Enzymatic Polymerization of Cyclic Monomers in Ionic Liquids as a Prospective Synthesis Method for Polyesters Used in Drug Delivery Systems
Abstract
:1. Introduction
2. Polymeric Carriers
3. Macromolecular Conjugates of Active Substances

4. Aliphatic Polyesters as a Carriers of Therapeutic Agents
| Polyesters | Therapeutic Agent | Ref. |
|---|---|---|
| poly(lactide-co-glycolide) (PLG) | Amphotericin B | [23,24] |
| Ciprofloxacin | [25,26] | |
| Cisplatin | [27] | |
| Docetaxel | [28] | |
| Doxorubicin | [29,30] | |
| Paclitaxel | [31,32] | |
| Rifampicin | [33] | |
| r-hGH (recombinant human growth hormone) | [34] | |
| polylactide (PLA) | Camptothecin | [35,36] |
| Doxorubicin | [37,38] | |
| 5-fluorouracil | [39] | |
| poly(ε-caprolactone) (PCL) | Amphotericin B | [40] |
| Ciprofloxacin | [41] | |
| Citropin | [42] | |
| Vancomycin | [43] |
5. Ring Opening Polymerization of Cyclic Esters
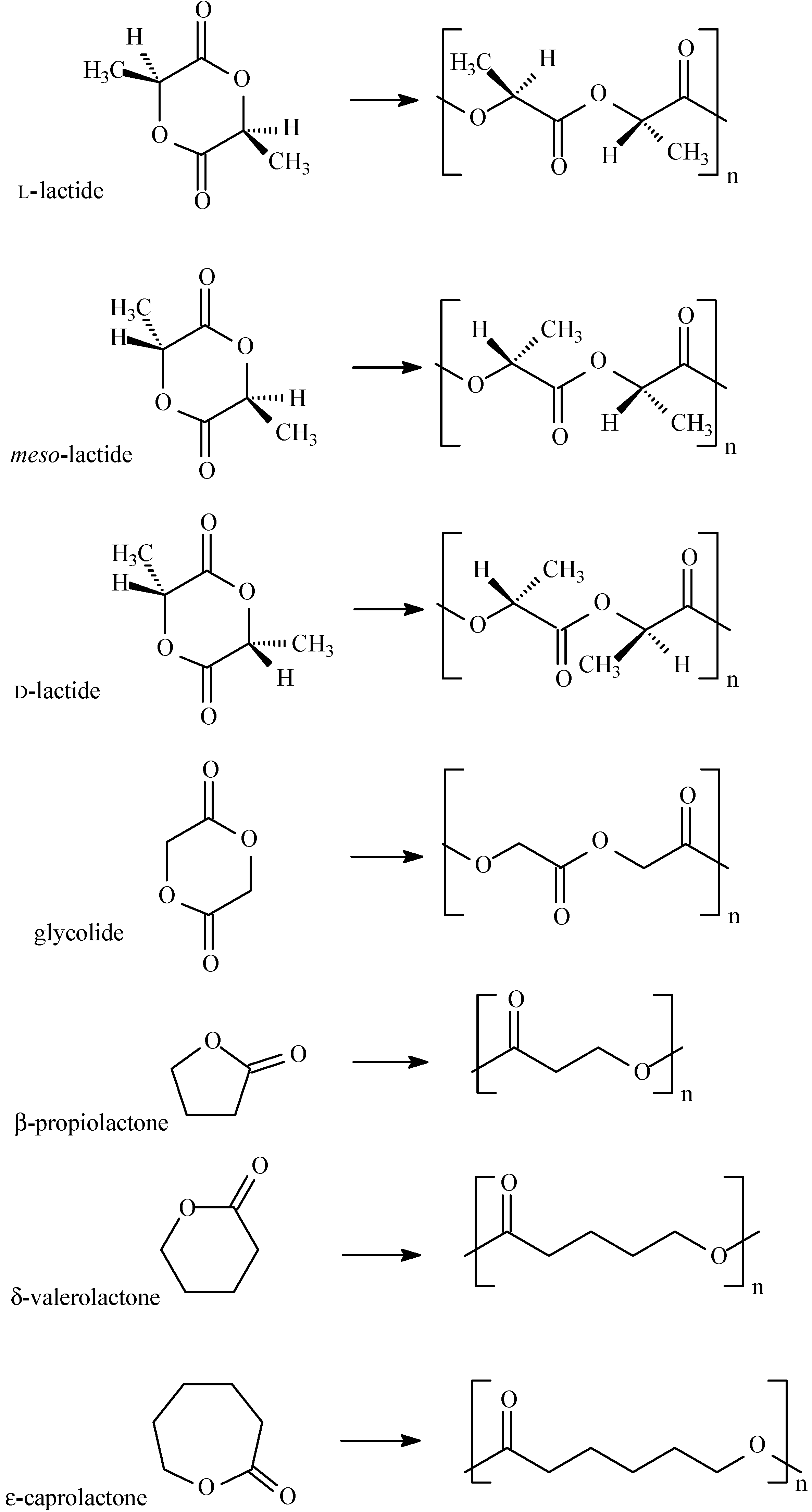
6. Biocatalytic Synthesis of Polymers for Biomedical Applications
| Origin of Lipases | Ref. |
|---|---|
| Aspergillus niger | [70] |
| Candida antarctica | [45,71,72,73,74,75,76,77,78] |
| Candida cylindracea | [45,70,78] |
| Candida rugosa | [70] |
| Penicillium roqueforti | [70] |
| Porcine pancreatic | [45,70,78,79,80,81,82] |
| Pseudomonas cepacia | [45,70,76,78] |
| Pseudomonas fluorescens | [45,70,76,78] |
| Yarrowia lipolytica | [83] |
6.1. Influence of the Reaction Media on Activity of Biocatalysts
6.2. Ionic Liquids
| ILs | Abbreviation |
|---|---|
| 1-Butyl-3-methylimidazolium tetrafluoroborate | [bmim][BF4] |
| 1-Butyl-3-methylimidazolium bis((trifluoromethyl)-sulfonyl)imide | [bmim][NTf2] |
| 1-Butyl-3-methylimidazolium hexafluorophosphate | [bmim][PF6] |
| 1-Butyl-3-methylimidazolium trifluoromethanesulfonate | [bmim][OTf] |
| 1-Ethyl-3-methylimidazolium tetrafluoroborate | [emim][BF4] |
| 1-Hexyl-3-methylimidazolium hexafluorophosphate | [hmim][PF6] |
| 1,4-bis(3-Hexylimidazolium-1-yl)butane bishexafluorophosphate | [C4(C6Im)2][PF6] |
| 1-Dodecyl-3-methylimidazolium bis((trifluoromethyl)-sulfonyl)imide | [C12MIm][NTf2] |
| 1-Dodecyl-3-methylimidazolium hexafluorophosphate | [C12MIm][PF6] |
6.2.1. Enzyme Stability in Ionic Liquids
6.2.2. Polarity of Ionic Liquids
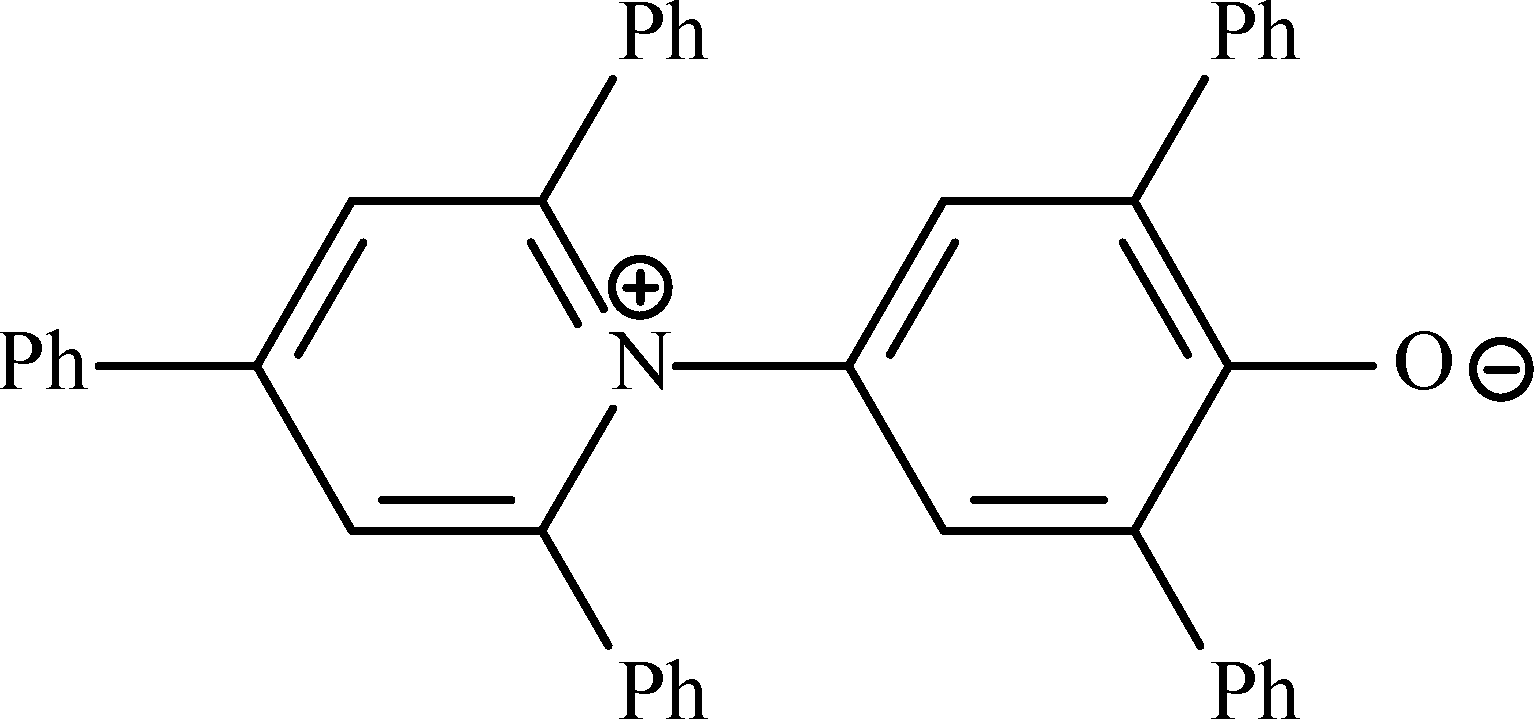
6.2.3. Hydrophobicity/Hydrophilicity
| ILs | Polarity ETN (RT) | Mw (g·mol−1) | Melting Point (°C) | Viscosity in 20 °C (cP) | Density (g·cm−3) | Conductivity (S·m−1) | Water Solubility (WS) (%w/v) |
|---|---|---|---|---|---|---|---|
| [bmim][BF4] | 0.680 | 226 | −82 | 233 | 1.17 | 0.17 | 100 |
| [bmim[Tf2N] | 0.645 | 419 | −4 | 52 | 1.43 | 0.39 | 1.40 |
| [bmim][PF6] | 0.676 | 284 | −8 | 312 | 1.362 | 0.14 | 0.13 |
6.2.4. Viscosity
6.2.5. Effect of Impurities
7. Ionic Liquids as a Medium for the Enzymatic Ring-Opening Polymerization of Cyclic Esters
| Monomer | ILs | Purity of ILs (%) | Enzyme | Temp. (°C) | Time (h) | Mn (Da) | PDI | Y/C (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| [bmim][BF4] | - | CA | 60 | 168 | 4200 | 2.7 | 97C | [123] | |
| [bmim][BF4] | ≥99 | CA | 60 | 24 | 12,700 | 1.8 | 35Y | [124] | |
| [bmim][BF4] | - | YLL | 100 | 16 | 1758 | 1.7 | - | [113] | |
| [bmim][BF4] | - | YLL | 150 | 6 | 3092 | 2.5 | - | [113] | |
| [bmim][PF6] | - | CA | 60 | 72 | 540 | 4.2 | 62C | [123] | |
| [bmim][PF6] | ≥99 | CA | 60 | 24 | 12,200 | 1.7 | 30Y | [124] | |
| CL | [bmim][NTf2] | ≥99 | CA | 60 | 24 | 10,500 | 2.1 | 44Y | [124] |
| [bmim][NTf2] | ≥97 | CA | 90 | 24 | 8100 | - | 85Y | [102] | |
| [C4(C6Im)2][PF6] | - | CA | 90 | 48 | 26,200 | - | 62Y | [125] | |
| [C12MIm][PF6] | - | CA | 90 | 48 | 11,700 | - | 37Y | [125] | |
| [C12MIm][NTf2] | - | CA/ILs | 60 | 48 | 35,600 | - | 62Y | [125] | |
| [C12MIm][NTf2] | - | CA | 60 | 48 | 20,300 | - | 54Y | [125] | |
| LLA | [bmim][BF4] | ≥99 | CA | 110 | 24 | 54,600 | 1.25 | 24.3Y96.2C | [126] |
| [bmim][PF6] | ≥99 | CA | 65 | 264 | 581 | 1.2 | 29.5Y | [127] | |
| [bmim][PF6] | - | CA | 90 | 120 | 19,600 | 1.2 | - | [128] | |
| [bmim][PF6] | ≥99 | CA | 120 | 24 | 3900 | 1.19 | 0.1Y90.4C | [126] | |
| [hmim][PF6] | ≥97 | CA | 90 | 168 | 37,800 | 1.3 | 63.2Y | [129] | |
| [hmim][PF6] | ≥97 | CA | 65 | 120 | 1700 | 1.3 | 16.5Y | [129] | |
| [bmim][NTf2] | ≥99 | CA | 120 | 24 | 50,100 | 1.42 | 10.5Y | [126] | |
| DO | [bmim][PF6] | ≥99 | CA/ILs | 70 | 18 | 182,100 | - | - | [130] |
| [bmim][PF6] | ≥99 | CA | 70 | 24 | 27,700 | - | - | [130] | |
| GA | [bmim][PF6] | ≥99 | CA | 65 | 96 | - | - | - | [127] |
| LLA:GA (1:3) | [hmim][PF6] | ≥97 | CA | 65 | 120 | 3500 | 1.3 | 40.2Y | [129] |
| [bmim][PF6] | ≥99 | CA | 65 | 96 | 2400 | 1.1 | 36.7Y | [127] | |
| LLA:GA (3:1) | [bmim][PF6] | ≥99 | CA | 90 | 144 | 18,500 | 1.1 | - | [127] |
7.1. Effect of Monomer and Polymer Solubility in ILs
7.2. Effect of Temperature
7.3. Effect of Enzyme Preparations
7.4. Structure of Polyesters Obtained in ILs
7.5. Kinetics of Polyester Enzymatic Synthesis in ILs
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rohini, N.A.; Joseph, A.; Mukerji, A. Polymeric prodrugs: Recent achievements and general strategies. J. Antivir. Antiretrovir. 2013. [Google Scholar] [CrossRef]
- Allen, T.M.; Cullis, P.R. Drug delivery systems: Entering the mainstream. Science 2004, 303, 1818–1822. [Google Scholar] [CrossRef] [PubMed]
- Tong, R.; Gabrielson, N.P.; Fan, T.M.; Cheng, J.J. Polymeric nanomedicines based on poly(lactide) and poly(lactide-co-glycolide). Curr. Opin. Solid State Mater. Sci. 2012, 16, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Sobczak, M.; Oledzka, E.; Kolodziejski, W.L.; Kuzmicz, R. Polymers for pharmaceutical applications. Polimery 2007, 52, 411–420. [Google Scholar]
- Hoste, K.; de Winne, K.; Schacht, E. Polymeric prodrugs. Int. J. Pharm. 2004, 277, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Vilar, G.; Tulla-Puche, J.; Albericio, F. Polymers and drug delivery systems. Curr. Drug Deliv. 2012, 9, 367–394. [Google Scholar] [CrossRef] [PubMed]
- Sobczak, M.; Debek, C.; Oledzka, E.; Kozlowski, R. Polymeric systems of antimicrobial peptides-strategies and potential applications. Molecules 2013, 18, 14122–14137. [Google Scholar] [CrossRef] [PubMed]
- Marinaro, W.; Stella, V. Macromolecular prodrugs of small molecules. In Prodrugs; Stella, V., Borchardt, R., Hageman, M., Oliyai, R., Maag, H., Tilley, J., Eds.; Springer: New York, NY, USA, 2007; Volume 5, pp. 989–1021. [Google Scholar]
- Elvira, C.; Gallardo, A.; San Roman, J.; Cifuentes, A. Covalent polymer-drug conjugates. Molecules 2005, 10, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Shu, J.Y.; Panganiban, B.; Xu, T. Peptide-polymer conjugates: From fundamental science to application. Annu. Rev. Phys. Chem. 2013, 64, 631–657. [Google Scholar] [CrossRef] [PubMed]
- Khandare, J.; Minko, T. Polymer-drug conjugates: Progress in polymeric prodrugs. Prog. Polym. Sci. 2006, 31, 359–397. [Google Scholar] [CrossRef]
- Ringsdorf, H. Structure and properties of pharmacologically active polymers. J. Polym. Sci. Symp. 1975, 51, 135–153. [Google Scholar] [CrossRef]
- Ringsdorf, H. Synthetic Polymeric Drugs. In Polymeric Delivery Systems; Kostelnik, R.J., Ed.; Gordon and Breach Science Publishers, Inc.: New York, NY, USA, 1978; pp. 197–225. [Google Scholar]
- Liu, Z.; Jiao, Y.; Wang, Y.; Zhou, C.; Zhang, Z. Polysaccharides-based nanoparticles as drug delivery systems. Adv. Drug Deliv. Rev. 2008, 60, 1650–1662. [Google Scholar] [CrossRef] [PubMed]
- Oledzka, E.; Sobczak, M. Polymers in the pharmaceutical applications—natural and bioactive initiators and catalysts in the synthesis of biodegradable and bioresorbable polyesters and polycarbonates. In Innovations in Biotechnology; InTech: Rijeka, Croatia, 2012; pp. 139–160. [Google Scholar]
- Solaro, R.; Chiellini, F.; Battisti, A. Targeted delivery of protein drugs by nanocarriers. Materials 2010, 3, 1928–1980. [Google Scholar] [CrossRef]
- Haag, R.; Kratz, F. Polymer therapeutics: Concepts and applications. Angew. Chem. Int. Ed. 2006, 45, 1198–1215. [Google Scholar] [CrossRef]
- Maeda, H.; Wu, J.; Sawa, T.; Matsumura, Y.; Hori, K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: A review. J. Control. Release 2000, 65, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. The enhanced permeability and retention (EPR) effect in tumor vasculature: The key role of tumor-selective macromolecular drug targeting. Adv. Enzym. Regul. 2001, 41, 189–207. [Google Scholar] [CrossRef]
- Goodarzi, N.; Varshochian, R.; Kamalinia, G.; Atyabi, F.; Dinarvand, R. A review of polysaccharide cytotoxic drug conjugates for cancer therapy. Carbohyd. Polym. 2013, 92, 1280–1293. [Google Scholar] [CrossRef]
- Ikada, Y.; Tsuji, H. Biodegradable polyesters for medical and ecological applications. Macromol. Rapid Commun. 2000, 21, 117–132. [Google Scholar] [CrossRef]
- Uhrich, K.E.; Cannizzaro, S.M.; Langer, R.S.; Shakesheff, K.M. Polymeric systems for controlled drug release. Chem. Rev. 1999, 99, 3181–3198. [Google Scholar] [CrossRef] [PubMed]
- Van de Ven, H.; Paulussen, C.; Feijens, P.B.; Matheeussen, A.; Rombaut, P.; Kayaert, P.; van den Mooter, G.; Weyenberg, W.; Cos, P.; Maes, L.; et al. PLGA nanoparticles and nanosuspensions with amphotericin B: Potent in vitro and in vivo alternatives to fungizone and ambisome. J. Control. Release 2012, 161, 795–803. [Google Scholar]
- Nahar, M.; Jain, N.K. Preparation, characterization and evaluation of targeting potential of amphotericin B-loaded engineered PLGA nanoparticles. Pharm. Res. 2009, 26, 2588–2598. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.I.; Na, H.S.; Nah, J.W.; Lee, H.C. Preparation of ciprofloxacin-encapsulated poly(DL-lactide-co-glycolide) microspheres and its antibacterial activity. J. Pharm. Sci. 2009, 98, 3659–3665. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.I.; Na, H.S.; Seo, D.H.; Kim, D.G.; Lee, H.C.; Jang, M.K.; Na, S.K.; Roh, S.H.; Kim, S.I.; Nah, J.W. Ciprofloxacin-encapsulated poly(DL-lactide-co-glycolide) nanoparticles and its antibacterial activity. Int. J. Pharm. 2008, 352, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Verrijk, R.; Smolders, I.J.; Bosnie, N.; Begg, A.C. Reduction of systemic exposure and toxicity of cisplatin by encapsulation in poly-lactide-co-glycolide. Cancer Res. 1992, 52, 6653–6656. [Google Scholar] [PubMed]
- Esmaeili, F.; Dinarvand, R.; Ghahremani, M.H.; Ostad, S.N.; Esmaily, H.; Atyabi, F. Cellular cytotoxicity and in vivo biodistribution of docetaxel poly (lactide-co-glycolide) nanoparticles. Anticancer Drugs 2010, 21, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Tewes, F.; Munnier, E.; Antoon, B.; Ngaboni Okassa, L.; Cohen-Jonathan, S.; Marchais, H.; Douziech-Eyrolles, L.; Soucé, M.; Dubois, P.; Chourpa, I. Comparative study of doxorubicin-loaded poly (lactide-co-glycolide) nanoparticles prepared by single and double emulsion methods. Eur.J. Pharm. Biopharm. 2007, 66, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.S.; Lee, E.A.; Park, T.G. Doxorubicin-conjugated biodegradable polymeric micelles having acid-cleavable linkages. J. Control. Release 2002, 82, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Feng, S.S. Poly(D,L-lactide-co-glycolide) (PLGA) nanoparticles prepared by high pressure homogenization for paclitaxel chemotherapy. Int. J. Pharm. 2007, 342, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Kou, G.; Gao, J.; Wang, H.; Chen, H.W.; Li, B.H.; Zhang, D.P.; Wang, S.H.; Hou, S.; Qian, W.Z.; Dai, J.X.; et al. Preparation and characterization of paclitaxel-loaded PLGA nanoparticles coated with cationic SM5-1 single-chain antibody. J. Biochem. Mol. Biol. 2007, 40, 731–739. [Google Scholar] [CrossRef]
- Esmaeili, F.; Hosseini-Nasr, M.; Rad-Malekshahi, M.; Samadi, N.; Atyabi, F.; Dinarvand, R. Preparation and antibacterial activity evaluation of rifampicin-loaded poly lactide-co-glycolide nanoparticles. Nanomedicine 2007, 3, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Rafi, M.; Singh, S.M.; Kanchan, V.; Anish, C.K.; Panda, A.K. Controlled release of bioactive recombinant human growth hormone from PLGA microparticles. J. Microencapsul. 2010, 27, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Tong, R.; Cheng, J.J. Controlled synthesis of camptothecin-polylactide conjugates and nanoconjugates. Bioconjugate Chem. 2010, 21, 111–121. [Google Scholar] [CrossRef]
- Sobczak, M.; Oledzka, E.; Kwietniewska, M.; Nalecz-Jawecki, G.; Kolodziejski, W. Promising macromolecular conjugates of camptothecin—The synthesis, characterization and in vitro studies. J. Macromol. Sci. A 2014, 51, 254–262. [Google Scholar] [CrossRef]
- Liu, S.; Zhou, G.Y.; Liu, D.X.; Xie, Z.G.; Huang, Y.B.; Wang, X.; Wu, W.B.; Jing, X.B. Inhibition of orthotopic secondary hepatic carcinoma in mice by doxorubicin-loaded electrospun polylactide nanofibers. J. Mater. Chem. 2013, 1, 101–109. [Google Scholar] [CrossRef]
- Tong, R.; Tang, L.; Yala, L.; Cheng, J.J. Controlled formulation of doxorubicin-polylactide nanoconjugates for cancer drug delivery. Conf. Proc. IEE Eng. Med. Biol. Soc. 2009, 2400–2402. [Google Scholar] [CrossRef]
- Nagarwal, R.C.; Singh, P.N.; Kant, S.; Maiti, P.; Pandit, J.K. Chitosan coated PLA nanoparticles for ophthalmic delivery: Characterization, in vitro and in vivo study in rabbit eye. J. Biomed. Nanotechnol. 2010, 6, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Espuelas, M.; Legrand, P.; Loiseau, P.; Bories, C.; Barratt, G.; Irache, J. In vitro antileishmanial activity of amphotericin B loaded in poly (ε-caprolactone) nanospheres. J. Drug Target. 2002, 10, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Dang, N.T.T.; Turner, M.S.; Coombes, A.G.A. Development of intra-vaginal matrices from polycaprolactone for sustained release of antimicrobial agents. J. Biomater. Appl. 2013, 28, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Sobczak, M.; Kamysz, W.; Tyszkiewicz, W.; Dębek, C.; Kozłowski, R.; Olędzka, E.; Piotrowska, U.; Nałęcz-Jawecki, G.; Plichta, A.; Grzywacz, D. Biodegradable macromolecular conjugates of citropin: Synthesis, characterization and in vitro efficiency study. React. Funct. Polym. 2014, 83, 54–61. [Google Scholar] [CrossRef]
- Le Ray, A.M.; Chiffoleau, S.; Iooss, P.; Grimandi, G.; Gouyette, A.; Daculsi, G.; Merle, C. Vancomycin encapsulation in biodegradable poly(epsilon-caprolactone) microparticles for bone implantation. Influence of the formulation process on size, drug loading, in vitro release and cytocompatibility. Biomaterials 2003, 24, 443–449. [Google Scholar]
- Jérôme, C.; Lecomte, P. Recent advances in the synthesis of aliphatic polyesters by ring-opening polymerization. Adv. Drug Deliv. Rev. 2008, 60, 1056–1076. [Google Scholar] [CrossRef] [PubMed]
- Namekawa, S.; Suda, S.; Uyama, H.; Kobayashi, S. Lipase-catalyzed ring-opening polymerization of lactones to polyesters and its mechanistic aspects. Int. J. Biol. Macromol. 1999, 25, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Nuyken, O.; Pask, S.D. Ring-opening polymerization-an introductory review. Polymers 2013, 5, 361–403. [Google Scholar] [CrossRef]
- Varma, I.K.; Albertsson, A.C.; Rajkhowa, R.; Srivastava, R.K. Enzyme catalyzed synthesis of polyesters. Prog. Polym. Sci. 2005, 30, 949–981. [Google Scholar] [CrossRef]
- Kobayashi, S.; Uyama, H.; Kimura, S. Enzymatic polymerization. Chem. Rev. 2001, 101, 3793–3818. [Google Scholar] [CrossRef] [PubMed]
- Albertsson, A.C.; Srivastava, R.K. Recent developments in enzyme-catalyzed ring-opening polymerization. Adv. Drug Deliv. Rev. 2008, 60, 1077–1093. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Uyama, H. Enzymatic polymerization to polyesters. Biopolym. Online 2002. [Google Scholar] [CrossRef]
- Jeon, B.W.; Lee, J.; Kim, H.S.; Cho, D.H.; Lee, H.; Chang, R.; Kim, Y.H. Lipase-catalyzed enantioselective synthesis of (R,R)-lactide from alkyl lactate to produce PDLA (poly D-lactic acid) and stereocomplex PLA (poly lactic acid). J. Biotechnol. 2013, 168, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Kadokawa, J.; Kobayashi, S. Polymer synthesis by enzymatic catalysis. Curr. Opin. Chem. Biol. 2010, 14, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Gross, R.A.; Kumar, A.; Kalra, B. Polymer synthesis by in vitro enzyme catalysis. Chem. Rev. 2001, 101, 2097–2124. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Uyama, H.; Ohmae, M. Enzymatic polymerization for precision polymer synthesis. Bull. Chem. Soc. Jpn. 2001, 74, 613–635. [Google Scholar] [CrossRef]
- Zhang, C.; Liao, L.; Gong, S. Recent developments in microwave-assisted polymerization with a focus on ring-opening polymerization. Green Chem. 2007, 9, 303–314. [Google Scholar] [CrossRef]
- Adrio, J.L.; Demain, A.L. Microbial enzymes: Tools for biotechnological processes. Biomolecules 2014, 4, 117–139. [Google Scholar] [CrossRef] [PubMed]
- Duda, A.; Kowalski, A.; Penczek, S.; Uyama, H.; Kobayashi, S. Kinetics of the ring-opening polymerization of 6-, 7-, 9-, 12-, 13-, 16-, and 17-membered lactones. Comparison of chemical and enzymatic polymerizations. Macromolecules 2002, 35, 4266–4270. [Google Scholar] [CrossRef]
- Uyama, H.; Takeya, K.; Kobayashi, S. Enzymatic ring-opening polymerization of lactones to polyesters by lipase catalyst—Unusually high reactivity of macrolides. Bull. Chem. Soc. Jpn. 1995, 68, 56–61. [Google Scholar] [CrossRef]
- Kobayashi, S. Lipase-catalyzed polyester synthesis—A green polymer chemistry. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2010, 86, 338–365. [Google Scholar] [CrossRef] [PubMed]
- Hasan, F.; Shah, A.A.; Hameed, A. Industrial applications of microbial lipases. Enzym. Microb. Technol. 2006, 39, 235–251. [Google Scholar] [CrossRef]
- Wehtje, E.; Costes, D.; Adlercreutz, P. Enantioselectivity of lipases: Effects of water activity. J. Mol. Catal. B 1997, 3, 221–230. [Google Scholar] [CrossRef]
- Mendes, A.A.; Oliveira, P.C.; de Castro, H.F. Properties and biotechnological applications of porcine pancreatic lipase. J. Mol. Catal. B 2012, 78, 119–134. [Google Scholar] [CrossRef]
- Gupta, R.; Gupta, N.; Rathi, P. Bacterial lipases: An overview of production, purification and biochemical properties. Appl. Microbiol. Biotechnol. 2004, 64, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Chisti, Y.; Banerjee, U.C. Production, purification, characterization, and applications of lipases. Biotechnol. Adv. 2001, 19, 627–662. [Google Scholar] [CrossRef] [PubMed]
- Idris, A.; Bukhari, A. Immobilized Candida antarctica lipase B: Hydration, stripping off and application in ring opening polyester synthesis. Biotechnol. Adv. 2012, 30, 550–563. [Google Scholar] [CrossRef] [PubMed]
- Miletic, N.; Nastasovic, A.; Loos, K. Immobilization of biocatalysts for enzymatic polymerizations: Possibilities, advantages, applications. Bioresour. Technol. 2012, 115, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Uyama, H.; Kuwabara, M.; Tsujimoto, T.; Kobayashi, S. High-performance immobilized lipase catalyst for polyester synthesis. Polym. J. 2002, 34, 970–972. [Google Scholar] [CrossRef]
- Chen, B.; Miller, M.E.; Gross, R.A. Effects of porous polystyrene resin parameters on Candida antarctica lipase B adsorption, distribution, and polyester synthesis activity. Langmuir 2007, 23, 6467–6474. [Google Scholar] [CrossRef] [PubMed]
- De Diego, T.; Lozano, P.; Abad, M.A.; Steffensky, K.; Vaultier, M.; Iborra, J.L. On the nature of ionic liquids and their effects on lipases that catalyze ester synthesis. J. Biotechnol. 2009, 140, 234–241. [Google Scholar]
- Kobayashi, S. Recent developments in lipase-catalyzed synthesis of polyesters. Macromol. Rapid Commun. 2009, 30, 237–266. [Google Scholar] [CrossRef] [PubMed]
- Cordova, A.; Iversen, T.; Hult, K. Lipase-catalyzed formation of end-functionalized poly(epsilon-caprolactone) by initiation and termination reactions. Polymer 1999, 40, 6709–6721. [Google Scholar] [CrossRef]
- Deng, F.; Gross, R.A. Ring-opening bulk polymerization of epsilon-caprolactone and trimethylene carbonate catalyzed by lipase Novozym 435. Int. J. Biol. Macromol. 1999, 25, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Hans, M.; Keul, H.; Moeller, M. Ring-opening polymerization of DD-lactide catalyzed by Novozyme 435. Macromol. Biosci. 2009, 9, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Bisht, K.S.; Henderson, L.A.; Gross, R.A.; Kaplan, D.L.; Swift, G. Enzyme-catalyzed ring-opening polymerization of omega-pentadecalactone. Macromolecules 1997, 30, 2705–2711. [Google Scholar] [CrossRef]
- Meyer, U.; Palmans, A.R.A.; Loontjens, T.; Heise, A. Enzymatic ring-opening polymerization and atom transfer radical polymerization from a bifunctional initiator. Macromolecules 2002, 35, 2873–2875. [Google Scholar] [CrossRef]
- Sobczak, M. Enzyme-catalyzed ring-opening polymerization of cyclic esters in the presence of poly(ethylene glycol). J. Appl. Polym. Sci. 2012, 125, 3602–3609. [Google Scholar] [CrossRef]
- Mei, Y.; Kumar, A.; Gross, R. Kinetics and mechanism of Candida antarctica lipase B catalyzed solution polymerization of epsilon-caprolactone. Macromolecules 2003, 36, 5530–5536. [Google Scholar] [CrossRef]
- Bisht, K.S.; Svirkin, Y.Y.; Henderson, L.A.; Gross, R.A.; Kaplan, D.L.; Swift, G. Lipase-catalyzed ring-opening polymerization of trimethylene carbonate. Macromolecules 1997, 30, 7735–7742. [Google Scholar] [CrossRef]
- Divakar, S. Porcine pancreas lipase catalyzed ring-opening polymerization of epsilon-caprolactone. J. Macromol. Sci. Pure 2004, A41, 537–546. [Google Scholar] [CrossRef]
- Yu, X.H.; Zhuo, R.X.; Feng, J. Ring-opening polymerization of trimethylene carbonate catalyzed by PPL immobilized on silica nanoparticles. Chem. J. Chin. Univ. 2005, 26, 978–981. [Google Scholar]
- Matsumura, S.; Tsukada, K.; Toshima, K. Novel lipase-catalyzed ring-opening copolymerization of lactide and trimethylene carbonate forming poly(ester carbonate)s. Int. J. Biol. Macromol. 1999, 25, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Henderson, L.A.; Svirkin, Y.Y.; Gross, R.A.; Kaplan, D.L.; Swift, G. Enzyme-catalyzed polymerizations of epsilon-caprolactone: Effects of initiator on product structure, propagation kinetics, and mechanism. Macromolecules 1996, 29, 7759–7766. [Google Scholar] [CrossRef]
- Barrera-Rivera, K.A.; Flores-Carreon, A.; Martinez-Richa, A. Enzymatic ring-opening polymerization of epsilon-caprolactone by a new lipase from yarrowia lipolytica. J. Appl. Polym. Sci. 2008, 109, 708–719. [Google Scholar] [CrossRef]
- Torres, S.; Castro, G.R. Non-aqueous biocatalysis in homogeneous solvent systems. Food Technol. Biotechnol. 2004, 42, 271–277. [Google Scholar]
- Sharma, S.; Kanwar, S.S. Organic solvent tolerant lipases and applications. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef]
- Dong, H.; Wang, H.D.; Cao, S.G.; Shen, J.C. Lipase-catalyzed polymerization of lactones and linear hydroxyesters. Biotechnol. Lett. 1998, 20, 905–908. [Google Scholar] [CrossRef]
- Zaks, A.; Klibanov, A.M. Enzymatic catalysis in organic media at 100 degrees C. Science 1984, 224, 1249–1251. [Google Scholar] [CrossRef] [PubMed]
- Zaks, A.; Klibanov, A.M. Substrate-specificity of enzymes in organic solvents vs. water is reversed. J. Am. Chem. Soc. 1986, 108, 2767–2768. [Google Scholar] [CrossRef]
- Zaks, A.; Klibanov, A.M. Enzyme-catalyzed processes in organic-solvents. Proc. Natl. Acad. Sci. USA 1985, 82, 3192–3196. [Google Scholar] [CrossRef] [PubMed]
- Zaks, A.; Klibanov, A.M. The effect of water on enzyme action in organic media. J. Biol. Chem. 1988, 263, 8017–8021. [Google Scholar] [PubMed]
- Madeira Lau, R.; van Rantwijk, F.; Seddon, K.R.; Sheldon, R.A. Lipase-catalyzed reactions in ionic liquids. Org. Lett. 2000, 2, 4189–4191. [Google Scholar]
- Kaar, J.L.; Jesionowski, A.M.; Berberich, J.A.; Moulton, R.; Russell, A.J. Impact of ionic liquid physical properties on lipase activity and stability. J. Am. Chem. Soc. 2003, 125, 4125–4131. [Google Scholar] [CrossRef] [PubMed]
- Tavares, A.P.M.; Rodríguez, O.; Macedo, E.A. New Generations of Ionic Liquids Applied to Enzymatic Biocatalysis. In Ionic Liquids-New Aspects for the Future; Kadokawa, J., Ed.; InTech: Rijeka, Croatia, 2013; pp. 537–556. [Google Scholar]
- Yang, Z.; Pan, W. Ionic liquids: Green solvents for nonaqueous biocatalysis. Enzym. Microb. Technol. 2005, 37, 19–28. [Google Scholar] [CrossRef]
- Van Rantwijk, F.; Madeira Lau, R.; Sheldon, R.A. Biocatalytic transformations in ionic liquids. Trends Biotechnol. 2003, 21, 131–138. [Google Scholar]
- Sureshkumar, M.; Lee, C.-K. Biocatalytic reactions in hydrophobic ionic liquids. J. Mol. Catal. B-Enzym. 2009, 60, 1–12. [Google Scholar] [CrossRef]
- Kubisa, P. Ionic liquids as solvents for polymerization processes—Progress and challenges. Prog. Polym. Sci. 2009, 34, 1333–1347. [Google Scholar] [CrossRef]
- Dupont, J.; de Souza, R.F.; Suarez, P.A. Ionic liquid (molten salt) phase organometallic catalysis. Chem. Rev. 2002, 102, 3667–3692. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Sun, N.; He, X.; Lu, X.; Zhang, X. Physical properties of ionic liquids: Database and evaluation. J. Phys. Chem. Ref. Data 2006, 35, 1475–1517. [Google Scholar] [CrossRef]
- Park, S.; Kazlauskas, R.J. Biocatalysis in ionic liquids–advantages beyond green technology. Curr. Opin. Biotechnol. 2003, 14, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Keskin, S.; Kayrak-Talay, D.; Akman, U.; Hortaçsu, Ö. A review of ionic liquids towards supercritical fluid applications. J. Supercrit. Fluids 2007, 43, 150–180. [Google Scholar] [CrossRef]
- Gorke, J.T.; Okrasa, K.; Louwagie, A.; Kazlauskas, R.J.; Srienc, F. Enzymatic synthesis of poly(hydroxyalkanoates) in ionic liquids. J. Biotechnol. 2007, 132, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Galonde, N.; Nott, K.; Debuigne, A.; Deleu, M.; Jerôme, C.; Paquot, M.; Wathelet, J.P. Use of ionic liquids for biocatalytic synthesis of sugar derivatives. J. Chem. Technol. Biot. 2012, 87, 451–471. [Google Scholar] [CrossRef]
- Dupont, J. On the solid, liquid and solution structural organization of imidazolium ionic liquids. J. Braz. Chem. Soc. 2004, 15, 341–350. [Google Scholar] [CrossRef]
- Lozano, P.; de Diego, T.; Carrie, D.; Vaultier, M.; Iborra, J. Over-stabilization of Candida antarctica lipase B by ionic liquids in ester synthesis. Biotechnol. Lett. 2001, 23, 1529–1533. [Google Scholar] [CrossRef]
- Reichardt, C. Solvatochromic dyes as solvent polarity indicators. Chem. Rev. 1994, 94, 2319–2358. [Google Scholar] [CrossRef]
- Reichardt, C. Polarity of ionic liquids determined empirically by means of solvatochromic pyridinium N-phenolate betaine dyes. Green Chem. 2005, 7, 339–351. [Google Scholar] [CrossRef]
- Carda–Broch, S.; Berthod, A.; Armstrong, D. Solvent properties of the 1-butyl-3-methylimidazolium hexafluorophosphate ionic liquid. Anal. Bioanal. Chem. 2003, 375, 191–199. [Google Scholar] [PubMed]
- Carmichael, A.J.; Seddon, K.R. Polarity study of some 1-alkyl-3-methylimidazolium ambient-temperature ionic liquids with the solvatochromic dye, Nile Red. J. Phys. Org. Chem. 2000, 13, 591–595. [Google Scholar]
- Bell, G.; Halling, P.J.; Moore, B.D.; Partridge, J.; Rees, D.G. Biocatalyst behaviour in low-water systems. Trends Biotechnol. 1995, 13, 468–473. [Google Scholar] [CrossRef]
- Ventura, S.P.; Santos, L.D.; Saraiva, J.A.; Coutinho, J.A. Concentration effect of hydrophilic ionic liquids on the enzymatic activity of Candida antarctica lipase B. World J Microb. Biot. 2012, 28, 2303–2310. [Google Scholar] [CrossRef]
- Martinelle, M.; Holmquist, M.; Hult, K. On the interfacial activation of Candida antarctica lipase A and B as compared with Humicola lanuginosa lipase. BBA—Lipids Lipid Metab. 1995, 1258, 272–276. [Google Scholar] [CrossRef]
- Barrera-Rivera, K.A.; Marcos-Fernandez, A.; Vera-Graziano, R.; Martinez-Richa, A. Enzymatic ring-opening polymerization of epsilon-caprolactone by Yarrowia lipolytica lipase in ionic liquids. J. Polym. Sci. A Polym. Chem. 2009, 47, 5792–5805. [Google Scholar] [CrossRef]
- Lozano, P.; de Diego, T.; Iborra, J. Hydrophobicity and water activity relationships of water-miscible aprotic solvents on kyotorphin synthesis catalyzed by α-chymotrypsin. Biotechnol. Lett. 1997, 19, 1005–1009. [Google Scholar] [CrossRef]
- Levitsky, V.Y.; Lozano, P.; Iborra, J.L. Designing enzymatic kyotorphin synthesis in organic media with low water content. Enzym. Microb. Technol. 2000, 26, 608–613. [Google Scholar] [CrossRef]
- Zhao, H.; Baker, G.A.; Song, Z.; Olubajo, O.; Zanders, L.; Campbell, S.M. Effect of ionic liquid properties on lipase stabilization under microwave irradiation. J. Mol. Catal. B-Enzym. 2009, 57, 149–157. [Google Scholar] [CrossRef]
- Huddleston, J.G.; Visser, A.E.; Reichert, W.M.; Willauer, H.D.; Broker, G.A.; Rogers, R.D. Characterization and comparison of hydrophilic and hydrophobic room temperature ionic liquids incorporating the imidazolium cation. Green Chem. 2001, 3, 156–164. [Google Scholar] [CrossRef]
- Kim, K.-W.; Song, B.; Choi, M.-Y.; Kim, M.-J. Biocatalysis in ionic liquids: Markedly enhanced enantioselectivity of lipase. Org. Lett. 2001, 3, 1507–1509. [Google Scholar] [CrossRef] [PubMed]
- Baudequin, C.; Baudoux, J.; Levillain, J.; Cahard, D.; Gaumont, A.-C.; Plaquevent, J.-C. Ionic liquids and chirality: Opportunities and challenges. Tetrahedron-Asymmetr 2003, 14, 3081–3093. [Google Scholar] [CrossRef]
- Van Rantwijk, F.; Sheldon, R.A. Biocatalysis in ionic liquids. Chem. Rev. 2007, 107, 2757–2785. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H. Methods for stabilizing and activating enzymes in ionic liquids—A review. J. Chem. Technol. Biot. 2010, 85, 891–907. [Google Scholar] [CrossRef]
- Seddon, K.R.; Stark, A.; Torres, M.-J. Influence of chloride, water, and organic solvents on the physical properties of ionic liquids. Pure Appl. Chem. 2000, 72, 2275–2287. [Google Scholar] [CrossRef]
- Uyama, H.; Takamoto, T.; Kobayashi, S. Enzymatic synthesis of polyesters in ionic liquids. Polym. J. 2002, 34, 94–96. [Google Scholar] [CrossRef]
- Marcilla, R.; de Geus, M.; Mecerreyes, D.; Duxbury, C.J.; Koning, C.E.; Heise, A. Enzymatic polyester synthesis in ionic liquids. Eur. Polym. J. 2006, 42, 1215–1221. [Google Scholar] [CrossRef]
- Wu, C.; Zhang, Z.; He, F.; Zhuo, R. Enzymatic synthesis of poly (ε-caprolactone) in monocationic and dicationic ionic liquids. Biotechnol. Lett. 2013, 35, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa-Fujita, M.; Saito, C.; Takeoka, Y.; Rikukawa, M. Lipase-catalyzed polymerization of l-lactide in ionic liquids. Polym. Adv. Technol. 2008, 19, 1396–1400. [Google Scholar] [CrossRef]
- Mena, M.; Chanfreau, S.; Gimeno, M.; Bárzana, E. Enzymatic synthesis of poly-l-lactide-co-glycolide in the ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate. Bioprocess Biosyst. Eng. 2010, 33, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Mena, M.; López-Luna, A.; Shirai, K.; Tecante, A.; Gimeno, M.; Bárzana, E. Lipase-catalyzed synthesis of hyperbranched poly-L-lactide in an ionic liquid. Bioprocess Biosyst. Eng. 2013, 36, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Chanfreau, S.; Mena, M.; Porras-Domínguez, J.R.; Ramírez-Gilly, M.; Gimeno, M.; Roquero, P.; Tecante, A.; Bárzana, E. Enzymatic synthesis of poly-L-lactide and poly-l-lactide-co-glycolide in an ionic liquid. Bioprocess Biosyst. Eng. 2010, 33, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.-X.; Zhang, L.; Tong, X.-Z.; Chen, H.-B.; Wang, X.-L.; Wang, Y.-Z. Ionic liquid coated lipase: Green synthesis of high molecular weight poly (1, 4-dioxan-2-one). J. Mol. Catal. B-Enzym. 2012, 77, 46–52. [Google Scholar] [CrossRef]
- Huijser, S.; Staal, B.B.; Huang, J.; Duchateau, R.; Koning, C.E. Topology characterization by MALDI-ToF-MS of enzymatically synthesized poly (lactide-co-glycolide). Biomacromolecules 2006, 7, 2465–2469. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhang, Z.; Chen, C.; He, F.; Zhuo, R. Synthesis of poly (ε-caprolactone) by an immobilized lipase coated with ionic liquids in a solvent-free condition. Biotechnol. Lett. 2013, 35, 1623–1630. [Google Scholar] [CrossRef] [PubMed]
- Córdova, A.; Iversen, T.; Hult, K.; Martinelle, M. Lipase-catalysed formation of macrocycles by ring-opening polymerisation of ε-caprolactone. Polymer 1998, 39, 6519–6524. [Google Scholar] [CrossRef]
- Skaria, S.; Smet, M.; Frey, H. Enzyme-catalyzed synthesis of hyperbranched aliphatic polyesters. Macromol. Rapid Commun. 2002, 23, 292–296. [Google Scholar] [CrossRef]
- López-Luna, A.; Gallegos, J.L.; Gimeno, M.; Vivaldo-Lima, E.; Bárzana, E. Lipase-catalyzed syntheses of linear and hyperbranched polyesters using compressed fluids as solvent media. J. Mol. Catal. B-Enzym. 2010, 67, 143–149. [Google Scholar] [CrossRef]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piotrowska, U.; Sobczak, M. Enzymatic Polymerization of Cyclic Monomers in Ionic Liquids as a Prospective Synthesis Method for Polyesters Used in Drug Delivery Systems. Molecules 2015, 20, 1-23. https://doi.org/10.3390/molecules20010001
Piotrowska U, Sobczak M. Enzymatic Polymerization of Cyclic Monomers in Ionic Liquids as a Prospective Synthesis Method for Polyesters Used in Drug Delivery Systems. Molecules. 2015; 20(1):1-23. https://doi.org/10.3390/molecules20010001
Chicago/Turabian StylePiotrowska, Urszula, and Marcin Sobczak. 2015. "Enzymatic Polymerization of Cyclic Monomers in Ionic Liquids as a Prospective Synthesis Method for Polyesters Used in Drug Delivery Systems" Molecules 20, no. 1: 1-23. https://doi.org/10.3390/molecules20010001







