNA.

Table 4.
Energy calculations for monomers with different scaffold.
| Base and OMe Orientation | Axial | Equatorial | Base and OMe Orientation | Axial | Equatorial |
|---|---|---|---|---|---|
| HNA | −205.31 | −205.79 | MANA | −195.22 | −200.35 |
| ANA | −200.58 | −198.19 | DMANA | −187.62 | −194.82 |
| Chair | 1C4 | 4C1 | Chair | 1C4 | 4C1 |
Conditions: Final energies in kcal/mol are shown following minimization using Amber force field. Nucleotides all have a thymine base, uncharged residues (phosphate groups protonated), with parametrization via antechamber, Gaff force field, 5000 steps of energy minimization, born solvation energy model.
3. Experimental Section
3.1. General
All chemicals including methylglucopyranoside were provided by Sigma-Aldrich (Diegem, Belgium) or Acros Organics (Geel, Belgium) and were of the highest quality. 1H and 13C-NMR spectra were determined with a 300, 500 and 600 MHz Varian Gemini apparatus (currently Agilent Technologies, Santa Clara, CA, USA) with tetramethylsilane as internal standard for the 1H NMR spectra (s = singlet, d = doublet, dd = double doublet, t = triplet, br. s = broad signal, m = multiplet) and the solvent signal; CD3OD-d4 (δ = 48.9 ppm), DMSO-d6 (δ = 39.6 ppm) or CDCl3 (δ = 76.9 ppm) for the 13C-NMR spectra. Exact mass measurements were performed with a quadrupole/orthogonal acceleration time-of-flight tandem mass spectrometer (qTOF2, Micromass, Manchester, UK) fitted with a standard electrospray ionization (ESI) interface. All solvents were carefully dried or bought as such.
3.2. 1',5';2',3'-Dianhydro-4',6'-O-benzylidene-1'-O-methyl-d-allopyranoside (2)
To a solution of the 4',6'-O-benzylidene-2',3'-ditosyl intermediate (10.0 g, 17 mmol) in CH2Cl2 (80 mL) was added NaOMe (5.3 M in MeOH, 12.8 mL, 67.8 mmol). After stirring for overnight at room temperature, the reaction mixture was concentrated and the residue was dissolved in CH2Cl2 (150 mL), and washed twice with brine (50 mL). The aqueous layer was again extracted with CH2Cl2 (2 × 80 mL). Combined organic layers were dried over anhydrous Na2SO4, filtered and evaporated to get pure epoxide 2 (4.45 g, 99%). 1H-NMR (300 MHz, CDCl3): δ 7.60–7.33 (m, 5H, Ar-H), 5.60 (d, J = 6.1 Hz, 1H, Ph-CH), 4.92 (dd, J = 6.1, 2.7 Hz, 1H, 1'-H), 4.32–4.22 (m, 1H, 6'-He), 4.18–4.05 (m, 1H, 5-H), 3.98 (dd, J = 9.0, 6.2 Hz, 1H, 4-H), 3.71 (m, 1H, 6-Ha), 3.58–3.45 (m, 5H, 2'-H, O-Me). 13C-NMR (75 MHz, CDCl3): δ 137.11 (Ar-Ci); 129.19; 128.28 (Ar-Cp+o); 126.27 (Ar-Cm); 102.71 (Ph-C); 95.27 (C-1'); 77.83 (C-4'); 68.86 (C-6'); 59.99 (C-5'); 55.82 (OMe); 53.07 (C-3'); 50.66 (C-2'); HRMS calcd. for C14H16O5Na+ [M+Na]+ 287.0890, found 287.0891.
3.3. 4',6'-O-Benzylidene-1'-O-methyl-2'-deoxy-2'-(uracil-l-yl)-d-altropyranoside (3)
To a solution of uracil (4.62 g, 38.6 mmol) in dry DMF (30 mL) was added NaH (60% dispersion in oil, 1.29 g, 41.2 mmol). The reaction mixture was heated at 120 °C under an argon atmosphere for 1 h and to this reaction mixture epoxide 2 (3.4 g, 12.9 mmol) in dry DMF (20 mL) was added and stirring was continued for overnight at same temperature. The reaction mixture was then cooled and evaporated to dryness. The residue was dissolved in ethyl acetate (150 mL) and the organic layer was washed with a saturated aqueous NaHCO3 solution (2 × 50 mL). The aqueous layer was again extracted with EtOAc (3 × 50 mL). The combined organic layers were washed with brine (2 × 50 mL), dried over Na2SO4, and concentrated under vacuo and purification by silica gel column chromatography (elution with 2% MeOH in DCM) afforded 3 (1.61 g, 33%) as a white foam while recovering a large part of the starting epoxide 2 (1.94 g, 40% yield). 1H-NMR (500 MHz, CDCl3): δ 7.75 (d, J = 8.1 Hz, 1H, 6-H), 7.49–7.41 (m, 2H, Ar-H), 7.39–7.30 (m, 3H, Ar-H), 5.77 (d, J = 8.1 Hz, 1H, 5-H), 5.62 (s, 1H, Ph-CH), 4.83 (s, 1H, 1'-H), 4.81 (s, 1H, 2'-H), 4.48 – 4.38 (m, 2H, 5'-H, 6'-He), 4.16 (brs, 1H, 3'-H), 3.81 (t, J = 9.9 Hz, 1H, 6'-Ha), 3.69 (dd, J = 9.9, 2.4 Hz, 1H, 4'-H), 3.41–3.51 (m, 4H, OMe, -OH). 13C-NMR (125 MHz, CDCl3): δ 163.08 (C-4); 150.60 (C-2); 141.14 (C-6); 136.81 (Ar-Ci); 129.26 (Ar-Cp); 128.28 (Ar-Cm); 126.18 (Ar-Co); 102.91 (C-5); 102.27 (Ph-C); 98.99 (C-1'); 75.61 (C-3'); 68.94 (C-4'); 67.02 (C-6'); 58.19 (C-5'); 57.91 (OMe); 55.97(C-2'). HRMS calcd. for C18H20N2O7Na+ [M+Na]+ 399.1163, found 399.1156.
3.4. 4',6'-O-Benzylidene-1',3'-di-O-methyl-2'-deoxy-2'-(uracil-l-yl)-d-altropyranoside (4)
To a solution of 3 (1.51 g, 4.0 mmol) in dry THF (15 mL) was added NaH (60% dispersion in oil, 404 mg, 12.04 mmol) at 0 °C and the reaction mixture was stirred at 0 °C for 1h under argon atmosphere. Methyl iodide (0.37 mL, 6.02 mmol) in dry THF (1 mL) was added and stirring was continued for another 1 h at the same temperature. The reaction mixture was quenched with 5 mL MeOH. The solution was concentrated and dissolved in ethyl acetate (150 mL) and washed with saturated aqueous NaHCO3 (150 mL). The aqueous layer was again extracted with ethyl acetate (3 × 50 mL). The combined the organic layers were dried over Na2SO4, filtered, concentrated under vacuo, and purification by silica gel column chromatography (elution with 2% MeOH in DCM) afforded the dimethylated nucleoside 4 (1.01 g, 65%). 1H-NMR (500 MHz, CDCl3): δ 9.41 (s, 1H, N3-H), 7.80 (d, J = 8.2 Hz, 1H, 6-H), 7.47–7.35 (m, 5H, Ar-H), 5.80 (dd, J = 8.1, 1.7 Hz, 1H, 5-H), 5.55 (s, 1H, Ph-CH), 4.90 (d, J = 1.7 Hz, 1H, 1'-H), 4.80 (s, 1H, 2'-H), 4.48–4.36 (m, 2H, 6'-He, 3'-H), 3.77 (t, J = 10.3 Hz, 1H, 6'-Ha),3.72–3.67 (m, 2H, 4'-H, 5'-H), 3.62 (s, 3H, OMe), 3.46 (s, 3H, OMe). 13C-NMR (125 MHz, CDCl3): δ 162.98 (C-4); 150.39 (C-2); 141.18 (C-6); 137.06 (Ar-Ci); 129.20 (Ar-Cp); 128.28 (Ar-Cm); 126.21 (Ar-Co); 102.86 (C-5); 102.48 (Ph-C); 98.84 (C-1'); 76.06 (C-3'); 75.95 (C-4'); 69.11 (C-5'); 59.59 (C-6'); 58.56 (OMe); 55.94 (OMe); 55.81(C-2'). HRMS calcd. for C19H23N2O7 [M+H]+ 391.1500, found 391.1494.
3.5. 4',6'-O-Benzylidene-1',3'-di-O-methyl-2-deoxy-2'-(cytosin-1-yl)-d-altropyranoside (5)
A solution of triazole (1.05 g, 15.2 mmol) and phosphorus oxychloride (0.3 mL, 3.18 mmol) was prepared in pyridine (8 mL) at 0 °C. Triethylamine (2.03 mL, 14.5 mmol) was added dropwise at 0 °C and the solution was stirred 30 min. The uracil derivative 4 (0.650 g, 1.66 mmol) dissolved in dry pyridine (8 mL) was added at 0 °C and the solution was stirred for 2 h at room temperature and concentrated and co-evaporated with toluene (2 × 20 mL). The crude product was dissolved with DCM (150 mL) and washed twice with brine (60 mL). The aqueous layer was extracted with DCM (30 mL). Combined organic layers were dried over anhydrous Na2SO4, filtered and evaporated. The residue was dissolved in 1,4-dioxane (30 mL), cooled to 0 °C and aqueous ammonia 25% (13 mL) were added. The solution was left overnight at RT. The solution was evaporated and co-evaporated with toluene (3 × 20 mL). The residue was purified by silica gel column chromatography (elution with 10% MeOH/dichloromethane) and afforded the dimethylated cytidine analog 5 (377 mg, 58%) as a white foam. 1H-NMR (500 MHz, CD3OD): δ 7.91 (d, J = 7.5 Hz, 1H, 6-H), 7.50–7.42 (m, 2H, Ar-H), 7.38–7.26 (m, 3H, Ar-H), 5.95 (d, J = 7.5 Hz, 1H, 5-H), 5.63 (s, 1H, Ph-CH), 4.93 (s, 1H, 2'-H), 4.91 (d, J = 2.1 Hz, 1H, 1'-H), 4.34–4.30 (m, 1H, 6'-He), 4.28 (dd, J = 9.9, 5.3 Hz, 1H, 5'-H), 3.84 (dd, J = 11.0, 2.3 Hz, 1H, 6'-Ha), 3.81 (dd, J = 6.6, 2.3 Hz, 1H, 4'-H), 3.67 (t, J = 2.3 Hz, 1H, 3'-H), 3.57 (s, 3H, OMe), 3.42 (s, 3H, OMe). 13C-NMR (125 MHz, CD3OD): δ 167.49 (C-4); 158.15 (C-2); 144.10 (C-6), 139.09 (Ar-Ci), 129.97 (Ar-Cp), 129.07 (Ar-Cm), 127.44 (Ar-Co), 103.41 (Ph-C), 100.28 (C-5), 96.46 (C-1'), 77.40 (C-3'), 76.86 (C-4'), 70.05 (C-6'), 59.93 (C-5'), 59.13 (OMe), 57.45 (OMe), 55.84 (C-2'). HRMS calcd. for C19H24N3O6+ [M+H]+ 390.1654, found 390.1653.
3.6. 4',6'-O-Benzylidene-1',3'-O-methyl-2'-deoxy-2'-(N6-benzoylcytosin-1-yl)-d-altropyranoside (6)
The analog 5 (0.35 g, 0.9 mmol) was co-evaporated with dry pyridine (6 mL), dissolved in dry pyridine (4 mL), and cooled at 0 °C. Benzoyl chloride (0.314 mL, 2.7 mmol) was added and the solution was allowed to come to RT. The solution was stirred for 3 h at RT. The mixture was cooled to 0 °C and water (0.25 mL) was added. Then, aqueous ammonia 25% (2 mL) was added and the solution was stirred for 30 min at RT. The volatiles were removed under reduced pressure and co-evaporated, each time with toluene (3 × 5 mL). The residue was adsorbed on silica by co-evaporation from DCM and purified by silica column chromatography (elution with 2% MeOH/DCM) affording the protected nucleoside 6 (370 mg, 83% yield) as a white foam. 1H-NMR (300 MHz, CDCl3): δ 8.24 (d, J = 7.5 Hz, 1H, 6-H), 7.94 (d, J = 7.3 Hz, 2H, Bz-H), 7.70–7.33 (m, 9H, 5-H, Ar-H, Bz-H), 5.56 (s, 1H, Ph-CH), 5.12 (d, J = 1.6 Hz, 1H, 1'-H), 4.90 (s, 1H, 2'-H), 4.55–4.37 (m, 2H, 5'-H, 6'-He), 3.86–3.71 (m, 2H, 6'-Ha, 3'-H), 3.74 (dd, J = 9.5, 3.1 Hz, 1H, 4'-H), 3.68 (s, 3H, OMe), 3.49 (s, 3H, OMe). 13C-NMR (75 MHz, CDCl3): δ 167.76 (PhCONH); 162.14 (C-4); 137.11 (C-2); 133.34 (C-6); 129.18, 129.08, 128.28, 127.60, 126.23 (Ar-C, Bz-C); 102.48 (Ph-C); 99.00 (C-5); 97.14 (C-1'), 78.35 (C-3'), 75.77 (C-4'); 75.33 (C-5'); 69.20 (C-6'); 59.51 (OMe); 58.72 (OMe); 56.03 (C2'). HRMS calcd. for C26H28N3O7+ [M+H]+ 494.1922, found 494.1919.
3.7. 4',6'-O-Benzylidene-1'-O-methyl-2'-deoxy-2'-(adenin-9-yl)-d-altropyranoside (7)
To a solution of adenine (3.22 g, 23.85 mmol) in dry DMF (40 mL) was added NaH (60% dispersion in oil, 890 mg, 22.26 mmol). The reaction mixture was heated at 90 °C under argon atmosphere for 1 h, after which the epoxide 2 (2.1 g, 7.95 mmol) in dry DMF (15 mL) was added and stirring was continued overnight at 120 °C. The reaction mixture was quenched with MeOH (15mL) and following evaporation the residue was partitioned between EtOAc and saturated aqueous NaHCO3 solution. The organic layer was separated and the aqueous layer was extracted with EtOAc (3 × 50 mL). The combined the organic layers were dried over Na2SO4, filtered, concentrated in vacuo, and purification by normal silica gel column chromatography (elution with 3% MeOH in DCM) afforded compound 7 (2.37 g, 75%). 1H-NMR (300 MHz, CDCl3): δ 8.30 (s, 1H, 8-H), 8.18 (s, 1H, 2-H), 7.50–7.29 (m, 5H, Ar-H), 6.00 (s, 2H, -NH2), 5.55 (s, 1H, Ph-CH), 5.15 (s, 1H, 1'-H), 5.07 (s, 1H, 2'-H), 4.64− 4.52 (m, 1H, 5'-H), 4.46 (dd, J = 9.9, 4.7 Hz, 1H, 6'-He), 4.33 (brs, 1H, 3'-H), 3.86 (t, J = 9.9 Hz, 1H, 6'-Ha), 3.73 (d, J = 8.3 Hz, 1H, 4'-H), 3.54 (s, 3H, OMe). 13C-NMR (75 MHz, CDCl3): δ 155.60 (C-6); 153.44 (C-2); 149.77 (C-4); 138.45 (C-8); 136.86 (C-5); 129.28, 128.26, 126.15, 118.89 (Ar-C); 102.35 (Ph-C); 99.66 (C-1'); 75.87 (C-5'); 69.14 (C-3'); 67.03 (C-4'); 58.52 (C-6'); 57.18 (OMe); 56.13 (C-2'). HRMS calcd. for C19H22N5O5+ [M+H]+ 400.1615, found 400.1613.
3.8. 4',6'-O-Benzylidene-1',3'-di-O-methyl-2'-deoxy-2'-(adenin-9-yl)-d-altropyranoside (8)
The adenine analog 7 (2.36 g, 5.9 mmol) was co-evaporated with dry DMF (40 mL).The foam was dissolved in dry DMF (70 mL), cooled to −78 °C and NaH (330 mg, 8.28 mmol) was added and the mixture was stirred at −78 °C for 30 min under argon. Subsequently, methyl iodide (0.590 mL, 9.46 mmol) was dissolved in dry DCM (20 mL) and the solution was added drop wise over 30 min to the reaction mixture at the same temperature. Stirring was continued for another 4.5 h at −78 °C and at −30 °C for an additional 1 h before quenching of the reaction with MeOH (10 mL). The solution was warmed to RT and concentrated to dryness under vacuum. The residue was dissolved in ethyl acetate (150 mL) and washed with aqueous saturated NaHCO3 (130 mL). The organic layer was then washed with brine (130 mL), and the combined aqueous layers were again extracted with ethyl acetate (2 × 150 mL). The combined organic layers were dried over Na2SO4, filtered, and concentrated. Purification by silica column chromatography (0%–3% MeOH/DCM) afforded the methylated nucleoside 8 (1.84 g, 75.4%). 1H-NMR (500 MHz, CDCl3): δ 8.39 (s, 1H, 8-H), 8.20 (s, 1H, 2-H), 7.47–7.40 (m, 2H, Ar-H), 7.37–7.31 (m, 3H, Ar-H), 5.92 (s, 2H, -NH2), 5.49 (s, 1H, Ph-CH), 5.15 (d, J = 2.5 Hz, 1H, 1'-H), 5.07 (s, 1H, 2'-H), 4.54 (td, J = 10.1, 5.3 Hz, 1H, 5'-H), 4.44 (dd, J = 10.5, 5.3 Hz, 1H, 6-He), 3.88 (t, J = 2.6 Hz, 1H, 3'-H), 3.83 (t, J = 10.5 Hz, 1H, 6'-Ha), 3.79 (dd, J = 9.8, 2.5 Hz, 1H, 4'-H), 3.71 (s, 3H, OMe), 3.52 (s, 3H, OMe). 13C-NMR (125 MHz, CDCl3): δ 155.57 (C-6); 153.54 (C-2); 149.99 (C-4); 138.56 (C-8); 137.11, 129.15, 128.26, 126.16 (Ar-C); 119.10 (C-5); 102.48 (Ph-C); 99.47 (C-1'); 76.25 (C-5'); 76.19 (C-3'); 69.26 (C-4'); 60.06 (C-6'); 58.81 (OMe); 56.06 (OMe); 55.05 (C-2'). HRMS calcd. for C20H24N5O5+ [M+H]+ 414.1772, found 414.1767.
3.9. 4',6'-O-Benzylidene-1',3'-di-O-methyl-2'-deoxy-2'-(N6-benzoyladenin-9-yl)-d-altropyranoside (9)
The obtained analog 8 (1.59 g, 3.85 mmol) was co-evaporated with dry pyridine (2 mL), dissolved in dry pyridine (16 mL), and cooled at 0 °C. Benzoyl chloride (1.34 mL, 11.54 mmol) was added and the solution was allowed to come to RT. The solution was stirred 3 h at RT. The mixture was cooled to 0 °C and water (4 mL) was added. Then, aqueous ammonia 25% (8 mL) was added and the solution was stirred 30 min at RT. The volatiles were removed under reduced pressure and co-evaporated, each time with toluene (3 × 25 mL). The residue was adsorbed on silica by co-evaporation and purified by silica column chromatography (elution with 2% MeOH/DCM) affording the protected nucleoside 9 (1.70 g, 83% yield) as a white foam. 1H-NMR (500 MHz, CDCl3): δ 9.43 (s, 1H, NH), 8.78 (s, 1H, 8-H), 8.43 (s, 1H, 2-H), 8.05 (d, J = 7.5 Hz, 2H, H-Bz), 7.66–7.29 (m, 8H, Bz-H, Ar-H), 5.49 (s, 1H, Ph-CH), 5.22 (d, J = 2.1 Hz, 1H, 1'-H), 5.09 (s, 1H, 2'-H), 4.55 (td, J = 10.0, 5.3 Hz, 1H, 5'-H), 4.44 (dd, J = 10.4, 5.2 Hz, 1H, 6'-He), 3.88 (brs, 1H, 3'-H), 3.85–3.76 (m, 2H, ,6'-Ha, 4'-H), 3.71 (s, 3H, OMe), 3.52 (s, 3H, OMe). 13C-NMR (125 MHz, CDCl3): δ 164.86 (C=O); 152.77 (C-6); 152.06 (C-2); 149.78 (C-4); 141.06 (C-8); 137.01, 133.35, 132.75, 129.09, 128.72, 128.19, 128.00, 126.11 (Ar-C, Bz-C); 122.75 (C-5); 102.43 (Ph-C); 99.19 (C-1'); 76.11 (C-5'); 76.08 (C-3'); 69.16 (C-4'); 59.98 (C-6'); 58.83 (OMe); 56.01 (OMe); 55.13 (C-2'). HRMS calcd. for C27H28N5O6+ [M+H]+ 518.2034, found 518.2031.
3.10. 4',6'-O-Benzylidene-1'-O-methyl-2'-deoxy-2'-(2-amino-6-(2-trimethylsilyl-ethoxy)purin-9-yl)-d-altropyranoside (11)
A mixture of 4.5 g (15.4 mmol) of 10 and 0.266 g (33.5 mmol) of lithium hydride in 60 mL of dry DMF was stirred under nitrogen at 90 °C for 2 h. After addition of 2.6 g (9.8 mmol) of the epoxide 2 dissolved in 20 mL of dry DMF, stirring was continued for 18 h at 130 °C. The reaction mixture was cooled and evaporated to dryness. The residue was dissolved in ethyl acetate (100 mL) and washed with brine (100 mL). A small part of the base precipitated during extraction and was filtered off and kept for recycling. The aqueous layer was again extracted with EtOAc (3 × 50 mL). The combined organic layers were dried over Na2SO4, filtered, concentrated under vacuo, and purification by silica gel column chromatography (elution with 3% MeOH in DCM) afforded 11 (2.11 g, 42%) while recovering a large point of the starting 10 (1.5 g, 26%). 1H-NMR (300 MHz, CDCl3): δ 8.03 (s, 1H, 8-H), 7.39 (m, 5H, Ar-H), 5.55 (s, 1H, Ph-CH), 5.27 (d, J = 1.8 Hz, 1H, 1'-H), 5.04 (s, 1H, 2'-H), 4.83–4.60 (m, 3H, -NH2, 5'-H), 4.59–4.38 (m, 3H, 6'-He, 6'-Ha, 3'-H), 4.36–4.29 (m, 1H, 4'-H), 3.89–3.73 (m, 2H, OCH2-), 3.55 (s, 3H, OMe), 1.21–1.10 (m, 9H, -SiC3H9). 13C-NMR (75 MHz, CDCl3): δ 161.74 (C-6); 159.43 (C-2); 152.79 (C-4); 137.10 (C-8); 136.53 (Ar-Ci), 129.53 (Ar-Cp), 128.34 (Ar-Cm), 126.46 (Ar-Co); 115.06 (C-5); 102.58 (Ph-C); 100.05 (C-1'); 76.14 (C-4'); 69.25 (C-6'); 66.10 (C-5'); 65.09 (C-3'); 58.63 (OCH2-); 57.19 (OMe); 56.40 (C-2'); 17.49 (CH2Si); −1.46 (-SiC3H9).
3.11. 4',6'-O-Benzylidene-1',3'-di-O-methyl-2'-deoxy-2'-(2-amino-6-(2-trimethylsilylethoxy)-purin-9-yl)-d-altropyranoside (12)
The obtained foam 11 (2.11 g, 4.1 mmol) was dried overnight under vacuum. The foam was dissolved in dry DMF (42 mL), cooled to −30 °C and NaH (1.3 g, 32 mmol) was added and the mixture was stirred at −30 °C for 1 h under argon. Subsequently, methyl iodide (1 mL, 16 mmol) was dissolved in dry DCM (10.5 mL) and the solution was added drop wise over 30 min to the reaction mixture at −30 °C which was stirred further at −30 °C for 1 h. The reaction was finally running 3 h more at −30 °C to −20 °C before quenching with 10 mL of MeOH for 20 min. The solution was concentrated and dissolved in DCM (50 mL) and washed with brine (50 mL). The aqueous layer was again extracted with DCM (3 × 50 mL). The combined organic layers were dried over Na2SO4, filtered and concentrated under vacuo, and purification by silica gel column chromatography (elution with 2% MeOH in DCM) afforded methylated nucleoside 12 (2.03 g, 94%) as a white foam. 1H-NMR (500 MHz, CDCl3): δ 7.98 (s, 1H, 8-H), 7.49–7.40 (m, 1H, 2H, Ar-H), 7.38–7.30 (m, 3H, Ar-H), 5.47 (s, 1H, Ph-CH), 5.01 (s, 1H, 1'-H), 4.98–4.92 (m, 1H, 5'-H), 4.66–4.55 (m, 1H, 3'-H), 4.53–4.46 (m, 1H, 4'-H), 4.41 (dd, J = 10.4, 5.3 Hz, 1H, 6'-Ha), 1.26 (t, J = 12.1 Hz, 1H). 13C-NMR (126 MHz, CDCl3): δ 161.59 (C-6); 159.61 (C-2); 153.62 (C-4); 137.21 (C-8); 136.93 (Ar-Ci); 129.07 (Ar-Cp); 128.21 (Ar-Cm); 126.16 (Ar-Co); 115.15 (C-5); 102.42 (Ph-C); 99.45 (C-1'); 76.26 (C-4'); 76.06 (C-6'); 69.26 (C-5'); 65.04 (C-3'); 59.98 (OCH2-); 58.73 (OMe); 55.97 (OMe); 54.82 (C-2'); 17.54 (CH2Si); −1.41 (3C, Si(CH3)3). HRMS calcd. For C25H36N5O6Si+ [M+H]+ 530.2429, found 530.2430.
3.12. 4',6'-O-Benzylidene-1',3'-di-O-methyl-2'-deoxy-2'-(guanin-9-yl)-d-altropyranoside (13)
A 1 M solution of tetrabutylammonium fluoride in dry THF (15.10 mL) was added to the guanine derivative 12 (2.0 g , 3.77 mmol) and the mixture was stirred at room temperature under N2 for 2 h after which water (15 mL) was added. The pH was adjusted to 5 with acetic acid and the mixture was evaporated. The residue was purified by silica gel column chromatography (elution with 10% MeOH in DCM) affording 13 (1.25 g, 75%). 1H-NMR (500 MHz, CDCl3): δ 11.99 (s, 1H, NH), 7.86 (s, 1H, 8-H), 7.42 (m, 2H, Ar-H), 7.31 (m, 3H, Ar-H), 6.73 (s, H, 2H, 2-NH2), 5.49 (s, 1H, Ph-CH), 5.07 (s, 1H, 1'-H), 4.81 (s, 1H, 2'-H), 4.50–4.42 (m, 1H, 5'-H), 4.37 (dd, J = 10.1, 5.0 Hz, 1H, 6'-He), 3.78 (s, 3H, 3'-H, 6-Ha, 4'-H), 3.58 (s, 3H, OMe), 3.53 (s, 3H, OMe). 13C-NMR (125 MHz, CDCl3): δ 159.03 (C-6); 154.05 (C-2); 151.58 (C-4); 137.20 (Ar-Ci); 135.42 (C-8); 129.08, 128.22, 126.19 (Ar-Cm+o+p); 116.24 (C-5); 102.40 (Ph-C); 99.23 (C-1'); 76.11 (C-4'); 76.04 (C-6'); 69.19 (C-5'); 59.76 (C-4'); 58.68 (OMe); 55.98 (OMe); 54.79 (C-2'). HRMS calcd. For C20H24N5O6+ [M+H]+ 430.1721, found 430.1711.
3.13. 4',6'-O-Benzylidene-1',3'-di-O-methyl-2'-deoxy-2'-(N2-(dimethylamino)methylene-guanin-9-yl))-d-altropyranoside (14)
An amount of 13 (0.66 g, 1.54 mmol) was co evaporated three times with pyridine, dissolved in 30 mL of dry MeOH (20 mL) and N,N-dimethylformamide diethyl acetal (0.824 mL, 6.16 mmol) was added. The mixture was stirred at reflux for 2 h under argon, after which it was evaporated and co-evaporated with toluene (3 × 30 mL). The residue was purified by silica gel column chromatography (2%–5% MeOH/dichloromethane) affording the analog 14 (0.634 g, 85%). 1H-NMR (500 MHz, CDCl3): δ 9.78 (s, 1H, N1-H), 8.65 (s, 1H, N=CH-N), 7.97 (s, 1H, 8-H), 7.45 (m, 2H, Ar-H), 7.40–7.30 (m, 3H, Ar-H), 5.51 (s, 1H, Ph-CH), 5.02 (s, 1H, 2'-H), 4.99 (d, J = 2.2 Hz, 1H, 1'-H), 4.50 (td, J = 10.4, 5.3 Hz, 1H, 5'-H), 4.41 (dd, J = 10.4, 5.3 Hz, 1H, 6-He), 3.90–3.80 (m, 3H, 3'-H, 6-Ha, 4'-H), 3.68 (s, 3H, OMe), 3.50 (s, 3H, OMe), 3.18 (s, 3H, NMe), 3.15 (s, 3H, NMe). 13C-NMR (125 MHz, CDCl3): δ 158.12 (N=CH-N); 157.97 (C-6); 157.04 (C-2); 150.18 (C-4); 137.20 (C-8); 136.08, 129.05, 128.19, 126.14 (Ar-C); 119.85 (C-4); 102.39 (Ph-CH); 99.31 (C-1'); 76.47 (C-4'); 76.12 (C-6'); 69.17 (C-3'); 60.14 (C-5'); 58.67 (OMe); 55.89 (OMe); 55.13 (C-2'); 41.44 (NMe); 35.23 (NMe). HRMS calcd. for C23H29N6O6+ [M+H]+ 485.2143, found 485.2145.
3.14. 1',3'-Di-O-methyl-2'-deoxy-2'-(uracil-1-yl)-d-altropyranoside (15a)
Compound 4 (0.9 g, 2.31 mmol) was dissolved in 60 mL of AcOH-H2O (3:1) at rt. The reaction mixture was slowly heated at 45 °C and reaction progress was monitored using TLC. After 12 h, the mixture was concentrated and co evaporated with toluene (30 mL). The crude residue was purification by flash silica gel column chromatography (elution with 5% MeOH in DCM) afforded compound 15a (0.66 g, 94%). 1H-NMR (600 MHz, DMSO-d6): δ 11.15 (s, 1H, NH), 7.71 (s, 1H, 6-H), 5.56 (d, J = 5.5 Hz, 1H, 5-H), 4.93 (d, J = 3.9 Hz, 1H, 1'-H), 4.88 (t, J = 5.0 Hz, 1H, 2'-H), 4.84–4.60 (m, 1H, 3'-H), 4.03 (dd, J = 9.8, 5.5 Hz, 1H, 5'-H), 3.77 (q, J = 5.5 Hz, 1H, 4'-H), 3.65–3.53 (m, 2H, 6'-H), 3.26, (s, 3H, OMe), 3.22 (s, 3H, OMe). 13C-NMR (150 MHz, DMSO-d6): δ 170.83 (C-4); 163.61 (C-2); 151.66 (C-6); 101.62 (C-5); 99.07 (C-1'); 77.52 (C-5'); 76.08 (C-3); 63.67 (C-4'); 61.12 (C-6'); 60.23 (OMe); 56.65 (OMe); 55.39 (C-2'). HRMS calcd. For C12H19N2O7+ [M+H]+ 325.1006, found 325.1005.
3.15. 1',3'-Di-O-methyl-2'-deoxy-2'-(N6-benzoylcytosin-1-yl)-d-altropyranoside (15b)
Compound 6 (0.36 g, 0.73 mmol) was dissolved in 24.1 mL of AcOH:H2O (3:1) at rt. The reaction mixture was slowly heated at 45 °C and further treated as per the synthesis of 15a affording the nucleoside analog 15b (0.28 g, 50%). 1H-NMR (300 MHz, CD3OD): 8.23–7.94 (m, 3H, 6-H, Bz-H), 7.68–7.51 (m, 4H, 5-H, Bz-H), 5.26 (brs, 1H, 1'-H), 4.87–4.81 (m, 1H, 2'-H), 4.23–4.09 (m, 1H, 5'-H), 4.03 (q, 1H, J = 5.5 Hz, 4'-H), 3.80–3.66 (m, 2H, 3'-H, 6'-He), 3.30 (s, 6H, 2OMe), 3.26–3.20 (m, 1H, 6'-Ha). 13C-NMR (150 MHz, CD3OD): 169.07 (PhCONH), 164.69 (C-4), 158.61 (C-2), 134.65 (C-6), 134.13, 130.47, 129.84, 129.59, 129.19 (Bz-C), 100.35 (C-5), 98.59 (C-1'), 64.94 (C-3'), 64.74 (C-5'), 63.46 (C-4'), 62.47 (C-6'), 57.75 (OMe), 56.05 (OMe), 49.85 (C-2'). HRMS calcd. for C19H24N3O7+ [M+H]+ 406.1609, found 406.1608.
3.16. 1',3'-Di-O-methyl-2'-deoxy-2'-(N6-benzoyladenin-9-yl)-d-altropyranoside (15c)
Following the procedure for 15a, compound 9 (1.65 g, 3.19 mmol) was treated 82.85 mL of AcOH:H2O (3:1) at rt. The reaction mixture was slowly heated at 45 °C and reaction progress was monitored using TLC. After 12 h, the mixture was concentrated and co evaporated with toluene, and after purification afforded (0.78 g, 57%) of 15c.
1H-NMR (300 MHz, CD3OD): δ 8.76 (s, 1H, 8-H), 8.52 (s, 1H, 2-H), 8.13 (d, J = 7.4 Hz, 2H, Bz-H), 7.77–7.53 (m, 3H, Bz-H), 5.39 (d, J = 5.5 Hz, 1H, 1'-H), 4.88–4.82 (m, 1H, 2'-H), 4.31 (dd, J = 8.8, 3.7 Hz, 1H, 5'-H), 4.26–4.21 (m, 1H, 3'-H), 4.13 (q, J = 5.5 Hz, 1H, 4'-H), 4.00–3.9 (m, 2H, 6'-H), 3.42 (s, 3H, OMe), 3.36 (s, 3H, OMe). 13C-NMR (75 MHz, DMSO-d6): δ 165.20 (C=O); 152.25 (C-6); 150.77 (C-2); 149.68 (C-4); 143.85 (C-8); 132.92 (C-5); 131.89, 127.95, 127.91, 125.07 (Bz-C); 97.91 (C-1'); 77.10 (C-5'); 75.44 (C-3'); 62.37 (C-4'); 59.78 (C-6'); 55.75 (OMe); 55.53 (OMe); 54.53 (C-2'). HRMS calcd. for C20H24N5O6+ [M+H]+ 430.1721, found 430.1716.
3.17. 1',3'-Di-O-methyl-2'-deoxy-2'-(N2-(dimethylamino)methylene-guanin-9-yl))-d-altro-pyranoside (15d)
Compound 14 (0.58 g, 1.2 mmol) was dissolved in 30 mL of AcOH:H2O (3:1) at rt. The reaction mixture was slowly heated at 45 °C and reaction progress was monitored using TLC. After 12 h, the mixture was concentrated and co-evaporated with toluene. The crude residue was purified by flash silica gel column chromatography (elution with 10% MeOH in DCM) afforded compound 15d (0.4 g, 81%). 1H-NMR (500 MHz, CD3OD): δ 8.65 (s, 1H, N=CH-N), 7.85 (s, 1H, 8-H), 5.21 (d, J = 5.3 Hz, 1H, 2'-H), 4.64–4.57 (m, 1H, 1'-H), 4.10 (m, 2H, 4'-H, 3'-H), 4.00 (dd, J = 8.8, 4.1 Hz, 1H, 5'-H), 3.84 (d, J = 4.6 Hz, 2H, 6'-H), 3.33 (s, 3H, OMe), 3.30 (s, 3H, OMe), 3.16 (s, 3H, OMe), 3.07 (s, 3H, OMe). 13C-NMR (125 MHz, CD3OD): δ 160.31 (N=C-N); 159.83 (C-6); 158.79 (C-2); 152.31 (C-4); 140.37 (C-8); 120.48 (C-5); 100.37 (C-1'); 78.41 (C-5'); 76.47 (C-3'); 65.24 (C-4'); 62.66 (C-6'); 57.94 (OMe); 57.74 (OMe); 56.17 (C-2'); 41.53 and 35.34 (NMe2). HRMS calcd. for C16H25N6O6+ [M+H]+ 397.1830, found 397.1827.
3.18. 6'-O-Dimethoxytrityl-1',3'-di-O-methyl-2'-deoxy-2'-(uracil-1-yl)-d-altropyranoside (16a)
To a solution of the nucleoside 15a (0.4 g, 1.32 mmol) in anhydrous pyridine (10 mL) and under argon atmosphere, dimethoxytrityl chloride (0.49 g, 1.45 mmol) was added under stirring on an ice bath. After stirring at room temperature for 3 h, 5% aqueous NaHCO3 solution (1 mL) was added, the reaction solvent was evaporated, diluted with CH2Cl2 (50 mL) and washed with 5% aqueous NaHCO3 (2 × 30 mL). The aqueous layers were back extracted once with 30 mL CH2Cl2. The combined organic layer was dried (Na2SO4), evaporated under reduced pressure and the crude residue was purified by flash chromatography (CH2Cl2/MeOH 96:4) affording the corresponding dimethoxytritylated nucleoside 16a (0.745 g, 93% yield) as a white foam. 1H-NMR (125 MHz, CDCl3): δ 9.31 (brs, 1H, NH), 7.48–7.43 (m, 2H, 6-H, Ar-H), 7.37–7.33 (m, 4H, Ar-H), 7.31–7.26 (m, 2H, Ar-H), 7.24–7.19 (m, 1H, Ar-H), 6.85–6.82 (m, 4H, Ar-H), 5.69 (dd, J = 8.0, 1.6 Hz, 1H, 5-H), 5.30 (brs, 1H, 1'-H), 4.94 (brs, 1H, 2'-H), 4.08–3.95 (m, 2H, 5'-H, 3'-H), 3.79 (s, 3H, OMe), 3.78–3.77 (m, 4H, OMe, 4'-H), 3.50–3.42 (brs, s, 6H, 2OMe), 3.42–3.40 (m, 2H, 6'-H). 13C-NMR (125 MHz, CDCl3): δ 163.28 (C-4); 158.50 (C-2); 150.68 (C-6); 144.85, 135.84, 130.09, 130.06, 128.10, 127.80, 126.81, 113.11 (Ar-C); 102.35 (C-5); 98.36 (C-1'); 86.20 (CTr-O); 77.14 (C-5'), 76.87 (C-3'); 64.05 (C-4'); 62.88 (C-6'); 57.93 (OMe); 55.69 (OMe); 55.19 (2OMe, C-2'). HRMS calcd. for C33H36N2O9Na+ [M+Na]+ 627.23186, found 627.2311.
3.19. 6'-O-Dimethoxytrityl-1',3'-di-O-methyl-2'-deoxy-2'-(N6-benzoylcytosin-1-yl)-d-altropyranoside (16b)
Compound 16b (0.346 g, 87% yield) was synthesized from compound 15b (0.228 g, 0.56 mmol) using dimethoxytrityl chloride (0.09 g, 1.29 mmol) in anhydrous pyridine (10 mL) according to the procedure used for the synthesis of compound 16a. 1H-NMR (500 MHz, CDCl3) : δ 8.75 (brs, 1H, NH), 7.90 (d, J = 7.3 Hz, 2H, 6-H, Ar-H), 7.64–7.45 (m, 6H, Ar-H), 7.40–7.19 (m, 8H, 5-H, Ar-H), 6.86 (d, J = 8.8 Hz, 4H, Ar-H), 5.29 (s, 1H, 1'-H), 5.03 (brs, 1H, 2'-H), 4.11–3.95 (m, 2H, 5'-H, 3'-H), 3.83–3.77 (m, 7H, 2OMe, 4'-H), 3.60–3.37 (m, 8H, 6'-H, 2OMe), 2.50 (s, 1H, 4-OH). 13C-NMR (126 MHz, CDCl3): δ 166.13 (C=0), 162.13 (C-4), 158.47 (C-4), 144.71 (C-6), 136.02, 133.17, 130.03, 129.01, 128.20, 127.79, 127.50, 126.80, 113.10 (Ar-C), 98.06 (C-5), 96.85 (C-1'), 86.11 (CTr-O), 77.13 (C-5'), 76.88 (C-3'), 65.62 (C-4'), 62.73 (C-6'), 58.04 (OMe), 55.72 (OMe), 55.18 (C-2, 2OMe). HRMS calcd. For C40H42N3 O9+ [M+H]+ 708.29208, found 708.2909.
3.20. 6'-O-Dimethoxytrityl-1',3'-di-O-methyl-2'-deoxy-2'-(N6-benzoyladenin-9-yl)-d-altropyranoside (16c)
Compound 16c (0.77 g, 91% yield) was synthesized from compound 15c(0.5 g, 1.16 mmol) using dimethoxytrityl chloride (0.44 g, 1.27 mmol) in anhydrous pyridine (10 mL) according to the procedure used for the synthesis of compound 16a. 1H-NMR (500 MHz, CDCl3): δ 9.15 (brs, 1H, 2-H), 8.12 (s, 1H, 8-H),8.03 (d, J = 8.0 Hz, 2H, Ar-H), 7.60 (t, J = 7.4 Hz, 1H, Ar-H), 7.52–7.49 (m, 3H, Ar-H), 7.40 (d, J = 8.8 Hz, 4H, Ar-H), 7.34–7.21 (m, 5H, Ar-H), 6.85 (d, J = 8.8 Hz, 3H, Ar-H), 5.29 (t, J = 1.1 Hz, 1H, 1'-H), 5.20 (d, J = 4.8 Hz, 1H, 2'-H), 4.79–4.68 (m, 1H, 6'-He), 4.27 (q, J = 5.5 Hz, 1H, 5'-H), 4.18 (dd, J = 7.9, 3.8 Hz, 1H, 4'-H), 4.07 (t, J = 4.7 Hz, 1H, 3'-H), 3.79 (s, 6H, OMe), 3.53 – 3.47 (m, 2H, 6'-Ha, OH), 3.41 (s, 3H, OMe), 3.29 (s, 3H, OMe). 13C-NMR (125 MHz, CDCl3): δ 164.63 (C-6); 158.52 (Ar-C); 152.46 (C-2); 151.78 (C-4); 149.56 (C-8); 144.66, 143.58, 135.84, 133.67, 132.75, 130.05, 128.84, 128.17, 127.85, 126.84, 123.11, 113.16 (Ar-C); 98.31 (C-1'); 86.33 (CTr-O); 76.26 (C-5'); 72.89 (C-3'); 64.68 (C-4'); 62.90 (C-6'); 57.89 (OMe); 56.87 (OMe); 55.94 (OMe); 55.19 (OMe); 53.38 (C-2'). HRMS calcd. for C41H42N5O8+ [M+H]+ 732.30332, found 732.3030.
3.21. 6'-O-Dimethoxytrityl-1',3'-di-O-methyl-2'-deoxy-2'-(N2-(dimethylamino)methylene-guanin-9-yl))-d-altropyranoside (16d)
Compound 16d (0.51 g, 74% yield) was synthesized from compound 15d (0.39 g, 0.974 mmol) using dimethoxytrityl chloride (0.362 g, 1.07 mmol) in anhydrous pyridine (10 mL) according to the procedure used for the synthesis of compound 16a. 1H-NMR (500 MHz, CDCl3): δ 9.20 (s, 1H, NH), 8.61 (d, J = 4.2 Hz, 1H, N=CH-N), 8.52 (s, 1H, 8-H), 7.79 (s, 1H, Ar-H), 7.68 (tt, J = 7.7, 1.8 Hz, 1H, Ar-H), 7.52–7.48 (m, 2H, Ar-H), 7.41–7.36 (m, 4H, Ar-H), 7.33–7.20 (m, 5H, Ar-H), 6.87–6.82 (m, 4H, Ar-H), 5.29 (s, 1H, 1'-H), 5.09 (d, J = 3.1 Hz, 1H, 2'-H), 4.79 (dd, J = 6.4, 3.1 Hz, 1H, 3'-H), 4.17–4.11 (m, 1H, 5'-H), 4.01 (brs, 1H, -OH), 3.88 (dd, J = 6.4, 4.0 Hz, 1H, 4'-H), 3.52 (dd, J = 10.2, 2.9 Hz, 1H, 6-He), 3.46 (s, 3H, OMe), 3.44 (s, 3H, OMe), 3.35 (dd, J = 10.2, 5.5 Hz, 1H, 6'-Ha), 3.05 (s, 3H, OMe), 2.93 (s, 3H, OMe). 13C-NMR (125 MHz, CDCl3): δ 158.50 (N=CH-N); 157.92 (MMTr); 157.78 (C-6); 156.55(C-2); 150.39(C-4); 149.81 (C-8); 144.84 (Ar-C), 137.33, 135.98, 135.87, 130.06, 130.02, 128.10, 127.85, 126.81, 123.70, 120.37, 113.13 (Ar-C); 99.83 (C-1'); 86.17 (CTr-O); 78.04 (C-5'); 71.31 (C-3'); 64.79 (C-4'); 63.87 (C-6'); 58.37 (OMe); 55.65 (OMe); 54.56 (C-2'); 41.12 and 35.12 (-NMe2). HRMS calcd. for C37H43N6O8+ [M+H]+ 699.3142, found 699.3121.
3.22. General Procedure for Nucleoside Phosphitylation
To a solution of the dimethoxytritylated nucleoside 16a (0.73 g, 1.2 mmol) in anhydrous CH2Cl2 (6 mL) at 0 °C and under argon atmosphere, freshly dried diisopropylethylamine (0.063 mL, 3.6 mmol) and 2-cyanoethyl-N,N-diisopropylchlorophosphoramidite (0.040 mL, 1.8 mmol) were added. The reaction mixture was stirred at 0 °C for 90 min after which completeness of the reaction was indicated by TLC. Saturated NaHCO3 solution (2 mL) was added, the solution was stirred for another10 min and partitioned between CH2Cl2 (50 mL) and aqueous NaHCO3 (30 mL). The organic layer was washed with brine (3 × 30 mL) and the aqueous phases were back extracted with CH2Cl2 (30 mL). After solvent evaporation, the resulting oil was purified by flash chromatography (hexane/acetone/TEA = 62/36/2). The yellow solid was then dissolved in CH2Cl2 (2 mL) and precipitated twice in cold hexane (160 mL, −30 °C) to afford the desired corresponding phosphoramidite nucleoside 17a (0.908 g, 93% yield) as a white powder. The obtained product was dried under vacuum and stored overnight under argon at −20 °C. 31P-NMR (CDCl3): δ = 150.84. HRMS calcd. for C42H54N4O10P1+ [M+H]+ 805.35773, found 805.3557; 31P-NMR (CDCl3): δ = 150.84.
Compound 17b (0.34 g, 79% yield) was synthesized from compound 16b (0.34 g, 0.47 mmol), dry diisopropylethylamine (0.025 mL, 1.42 mmol), 2-cyanoethyl-N,N-diisopropylchloro-phosphoramidite (0.016 mL, 0.71 mmol) and anhydrous CH2Cl2 (10 mL) according to procedure used for the synthesis of compound 17a. 31P-NMR (CDCl3): δ = 150.96. HRMS calcd. for C49H59N5O10P1 [M+H]+ 908.39993, found 908.3981.
Compound 17c (0.83 g, 85% yield) was synthesized from compound 16c (0.76 g, 1.03 mmol), dry diisopropylethylamine (0.054 mL, 3.09 mmol), 2-cyanoethyl-N,N-diisopropylchloro-phosphoramidite (0.034 mL, 1.54 mmol) and anhydrous CH2Cl2 (10 mL) according to procedure used for the synthesis of compound 17a. 31P-NMR (CDCl3): δ = 150.287 and 151.231. HRMS calcd. for C50H59N7O9P1+ [M+H]+ 932.41116, found 932.4103.
Compound 17d (0.42 g, 65% yield) was synthesized from compound 16d (0.5 g, 0.71 mmol), dry diisopropylethylamine (0.037 mL, 2.13 mmol), 2-cyanoethyl-N,N-diisopropylchloro-phosphoramidite (0.025 mL, 1.15 mmol) and anhydrous CH2Cl2 (10 mL) according to procedure used for the synthesis of compound 17a. 31P-NMR (CDCl3): δ = 150.065 and 151.206. HRMS calcd. for C46H60N8O9P1+ [M+H]+ 899.42206, found 899.4240.
4. Conclusions
A new nucleoside analogue scaffold for incorporation into oligonucleotides was developed and all four monomers with the natural heterocyclic bases have been prepared and evaluated on their hybridization potential with natural DNA and RNA. While it was anticipated that the constraint imposed by the 6-membered ring structure could afford the entropic advantage as seen with HNA and ANA constructs, an entropic penalty to preserve the 1C4 conformation required for pairing with RNA annulated the affinity gain which one could expect from a pre-organized structure.
Acknowledgments
This work was supported by a FWO (Flemish Scientific Research) grant G.0784.11 and KU Leuven financial support (GOA/10/13). Mass spectrometry was made possible by the support of the Hercules Foundation of the Flemish Government (grant 20100225-7). We are indebted to Chantal Biernaux for final typesetting.
Author Contributions
A.V. and D.K. synthesized and analyzed the different monomers; G.S. assembled oligonucleotides and carried out Tm analysis; J.R. carried out all mass spectrometric analysis; M.F. performed the modeling experiments; A.V.A. conceived and supervised research and wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Helene, C. The anti-gene strategy: Control of gene expression by triplex-forming-oligonucleotides. Anti-Cancer Drug Des. 1991, 6, 569–584. [Google Scholar]
- Wagner, R.W. Gene inhibition using antisense oligodeoxynucleotides. Nature 1994, 372, 333–335. [Google Scholar] [CrossRef] [PubMed]
- Opalinska, J.B.; Gewirtz, A.M. Nucleic-acid therapeutics: Basic principles and recent applications. Nat. Rev. Drug. Discov. 2002, 1, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Castanotto, D.; Stein, C.A. Antisense oligonucleotides in cancer. Curr. Opin. Oncol. 2014, 26, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Dean, N.M.; Bennett, C.F. Antisense oligonucleotide-based therapeutics for cancer. Oncogene 2003, 22, 9087–9096. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.Q.; Zhang, Y. RNAi-mediated gene silencing in cancer therapy. Expert Opin. Biol. Ther. 2012, 12, 1495–1504. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Yang, C.; Jiang, S.; Xu, X.-N.; Lu, X.; He, Y.-W.; Cheung, A.; Wang, H. Applications of RNA interference high-throughput screening technology in cancer biology and virology. Protein Cell 2014, 5, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhu, X.; Lu, P.Y.; Rosato, R.R.; Tan, W.; Zu, Y. Oligonucleotide aptamers: New tools for targeted cancer therapy. Mol. Ther. Nucleic Acids 2014, 3, e182. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K. Biotech Comes to Its “Antisenses” after Hard-Won Drug Approval. (Archived by WebCite® at http://www.webcitation.org/6WNb4M1rP). Available online: http://blogs.nature.com/spoonful/2013/02/biotech-comes-to-its-antisenses-after-hard-won-drug-approval.html (accessed on 6 January 2015).
- Lu, Q.L.; Cirak, S.; Partridge, T. What Can We Learn From Clinical Trials of Exon Skipping for DMD? Mol. Ther. Nucl. Acids 2014, 3, e152. [Google Scholar] [CrossRef]
- Soutschek, J.; Akinc, A.; Bramlage, B.; Charisse, K.; Constien, R.; Donoghue, M.; Elbashir, S.; Geick, A.; Hadwiger, P.; Harborth, J.; et al. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature 2004, 432, 173–178. [Google Scholar] [CrossRef] [PubMed]
- De Fougerolles, A.; Vornlocher, H.P.; Maraganore, J.; Lieberman, J. Interfering with disease: A progress report on siRNA-based therapeutics. Nat. Rev. Drug. Discov. 2007, 6, 443–453. [Google Scholar]
- Herdewijn, P. Heterocyclic modifications of oligonucleotides and antisense technology. Antisense Nucl. Acid. Drug Dev. 2000, 10, 297–310. [Google Scholar] [CrossRef]
- Herdewijn, P. Conformationally restricted carbohydrate-modified nucleic acids and antisense technology. Biochim. Biophys. Acta 1999, 1489, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Manoharan, M. RNA interference and chemically modified small interfering RNAs. Curr. Opin. Chem. Biol. 2004, 8, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Van Aerschot, A.; Verheggen, I.; Hendrix, C.; Herdewijn, P. 1,5-Anhydrohexitol nucleic acids, a new promising antisense construct. Angew. Chem. Int. Ed. Engl. 1995, 34, 1338–1339. [Google Scholar] [CrossRef]
- Hendrix, C.; Rosemeyer, H.; Verheggen, I.; Seela, F.; van Aerschot, A.; Herdewijn, P. 1',5'-Anhydrohexitol oligonucleotides: Synthesis, base pairing and recognition by regular oligodeoxyribonucleotides and oligoribonucleotides. Chem. Eur. J. 1997, 3, 110–120. [Google Scholar] [CrossRef]
- Allart, B.; Khan, K.; Rosemeyer, H.; Schepers, G.; Hendrix, C.; Rothenbacher, K.; Seela, F.; Van Aerschot, A.; Herdewijn, P. d-Altritol nucleic acids (ANA): Hybridisation properties, stability, and initial structural analysis. Chem. Eur. J. 1999, 5, 2424–2431. [Google Scholar] [CrossRef]
- Wang, J.; Verbeure, B.; Luyten, I.; Lescrinier, E.; Froeyen, M.; Hendrix, C.; Rosemeyer, H.; Seela, F.; van Aerschot, A.; Herdewijn, P. Cyclohexene nucleic acids (CeNA): Serum stable oligonucleotides that activate RNase H and increase duplex stability with complementary RNA. J. Am. Chem. Soc. 2000, 122, 8595–8602. [Google Scholar] [CrossRef]
- Singh, S.K.; Nielsen, P.; Koshkin, A.A.; Wengel, J. LNA (locked nucleic acids): Synthesis and high-affinity nucleic acid recognition. Chem. Commun. 1998, 455–456. [Google Scholar] [CrossRef]
- Veedu, R.N.; Wengel, J. Locked nucleic acids: Promising nucleic acid analogs for therapeutic applications. Chem. Biodivers. 2010, 7, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.; Abramov, M.; van Aerschot, A.; Xu, D.; Juliano, R.L.; Herdewijn, P. Inhibition of MDR1 expression with altritol-modified siRNAs. Nucl. Acids Res. 2007, 35, 1064–1074. [Google Scholar] [CrossRef] [PubMed]
- Chatelain, G.; Schepers, G.; Rozenski, J.; van Aerschot, A. Hybridization potential of oligonucleotides comprising 3'-O-methylated altritol nucleosides. Mol. Divers. 2012, 16, 825–837. [Google Scholar] [CrossRef] [PubMed]
- Richtmyer, N.K.; Hudson, C.S. Crystalline α-methyl-d-altroside and some new derivatives of d-altrose1. J. Am. Chem. Soc. 1941, 63, 1727–1731. [Google Scholar] [CrossRef]
- Teste, K.; Colombeau, L.; Hadj-Bouazza, A.; Lucas, R.; Zerrouki, R.; Krausz, P.; Champavier, Y. Solvent-controlled regioselective protection of 5'-O-protected thymidine. Carbohydr. Res. 2008, 343, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.; Teste, K.; Zerrouki, R.; Champavier, Y.; Guilloton, M. Chelation-controlled regioselective alkylation of pyrimidine 2'-deoxynucleosides. Carbohydr. Res. 2010, 345, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Divakar, K.J.; Reese, C.B. 4-(1,2,4-Triazol-1-yl) and 4-(3-nitro-1,2,4-triazol-1-yl)-1-(beta-d-2,3,5-tri-O-acetylarabinofuranosyl)pyrimidin-2(1H)-ones—Valuable intermediates in the synthesis of derivatives of 1-(beta-d-arabinofuranosyl)cytosine (Ara-C). J. Chem. Soc. Perk. Trans. 1 1982, 1171–1176. [Google Scholar] [CrossRef]
- Pulido, D.; Sánchez, A.; Robles, J.; Pedroso, E.; Grandas, A. Guanine-containing DNA minor-groove binders. Eur. J. Org. Chem. 2009, 2009, 1398–1406. [Google Scholar] [CrossRef]
- Jenny, T.F.; Schneider, K.C.; Benner, S.A. N-2-Isobutyrl-O-6-[2-(para-nitrophenyl)ethyl]guanine—A new building block for the efficient synthesis of carbocyclic guanosine analogs. Nucleos. Nucleot. 1992, 11, 1257–1261. [Google Scholar] [CrossRef]
- Mcbride, L.J.; Kierzek, R.; Beaucage, S.L.; Caruthers, M.H. Amidine protecting groups for oligonucleotide synthesis 16. J. Am. Chem. Soc. 1986, 108, 2040–2048. [Google Scholar] [CrossRef]
- Vu, H.; Mccollum, C.; Jacobson, K.; Theisen, P.; Vinayak, R.; Spiess, E.; Andrus, A. Fast Oligonucleotide deprotection phosphoramidite chemistry for DNA-synthesis. Tetrahedron Lett. 1990, 31, 7269–7272. [Google Scholar] [CrossRef]
- Theisen, P.; McCollum, C.; Andrus, A. N-6-Dialkylformamidine-2'-deoxyadenosine phosphoramidites in oligodeoxynucleotide synthesis—Rapid deprotection of oligodeoxynucleotides. Nucleos. Nucleot. 1993, 12, 1033–1046. [Google Scholar] [CrossRef]
- D’Alonzo, D.; van Aerschot, A.; Guaragna, A.; Palumbo, G.; Schepers, G.; Capone, S.; Rozenski, J.; Herdewijn, P. Synthesis and base pairing properties of 1',5'-anhydro-l-hexitol nucleic acids (l-HNA). Chem. Eur. J. 2009, 15, 10121–10131. [Google Scholar] [CrossRef] [PubMed]
- Van Aerschot, A.; Meldgaard, M.; Schepers, G.; Volders, F.; Rozenski, J.; Busson, R.; Herdewijn, P. Improved hybridisation potential of oligonucleotides comprising O-methylated anhydrohexitol nucleoside congeners. Nucl. Acids Res. 2001, 29, 4187–4194. [Google Scholar] [CrossRef] [PubMed]
- Vandermeeren, M.; Preveral, S.; Janssens, S.; Geysen, J.; Saison-Behmoaras, E.; Van Aerschot, A.; Herdewijn, P. Biological activity of hexitol nucleic acids targeted at Ha-ras and intracellular adhesion molecule-1 mRNA. Biochem. Pharmacol. 2000, 59, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.; Abramov, M.; Van Aerschot, A.; Rozenski, J.; Dixit, V.; Juliano, R.L.; Herdewijn, P. Biological effects of hexitol and altritol-modified siRNAs targeting B-Raf. Eur. J. Pharmacol. 2009, 606, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, V.B.; Taylor, A.I.; Cozens, C.; Abramov, M.; Renders, M.; Zhang, S.; Chaput, J.C.; Wengel, J.; Peak-Chew, S.Y.; McLaughlin, S.H.; et al. Synthetic genetic polymers capable of heredity and evolution. Science 2012, 336, 341–344. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.I.; Pinheiro, V.B.; Smola, M.J.; Morgunov, A.S.; Peak-Chew, S.; Cozens, C.; Weeks, K.M.; Herdewijn, P.; Holliger, P. Catalysts from synthetic genetic polymers. Nature 2015, 518, 427–430. [Google Scholar] [CrossRef] [PubMed]
- Mooers, B.H.; Singh, A. The crystal structure of an oligo(U):pre-mRNA duplex from a trypanosome RNA editing substrate. RNA 2011, 17, 1870–1883. [Google Scholar] [CrossRef] [PubMed]
- Salomon-Ferrer, R.; Case, D.A.; Walker, R.C. An overview of the Amber biomolecular simulation package. Wires Comput. Mol. Sci. 2013, 3, 198–210. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Only small amounts of the different amidites are still available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).