Carbon Nanotubes as Supports for Inulinase Immobilization
Abstract
:1. Introduction
2. Results and Discussion
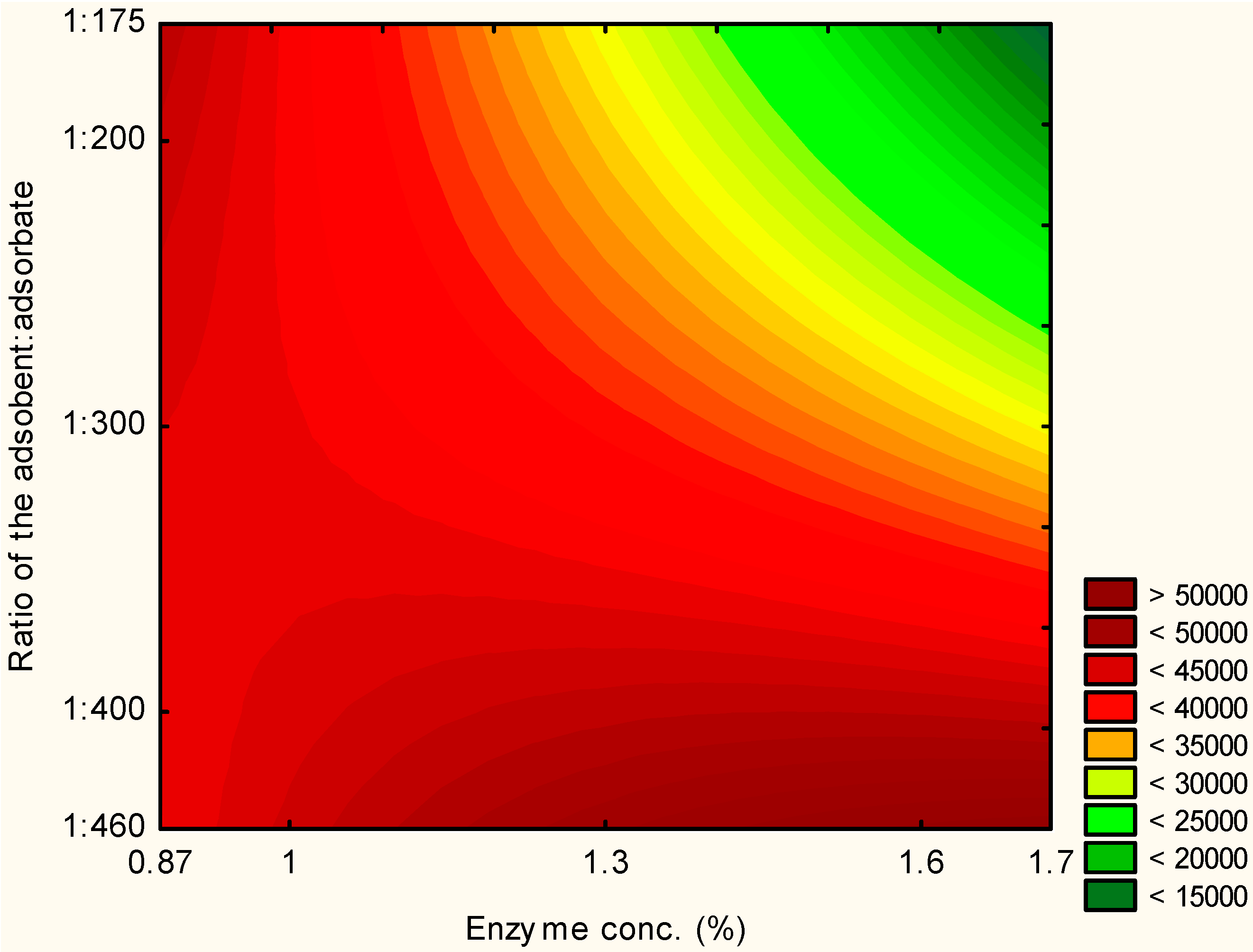
2.1. Experimental Design
| Assays | Enzyme Concentration (%) | Ratio Adsorbent:Adsorbate | Immobilization (U/g) |
|---|---|---|---|
| 1 | −1 (1.0) | −1 (1:400) | 45,774.44 |
| 2 | 1 (1.6) | −1 (1:400) | 45,286.32 |
| 3 | −1 (1.0) | 1 (1:200) | 49,570.90 |
| 4 | 1 (1.6) | 1 (1:200) | 26,665.60 |
| 5 | −1.41 (0.87) | 0 (1:300) | 39,331.28 |
| 6 | 1.41 (1.7) | 0 (1:300) | 30,784.64 |
| 7 | 0 (1.3) | −1.41 (1:460) | 51,047.76 |
| 8 | 0 (1.3) | 1.41 (1:175) | 24,603.97 |
| 9 | 0 (1.3) | 0 (1:300) | 38,715.53 |
| 10 | 0 (1.3) | 0 (1:300) | 40,217.43 |
| 11 | 0 (1.3) | 0 (1:300) | 40,175.71 |

| SS | df | MS | F | R2 | |
|---|---|---|---|---|---|
| Regression | 623,581,895 | 3 | 207,860,631 | 9.84 | 81.0 |
| Residual | 147,736,259 | 7 | 21,105,179 | ||
| Total | 771,318,154 | 10 |
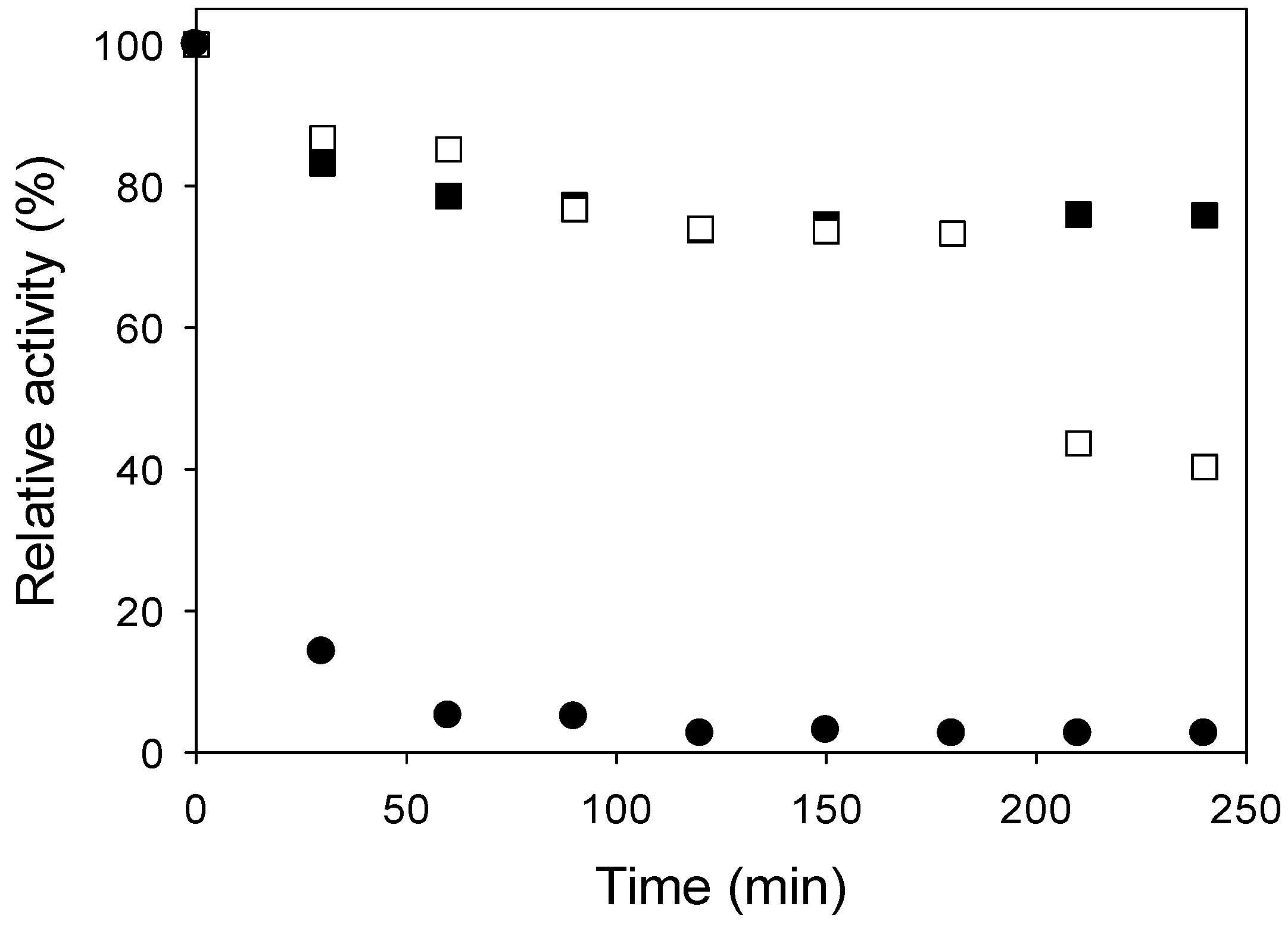
2.2. Thermal and Shelf Stabilities

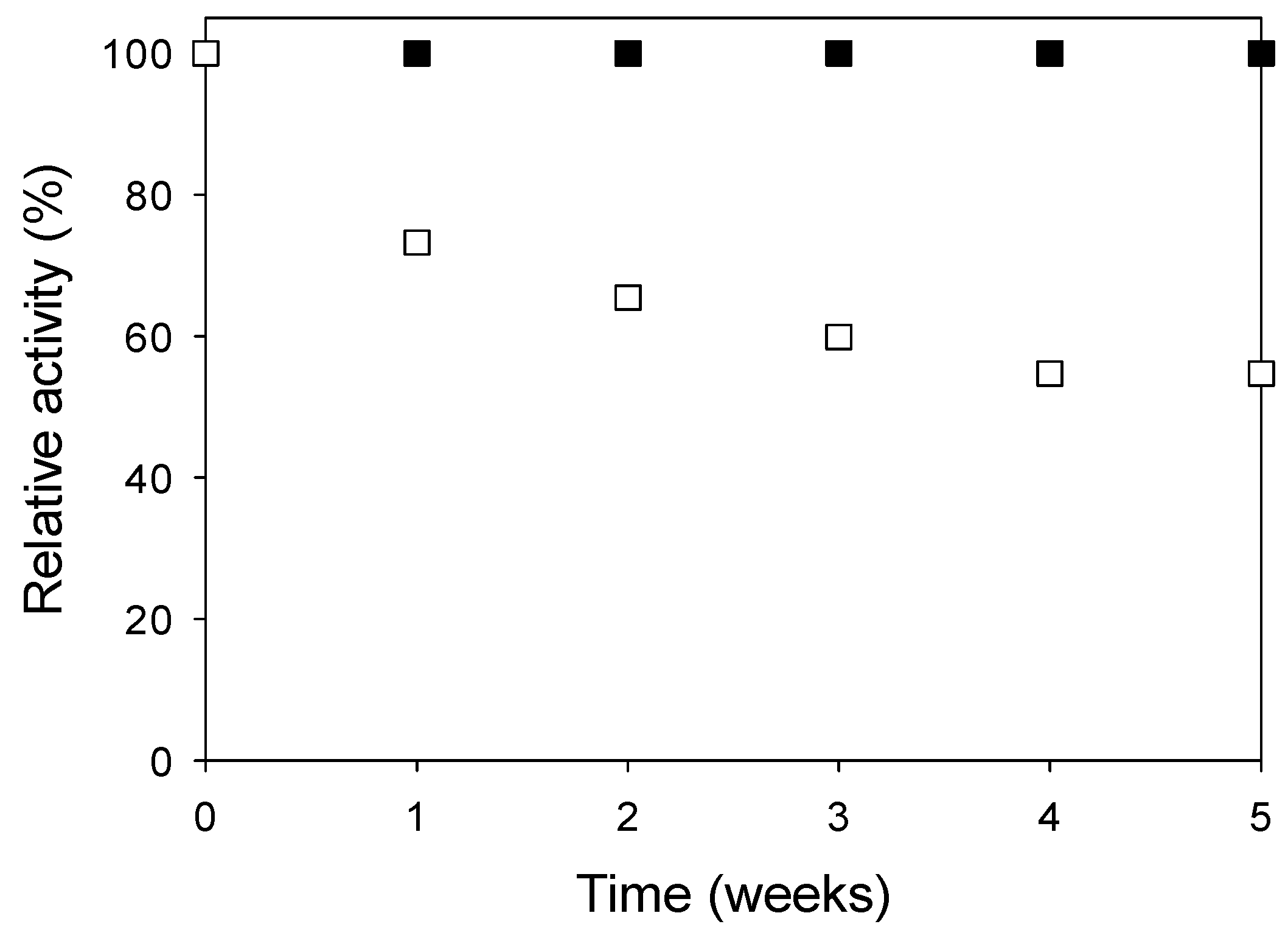
3. Experimental Section
3.1. Materials
3.2. Enzyme Immobilization
3.3. Inulinase Activity Assay
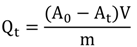
3.4. Experimental Design
3.5. Thermal Stability
3.6. Shelf Stability
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Silva, M.F.; Rigo, D.; Mossi, V.; Dallago, R.M.; Henrick, P.; Kuhn, G.O.; Rosa, C.D.; Oliveira, D.; Oliveira, J.V.; Treichel, H. Evaluation of enzymatic activity of commercial inulinase from Aspergillus niger immobilized in polyurethane foam. Food Bioprod. Process. 2013, 91, 54–59. [Google Scholar] [CrossRef]
- Ye, P.; Jiang, J.; Xu, Z.-K. Adsorption and activity of lipase from Candida rugosa on the chitosan-modified poly (acrylonitrile-co-maleic acid) membrane surface. Colloids Surf. B 2007, 60, 62–67. [Google Scholar] [CrossRef]
- Karimi, M.; Chaudhury, I.; Jianjun, C.; Safari, M.; Sadeghi, R.; Habibi-Rezaei, M.; Kokini, J. Immobilization of endo-inulinase on non-porous amino functionalized silica nanoparticles. J. Mol. Catal. B 2014, 104, 48–55. [Google Scholar] [CrossRef]
- Fernandes, P.; Marques, M.P.C.; Carvalho, F.; Cabral, J.M.S. A simple method for biocatalyst immobilization using PVA-based hydrogel particles. J. Chem. Technol. Biotechnol. 2009, 84, 561–564. [Google Scholar] [CrossRef]
- Ettalibi, M.; Baratti, J.C. Sucrose hydrolysis by thermostable immobilized inulinases from Aspergillus ficcum. Enzym. Microb. Technol. 2001, 28, 596–601. [Google Scholar]
- Yewale, T.; Singhal, R.S.; Vaidja, A.A. Immobilization of inulinase from Aspergillus niger NCIM 945 on chitosan and its application in continuous inulin hydrolysis. Biocatal. Agric. Biotechnol. 2013, 2, 96–101. [Google Scholar]
- Prlainovic, N.Z.; Bezbradica, D.I.; Knezevic-Jugovic, Z.D.; Stevanovic, S.I.; Ivic, M.L.A.; Uskokovic, P.S.; Mijin, D.Z. Adsorption of lipase from Candida rugosa on multi walled carbon nanotubes. J. Ind. Eng. Chem. 2013, 19, 279–285. [Google Scholar] [CrossRef]
- Brady, D.; Jordaan, J. Advances in enzyme immobilization. Biotechnol. Lett. 2009, 31, 1639–1650. [Google Scholar] [CrossRef]
- Garcia-Galan, C.; Berenguer-Murcia, A.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- Feng, W.; Ji, P. Enzymes immobilized on carbon nanotubes. Biotechnol. Adv. 2011, 29, 889–895. [Google Scholar] [CrossRef]
- Tasis, D.; Tagmatarchis, N.; Bianco, A.; Prato, M. Chemistry of carbon nanotubes. Chem. Ver. 2006, 106, 1105–1136. [Google Scholar]
- Gao, Y.; Kyratzis, I. Covalent immobilization of proteins on carbon nanotubes using the cross-linker 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide—A critical assessment. Bioconjugate Chem. 2008, 19, 1945–1950. [Google Scholar]
- Brena, B.; González-Pombo, P.; Batista-Viera, F. Immobilization of enzymes: A literature survey. Methods Mol. Biol. 2013, 1051, 15–31. [Google Scholar]
- Ji, P.; Tan, H.; Xu, X.; Feng, W. Lipase covalently attached to multi-walled carbon nanotubes as an efficient catalyst in organic solvent. AIChE J. 2010, 56, 3005–3011. [Google Scholar] [CrossRef]
- Asuri, P.; Bale, S.S.; Pangule, R.C.; Shah, D.A.; Kane, R.S.; Dordick, J.S. Structure, function, and stability of enzymes covalently attached to single-walled carbon nanotubes. Langmuir 2007, 23, 12318–12321. [Google Scholar] [CrossRef]
- Leaes, E.; Zimmermann, E.; Souza, M.; Ramon, A.; Mezadri, E.; Dal Prá, V.; Terra, L.; Mazutti, M. Ultrasound-assisted enzymatic hydrolysis of cassava waste to obtain fermentable sugars. Biol. Eng. 2013, 115, 1–6. [Google Scholar]
- Rocha, J.R.; Catana, R.; Ferreira, B.S.; Cabral, J.M.S.; Fernandes, P. Design and characterization of an enzyme system from inulin hydrolysis. Food Chem. 2006, 95, 77–82. [Google Scholar] [CrossRef]
- Danial, E.N.; Elnashar, M.M.M.; Awad, G.E.A. Immobilized inulinase on grafted alginate beads prepared by the one-step and the two-steps methods. Ind. Eng. Chem. Res. 2010, 49, 3120–3125. [Google Scholar] [CrossRef]
- Richeti, A.; Munaretto, C.B.; Lerin, L.A.; Batistella, L.; Oliveira, J.V.; Dallago, R.M.; Astolfi, V.; di Luccio, M.; Mazutti, M.; de Oliveira, D.; et al. Immobilization of inulinase from Kluyveromyces marxianus NRRL Y-7571 using modified sodium alginate beads. Bioproc. Biosyst. Eng. 7571, 35, 383–388. [Google Scholar]
- Asuri, P.; Karajanagi, S.S.; Yang, H.; Yim, T.-J.; Kane, R.S.; Dordick, J.S. Increasing protein stability through control of the nanoscale environment. Langmuir 2006, 22, 5833–5836. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalisylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Garlet, T.B.; Weber, C.T.; Klaic, R.; Foletto, E.L.; Jahn, S.L.; Mazutti, M.A.; Kuhn, R.C. Carbon Nanotubes as Supports for Inulinase Immobilization. Molecules 2014, 19, 14615-14624. https://doi.org/10.3390/molecules190914615
Garlet TB, Weber CT, Klaic R, Foletto EL, Jahn SL, Mazutti MA, Kuhn RC. Carbon Nanotubes as Supports for Inulinase Immobilization. Molecules. 2014; 19(9):14615-14624. https://doi.org/10.3390/molecules190914615
Chicago/Turabian StyleGarlet, Tais B., Caroline T. Weber, Rodrigo Klaic, Edson L. Foletto, Sergio L. Jahn, Marcio A. Mazutti, and Raquel C. Kuhn. 2014. "Carbon Nanotubes as Supports for Inulinase Immobilization" Molecules 19, no. 9: 14615-14624. https://doi.org/10.3390/molecules190914615
APA StyleGarlet, T. B., Weber, C. T., Klaic, R., Foletto, E. L., Jahn, S. L., Mazutti, M. A., & Kuhn, R. C. (2014). Carbon Nanotubes as Supports for Inulinase Immobilization. Molecules, 19(9), 14615-14624. https://doi.org/10.3390/molecules190914615




