Simultaneous Quantification of Three Polymorphic Forms of Carbamazepine in the Presence of Excipients Using Raman Spectroscopy
Abstract
:1. Introduction
2. Results and Discussion
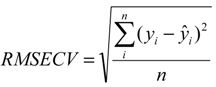
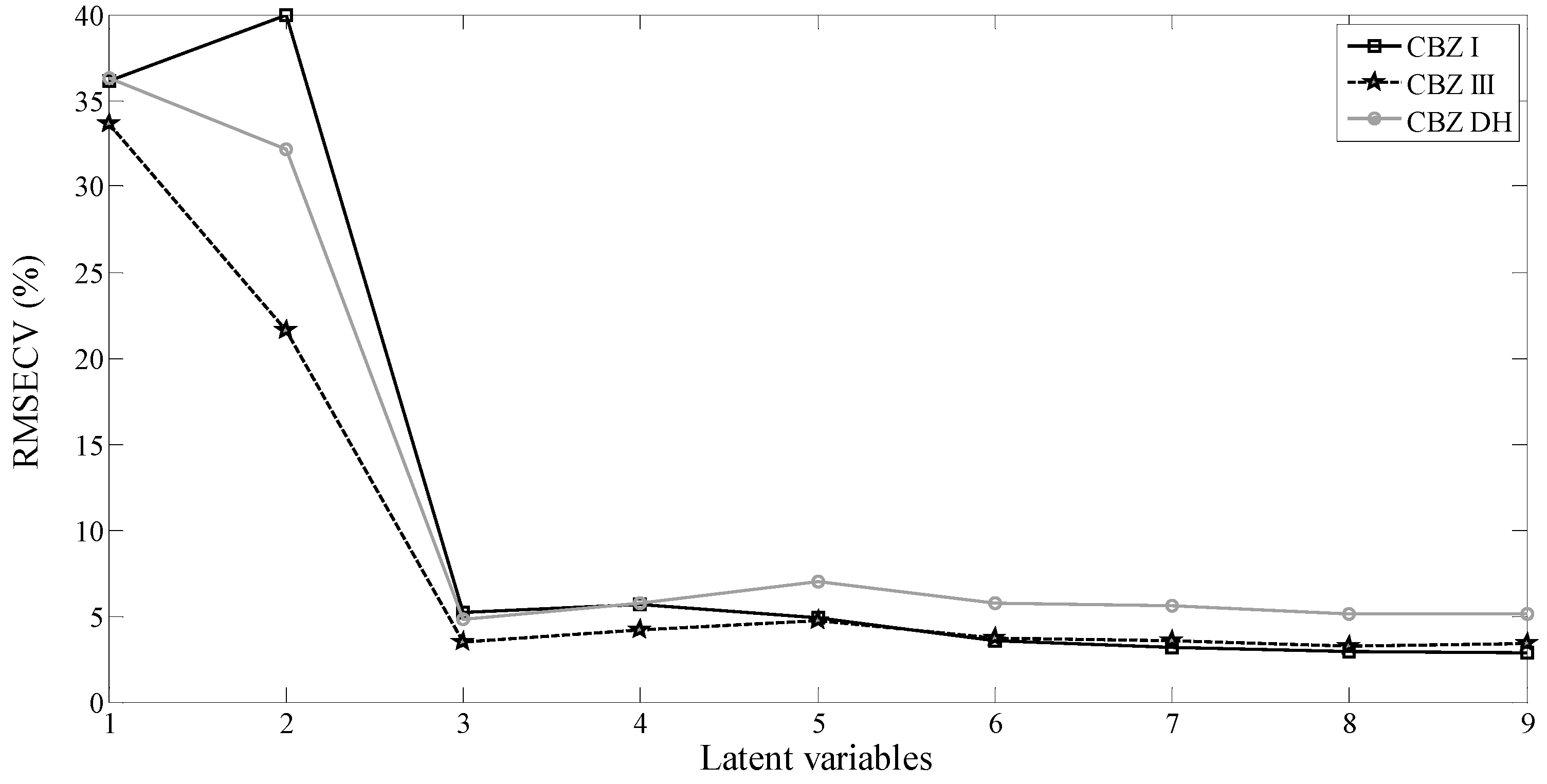
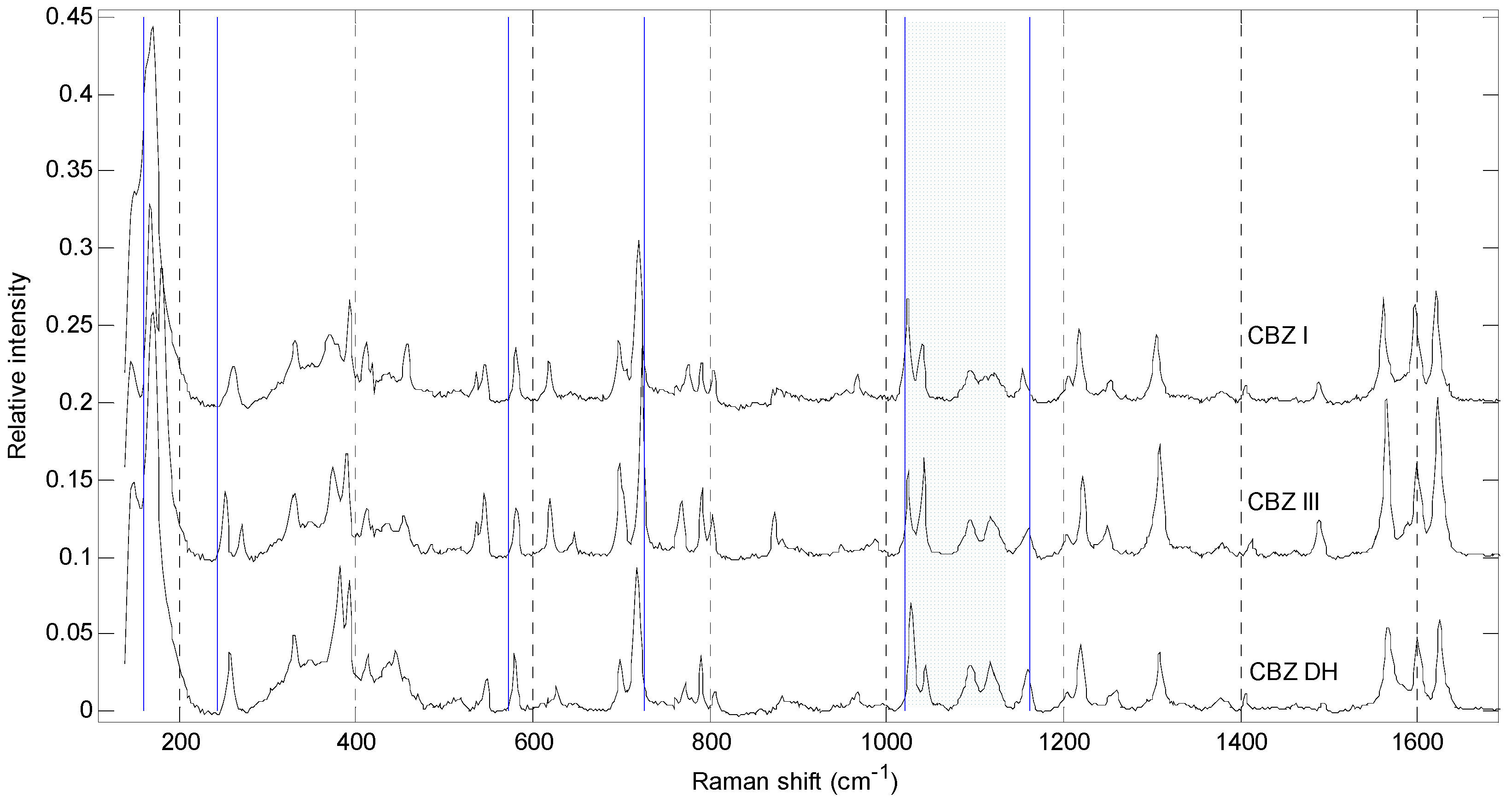
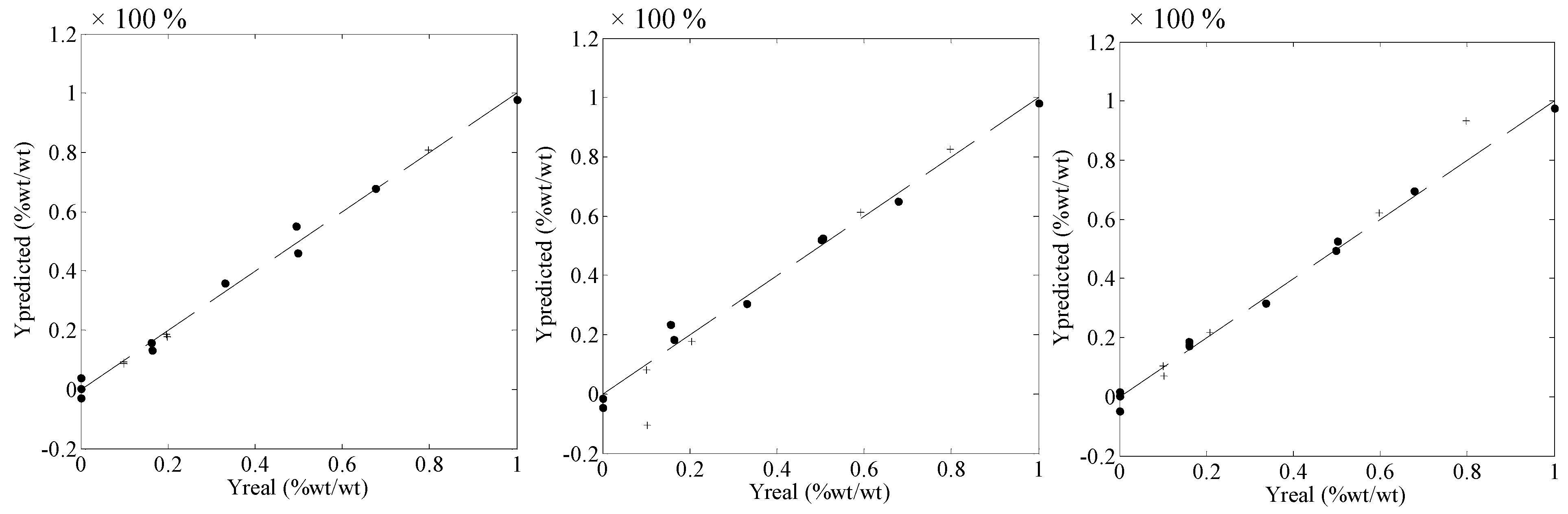
| Parameters | Polymorphs | ||
|---|---|---|---|
| CBZ I | CBZ III | CBZ DH | |
| RMSEC | 3.38 (0.56) | 2.30 (0.38) | 3.02 (0.50) |
| RMSECV | 5.19 (0.86) | 3.49 (0.58) | 4.82 (0.80) |
| RMSEV | 9.49 (1.58) | 6.27 (1.04) | 1.34 (0.22) |
| R^2 | 0.965 | 0.994 | 0.999 |
3. Experimental Section
3.1. Preparation of Polymorphs
3.2. Characterization of the Polymorphs
3.3. Preparation of the Tablets
| Samples | Polymorphs | ||
|---|---|---|---|
| CBZ I | CBZ III | CBZ DH | |
| 1 | 49.4 | 0 | 0 |
| 2 | 0 | 50 | 0 |
| 3 | 0 | 0 | 50.8 |
| 4 | 25.3 | 25.2 | 0 |
| 5 | 25.2 | 0 | 24.7 |
| 6 | 0 | 25.1 | 24.9 |
| 7 | 34.3 | 8.1 | 8.2 |
| 8 | 7.9 | 34.3 | 8.3 |
| 9 | 8.2 | 8 | 33.9 |
| 10 | 16.4 | 16.2 | 16.4 |
| 11 | 40.2 | 5.2 | 5 |
| 12 | 5.1 | 40 | 5 |
| 13 | 5.1 | 5.1 | 40.1 |
| 14 | 30 | 10.6 | 10.1 |
| 15 | 10.2 | 29.8 | 9.8 |
3.4. Acquisition and Treatment of the Data
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bauer, J.; Spanton, S.; Henry, R.; Quick, J.; Dziki, W.; Porter, W.; Morris, J. Ritonavir: An extraordinary example of conformational polymorphism. Pharm. Res. 2001, 18, 859–866. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Pharmaceutical Solid Polymorphism Chemistry, Manufacturing, and Controls Information. Available online: http://www.fda.gov/downloads/Drugs/Guidances/UCM072866.pdf (accessed on 4 September 2014).
- Threlfall, T.L. Analysis of organic polymorphs. A review. Analyst 1995, 120, 2435–2460. [Google Scholar] [CrossRef]
- Haleblian, J.; McCrone, W. Pharmaceutical applications of polymorphism. J. Pharm. Sci. 1969, 58, 911–929. [Google Scholar]
- Stahly, G.P. Diversity in single- and multiple-component crystals. The search for and prevalence of polymorphs and cocrystals. Cryst. Growth Des. 2007, 7, 1007–1026. [Google Scholar] [CrossRef]
- Taylor, L.S.; Langkilde, F.W. Evaluation of solid-state forms present in tablets by raman spectroscopy. J. Pharm. Sci. 2000, 89, 1342–1353. [Google Scholar] [CrossRef]
- Byrn, S.; Pfeiffer, R.; Ganey, M.; Hoiberg, C.; Poochikian, G. Pharmaceutical solids: A strategic approach to regulatory considerations. Pharm. Res. 1995, 12, 945–954. [Google Scholar] [CrossRef]
- Arlin, J.B.; Price, L.S.; Price, S.L.; Florence, A.J. A strategy for producing predicted polymorphs: Catemeric carbamazepine form V. Chem. Commun. 2011, 47, 7074–7076. [Google Scholar]
- Lang, M.; Kampf, J.W.; Matzger, A.J. Form iv of carbamazepine. J. Pharm. Sci. 2002, 91, 1186–1190. [Google Scholar] [CrossRef]
- Grzesiak, A.L.; Lang, M.; Kim, K.; Matzger, A.J. Comparison of the four anhydrous polymorphs of carbamazepine and the crystal structure of form i. J. Pharm. Sci. 2003, 92, 2260–2271. [Google Scholar] [CrossRef]
- Caliandro, R.; Profiob, G.; Nicolottic, O. Multivariate analysis of quaternary carbamazepine-saccharin mixtures by X-ray diffraction and infrared spectroscopy. J. Pharm. Biomed. Anal. 2013, 78–79, 269–279. [Google Scholar] [CrossRef]
- Flicker, F.; Eberle, V.A.; Betz, G. Variability in commercial carbamazepine samples-impact on drug release. Int. J. Pharm. 2011, 410, 99–106. [Google Scholar] [CrossRef]
- Meyer, M.C.; Straughn, A.B.; Mhatre, R.M.; Shah, V.P.; Williams, R.L.; Lesko, L.J. The relative bioavailability and in vivo-in vitro correlations for four marketed carbamazepine tablets. Pharm. Res. 1998, 15, 1787–1791. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. The Biopharmaceutics Classification System (BCS) Guidance. Available online: Http://www.Fda.Gov/aboutfda/centersoffices/officeofmedicalproductsandtobacco/cder/ucm128219.Htm (accessed on 27 August 2014).
- Kobayashi, Y.; Ito, S.; Itai, S.; Yamamoto, K. Physicochemical properties and bioavailability of carbamazepine polymorphs and dihydrate. Int. J. Pharm. 2000, 193, 137–146. [Google Scholar] [CrossRef]
- Chieng, N.; Rades, T.; Aaltonen, J. An overview of recent studies on the analysis of pharmaceutical polymorphs. J. Pharm. Biomed. Anal. 2011, 55, 618–644. [Google Scholar] [CrossRef]
- Offerdahl, T.J.; Salsbury, J.S.; Dong, Z.; Grant, D.J.; Schroeder, S.A.; Prakash, I.; Gorman, E.M.; Barich, D.H.; Munson, E.J. Quantitation of crystalline and amorphous forms of anhydrous neotame using 13c cpmas nmr spectroscopy. J. Pharm. Sci. 2005, 94, 2591–2605. [Google Scholar] [CrossRef]
- Tian, F.; Zhang, F.; Sandler, N.; Gordon, K.C.; McGoverin, C.M.; Strachan, C.J.; Saville, D.J.; Rades, T. Influence of sample characteristics on quantification of carbamazepine hydrate formation by X-ray powder diffraction and raman spectroscopy. Eur. J. Pharm. Biopharm. 2007, 66, 466–474. [Google Scholar] [CrossRef]
- Soares, F.L.F.; Carneiro, R.L. Evaluation of analytical tools and multivariate methods for quantification of co-former crystals in ibuprofen-nicotinamide co-crystals. J. Pharm. Biomed. Anal. 2013, 89, 166–175. [Google Scholar] [CrossRef]
- Atef, E.; Chauhan, H.; Prasad, D.; Kumari, D.; Pidgeon, C. Quantifying solid-state mixtures of crystalline indomethacin by raman spectroscopy comparison with thermal analysis. ISRN Chromatogr. 2012, 2012. [Google Scholar] [CrossRef]
- Bugay, D.E. Characterization of the solid-state: Spectroscopic techniques. Adv. Drug Deliv. Rev. 2001, 48, 43–65. [Google Scholar] [CrossRef]
- Langkilde, F.W.; Sjoblom, J.; Tekenbergs-Hjelte, L.; Mrak, J. Quantitative ft-raman analysis of two crystal forms of a pharmaceutical compound. J. Pharm. Biomed. Anal. 1997, 15, 687–696. [Google Scholar] [CrossRef]
- Vergote, G.J.; Vervaet, C.; Remon, J.P.; Haemers, T.; Verpoort, F. Near-infrared ft-raman spectroscopy as a rapid analytical tool for the determination of diltiazem hydrochloride in tablets. Eur. J. Pharm. Sci. 2002, 16, 63–67. [Google Scholar]
- Tudor, A.M.; Melia, C.D.; Binns, J.S.; Hendra, P.J.; Church, S.; Davies, M.C. The application of fourier-transform raman spectroscopy to the analysis of pharmaceuticals and biomaterials. J. Pharm. Biomed. Anal. 1990, 8, 717–720. [Google Scholar]
- Strachan, C.J.; Pratiwi, D.; Gordon, K.C.; Rades, T. Quantitative analysis of polymorphic mixtures of carbamazepine by raman spectroscopy and principal components analysis. J. Raman Spectrosc. 2004, 35, 347–352. [Google Scholar] [CrossRef]
- Farias, M.A.D.S.; Carneiro, R.L. Simultaneous quantification of three polymorphic forms of carbamazepine using raman spectroscopy and multivariate calibration. Anal. Lett. 2014, 47, 1043–1051. [Google Scholar] [CrossRef]
- McMahon, L.E.; Timmins, P.; Williams, A.C.; York, P. Characterization of dihydrates prepared from carbamazepine polymorphs. J. Pharm. Sci. 1996, 85, 1064–1069. [Google Scholar] [CrossRef]
- Brereton, R.G. Applied chemometrics for Scientists; John Wiley & Sons: Chichester, UK, 2007. [Google Scholar]
- Rowe, R.C.; Sheskey, P.J.; Quinn, M.E. Handbook of Pharmaceutical Excipients, 6 ed.; Pharmaceutical Press and American Pharmacists Association: Washington, DC, USA, 2009; p. 888. [Google Scholar]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Farias, M.; Carneiro, R. Simultaneous Quantification of Three Polymorphic Forms of Carbamazepine in the Presence of Excipients Using Raman Spectroscopy. Molecules 2014, 19, 14128-14138. https://doi.org/10.3390/molecules190914128
Farias M, Carneiro R. Simultaneous Quantification of Three Polymorphic Forms of Carbamazepine in the Presence of Excipients Using Raman Spectroscopy. Molecules. 2014; 19(9):14128-14138. https://doi.org/10.3390/molecules190914128
Chicago/Turabian StyleFarias, Marco, and Renato Carneiro. 2014. "Simultaneous Quantification of Three Polymorphic Forms of Carbamazepine in the Presence of Excipients Using Raman Spectroscopy" Molecules 19, no. 9: 14128-14138. https://doi.org/10.3390/molecules190914128
APA StyleFarias, M., & Carneiro, R. (2014). Simultaneous Quantification of Three Polymorphic Forms of Carbamazepine in the Presence of Excipients Using Raman Spectroscopy. Molecules, 19(9), 14128-14138. https://doi.org/10.3390/molecules190914128





